Neutralization of Industrial Water by Electrodialysis
Abstract
1. Introduction
- ✓
- the formation of a large amount of sediments as a result of changes in acidity;
- ✓
- disruption of dosing mechanism operation due to clogging of holes;
- ✓
- the need to prepare reagent solutions of a certain concentration;
- ✓
- inaccuracy in predicting and obtaining acidity values.
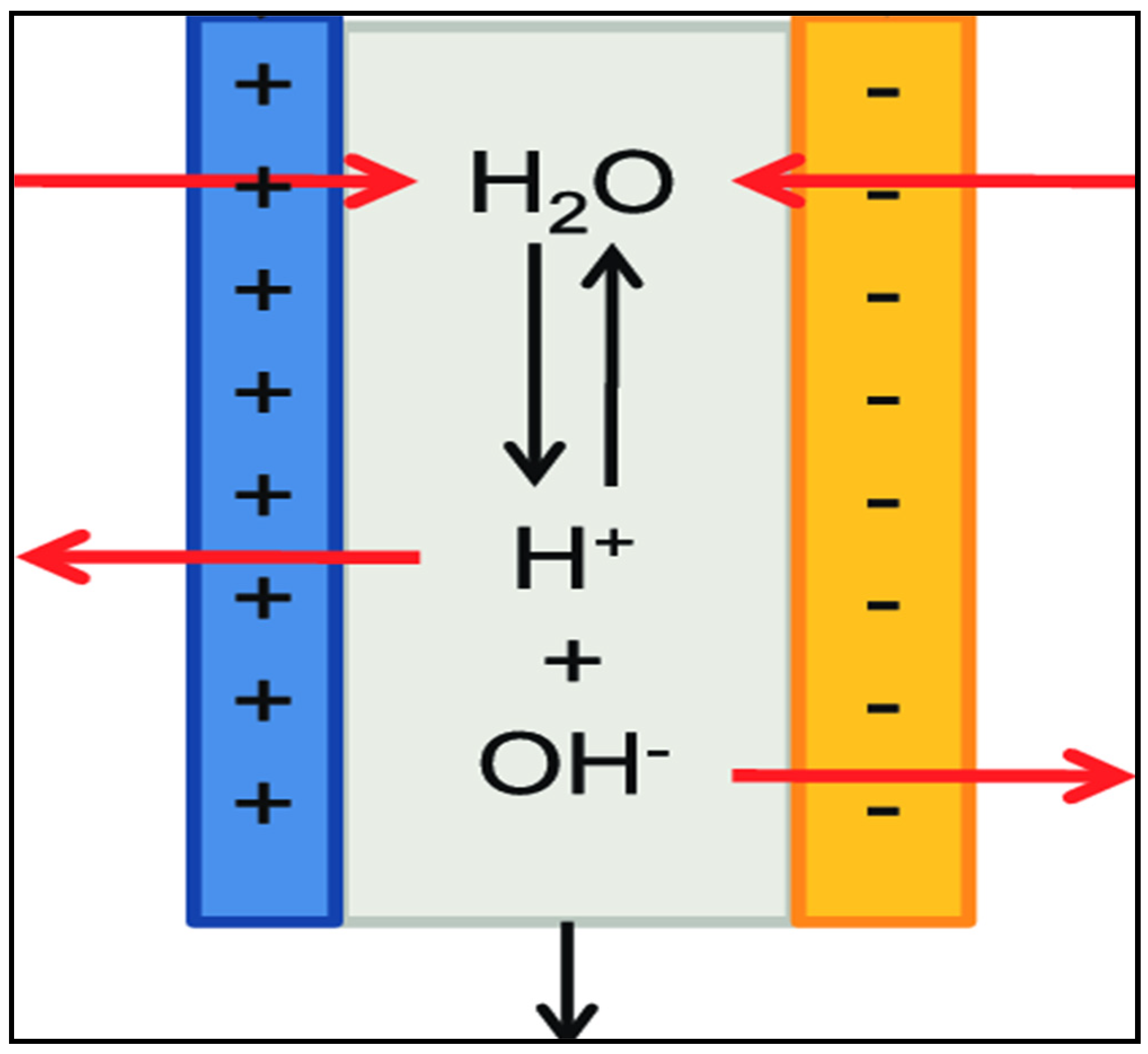
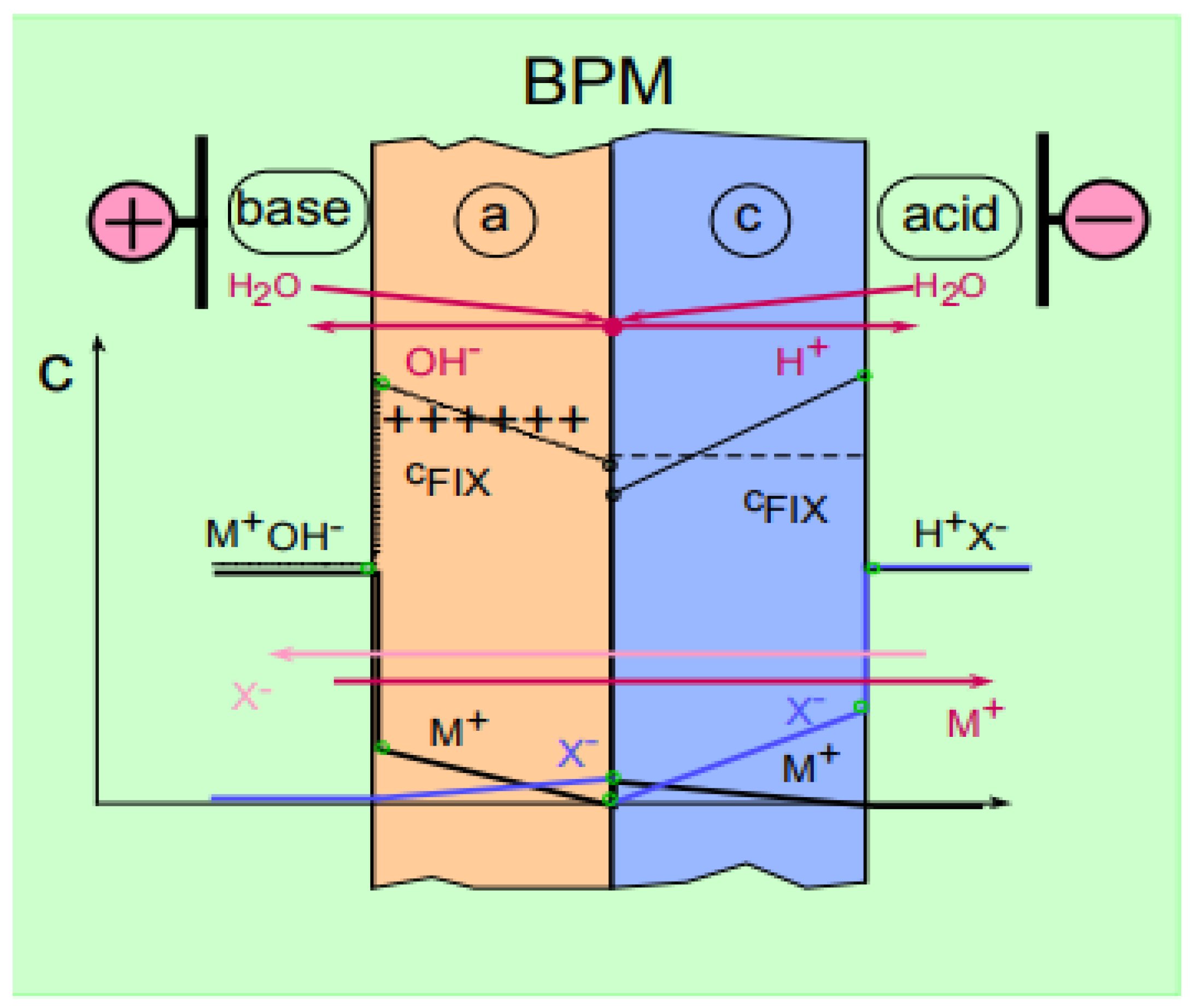
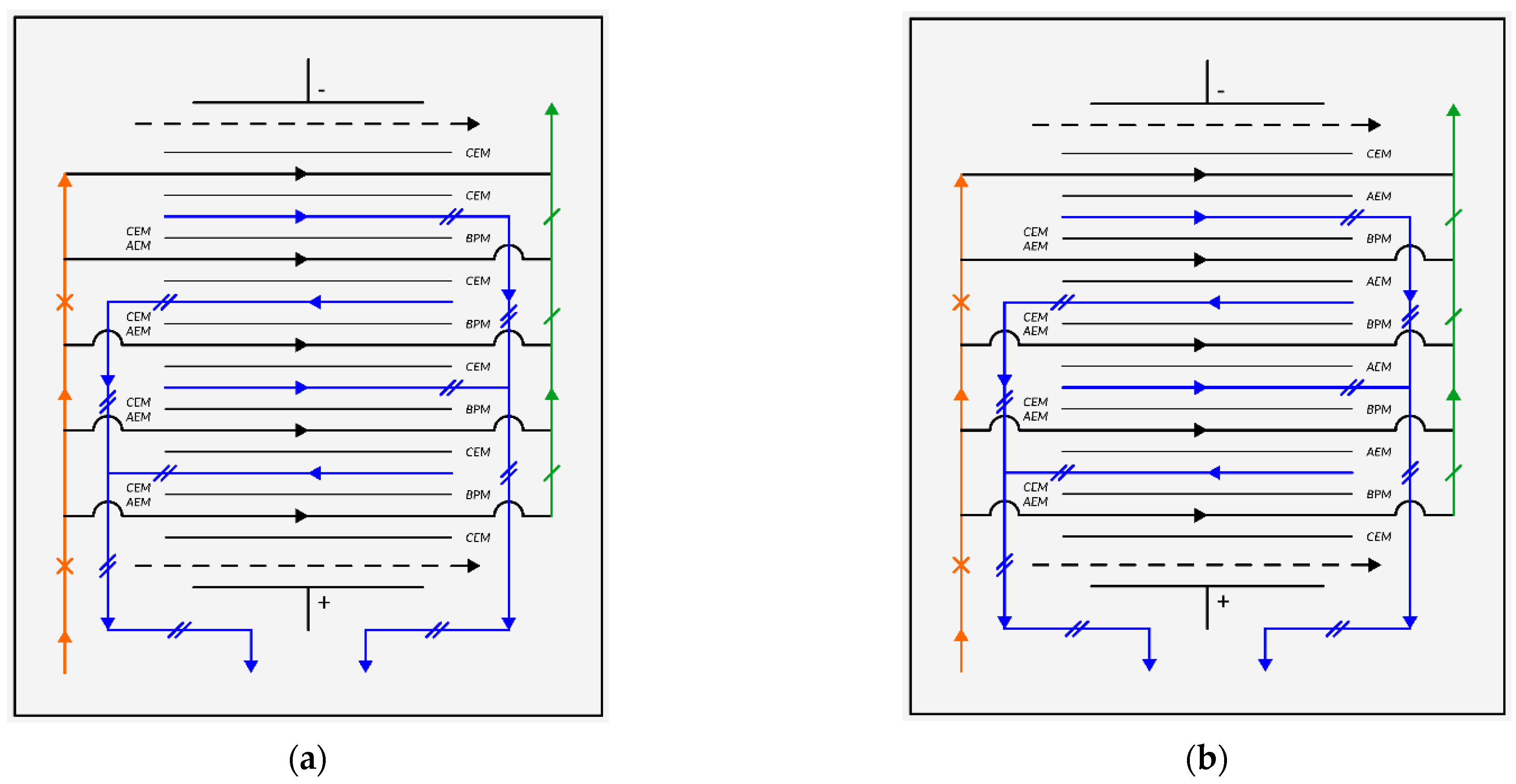
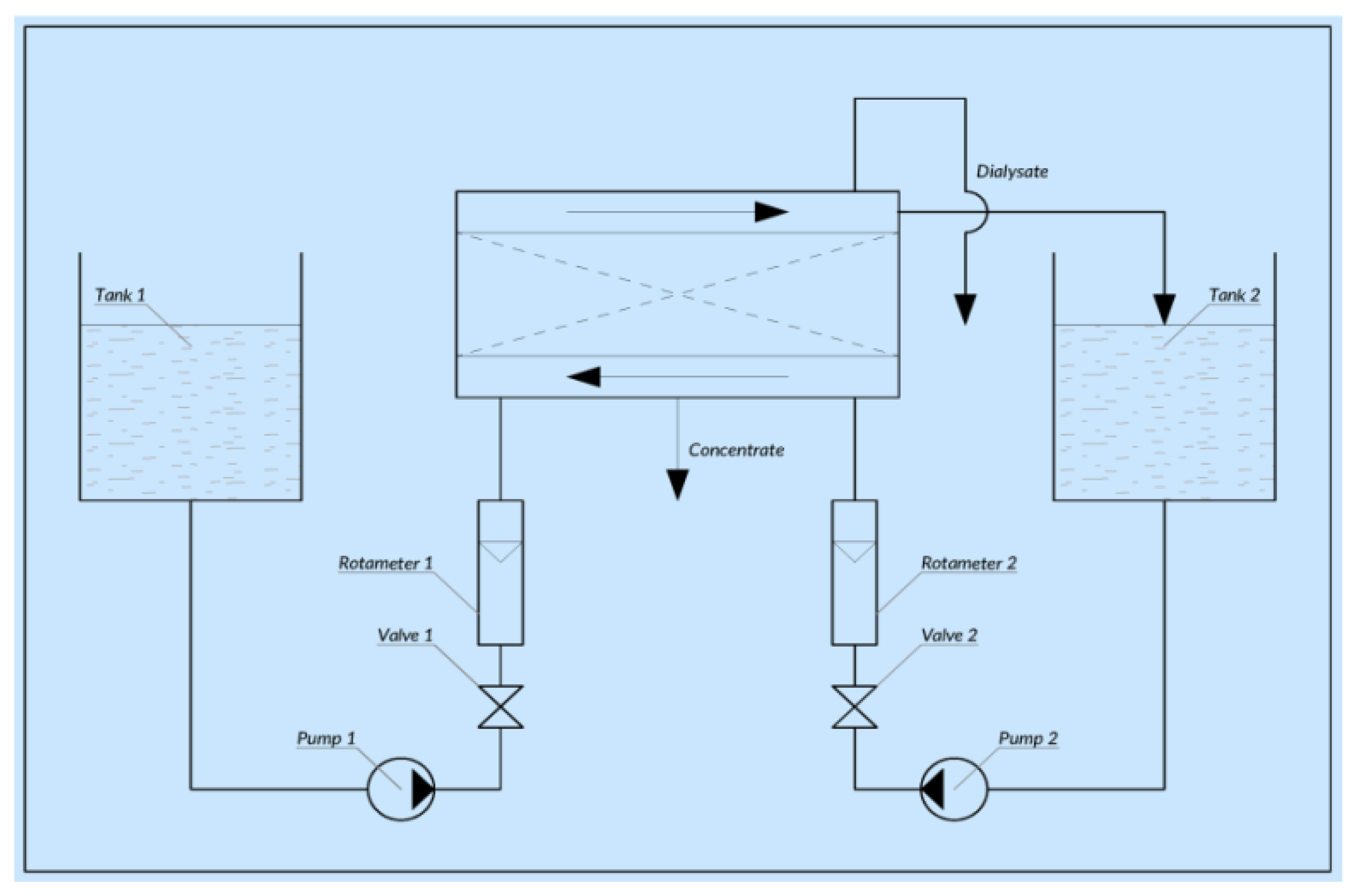
2. Methods and Materials
- Process performance:
- ✓
- V (product)—product volume, l;
- ✓
- τ—time, h;
- ✓
- S—membrane area, m2
- 2.
- Energy consumption:
- ✓
- I—amperage, kW;
- ✓
- τ—time, h;
- ✓
- V—product volume, m3;
- ✓
- U—voltage across the electrodialyzer
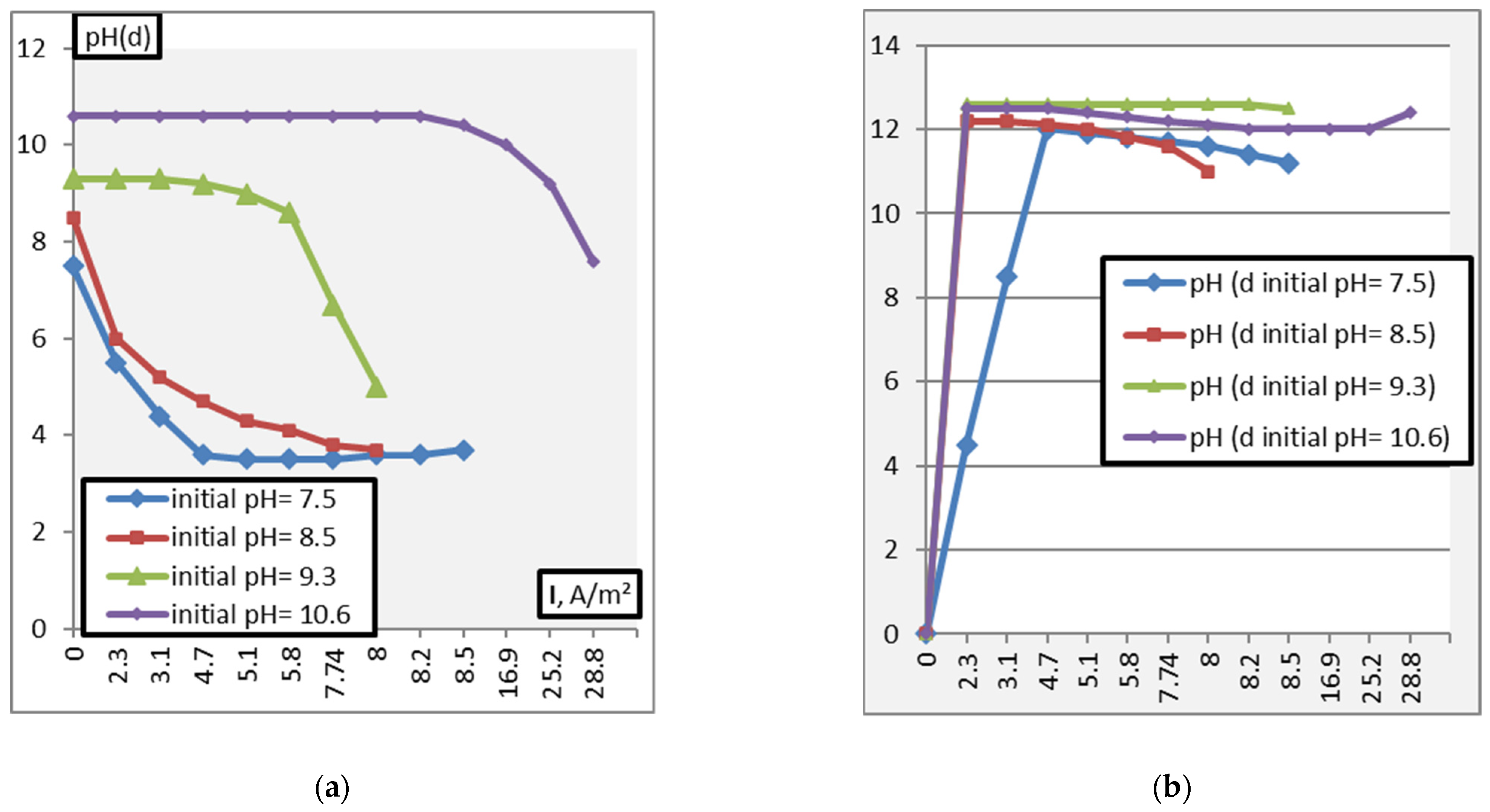
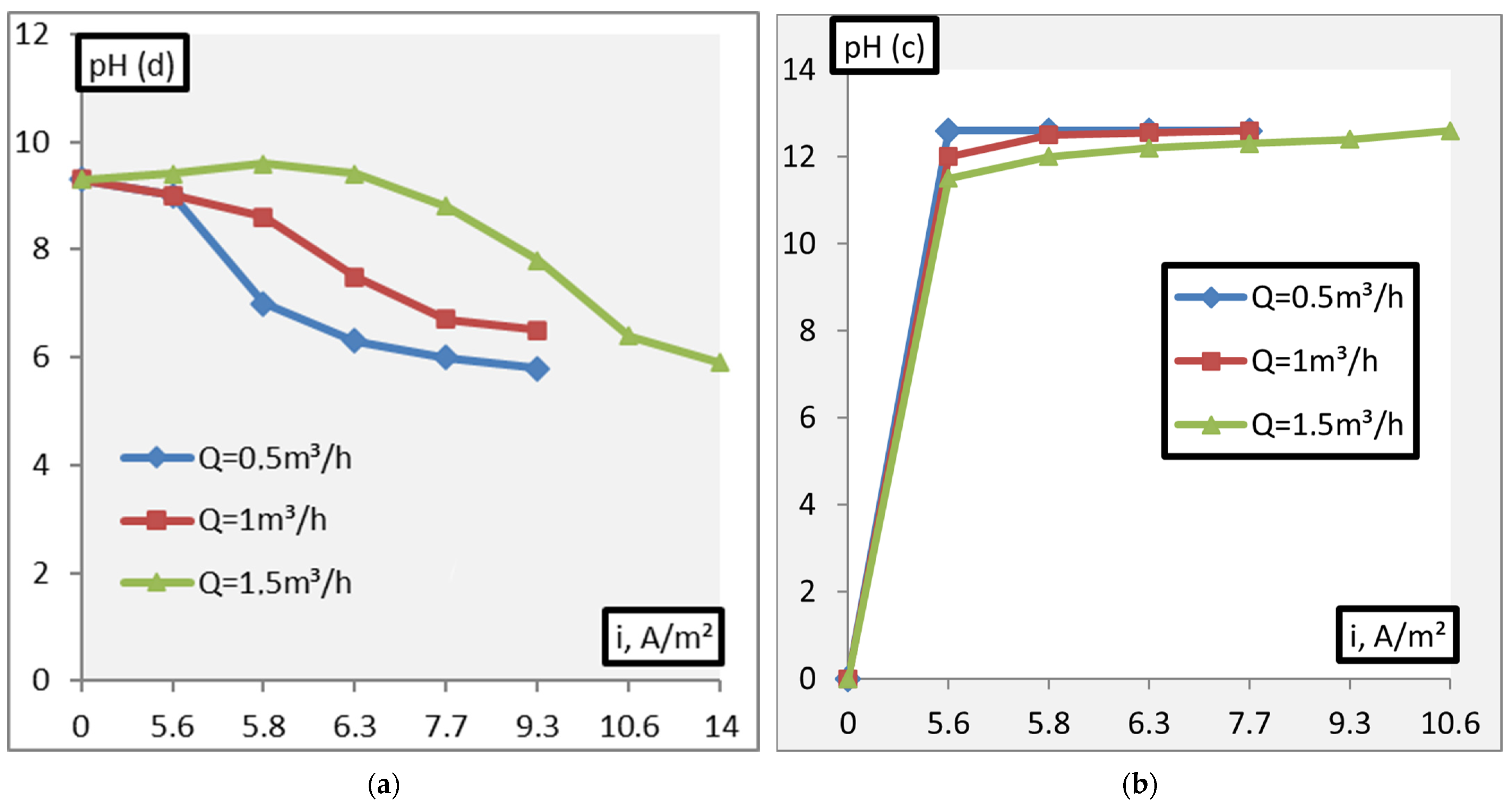
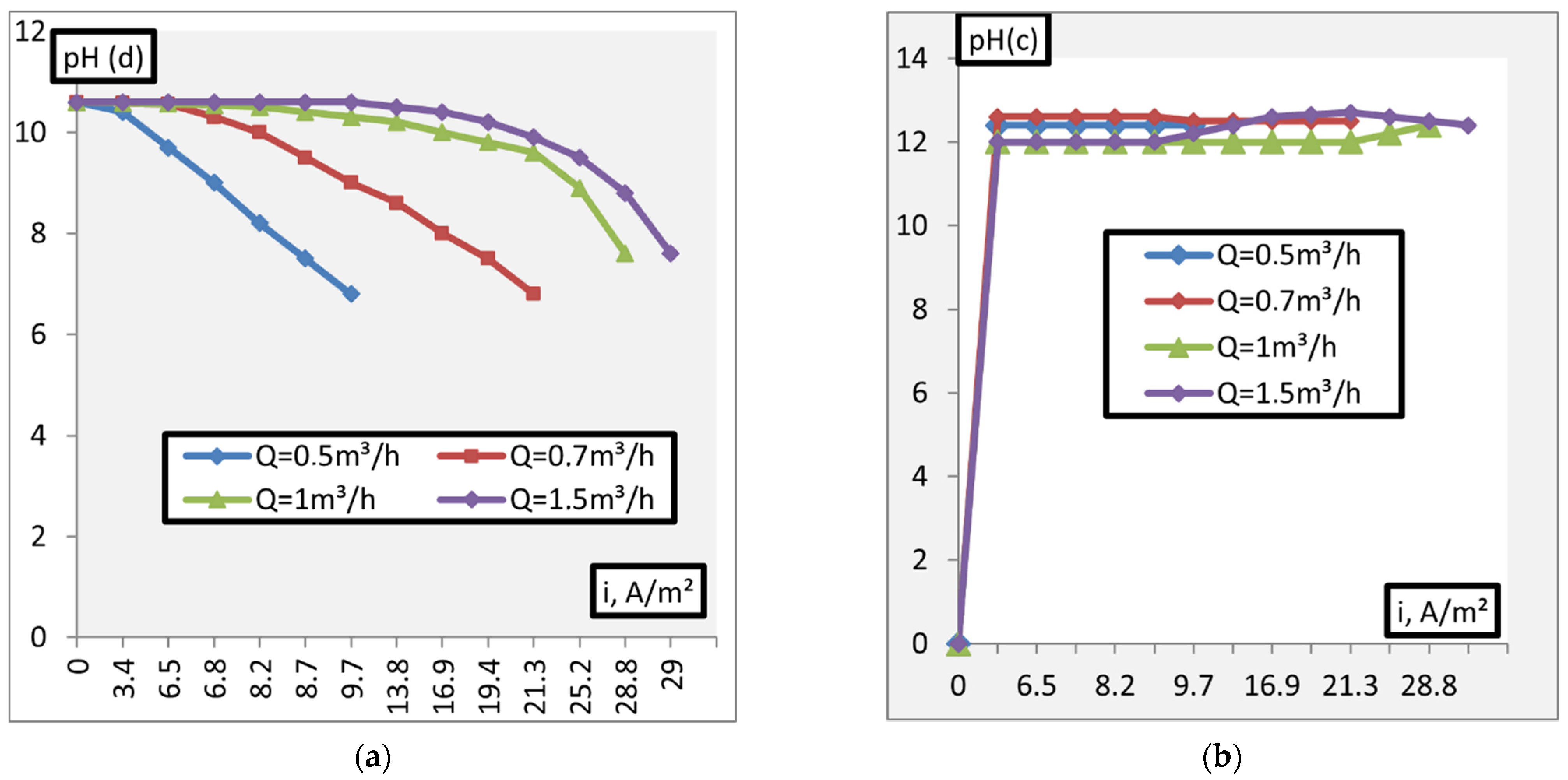
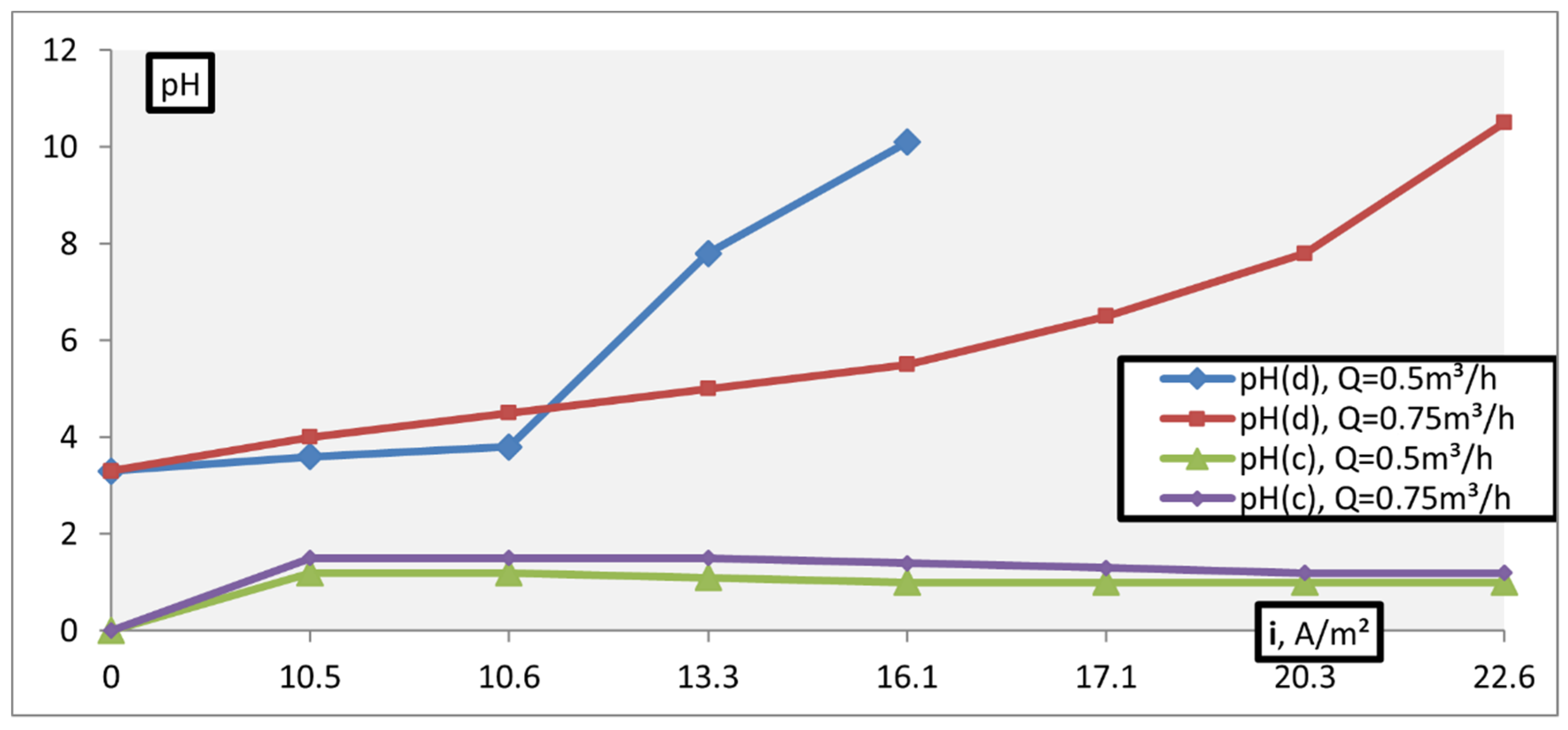
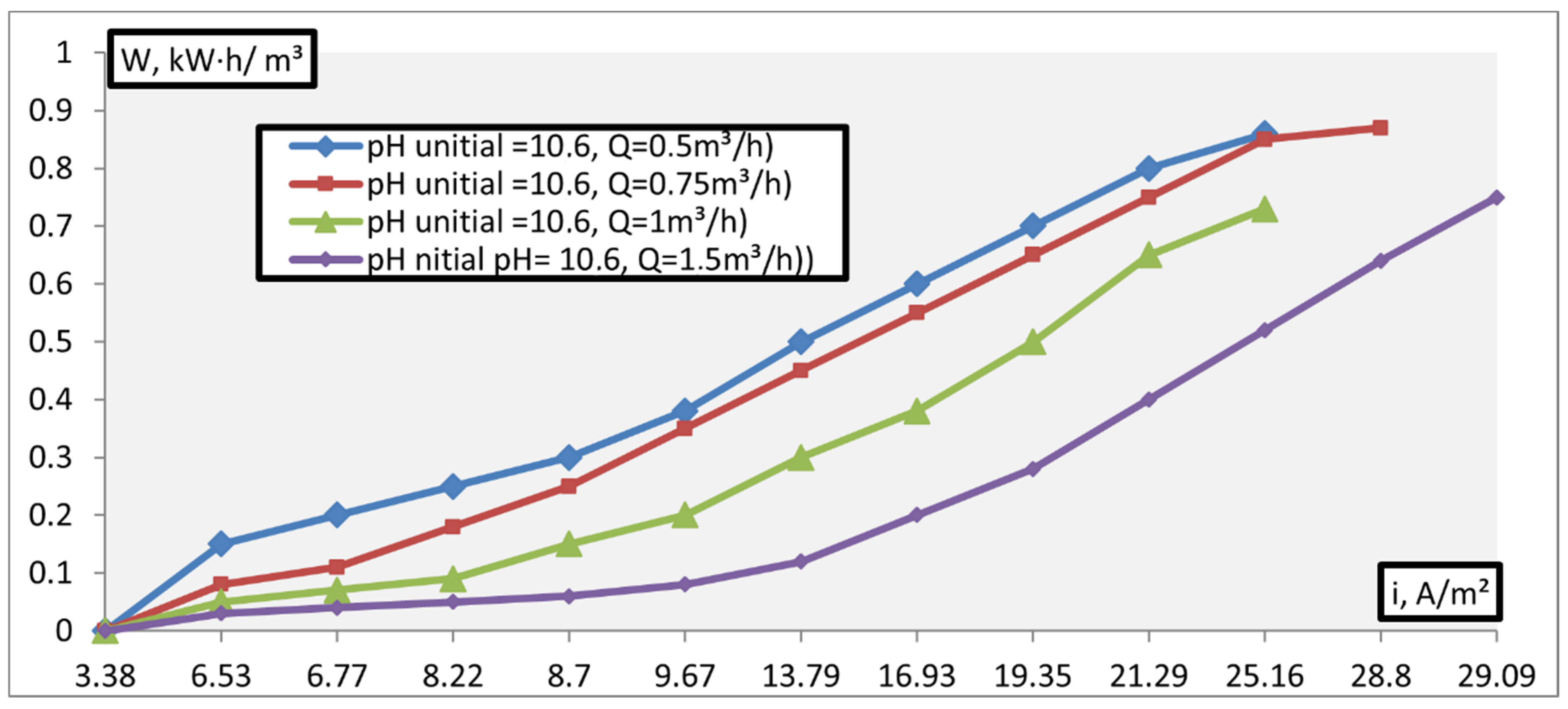
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Frenkel, V. Membrane technologies: Past, present and future (on the example of North America). Water Supply Sanit. Eng. 2010, 8, 48–54. [Google Scholar]
- Lobovko, A. Ways of solving the problem of regulation of acidity of technological liquids. Young Sci. 2018, 44, 19–22. [Google Scholar]
- Laptev, A.; Sergeeva, E. Water treatment and water purification in the energy sector. Part 1. Water: Chemistry and Ecology 2011, 3, 33–40. [Google Scholar]
- Bejanidze, I.; Pohrebennyk, V.; Kharebava, T.; Koncelidze, L.; Jun, C. Correction of the chemical composition of the washing waters received as a result of pH cation exchange of ionex change resin. In Proceedings of the International International Multidisciplinary Scientific GeoConference SGEM, Albena, Bulgaria, 30 June–6 July 2019; pp. 133–140. [Google Scholar]
- Zabolotskiy, V.I.; Utin, S.V.; Sheldeshov, N.V.; Lebedev, K.A.; Vasilenko, P.A. Reseach of the process of pH correction of diluted electrolyte solutions by electrodialysis with bipolar membranes. Electochemystry 2011, 47, 343–348. [Google Scholar]
- Bejanidze, I.; Petrov, O.; Pohrebennyk, V.; Kharebava, T.; Nakashidze, N.; Davitadze, N.D.; Petrov, A. Sorption of organic electrolytes and surfactants from natural waters by heterogeneous membranes. Appl. Sci. 2020, 10, 7383. [Google Scholar] [CrossRef]
- Strathmann, H. Ion-Exchange Membrane Separation Processes, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2004; ISBN 044450236X. [Google Scholar]
- Petrov, O.; Petrichenko, S.; Yushchishina, A.; Mitryasova, O.; Pohrebennyk, V. Electrospark method in galvanic wastewater treatment for heavy metal removal. Appl. Sci. 2020, 10, 5148. [Google Scholar]
- Strathmann, H. Electrodialysis, a mature technology with a multitude of new applications. Desalination 2010, 264, 268–288. [Google Scholar] [CrossRef]
- Przystupa, K.; Petrichenko, S.; Yushchishina, A.; Mitryasova, O.; Pohrebennyk, V.; Kochan, O. Electric spark method of purification of galvanic waste waters. Przegląd Elektrotechniczny 2020, 12, 230–233. [Google Scholar]
- Sajjad, A.-A.; Yunus, M.Y.B.M.; Azoddein, A.A.M.; Hassell, D.G.; Dakhil, I.H.; Hasan, H.A. Electrodialysis Desalination for Water and Wastewater: A Review. Chem. Eng. J. 2020, 380, 122231. [Google Scholar]
- Bejanidze, I. Save natural-save your life. Ga. Natl. Sci. Acad. Bull. 2010, 36, 76–84. [Google Scholar]
- Ezugbe, E.O.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef]
- Gurreri, L.; Tamburini, A.; Cipollina, A.; Micale, G. Electrodialysis Applications in Wastewater Treatment for Environmental Protection and Resources Recovery: A Systematic Review on Progress and Perspectives. Membranes 2020, 10, 146. [Google Scholar] [CrossRef]
- Niftaliev, S.; Kozaderova, O.; Kim, K.; Velho, F. The use of electrodialysis to obtain acid and alkali from a concentrated solution of sodium sulfate. Bull. VGUIT 2014, 4, 175–178. (In Russian) [Google Scholar]
- Zhao, W.Y.; Zhou, M.; Yan, B.; Sun, X.; Liu, Y.; Wang, Y.; Xu, T.; Zhang, Y. Waste conversion and resource recovery from wastewater by ion exchange membranes: State-of-the-art and perspective. Ind. Eng. Chem. Res. 2018, 57, 6025–6039. [Google Scholar] [CrossRef]
- Xu, T.; Huang, C. Electrodialysis-Based separation technologies: A critical review. AiCHE J. 2008, 54, 3147–3159. [Google Scholar] [CrossRef]
- Xu, T. Ion exchange membranes: State of their development and perspective. J. Membr. Sci. 2005, 263, 1–29. [Google Scholar] [CrossRef]
- Nagarale, R.K.; Gohil, G.S.; Shahi, V.K. Recent developments on ion-exchange membranes and electro-membrane processes. Adv. Colloid Interface Sci. 2006, 119, 97–130. [Google Scholar] [CrossRef]
- Basile, A.; Cassano, A.; Rastogi, N. Advances in Membrane Technologies for Water Treatment. Materials, Processes and Applications; Woodhead Publishing: Cambridge, UK, 2015; 666p, ISBN 9781782421214. [Google Scholar]
- Tanaka, Y. Ion Exchange Membranes: Fundamentals and Applications; Elsevier: Amsterdam, The Netherlands, 2007; Volume 12, ISBN 0927-5193. [Google Scholar]
- Ran, J.; Wu, L.; He, Y.; Yang, Z.; Wang, Y.; Jiang, C.; Ge, L.; Bakangura, E.; Xu, T. Ion exchange membranes: New developments and applications. J. Membr. Sci. 2017, 522, 267–291. [Google Scholar] [CrossRef]
- Pal, P. Membrane-Based Technologies for Environmental Pollution Control; Butterworth-Heinemann: Oxford, UK, 2020; 784p, ISBN 9780128194553. [Google Scholar]
- Nikonenko, V.; Pismenskaya, N.; Belova, E.; Sistat, P.; Huguet, P.; Pourcelly, G.; Larchet, I. Intensive current transfer in membrane systems: Modelling, mechanisms and application in electrodialysis. Adv. Colloid Interface Sci. 2010, 160, 101–123. [Google Scholar] [CrossRef]
- Bejanidze, I.; Kharebava, T.; Davitadze, N.; Kontselidze, Z.; Kontselidze, L. Purification of Natural and Wastewater by Electromembrane Technology: Monograph; Series Membrane Technology; Shota Rustaveli State University: Batumi, Georgia, 2019; p. 178. (In Georgian) [Google Scholar]
- Campione, A.; Gurreri, L.; Ciofalo, M.; Micale, G.; Tamburini, A.; Cipollina, A. Electrodialysis for water desalination: A critical assessment of recent developments on process fundamentals, models and applications. Desalination 2018, 434, 121–160. [Google Scholar] [CrossRef]
- Bobylev, V.P.; Matukhno, Y.V.; Turishchev, V.V.; Belokon, K.V. Methodical approach for selection of design parameters of electrodialisys diaphragmless apparatus for regeneration of electrolytecontaining solution. Metall. Min. Ind. 2013, 2, 77. [Google Scholar]
- Singh, R.; Hankins, N. Emerging Membrane Technology for Sustainable Water Treatment; Elsevier Science: Amsterdam, The Netherlands, 2016; 480p, ISBN 9780444633125. [Google Scholar]
- Bernardes, A.M.; Ferreira, J.Z.; Rodrigues, M.S. Electrodialysis and Water Reuse: Novel Approaches; Springer: Berlin/Heidelberg, Germany, 2014; ISBN 9783642402494. [Google Scholar]
- Bejanidze, I.; Kharebava, T.; Pohrebennyk, V.; Didmanidze, N.; Davitadze, N. The study of composition and physical and chemical properties of some natural waters of Georgia. In Water Supply and Wastewater Disposal. Designing, Construction, Operation and Monitoring: Monograph; Politechnika Lubelska: Lublin, Poland, 2020; pp. 8–23. ISBN 978837947409. [Google Scholar]
- Bejanidze, I.; Petrov, O.; Kharebava, T.; Pohrebennyk, V.; Davitadze, N.; Didmanidze, N. Study of the healing properties of natural sources of Georgia and modeling of their purification processes. Appl. Sci. 2020, 10, 6529. [Google Scholar] [CrossRef]
- Merkel, A.; Ashrafi, M.; Ecer, J. Bipolar membrane electrodialysis assisted pH correction of milk whey. J. Membr. Sci. 2018, 555, 185–196. [Google Scholar] [CrossRef]
- Scarazzato, T.; Panossian, Z.; Tenório, J.A.S.; Pérez-Herranz, V.; Espinosa, D.C.R. A review of cleaner production in electroplating industries using electrodialysis. J. Clean. Prod. 2017, 168, 1590–1602. [Google Scholar] [CrossRef]
- Pawlowski, S.; Crespo, J.; Velizarov, S. Profiled Ion Exchange Membranes: A Comprehensible Review. Int. J. Mol. Sci. 2019, 20, 165. [Google Scholar] [CrossRef]
- Mareev, S.A.; Evdochenko, E.; Wessling, M.; Kozaderova, O.A.; Niftaliev, S.I.; Pismenskaya, N.D.; Nikonenko, V.V. A comprehensive mathematical model of water splitting in bipolar membranes: Impact of the spatial distribution of fixed charges and catalyst at bipolar junction. J. Membr. Sci. 2020, 603, 118010. [Google Scholar] [CrossRef]
- Bejanidze, I.; Kharebava, T.; Koncelidze, Z. The use of bipolar electrodialysis for processing juice production waste. Sci. Rev. 2018, 2, 35–38. [Google Scholar]
- Nikonenko, V.; Urtenov, M.; Mareev, S. Pourcelly Mathematical Modeling of the Effect of Water Splitting on Ion Transfer in the Depleted Diffusion Layer Near an Ion-Exchange Membrane. Membranes 2020, 10, 22. [Google Scholar] [CrossRef]
- Donskikh, A.N. A Method for Regulating the Acidity of Whey in the Process of Electrodialysis. Russian Patent No. 2515096, 5 October 2014. Bul. No. 13. [Google Scholar]
- Kamcev, J.; Sujanani, R.; Jang, E.S.; Yan, N.; Moe, N.; Paul, D.R.; Freeman, B.D. Salt concentration dependence of ionic conductivity in ion exchange membranes. J. Membr. Sci. 2018, 547, 123–133. [Google Scholar] [CrossRef]
- Veerman, J. The Effect of the NaCl Bulk Concentration on the Resistance of Ion Exchange Membranes—Measuring and Modeling. Energies 2020, 13, 1946. [Google Scholar] [CrossRef]
- Kamcev, J.; Doherty, C.M.; Lopez, K.P.; Hill, A.J.; Paul, D.R.; Freeman, B.D. Effect of fixed charge group concentration on salt permeability and diffusion coefficients in ion exchange membranes. J. Membr. Sci. 2018, 566, 307–316. [Google Scholar] [CrossRef]
- La Cerva, M.; Gurreri, L.; Tedesco, M.; Cipollina, A.; Ciofalo, M.; Tamburini, A.; Micale, G. Determination of limiting current density and current efficiency in electrodialysis units. Desalination 2018, 445, 138–148. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrov, O.; Iwaszczuk, N.; Kharebava, T.; Bejanidze, I.; Pohrebennyk, V.; Nakashidze, N.; Petrov, A. Neutralization of Industrial Water by Electrodialysis. Membranes 2021, 11, 101. https://doi.org/10.3390/membranes11020101
Petrov O, Iwaszczuk N, Kharebava T, Bejanidze I, Pohrebennyk V, Nakashidze N, Petrov A. Neutralization of Industrial Water by Electrodialysis. Membranes. 2021; 11(2):101. https://doi.org/10.3390/membranes11020101
Chicago/Turabian StylePetrov, Oleksandr, Natalia Iwaszczuk, Tina Kharebava, Irina Bejanidze, Volodymyr Pohrebennyk, Nunu Nakashidze, and Anton Petrov. 2021. "Neutralization of Industrial Water by Electrodialysis" Membranes 11, no. 2: 101. https://doi.org/10.3390/membranes11020101
APA StylePetrov, O., Iwaszczuk, N., Kharebava, T., Bejanidze, I., Pohrebennyk, V., Nakashidze, N., & Petrov, A. (2021). Neutralization of Industrial Water by Electrodialysis. Membranes, 11(2), 101. https://doi.org/10.3390/membranes11020101






