X-ray Reflectivity Study of Polylysine Adsorption on the Surface of DMPS Monolayers
Abstract
1. Introduction
2. Materials and Methods
3. Results
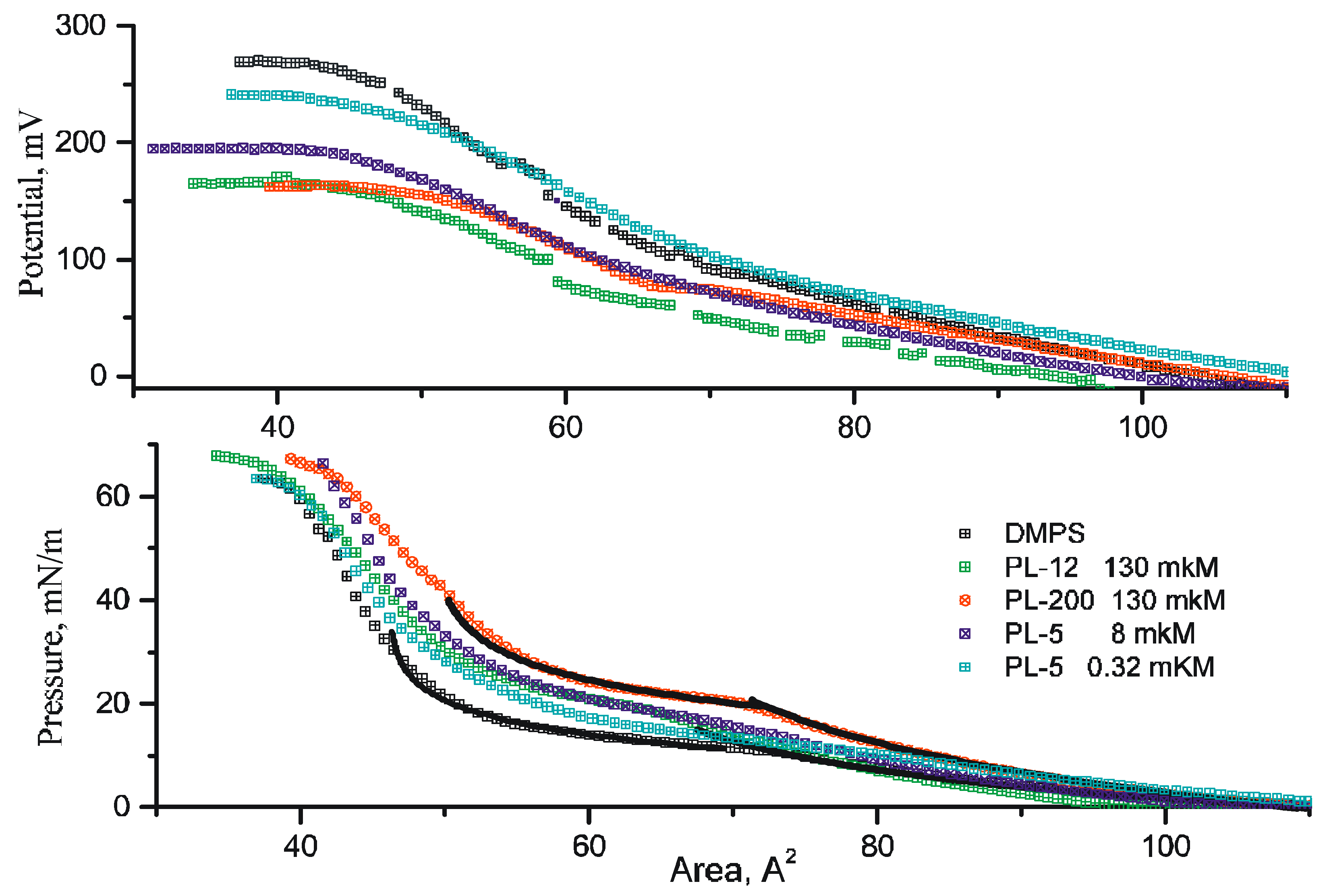
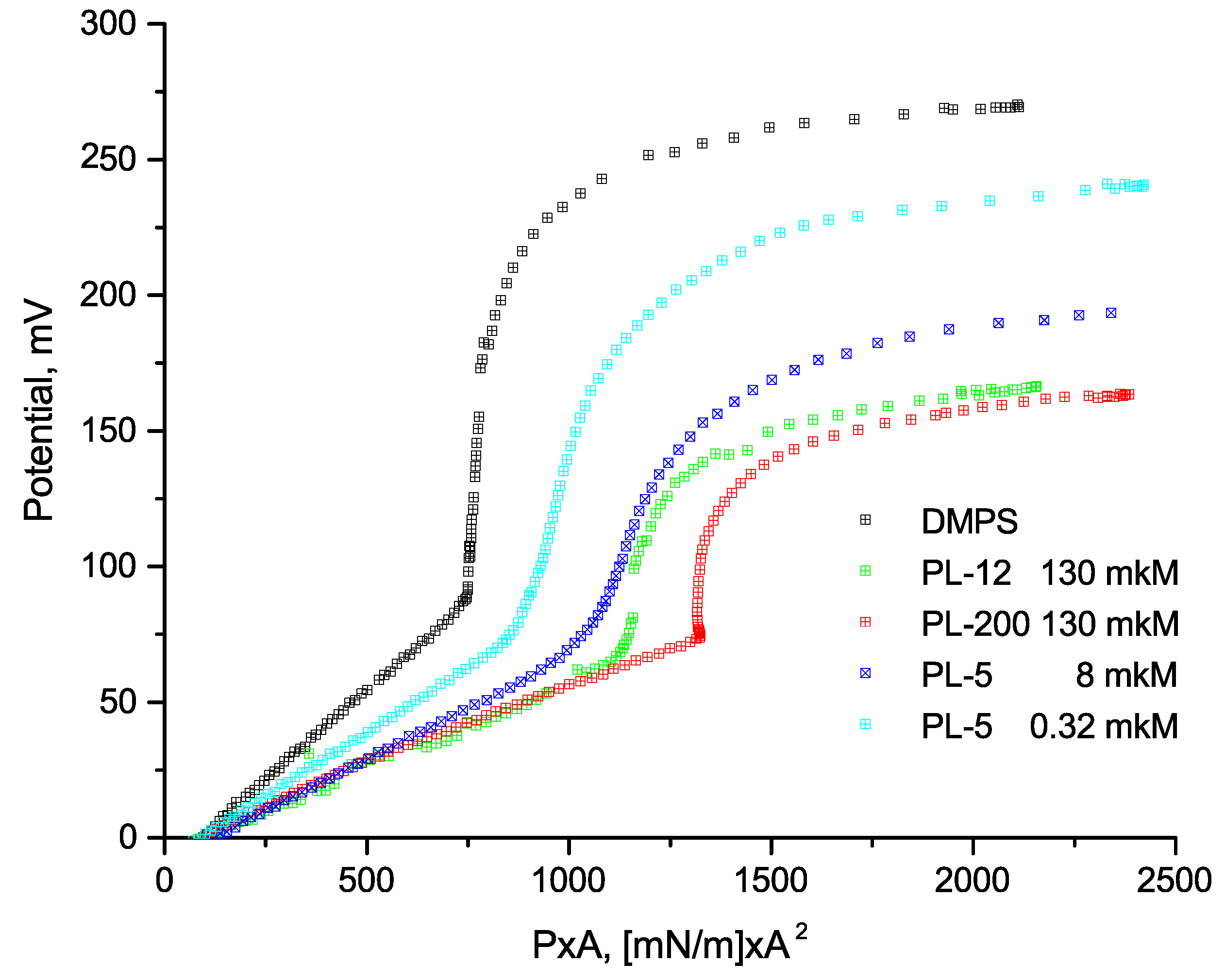
3.1. Compression of Langmuir Monolayers
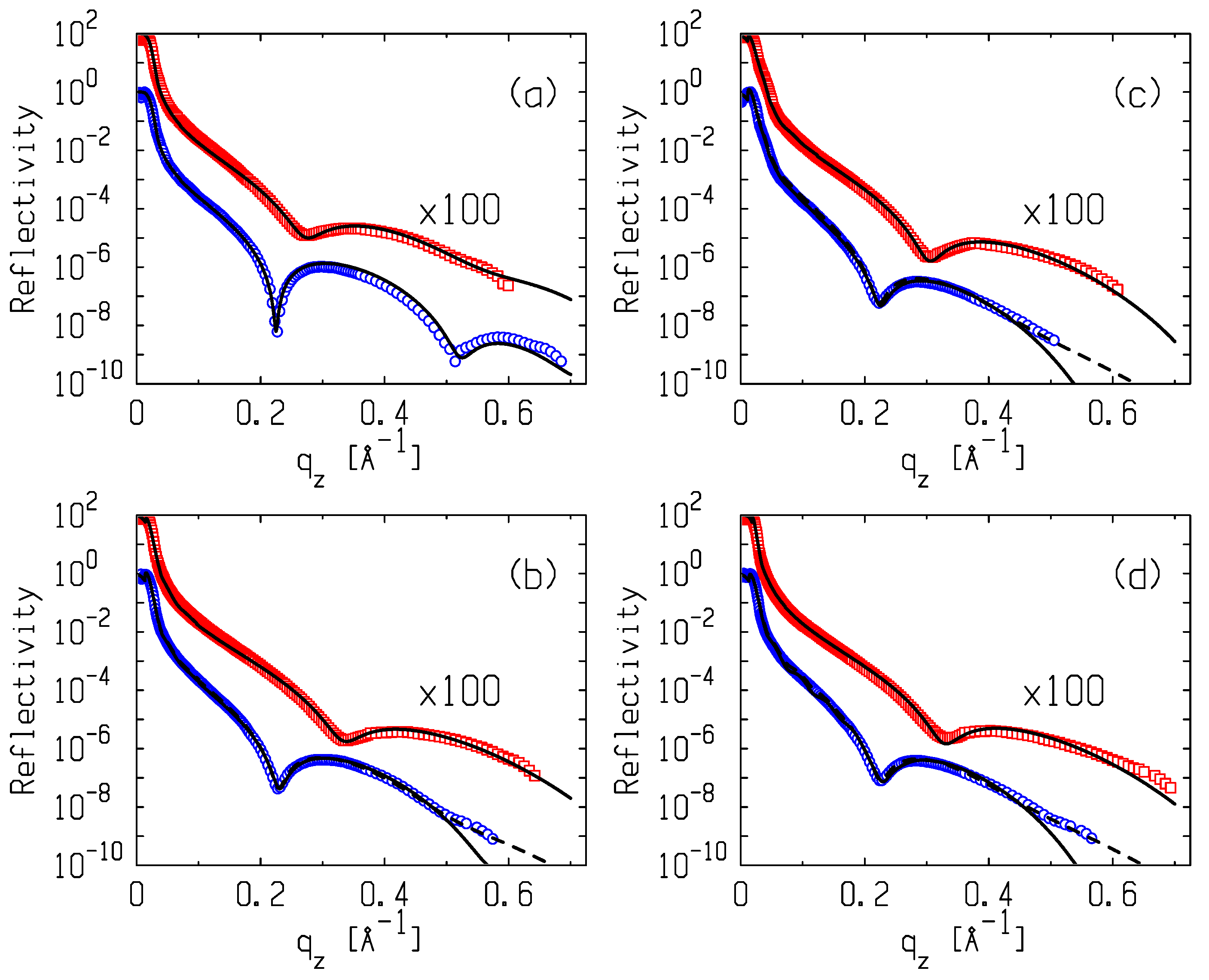
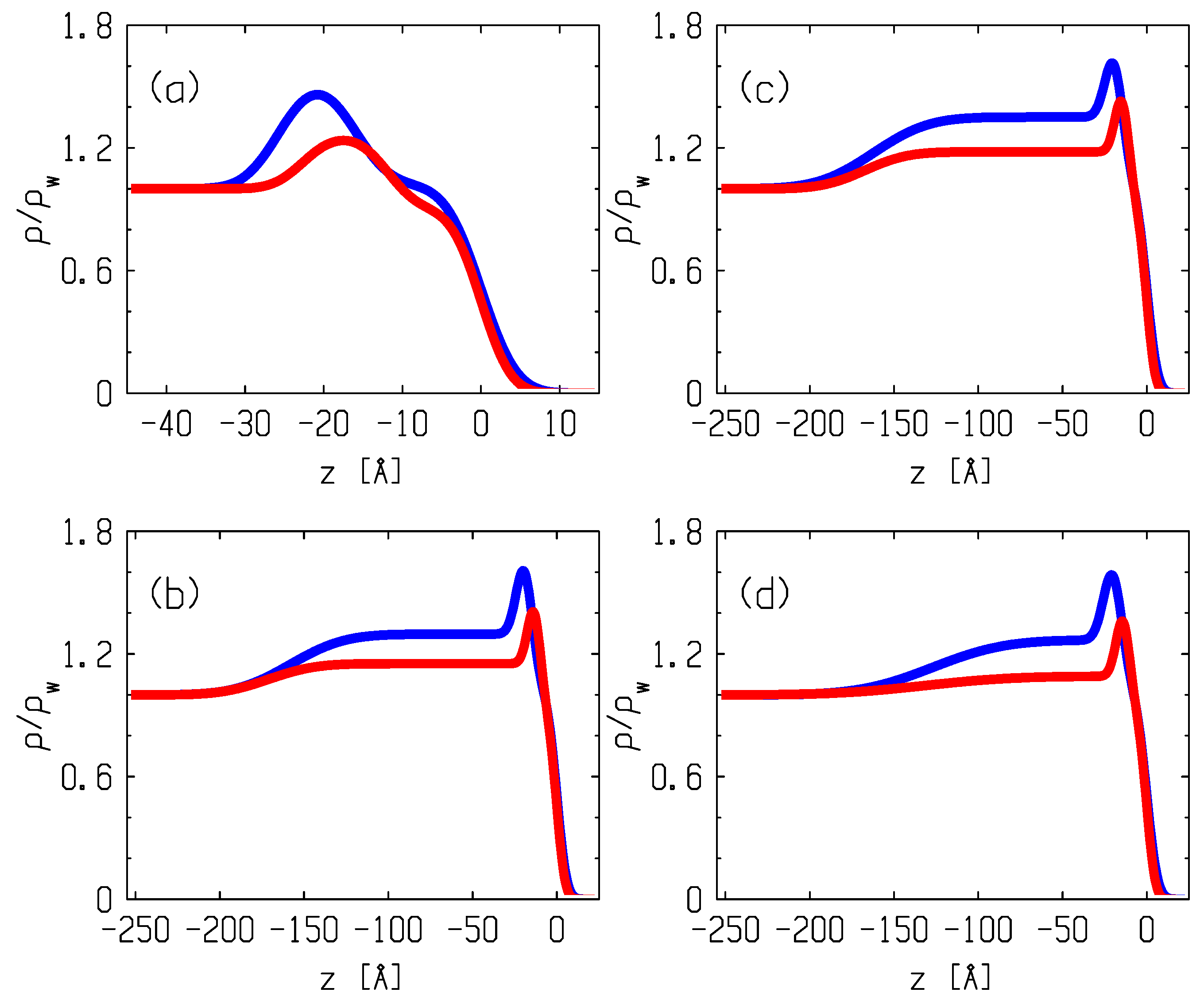
3.2. X-ray Reflectometry Data and Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Harries, D.; May, S.; Ben Shaul, A. Adsorption of charged macromolecules on mixed fluid membranes. Colloids Surf. A Physicochem. Eng. Asp. 2002, 208, 41–50. [Google Scholar] [CrossRef]
- Kim, J.; Mosior, M.; Chung, L.A.; Wu, H.; McLaughlin, S. Binding of peptides with basic residues to membranes containing acidic phospholipids. Biophys. J. 1991, 60, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Vorobyov, I.; Allen, T.W. On the role of anionic lipids in charged protein interactions with membranes. Biochim. Biophys. Acta 2010, 1808, 1673–1683. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y. Investigation of organic films by atomic force microscopy: Structural, nanotribological and electrical properties. Surf. Sci. Rep. 2011, 66, 379–393. [Google Scholar] [CrossRef]
- Wu, Z.; Cui, Q.; Yethiraj, A. Why Do Arginine and Lysine Organize Lipids Differently? Insights from Coarse-Grained and Atomistic Simulations. J. Phys. Chem. B 2013, 117, 12145–12156. [Google Scholar] [CrossRef] [PubMed]
- Doltchinkova, V.; Vitkova, V. Polylysine effect on thylakoid membranes. Biophys. Chem. 2020, 266, 106440. [Google Scholar] [CrossRef]
- Ikeda, T.; Tazuke, S.; Watanabe, B. Interaction of biologically active molecules with phospholipid membranes I. Fluorescence depolarisation studies on the effect of polymeric biocide bearing biguanin groups in the main chain. Biochem. Biophys. Acta 1983, 735, 380–386. [Google Scholar] [CrossRef]
- Rink, T.; Bartel, H.; Jung, G.; Bannwarth, W.; Boheim, G. Effects of polycations on ion channels formed by neutral and negatively charged alamethicins. Eur. Biophys. J. 1994, 23, 155–165. [Google Scholar] [CrossRef]
- Cetuk, H.; Anishkin, A.; Scott, A.J.; Rempe, S.B.; Ernst, R.K.; Sukharev, S. Partitioning of Seven Different Classes of Antibiotics into LPS Monolayers Supports Three Different Permeation Mechanisms through the Outer Bacterial Membrane. Langmuir 2021, 37, 1372–1385. [Google Scholar] [CrossRef]
- Timofeeva, L.M.; Kleshcheva, N.A.; Shleeva, M.O.; Filatova, M.P.; Simonova, Y.A.; Ermakov, Y.A.; Kaprelyants, A.S. Nonquaternary poly(diallylammonium) polymers with different amine structure and their biocidal effect on Mycobacterium tuberculosis and Mycobacterium smegmatis. Appl. Microbiol. Biotechnol. 2015, 99, 2557–2571. [Google Scholar] [CrossRef]
- Alvares, D.S.; Cabrera, M.P.S.; Neto, J.R. Strategies for Exploring Electrostatic and Nonelectrostatic Contributions to the Interaction of Helical Antimicrobial Peptides with Model Membranes. In Advances in Biomembranes and Lipid Self-Assembly; Elsevier Inc.: London, UK, 2016; pp. 43–73. [Google Scholar]
- Ermakov, Y.A.; Kamaraju, K.; Sengupta, K.; Sukharev, S. Gadolinium Ions Block Mechanosensitive Channels by Altering the Packing and Lateral Pressure of Anionic Lipids. Biophys. J. 2010, 98, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Ermakov, Y.A.; Asadchikov, V.E.; Roshchin, B.S.; Volkov, Y.O.; Khomich, D.A.; Nesterenko, A.M.; Tikhonov, A.M. Comprehensive Study of the LE-LC phase Transition in DMPS monolayers: Surface Pressure, Volta Potential, X-ray Reflectivity, and MD Modeling. Langmuir 2019, 35, 12326–12338. [Google Scholar] [CrossRef] [PubMed]
- Marukovich, N.I.; Nesterenko, A.M.; Ermakov, Y.A. Structural factors of lysine and polylysine interaction with lipid membranes. Biochem. (Mosc.) Suppl. Ser. A Membr. Cell Biol. 2015, 9, 40–47. [Google Scholar] [CrossRef]
- Molotkovsky, R.J.; Ermakov, Y.A. Two Possible Approaches to Quantitative Analysis of Compression Diagrams of Lipid Monolayers. Biochem. (Mosc.) Suppl. Ser. A Membr. Cell Biol. 2015, 9, 48–52. [Google Scholar] [CrossRef]
- Finogenova, O.A.; Batishchev, O.V.; Ermakov, Y.A. Polylysine adsorption at charged surface of lipid membranes and mica. Eur. Biophys. J. 2009, 38 (Suppl. S1), 574. [Google Scholar]
- Finogenova, O.A.; Filinsky, D.V.; Ermakov, Y.A. Electrostatic effects upon adsorption and desorption of polylysines on the surface of lipid membranes of different composition. Biochem. (Mosc.) Suppl. Ser. A Membr. Cell Biol. 2008, 2, 181–188. [Google Scholar] [CrossRef]
- Ermakov, Y.A.; Asadchikov, V.E.; Volkov, Y.O.; Nuzhdin, A.D.; Roshchin, B.S.; Honkimaki, V.; Tikhonov, A.M. Electrostatic and Structural Effects at the Adsorption of Polylysine on the Surface of the DMPS Monolayer. J. Exp. Theor. Phys. Lett. 2019, 109, 334–339. [Google Scholar] [CrossRef]
- Asadchikov, V.E.; Tikhonov, A.M.; Volkov, Y.O.; Roshchin, B.S.; Ermakov, Y.A.; Rudakova, E.B.; D’Yachkova, I.G.; Nuzhdin, A.D. X-ray study of the structure of phospholipid monolayers on the water surface. J. Exp. Theor. Phys. Lett. 2017, 106, 534–539. [Google Scholar] [CrossRef]
- Tikhonov, A.M.; Asadchikov, V.E.; Volkov, Y.O.; Roshchin, B.S.; Ermakov, Y.A. X-ray Reflectometry of DMPS Monolayers on a Water Substrate. J. Exp. Theor. Phys. 2017, 125, 1051–1057. [Google Scholar] [CrossRef][Green Version]
- Braslau, A.; Deutsch, M.; Pershan, P.S.; Weiss, A.H.; Als-Nielsen, J.; Bohr, J. Surface Roughness of Water Measured by X-ray Reflectivity. Phys. Rev. Lett. 1985, 54, 114–117. [Google Scholar] [CrossRef]
- Delcea, M.; Helm, C.A. X-ray and Neutron Reflectometry of Thin Films at Liquid Interfaces. Langmuir 2019, 35, 8519–8530. [Google Scholar] [CrossRef] [PubMed]
- Kaganer, V.M.; Mohwald, H.; Dutta, P. Structure and phase transitions in Langmuir monolayers. Rev. Mod. Phys. 1999, 71, 779–819. [Google Scholar] [CrossRef]
- Malkova, S.; Long, F.; Stahelin, R.V.; Pingali, S.V.; Murray, D.; Cho, W.H.; Schlossman, M.L. X-ray Reflectivity Studies of cPLA2α-C2 Domains Adsorbed onto Langmuir Monolayers of SOPC. Biophys. J. 2005, 89, 1861–1873. [Google Scholar] [CrossRef] [PubMed]
- Bitto, E.; Li, M.; Tikhonov, A.M.; Schlossman, M.L.; Cho, W. Mechanism of Annexin I-Mediated Membrane Aggregation. Biochemistry 2000, 39, 13469–13477. [Google Scholar] [CrossRef] [PubMed]
- Marukovich, N.; McMurray, M.; Finogenova, O.; Nesterenko, A.; Batishchev, O.; Ermakov, Y. Interaction of polylysines with the surface of lipid membranes: The electrostatic and structural aspects. In Advances in Planar Lipid Bilayers and Liposomes; Iglic, A., Kulkarni, C., Rappolt, M., Eds.; Elsevier: Amsterdam, The Netherlands; London, UK; San Diego, CA, USA; Academic Press: Amsterdam, The Netherlands; London, UK; San Diego, CA, USA, 2013; pp. 139–166. [Google Scholar]
- Honkimaki, V.; Reichert, H.; Okasinski, J.S.; Dosch, H. X-ray optics for liquid surface/interface spectrometers. J. Synchrotron Radiat. 2006, 13, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Tikhonov, A.M.; Asadchikov, V.E.; Volkov, Y.O.; Nuzhdin, A.D.; Roshchin, B.S. Thermostatic Chamber for X-ray Studies of Thin Film Structures at Liquid Substrates. Instrum. Exp. Tech. 2021, 64, 172–176. [Google Scholar] [CrossRef]
- Ivashkov, O.V.; Sybachin, A.V.; Efimova, A.A.; Pergushov, D.V.; Orlov, V.N.; Schmalz, H.; Yaroslavov, A.A. The Influence of the Chain Length of Polycations on their Complexation with Anionic Liposomes. ChemPhysChem 2015, 16, 2849–2853. [Google Scholar] [CrossRef] [PubMed]
- Ermakov, Y.A. Boundary Potential and the Energy of Lipid Monolayer Compression at the Liquid Expanded State. Biochem. (Mosc.) Suppl. Ser. A Membr. Cell Biol. 2021, 15, 130–141. [Google Scholar] [CrossRef]
- Sinha, S.K.; Sirota, E.B.; Garoff, S.; Stanley, H.B. X-ray and neutron scattering from rough surfaces. Phys. Rev. B 1988, 38, 2297–2312. [Google Scholar] [CrossRef]
- Buff, F.P.; Lovett, R.A.; Stillinger, F.H., Jr. Interface Density Profile for Fluids in the Critical Region. Phys. Rev. Lett. 1965, 15, 621–623. [Google Scholar] [CrossRef]
- Braslau, A.; Pershan, P.S.; Swislow, G.; Ocko, B.M.; Als-Nielsen, J. Capillary waves on the surface of simple liquids measured by x-ray reflectivity. Phys. Rev. A 1988, 38, 2457–2473. [Google Scholar] [CrossRef] [PubMed]
- Tikhonov, A.M.; Schlossman, M.L. Vaporization and layering of alkanols at the oil/water interface. Condens. Matter 2007, 19, 375101. [Google Scholar] [CrossRef][Green Version]
- Tikhonov, A.M. Ion-size effect at the surface of a silica hydrosol. J. Chem. Phys. 2009, 130, 024512. [Google Scholar] [CrossRef] [PubMed]
- Molotkovsky, R.J.; Galimzyanov, T.R.; Khomich, D.A.; Nesterenko, A.M.; Ermakov, Y.A. Inhomogeneity of polylysine adsorption layer on the lipid membrane revealed by theoretical analysis of electrokinetic data and molecular dynamics simulations. Bioelectrochemistry 2021, 141, 107828. [Google Scholar] [CrossRef] [PubMed]
- Molotkovsky, R.J.; Galimzyanov, T.R.; Ermakov, Y.A. Heterogeneity in Lateral Distribution of Polycations at the Surface of Lipid Membrane: From the Experimental Data to the Theoretical Model. Materials 2021, 14, 6623. [Google Scholar] [CrossRef]
- Li, M.; Tikhonov, A.M.; Schlossman, M.L. An X-ray diffuse scattering study of domains in F(FC2)10(CH2)2OH OH monolayers at the hexane-water interface. J. Eur. Phys. Lett. 2002, 58, 80–86. [Google Scholar] [CrossRef]
- Schwartz, D.K.; Schlossman, M.L.; Kawamoto, E.H.; Kellogg, G.J.; Pershan, P.S.; Ocko, B.M. Thermal diffuse X-ray-scattering studies of the water-vapor interface. Phys. Rev. A 1990, 41, 5687–5690. [Google Scholar] [CrossRef]
- Tikhonov, A.M.; Asadchikov, V.E.; Volkov, Y.O.; Roshchin, B.S.; Ermakov, Y.A. On the Spectrum of Interlayer Roughnesses in a Phospholipid Multilayer. J. Exp. Theor. Phys. Lett. 2021, 114, 620–624. [Google Scholar] [CrossRef]

| System/Parameters | A0, Ǻ2 | Ae, Ǻ2 | KP, mN/m | t0, mN/m | S |
|---|---|---|---|---|---|
| DMPS (LE) | 59.8 | 49.3 | 8.1 | 0.08 | 0.132 |
| DMPS + PL-200 (LE) | 60.8 | 48.9 | 13.6 | 0.07 | 0.057 |
| DMPS (LC) | 46 | 30.6 | 5.2 | - | - |
| DMPS + PL-200 (LC) | 49 | 24.4 | 7.0 | - | - |
| Hydrophobic Tail Region | Headgroup Region | Polymer Layer | |||||||
|---|---|---|---|---|---|---|---|---|---|
| L1 (Å) | σ0 (Å) | L2 (Å) | n | L3 (Å) | σ3 (Å) | ||||
| A ≈ 40 Å2 | |||||||||
| Water | 16.1 ± 0.4 | 1.02 ± 0.01 | 3.6 ± 0.2 | 9.5 ± 1 | 1.56 ± 0.01 | 3 ± 1 | |||
| PL-5 | 4.6 ± 0.2 | 6.0 ± 0.5 | 2.1 ± 0.1 | 0 ± 1 | 144 ± 3 | 1.29 ± 0.02 | 40 ± 3 | ||
| PL-12 | 5.0 ± 0.2 | 6.0 ± 0.5 | 2.1 ± 0.1 | 0 ± 1 | 112 ± 3 | 1.29 ± 0.04 | 25 ± 2 | ||
| PL-200 | 4.9 ± 0.2 | 6.9 ± 0.5 | 2.0 ± 0.1 | 1 ± 1 | 103 ± 3 | 1.27 ± 0.02 | 33 ± 3 | ||
| A ≈ 70 Å2 | |||||||||
| Water | 12 ± 0.3 | 0.90 ± 0.02 | 3.0 ± 0.2 | 11 ± 1 | 1.25 ± 0.04 | 18 ± 3 | |||
| PL-5 | 3.9 ± 0.2 | 2.5 ± 0.5 | 2.5 ± 0.5 | 1 ± 1 | 167 ± 5 | 1.21 ± 0.02 | 29 ± 3 | ||
| PL-12 | 3.6 ± 0.2 | 5.2 ± 0.4 | 1.5 ± 0.1 | 4 ± 1 | 165 ± 5 | 1.18 ± 0.04 | 19 ± 2 | ||
| PL-200 | 3.9 ± 0.2 | 3.4 ± 0.5 | 2.1 ± 0.5 | 3 ± 1 | 115 ± 3 | 1.09 ± 0.02 | 41 ± 3 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tikhonov, A.M.; Asadchikov, V.E.; Volkov, Y.O.; Roshchin, B.S.; Nuzhdin, A.D.; Makrinsky, K.I.; Ermakov, Y.A. X-ray Reflectivity Study of Polylysine Adsorption on the Surface of DMPS Monolayers. Membranes 2022, 12, 1223. https://doi.org/10.3390/membranes12121223
Tikhonov AM, Asadchikov VE, Volkov YO, Roshchin BS, Nuzhdin AD, Makrinsky KI, Ermakov YA. X-ray Reflectivity Study of Polylysine Adsorption on the Surface of DMPS Monolayers. Membranes. 2022; 12(12):1223. https://doi.org/10.3390/membranes12121223
Chicago/Turabian StyleTikhonov, Aleksey M., Victor E. Asadchikov, Yury O. Volkov, Boris S. Roshchin, Alexander D. Nuzhdin, Kirill I. Makrinsky, and Yury A. Ermakov. 2022. "X-ray Reflectivity Study of Polylysine Adsorption on the Surface of DMPS Monolayers" Membranes 12, no. 12: 1223. https://doi.org/10.3390/membranes12121223
APA StyleTikhonov, A. M., Asadchikov, V. E., Volkov, Y. O., Roshchin, B. S., Nuzhdin, A. D., Makrinsky, K. I., & Ermakov, Y. A. (2022). X-ray Reflectivity Study of Polylysine Adsorption on the Surface of DMPS Monolayers. Membranes, 12(12), 1223. https://doi.org/10.3390/membranes12121223








