Applications of Human Amniotic Membrane for Tissue Engineering
Abstract
1. Introduction
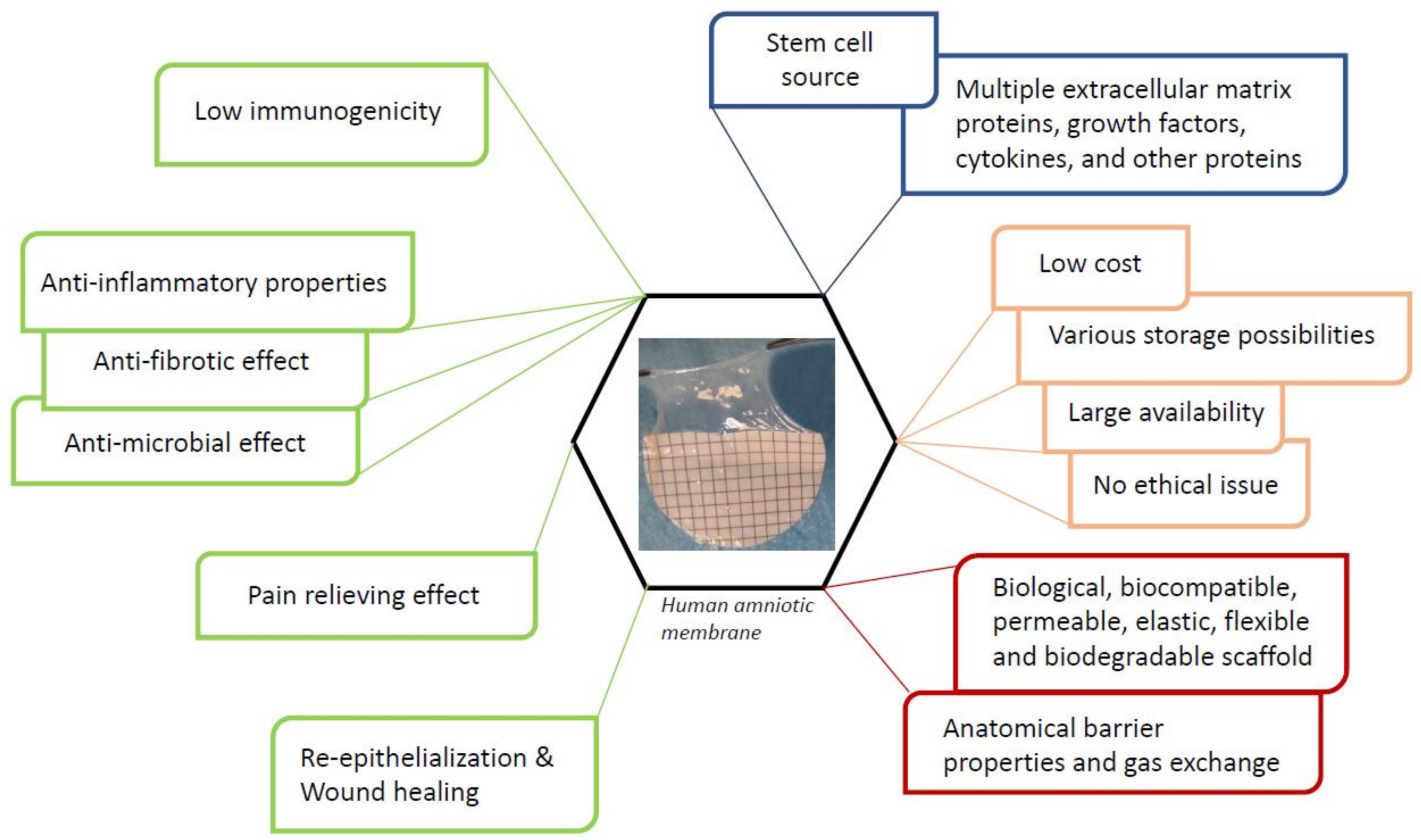
2. Human Amniotic Membrane
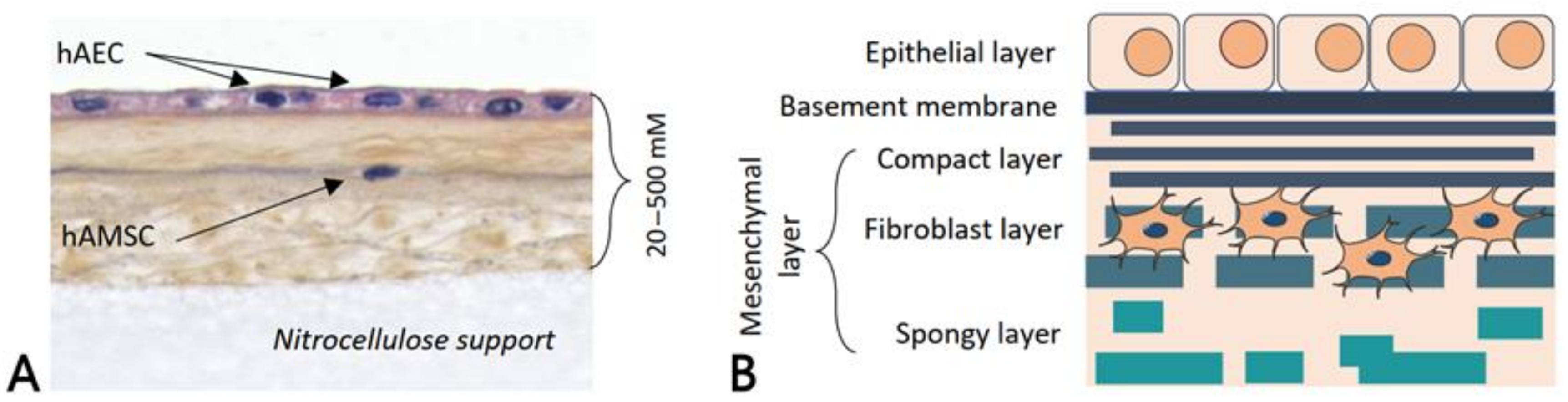
2.1. Anatomy and Physiology
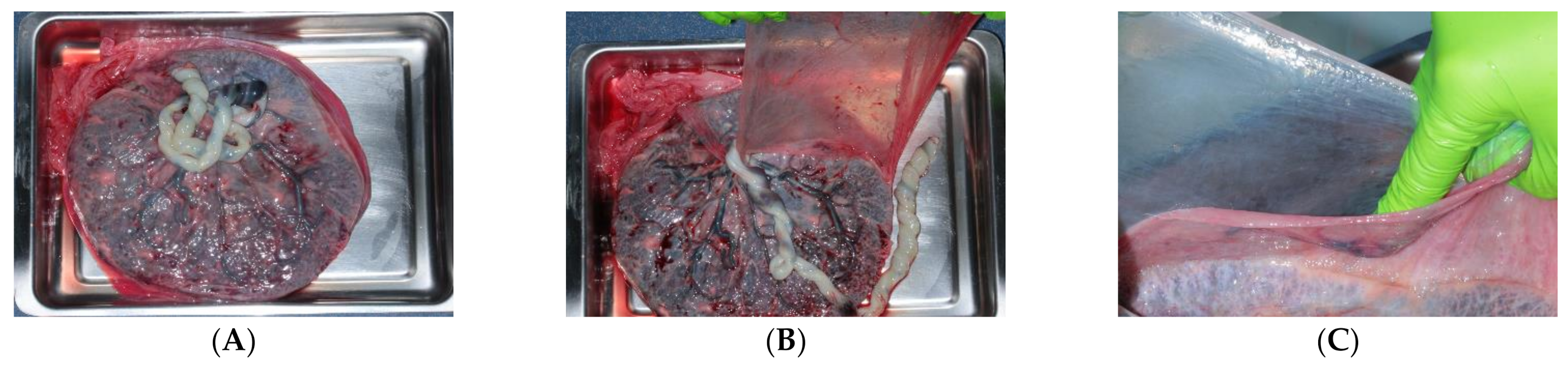
2.2. Collection and Preservation Methods
2.3. Biological Properties
2.4. Mechanical Properties
2.5. Biocompatibility
2.6. Cell Adhesion, Proliferation, and Differentiation
2.7. Biodegradation
3. Tissue Engineering Applications
3.1. Eye
3.2. Skin
3.3. Vascular System
3.4. Bladder and Vagina
3.5. Urethral
3.6. Cartilage
3.7. Bone
3.8. Oral, Periodontal, and Maxillofacial Surgery
3.9. Nerve
3.10. Ligament and Tendon
3.11. Heart
3.12. Clinical Trials
| Conditions | Clinical Trials Id | Phase | Tissue Engineering Product Evaluated | Status | Sponsor | Results/Status or Remarks |
|---|---|---|---|---|---|---|
| OCULAR SURFACE DISEASE | NCT00348114 | 2 | Amnion + ex vivo expanded limbal epithelial stem cells | Suspended | Singapore National Eye Centre | Estimated Enrolment: 8 participants Estimated Study Completion Date: May 2006 |
| LIMBAL STEM CELL DEFICIENCY | NCT00736307 | 1 2 | Amnion + cultured limbal epithelial stem cells | Completed | Royan Institute, Tehran, Iran | Enrolment: 10 participants Study Completion Date: October 2009 |
| UNILATERALLIMBAL STEM CELL INSUFFICIENCY | NCT01377311 | 1 | Amnion + cultured limbal stem cells | Terminated | National Taiwan University Hospital | Enrolment: 0 participants Study Completion Date: April 2010 |
| LIMBAL INSUFFICIENCY SYMBLEPHARON | NCT00491959 | 1 | Amnion + oral mucosal epithelial cells | Terminated (Due to unstable cell sheet quality, the technique was not tested on patients) | National Taiwan University Hospital | Enrolment: 0 participants Study Completion Date: April 2010 |
| SYMBLEPHARON | NCT00799526 | 1 | Amnion + ex vivo cultivated autologous conjunctival epithelial cells | Unknown | Federal University of São Paulo | Estimated Enrolment: 10 participants Estimated Study Completion Date: November 2010 |
| EYE INJURY | NCT01123044 | 3 | Amnion + autologous limbal epithelial cells | Unknown | Ministry of Health, Malaysia | Enrolment: 42 participants Estimated Study Completion Date: September 2012 |
| EPIDERMOLYSIS BULLOSA WITH MITTEN HANDS | NCT01908088 | 1 | Amnion + autologous fibroblasts | Completed | Royan Institute | Enrolment: 6 participants Study Completion Date: July 2013 |
| CORNEAL DISEASE PTERYGIUM MYOPIA HYPEROPIA | NCT02148016 | 1 2 | Autologous limbal stem cell + amnion as a protective contact lens | Unknown | Sun Yat-sen University | Estimated Enrolment: 30 participants Estimated Study Completion Date: September 2014 |
| LIMBUS CORNEAE INSUFFICIENCY SYNDROME | NCT01562002 | 1 2 | Amnion + allogenic bone marrow MSC versus amnion + allogenic limbal stem cells | Completed | Instituto Universitario de Oftalmobiología Aplicada (Institute of Applied Ophthalmobiology)—IOBA | Enrolment: 27 participants Study Completion Date: December 2014 |
| OCULAR SURFACE RECONSTRUCTION | NCT01341223 | Observational | Amnion as a carrier for ex vivo cell culture | Unknown | National Taiwan University Hospital | Estimated Enrolment: 50 participants Estimated Study Completion Date: Mars 2016 |
| LIMBAL STEM CELL DEFICIENCY | NCT03226015 | Observational | Amnion + autologous oral mucosa | Completed | Klinikum Chemnitz gGmbH | Enrolment: 27 participants Study Completion Date: May 2017 |
| LIMBAL STEM CELL DEFICIENCY | NCT01619189 | 2 | Amnion + allogeneic or autologous limbal epithelial stem cells | Completed | CHNO des quinze-vingtsParis, France | Enrollment: 14 participants Study Completion Date: 6 March 2017 |
| LIMBAL STEM CELL DEFICIENCY | NCT02579993 | Interventional | Amnion + in vitro expanded limbal stem cells | Terminated (Preliminary results not favorable) | Instituto de Oftalmologia Conde de Valenciana | Enrolment: 10 participants Study Completion Date: March 2018 |
| LIMBAL STEM CELL DEFICIENCY | NCT02592330 | 1 | Amnion + expanded autologous limbal epithelial cells | Recruiting | Massachusetts Eye and Ear Infirmary | Estimated Enrollment: 17 participants Estimated Study Completion Date: 30 June 2023 |
| WOUNDS | NCT02314416 | 4 | Amniotic stem cells + collagen matrix | Withdrawn | Augusta University | Enrolment: 0 participant Study Completion Date: May 2015 |
| ASHERMAN’S SYNDROME | NCT03223454 | 1 | Amnion + AEC | Unknown | The Second Affiliated Hospital of Chongqing Medical University | Estimated Enrolment: 50 participants Estimated Study Completion Date: March 2021 |
| ENDOMETRIUM INFERTILE PATIENTS | NCT04676269 | 1 | Amnion + autologous endometrium cells or allogenic AEC or both type of cells | Recruiting | Indonesia University | Estimated Enrolment: 40 participants Estimated Study Completion Date: 15 December 2021 |
| ANTERIOR CRUCIATE LIGAMENT RUPTURE | NCT03294759 | Interventional | Amnion collagen matrix wrap + bone MSC | Active, not recruiting | Andrews Research & Education Foundation | Actual Enrolment: 40 participants Estimated Study Completion Date: 25 September 2021 |
| ANTERIOR CRUCIATE LIGAMENT RUPTURE | NCT03294720 | Interventional | Amnion collagen matrix wrap + bone MSC | Active, not recruiting | Andrews Research & Education Foundation | Actual Enrolment: 10 participants Estimated Study Completion Date: 20 March 2021 |
| NONUNION FRACTURE | NCT03031509 | 1 2 | AEC | Not yet recruiting | Shanghai East Hospital | Estimated Enrollment: 36 participants Estimated Study Completion Date: December 2020 |
4. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Langer, R.; Vacanti, J. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Vacanti, J.P.; Langer, R. Tissue engineering: The design and fabrication of living replacement devices for surgical reconstruction and transplantation. Lancet 1999, 354, S32–S34. [Google Scholar] [CrossRef]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Naung, N.Y.; Shehata, E.; Van Sickels, J.E. Resorbable Versus Nonresorbable Membranes. Dent. Clin. N. Am. 2019, 63, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Gindraux, F.; Obert, L.; Laganier, L.; Barnouin, L. Industrial approach in developing an advanced therapy product for bone repair. J. Tissue Eng. Regen. Med. 2010, 4, 194–204. [Google Scholar] [CrossRef]
- Louvrier, A.; Euvrard, E.; Nicod, L.P.; Rolin, G.; Gindraux, F.; Pazart, L.; Houdayer, C.; Risold, P.Y.; Meyer, F. Odontoblastic differentiation of dental pulp stem cells from healthy and carious teeth on an original PCL-based 3D scaffold. Int. Endod. J. 2017, 51, e252–e263. [Google Scholar] [CrossRef]
- Gindraux, F.; Obert, L. Human amniotic membranes: Benefits for bone repair/regeneration. In Bioreconstruction: De l’os à la peau Tome 2; Sauramps Médical: Montpellier, France, 2010; pp. 85–91. [Google Scholar]
- Obert, L.; Genestier, L.; Froidevaux, L.; Averlant, E.; Laurent, R.; Wajszczak, L.; Zwetyenga, N.; Pouthier, F.; Malugani, C.; Gindraux, F. Amniotic membrane for bone repair? Reflection around of the Masquelet technique to one stage/Membrane amniotique pour la réparation osseuse? Réflexion autour de la simplification de la technique de Masquelet à une chirurgie. In Technique de Masquelet; Sauramps Médical: Montpellier, France, 2012. [Google Scholar]
- Gindraux, F.; Laurent, R.; Nicod, L.; Billy, B.; Meyer, C.; Zwetyenga, N.; Wajszczak, L.; Garbuio, P.; Obert, L. Human Amniotic Membrane: Clinical Uses, Patents And Marketed Products. Recent Pat. Regen. Med. 2013, 3, 193–214. [Google Scholar] [CrossRef]
- Laurent, R.; Brennan, M.; Renaud, A.; D’arros, C.; Obert, L.; Layrolle, P.; Gindraux, F. Osteodifferentation of intact human amniotic membrane through a jet sprayed polycaprolactone nanofibre scaffold. Bone Jt. J. Orthop. Proc. Suppl. 2014, 96-B, 113. [Google Scholar]
- Laurent, R.; Nallet, A.; Obert, L.; Nicod, L.; Gindraux, F. Storage and qualification of viable intact human amniotic graft and technology transfer to a tissue bank. Cell Tissue Bank 2014, 15, 267–275. [Google Scholar] [CrossRef]
- Gindraux, F.; Rondot, T.; de Billy, B.; Zwetyenga, N.; Fricain, J.-C.; Pagnon, A.; Obert, L. Similarities between induced membrane and amniotic membrane: Novelty for bone repair. Placenta 2017, 59, 116–123. [Google Scholar] [CrossRef]
- Laurent, R.; Nallet, A.; Obert, L.; Nicod, L.P.; Meyer, C.; Layrolle, P.; Zwetyenga, N.; Gindraux, F.; De Billy, B. Fresh and in vitro osteodifferentiated human amniotic membrane, alone or associated with an additional scaffold, does not induce ectopic bone formation in Balb/c mice. Cell Tissue Bank 2017, 18, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Fénelon, M.; Chassande, O.; Kalisky, J.; Gindraux, F.; Brun, S.; Bareille, R.; Ivanovic, Z.; Fricain, J.-C.; Boiziau, C. Human amniotic membrane for guided bone regeneration of calvarial defects in mice. J. Mater. Sci. Mater. Med. 2018, 29, 78. [Google Scholar] [CrossRef] [PubMed]
- Laurent, R.; Nicod, L.; Layrolle, P.; de Billy, B.; Obert, L.; Gindraux, F. Osteogenic potential and immunogenicity of human amniotic membrane: In vivo and in vivo studies. Orthop. Proc. 2018, 96-B, 112. [Google Scholar]
- Fénelon, M.; Catros, S.; Fricain, J.C. What is the benefit of using amniotic membrane in oral surgery? A comprehensive review of clinical studies. Clin. Oral Investig. 2018, 22, 1881–1891. [Google Scholar] [CrossRef] [PubMed]
- Bourgeois, M.; Loisel, F.; Obert, L.; Pluvy, I.; Gindraux, F. Can the amniotic membrane be used to treat peripheral nerve defects? A review of literature. Hand Surg. Rehabil. 2019, 38, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Fenelon, M.; Maurel, D.B.; Siadous, R.; Gremare, A.; Delmond, S.; Durand, M.; Brun, S.; Catros, S.; Gindraux, F.; L’Heureux, N.; et al. Comparison of the impact of preservation methods on amniotic membrane properties for tissue engineering applications. Mater. Sci. Eng. C 2019, 104, 109903. [Google Scholar] [CrossRef] [PubMed]
- Gualdi, T.; Laurent, R.; Moutarlier, V.; Fenelon, M.; Nallet, A.; Pouthier, F.; Obert, L.; De Billy, B.; Meyer, C.; Gindraux, F. In vitro osteodifferentiation of intact human amniotic membrane is not beneficial in the context of bone repair. Cell Tissue Bank 2019, 20, 435–446. [Google Scholar] [CrossRef]
- Grémare, A.; Jean-Gilles, S.; Musqui, P.; Magnan, L.; Torres, Y.; Fénelon, M.; Brun, S.; Fricain, J.-C.; L’Heureux, N. Cartography of the mechanical properties of the human amniotic membrane. J. Mech. Behav. Biomed. Mater. 2019, 99, 18–26. [Google Scholar] [CrossRef]
- Fenelon, M.; Etchebarne, M.; Siadous, R.; Grémare, A.; Durand, M.; Sentilhes, L.; Torres, Y.; Catros, S.; Gindraux, F.; L’Heureux, N.; et al. Assessment of fresh and preserved amniotic membrane for guided bone regeneration in mice. J. Biomed. Mater. Res. Part A 2020, 108, 2044–2056. [Google Scholar] [CrossRef]
- Etchebarne, M.; Fricain, J.-C.; Kerdjoudj, H.; Di Pietro, R.; Wolbank, S.; Gindraux, F.; Fenelon, M. Use of Amniotic Membrane and Its Derived Products for Bone Regeneration: A Systematic Review. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef]
- Fenelon, M.; Etchebarne, M.; Siadous, R.; Grémare, A.; Durand, M.; Sentilhes, L.; Catros, S.; Gindraux, F.; L’Heureux, N.; Fricain, J.-C. Comparison of amniotic membrane versus the induced membrane for bone regeneration in long bone segmental defects using calcium phosphate cement loaded with BMP-2. Mater. Sci. Eng. C 2021, 124, 112032. [Google Scholar] [CrossRef] [PubMed]
- Odet, S.; Louvrier, A.; Meyer, C.; Nicolás, F.J.; Hofmann, N.; Chatelain, B.; Mauprivez, C.; Laurence, S.; Kerdjoudj, H.; Zwetyenga, N.; et al. Surgical application of human amniotic membrane and amnion-chorion membrane in the oral cavity and efficacy evaluation: Corollary with ophthalmological and wound healing experiences. Front. Bioeng. Biotechnol. 2021, 9, 443. [Google Scholar] [CrossRef]
- Davis, J.S. Skin grafting at the johns hopkins hospital. Ann. Surg. 1909, 50, 542–549. [Google Scholar] [CrossRef] [PubMed]
- Dua, H.S.; Gomes, J.A.; King, A.J.; Maharajan, V. The amniotic membrane in ophthalmology. Surv. Ophthalmol. 2004, 49, 51–77. [Google Scholar] [CrossRef] [PubMed]
- Ricci, E.; Vanosi, G.; Lindenmair, A.; Hennerbichler, S.; Peterbauer-Scherb, A.; Wolbank, S.; Cargnoni, A.; Signoroni, P.B.; Campagnol, M.; Gabriel, C.; et al. Anti-fibrotic effects of fresh and cryopreserved human amniotic membrane in a rat liver fibrosis model. Cell Tissue Bank 2012, 14, 475–488. [Google Scholar] [CrossRef] [PubMed]
- Kubo, M.; Sonoda, Y.; Muramatsu, R.; Usui, M. Immunogenicity of human amniotic membrane in experimental xenotransplantation. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1539–1546. [Google Scholar]
- Parolini, O.; Alviano, F.; Bagnara, G.P.; Bilic, G.; Bühring, H.-J.; Evangelista, M.; Hennerbichler, S.; Liu, B.; Magatti, M.; Mao, N.; et al. Concise Review: Isolation and Characterization of Cells from Human Term Placenta: Outcome of the First International Workshop on Placenta Derived Stem Cells. Stem Cells 2008, 26, 300–311. [Google Scholar] [CrossRef]
- Silini, A.R.; Di Pietro, R.; Lang-Olip, I.; Alviano, F.; Banerjee, A.; Basile, M.; Borutinskaite, V.; Eissner, G.; Gellhaus, A.; Giebel, B.; et al. Perinatal Derivatives: Where Do We Stand? A Roadmap of the Human Placenta and Consensus for Tissue and Cell Nomenclature. Front. Bioeng. Biotechnol. 2020, 8, 610544. [Google Scholar] [CrossRef]
- Leal-Marin, S.; Kern, T.; Hofmann, N.; Pogozhykh, O.; Framme, C.; Börgel, M.; Figueiredo, C.; Glasmacher, B.; Gryshkov, O. Human Amniotic Membrane: A review on tissue engineering, application, and storage. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 1–18. [Google Scholar] [CrossRef]
- Tehrani, F.D.; Firouzeh, A.; Shabani, I.; Shabani, A. A Review on Modifications of Amniotic Membrane for Biomedical Applications. Front. Bioeng. Biotechnol. 2021, 8, 606982. [Google Scholar] [CrossRef]
- Ilic, D.; Vićovac, L.; Nikolic, M. Human amniotic membrane grafts in therapy of chronic non-healing wounds. Br. Med. Bull. 2016, 117, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Toda, A.; Okabe, M.; Yoshida, T.; Nikaido, T. The Potential of Amniotic Membrane/Amnion-Derived Cells for Regeneration of Various Tissues. J. Pharmacol. Sci. 2007, 105, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Niknejad, H.; Peirovi, H.; Jorjani, M.; Ahmadiani, A.; Ghanavi, J.; Seifalian, A.M. Properties of the amniotic membrane for potential use in tissue engineering. Eur. Cells Mater. 2008, 15, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Arrizabalaga, J.H.; Nollert, M.U. Human Amniotic Membrane: A Versatile Scaffold for Tissue Engineering. ACS Biomater. Sci. Eng. 2018, 4, 2226–2236. [Google Scholar] [CrossRef]
- Nejad, A.R.; Hamidieh, A.A.; Amirkhani, M.A.; Sisakht, M.M. Update review on five top clinical applications of human amniotic membrane in regenerative medicine. Placenta 2021, 103, 104–119. [Google Scholar] [CrossRef]
- Bourne, G. The Foetal Membranes: A Review of the Anatomy of Normal Amnion and Chorion and Some Aspects of Their Function. Postgrad. Med. J. 1962, 38, 193–201. [Google Scholar] [CrossRef]
- Miki, T.; Strom, S.C. Amnion-derived pluripotent/multipotent stem cells. Stem Cell Rev. Rep. 2006, 2, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Pasquier, J.-C.; Doret, M. Les membranes fœtales: Développement embryologique, structure et physiopathologie de la rupture prématurée avant terme. J. Gynécologie Obs. Biol. Reprod. 2008, 37, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Mamede, A.C.; Carvalho, M.J.; Abrantes, A.M.; Laranjo, M.; Maia, C.J.; Botelho, M.F. Amniotic membrane: From structure and functions to clinical applications. Cell Tissue Res. 2012, 349, 447–458. [Google Scholar] [CrossRef]
- Bryant-Greenwood, G.D. The extracellular matrix of the human fetal membranes: Structure and function. Placenta 1998, 19, 1–11. [Google Scholar] [CrossRef]
- Malak, T.; Ockleford, C.; Bell, S.; Dalgleish, R.; Bright, N.; MacVicar, J. Confocal immunofluorescence localization of collagen types I, III, IV, V and VI and their ultrastructural organization in term human fetal membranes. Placenta 1993, 14, 385–406. [Google Scholar] [CrossRef]
- Miki, T.; Marongiu, F.; Dorko, K.; Ellis, E.C.; Strom, S.C. Isolation of Amniotic Epithelial Stem Cells. Curr. Protoc. Stem Cell Biol. 2010, 12, 1E.3.1–1E.3.10. [Google Scholar] [CrossRef]
- Kim, J.; Kang, H.M.; Kim, H.; Kim, M.R.; Kwon, H.C.; Gye, M.C.; Kang, S.G.; Yang, H.S.; You, J. Ex Vivo Characteristics of Human Amniotic Membrane-Derived Stem Cells. Cloning Stem Cells 2007, 9, 581–594. [Google Scholar] [CrossRef]
- Riau, A.K.; Beuerman, R.W.; Lim, L.S.; Mehta, J.S. Preservation, sterilization and de-epithelialization of human amniotic membrane for use in ocular surface reconstruction. Biomaterials 2010, 31, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Von Versen-Hoeynck, F.; Steinfeld, A.P.; Becker, J.; Hermel, M.; Rath, W.; Hesselbarth, U. Sterilization and preservation influence the biophysical properties of human amnion grafts. Biologicals 2008, 36, 248–255. [Google Scholar] [CrossRef]
- Jirsova, K.; Jones, G.L.A. Amniotic membrane in ophthalmology: Properties, preparation, storage and indications for grafting—A review. Cell Tissue Bank 2017, 18, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ares, M.T.; López-Valladares, M.J.; Touriño, R.; Vieites, B.; Gude, F.; Silva, M.T.; Couceiro, J. Effects of lyophilization on human amniotic membrane. Acta Ophthalmol. 2009, 87, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Hennerbichler, S.; Reichl, B.; Pleiner, D.; Gabriel, C.; Eibl, J.; Redl, H. The influence of various storage conditions on cell viability in amniotic membrane. Cell Tissue Bank 2006, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Fairbairn, N.; Randolph, M.; Redmond, R. The clinical applications of human amnion in plastic surgery. J. Plast. Reconstr. Aesthetic Surg. 2014, 67, 662–675. [Google Scholar] [CrossRef]
- Laranjo, M. Preservation of Amniotic Membrane. In Amniotic Membrane: Origin, Characterization and Medical Applications; Mamede, A.C., Botelho, M.F., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 209–230. [Google Scholar]
- Nakamura, T.; Sekiyama, E.; Takaoka, M.; Bentley, A.J.; Yokoi, N.; Fullwood, N.J.; Kinoshita, S. The use of trehalose-treated freeze-dried amniotic membrane for ocular surface reconstruction. Biomaterials 2008, 29, 3729–3737. [Google Scholar] [CrossRef]
- Thomasen, H.; Pauklin, M.; Steuhl, K.-P.; Meller, D. Comparison of cryopreserved and air-dried human amniotic membrane for ophthalmologic applications. Graefe’s Arch. Clin. Exp. Ophthalmol. 2009, 247, 1691–1700. [Google Scholar] [CrossRef] [PubMed]
- Shortt, A.J.; Secker, G.A.; Lomas, R.J.; Wilshaw, S.-P.; Kearney, J.N.; Tuft, S.J.; Daniels, J.T. The effect of amniotic membrane preparation method on its ability to serve as a substrate for the ex-vivo expansion of limbal epithelial cells. Biomaterials 2009, 30, 1056–1065. [Google Scholar] [CrossRef] [PubMed]
- Arai, N.; Tsuno, H.; Okabe, M.; Yoshida, T.; Koike, C.; Noguchi, M.; Nikaido, T. Clinical Application of a Hyperdry Amniotic Membrane on Surgical Defects of the Oral Mucosa. J. Oral Maxillofac. Surg. 2012, 70, 2221–2228. [Google Scholar] [CrossRef]
- Koob, T.J.; Young, C.S.; Lim, J.J.; Chinn, K.; Massee, M.; Carter, M.; Spencer, R.; Mason, D.; Fetterolf, D. A Primer on Amniotic Membrane Regenerative Healing; Mimedx: Marietta, GA, USA, 2016. [Google Scholar]
- Koizumi, N.; Fullwood, N.J.; Bairaktaris, G.; Inatomi, T.; Kinoshita, S.; Quantock, A.J. Cultivation of corneal epithelial cells on intact and denuded human amniotic membrane. Investig. Ophthalmol. Vis. Sci. 2000, 41, 2506–2513. [Google Scholar]
- Koizumi, N.; Rigby, H.; Fullwood, N.J.; Kawasaki, S.; Tanioka, H.; Koizumi, K.; Kociok, N.; Joussen, A.M.; Kinoshita, S. Comparison of intact and denuded amniotic membrane as a substrate for cell-suspension culture of human limbal epithelial cells. Graefe’s Arch. Clin. Exp. Ophthalmol. 2006, 245, 123–134. [Google Scholar] [CrossRef]
- Wilshaw, S.P.; Kearney, J.N.; Fisher, J.; Ingham, E. Production of an acellular amniotic membrane matrix for use in tissue engineering. Tissue Eng. 2006, 12, 2117–2129. [Google Scholar] [CrossRef] [PubMed]
- Courtman, D.W.; Pereira, C.A.; Kashef, V.; McComb, D.; Lee, J.M.; Wilson, G.J. Development of a pericardial acellular matrix biomaterial: Biochemical and mechanical effects of cell extraction. J. Biomed. Mater. Res. 1994, 28, 655–666. [Google Scholar] [CrossRef]
- Ilancheran, S.; Moodley, Y.; Manuelpillai, U. Human Fetal Membranes: A Source of Stem Cells for Tissue Regeneration and Repair? Placenta 2009, 30, 2–10. [Google Scholar] [CrossRef]
- Grzywocz, Z.; Pius-Sadowska, E.; Klos, P.; Gryzik, M.; Wasilewska, D.; Aleksandrowicz, B.; Dworczynska, M.; Sabalinska, S.; Hoser, G.; Machalinski, B.; et al. Growth factors and their receptors derived from human amniotic cells in vitro. Folia Histochem. Cytobiol. 2014, 52, 163–170. [Google Scholar] [CrossRef]
- Koizumi, N.J.; Inatomi, T.J.; Sotozono, C.J.; Fullwood, N.J.; Quantock, A.J.; Kinoshita, S. Growth factor mRNA and protein in preserved human amniotic membrane. Curr. Eye Res. 2000, 20, 173–177. [Google Scholar] [CrossRef]
- Hao, Y.; Ma, D.H.-K.; Hwang, D.G.; Kim, W.-S.; Zhang, F. Identification of Antiangiogenic and Antiinflammatory Proteins in Human Amniotic Membrane. Cornea 2000, 19, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Niknejad, H.; Paeini-Vayghan, G.; Tehrani, F.; Khayat-Khoei, M.; Peirovi, H. Side dependent effects of the human amnion on angiogenesis. Placenta 2013, 34, 340–345. [Google Scholar] [CrossRef]
- Niknejad, H.; Yazdanpanah, G. Anticancer effects of human amniotic membrane and its epithelial cells. Med. Hypotheses 2014, 82, 488–489. [Google Scholar] [CrossRef] [PubMed]
- Niknejad, H.; Khayat-Khoei, M.; Peirovi, H.; Abolghasemi, H. Human amniotic epithelial cells induce apoptosis of cancer cells: A new anti-tumor therapeutic strategy. Cytotherapy 2014, 16, 33–40. [Google Scholar] [CrossRef]
- Lee, S.B.; Li, D.Q.; Tan, D.T.; Meller, D.C.; Tseng, S.C. Suppression of TGF-beta signaling in both normal conjunctival fibroblasts and pterygial body fibroblasts by amniotic membrane. Curr. Eye Res. 2000, 20, 325–334. [Google Scholar] [CrossRef]
- Chopra, A.; Thomas, B.S. Amniotic Membrane: A Novel Material for Regeneration and Repair. J. Biomim. Biomater. Tissue Eng. 2013, 18, 1–8. [Google Scholar]
- King, A.; Paltoo, A.; Kelly, R.; Sallenave, J.-M.; Bocking, A.; Challis, J. Expression of Natural Antimicrobials by Human Placenta and Fetal Membranes. Placenta 2007, 28, 161–169. [Google Scholar] [CrossRef]
- Kjaergaard, N.; Hein, M.; Hyttel, L.; Helmig, R.; Schønheyder, H.; Uldbjerg, N.; Madsen, H. Antibacterial properties of human amnion and chorion in vitro. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001, 94, 224–229. [Google Scholar] [CrossRef]
- Díaz-Prado, S.; Rendal-Vázquez, M.E.; Muiños-López, E.; Hermida-Gómez, T.; Rodríguez-Cabarcos, M.; Fuentes-Boquete, I.; De Toro, F.J.; Blanco, F.J. Potential use of the human amniotic membrane as a scaffold in human articular cartilage repair. Cell Tissue Bank 2010, 11, 183–195. [Google Scholar] [CrossRef]
- Farhadihosseinabadi, B.; Farahani, M.; Tayebi, T.; Jafari, A.; Biniazan, F.; Modaresifar, K.; Moravvej, H.; Bahrami, S.; Redl, H.; Tayebi, L.; et al. Amniotic membrane and its epithelial and mesenchymal stem cells as an appropriate source for skin tissue engineering and regenerative medicine. Artif. Cells Nanomed. Biotechnol. 2018, 46, 431–440. [Google Scholar] [CrossRef]
- Herndon, D.N.; Branski, L.K. Contemporary Methods Allowing for Safe and Convenient Use of Amniotic Membrane as a Biologic Wound Dressing for Burns. Ann. Plast. Surg. 2017, 78, S9–S10. [Google Scholar] [CrossRef]
- Hopkinson, A.; McIntosh, R.S.; Tighe, P.J.; James, D.K.; Dua, H.S. Amniotic Membrane for Ocular Surface Reconstruction: Donor Variations and the Effect of Handling on TGF-β Content. Investig. Opthalmol. Vis. Sci. 2006, 47, 4316–4322. [Google Scholar] [CrossRef] [PubMed]
- López-Valladares, M.J.; Rodríguez-Ares, M.T.; Touriño, R.; Gude, F.; Silva, M.T.; Couceiro, J. Donor age and gestational age influence on growth factor levels in human amniotic membrane. Acta Ophthalmol. 2010, 88, e211–e216. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I.; Said, D.G.; Maharajan, V.; Dua, H.S. Amniotic membrane in ophthalmology: Indications and limitations. Eye 2009, 23, 1954–1961. [Google Scholar] [CrossRef] [PubMed]
- Weidinger, A.; Poženel, L.; Wolbank, S.; Banerjee, A. Sub-Regional Differences of the Human Amniotic Membrane and Their Potential Impact on Tissue Regeneration Application. Front. Bioeng. Biotechnol. 2021, 8, 613804. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Bonci, P.; Bonci, P. The effects of different preservation processes on the total protein and growth factor content in a new biological product developed from human amniotic membrane. Cell Tissue Bank 2011, 13, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Jabareen, M.; Mallik, A.S.; Bilic, G.; Zisch, A.H.; Mazza, E. Relation between mechanical properties and microstructure of human fetal membranes: An attempt towards a quantitative analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 144, S134–S141. [Google Scholar] [CrossRef]
- Li, W.; Ma, G.; Brazile, B.; Li, N.; Dai, W.; Butler, J.R.; Claude, A.A.; Wertheim, J.A.; Liao, J.; Wang, B. Investigating the Potential of Amnion-Based Scaffolds as a Barrier Membrane for Guided Bone Regeneration. Langmuir 2015, 31, 8642–8653. [Google Scholar] [CrossRef]
- Rohleder, N.H.; Loeffelbein, D.J.; Feistl, W.; Eddicks, M.; Wolff, K.-D.; Gulati, A.; Steinstraesser, L.; Kesting, M.R. Repair of Oronasal Fistulae by Interposition of Multilayered Amniotic Membrane Allograft. Plast. Reconstr. Surg. 2013, 132, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Ramuta, T.Ž.; Kreft, M.E. Human Amniotic Membrane and Amniotic Membrane–Derived Cells. Cell Transplant. 2018, 27, 77–92. [Google Scholar] [CrossRef]
- Adamowicz, J.; Pokrywczyńska, M.; Tworkiewicz, J.; Kowalczyk, T.; Van Breda, S.V.; Tyloch, D.; Kloskowski, T.; Bodnar, M.; Skopinska-Wisniewska, J.; Marszałek, A.; et al. New Amniotic Membrane Based Biocomposite for Future Application in Reconstructive Urology. PLoS ONE 2016, 11, e0146012. [Google Scholar] [CrossRef] [PubMed]
- Gholipourmalekabadi, M.; Samadikuchaksaraei, A.; Seifalian, A.M.; Urbanska, A.M.; Ghanbarian, H.; Hardy, J.G.; Omrani, M.D.; Mozafari, M.; Reis, R.L.; Kundu, S.C. Silk fibroin/amniotic membrane 3D bi-layered artificial skin. Biomed. Mater. 2017, 13, 035003. [Google Scholar] [CrossRef] [PubMed]
- Niknejad, H.; Deihim, T.; Solati-Hashjin, M.; Peirovi, H. The effects of preservation procedures on amniotic membrane’s ability to serve as a substrate for cultivation of endothelial cells. Cryobiology 2011, 63, 145–151. [Google Scholar] [CrossRef]
- Chuck, R.S.; Graff, J.M.; Bryant, M.R.; Sweet, P.M. Biomechanical Characterization of Human Amniotic Membrane Preparations for Ocular Surface Reconstruction. Ophthalmic Res. 2004, 36, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Connon, C.J.; Doutch, J.; Chen, B.; Hopkinson, A.; Mehta, J.S.; Nakamura, T.; Kinoshita, S.; Meek, K.M. The variation in transparency of amniotic membrane used in ocular surface regeneration. Br. J. Ophthalmol. 2009, 94, 1057–1061. [Google Scholar] [CrossRef] [PubMed]
- Baguneid, M.S.; Seifalian, A.M.; Salacinski, H.J.; Murray, D.; Hamilton, G.; Walker, M.G. Tissue engineering of blood vessels. BJS 2006, 93, 282–290. [Google Scholar] [CrossRef]
- Kamarul, T.; Krishnamurithy, G.; Salih, N.D.; Ibrahim, N.S.; Raghavendran, H.R.B.; Suhaeb, A.R.; Choon, D.S.K. Biocompatibility and Toxicity of Poly(vinyl alcohol)/N,O-Carboxymethyl Chitosan Scaffold. Sci. World J. 2014, 2014, 905103. [Google Scholar] [CrossRef]
- Wilshaw, S.-P.; Kearney, J.; Fisher, J.; Ingham, E. Biocompatibility and Potential of Acellular Human Amniotic Membrane to Support the Attachment and Proliferation of Allogeneic Cells. Tissue Eng. Part A 2008, 14, 463–472. [Google Scholar] [CrossRef]
- Nakamura, T.; Inatomi, T.; Sekiyama, E.; Ang, L.P.K.; Yokoi, N.; Kinoshita, S. Novel clinical application of sterilized, freeze-dried amniotic membrane to treat patients with pterygium. Acta Ophthalmol. Scand. 2006, 84, 401–405. [Google Scholar] [CrossRef]
- Zhang, T.; Yam, G.H.-F.; Riau, A.K.; Poh, R.; Allen, J.C.; Peh, G.S.; Beuerman, R.W.; Tan, D.T.; Mehta, J.S. The Effect of Amniotic Membrane De-Epithelialization Method on its Biological Properties and Ability to Promote Limbal Epithelial Cell Culture. Investig. Opthalmol. Vis. Sci. 2013, 54, 3072–3081. [Google Scholar] [CrossRef]
- Roux, S.; Bodivit, G.; Bartis, W.; Lebouvier, A.; Chevallier, N.; Fialaire-Legendre, A.; Bierling, P.; Rouard, H. In Vitro Characterization of Patches of Human Mesenchymal Stromal Cells. Tissue Eng. Part A 2015, 21, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.-L.; Sulaiman, S.; Pingguan-Murphy, B.; Selvaratnam, L.; Tai, C.-C.; Kamarul, T. Human amnion as a novel cell delivery vehicle for chondrogenic mesenchymal stem cells. Cell Tissue Bank 2009, 12, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Gholipourmalekabadi, M.; Sameni, M.; Radenkovic, D.; Mozafari, M.; Mossahebi-Mohammadi, M.; Seifalian, A. Decellularized human amniotic membrane: How viable is it as a delivery system for human adipose tissue-derived stromal cells? Cell Prolif. 2016, 49, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.Z.; Park, S.R.; Choi, B.H.; Lee, K.-Y.; Kang, C.K.; Min, B.-H. Human Amniotic Membrane as a Delivery Matrix for Articular Cartilage Repair. Tissue Eng. 2007, 13, 693–702. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Chung, M.-C.; Yao, C.-C.J.; Huang, C.-H.; Chang, H.-H.; Jeng, J.-H.; Young, T.-H. The effects of acellular amniotic membrane matrix on osteogenic differentiation and ERK1/2 signaling in human dental apical papilla cells. Biomaterials 2012, 33, 455–463. [Google Scholar] [CrossRef]
- Jerman, U.D.; Veranič, P.; Kreft, M.E. Amniotic Membrane Scaffolds Enable the Development of Tissue-Engineered Urothelium with Molecular and Ultrastructural Properties Comparable to that of Native Urothelium. Tissue Eng. Part C Methods 2014, 20, 317–327. [Google Scholar] [CrossRef]
- Iranpour, S.; Mahdavi-Shahri, N.; Miri, R.; Hasanzadeh, H.; Bidkhori, H.R.; Naderi-Meshkin, H.; Zahabi, E.; Matin, M.M. Supportive properties of basement membrane layer of human amniotic membrane enable development of tissue engineering applications. Cell Tissue Bank 2018, 19, 357–371. [Google Scholar] [CrossRef]
- Schwab, I.R.; Reyes, M.; Isseroff, R.R. Successful Transplantation of Bioengineered Tissue Replacements in Patients with Ocular Surface Disease. Cornea 2000, 19, 421–426. [Google Scholar] [CrossRef]
- Tsai, R.J.-F.; Li, L.-M.; Chen, J.-K. Reconstruction of Damaged Corneas by Transplantation of Autologous Limbal Epithelial Cells. N. Engl. J. Med. 2000, 343, 86–93. [Google Scholar] [CrossRef]
- Koizumi, N.; Inatomi, T.; Suzuki, T.; Sotozono, C.; Kinoshita, S. Cultivated corneal epithelial stem cell transplantation in ocular surface disorders11The authors have no financial interest in this work. Ophthalmology 2001, 108, 1569–1574. [Google Scholar] [CrossRef]
- Malhotra, C.; Jain, A.K. Human amniotic membrane transplantation: Different modalities of its use in ophthalmology. World J. Transplant. 2014, 4, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Inatomi, T.; Sotozono, C.; Amemiya, T.; Kanamura, N.; Kinoshita, S. Transplantation of cultivated autologous oral mucosal epithelial cells in patients with severe ocular surface disorders. Br. J. Ophthalmol. 2004, 88, 1280–1284. [Google Scholar] [CrossRef]
- Walkden, A. Amniotic Membrane Transplantation in Ophthalmology: An Updated Perspective. Clin. Ophthalmol. 2020, 14, 2057–2072. [Google Scholar] [CrossRef]
- Che, X.; Wu, H.; Jia, C.; Sun, H.; Ou, S.; Wang, J.; Jeyalatha, M.V.; Yu, J.; Zuo, C.; Liu, Z.; et al. A Novel Tissue-Engineered Corneal Stromal Equivalent Based on Amniotic Membrane and Keratocytes. Investig. Opthalmol. Vis. Sci. 2019, 60, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Bandeira, F.; Yam, G.H.; Fuest, M.; Ong, H.S.; Liu, Y.; Seah, X.; Shen, S.Y.; Mehta, J.S. Urea-De-Epithelialized Human Amniotic Membrane for Ocular Surface Reconstruction. Stem Cells Transl. Med. 2019, 8, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Ben M’Barek, K.; Habeler, W.; Plancheron, A.; Jarraya, M.; Regent, F.; Terray, A.; Yang, Y.; Chatrousse, L.; Domingues, S.; Masson, Y.; et al. Human ESC–derived retinal epithelial cell sheets potentiate rescue of photoreceptor cell loss in rats with retinal degeneration. Sci. Transl. Med. 2017, 9, eaai7471. [Google Scholar] [CrossRef]
- Ben M’Barek, K.; Bertin, S.; Brazhnikova, E.; Jaillard, C.; Habeler, W.; Plancheron, A.; Fovet, C.-M.; Demilly, J.; Jarraya, M.; Bejanariu, A.; et al. Clinical-grade production and safe delivery of human ESC derived RPE sheets in primates and rodents. Biomaterials 2020, 230, 119603. [Google Scholar] [CrossRef]
- Laurent, I.; Astère, M.; Wang, K.R.; Cheng, Q.-F.; Li, Q.F. Efficacy and Time Sensitivity of Amniotic Membrane treatment in Patients with Diabetic Foot Ulcers: A Systematic Review and Meta-analysis. Diabetes Ther. 2017, 8, 967–979. [Google Scholar] [CrossRef]
- Murphy, S.V.; Skardal, A.; Song, L.; Sutton, K.; Haug, R.; Mack, D.L.; Jackson, J.; Soker, S.; Atala, A. Solubilized Amnion Membrane Hyaluronic Acid Hydrogel Accelerates Full-Thickness Wound Healing. Stem Cells Transl. Med. 2017, 6, 2020–2032. [Google Scholar] [CrossRef]
- Yang, L.; Shirakata, Y.; Shudou, M.; Dai, X.; Tokumaru, S.; Hirakawa, S.; Sayama, K.; Hamuro, J.; Hashimoto, K. New skin-equivalent model from de-epithelialized amnion membrane. Cell Tissue Res. 2006, 326, 69–77. [Google Scholar] [CrossRef]
- Kim, S.S.; Song, C.K.; Shon, S.K.; Lee, K.Y.; Kim, C.H.; Lee, M.J.; Wang, L. Effects of human amniotic membrane grafts combined with marrow mesenchymal stem cells on healing of full-thickness skin defects in rabbits. Cell Tissue Res. 2009, 336, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Redondo, P.; De Azcarate, A.G.; Marqués, L.; García-Guzman, M.; Andreu, E.; Prósper, F. Amniotic Membrane as a Scaffold for Melanocyte Transplantation in Patients with Stable Vitiligo. Dermatol. Res. Pract. 2011, 2011, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.-H.; Liu, Y.-W.; Tang, W.-C.; Zhou, Z.-W.; Hwang, C.-Y.; Hwang, G.-Y.; Ou, B.-R.; Hu, C.-P.; Yang, V.C.; Chen, J.-K. Characterization of porcine arterial endothelial cells cultured on amniotic membrane, a potential matrix for vascular tissue engineering. Biochem. Biophys. Res. Commun. 2007, 357, 984–990. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.-H.; Tsai, S.-H.; Kuo, L.; Hwang, C.-Y.; Kuo, C.-Y.; Yang, V.C.; Chen, J.-K. A prototype tissue engineered blood vessel using amniotic membrane as scaffold. Acta Biomater. 2012, 8, 3342–3348. [Google Scholar] [CrossRef]
- Peirovi, H.; Rezvani, N.; Hajinasrollah, M.; Mohammadi, S.S.; Niknejad, H. Implantation of amniotic membrane as a vascular substitute in the external jugular vein of juvenile sheep. J. Vasc. Surg. 2012, 56, 1098–1104. [Google Scholar] [CrossRef]
- Amensag, S.; McFetridge, P.S. Rolling the Human Amnion to Engineer Laminated Vascular Tissues. Tissue Eng. Part C Methods 2012, 18, 903–912. [Google Scholar] [CrossRef]
- Amensag, S.; Goldberg, L.A.; O’Malley, K.A.; Rush, D.S.; Berceli, S.A.; McFetridge, P.S. Pilot assessment of a human extracellular matrix-based vascular graft in a rabbit model. J. Vasc. Surg. 2017, 65, 839–847.e1. [Google Scholar] [CrossRef]
- Swim, M.M.; Albertario, A.; Iacobazzi, D.; Caputo, M.; Ghorbel, M.T. Amnion-Based Scaffold with Enhanced Strength and Biocompatibility for In Vivo Vascular Repair. Tissue Eng. Part A 2019, 25, 603–619. [Google Scholar] [CrossRef]
- Kakavand, M.; Yazdanpanah, G.; Ahmadiani, A.; Niknejad, H. Blood compatibility of human amniotic membrane compared with heparin-coated ePTFE for vascular tissue engineering. J. Tissue Eng. Regen. Med. 2015, 11, 1701–1709. [Google Scholar] [CrossRef]
- Norris, M.A.; Cohen, M.S.; Warren, M.M.; Becker, S.N.; Baur, P.S.; Seybold, H.M. Bladder reconstruction in rabbits with glutaraldehyde-stablilized amniotic membranes. Urology 1982, 19, 631–635. [Google Scholar] [CrossRef]
- Fishman, I.J.; Flores, F.; Scott, F.B.; Spjut, H.J.; Morrow, B. Use of Fresh Placental Membranes for Bladder Reconstruction. J. Urol. 1987, 138, 1291–1294. [Google Scholar] [CrossRef]
- Shakeri, S.; Masoudi, P.; Yazdani, M.; Monabbati, A.; Mehrabani, D.; Tanideh, N. Evaluation of human amniotic membrane as a substitute for transitional epithelium of bladder in dog. J. Appl. Anim. Res. 2008, 33, 55–59. [Google Scholar] [CrossRef]
- Barski, D.; Gerullis, H.; Ecke, T.; Varga, G.; Boros, M.; Pintelon, I.; Timmermans, J.-P.; Winter, A.; Bagner, J.-W.; Otto, T. Repair of a vesico-vaginal fistula with amniotic membrane—Step 1 of the IDEAL recommendations of surgical innovation. Central Eur. J. Urol. 2015, 68, 459–461. [Google Scholar] [CrossRef]
- Brandt, F.T.; Albuquerque, C.D.; Lorenzato, F.R. Female urethral reconstruction with amnion grafts. Int. J. Surg. Investig. 2000, 1, 409–414. [Google Scholar]
- Mhaskar, R. Amniotic membrane for cervical reconstruction. Int. J. Gynecol. Obstet. 2005, 90, 123–127. [Google Scholar] [CrossRef]
- Fotopoulou, C.; Sehouli, J.; Gehrmann, N.; Schoenborn, I.; Lichtenegger, W. Functional and anatomic results of amnion vaginoplasty in young women with Mayer-Rokitansky-Küster-Hauser syndrome. Fertil. Steril. 2010, 94, 317–323. [Google Scholar] [CrossRef]
- Vatsa, R.; Bharti, J.; Roy, K.K.; Kumar, R.K.; Sharma, J.B.; Singh, N.; Singhal, S.; Meena, J. Evaluation of amnion in creation of neovagina in women with Mayer-Rokitansky-Kuster-Hauser syndrome. Fertil. Steril. 2017, 108, 341–345. [Google Scholar] [CrossRef]
- Sharifiaghdas, F.; Moghadasali, R.; Baharvand, H.; Hosseini-Moghaddam, S.M.; Mahmoudnejad, N. Special characteristics of culturing mature human bladder smooth muscle cells on human amniotic membrane as a suitable matrix. Urol. J. 2009, 6, 283–288. [Google Scholar] [PubMed]
- Seyed-Forootan, K.; Karimi, H.; Seyed-Forootan, N.-S. Autologous Fibroblast-Seeded Amnion for Reconstruction of Neo-vagina in Male-to-Female Reassignment Surgery. Aesthetic Plast. Surg. 2018, 42, 491–497. [Google Scholar] [CrossRef]
- Adamowicz, J.; Van Breda, S.; Tyloch, D.; Pokrywczynska, M.; Drewa, T. Application of amniotic membrane in reconstructive urology; the promising biomaterial worth further investigation. Expert Opin. Biol. Ther. 2018, 19, 9–24. [Google Scholar] [CrossRef]
- Oottamasathien, S.; Hotaling, J.M.; Craig, J.R.; Myers, J.B.; Brant, W.O. Amniotic therapeutic biomaterials in urology: Current and future applications. Transl. Androl. Urol. 2017, 6, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, S.; Haghpanah, A.; Khezri, A.; Yazdani, M.; Monabbati, A.; Haghpanah, S.; Malekmakan, L.; Ayrempour, S. Application of amniotic membrane as xenograft for urethroplasty in rabbit. Int. Urol. Nephrol. 2009, 41, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Sartoneva, R.; Haimi, S.; Miettinen, S.; Mannerström, B.; Haaparanta, A.-M.; Sándor, G.K.; Kellomäki, M.; Suuronen, R.; Lahdes-Vasama, T. Comparison of a poly- l -lactide-co- ɛ -caprolactone and human amniotic membrane for urothelium tissue engineering applications. J. R. Soc. Interface 2010, 8, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Salehipour, M.; Mohammadian, R.; Jahanbini, S.; Emadmarvasti, V.; Geramizadeh, B.; Tanideh, N. Is amniotic membrane a suitable biomaterial for reconstruction of long ureteral defects? Saudi J. Kidney Dis. Transplant. 2013, 24, 135–138. [Google Scholar] [CrossRef]
- Koziak, A.; Marcheluk, A.; Dmowski, T.; Szcześniewski, R.; Kania, P.; Dorobek, A. Reconstructive surgery of male urethra using human amnion membranes (grafts)—First announcement. Ann. Transplant. 2004, 9, 21–24. [Google Scholar]
- Koziak, A.; Salagierski, M.; Marcheluk, A.; Szcześniewski, R.; Sosnowski, M. Early experience in reconstruction of long ureteral strictures with allogenic amniotic membrane. Int. J. Urol. 2007, 14, 607–610. [Google Scholar] [CrossRef]
- Sharifiaghdas, F.; Hamzehiesfahani, N.; Moghadasali, R.; Ghaemimanesh, F.; Baharvand, H. Human amniotic membrane as a suitable matrix for growth of mouse urothelial cells in comparison with human peritoneal and omentum membranes. Urol. J. 2007, 4, 71–78. [Google Scholar]
- Yuan, J.; Wang, F.; Liu, T.; Yang, L.; Zhang, G.; Liu, H.; Yi, X.; Yang, X.; Lin, T.-Y.; Qin, W. Urethral Reconstruction with Tissue-Engineered Human Amniotic Scaffold in Rabbit Urethral Injury Models. Med. Sci. Monit. 2014, 20, 2430–2438. [Google Scholar] [CrossRef]
- Chen, C.; Zheng, S.; Zhang, X.; Dai, P.; Gao, Y.; Nan, L.; Zhang, Y. Transplantation of Amniotic Scaffold-Seeded Mesenchymal Stem Cells and/or Endothelial Progenitor Cells From Bone Marrow to Efficiently Repair 3-cm Circumferential Urethral Defect in Model Dogs. Tissue Eng. Part A 2018, 24, 47–56. [Google Scholar] [CrossRef]
- Garcia, D.; Longo, U.; Vaquero, J.; Forriol, F.; Loppini, M.; Khan, W.; Denaro, V. Amniotic Membrane Transplant for Articular Cartilage Repair: An Experimental Study in Sheep. Curr. Stem Cell Res. Ther. 2014, 10, 77–83. [Google Scholar] [CrossRef]
- Krishnamurithy, G.; Shilpa, P.N.; Ahmad, R.E.; Sulaiman, S.; Ng, C.L.L.; Kamarul, T. Human amniotic membrane as a chondrocyte carrier vehicle/substrate: In vitro study. J. Biomed. Mater. Res. Part A 2011, 99, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Hussin, I.H.; Pingguan-Murphy, B.; Osman, S.Z. The Fabrication of Human Amniotic Membrane Based Hydrogel for Cartilage Tissue Engineering Applications: A Preliminary Study. In Proceedings of the 5th Kuala Lumpur International Conference on Biomedical Engineering 2011, Kuala Lumpur, Malaysia, 20–23 June 2011; Springer: Berlin/Heidelberg, Germany, 2011; Volume 35, pp. 841–844. [Google Scholar]
- Nogami, M.; Kimura, T.; Seki, S.; Matsui, Y.; Yoshida, T.; Koike-Soko, C.; Okabe, M.; Motomura, H.; Gejo, R.; Nikaido, T. A Human Amnion-Derived Extracellular Matrix-Coated Cell-Free Scaffold for Cartilage Repair: In Vitro and In Vivo Studies. Tissue Eng. Part A 2016, 22, 680–688. [Google Scholar] [CrossRef]
- Lindenmair, A.; Wolbank, S.; Stadler, G.; Meinl, A.; Peterbauer-Scherb, A.; Eibl, J.; Polin, H.; Gabriel, C.; Van Griensven, M.; Redl, H. Osteogenic differentiation of intact human amniotic membrane. Biomaterials 2010, 31, 8659–8665. [Google Scholar] [CrossRef] [PubMed]
- Ghanmi, S.; Trigui, M.; Baya, W.; Ellouz, Z.; Elfeki, A.; Charfi, S.; Fricain, J.C.; Keskes, H. The periosteum-like effect of fresh human amniotic membrane on bone regeneration in a rabbit critical-sized defect model. Bone 2018, 110, 392–404. [Google Scholar] [CrossRef]
- Tang, K.; Wu, J.; Xiong, Z.; Ji, Y.; Sun, T.; Guo, X. Human acellular amniotic membrane: A potential osteoinductive biomaterial for bone regeneration. J. Biomater. Appl. 2017, 32, 754–764. [Google Scholar] [CrossRef]
- Bourgeois, M.; Loisel, F.; Bertrand, D.; Nallet, J.; Gindraux, F.; Adam, A.; Lepage, D.; Sergent, P.; Leclerc, G.; Rondot, T.; et al. Management of forearm bone loss with induced membrane technique. Hand Surg. Rehabil. 2020, 39, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Gindraux, F.; Loisel, F.; Bourgeois, M.; Oudina, K.; Melin, M.; De Billy, B.; Sergent, P.; Leclerc, G.; Petite, H.; Auber, F.; et al. Induced membrane maintains its osteogenic properties even when the second stage of Masquelet’s technique is performed later. Eur. J. Trauma Emerg. Surg. 2019, 46, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Masquelet, A.C.; Obert, L. Induced membrane technique for bone defects in the hand and wrist. Chir. Main 2010, 29 (Suppl. 1), S221–S224. [Google Scholar] [CrossRef]
- Masquelet, A.-C.; Fitoussi, F.; Begue, T.; Muller, G.P. Reconstruction of the long bones by the induced membrane and spongy autograft. Ann. Chir. Plast. Esthet. 2000, 45, 346–353. [Google Scholar]
- Tsugawa, J.; Komaki, M.; Yoshida, T.; Nakahama, K.-I.; Amagasa, T.; Morita, I. Cell-printing and transfer technology applications for bone defects in mice. J. Tissue Eng. Regen. Med. 2011, 5, 695–703. [Google Scholar] [CrossRef]
- Semyari, H.; Rajipour, M.; Sabetkish, S.; Sabetkish, N.; Abbas, F.M.; Kajbafzadeh, A.-M. Evaluating the bone regeneration in calvarial defect using osteoblasts differentiated from adipose-derived mesenchymal stem cells on three different scaffolds: An animal study. Cell Tissue Bank 2015, 17, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Akazawa, K.; Iwasaki, K.; Nagata, M.; Yokoyama, N.; Ayame, H.; Yamaki, K.; Tanaka, Y.; Honda, I.; Morioka, C.; Kimura, T.; et al. Double-layered cell transfer technology for bone regeneration. Sci. Rep. 2016, 6, 33286. [Google Scholar] [CrossRef] [PubMed]
- Akhlaghi, F.; Hesami, N.; Rad, M.R.; Nazeman, P.; Fahimipour, F.; Khojasteh, A. Improved bone regeneration through amniotic membrane loaded with buccal fat pad-derived MSCs as an adjuvant in maxillomandibular reconstruction. J. Cranio-Maxillofac. Surg. 2019, 47, 1266–1273. [Google Scholar] [CrossRef] [PubMed]
- Uppada, U.K.; Kalakonda, B.; Koppolu, P.; Varma, N.; Palakurthy, K.; Manchikanti, V.; Prasad, S.; Samar, S.; Swapna, L.A. Combination of hydroxyapatite, platelet rich fibrin and amnion membrane as a novel therapeutic option in regenerative periapical endodontic surgery: Case series. Int. J. Surg. Case Rep. 2017, 37, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Lawson, V.G. Oral Cavity Reconstruction Using Pectoralis Major Muscle and Amnion. Arch. Otolaryngol. Head Neck Surg. 1985, 111, 230–233. [Google Scholar] [CrossRef]
- Kesting, M.R.; Loeffelbein, D.J.; Classen, M.; Slotta-Huspenina, J.; Hasler, R.J.; Jacobsen, F.; Kreutzer, K.; Al-Benna, S.; Wolff, K.-D.; Steinstraesser, L. Repair of oronasal fistulas with human amniotic membrane in minipigs. Br. J. Oral Maxillofac. Surg. 2010, 48, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Gulameabasse, S.; Gindraux, F.; Catros, S.; Fricain, J.; Fenelon, M. Chorion and amnion/chorion membranes in oral and periodontal surgery: A systematic review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020. [Google Scholar] [CrossRef]
- Lai, D.R.; Chen, H.R.; Lin, L.M.; Huang, Y.L.; Tsai, C.C. Clinical evaluation of different treatment methods for oral submucous fibrosis. A 10-year experience with 150 cases. J. Oral Pathol. Med. 1995, 24, 402–406. [Google Scholar] [CrossRef]
- Khademi, B.; Bahranifard, H.; Azarpira, N.; Behboodi, E. Clinical application of amniotic membrane as a biologic dressing in oral cavity and pharyngeal defects after tumor resection. Arch. Iran. Med. 2013, 16, 503–506. [Google Scholar]
- Ahn, K.-M.; Lee, J.-H.; Hwang, S.-J.; Choung, P.-H.; Kim, M.-J.; Park, H.-J.; Park, J.-K.; Jahng, J.; Yang, E.-K. Fabrication of Myomucosal Flap Using Tissue-engineered Bioartificial Mucosa Constructed with Oral Keratinocytes Cultured on Amniotic Membrane. Artif. Organs 2006, 30, 411–423. [Google Scholar] [CrossRef]
- Amemiya, T.; Nakamura, T.; Yamamoto, T.; Kinoshita, S.; Kanamura, N. Immunohistochemical study of oral epithelial sheets cultured on amniotic membrane for oral mucosal reconstruction. Bio-Med. Mater. Eng. 2010, 20, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, Y.-J.; Huang, S.-F.; Lai, J.-Y.; Ma, S.-C.; Chen, H.-C.; Wu, S.-E.; Wang, T.-K.; Sun, C.-C.; Ma, K.S.-K.; Chen, J.-K.; et al. Preservation of epithelial progenitor cells from collagenase-digested oral mucosa during ex vivo cultivation. Sci. Rep. 2016, 6, 36266. [Google Scholar] [CrossRef] [PubMed]
- Amemiya, T.; Nakamura, T.; Yamamoto, T.; Kinoshita, S.; Kanamura, N. Tissue engineering by transplantation of oral epithelial sheets cultivated on amniotic membrane for oral mucosal reconstruction. Inflamm. Regen. 2010, 30, 176–180. [Google Scholar] [CrossRef][Green Version]
- Amemiya, T.; Nakamura, T.; Yamamoto, T.; Kinoshita, S.; Kanamura, N. Autologous Transplantation of Oral Mucosal Epithelial Cell Sheets Cultured on an Amniotic Membrane Substrate for Intraoral Mucosal Defects. PLoS ONE 2015, 10, e0125391. [Google Scholar] [CrossRef] [PubMed]
- Gurinsky, B. A novel dehydrated amnion allograft for use in the treatment of gingival recession: An observational case series. J. Implant Adv. Clin. Dent. 2009, 1, 65–73. [Google Scholar]
- Shetty, S.S.; Chatterjee, A.; Bose, S. Bilateral multiple recession coverage with platelet-rich fibrin in comparison with amniotic membrane. J. Indian Soc. Periodontol. 2014, 18, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Yadav, K. Amniotic membrane—A Novel material for the root coverage: A case series. J. Indian Soc. Periodontol. 2015, 19, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Orzechowska-Wylęgała, B.; Dobrowolski, D.; Puzzolo, D.; Wowra, B.; Niemiec, W.; Wylęgała, A.; Szczubiałka, K. Use of Autologous Epithelium Transplantation on Various Scaffolds to Cover Tissue Loss in Oral Cavity: Long-Term Observation. J. Appl. Biomater. Funct. Mater. 2017, 15, 25–30. [Google Scholar] [CrossRef]
- Amemiya, T.; Adachi, K.; Nishigaki, M.; Yamamoto, T.; Kanamura, N. Experiences of preclinical use of periodontal ligament-derived cell sheet cultured on human amniotic membrane. J. Oral. Tissue Eng. 2008, 6, 106–112. [Google Scholar]
- Iwasaki, K.; Komaki, M.; Yokoyama, N.; Tanaka, Y.; Taki, A.; Honda, I.; Kimura, Y.; Takeda, M.; Akazawa, K.; Oda, S.; et al. Periodontal Regeneration Using Periodontal Ligament Stem Cell-Transferred Amnion. Tissue Eng. Part A 2013, 20, 693–704. [Google Scholar] [CrossRef]
- Wu, P.-H.; Chung, H.-Y.; Wang, J.-H.; Shih, J.-C.; Kuo, M.Y.-P.; Chang, P.-C.; Huang, Y.-D.; Wang, P.-C.; Chang, C.-C. Amniotic membrane and adipose-derived stem cell co-culture system enhances bone regeneration in a rat periodontal defect model. J. Formos. Med Assoc. 2016, 115, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Honjo, K.-I.; Yamamoto, T.; Adachi, T.; Amemiya, T.; Mazda, O.; Kanamura, N.; Kita, M. Evaluation of a dental pulp-derived cell sheet cultured on amniotic membrane substrate. Bio-Med. Mater. Eng. 2015, 25, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Tuncel, U.; Ozgenel, G. Use of Human Amniotic Membrane as an Interpositional Material in Treatment of Temporomandibular Joint Ankylosis. J. Oral Maxillofac. Surg. 2011, 69, e58–e66. [Google Scholar] [CrossRef] [PubMed]
- Tuncel, U.; Kostakoglu, N.; Turan, A.; Markoc, F.; Gokce, E.; Erkorkmaz, U.; Turan, A. The use of temporalis muscle graft, fresh and cryopreserved amniotic membrane in preventing temporomandibular joint ankylosis after discectomy in rabbits. J. Cranio-Maxillofac. Surg. 2014, 42, 1868–1876. [Google Scholar] [CrossRef]
- Guarda-Nardini, L.; Trojan, D.; Montagner, G.; Cogliati, E.; Bendini, M.; Manfredini, D. Human Amniotic Membrane Positioning in the Surgical Treatment of Temporomandibular Joint Degenerative Disorder. Case Rep. Surg. 2019, 2019, 6037191. [Google Scholar] [CrossRef] [PubMed]
- Ragazzo, M.; Trojan, D.; Spagnol, L.; Paolin, A.; Nardini, L.G. Use of amniotic membrane in the treatment of patients with BRONJ: Two case reports. J. Surg. Case Rep. 2018, 2018, rjy073. [Google Scholar] [CrossRef] [PubMed]
- Lemke, A.; Ferguson, J.; Gross, K.; Penzenstadler, C.; Bradl, M.; Mayer, R.; Gerner, C.; Redl, H.; Wolbank, S. Transplantation of human amnion prevents recurring adhesions and ameliorates fibrosis in a rat model of sciatic nerve scarring. Acta Biomater. 2018, 66, 335–349. [Google Scholar] [CrossRef]
- Meng, H.; Li, M.; You, F.; Du, J.; Luo, Z. Assessment of processed human amniotic membrane as a protective barrier in rat model of sciatic nerve injury. Neurosci. Lett. 2011, 496, 48–53. [Google Scholar] [CrossRef]
- Özgenel, G.Y.; Filiz, G. Combined Application of Human Amniotic Membrane Wrapping and Hyaluronic Acid Injection in Epineurectomized Rat Sciatic Nerve. J. Reconstr. Microsurg. 2004, 20, 153–157. [Google Scholar] [CrossRef]
- Marchesini, A.; Raimondo, S.; Zingaretti, N.; Riccio, V.; Battiston, B.; Provinciali, M.; Geuna, S.; Riccio, M. The amnion muscle combined graft (AMCG) conduits in nerves repair: An anatomical and experimental study on a rat model. J. Mater. Sci. Mater. Med. 2018, 29, 120. [Google Scholar] [CrossRef]
- Riccio, E.; Esposito, G.; Franzone, A.; Imbriaco, M.; Santangelo, M.; Pisani, A. Renal Sympathetic-Nerve Ablation for Uncontrolled Hypertension in a Patient With Single-Kidney Autosomal Dominant Polycystic Kidney Disease. J. Clin. Hypertens. 2014, 16, 385–386. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, G.; Shenaq, S.; Spira, M. Vascularized Nerve Tube: An Experimental Alternative for Vascularized Nerve Grafts Over Short Gaps. J. Reconstr. Microsurg. 1993, 9, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, J.; Shenaq, J.; Rabinovsky, E.; Shenaq, S. Modulation of Peripheral Nerve Regeneration: A Tissue-Engineering Approach. The Role of Amnion Tube Nerve Conduit across a 1-Centimeter Nerve Gap. Plast. Reconstr. Surg. 2000, 105, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Mligiliche, N.; Endo, K.; Okamoto, K.; Fujimoto, E.; Ide, C. Extracellular matrix of human amnion manufactured into tubes as conduits for peripheral nerve regeneration. J. Biomed. Mater. Res. 2002, 63, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Liu, C.; Tian, S.; Bai, J.; Yu, K.; Liu, L.; Tian, D. Electrospun polycaprolactone (PCL)-amnion nanofibrous membrane prevents adhesions and promotes nerve repair in a rat model of sciatic nerve compression. PLoS ONE 2020, 15, e0244301. [Google Scholar] [CrossRef]
- Zhang, Q.; Gu, X.-M.; Yu, G.-Y.; Mao, T.-Q.; Zheng, J.-C.; Tong, Q.-Y. Repair of peripheral nerve defect by a scroll of amnion derivative compound with cultured autogenous Schwann cell in a rat model. Zhonghua Kou Qiang Yi Xue Za Zhi 2006, 41, 98–101. [Google Scholar]
- Li, Z.; Qin, H.; Feng, Z.; Liu, W.; Zhou, Y.; Yang, L.; Zhao, W.; Li, Y. Human umbilical cord mesenchymal stem cell-loaded amniotic membrane for the repair of radial nerve injury. Neural Regen. Res. 2013, 8, 3441–3448. [Google Scholar]
- Riboh, J.C.; Saltzman, B.M.; Yanke, A.B.; Cole, B.J. Human Amniotic Membrane–Derived Products in Sports Medicine. Am. J. Sports Med. 2016, 44, 2425–2434. [Google Scholar] [CrossRef]
- Heckmann, N.; Auran, R.; Mirzayan, R. Application of Amniotic Tissue in Orthopedic Surgery. Am. J. Orthop. 2016, 45, E421–E425. [Google Scholar]
- Demirkan, F.; Colakoglu, N.; Herek, O.; Erkula, G. The use of amniotic membrane in flexor tendon repair: An experimental model. Arch. Orthop. Trauma Surg. 2002, 122, 396–399. [Google Scholar] [CrossRef]
- Özgenel, G.Y. The effects of a combination of hyaluronic and amniotic membrane on the formation of peritendinous adhesions after flexor tendon surgery in chickens. J. Bone Jt. Surg. Br. Vol. 2004, 86, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yu, K.; Bai, J.; Tian, D.; Liu, G. Experimental study of tendon sheath repair via decellularized amnion to prevent tendon adhesion. PLoS ONE 2018, 13, e0205811. [Google Scholar] [CrossRef] [PubMed]
- Warner, M.; LaSyone, L. An Open-label, Single-center, Retrospective Study of Cryopreserved Amniotic Membrane and Umbilical Cord Tissue as an Adjunct for Foot and Ankle Surgery. Surg. Technol. Int. 2014, 25, 251–255. [Google Scholar] [PubMed]
- DeMill, S.L.; Granata, J.D.; McAlister, J.; Berlet, G.C.; Hyer, C.F. Safety analysis of cryopreserved amniotic membrane/umbilical cord tissue in foot and ankle surgery: A consecutive case series of 124 patients. Surg. Technol. Int. 2014, 25, 257–261. [Google Scholar]
- Seo, Y.-K.; Kim, J.-H.; Eo, S.-R. Co-effect of silk and amniotic membrane for tendon repair. J. Biomater. Sci. Polym. Ed. 2016, 27, 1232–1247. [Google Scholar] [CrossRef]
- He, Q.; Li, Q.; Chen, B.; Wang, Z. Repair of flexor tendon defects of rabbit with tissue engineering method. Chin. J. Traumatol. 2002, 5, 200–208. [Google Scholar]
- Cargnoni, A.; Di Marcello, M.; Campagnol, M.; Nassuato, C.; Albertini, A.; Parolini, O. Amniotic Membrane Patching Promotes Ischemic Rat Heart Repair. Cell Transplant. 2009, 18, 1147–1159. [Google Scholar] [CrossRef]
- Roy, R.; Haase, T.; Ma, N.; Bader, A.; Becker, M.; Seifert, M.; Choi, Y.-H.; Falk, V.; Stamm, C. Decellularized amniotic membrane attenuates postinfarct left ventricular remodeling. J. Surg. Res. 2016, 200, 409–419. [Google Scholar] [CrossRef]
- Lim, J.J.; Koob, T.J.; Fonger, J.; Koob, T.J. Dehydrated human amnion/chorionmembrane allograft promotes cardiac repair following myocardial infarction. J. Cardiol. Cardiovasc. Ther. 2017, 2, 2–7. [Google Scholar]
- Francisco, J.; Cunha, R.C.; Cardoso, M.; Simeoni, R.B.; Mogharbel, B.; Picharski, G.; Dziedzic, D.S.M.; Guarita-Souza, L.; Carvalho, K. Decellularized Amniotic Membrane Scaffold as a Pericardial Substitute: An In Vivo Study. Transplant. Proc. 2016, 48, 2845–2849. [Google Scholar] [CrossRef]
- Khalpey, Z.; Marsh, K.M.; Ferng, A.; Bin Riaz, I.; Friedman, M.; Indik, J.; Avery, R.; Jokerst, C.; Oliva, I. First in Man: Amniotic Patch Reduces Postoperative Inflammation. Am. J. Med. 2015, 128, e5–e6. [Google Scholar] [CrossRef] [PubMed]
- Marsh, K.M.; Ferng, A.S.; Pilikian, T.; Desai, A.; Avery, R.; Friedman, M.; Oliva, I.; Jokerst, C.; Schipper, D.; Khalpey, Z. Anti-inflammatory properties of amniotic membrane patch following pericardiectomy for constrictive pericarditis. J. Cardiothorac. Surg. 2017, 12, 6. [Google Scholar] [CrossRef] [PubMed]
- Becker, M.; Maring, J.A.; Schneider, M.; Martin, A.X.H.; Seifert, M.; Klein, O.; Braun, T.; Falk, V.; Stamm, C. Towards a Novel Patch Material for Cardiac Applications: Tissue-Specific Extracellular Matrix Introduces Essential Key Features to Decellularized Amniotic Membrane. Int. J. Mol. Sci. 2018, 19, 1032. [Google Scholar] [CrossRef] [PubMed]
- Parveen, S.; Singh, S.P.; Panicker, M.M.; Gupta, P.K. Amniotic membrane as novel scaffold for human iPSC-derived cardiomyogenesis. Vitr. Cell. Dev. Biol. Anim. 2019, 55, 272–284. [Google Scholar] [CrossRef]

| Authors | Tissue Engineering Applications | Amniotic Membrane Formats | Modalities of Amniotic Membrane Usage | Cells Seeded on Amniotic Membrane | Sides of Cells Seeding | Assessment |
|---|---|---|---|---|---|---|
| Shortt et al., 2009 | Ocular surface | Cryopreserved or Decellularized + Cryopreserved | Single membrane | Human limbal epithelial stem cells | Basement membrane (?) | In vitro/Ex vivo |
| Zhang et al., 2013 | Ocular surface | Cryopreserved or De-epithelialized | Single membrane | Human limbal epithelial cells | Basement membrane | In vitro/Ex vivo |
| Che et al., 2019 | Ocular surface | De-epithelialized | Multilayer ultrathin amnion (3–4 layers) | Human corneal stromal cells Keratocytes | Basement membrane Cells between the layers | In vitro/Ex vivo |
| Bandeira et al., 2019 | Ocular surface | Cryopreserved + De-epithelialized | Single membrane/Cover | Human conjunctival epithelial cells | Basement membrane | Clinical study |
| Yang et al., 2006 | Skin | Cryopreserved + De-epithelialized | Single membrane/Cover | Human keratinocytes | Basement membrane | In vitro/Ex vivo + In vivo |
| Kim et al., 2008 | Skin | Cryopreserved + De-epithelialized | Single membrane/Cover | Rabbit bone marrow autologous or allologous MSC | Basement membrane | In vivo |
| Redondo et al., 2011 | Skin | Cryopreserved + De-epithelialized | Single membrane/Cover | Human melanocytes | Basement membrane | Clinical study |
| Tsai et al., 2007 | Vascular system | Cryopreserved + De-epithelialized sow amnion | Single membrane | Porcine vascular endothelial cells | Basement membrane | In vitro/Ex vivo |
| Niknejad et al., 2011 | Vascular system | Fresh or Cryopreserved or Lyophilized | Single membrane | Rat vascular endothelial cells | Epithelial | In vitro/Ex vivo |
| Lee et al., 2012 | Vascular system | Air-dried + De-epithelialized + Glutaraldehyde | Tube of amnion | Porcine vascular endothelial cells | NS | In vitro/Ex vivo |
| Amensag et al., 2012 | Vascular system | Two cycles of freezing and thawing + Decellularized | Tube of six-layered amnion | Human umbilical vein endothelial cells Human vascular smooth muscle cells | Stromal | In vitro/Ex vivo |
| Amensag et al., 2017 | Vascular system | Two cycles of freezing and thawing + Decellularized | Tube of six-layered amnion | Human vascular smooth muscle cells | NS | In vitro/Ex vivo + In vivo |
| Swim et al., 2018 | Vascular system | Decellularized + Lyophilized | Multilayer amnion/Cover | Human thymus-derived MSC Human umbilical cord blood MSC Human umbilical vein endothelial cells Cardiac myocytes Arterial smooth muscle cells | NS | In vitro/Ex vivo + In vivo |
| Sharifiaghdas et al., 2009 | Vaginal and bladder | Fresh + De-epithelialized | Single membrane | Human bladder smooth muscle cells | Basement membrane | In vitro/Ex vivo |
| Seyed-Forootan et al., 2018 | Vaginal and bladder | Fresh | Two layers of amnion/Cover | Autologous skin fibroblasts | NS | Clinical study |
| Sharifiaghdas et al., 2007 | Urethra | Fresh + De-epithelialized | Single membrane | Mouse urothelial cells | Basement membrane | In vitro/Ex vivo |
| Sartoneva et al., 2011 | Urethra | Fresh + De-epithelialized | Amnion attached to a membrane fixation device (cell crowns) | Human urothelial cell | NS | In vitro/Ex vivo |
| Jerman et al., 2014 | Urethra | Cryopreserved | Single membrane | Porcine urethral cells | Epithelial or Basement membrane or Stromal | In vitro/Ex vivo |
| Wang et al., 2014 | Urethra | De-epithelialized | Single membrane/Cover | Rabbit urethral epithelial cells | NS | In vitro/Ex vivo + In vivo |
| Chen et al., 2018 | Urethra | Decellularized + Lyophilized | Tube of amnion | Allogenic canine endothelial progenitor cells +/− bone marrow MSC | NS | In vitro/Ex vivo + In vivo |
| Jin et al., 2007 | Cartilage | Cryopreserved or Cryopreserved + De-epithelialized | Single membrane/Cover | Rabbit chondrocytes | Epithelial or Basement membrane or Stromal | In vitro/Ex vivo + In vivo |
| Díaz-Prado et al., 2010 | Cartilage | Cryopreserved or Cryopreserved + De-epithelialized | Single membrane | Human chondrocytes | Epithelial or Basement membrane or Stromal | In vitro/Ex vivo |
| Krishnamurithy et al., 2011 | Cartilage | Air-dried or Lyophilized | Single membrane | Rabbit chondrocytes | Basement membrane | In vitro/Ex vivo |
| Tan et al., 2011 | Cartilage | Air-dried or Lyophilized | Single membrane | Rabbit bone marrow MSC | NS | In vitro/Ex vivo |
| Garcia et al., 2015 | Cartilage | Fresh or cryopreserved or and cryopreserved | Single membrane/Cover | Sheep bone marrow MSC | Stromal | In vitro/Ex vivo + In vivo |
| Tsugawa et al., 2011 | Bone | Cryopreserved + De-epithelialized | Single membrane/Cover | Mouse bone marrow-derived osteoblast cells | Stromal | In vitro/Ex vivo + In vivo |
| Chen et al., 2012 | Bone | Decellularized + Dried | Single membrane | Human dental apical papilla cells | Basement membrane or Stromal | In vitro/Ex vivo |
| Semyari et al., 2015. | Bone | Fresh decellularized rabbit amnion | Single membrane/Cover | Rabbit adipose-derived MSC | NS | In vitro/Ex vivo + In vivo |
| Akazawa et al., 2016 | Bone | Cryopreserved + Decellularized | Single membrane/Cover | Human calvaria osteoblasts Human dermal fibroblasts Human umbilical vein endothelial cells Mouse osteoblasts Human periodontal ligament stem cells | NS | In vitro/Ex vivo + In vivo |
| Tang et al., 2017 | Bone | Fresh + De-epithelialized | Single membrane | Human umbilical vein endothelial cells Rat bone marrow MSC | NS | In vitro/Ex vivo |
| Akhlaghi et al., 2019 | Bone | Decellularized + Lyophilized | Single membrane/Cover | Buccal fat pad-derived stem cells | NS | Clinical study |
| Ahn et al., 2006 | Oral mucosa | De-epithelialized + Lyophilized | Single membrane/Cover | Rabbit oral keratinocytes | Basement membrane | In vitro/Ex vivo + In vivo |
| Amemiya et al., 2010 | Oral mucosa | Cryopreserved + De-epithelialized | Single membrane/Cover | Human oral mucosal epithelial cells | Basement membrane | In vitro/Ex vivo + In vivo |
| Amemiya et al., 2009/2015 | Oral mucosa | Cryopreserved + De-epithelialized | Single membrane/Cover | Human oral mucosal epithelial cells | Basement membrane | Clinical study |
| Hsueh et al., 2016 | Oral mucosa | De-epithelialized + air dried | Single membrane | Human oral mucosal epithelial cells | Basement membrane | In vitro/Ex vivo |
| Amemiya et al., 2008 | Periodontal | Cryopreserved + De-epithelialized | Single membrane/Cover | Dog periodontal ligament cells | Basement membrane | In vivo |
| Iwasaki et al., 2013 | Periodontal | Decellularized + Cryopreserved | Single membrane/Cover | Human periodontal ligament stem cells | NS | In vitro/Ex vivo + In vivo |
| Amemiya et al., 2014 | Periodontal | De-epithelialized | Single membrane/Cover | Human periosteum derived stem cells | NS | In vitro/Ex vivo + In vivo |
| Wu et al., 2015 | Periodontal | De-epithelialized | Single membrane/Cover | Human adipose-derived MSC | Basement membrane | In vitro/Ex vivo + In vivo |
| Honjo et al., 2015 | Periodontal | Cryopreserved + De-epithelialized | Amnion placed on a cell culture insert | dental pulp-derived cell sheet | Basement membrane | In vitro/Ex vivo |
| Zhang et al., 2006 | Nerve | NS in the abstract/Not translated to English | A scroll/wrap of amnion | Autogenous Schwann cell | NS in the abstract/Not translated to English | In vivo |
| Li et al., 2013 | Nerve | Fresh | A scroll/wrap of amnion | Allogenic human umbilical cord MSC | NS | Clinical study |
| He et al., 2002 | Tendon | De-epithelialized + Cryopreserved | A scroll/wrap of amnion | Fetal rabbit skin fibroblasts | Attachment on ECM and proliferation on stromal layer | In vitro/Ex vivo + In vivo |
| Parveen et al., 2019 | Cardiac | Trypsinized + Cryopreserved | Single membrane | Human-induced pluripotent stem cell-derived cardiomyocytes | Basement membrane (?) | In vitro/Ex vivo |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fénelon, M.; Catros, S.; Meyer, C.; Fricain, J.-C.; Obert, L.; Auber, F.; Louvrier, A.; Gindraux, F. Applications of Human Amniotic Membrane for Tissue Engineering. Membranes 2021, 11, 387. https://doi.org/10.3390/membranes11060387
Fénelon M, Catros S, Meyer C, Fricain J-C, Obert L, Auber F, Louvrier A, Gindraux F. Applications of Human Amniotic Membrane for Tissue Engineering. Membranes. 2021; 11(6):387. https://doi.org/10.3390/membranes11060387
Chicago/Turabian StyleFénelon, Mathilde, Sylvain Catros, Christophe Meyer, Jean-Christophe Fricain, Laurent Obert, Frédéric Auber, Aurélien Louvrier, and Florelle Gindraux. 2021. "Applications of Human Amniotic Membrane for Tissue Engineering" Membranes 11, no. 6: 387. https://doi.org/10.3390/membranes11060387
APA StyleFénelon, M., Catros, S., Meyer, C., Fricain, J.-C., Obert, L., Auber, F., Louvrier, A., & Gindraux, F. (2021). Applications of Human Amniotic Membrane for Tissue Engineering. Membranes, 11(6), 387. https://doi.org/10.3390/membranes11060387









