Performance Comparison of Proton Exchange Membrane Water Electrolysis Cell Using Channel and PTL Flow Fields through Three-Dimensional Two-Phase Flow Simulation
Abstract
1. Introduction
2. Methodology
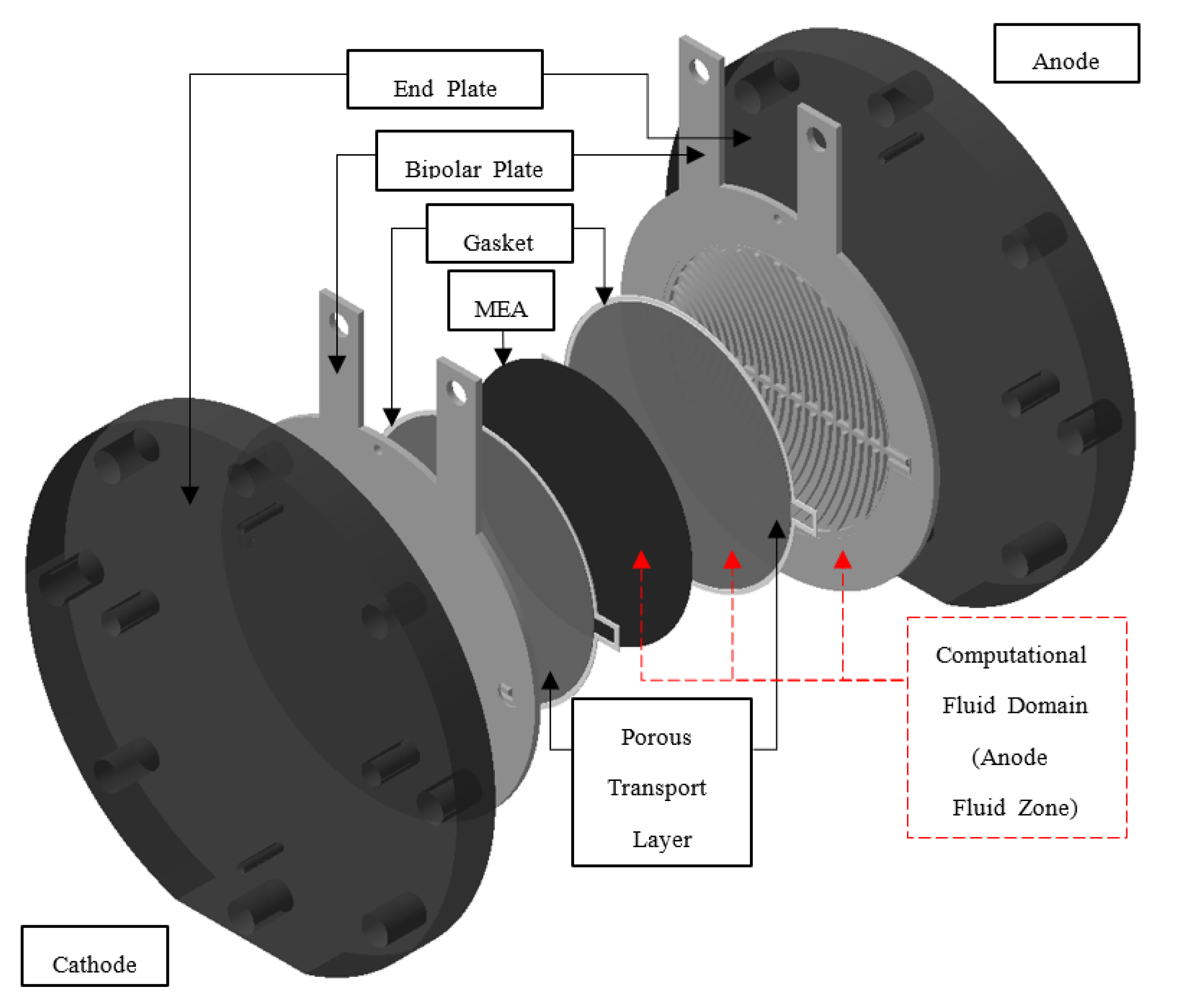
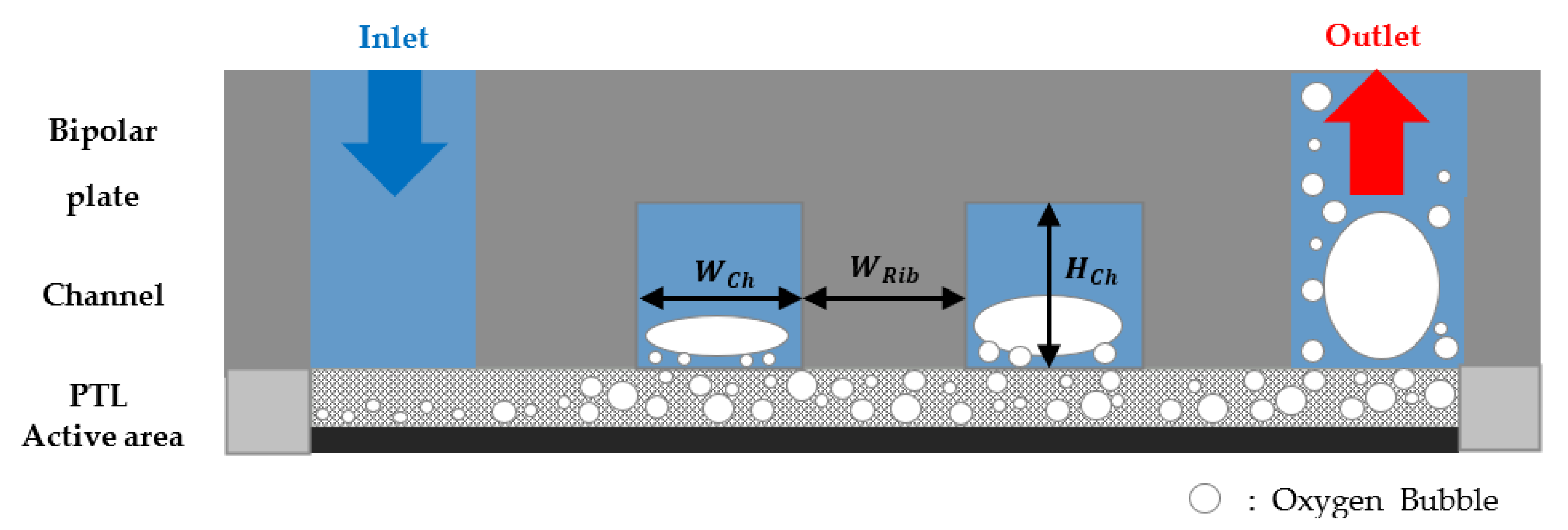
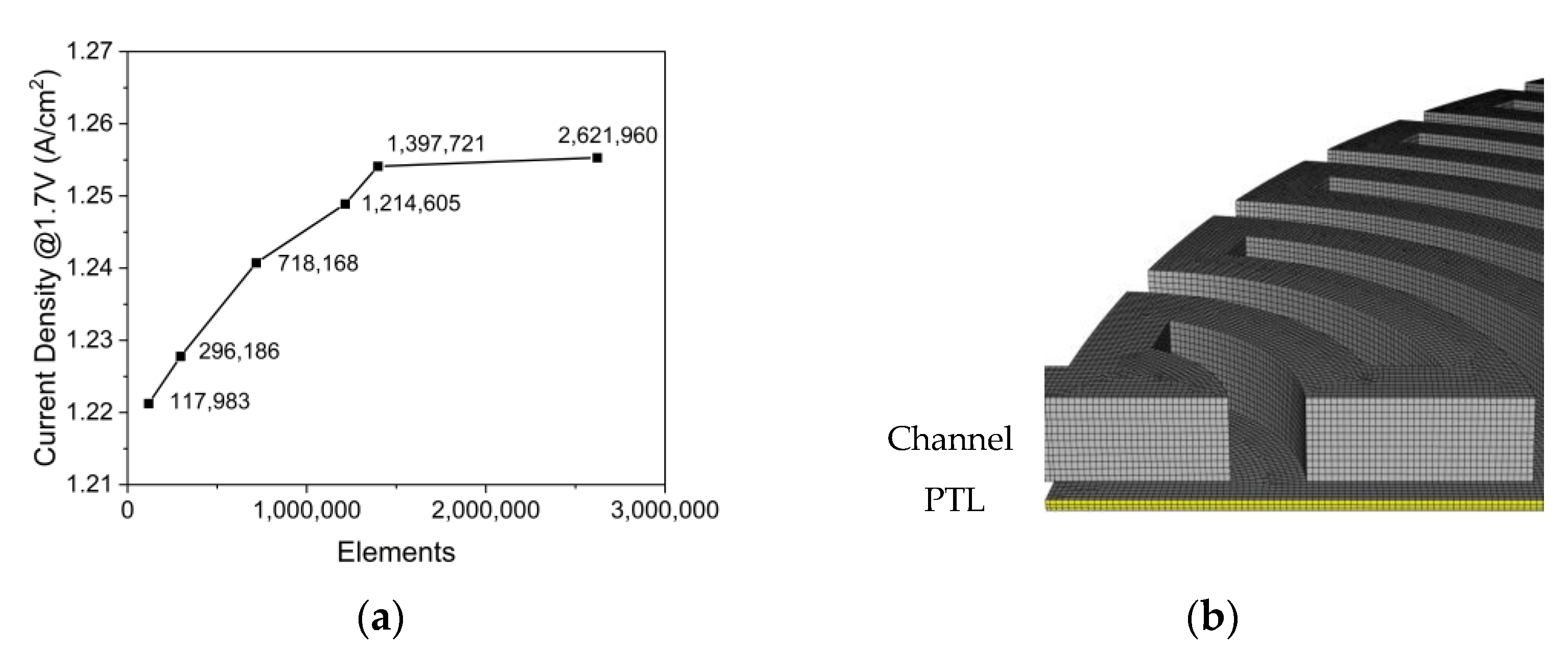
2.1. Simulation Domain
2.2. Model Assumptions
- The activation voltage of the cathode was ignored because of the fast reaction rate of the cathode [24].
- The flow through the channel and PTLs were assumed as laminar flow because of the low Reynolds number.
- It is assumed that the properties of the PTL are isotropic and identical in all porous media regions.
- The anode was not pressurized and the oxygen flow from anode to cathode was ignored [25].
2.3. Governing Equations
2.4. Boundary Conditions
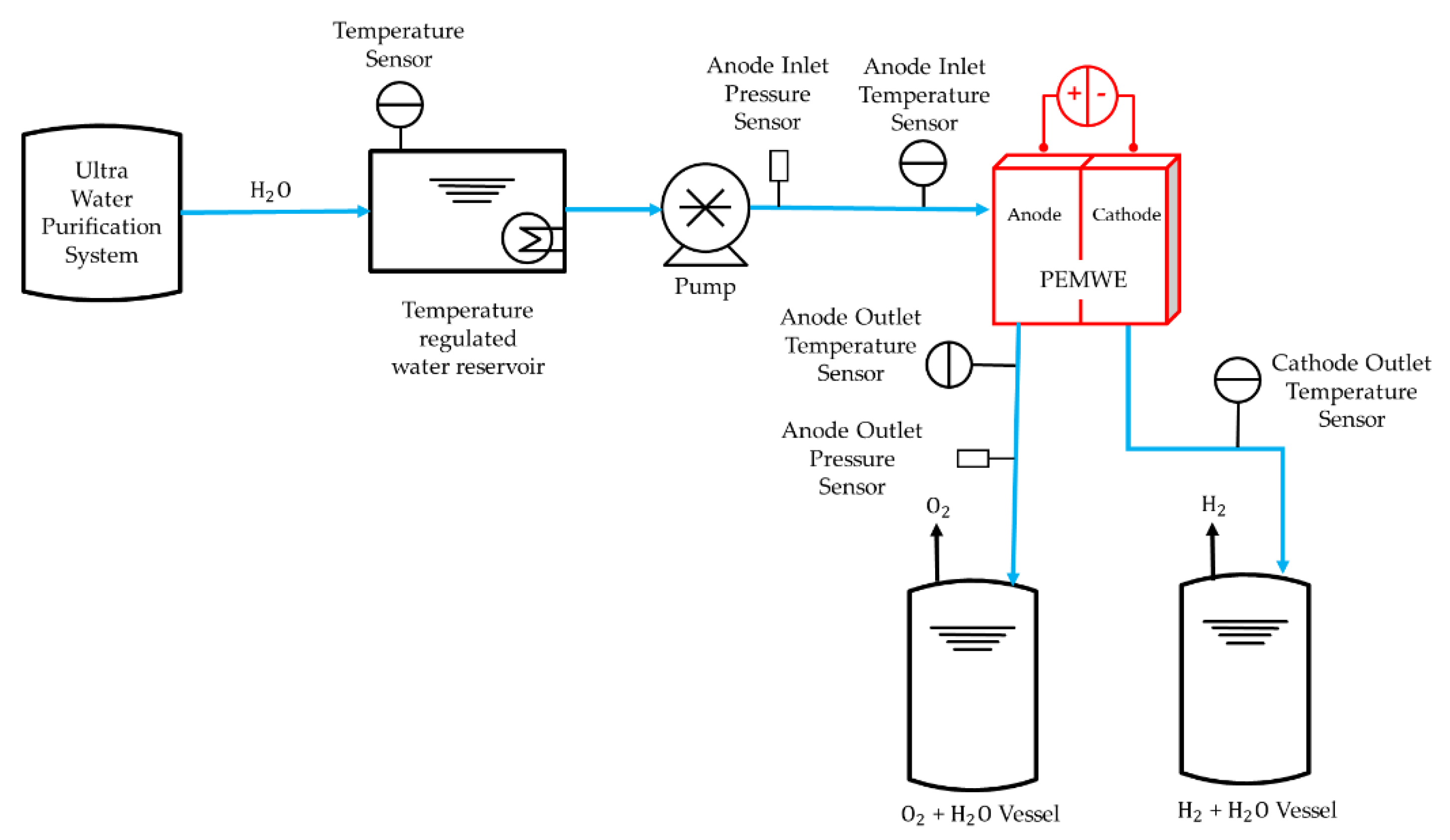
2.5. Experimental Setup
3. Results and Discussion
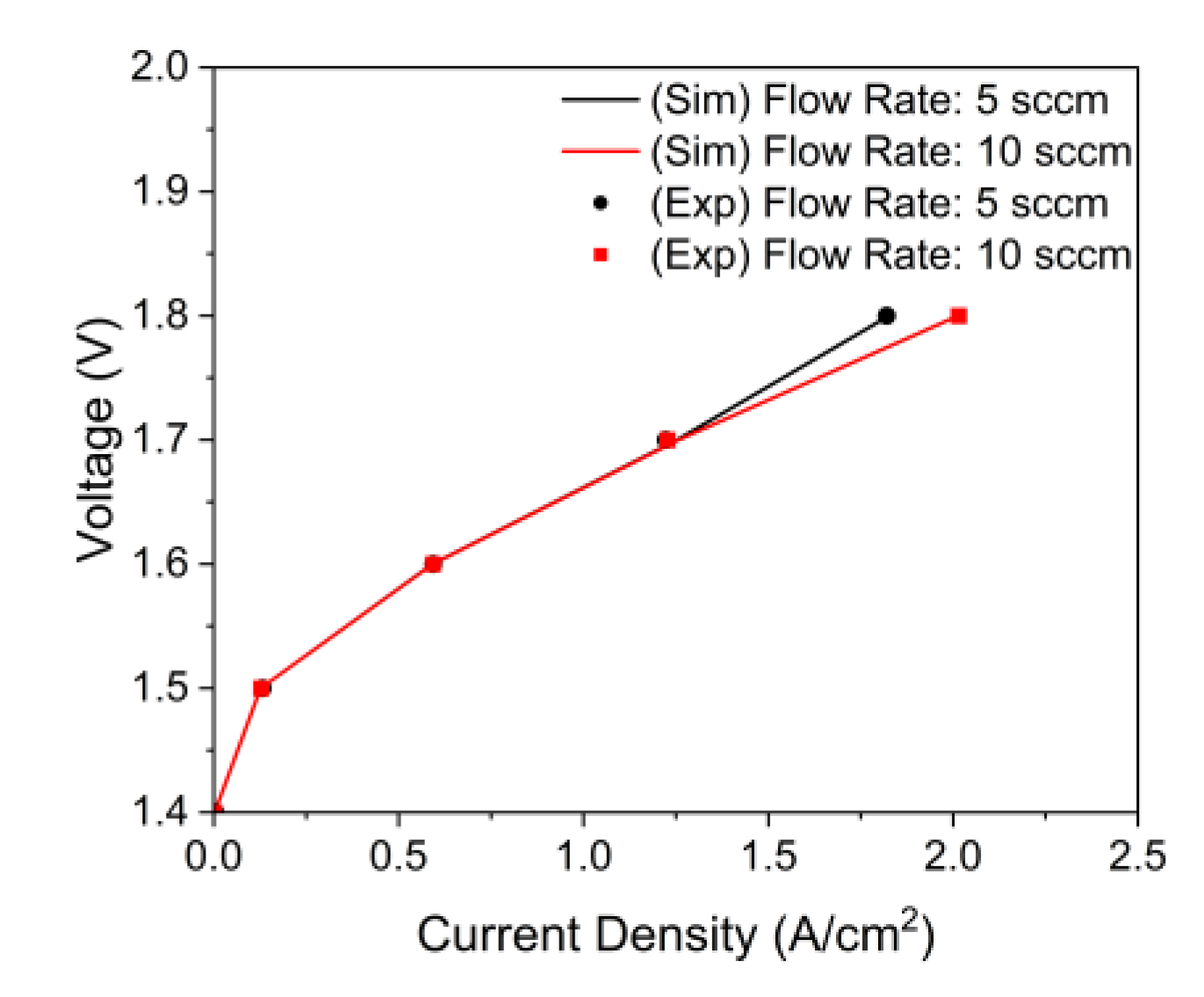
3.1. Model Validation
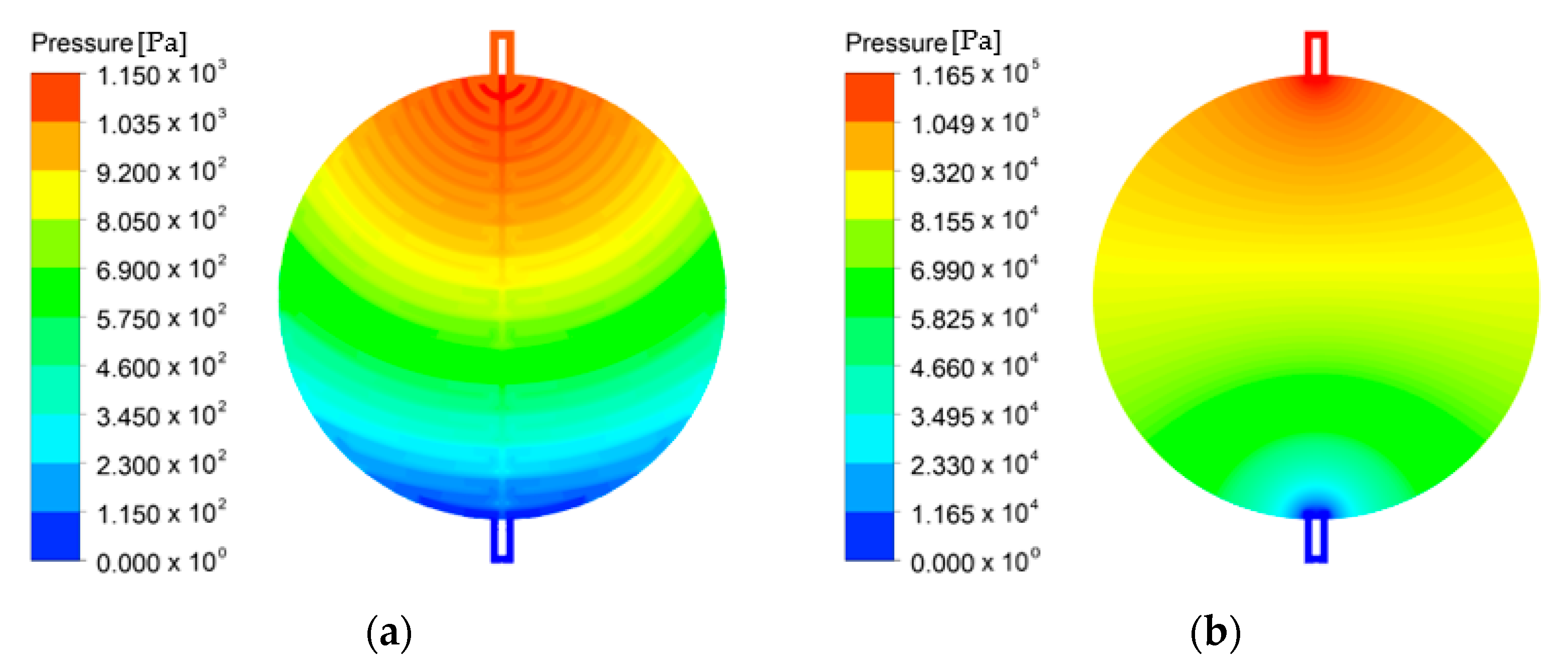
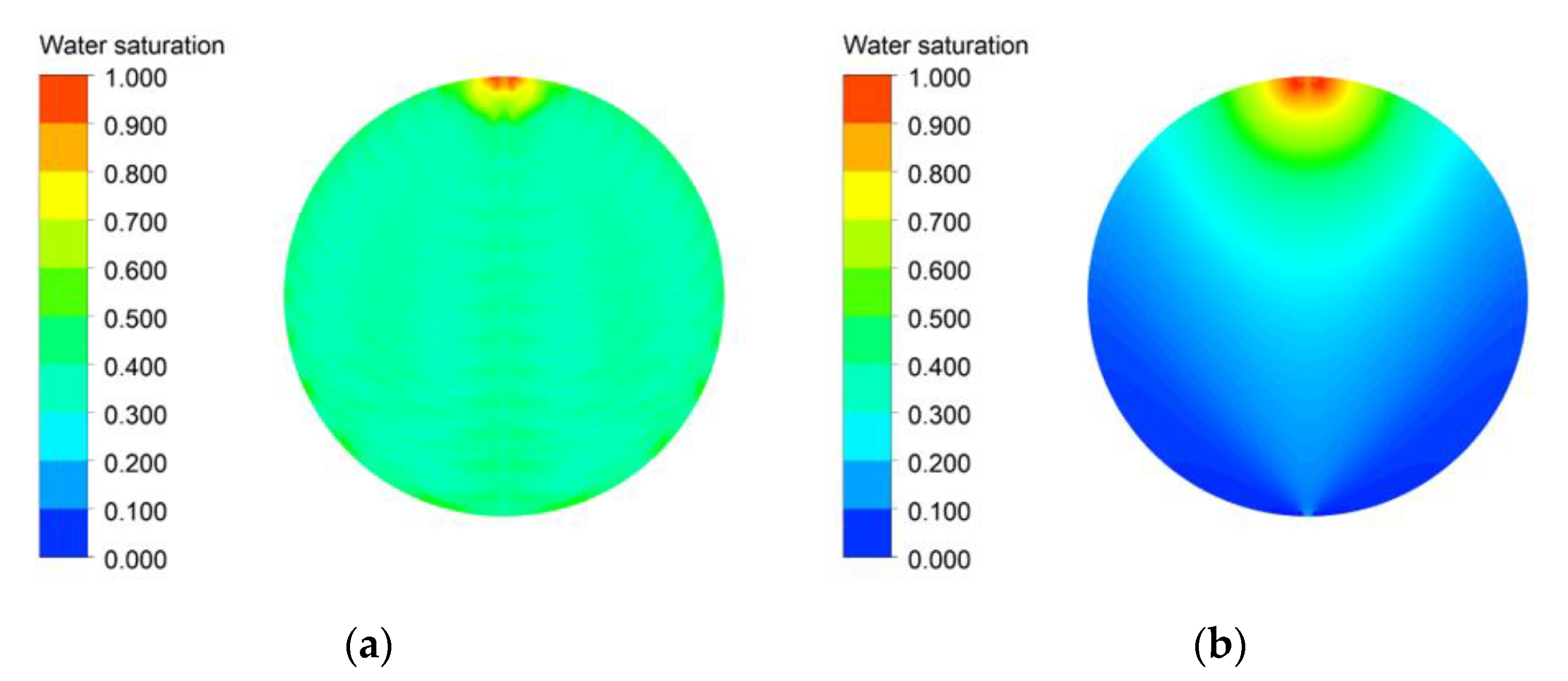
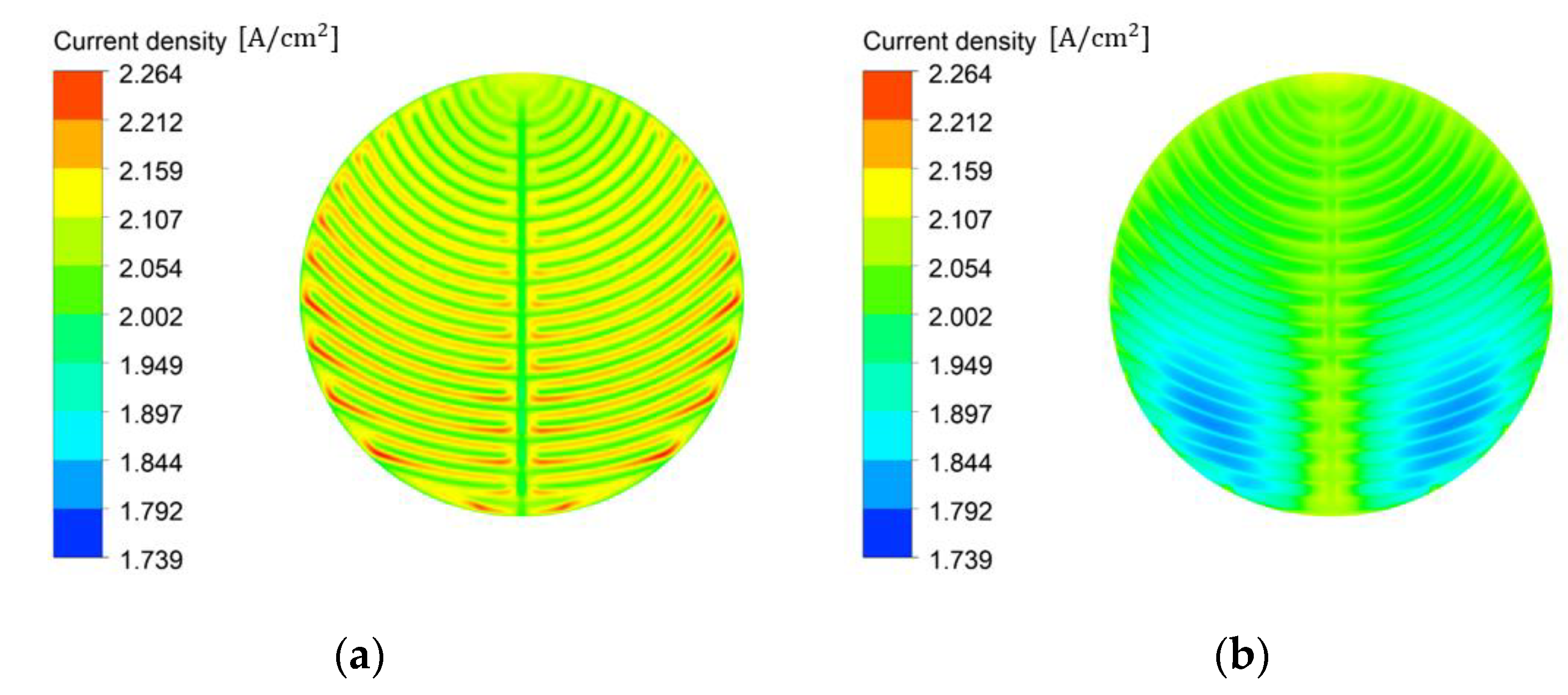
3.2. Effects of the Flow Field
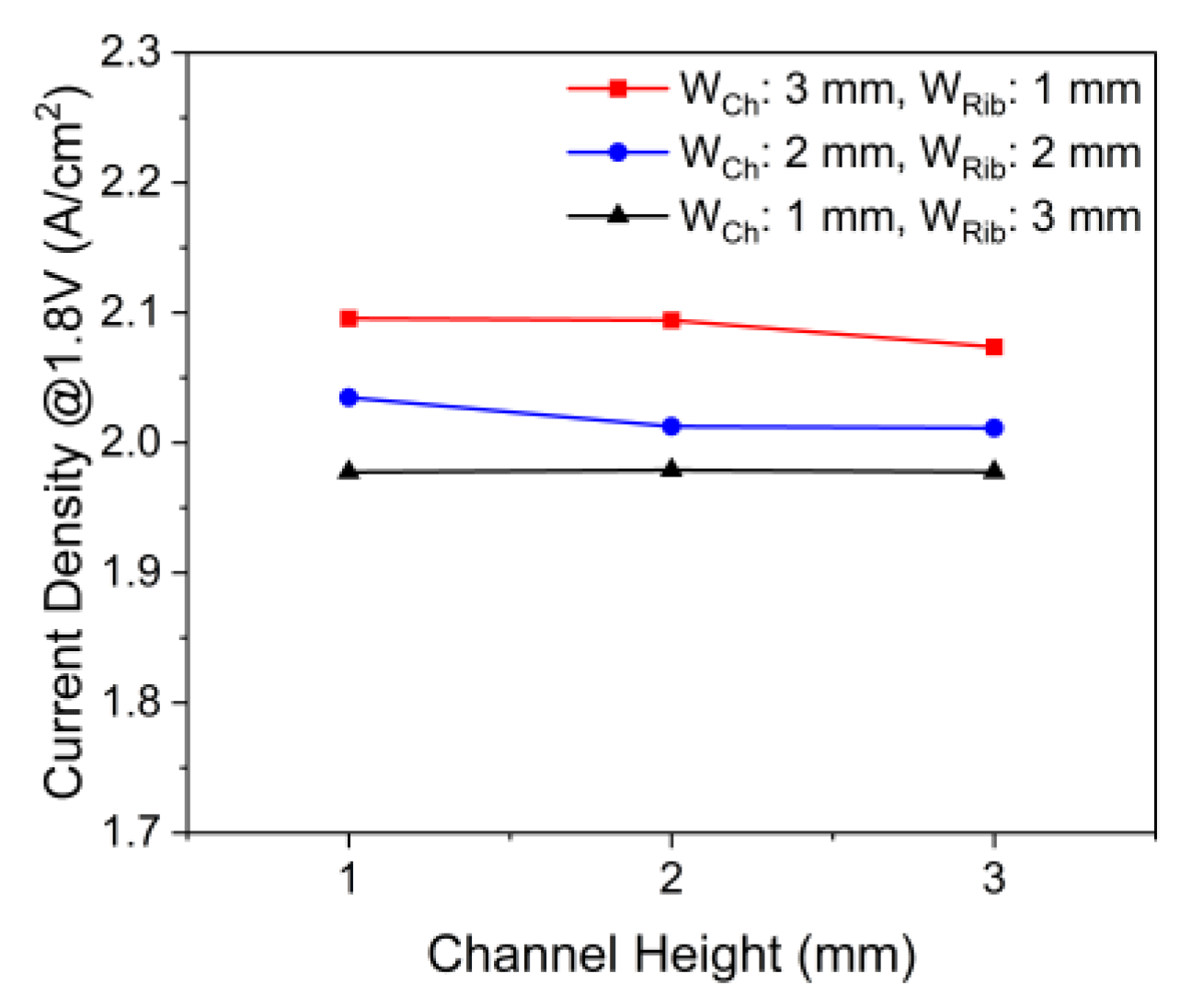
3.3. Effects of the Channel Geometry
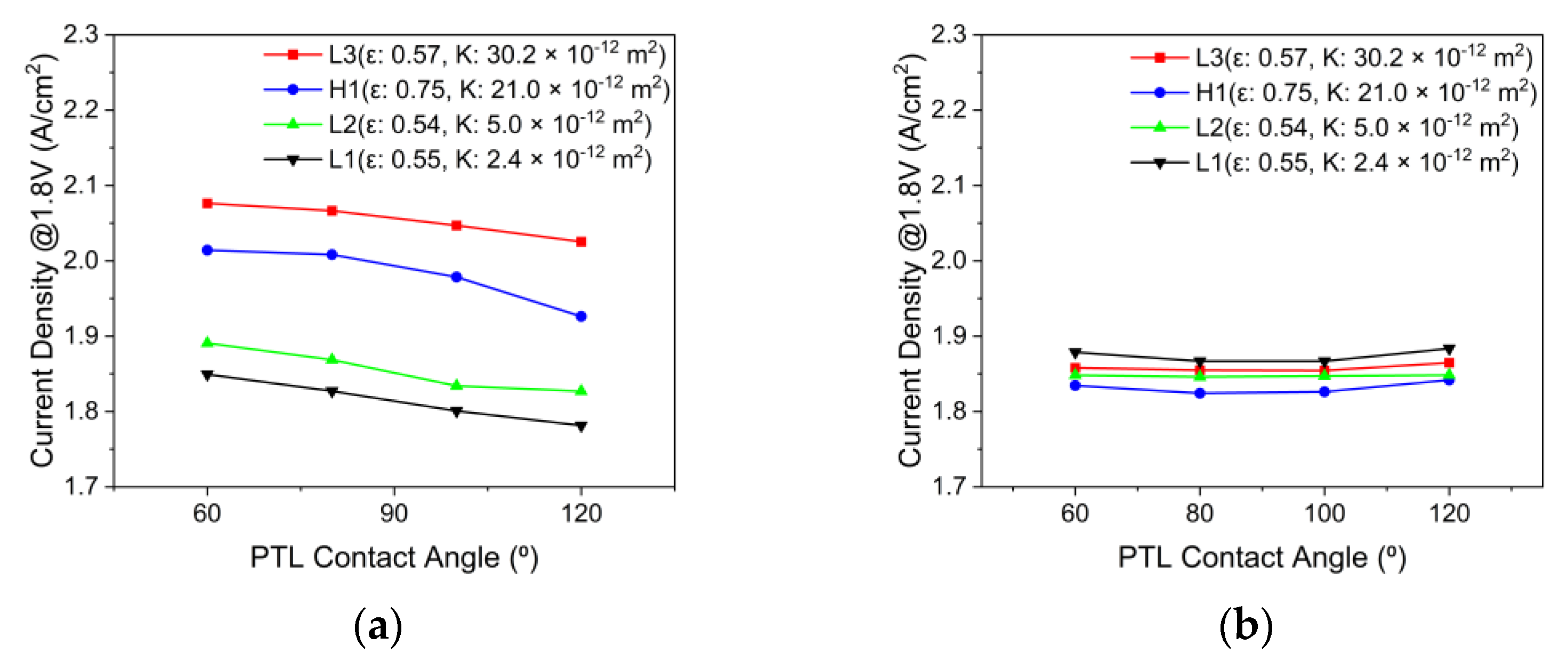
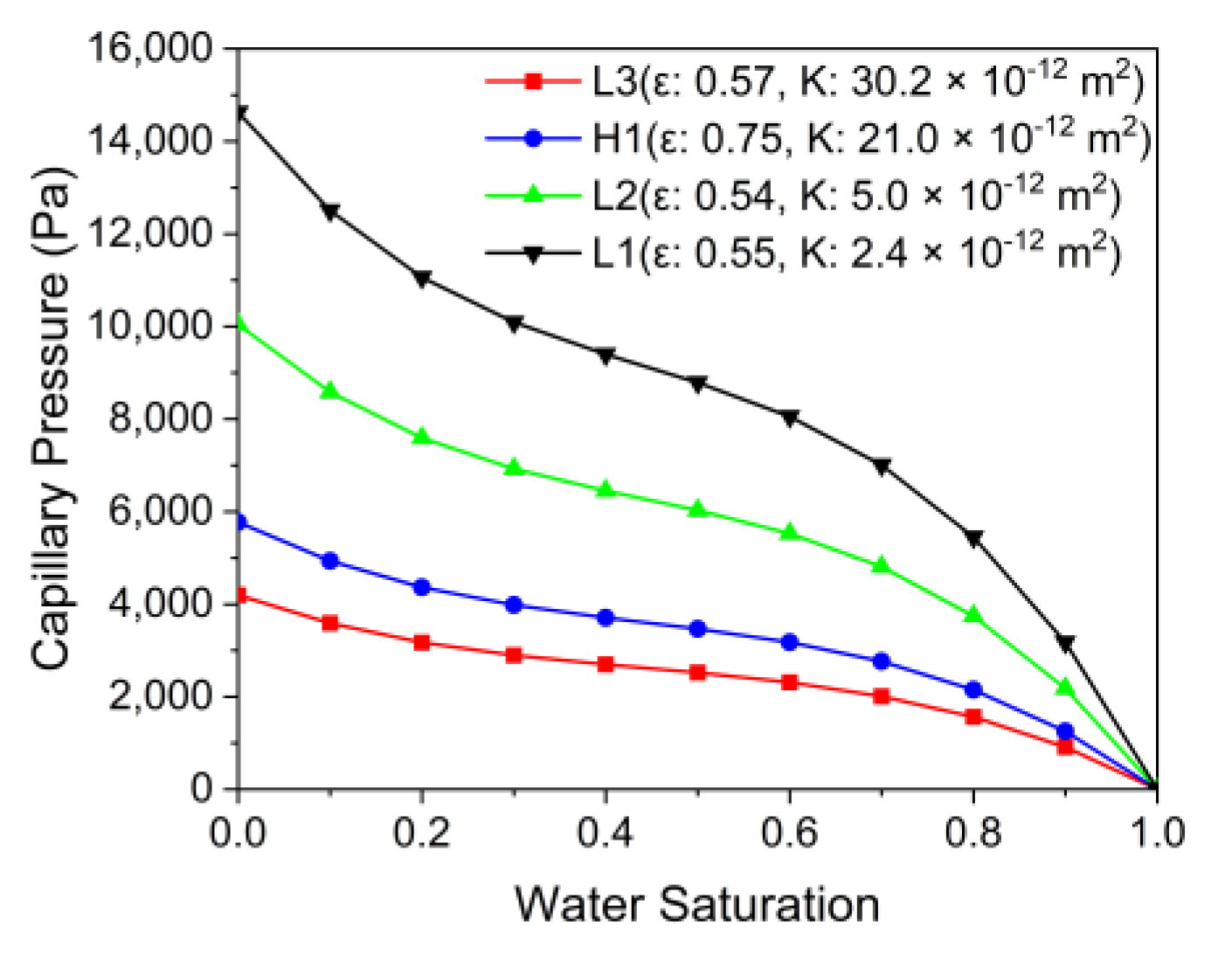
3.4. Effects of the PTL Properties
3.5. Effects of the Gravity
3.6. Effects of the Flow Rate
4. Conclusions
- The main factor in the performance differences is the bubble overvoltage caused by the lower water saturation on the active area. A lower stoichiometric ratio increased the bubble overvoltage, and the polarization curve results of the simulation and experiment showed high consistency.
- At a low stoichiometric ratio, a high and uniform current density was obtained using the channel flow field. Moreover, the performance can be improved by reducing the distance between the channel and area under the rib where the water and oxygen must flow, reducing the resistance of the flow in the PTL. The direction of gravity helped improve performance when setting the direction to help water flow from the channel to the active area or from the inlet to the outlet.
- At a high stoichiometric ratio, the performances of channel and PTL flow fields become similar because of the gradually disappearing bubble overvoltage. However, a significantly large pressure drop must be considered in the PTL flow field.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chu, S.; Cui, Y.; Liu, N. The path towards sustainable energy. Nat. Mater. 2016, 16, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.A. A realizable renewable energy future. Science 1999, 285, 687–689. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, J.D.; Camargo, M.; Commenge, J.M.; Falk, L.; Gil, I.D. Trends in design of distributed energy systems using hydrogen as energy vector: A systematic literature review. Int. J. Hydrog. Energy 2019, 4, 9486–9504. [Google Scholar] [CrossRef]
- Kazim, A.; Veziroglu, T.N. Utilization of solar-hydrogen energy in the UAE to maintain its share in the world energy market for the 21st century. Renew. Energy 2001, 24, 259–274. [Google Scholar] [CrossRef]
- Shiva Kumar, S.; Himabindu, V. Hydrogen production by PEM water electrolysis—A review. Mater. Sci. Energy Technol. 2019, 2, 442–454. [Google Scholar] [CrossRef]
- Uddin, M.N.; Nageshkar, V.V.; Asmatulu, R. Improving water-splitting efficiency of water electrolysis process via highly conductive nanomaterials at lower voltages. Energy Ecol. Environ. 2020, 5, 108–117. [Google Scholar] [CrossRef]
- Schmidt, O.; Gambhir, A.; Staffell, I.; Hawkes, A.; Nelson, J.; Few, S. Future cost and performance of water electrolysis: An expert elicitation study. Int. J. Hydrog. Energy 2017, 42, 30470–30492. [Google Scholar] [CrossRef]
- Teuku, H.; Alshami, I.; Goh, J.; Masdar, M.S.; Loh, K.S. Review on bipolar plates for low-temperature polymer electrolyte membrane water electrolyzer. Int. J. Energy Res. 2021, 45, 20583–20600. [Google Scholar] [CrossRef]
- Carmo, M.; Fritz, D.L.; Mergel, J.; Stolten, D. A comprehensive review on PEM water electrolysis. Int. J. Hydrog. Energy 2013, 38, 4901–4934. [Google Scholar] [CrossRef]
- Bertuccioli, L.; Chan, A.; Hart, D.; Lehner, F.; Madden, B.; Standen, E. Study on development of water electrolysis in the EU, Fuel Cells and hydrogen Joint Undertaking. Fuel Cells Hydrog. Jt. Undert. 2014, 1, 1–160. Available online: https://www.fch.europa.eu/sites/default/files/FCHJUElectrolysisStudy_FullReport(ID 199214).pdf (accessed on 20 April 2015).
- Selamet, F.; Acar, M.C.; Mat, M.D.; Kaplan, Y. Effects of operating parameters on the performance of a high-pressure proton exchange membrane electrolyzer. Int. J. Energy Res. 2013, 37, 457–467. [Google Scholar] [CrossRef]
- Lee, C.H.; Banerjee, R.; Arbabi, F.; Hinebaugh, J.; Bazylak, A. Porous transport layer related mass transport losses in polymer electrolyte membrane electrolysis—A review. In Proceedings of the ASME 2016 14th International Conference on Nanochannels, Microchannels, and Minichannels collocated with the ASME 2016 Heat Transfer Summer Conference and the ASME 2016 Fluids Engineering Division Summer Meeting, Washington, DC, USA, 10–14 July 2016. [Google Scholar] [CrossRef]
- Suermann, M.; Schmidt, T.J.; Büchi, F.N. Investigation of Mass Transport Losses in Polymer Electrolyte Electrolysis Cells. Ecs Trans. 2015, 69, 1141. [Google Scholar] [CrossRef]
- Sun, S.; Xiao, Y.; Liang, D.; Shao, Z.; Yu, H.; Hou, M.; Yi, B. Behaviors of a proton exchange membrane electrolyzer under water starvation. RSC Adv. 2015, 5, 14506–14513. [Google Scholar] [CrossRef]
- Maier, M.; Smith, K.; Dodwell, J.; Hinds, G.; Shearing, P.R.; Brett, D.J.L. Mass Transport in PEM Water Electrolysers: A Review. Int. J. Hydrog. Energy 2022, 47, 30–56. [Google Scholar] [CrossRef]
- Harrison, K.W.; Martin, G.D.; Ramsden, T.G.; Kramer, W.E.; Novachek, F.J. The wind-to-hydrogen project: Operational experience, performance testing, and systems integration. Hydrog. Energy Mix. 2011, 89–168. [Google Scholar] [CrossRef]
- Immerz, C.; Bensmann, B.; Trinke, P.; Suermann, M.; Hanke-Rauschenbach, R. Local Current Density and Electrochemical Impedance Measurements within 50 cm Single-Channel PEM Electrolysis Cell. J. Electrochem. Soc. 2018, 165, F1292–F1299. [Google Scholar] [CrossRef]
- Olesen, A.C.; Frensch, S.H.; Kær, S.K. Towards uniformly distributed heat, mass and charge: A flow field design study for high pressure and high current density operation of PEM electrolysis cells. Electrochim. Acta 2019, 293, 476–495. [Google Scholar] [CrossRef]
- Nie, J.; Chen, Y.; Cohen, S.; Carter, B.D.; Boehm, R.F. Numerical and experimental study of three-dimensional fluid flow in the bipolar plate of a PEM electrolysis cell. Int. J. Therm. Sci. 2009, 48, 1914–1922. [Google Scholar] [CrossRef]
- Majasan, J.O.; Cho, J.I.S.; Dedigama, I.; Tsaoulidis, D.; Shearing, P.; Brett, D.J.L. Two-Phase Flow Behaviour and Performance of Polymer Electrolyte Membrane Electrolysers: Electrochemical and Optical Characterisation. Int. J. Hydrog. Energy 2018, 43, 15659–15672. [Google Scholar] [CrossRef]
- Ruiz, D.D.H.; Sasmito, A.P.; Shamim, T. Numerical Investigation of the High Temperature PEM Electrolyzer: Effect of Flow Channel Configurations. ECS Trans. 2013, 58, 99. [Google Scholar] [CrossRef]
- Kang, Z.; Alia, S.M.; Young, J.L.; Bender, G. Effects of Various Parameters of Different Porous Transport Layers in Proton Exchange Membrane Water Electrolysis. Electrochim. Acta 2020, 354, 136641. [Google Scholar] [CrossRef]
- Sazali, N.; Salleh, W.N.W.; Jamaludin, A.S.; Razali, M.N.M. New Perspectives on Fuel Cell Technology. Membranes 2020, 10, 99. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Liang, X.; Wang, L.; Sun, K.; Wang, Y.; Xie, Z.; Wu, Q.; Bai, X.; Hamdy, M.S.; Chen, H.; et al. Status and Perspectives of Key Materials for PEM Electrolyzer. Nano Res. Energy 2022, 1, e9120032. [Google Scholar] [CrossRef]
- Müller, M.; Carmo, M.; Glüsen, A.; Hehemann, M.; Saba, S.; Zwaygardt, W.; Stolten, D. Water Management in Membrane Electrolysis and Options for Advanced Plants. Int. J. Hydrog. Energy 2019, 44, 10147–10155. [Google Scholar] [CrossRef]
- Kulkarni, D.; Huynh, A.; Satjaritanun, P.; O’Brien, M.; Shimpalee, S.; Parkinson, D.; Shevchenko, P.; DeCarlo, F.; Danilovic, N.; Ayers, K.E.; et al. Elucidating Effects of Catalyst Loadings and Porous Transport Layer Morphologies on Operation of Proton Exchange Membrane Water Electrolyzers. Appl. Catal. B Environ. 2022, 308, 121213. [Google Scholar] [CrossRef]
- O’hayre, R.; Cha, S.W.; Colella, W.; Prinz, F.B. Fuel Cell Fundamentals, 3rd ed.; Wiley: Hoboken, NY, USA, 2016; pp. 48, 98, 124, 139, 140. [Google Scholar]
- Ojong, E.T.; Kwan, J.T.H.; Nouri-Khorasani, A.; Bonakdarpour, A.; Wilkinson, D.P.; Smolinka, T. Development of an Experimentally Validated Semi-Empirical Fully-Coupled Performance Model of a PEM Electrolysis Cell with a 3-D Structured Porous Transport Layer. Int. J. Hydrog. Energy 2017, 42, 25831–25847. [Google Scholar] [CrossRef]
- Marangio, F.; Santarelli, M.; Calì, M. Theoretical Model and Experimental Analysis of a High Pressure PEM Water Electrolyser for Hydrogen Production. Int. J. Hydrog. Energy 2009, 34, 1143–1158. [Google Scholar] [CrossRef]
- Rajora, A.; Haverkort, J.W. An Analytical Model for Liquid and Gas Diffusion Layers in Electrolyzers and Fuel Cells. J. Electrochem. Soc. 2021, 168, 034506. [Google Scholar] [CrossRef]
- Lopata, J.; Kang, Z.; Young, J.; Bender, G.; Weidner, J.W.; Shimpalee, S. Effects of the Transport/Catalyst Layer Interface and Catalyst Loading on Mass and Charge Transport Phenomena in Polymer Electrolyte Membrane Water Electrolysis Devices. J. Electrochem. Soc. 2020, 167, 064507. [Google Scholar] [CrossRef]
- ANSYS Inc. Ansys Fluent Theory Guide; ANSYS Inc.: Canonsburg, PA, USA, 2021; Volume 15317, pp. 724–746. [Google Scholar]
- Schuler, T.; De Bruycker, R.; Schmidt, T.J.; Büchi, F.N. Polymer Electrolyte Water Electrolysis: Correlating Porous Transport Layer Structural Properties and Performance: Part I. Tomographic Analysis of Morphology and Topology. J. Electrochem. Soc. 2019, 166, F270–F281. [Google Scholar] [CrossRef]
- Leverett, M.C. Capillary Behavior in Porous Solids. Trans. AIME 1941, 142, 152–169. [Google Scholar] [CrossRef]
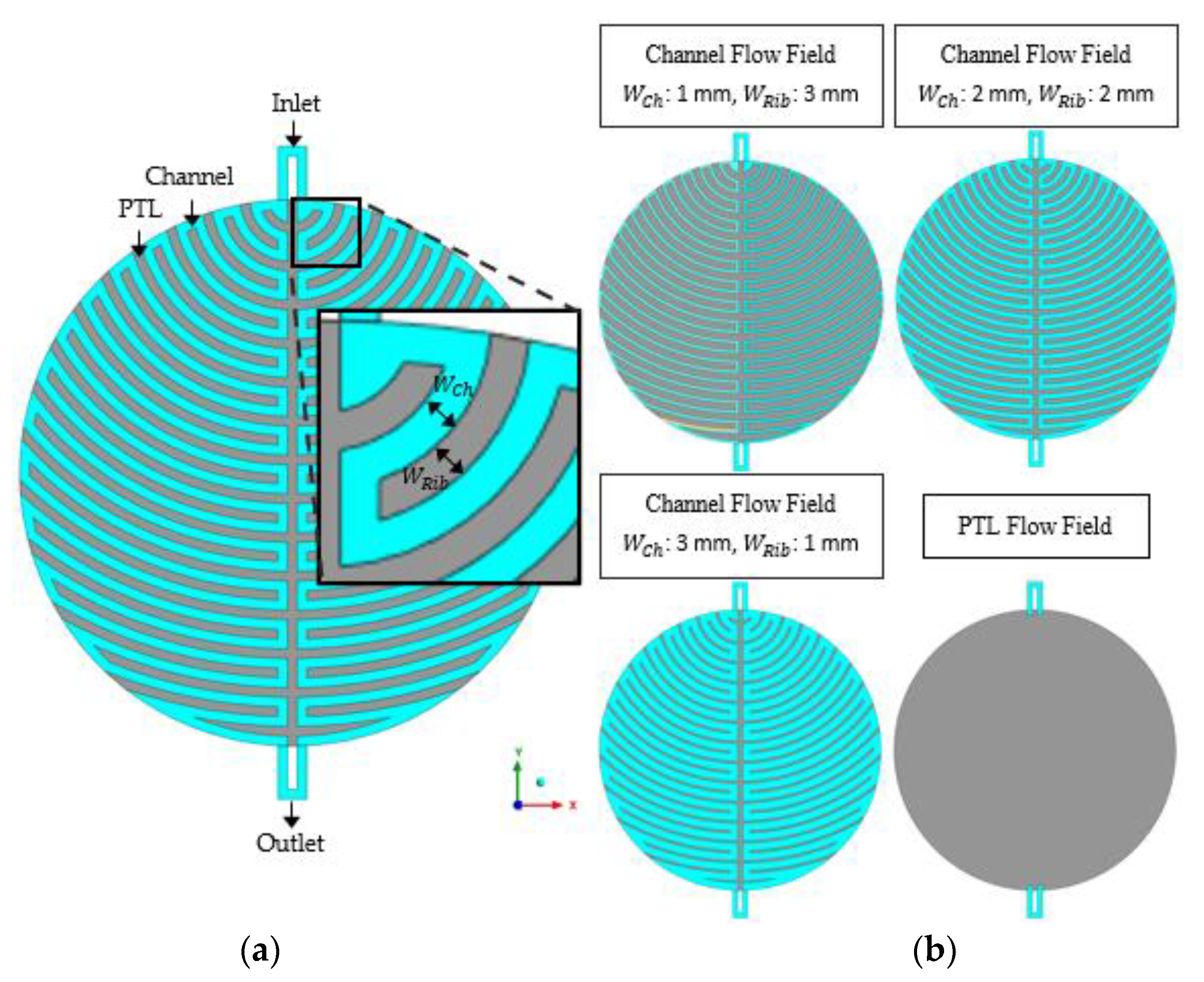




| Parameter | Symbol | Value |
|---|---|---|
| Channel width | 1 mm, 2 mm, 3 mm | |
| Rib width | 3 mm, 2 mm, 1 mm | |
| Channel height | 1 mm, 2 mm, 3 mm | |
| PTL thickness | 0.25 mm |
| Parameter | Symbol | Value |
|---|---|---|
| Reversible voltage in standard state | 1.229 V | |
| Amount of change in entropy | 163.23 J/K | |
| Faraday’s constant | 96,485 C/mol | |
| Temperature in standard state | 298.15 K | |
| Universal gas constant | 8.314 J/K∙mol | |
| Tafel coefficient | 1.328 | |
| Exchange current density | 1.6 × 10−3 A/cm2 | |
| Membrane thickness | 0.175 mm | |
| Water content | 22 |
| Parameter | Symbol | Value |
|---|---|---|
| Water density | 985.64 kg/m3 | |
| Oxygen density | 1.191 kg/m3 | |
| Water viscosity | 0.000504 kg/m∙s | |
| Oxygen viscosity | 0.000022 kg/m∙s | |
| Surface tension coefficient | 0.063 N/m | |
| Gravity direction | X+, X-, Y+, Y-, Z+, Z- |
| Parameter | Symbol | Value | |||
|---|---|---|---|---|---|
| Type | L1 | L2 | L3 | H1 | |
| Porosity | 0.55 | 0.54 | 0.57 | 0.75 | |
| Permeability | 2.4 × 10−12 m2 | 5.0 × 10−12 m2 | 21.0 × 10−12 m2 | 30.2 × 10−12 m2 | |
| Contact angle | 30°, 60°, 80°, 100°, 120° | ||||
| Parameter | Symbol | Value |
|---|---|---|
| Inlet water flow rate | 5 sccm, 10 sccm, 30 sccm, 100 sccm, 300 sccm | |
| Inlet water temperature | 328.15 K | |
| Outlet pressure | 0 Pa | |
| Cell temperature | 328.15 K | |
| Water molecular weight | 18 kg/kmol | |
| Oxygen molecular weight | 32 kg/kmol | |
| Operating voltage | 1.4 V, 1.5 V, 1.6 V, 1.7 V, 1.8 V | |
| Thermal neutral voltage | 1.48 V |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.; Lee, W.; Na, Y. Performance Comparison of Proton Exchange Membrane Water Electrolysis Cell Using Channel and PTL Flow Fields through Three-Dimensional Two-Phase Flow Simulation. Membranes 2022, 12, 1260. https://doi.org/10.3390/membranes12121260
Park S, Lee W, Na Y. Performance Comparison of Proton Exchange Membrane Water Electrolysis Cell Using Channel and PTL Flow Fields through Three-Dimensional Two-Phase Flow Simulation. Membranes. 2022; 12(12):1260. https://doi.org/10.3390/membranes12121260
Chicago/Turabian StylePark, Seongsoon, Woojung Lee, and Youngseung Na. 2022. "Performance Comparison of Proton Exchange Membrane Water Electrolysis Cell Using Channel and PTL Flow Fields through Three-Dimensional Two-Phase Flow Simulation" Membranes 12, no. 12: 1260. https://doi.org/10.3390/membranes12121260
APA StylePark, S., Lee, W., & Na, Y. (2022). Performance Comparison of Proton Exchange Membrane Water Electrolysis Cell Using Channel and PTL Flow Fields through Three-Dimensional Two-Phase Flow Simulation. Membranes, 12(12), 1260. https://doi.org/10.3390/membranes12121260







