Impact of the Recovery on Concentrating Acetic Acid with Low-Pressure Reverse-Osmosis Membranes
Abstract
:1. Introduction
2. Materials and Methods
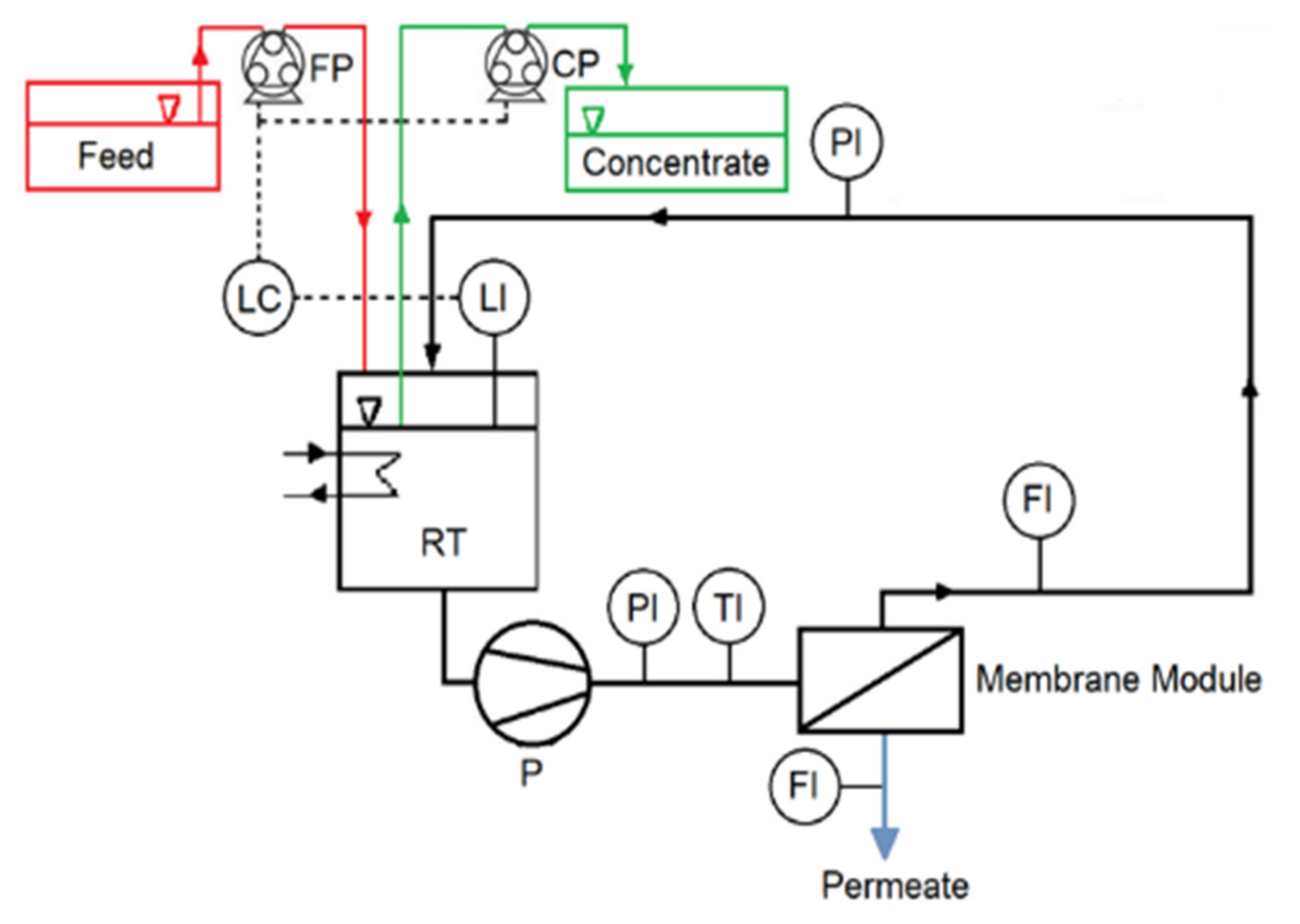
2.1. Experimental Setup
2.2. Feed Solution
2.3. Sampling and Analytics
2.4. Development of Parameters
3. Results and Discussion
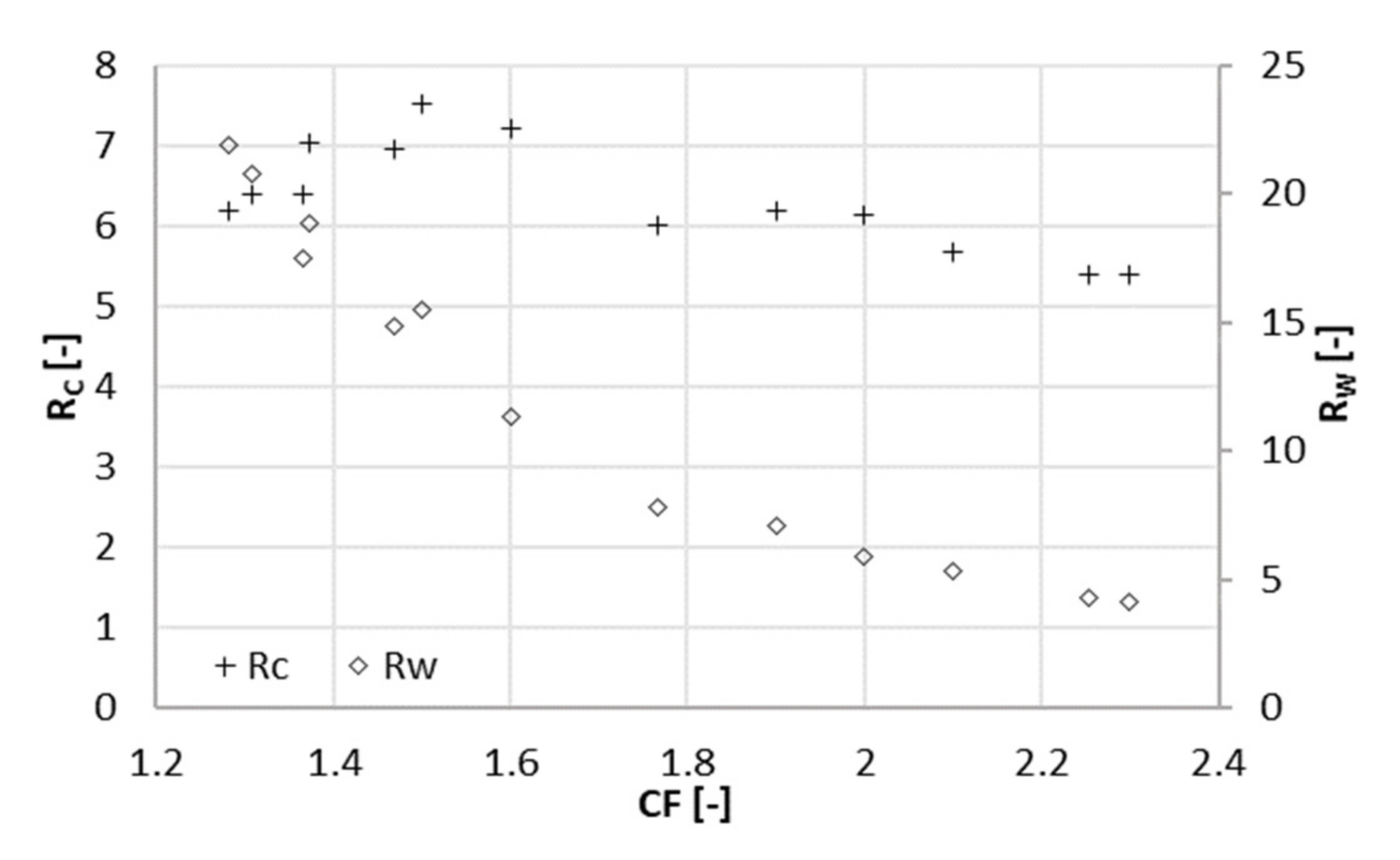
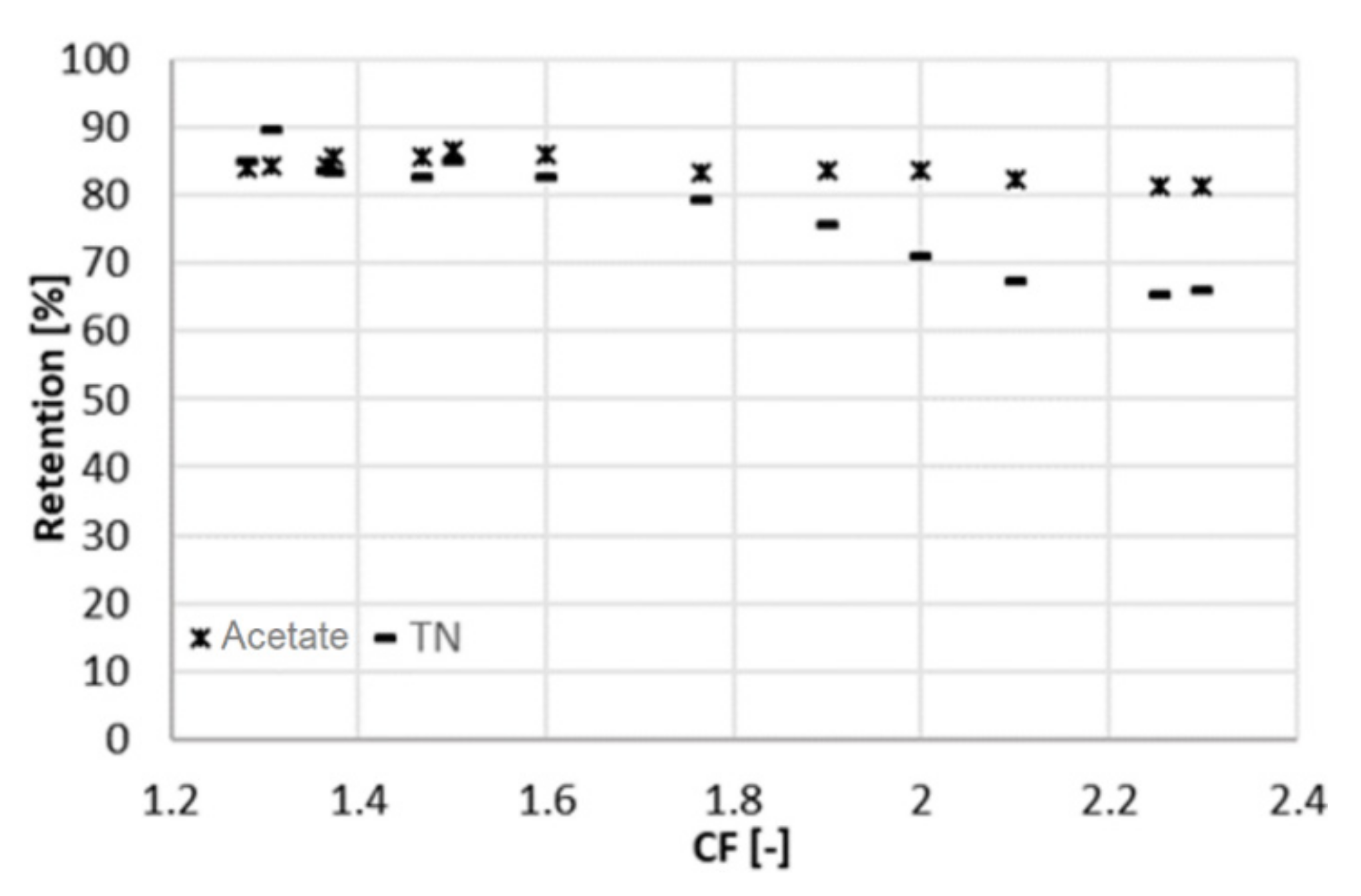
3.1. Experiment at 22 Bar
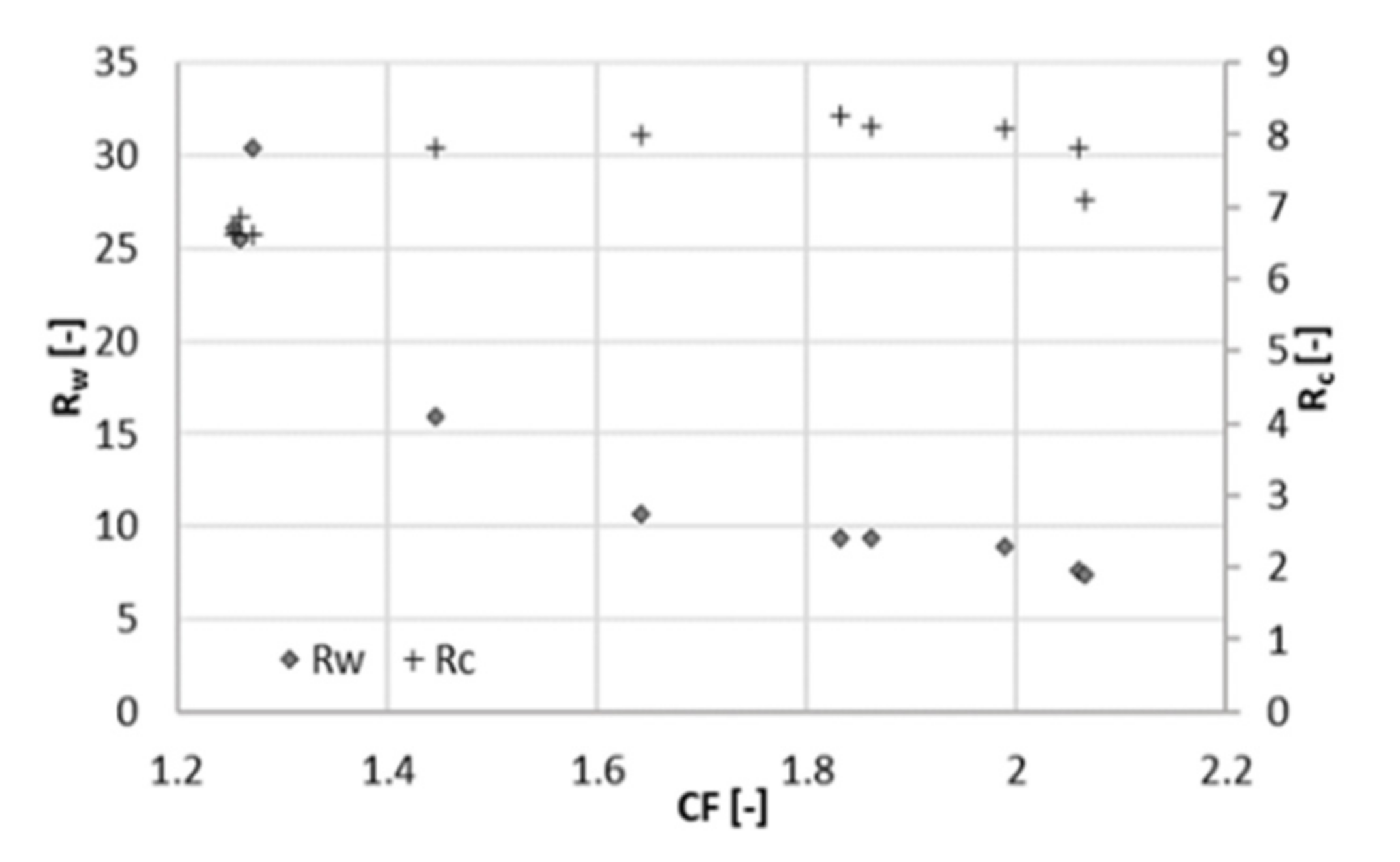
3.2. Experiment at 25 Bar
3.3. Economic Considerations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Y.; Shahbazi, A.; Williams, K.; Wan, C. Separate and concentrate lactic acid using combination of nanofiltration and reverse osmosis membranes. Appl. Biochem. Biotechnol. 2008, 147, 1–9. [Google Scholar] [CrossRef]
- Valentino, F.; Moretto, G.; Gottardo, M.; Pavan, P.; Bolzonella, D.; Majone, M. Novel routes for urban bio-waste management: A combined acidic fermentation and anaerobic digestion process for platform chemicals and biogas production. J. Clean. Prod. 2019, 220, 368–375. [Google Scholar] [CrossRef]
- Ali, R.; Saravia, F.; Hille-Reichel, A.; Härrer, D.; Gescher, J.; Horn, H. Enhanced production of propionic acid through acidic hydrolysis by choice of inoculum. J. Chem. Technol. Biotechnol. 2020, 96, 207–216. [Google Scholar] [CrossRef]
- Kumanowska, E.; Saldaña, M.U.; Zielonka, S.; Oechsner, H. Two-stage anaerobic digestion of sugar beet silage: The effect of the pH-value on process parameters and process efficiency. Bioresour. Technol. 2017, 245, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Ravi, P.P.; Merkle, W.; Tuczinski, M.; Saravia, F.; Horn, H.; Lemmer, A. Integration of membrane filtration in two-stage anaerobic digestion system: Specific methane yield potentials of hydrolysate and permeate. Bioresour. Technol. 2018, 275, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Aktija, S.A.; Zirehpour, A.; Mollahosseini, A.; Taherzadeh, M.J.; Tiraferri, A.; Rahimpour, A. Feasibility of membrane processes for the recovery and purification of bio-based volatile fatty acids: A comprehensive review. J. Ind. Eng. Chem. 2020, 81, 24–40. [Google Scholar] [CrossRef]
- Zhu, X.; Leininger, A.; Jassby, D.; Tsesmetzis, N.; Ren, Z.J. Will membranes break barriers on volatile fatty acid recovery from anaerobic digestion? ACS EST Eng. 2020, 1, 141–153. [Google Scholar] [CrossRef]
- Blandi, G.; Rosselló, B.; Monsalvo, V.M.; Batlle-Vilanova, P.; Viñas, J.M.; Rogalla, F.; Comas, J. Volatile fatty acids concentration in real wastewater by forward osmosis. J. Membr. Sci. 2019, 575, 60–70. [Google Scholar] [CrossRef]
- Atasoya, M.; Owusu-Agyemana, I.; Plazab, E.; Cetecioglu, Z. Bio-based volatile fatty acid production and recovery from waste streams: Current status and future challenges. Bioresour. Technol. 2018, 268, 773–786. [Google Scholar] [CrossRef]
- Choi, J.H.; Fukushi, K.; Yamamoto, K. A study on the removal of organic acids from wastewaters using nanofiltration membranes. Sep. Purif. Technol. 2008, 59, 17–25. [Google Scholar] [CrossRef]
- Bellona, C.; Drews, J.E. The role of membrane surface charge and solute physico-chemicalproperties in the rejection of organic acids by NF membranes. J. Membr. Sci. 2005, 249, 227–234. [Google Scholar] [CrossRef]
- Verliefde, A.R.D.; Cornelissen, E.R.; Heijman, S.G.J.; Verberk, J.Q.J.C.; Amy, G.L.; Van der Bruggen, B.; van Dijk, J.C. The role of electrostatic interactions on the rejection of organic solutes in aqueous solutions with nanofiltration. J. Membr. Sci. 2008, 322, 52–66. [Google Scholar] [CrossRef]
- Umpuch, C.; Galier, S.; Kanchanatawee, S.; Roux-de Balmann, H. Nanofiltration as a purification step in production process of organic acids: Selectivity improvement by addition of an inorganic salt. Process. Biochem. 2010, 45, 1763–1768. [Google Scholar] [CrossRef] [Green Version]
- Zacharof, M.P.; Mandale, S.J.; Williams, P.M.; Lovitt, R.W. Nanofiltration of treated digested agricultural wastewater for recovery of carboxylic acids. J. Clean. Prod. 2015, 112, 4749–4761. [Google Scholar] [CrossRef]
- Zhu, Y.; Galier, S.; Roux-de Balmann, H. Nanofiltration of solutions containing organic and inorganic salts: Relationship between feed and permeate proportions. J. Membr. Sci. 2020, 613, 118380. [Google Scholar] [CrossRef]
- Laufenberg, G.; Hausmanns, S.; Kunz, B. The influence of intermolecular interactions on the selectivity of several organic acids in aqueous multi-component systems during reverse osmosis. J. Membr. Sci. 1996, 110, 59–68. [Google Scholar] [CrossRef]
- Bóna, Á.; Bakonyi, P.; Galambos, I.; Bélafi-Bakó, K.; Nemestóthy, N. Separation of Volatile Fatty Acids from model anaerobic effuents using various membrane technologies. Membranes 2020, 10, 252. [Google Scholar] [CrossRef]
- Van der Bruggen, B.; Schaep, J.; Wilms, D.; Vandecasteele, C. Infuence of molecular size, polarity and charge on the retention of organic molecules by nanofiltration. J. Membr. Sci. 1999, 156, 29–41. [Google Scholar] [CrossRef]
- Braeken, L.; Ramaekers, R.; Zhang, Y.; Maes, G.; Van der Bruggen, B.; Vandecasteele, C. Influence of hydrophobicity on retention in nanofiltration of aqueous solutions containing organic compounds. J. Membr. Sci. 2005, 252, 195–203. [Google Scholar] [CrossRef]
- Zhou, F.; Wang, C.; Wei, J. Separation of acetic acid from monosaccharides by NF and RO membranes: Performance comparison. J. Membr. Sci. 2013, 429, 243–251. [Google Scholar] [CrossRef]
- Zhou, F.; Wang, C.; Wei, J. Simultaneous acetic acid separation and monosaccharide concentration by reverse osmosis. Bioresour. Technol. 2013, 131, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Roman, R. Eliminierung von Organozinnverbindungen aus Industrieabwasser im Labor- und Technikumsmaßstab. Ph.D. Thesis, Paderborn University, Paderborn, Germany, 2006. [Google Scholar]
- Warnecke, H.J. Reduktion des Eintrags von Organozinnverbindungen in Die Umwelt. Available online: https://www.lanuv.nrw.de/fileadmin/lanuv/wasser/abwasser/forschung/pdf/Abschlussberichtreduktion.pdf (accessed on 23 August 2021).
- West, S.; Wagner, M.; Engelke, C.; Horn, H. Optical coherence tomography for the in situ three-dimensional visualization and quantification of feed spacer channel fouling in reverse osmosis membrane modules. J. Membr. Sci. 2016, 498, 345–352. [Google Scholar] [CrossRef]
- Jänisch, T.; Reinhardt, S.; Pohsner, U.; Böringer, S.; Bolduan, R.; Steinbrenner, J.; Oechsner, H. Separation of volatile fatty acids from biogas plant hydrolysates. Sep. Purif. Technol. 2019, 223, 264–273. [Google Scholar] [CrossRef]
- Nagy, E.; Hegedüs, I.; Rehman, D.; Wei, Q.J.; Ahdab, Y.D.; Lienhard, J.H. The need for accurate osmotic pressure and mass transfer resistances in modeling osmotically driven membrane processes. Membranes 2021, 11, 128. [Google Scholar] [CrossRef]
- Salcedo-Díaz, R.; García-Algado, P.; García-Rodríguez, M.; Fernández-Sempere, J.; Ruiz-Beviá, F. Visualization and modeling of the polarization layer in crossflow reverse osmosis in a slit-type channel. J. Membr. Sci. 2014, 456, 21–30. [Google Scholar] [CrossRef] [Green Version]
- Jung, O. Raman Microspectroscopy for In-Situ Measurement of Concentration Polarization in Nanofiltration. Ph.D. Thesis, Karlsruhe Institute of Technology, Karlsruhe, Germany, 2020. [Google Scholar]



| Membrane | Material | VFA Recovered | NF/LPRO | Manufacturer | Reference |
|---|---|---|---|---|---|
| NF-90 | Polyamide | Acetic acid | NF | FilmTec-Dow | [11] |
| NF-270 | Polyamide | Formic, acetic, butyric acids | NF | FilmTec-Dow | [10] |
| ES10 | Aromatic polyamide | Formic, acetic, butyric acids | NF | Nitto Denko | [10] |
| Desal-5 DK | Cross-linked aromatic polyamide | Lactic acid | LPRO | GE | [13] |
| SW30 | Polyamide | Acetic, propionic, butyric acids | LPRO | FilmTec-Dow | [17] |
| dNF40 | Polyethersulfone | Acetic, propionic, butyric acids | NF | NX Filtration | [17] |
| RO98pHt | Aromatic polyamide | Acetic acid | LPRO | Alfa Laval | [20,21] |
| Trisep TS80 | Aromatic polyamide | Formic, acetic, lactic acids | NF | Trisep | [12] |
| Desal HL | Cross-linked aromatic polyamide | Formic, acetic, lactic acids | NF | GE | [12] |
| XLE | Polyamide | Acetic, propionic, butyric acids | LPRO | FilmTec-Dow | [15] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pratofiorito, G.; Horn, H.; Saravia, F. Impact of the Recovery on Concentrating Acetic Acid with Low-Pressure Reverse-Osmosis Membranes. Membranes 2021, 11, 742. https://doi.org/10.3390/membranes11100742
Pratofiorito G, Horn H, Saravia F. Impact of the Recovery on Concentrating Acetic Acid with Low-Pressure Reverse-Osmosis Membranes. Membranes. 2021; 11(10):742. https://doi.org/10.3390/membranes11100742
Chicago/Turabian StylePratofiorito, Giorgio, Harald Horn, and Florencia Saravia. 2021. "Impact of the Recovery on Concentrating Acetic Acid with Low-Pressure Reverse-Osmosis Membranes" Membranes 11, no. 10: 742. https://doi.org/10.3390/membranes11100742
APA StylePratofiorito, G., Horn, H., & Saravia, F. (2021). Impact of the Recovery on Concentrating Acetic Acid with Low-Pressure Reverse-Osmosis Membranes. Membranes, 11(10), 742. https://doi.org/10.3390/membranes11100742






