Modeling and Simulation of Either Co-Current or Countercurrent Operated Reverse-Osmosis-Based Air Water Generator
Abstract
:1. Introduction
2. Materials and Methods
2.1. Absorbents
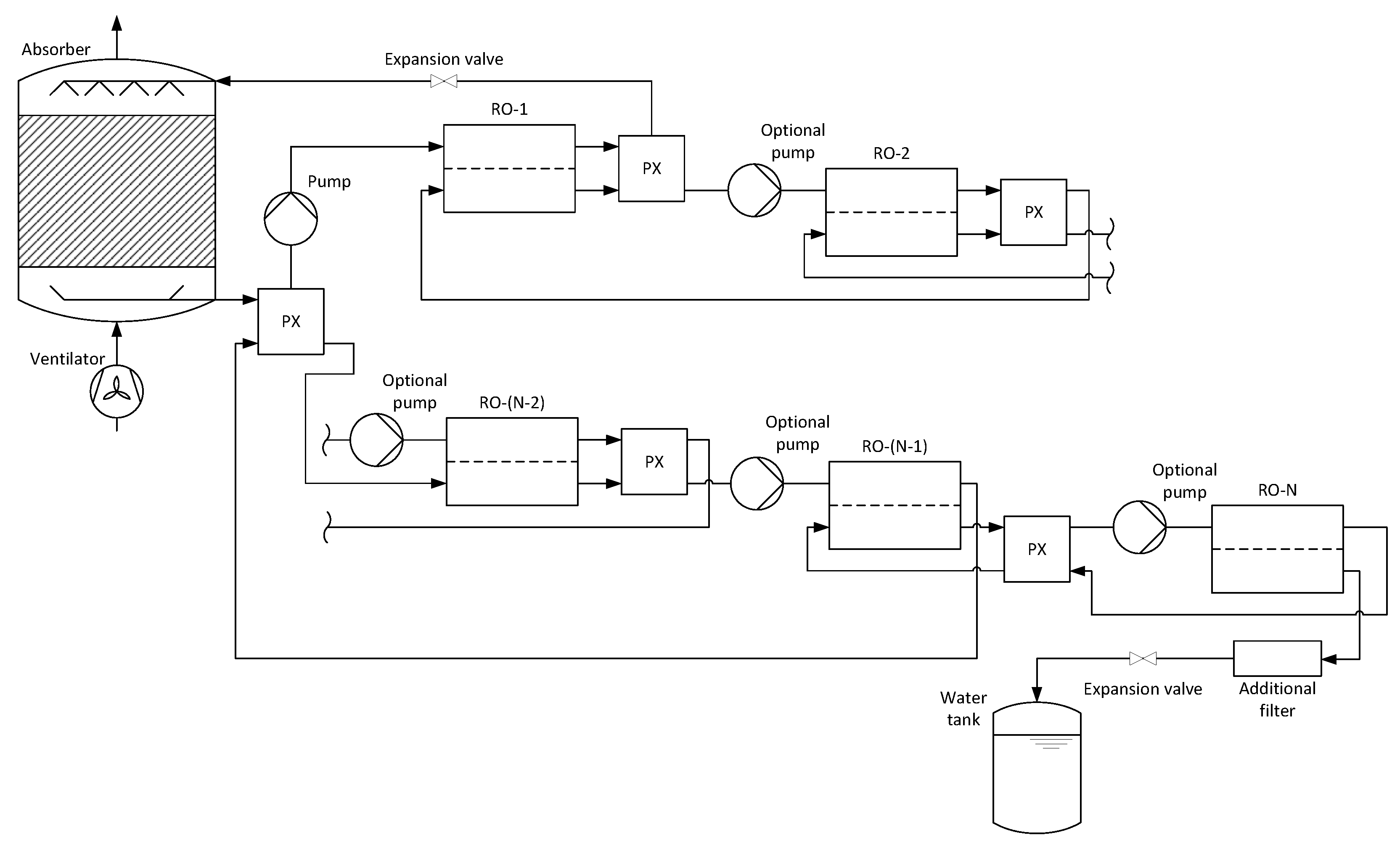
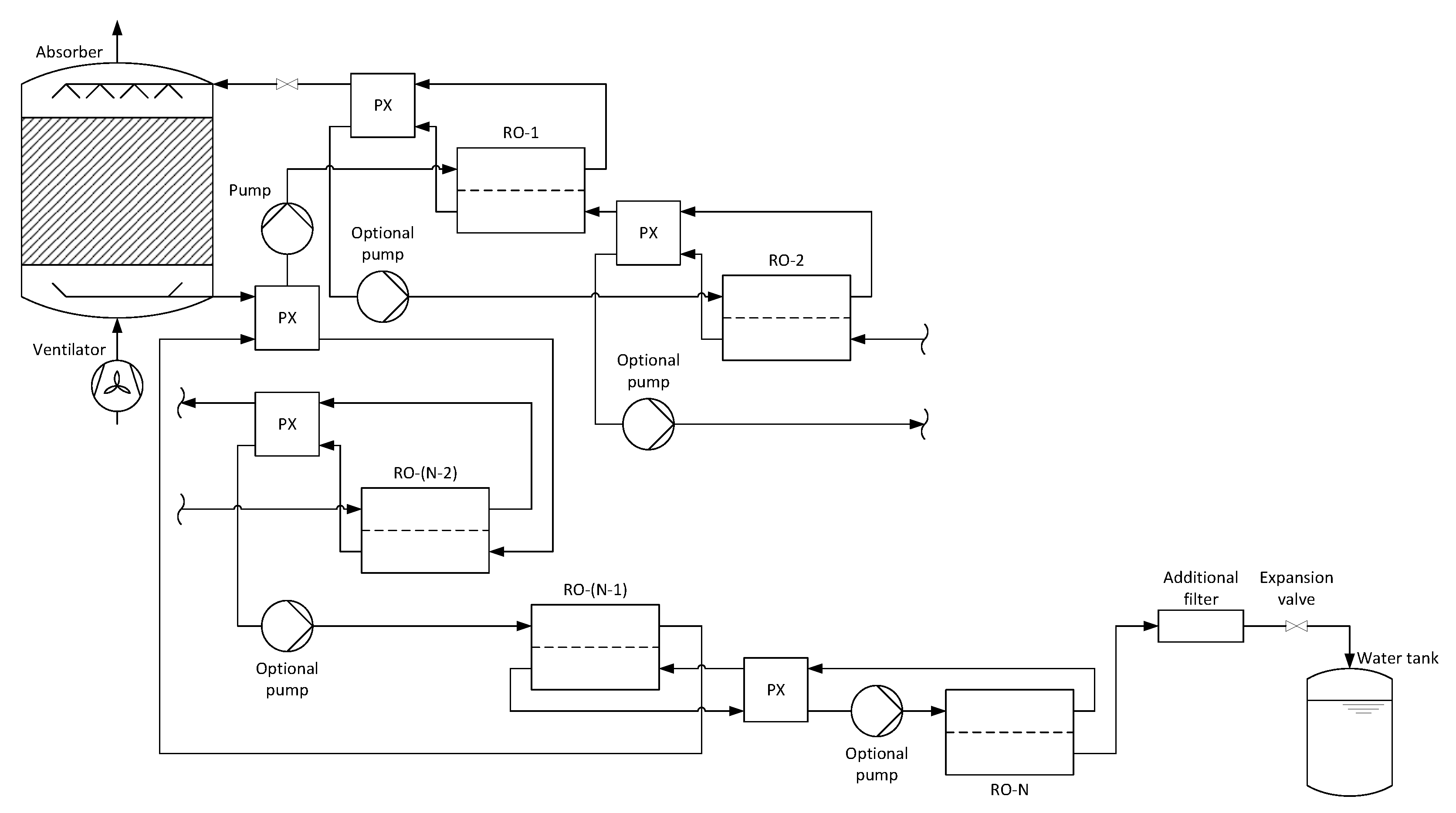
2.2. Conceptual Design of Reverse Osmosis Based Air Water Generator
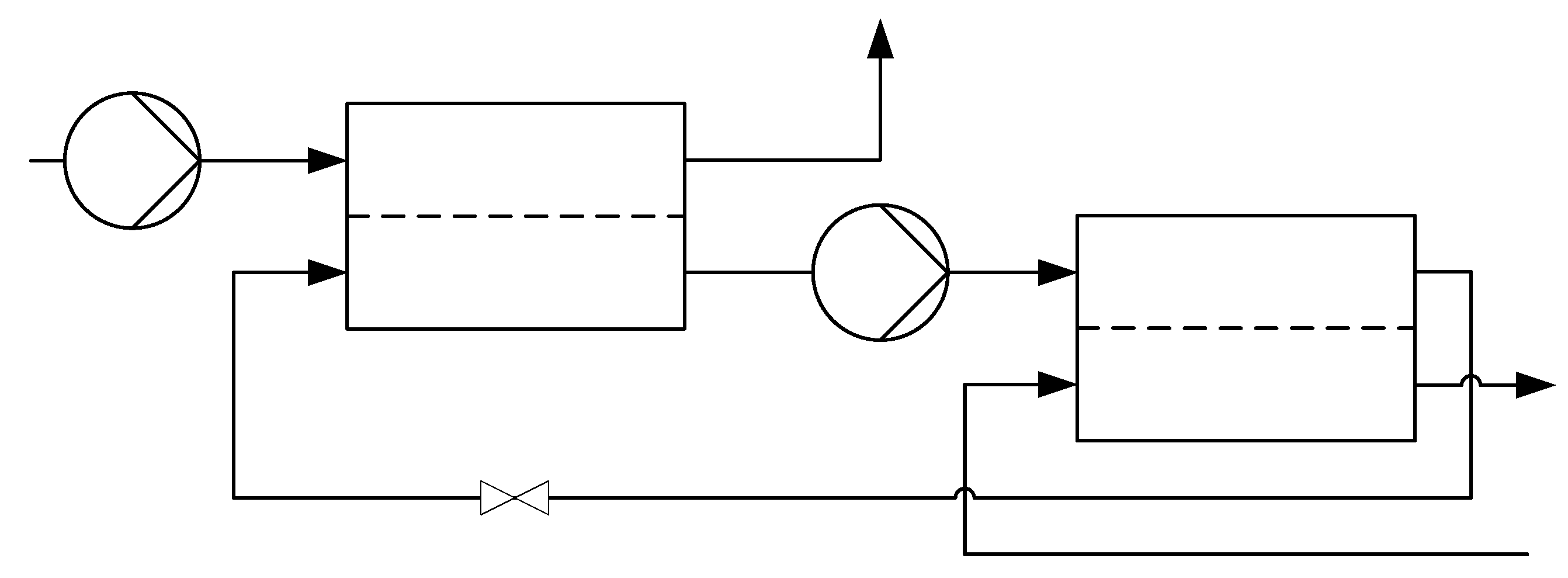
2.2.1. Co-Current Multi-Stage Reverse Osmosis
2.2.2. Countercurrent Multi-Stage Reverse Osmosis
2.3. Modeling
2.3.1. Absorber
Assumptions
- The pressure p in the aqueous lithium bromide solution is constant.
- The total pressure of the air is constant.
- The liquid film is flat and has no surface waves.
- The film thickness is considered constant along the height of the absorber column.
- The inlet mass flow rate of the solution and the inlet volume flow rate of the air are assumed to be constant and are calculated according to Appendices B and C of [9].
- The conditions of the air and solution are constant at a given height of the absorber.
Correlations
Calculations of the Absorber
Calculations of the Ventilator
Solution Algorithm
2.3.2. Reverse Osmosis Process
Assumptions
- No temperature changes over the membranes, the pressure exchangers or the pumps.
- The membrane has a salt rejection of 100%, so no salt flows through the membrane.
- Concentration polarization phenomena in the membrane are not considered.
- Water mass transfer through the membrane is calculated using a membrane constant.
- The representative membrane module used has a pressure drop of 1 bar; therefore, this pressure drop is distributed linearly over the membrane.
- No leakages between the streams in the pressure exchangers.
Calculations of the Reverse Osmosis Membrane Modules
Calculations of the Pressure Exchangers and Pumps
Solution Algorithm
2.4. Simulations
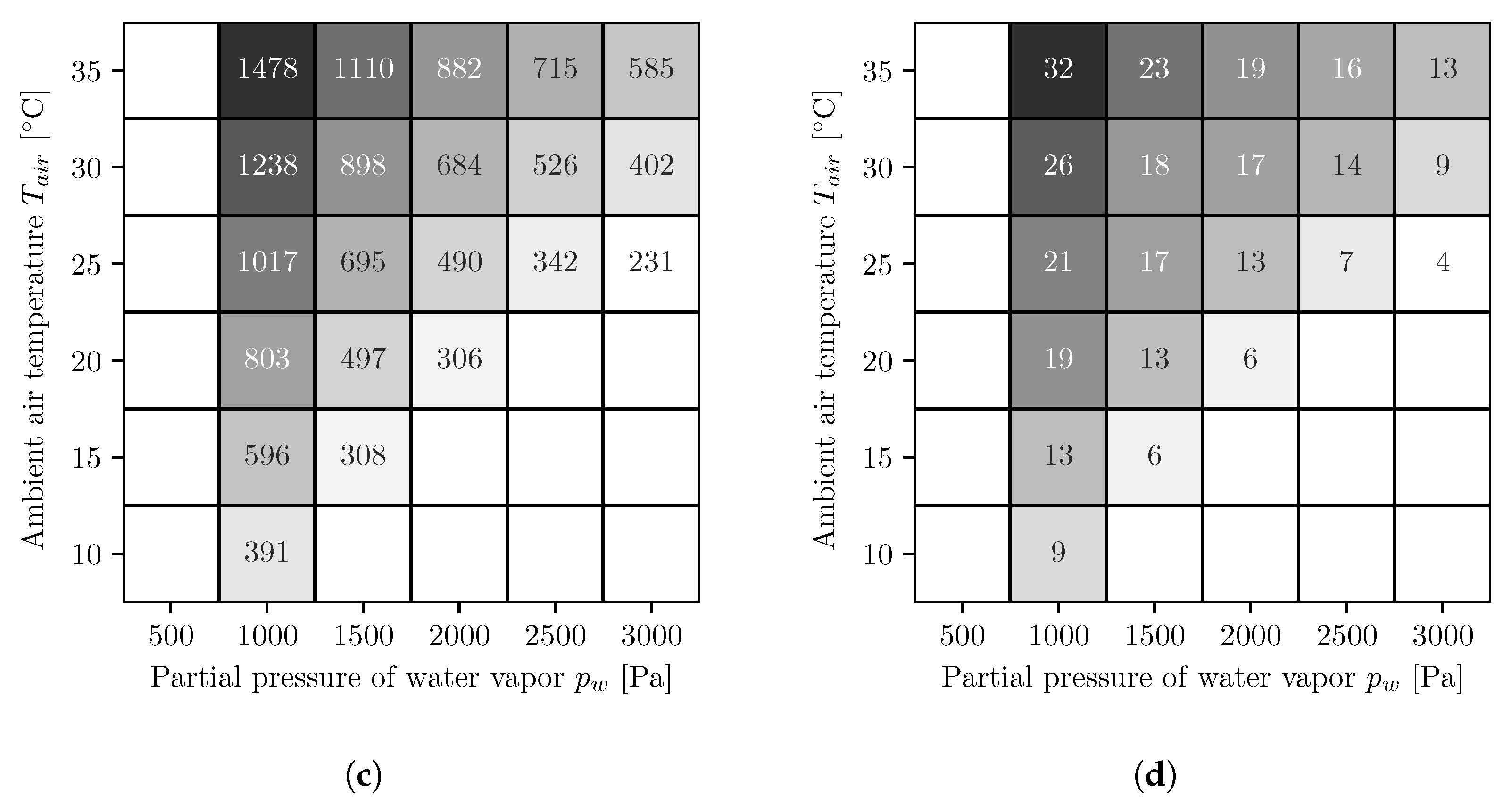
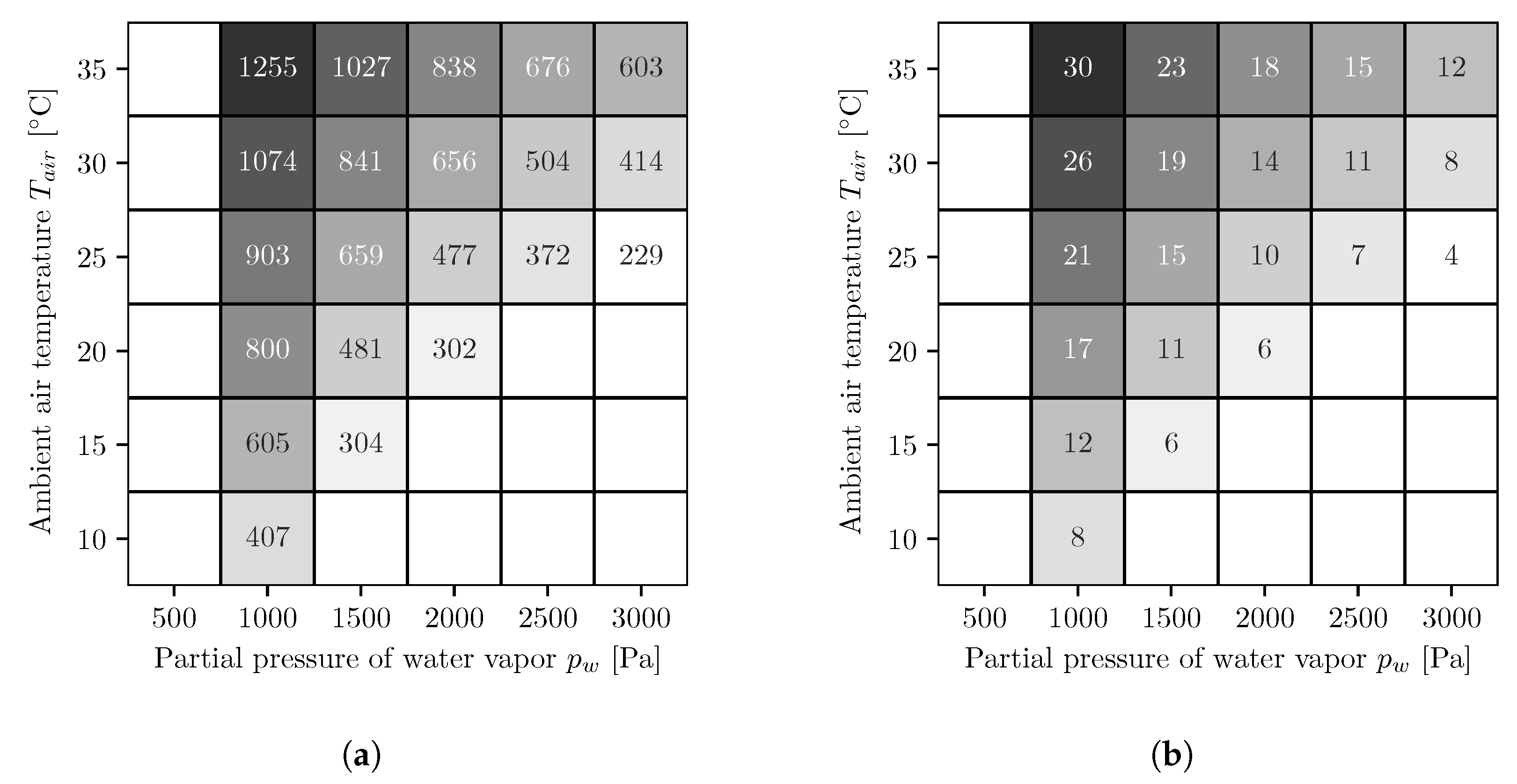
3. Results
3.1. Co-Current Multi-Stage Reverse Osmosis
3.2. Countercurrent Multi-Stage Reverse Osmosis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AWG | Air water generator/air water generation |
| LCOW | Levelized cost of water |
| PX | Pressure exchanger |
| Symbols | |
| a | Activity |
| A | Area |
| Membrane constant | |
| c | Concentration |
| Specific heat capacity (at constant pressure) | |
| d | Diameter |
| D | Diffusion coefficient |
| h | Specific enthalpy |
| Enthalpy flow | |
| J | Mass flux |
| Characteristic length | |
| Mass flow rate | |
| N | Number of elements |
| p | Pressure |
| P | Power |
| Heat flow | |
| R | Universal gas constant |
| s | Gap thickness |
| T | Temperature |
| Velocity | |
| V | Volume |
| Molar volume | |
| Volume flow rate | |
| Mass fraction of component i | |
| x | Position |
| Mole fraction of component i | |
| Mass load of water per component i | |
| Indices | |
| a | Air |
| Absorption | |
| Average | |
| Electric | |
| Element | |
| f | Feed |
| g | Gas |
| h | Hydraulic |
| i | i-th element |
| Inlet | |
| j | Solvent |
| Outlet | |
| p | Permeate |
| r | Retentate |
| Solution | |
| Total | |
| Vapor | |
| w | Water |
| Greek Symbols | |
| Heat transfer coefficient | |
| Mass transfer coefficient | |
| Activity coefficient | |
| Film thickness | |
| Drag coefficient | |
| Efficiency | |
| Temperature | |
| Thermal conductivity | |
| Chemical potential | |
| Kinematic viscosity | |
| Density | |
| Osmotic pressure | |
| Dimensionless Numbers | |
| Lewis number | |
| Nusselt number | |
| Prandtl number | |
| Reynolds number |
References
- IPCC. Summary for Policymakers. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S.L., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M.I., et al., Eds.; Cambridge University Press: Cambridge, UK, 2021. [Google Scholar]
- Markonis, Y.; Kumar, R.; Hanel, M.; Rakovec, O.; Máca, P.; AghaKouchak, A. The rise of compound warm-season droughts in Europe. Sci. Adv. 2021, 7, eabb9668. [Google Scholar] [CrossRef]
- Progress on Household Drinking Water, Sanitation and Hygiene 2000–2020: Five Years into the SDGs; Report; World Health Organization (WHO): Geneva, Switzerland; United Nations Children’s Fund (UNICEF): Geneva, Switzerland, 2021.
- Shiklomanov, I. Water in Crisis: A Guide to the World’s Fresh Water Resources; Chapter World Fresh Water Resources; Oxford University Press: New York, NY, USA, 1993. [Google Scholar]
- Baker, R.W. Membrane Technology and Applications; J. Wiley: Chichester, UK, 2004. [Google Scholar]
- Zheng, X.; Wen, J.; Shi, L.; Cheng, R.; Zhang, Z. A top-down approach to estimate global RO desalination water production considering uncertainty. Desalination 2020, 488, 114523. [Google Scholar] [CrossRef]
- Mirmanto, M.; Syahrul, S.; Wijayanta, A.; Mulyanto, A.; Winata, L. Effect of evaporator numbers on water production of a free convection air-water harvester. Case Stud. Therm. Eng. 2021, 27, 101253. [Google Scholar] [CrossRef]
- Li, R.; Shi, Y.; Wu, M.; Hong, S.; Wang, P. Improving atmospheric water production yield: Enabling multiple water harvesting cycles with nano sorbent. Nano Energy 2020, 67, 104255. [Google Scholar] [CrossRef]
- Fill, M.; Muff, F.; Kleingries, M. Evaluation of a new air water generator based on absorption and reverse osmosis. Heliyon 2020, 6, e05060. [Google Scholar] [CrossRef] [PubMed]
- DuPont de Nemours, Inc. DuPont™XUS180808 Reverse Osmosis Element. 2020. Available online: https://www.dupont.com/content/dam/dupont/amer/us/en/water-solutions/public/documents/en/45-D01736-en.pdf (accessed on 8 March 2021).
- Kraume, M. Transportvorgänge in der Verfahrenstechnik; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Cameron, I.B.; Clemente, R.B. SWRO with ERI’s PX Pressure Exchanger device—A global survey. Desalination 2008, 221, 136–142. [Google Scholar] [CrossRef]
- Hirschberg, H.G. Handbuch Verfahrenstechnik und Anlagenbau; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- University of Maryland. LiBrSSC (Aqueous Lithium Bromide) Property Routines. Available online: http://fchart.com/ees/libr_help/ssclibr.pdf (accessed on 6 December 2019).
- Rönsch, S. Anlagenbilanzierung in der Energietechnik; Springer: Wiesbaden, Germany, 2015. [Google Scholar] [CrossRef]
- Bo, S.; Ma, X.; Lan, Z.; Chen, J.; Chen, H. Numerical simulation on the falling film absorption process in a counter-flow absorber. Chem. Eng. J. 2010, 156, 607–612. [Google Scholar] [CrossRef]
- Xu, Z.F.; Khoo, B.C.; Wijeysundera, N.E. Mass transfer across the falling film: Simulations and experiments. Chem. Eng. Sci. 2008, 63, 2559–2575. [Google Scholar] [CrossRef]
- Stephan, P.; Kabelac, S.; Kind, M.; Mewes, D.; Schaber, K.; Wetzel, T. (Eds.) VDI-Wärmeatlas; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar] [CrossRef]
- Thomas Melin, R.R. Membranverfahren; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Jiang, A.; Ding, Q.; Wang, J.; Jiangzhou, S.; Cheng, W.; Xing, C. Mathematical Modeling and Simulation of SWRO Process Based on Simultaneous Method. J. Appl. Math. 2014, 2014, 908569. [Google Scholar] [CrossRef]
- Sundaramoorthy, S.; Srinivasan, G.; Murthy, D.V.R. An analytical model for spiral wound reverse osmosis membrane modules: Part I—Model development and parameter estimation. Desalination 2011, 280, 403–411. [Google Scholar] [CrossRef]
- Moser, M.; Micari, M.; Fuchs, B.; Farnós, J. Software Tool for the Simulation of Selected Brine Treatment Technologies; Technical Report; Horizon 2020 Framework Programme: Delft, The Netherlands, 2018. [Google Scholar]
- Böswirth, L.; Bschorer, S. Technische Strömungslehre; Springer: Wiesbaden, Germany, 2014. [Google Scholar] [CrossRef]
- Van Rossum, G.; Drake, F.L. Python 3 Reference Manual; CreateSpace: Scotts Valley, CA, USA, 2009. [Google Scholar]
- Vostermans Ventilation. Fiberglass Cone Fans—Efficient Ventilation with High Air Yields. 2020. Available online: https://www.vostermans.com/hubfs/Brochures/Fans/Fiberglass%20Cone%20Fans/Multifan%20Fiberglass%20Cone%20fan%20EN.pdf?hsLang=en (accessed on 8 March 2021).
- Conde-Petit, M.R. Solid—Liquid Equilibria (SLE) and Vapour—Liquid Equilibria (VLE) of Aqueous LiBr. 2014. Available online: http://www.aldacs.com/DocBase/AqLiBrSLEVLE.pdf (accessed on 6 December 2019).
- Wahlgren, R.V. Atmospheric water vapour processor designs for potable water production: A review. Water Res. 2001, 35, 1–22. [Google Scholar] [CrossRef]
- Leiva-Illanes, R.; Escobar, R.; Cardemil, J.M.; Alarcón-Padilla, D.C. Comparison of the levelized cost and thermoeconomic methodologies—Cost allocation in a solar polygeneration plant to produce power, desalted water, cooling and process heat. Energy Convers. Manag. 2018, 168, 215–229. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fill, M.; Kleingries, M. Modeling and Simulation of Either Co-Current or Countercurrent Operated Reverse-Osmosis-Based Air Water Generator. Membranes 2021, 11, 913. https://doi.org/10.3390/membranes11120913
Fill M, Kleingries M. Modeling and Simulation of Either Co-Current or Countercurrent Operated Reverse-Osmosis-Based Air Water Generator. Membranes. 2021; 11(12):913. https://doi.org/10.3390/membranes11120913
Chicago/Turabian StyleFill, Marc, and Mirko Kleingries. 2021. "Modeling and Simulation of Either Co-Current or Countercurrent Operated Reverse-Osmosis-Based Air Water Generator" Membranes 11, no. 12: 913. https://doi.org/10.3390/membranes11120913
APA StyleFill, M., & Kleingries, M. (2021). Modeling and Simulation of Either Co-Current or Countercurrent Operated Reverse-Osmosis-Based Air Water Generator. Membranes, 11(12), 913. https://doi.org/10.3390/membranes11120913





