A Novel Electrospinning Polyacrylonitrile Separator with Dip-Coating of Zeolite and Phenoxy Resin for Li-ion Batteries
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Electrospun PAN Separator
2.3. Preparation of Composite Separator
2.4. Characterization of Composite Separator
2.5. Electrochemical Performance of the Composite Separator
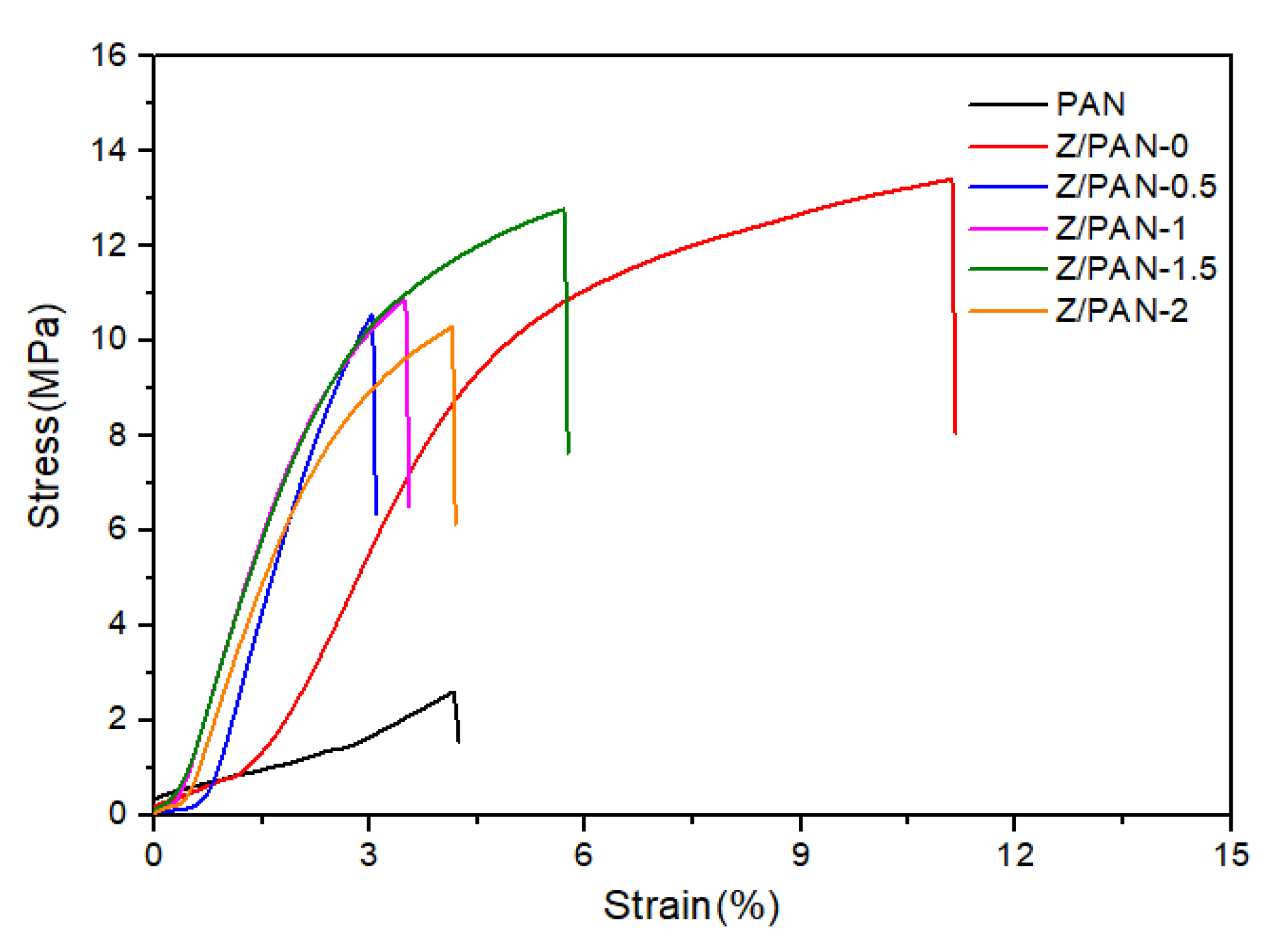
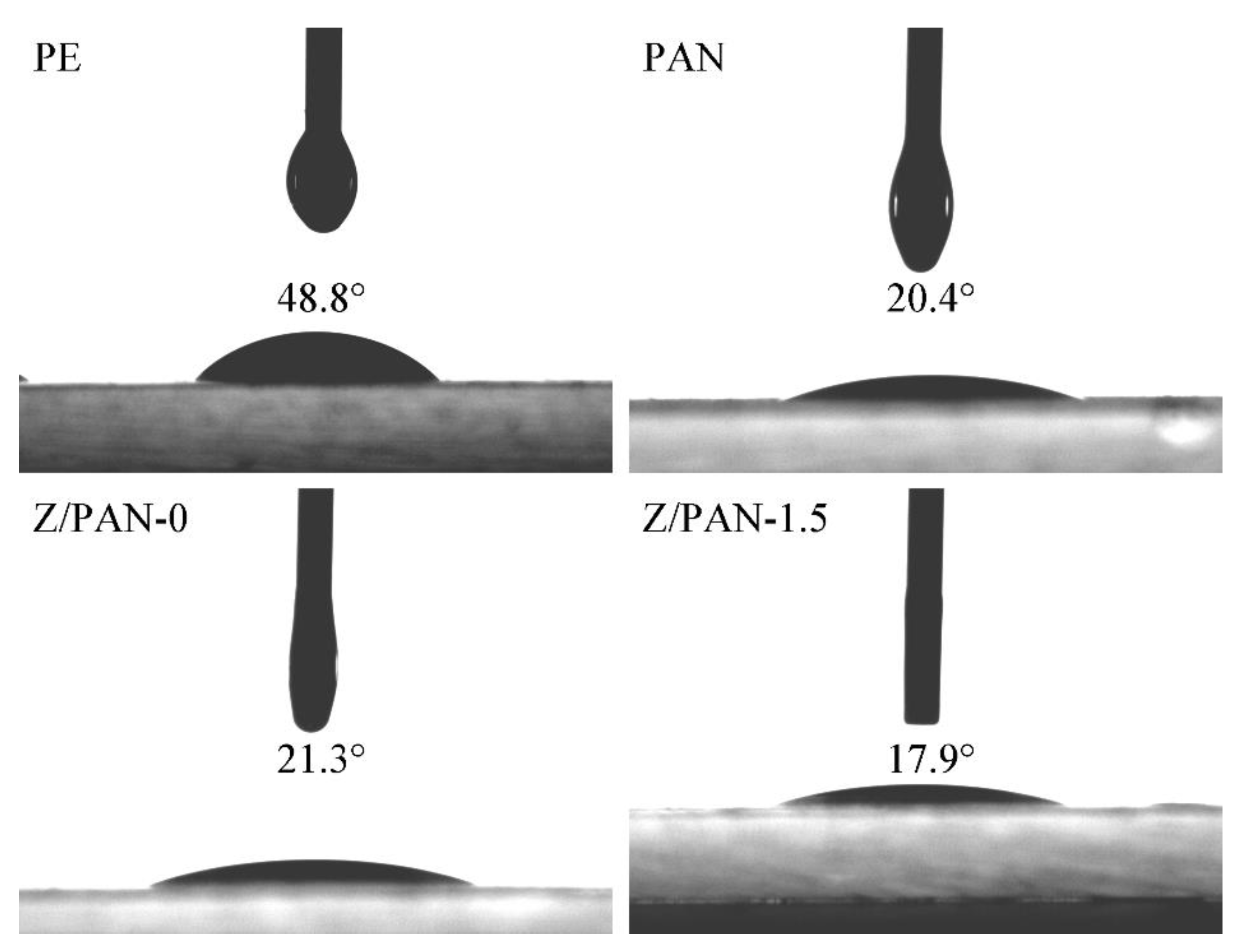
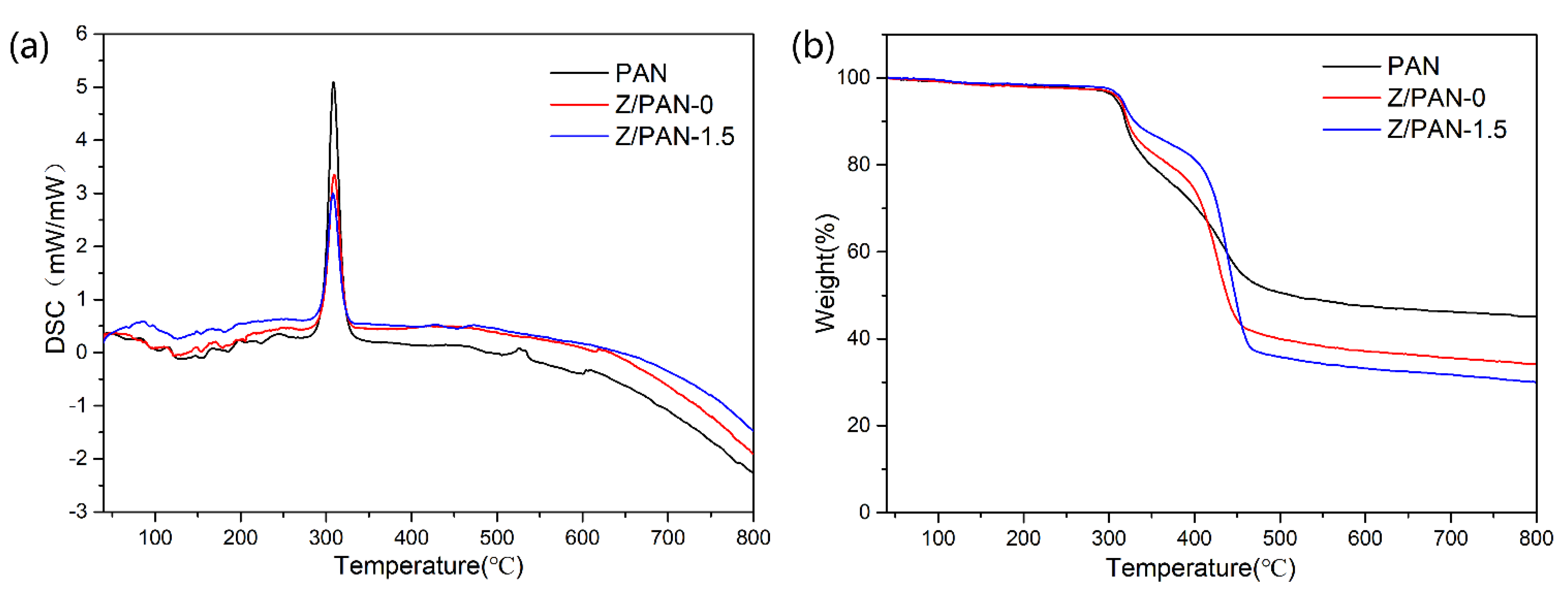
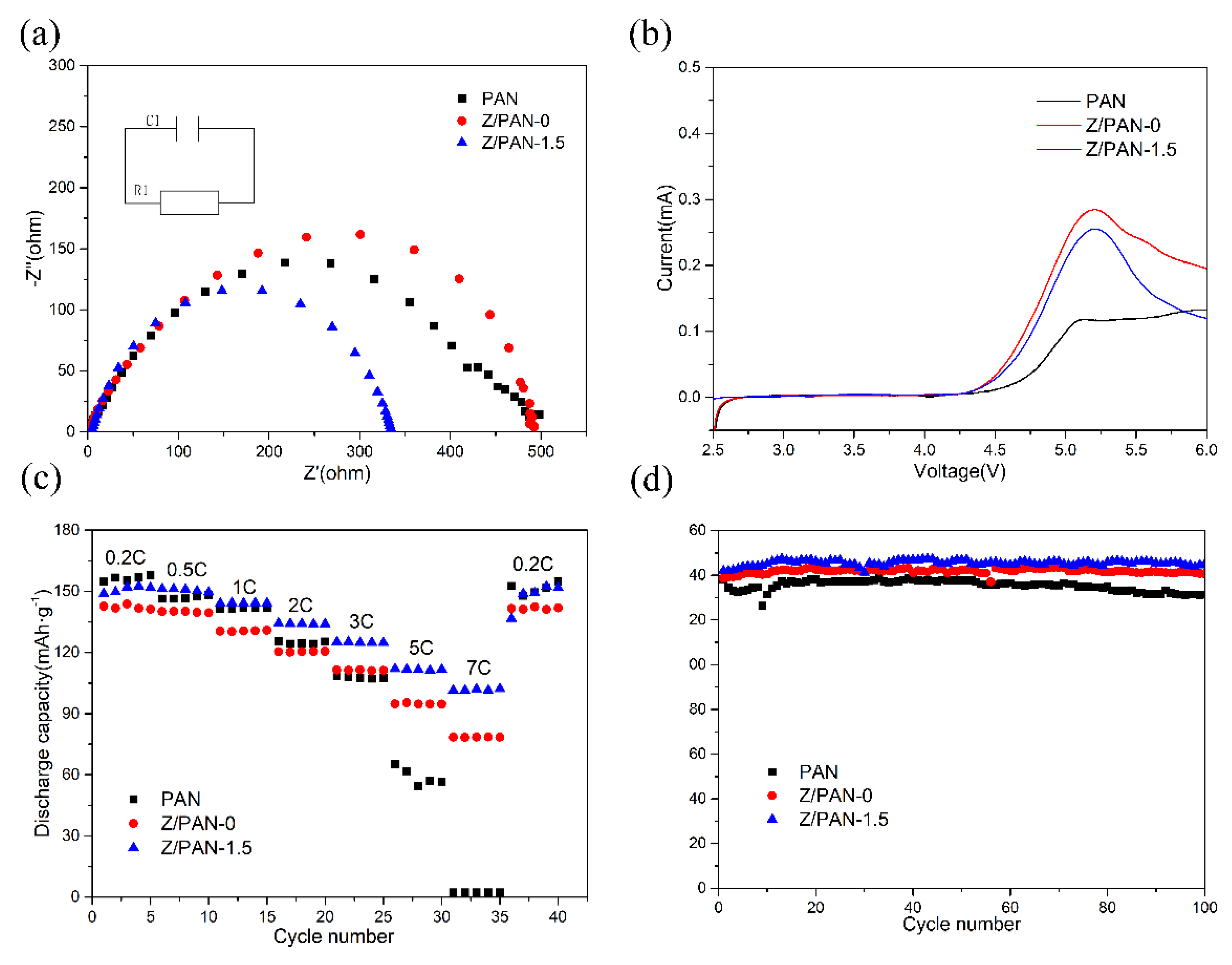
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Naik, K.M.; Sampath, S. Two-Step Oxygen Reduction on Spinel NiFe2O4 Catalyst: Rechargeable, Aqueous Solution- and Gel-Based, Zn-Air Batteries. Electrochim. Acta 2018, 292, 268–275. [Google Scholar] [CrossRef]
- Lee, H.; Yanilmaz, M.; Toprakci, O.; Fu, K.; Zhang, X. A Review of Recent Developments in Membrane Separators for Rechargeable Lithium-Ion Batteries. Energy Environ. Sci. 2014, 7, 3857–3886. [Google Scholar] [CrossRef]
- Waqas, M.; Ali, S.; Feng, C.; Chen, D.; Han, J.; He, W. Recent Development in Separators for High-Temperature Lithium-Ion Batteries. Small 2019, 15. [Google Scholar] [CrossRef]
- Costa, C.M.; Lee, Y.H.; Kim, J.H.; Lee, S.Y.; Lanceros-Méndez, S. Recent Advances on Separator Membranes for Lithium-Ion Battery Applications: From Porous Membranes to Solid Electrolytes. Energy Storage Mater. 2019, 22, 346–375. [Google Scholar] [CrossRef]
- Zheng, W.; Zhu, Y.; Na, B.; Lv, R.; Liu, H.; Li, W.; Zhou, H. Hybrid Silica Membranes with a Polymer Nanofiber Skeleton and Their Application as Lithium-Ion Battery Separators. Compos. Sci. Technol. 2017, 144, 178–184. [Google Scholar] [CrossRef]
- Kukunuri, S.; Naik, K.; Sampath, S. Effects of Composition and Nanostructuring of Palladium Selenide Phases, Pd4Se, Pd7Se4 and Pd17Se15, on ORR Activity and Their Use in Mg-Air Batteries. J. Mater. Chem. A 2017, 5, 4660–4670. [Google Scholar] [CrossRef]
- Naik, K.M. Highly Stable Spinel Oxide Cathode for Rechargeable Li-O2Batteries in Non-Aqueous Liquid and Gel-Based Electrolytes. ACS Appl. Energy Mater. 2021, 4, 1014–1020. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, S.; Fan, W.; Wang, J.; Li, C. Long Cycling, Thermal Stable, Dendrites Free Gel Polymer Electrolyte for Flexible Lithium Metal Batteries. Electrochim. Acta 2019, 301, 304–311. [Google Scholar] [CrossRef]
- Chao, C.Y.; Feng, Y.F.; Hua, K.; Li, H.; Wu, L.J.; Zhou, Y.S.; Dong, Z.W. Enhanced Wettability and Thermal Stability of Polypropylene Separators by Organic–Inorganic Coating Layer for Lithium-Ion Batteries. J. Appl. Polym. Sci. 2018, 135, 1–7. [Google Scholar] [CrossRef]
- Jia, S.; Long, J.; Li, J.; Yang, S.; Huang, K.; Yang, N.; Liang, Y.; Xiao, J. Biomineralized Zircon-Coated PVDF Nanofiber Separator for Enhancing Thermo- and Electro-Chemical Properties of Lithium Ion Batteries. J. Mater. Sci. 2020, 55, 14907–14921. [Google Scholar] [CrossRef]
- Arora, P.; Zhang, Z. Battery Separators. Chem. Rev. 2004, 104, 4419–4462. [Google Scholar] [CrossRef]
- Deimede, V.; Elmasides, C. Separators for Lithium-Ion Batteries: A Review on the Production Processes and Recent Developments. Energy Technol. 2015, 3, 453–468. [Google Scholar] [CrossRef]
- Dai, J.; Shi, C.; Li, C.; Shen, X.; Peng, L.; Wu, D.; Sun, D.; Zhang, P.; Zhao, J. A Rational Design of Separator with Substantially Enhanced Thermal Features for Lithium-Ion Batteries by the Polydopamine-Ceramic Composite Modification of Polyolefin Membranes. Energy Environ. Sci. 2016, 9, 3252–3261. [Google Scholar] [CrossRef]
- Zhang, Y.; Yuan, J.J.; Song, Y.Z.; Yin, X.; Sun, C.C.; Zhu, L.P.; Zhu, B.K. Tannic Acid/Polyethyleneimine-Decorated Polypropylene Separators for Li-Ion Batteries and the Role of the Interfaces between Separator and Electrolyte. Electrochim. Acta 2018, 275, 25–31. [Google Scholar] [CrossRef]
- Yu, L.; Miao, J.; Jin, Y.; Lin, J.Y.S. A Comparative Study on Polypropylene Separators Coated with Different Inorganic Materials for Lithium-Ion Batteries. Front. Chem. Sci. Eng. 2017, 11, 346–352. [Google Scholar] [CrossRef]
- Liu, L.; Wang, Y.; Gao, C.; Yang, C.; Wang, K.; Li, H.; Gu, H. Ultrathin ZrO2-Coated Separators Based on Surface Sol-Gel Process for Advanced Lithium Ion Batteries. J. Memb. Sci. 2019, 592, 117368. [Google Scholar] [CrossRef]
- Fan, H.; Yip, N.Y. An AlOOH-Coated Polyimide Electrospun Fibrous Membrane as a High-Safety Lithium-Ion Battery Separator. J. Memb. Sci. 2019, 572, 512–519. [Google Scholar] [CrossRef]
- Xie, Y.; Pan, Y.; Cai, P. Novel PVA-Based Porous Separators Prepared via Freeze-Drying for Enhancing Performance of Lithium-Ion Batteries. Ind. Eng. Chem. Res. 2020, 59, 15242–15254. [Google Scholar] [CrossRef]
- Al Zoubi, W.; Kamil, M.P.; Fatimah, S.; Nisa, N.; Ko, Y.G. Recent Advances in Hybrid Organic-Inorganic Materials with Spatial Architecture for State-of-the-Art Applications. Prog. Mater. Sci. 2020, 112, 100663. [Google Scholar] [CrossRef]
- Barbosa, J.C.; Dias, J.P.; Lanceros-Méndez, S.; Costa, C.M. Recent Advances in Poly(Vinylidene Fluoride) and Its Copolymers for Lithium-Ion Battery Separators. Membranes 2018, 8, 45. [Google Scholar] [CrossRef]
- Cao, L.; An, P.; Xu, Z.; Huang, J. Performance Evaluation of Electrospun Polyimide Non-Woven Separators for High Power Lithium-Ion Batteries. J. Electroanal. Chem. 2016, 767, 34–39. [Google Scholar] [CrossRef]
- Shayapat, J.; Chung, O.H.; Park, J.S. Electrospun Polyimide-Composite Separator for Lithium-Ion Batteries. Electrochim. Acta 2015, 170, 110–121. [Google Scholar] [CrossRef]
- Baskoro, F.; Wong, H.Q.; Yen, H.J. Strategic Structural Design of a Gel Polymer Electrolyte toward a High Efficiency Lithium-Ion Battery. ACS Appl. Energy Mater. 2019, 2, 3937–3971. [Google Scholar] [CrossRef]
- Li, Y.; Li, Q.; Tan, Z. A Review of Electrospun Nanofiber-Based Separators for Rechargeable Lithium-Ion Batteries. J. Power Sources 2019, 443, 227262. [Google Scholar] [CrossRef]
- Yu, H.; Shi, Y.; Yuan, B.; He, Y.; Qiao, L.; Wang, J.; Lin, Q.; Chen, Z.; Han, E. Recent Developments of Polyimide Materials for Lithium-Ion Battery Separators. Ionics 2021, 907–923. [Google Scholar] [CrossRef]
- Ngai, K.S.; Ramesh, S.; Ramesh, K.; Juan, J.C. A Review of Polymer Electrolytes: Fundamental, Approaches and Applications. Ionics 2016, 22, 1259–1279. [Google Scholar] [CrossRef]
- Tong, Y.; Xu, Y.; Chen, D.; Xie, Y.; Chen, L.; Que, M.; Hou, Y. Deformable and Flexible Electrospun Nanofiber-Supported Cross-Linked Gel Polymer Electrolyte Membranes for High Safety Lithium-Ion Batteries. RSC Adv. 2017, 7, 22728–22734. [Google Scholar] [CrossRef]
- Pi, J.K.; Wu, G.P.; Yang, H.C.; Arges, C.G.; Xu, Z.K. Separators with Biomineralized Zirconia Coatings for Enhanced Thermo- and Electro-Performance of Lithium-Ion Batteries. ACS Appl. Mater. Interfaces 2017, 9, 21971–21978. [Google Scholar] [CrossRef]
- Lee, Y.Y.; Liu, Y.L. Crosslinked Electrospun Poly(Vinylidene Difluoride) Fiber Mat as a Matrix of Gel Polymer Electrolyte for Fast-Charging Lithium-Ion Battery. Electrochim. Acta 2017, 258, 1329–1335. [Google Scholar] [CrossRef]
- Verdier, N.; Lepage, D.; Zidani, R.; Prébé, A.; Aymé-Perrot, D.; Pellerin, C.; Dollé, M.; Rochefort, D. Cross-Linked Polyacrylonitrile-Based Elastomer Used as Gel Polymer Electrolyte in Li-Ion Battery. ACS Appl. Energy Mater. 2020, 3, 1099–1110. [Google Scholar] [CrossRef]
- Zhang, J.; Xiang, Y.; Jamil, M.I.; Lu, J.; Zhang, Q.; Zhan, X.; Chen, F. Polymers/Zeolite Nanocomposite Membranes with Enhanced Thermal and Electrochemical Performances for Lithium-Ion Batteries. J. Memb. Sci. 2018, 564, 753–761. [Google Scholar] [CrossRef]
- Xiao, W.; Gao, Z.; Wang, S.; Liu, J.; Yan, C. A Novel NaA-Type Zeolite-Embedded Composite Separator for Lithium-Ion Battery. Mater. Lett. 2015, 145, 177–179. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Liang, J.; Wu, K.; Xu, L.; Wang, J. Design of a High Performance Zeolite/Polyimide Composite Separator for Lithium-Ion Batteries. Polymers 2020, 12, 764. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Hao, H.U.; Bo, G.U.; Zhang, H. Preparation and Performance of PVDF-Zeolite Composite Separator for Lithium-Ion Batteries. Acta Mater. Compos. Sin. 2017, 34, 625–631. [Google Scholar]
- Jie, J.; Liu, Y.; Cong, L.; Zhang, B.; Lu, W.; Zhang, X.; Liu, J.; Xie, H.; Sun, L. High-Performance PVDF-HFP Based Gel Polymer Electrolyte with a Safe Solvent in Li Metal Polymer Battery. J. Energy Chem. 2020, 49, 80–88. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, F.; Liu, L.; Xiao, S.; Chang, Z.; Wu, Y. Composite of a Nonwoven Fabric with Poly(Vinylidene Fluoride) as a Gel Membrane of High Safety for Lithium Ion Battery. Energy Environ. Sci. 2013, 6, 618–624. [Google Scholar] [CrossRef]
- Li, D.; Xu, H.; Liu, Y.; Jiang, Y.; Li, F.; Xue, B. Fabrication of Diatomite/Polyethylene Terephthalate Composite Separator for Lithium-Ion Battery. Ionics 2019, 25, 5341–5351. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, W.; Zhang, J. A Waterborne SuperLEphilic and Thermostable Separator Based on Natural Clay Nanorods for High-Voltage Lithium-Ion Batteries. Mater. Today Energy 2020, 16, 1–9. [Google Scholar] [CrossRef]
- Zhu, X.; Jiang, X.; Ai, X.; Yang, H.; Cao, Y. TiO2 Ceramic-Grafted Polyethylene Separators for Enhanced Thermostability and Electrochemical Performance of Lithium-Ion Batteries. J. Memb. Sci. 2016, 504, 97–103. [Google Scholar] [CrossRef]




| Ionic Conductivity, mS·cm−1 | Electrolyte Uptake, % | Porosity, % | |
|---|---|---|---|
| PE | 0.592 | 45.2 | 41.4 |
| PAN | 1.982 | 385.7 | 73.2 |
| Z/PAN-0 | 1.712 | 195.2 | 55.5 |
| Z/PAN-1.5 | 2.158 | 308.1 | 68.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, D.; Wang, X.; Liang, J.; Zhang, Z.; Chen, W. A Novel Electrospinning Polyacrylonitrile Separator with Dip-Coating of Zeolite and Phenoxy Resin for Li-ion Batteries. Membranes 2021, 11, 267. https://doi.org/10.3390/membranes11040267
Chen D, Wang X, Liang J, Zhang Z, Chen W. A Novel Electrospinning Polyacrylonitrile Separator with Dip-Coating of Zeolite and Phenoxy Resin for Li-ion Batteries. Membranes. 2021; 11(4):267. https://doi.org/10.3390/membranes11040267
Chicago/Turabian StyleChen, Danxia, Xiang Wang, Jianyu Liang, Ze Zhang, and Weiping Chen. 2021. "A Novel Electrospinning Polyacrylonitrile Separator with Dip-Coating of Zeolite and Phenoxy Resin for Li-ion Batteries" Membranes 11, no. 4: 267. https://doi.org/10.3390/membranes11040267
APA StyleChen, D., Wang, X., Liang, J., Zhang, Z., & Chen, W. (2021). A Novel Electrospinning Polyacrylonitrile Separator with Dip-Coating of Zeolite and Phenoxy Resin for Li-ion Batteries. Membranes, 11(4), 267. https://doi.org/10.3390/membranes11040267






