Water Pores in Planar Lipid Bilayers at Fast and Slow Rise of Transmembrane Voltage
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Experimental Setups
2.3. Measurement Protocols
2.4. Experimental Data Analysis
2.5. The Rate of the Planar Lipid Bilayer Capacitance Change at
2.6. Calculation of a Fraction of the Planar Lipid Bilayer That Is Occupied By Pores
3. Results
3.1. Experimental Results
3.2. Modeling Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| POPC | 1-pamitoyl 2-oleoyl phosphatidylcholine |
| POPS | 1-pamitoyl 2-oleoyl phosphatidylserine |
| MD | Molecular dynamics simulation |
| GUV | Giant unilamellar vesicles |
References
- Siontorou, C.; Nikoleli, G.P.; Nikolelis, D.; Karapetis, S. Artificial Lipid Membranes: Past, Present, and Future. Membranes 2017, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Brosseau, C.; Sabri, E. Resistor–capacitor modeling of the cell membrane: A multiphysics analysis. J. Appl. Phys. 2021, 129, 011101. [Google Scholar] [CrossRef]
- Naumowicz, M.; Figaszewski, Z.A. Pore Formation in Lipid Bilayer Membranes made of Phosphatidylcholine and Cholesterol Followed by Means of Constant Current. Cell Biochem. Biophys. 2013, 66, 109–119. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kramar, P.; Miklavčič, D.; Kotulska, M.; Lebar, A. Voltage- and Current-Clamp Methods for Determination of Planar Lipid Bilayer Properties; Elsevier Inc.: Amsterdam, The Netherlands, 2010; Volume 11. [Google Scholar] [CrossRef]
- Naumowicz, M.; Kotynska, J.; Petelska, A.; Figaszewski, Z. Impedance analysis of phosphatidylcholine membranes modified with valinomycin. Eur. Biophys. J. 2006, 35, 239–246. [Google Scholar] [CrossRef]
- Shlyonsky, V.; Dupuis, F.; Gall, D. The OpenPicoAmp: An Open-Source Planar Lipid Bilayer Amplifier for Hands-On Learning of Neuroscience. PLoS ONE 2014, 9, e108097. [Google Scholar] [CrossRef]
- Mosgaard, L.D.; Zecchi, K.A.; Heimburg, T.; Budvytyte, R. The effect of the nonlinearity of the response of lipid membranes to voltage perturbations on the interpretation of their electrical properties. A new theoretical description. Membranes 2015, 5, 495–512. [Google Scholar] [CrossRef]
- Pavlin, M.; Kotnik, T.; Miklavčič, D.; Kramar, P.; Maček Lebar, A. Chapter Seven Electroporation of Planar Lipid Bilayers and Membranes. Adv. Planar Lipid Bilayers Liposomes 2008, 6, 165–226. [Google Scholar] [CrossRef]
- Kotnik, T.; Rems, L.; Tarek, M.; Miklavčič, D. Membrane Electroporation and Electropermeabilization: Mechanisms and Models. Annu. Rev. Biophys. 2019, 48, 63–91. [Google Scholar] [CrossRef]
- Hu, Y.; Sinha, S.K.; Patel, S. Investigating Hydrophilic Pores in Model Lipid Bilayers Using Molecular Simulations: Correlating Bilayer Properties with Pore-Formation Thermodynamics. Langmuir 2015, 31, 6615–6631. [Google Scholar] [CrossRef]
- Alvarez, O.; Latorre, R. Voltage-dependent capacitance in lipid bilayers made from monolayers. Biophys. J. 1978, 21, 1–17. [Google Scholar] [CrossRef]
- Heimburg, T. The capacitance and electromechanical coupling of lipid membranes close to transitions: The effect of electrostriction. Biophys. J. 2012, 103, 918–929. [Google Scholar] [CrossRef]
- Sabri, E.; Lasquellec, S.; Brosseau, C. Electromechanical modeling of the transmembrane potential-dependent cell membrane capacitance. Appl. Phys. Lett. 2020, 117, 043701. [Google Scholar] [CrossRef]
- Freeman, S.A.; Wang, M.A.; Weaver, J.C. Theory of electroporation of planar bilayer membranes: Predictions of the aqueous area, change in capacitance, and pore-pore separation. Biophys. J. 1994, 67, 42–56. [Google Scholar] [CrossRef]
- Akimov, S.A.; Volynsky, P.E.; Galimzyanov, T.R.; Kuzmin, P.I.; Pavlov, K.V.; Batishchev, O.V. Pore formation in lipid membrane II: Energy landscape under external stress. Sci. Rep. 2017, 7, 12509. [Google Scholar] [CrossRef]
- Abidor, I.G.; Arakelyan, V.B.; Chernomordik, L.V.; Chizmadzhev, Y.A.; Pastushenko, V.F.; Tarasevich, M.P. Electric breakdown of bilayer lipid membranes. I. The main experimental facts and their qualitative discussion. J. Electroanal. Chem. 1979, 104, 37–52. [Google Scholar] [CrossRef]
- Tieleman, D.P.; Leontiadou, H.; Mark, A.E.; Marrink, S.J. Simulation of Pore Formation in Lipid Bilayers by Mechanical Stress and Electric Fields. J. Am. Chem. Soc. 2003, 125, 6382–6383. [Google Scholar] [CrossRef]
- Anosov, A.A.; Smirnova, E.Y.; Sharakshane, A.A.; Nikolayeva, E.A.; Zhdankina, Y.S. Increase in the current variance in bilayer lipid membranes near phase transition as a result of the occurrence of hydrophobic defects. Biochim. Biophys. Acta Biomembr. 2020, 1862, 183147. [Google Scholar] [CrossRef]
- Melikov, K.C.; Frolov, V.A.; Shcherbakov, A.; Samsonov, A.V.; Chizmadzhev, Y.A.; Chernomordik, L.V. Voltage-Induced Nonconductive Pre-Pores and Metastable Single Pores in Unmodified Planar Lipid Bilayer. Biophys. J. 2001, 80, 1829–1836. [Google Scholar] [CrossRef]
- Kalinowski, S.; Ibron, G.; Bryl, K.; Figaszewski, Z. Chronopotentiometric studies of electroporation of bilayer lipid membranes. Biochim. Biophys. Acta Biomembr. 1998, 1369, 204–212. [Google Scholar] [CrossRef][Green Version]
- Kotulska, M. Natural fluctuations of an electropore show fractional Lévy stable motion. Biophys. J. 2007, 92, 2412–2421. [Google Scholar] [CrossRef]
- Kramar, P.; Delemotte, L.; Lebar, A.M.; Kotulska, M.; Tarek, M.; Miklavčič, D. Molecular-level characterization of lipid membrane electroporation using linearly rising current. J. Membr. Biol. 2012, 245, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Akimov, S.A.; Volynsky, P.E.; Galimzyanov, T.R.; Kuzmin, P.I.; Pavlov, K.V.; Batishchev, O.V. Pore formation in lipid membrane I: Continuous reversible trajectory from intact bilayer through hydrophobic defect to transversal pore. Sci. Rep. 2017, 7, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Evans, E.; Heinrich, V.; Ludwig, F.; Rawicz, W. Dynamic tension spectroscopy and strength of biomembranes. Biophys. J. 2003, 85, 2342–2350. [Google Scholar] [CrossRef]
- Kramar, P.; Miklavcic, D.; Lebar, A.M. Determination of the lipid bilayer breakdown voltage by means of linear rising signal. Bioelectrochemistry 2007, 70, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Kalinowski, S.; Figaszewski, Z. A four-electrode system for measurement of bilayer lipid membrane capacitance. Meas. Sci. Technol. 1995, 6, 1043–1049. [Google Scholar] [CrossRef]
- Kalinowski, S.; Figaszewski, Z. A four-electrode potentiostat-galvanostat for studies of bilayer lipid membranes. Meas. Sci. Technol. 1995, 6, 1050–1055. [Google Scholar] [CrossRef]
- Montal, M.; Mueller, P. Formation of Bimolecular Membranes from Lipid Monolayers and a Study of Their Electrical Properties. Proc. Natl. Acad. Sci. USA 1972, 69, 3561–3566. [Google Scholar] [CrossRef]
- Kramar, P.; Miklavčič, D.; Lebar, A.M. A system for the determination of planar lipid bilayer breakdown voltage and its applications. IEEE Trans. Nanobiosci. 2009, 8, 132–138. [Google Scholar] [CrossRef]
- Dimitrov, D.S. Electric field-induced breakdown of lipid bilayers and cell membranes: A thin viscoelastic film model. J. Membr. Biol. 1984, 78, 53–60. [Google Scholar] [CrossRef]
- Sabotin, I.; Lebar, A.; Miklavčič, D.; Kramar, P. Measurement protocol for planar lipid bilayer viscoelastic properties. IEEE Trans. Dielectr. Electr. Insul. 2009, 16, 1236–1242. [Google Scholar] [CrossRef]
- Levine, Z.A.; Vernier, P.T. Life cycle of an electropore: Field-dependent and field-independent steps in pore creation and annihilation. J. Membr. Biol. 2010, 236, 27–36. [Google Scholar] [CrossRef]
- Shahane, G.; Ding, W.; Palaiokostas, M.; Orsi, M. Physical properties of model biological lipid bilayers: Insights from all-atom molecular dynamics simulations. J. Mol. Model. 2019, 25, 1–13. [Google Scholar] [CrossRef]
- Jurkiewicz, P.; Cwiklik, L.; Vojtíšková, A.; Jungwirth, P.; Hof, M. Structure, dynamics, and hydration of POPC/POPS bilayers suspended in NaCl, KCl, and CsCl solutions. Biochim. Biophys. Acta Biomembr. 2012, 1818, 609–616. [Google Scholar] [CrossRef]
- Kastl, K.; Menke, M.; Lüthgens, E.; Faiß, S.; Gerke, V.; Janshoff, A.; Steinem, C. Partially reversible adsorption of annexin A1 on POPC/POPS bilayers investigated by QCM measurements, SFM, and DMC simulations. ChemBioChem 2006, 7, 106–115. [Google Scholar] [CrossRef]
- Troiano, G.C.; Tung, L.; Sharma, V.; Stebe, K.J. The reduction in electroporation voltages by the addition of a surfactant to planar lipid bilayers. Biophys. J. 1998, 75, 880–888. [Google Scholar] [CrossRef][Green Version]
- Naumowicz, M.; Petelska, A.D.; Figaszewski, Z.A. Physicochemical analysis of phosphatidylcholine-ceramide system in bilayer lipid membranes. Acta Biochim. Pol. 2008, 55, 721–730. [Google Scholar] [CrossRef]
- Cannon, B.; Hermansson, M.; Györke, S.; Somerharju, P.; Virtanen, J.A.; Cheng, K.H. Regulation of calcium channel activity by lipid domain formation in planar lipid bilayers. Biophys. J. 2003, 85, 933–942. [Google Scholar] [CrossRef]
- Meleleo, D. Study of Resveratrol’s Interaction with Planar Lipid Models: Insights into Its Location in Lipid Bilayers. Membranes 2021, 11, 132. [Google Scholar] [CrossRef]
- Naumowicz, M.; Figaszewski, Z.A. Impedance Spectroscopic Investigation of the Bilayer Lipid Membranes Formed from the Phosphatidylserine–Ceramide Mixture. J. Membr. Biol. 2009, 227, 67–75. [Google Scholar] [CrossRef]
- Naumowicz, M.; Figaszewski, Z.A. The effect of pH on the electrical capacitance of phosphatidylcholine-phosphatidylserine system in bilayer lipid membrane. J. Membr. Biol. 2014, 247, 361–369. [Google Scholar] [CrossRef]
- Micelli, S.; Gallucci, E.; Meleleo, D.; Stipani, V.; Picciarelli, V. Mitochondrial porin incorporation into black lipid membranes: Ionic and gating contribution to the total current. Bioelectrochemistry 2002, 57, 97–106. [Google Scholar] [CrossRef]
- Kotulska, M.; Basalyga, J.; Derylo, M.B.; Sadowski, P. Metastable pores at the onset of constant-current electroporation. J. Membr. Biol. 2010, 236, 37–41. [Google Scholar] [CrossRef][Green Version]
- Dehez, F.; Delemotte, L.; Kramar, P.; Miklavčič, D.; Tarek, M. Evidence of conducting hydrophobic nanopores across membranes in response to an electric field. J. Phys. Chem. C 2014, 118, 6752–6757. [Google Scholar] [CrossRef]
- Sengel, J.T.; Wallace, M.I. Imaging the dynamics of individual electropores. Proc. Natl. Acad. Sci. USA 2016, 113, 5281–5286. [Google Scholar] [CrossRef]
- What Is the Electric Potential Difference Across Membranes? Available online: http://book.bionumbers.org/what-is-the-electric-potential-difference-across-membranes/ (accessed on 27 March 2021).
- Kinoshita, N.; Unemoto, T.; Kobayashi, H. Proton motive force is not obligatory for growth of Escherichia coli. J. Bacteriol. 1984, 160, 1074–1077. [Google Scholar] [CrossRef]
- Justice, M.J.; Petrusca, D.N.; Rogozea, A.L.; Williams, J.A.; Schweitzer, K.S.; Petrache, I.; Wassall, S.R.; Petrache, H.I. Effects of Lipid Interactions on Model Vesicle Engulfment by Alveolar Macrophages. Biophys. J. 2014, 106, 598–609. [Google Scholar] [CrossRef]
- Sahoo, A.; Matysiak, S. Microscopic Picture of Calcium-Assisted Lipid Demixing and Membrane Remodeling Using Multiscale Simulations. J. Phys. Chem. B 2020, 124, 7327–7335. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, S.J.; Xu, H.; Matysiak, S. Influence of Monovalent Cation Size on Nanodomain Formation in Anionic–Zwitterionic Mixed Bilayers. J. Phys. Chem. B 2017, 121, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Bobone, S.; Hilsch, M.; Storm, J.; Dunsing, V.; Herrmann, A.; Chiantia, S. Phosphatidylserine Lateral Organization Influences the Interaction of Influenza Virus Matrix Protein 1 with Lipid Membranes. J. Virol. 2017, 91, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Phase Transition Temperatures for Glycerophospholipids. Available online: https://avantilipids.com/tech-support/physical-properties/phase-transition-temps (accessed on 27 March 2021).
- Huster, D.; Arnold, K.; Gawrisch, K. Influence of docosahexaenoic acid and cholesterol on lateral lipid organization in phospholipid mixtures. Biochemistry 1998, 37, 17299–17308. [Google Scholar] [CrossRef]
- Reigada, R. Electroporation of heterogeneous lipid membranes. Biochim. Biophys. Acta Biomembr. 2014, 1838, 814–821. [Google Scholar] [CrossRef]
- López Martí, J.M.; English, N.J.; Del Pópolo, M.G. Elucidating mysteries of phase-segregated membranes: Mobile-lipid recruitment facilitates pores’ passage to the fluid phase. Phys. Chem. Chem. Phys. 2018, 20, 19234–19239. [Google Scholar] [CrossRef]
- Kirsch, S.A.; Böckmann, R.A. Coupling of Membrane Nanodomain Formation and Enhanced Electroporation near Phase Transition. Biophys. J. 2019, 116, 2131–2148. [Google Scholar] [CrossRef]
- Cordeiro, R.M. Molecular Structure and Permeability at the Interface between Phase-Separated Membrane Domains. J. Phys. Chem. B 2018, 122, 6954–6965. [Google Scholar] [CrossRef]
- Engberg, O.; Yasuda, T.; Hautala, V.; Matsumori, N.; Nyholm, T.K.; Murata, M.; Slotte, J.P. Lipid Interactions and Organization in Complex Bilayer Membranes. Biophys. J. 2016, 110, 1563–1573. [Google Scholar] [CrossRef]
- Lebar, A.M.; Velikonja, A.; Kramar, P.; Iglič, A. Internal configuration and electric potential in planar negatively charged lipid head group region in contact with ionic solution. Bioelectrochemistry 2016, 111, 49–56. [Google Scholar] [CrossRef]
- Iglič, A.; Gongadze, E.; Kralj-Iglič, V. Differential capacitance of electric double layer – Influence of asymmetric size of ions, thickness of stern layer and orientational ordering of water dipoles. Acta Chim. Slov. 2019, 66, 534–541. [Google Scholar] [CrossRef]
- Drab, M.; Gongadze, E.; Kralj-Iglič, V.; Iglič, A. Electric double layer and orientational ordering of water dipoles in narrow channels within a modified Langevin Poisson-Boltzmann model. Entropy 2020, 22, 1054. [Google Scholar] [CrossRef]
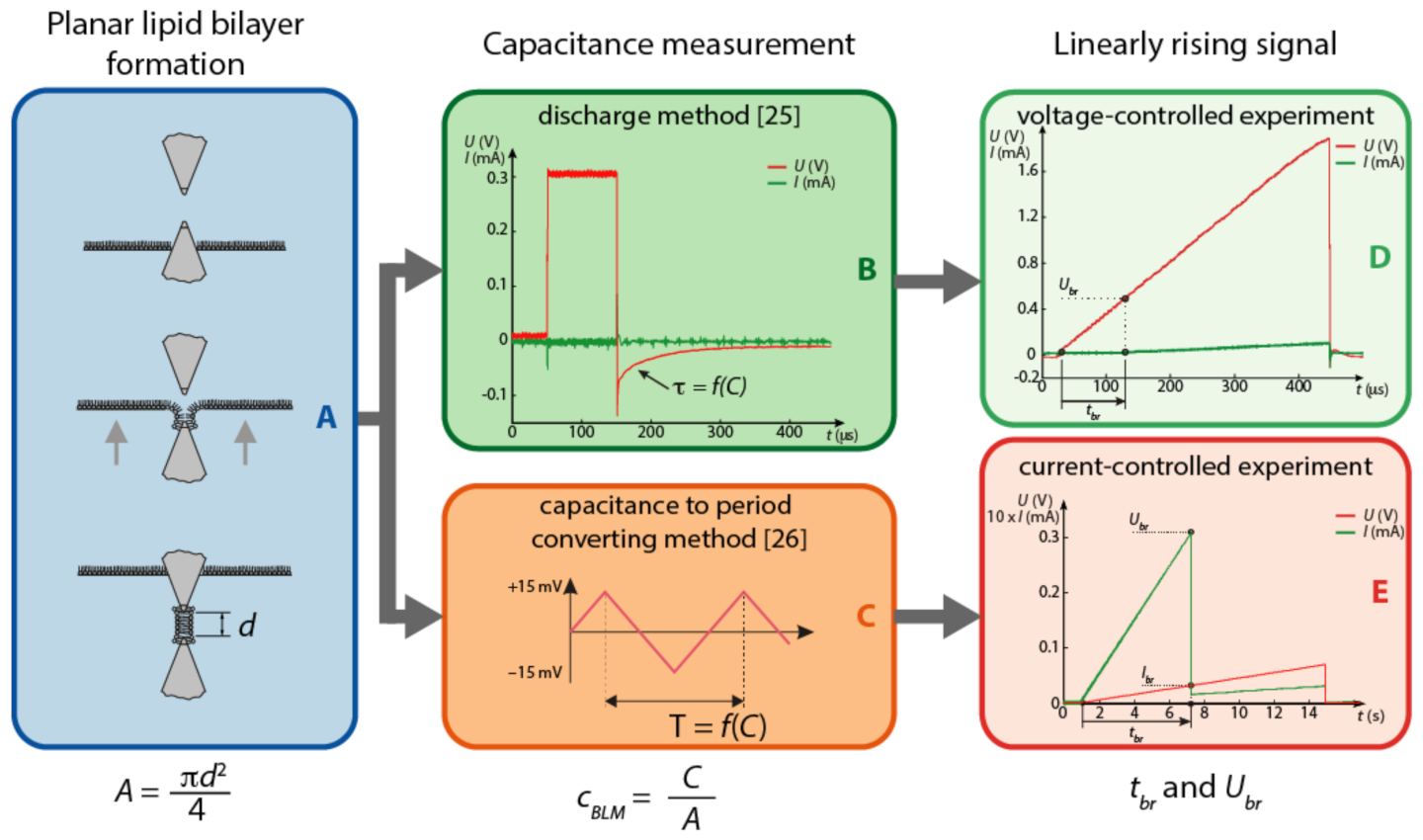
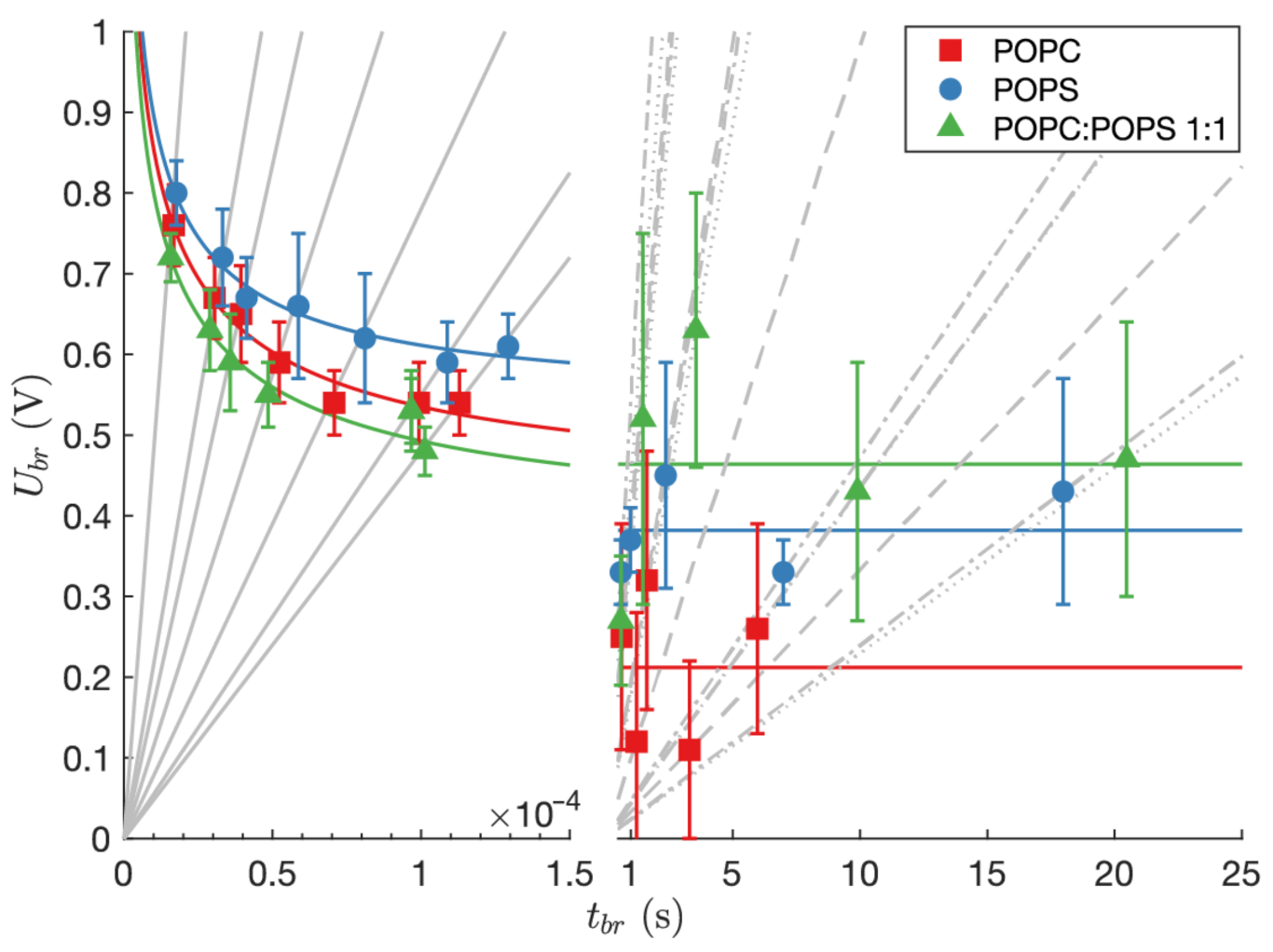
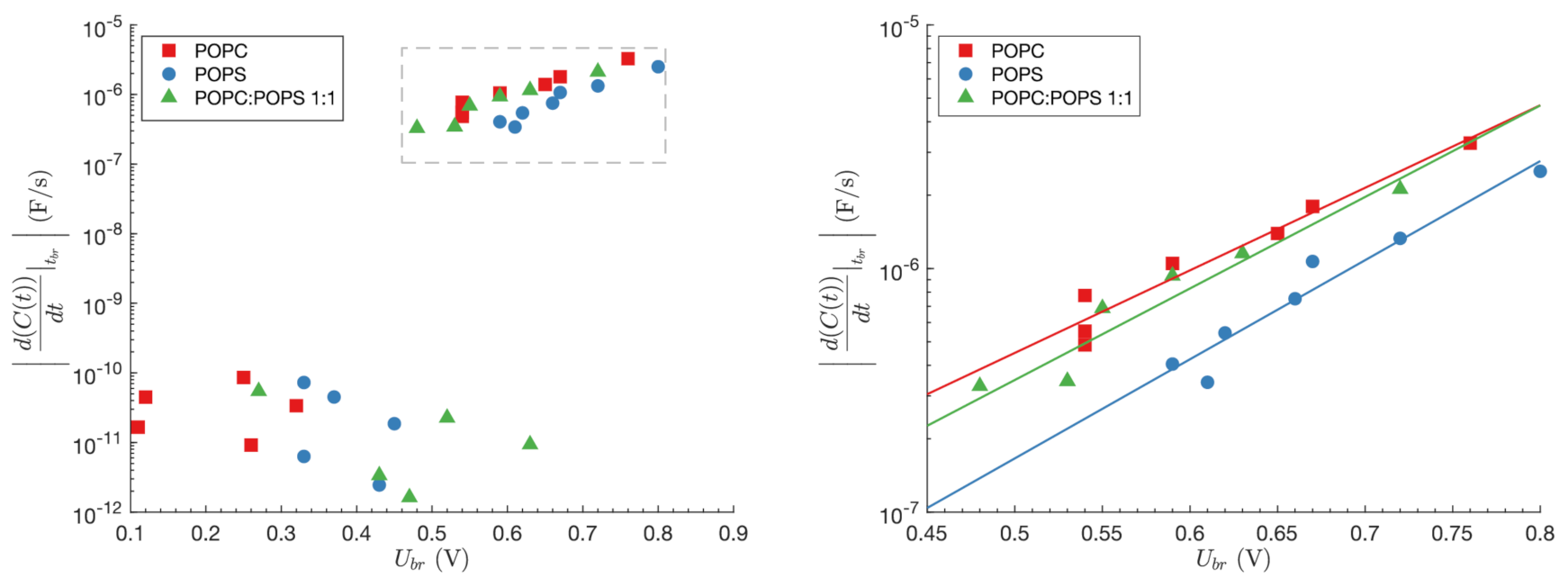

| Lipid Mixtures | Discharge Method [25] [F/cm ] | Capacitance to Period Converting Method [26] [F/cm ] |
|---|---|---|
| POPC | 0.51 ± 0.17 (80) | 0.51 ± 0.16 (58) * |
| POPS | 0.41 ± 0.14 (76) | 0.41 ± 0.13 (34) * |
| POPC:POPS 1:1 | 0.31 ± 0.07 (60) | 0.34 ± 0.17 (25) * |
| POPC | |||||
|---|---|---|---|---|---|
| (kV/s) | (V) | (s) | (%) | ||
| voltage-cont. | 48.1 | 16 | 0.76 ± 0.05 | 16.76 ± 1.14 | 1.72 |
| 21.6 | 17 | 0.67 ± 0.05 | 30.50 ± 2.60 | 0.95 | |
| 16.7 | 12 | 0.65 ± 0.06 | 39.38 ± 3.87 | 0.73 | |
| 11.5 | 12 | 0.59 ± 0.05 | 52.25 ± 4.65 | 0.55 | |
| 7.8 | 16 | 0.54 ± 0.04 | 70.74 ± 5.07 | 0.41 | |
| 5.5 | 16 | 0.54 ± 0.05 | 99.24 ± 9.93 | 0.29 | |
| 4.8 | 17 | 0.54 ± 0.04 | 112.92 ± 8.32 | 0.27 | |
| (A/s) | (V) | (s) | (%) | ||
| current-cont. | 10 | 8 | 0.25 ± 0.14 | 0.64 ± 0.55 | 45.20 |
| 8 | 9 | 0.12 ± 0.16 | 1.22 ± 1.16 | 23.63 | |
| 4 | 7 | 0.32 ± 0.16 | 1.63 ± 1.35 | 17.76 | |
| 1 | 11 | 0.11 ± 0.11 | 3.30 ± 0.98 | 8.74 | |
| 0.5 | 16 | 0.26 ± 0.13 | 5.97 ± 2.09 | 4.83 | |
| POPS | |||||
|---|---|---|---|---|---|
| (kV/s) | (V) | (s) | (%) | ||
| voltage-cont. | 48.1 | 18 | 0.80 ± 0.04 | 17.59 ± 0.86 | 0.65 |
| 21.6 | 14 | 0.72 ± 0.06 | 33.15 ± 2.76 | 0.35 | |
| 16.7 | 13 | 0.67 ± 0.05 | 41.24 ± 3.34 | 0.28 | |
| 11.5 | 12 | 0.66 ± 0.09 | 58.66 ± 7.71 | 0.20 | |
| 7.8 | 15 | 0.62 ± 0.08 | 81.05 ± 10.92 | 0.14 | |
| 5.5 | 13 | 0.59 ± 0.05 | 108.78 ± 8.72 | 0.11 | |
| 4.8 | 15 | 0.61 ± 0.04 | 129.24 ± 7.86 | 0.09 | |
| (A/s) | (V) | (s) | (%) | ||
| current-cont. | 10 | 5 | 0.33 ± 0.04 | 0.61 ± 0.26 | 18.91 |
| 8 | 5 | 0.37 ± 0.04 | 0.98 ± 0.08 | 11.72 | |
| 4 | 6 | 0.45 ± 0.14 | 2.37 ± 0.71 | 4.84 | |
| 1 | 5 | 0.33 ± 0.04 | 6.99 ± 0.91 | 1.64 | |
| 0.5 | 6 | 0.43 ± 0.14 | 17.98 ± 5.85 | 0.64 | |
| POPC:POPS 1:1 | |||||
|---|---|---|---|---|---|
| (kV/s) | (V) | (s) | (%) | ||
| voltage-cont. | 48.1 | 7 | 0.72 ± 0.03 | 15.73 ± 0.73 | 1.49 |
| 21.6 | 7 | 0.63 ± 0.05 | 28.90 ± 2.30 | 0.81 | |
| 16.7 | 6 | 0.59 ± 0.06 | 35.73 ± 3.70 | 0.66 | |
| 11.5 | 7 | 0.55 ± 0.04 | 48.49 ± 3.55 | 0.48 | |
| 7.8 | 11 | 0.53 ± 0.04 | 96.66 ± 4.60 | 0.24 | |
| 5.5 | 9 | 0.53 ± 0.05 | 96.68 ± 9.12 | 0.24 | |
| 4.8 | 13 | 0.48 ± 0.03 | 101.24 ± 7.39 | 0.23 | |
| (A/s) | (V) | (s) | (%) | ||
| current-cont. | 10 | 3 | 0.27 ± 0.08 | 0.61 ± 0.19 | 15.92 |
| 8 | 5 | 0.52 ± 0.23 | 1.47 ± 0.51 | 38.47 | |
| 4 | 5 | 0.63 ± 0.17 | 3.56 ± 0.94 | 6.59 | |
| 1 | 6 | 0.43 ± 0.16 | 9.90 ± 3.70 | 2.37 | |
| 0.5 | 5 | 0.47 ± 0.17 | 20.47 ± 8.92 | 1.15 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maček Lebar, A.; Miklavčič, D.; Kotulska, M.; Kramar, P. Water Pores in Planar Lipid Bilayers at Fast and Slow Rise of Transmembrane Voltage. Membranes 2021, 11, 263. https://doi.org/10.3390/membranes11040263
Maček Lebar A, Miklavčič D, Kotulska M, Kramar P. Water Pores in Planar Lipid Bilayers at Fast and Slow Rise of Transmembrane Voltage. Membranes. 2021; 11(4):263. https://doi.org/10.3390/membranes11040263
Chicago/Turabian StyleMaček Lebar, Alenka, Damijan Miklavčič, Malgorzata Kotulska, and Peter Kramar. 2021. "Water Pores in Planar Lipid Bilayers at Fast and Slow Rise of Transmembrane Voltage" Membranes 11, no. 4: 263. https://doi.org/10.3390/membranes11040263
APA StyleMaček Lebar, A., Miklavčič, D., Kotulska, M., & Kramar, P. (2021). Water Pores in Planar Lipid Bilayers at Fast and Slow Rise of Transmembrane Voltage. Membranes, 11(4), 263. https://doi.org/10.3390/membranes11040263







