A Review on the Mechanism, Impacts and Control Methods of Membrane Fouling in MBR System
Abstract
1. Introduction
2. Classification of Membrane Fouling
3. Factors Affecting the Fouling of Membranes
3.1. Influence of Membrane Intrinsic Properties on Membrane Fouling
3.1.1. Effect of Membrane Material on Membrane Fouling
3.1.2. Effect of Hydrophilicity/Hydrophobicity on Membrane Fouling
3.1.3. Effect of Membrane Surface Charge on Membrane Fouling
3.1.4. Effect of Membrane Pore Size, Distribution and Structure on Membrane Fouling
3.1.5. Effect of Porosity and Roughness on Membrane Fouling
3.1.6. Effect of Membrane Module Structure on Membrane Fouling
3.2. Effect of Operating Conditions on Membrane Fouling
3.2.1. Effect of Membrane Flux and TMP on Membrane Fouling
3.2.2. Effect of Aeration and CFV on Membrane Fouling
3.2.3. Effect of SRT and HRT on Membrane Fouling
3.2.4. Effect of Temperature on Membrane Fouling
3.2.5. Effect of the Mode of Operation on Membrane Fouling
3.3. Effect of Character of Activated Sludge Mixture on Membrane Fouling
3.3.1. Effect of Activated Sludge Components on Membrane Fouling
3.3.2. Effect of MLSS on Membrane Fouling
3.3.3. Effect of Sludge Viscosity on Membrane Fouling
3.3.4. Effect of EPS and SMP on Membrane Fouling
3.3.5. The Effect of Microorganisms on Membrane Fouling
4. Membrane Fouling Control
4.1. Modification of Membrane Material Body
4.1.1. Physical Blending
4.1.2. Chemical Copolymerization
4.2. Hydrophilic Modification of the Surface of Membrane Material
4.2.1. Surface Coating
4.2.2. Membrane Modification by Low Temperature Plasma Surface Treatment
4.2.3. Surface Grafting
4.3. Optimization of Membrane Modules
4.4. Changing the Properties of the Feed Water
4.5. Control of Operating Conditions
4.6. Cleaning of Membrane Fouling
4.6.1. Physical Cleaning
4.6.2. Chemical Cleaning
4.6.3. Electric Cleaning
4.6.4. Ultrasonic Cleaning
5. Conclusions
Funding
Conflicts of Interest
Abbreviations
| AnMBR | anaerobic membrane bioreactor |
| BEPS | blend extracellular polymer |
| BPC | biopolymer clusters |
| CFD | computational fluid dynamics |
| CFV | cross-flow velocity |
| CLSM | confocal laser scanning microscopy |
| CMPSF | chloromethylated polysulfone |
| COD | chemical oxygen demand |
| CST | capillary suction time |
| DO | dissolved oxygen |
| DMAEMA | dimethylaminoethyl methacrylate |
| DPM | discrete phase model |
| ECM | extracellular matrix |
| EDTA | ethylene diamine tetraacetic |
| EPS | extracellular polymers |
| F/M | food to microorganism ratio |
| FO | forward osmosis |
| HA | humicacid |
| HPI | hydrophilic |
| HRT | hydraulic retention time |
| HPO | hydrophobic |
| MBR | membrane bioreactor |
| MF | microfiltration |
| MLSS | mixed liquid suspended solids |
| OMBR | osmotic membrane bioreactor |
| OPMT | only plasma modify treatment; |
| OLR | organic loading rate |
| PAC | powdered activated carbon |
| PAN | polyacrylonitrile |
| pH | hydrogen ion concentration |
| PE | polyethylene |
| PES | polyethersulfone |
| POEM | polyoxyethylene methacrylate |
| PPG-CT | plasma polymerization graft coating treatment |
| PTRGP | plasma trigger radical graft polymerization |
| PVDF | polyvinylidene fluoride |
| PSD | particle size distribution |
| PVC | Polyvinyl chloride |
| PVP | Polyvinyl pyrrolidone |
| PS | polysulfone |
| PVAc | polyvinyl acetate |
| PVA | polyvinyl alcohol |
| SA | sodium alginate |
| SEC-OCD | size exclusion chromatography with organic carbon detector |
| SF-DMBR | self-forming dynamic membrane bioreactor |
| SMB | sponge-based moving bed |
| SMBR | small-scale immersed MBR |
| SMP | microbial metabolites products |
| SRT | sludge retention time |
| SG | suspended-growth |
| SPS | sulfonated polystyrene |
| SPES-C | sulfonated polyaryl ether sulfone |
| TMP | transmembrane pressure |
| TOC | total organic carbon |
| UV | under voltage |
| WOM | wastewater organic matter |
| XPS | X-ray photoelectron spectroscopy |
Appendix A
| Research Area | References |
|---|---|
| FO; RO; Driven membrane processes; Biofilm dynamics; Membrane performance; Concentration polarization | [2,12,26,56,105] |
| EPS; SMP; Microbial community structure; Microbial flocs; Microbial soluble substances; Membrane modification | [7,8,15,34,63] |
| Membrane cleaning; Membrane fouling control; Cross-flow membrane filtration; osmotic pressure | [2,4,10,11,13] |
| Inherent properties of membrane; Operating conditions; Mixed liquid properties; Fouling mechanisms | [14,22,34,81,88] |
| Anaerobic membrane bioreactor; Influencing factors; Domestic wastewater; Biosolids production; Energy; Reuse | [23,30,43,45,47,166] |
| Chemical oxygen demand; SRT; HRT | [22,24,37,60] |
| Ultrasonication; Hollow fiber membrane; Mathematical model; Emerging micropollutants | [27,44,60,96] |
| Nutrient recovery; Phosphate recovery; Ammonia recovery; Hybrid system; Direct membrane; | [22,60,112,116,128] |
References
- Li, C.; Deng, W.; Gao, C.; Xiang, X.M.; Feng, X.H.; Batchelor, B.; Li, Y. Membrane distillation coupled with a novel two-stage pretreatment process for petrochemical wastewater treatment and reuse. Sep. Purif. Technol. 2019, 224, 23–32. [Google Scholar] [CrossRef]
- Abdelrasoul, A.; Doan, H.; Lohi, A. Fouling in forward osmosis membranes: Mechanisms, control, and challenges. In Osmotically Driven Membrane Processes: Approach, Development and Current Status; IntechOpen Limited: London, UK, 2018; pp. 151–177. [Google Scholar]
- Gong, H.; Jin, Z.; Wang, Q.; Zuo, J.; Wu, J.; Wang, K.J. Effects of adsorbent cake layer on membrane fouling during hybrid coagulation/adsorption microfiltration for wastewater organic recovery. Chem. Eng. J. 2017, 317, 751–757. [Google Scholar] [CrossRef]
- Choudhury, M.R.; Anwar, N.; Jassby, D.; Rahaman, M.S. Fouling and wetting in the membrane distillation driven wastewater reclamation process—A review. Adv. Colloid Interf. Sci. 2019, 269, 370–399. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.Y.; Li, X.F.; Feng, L. Characteristics of Extracellular Polysaccharide in Bio-fouling Layer of MBR. Environ. Sci. Technol. 2008, 31, 99–102. [Google Scholar]
- Zhang, K.; Choi, H.; Dionysiou, D.D.; Sorial, G.A.; Oerther, D.B. Identifying pioneer bacterial species responsible for biofouling membrane bioreactors. Environ. Microbiol. 2006, 8, 433–440. [Google Scholar] [CrossRef]
- Wu, B.; Fane, A.G. Microbial relevant fouling in membrane bioreactors: Influencing factors, characterization, and fouling control. Membranes 2012, 2, 565–584. [Google Scholar] [CrossRef]
- Meng, F.G.; Zhang, S.Q.; Oh, Y.; Zhou, Z.B.; Shin, H.S.; Chae, S.R. Fouling in membrane bioreactors: An updated review. Water Res. 2017, 114, 151–180. [Google Scholar] [CrossRef]
- Chen, Y.Q.; Li, F.; Qiao, T.J. Research on ultrafiltration membrane fouling based on chemical cleaning. China Water Wastewater 2013, 29, 51–54. [Google Scholar]
- Blandin, G.; Verliefde, A.R.D.; Comas, J. Efficiently combining water reuse and desalination through forward osmosis-reverse osmosis (FO-RO) hybrids: A critical review. Membranes 2016, 6, 37. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Kim, A.S.; Elimelech, M. Concentration polarization of interacting solute particles in cross-flow membrane filtration. Colloid Interface Sci. 1999, 212, 81–99. [Google Scholar] [CrossRef]
- Li, C.; Yang, Y.; Ding, S.Y. Dynamics of bio-fouling development on the conditioned membrane and its relationship with membrane performance. J. Membr. Sci. 2016, 514, 264–273. [Google Scholar] [CrossRef]
- Zhou, X.L.; Chen, J.R.; Yu, G.Y.; Hong, H.C.; Jin, L.; Lu, X.F.; Lin, H. Review on mechanism and control of membrane fouling in membrane bioreactor. Environ. Sci. Technol. 2012, 35, 86–91. [Google Scholar]
- Le Clech, P.; Chen, V.; Fane, T.A.G. Fouling in membrane bioreactors used in wastewater treatment. J. Membr. Sci. 2006, 284, 17–53. [Google Scholar] [CrossRef]
- Meng, F.; Chae, S.R.; Drews, A.; Kraume, M.; Shin, H.S.; Yang, F.L. Recent advances in membrane bioreactors (MBRs): Membrane fouling and membrane material. Water Res. 2009, 43, 1489–1512. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.M.; Li, X.Y. Accumulation of biopolymer clusters in a submerged membrane bioreactor and its effect on membrane fouling. Water Res. 2008, 42, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.J.; Xie, K.; Mahendran, B. Sludge properties and their effects on membrane fouling in submerged anaerobic membrane bioreactors (SAnMBRs). Water Res. 2009, 43, 3827–3837. [Google Scholar] [CrossRef]
- Wang, L.; Liu, H.B.; Zhang, W.D.; Yu, T.T.; Jin, Q.; Fu, B.; Liu, H. Recovery of organic matters in wastewater by self-forming dynamic membrane bioreactor: Performance and membrane fouling. Chemosphere 2018, 203, 123–131. [Google Scholar] [CrossRef]
- Gao, W.J.; Lin, H.J.; Leung, K.T.; Liao, B.Q. Influence of elevated pH shocks on the performance of a submerged anaerobic membrane bioreactor. Process Biochem. 2010, 45, 1279–1287. [Google Scholar] [CrossRef]
- Arena, J.T.; McCloskey, B.; Freeman, B.D.; McCutcheon, J.R. Surface modification of thin film composite membrane support layers with polydopamine: Enabling use of reverse osmosis membranes in pressure retarded osmosis. J. Membr. Sci. 2011, 375, 55–62. [Google Scholar] [CrossRef]
- Li, X.H. Study of Modified PVDF UF Membrane Anti-Fouling Performance. Master’s Thesis, Harbin Institute of Technology, Harbin, China, 2010. [Google Scholar]
- Xie, Y.H.; Zhu, T.; Xu, C.H. Research progress in influence factors on membrane fouling in membrane bioreactor. Chem Eng. (CHINA) 2010, 38, 26–31. [Google Scholar]
- Zheng, Y.L.; Li, H.Q.; Liu, L. Research progress in influence factors and control technologies of membrane fouling in anaerobic membrane bioreactor. Environ. Sci. Technol. 2015, 28, 71–75. [Google Scholar]
- Jegatheesan, V.; Pramanik, B.K.; Chen, J.; Navaratna, D.; Chang, C.Y.; Shu, L. Treatment of textile wastewater with membrane bioreactor: A critical review. Bioresour. Technol. 2016, 204, 202–212. [Google Scholar] [CrossRef]
- Yang, F.L.; Zhang, S.T.; Zhang, X.W.; Qu, Y.B.; Liu, Y.H. Experimental study of domestic wastewater treatment with a metal membrane bioreactor. Desalination 2005, 177, 83–93. [Google Scholar]
- Xiao, Q.Q.; Xu, S.C.; Wang, Y. Research and analysis on influencing factors of forward osmosis membrane fouling. Chem. Ind. Eng. Prog. 2018, 37, 59–367. [Google Scholar]
- Mu, S.T.; Fan, H.J.; Han, B.J. Research progress on the process and mathematical model of hollow fiber membrane fouling. Membr. Sci. Technol. 2018, 38, 114–121. [Google Scholar]
- Qu, F.S.; Liang, H.; Wang, Z.Z.; Wang, H.; Yu, H.; Li, G. Ultrafiltration membrane fouling by extracellular organic matters(EOM) of microcystis aeruginosa in stationary phase: Influences of interfacial characteristics of foulants and fouling mechanisms. Water Res. 2012, 46, 1490–1500. [Google Scholar] [CrossRef]
- Katsoufidou, K.S.; Sioutopoulos, D.C.; Yiantsios, S.G.; Karabelas, A.J. UF membrane fouling by mixtures of humic acids and sodium alginate: Fouling mechanisms and reversibility. Desalination 2010, 64, 220–227. [Google Scholar] [CrossRef]
- Shin, C.; Jaeho, B. Current status of the pilot-scale anaerobic membrane bioreactor treatments of domestic wastewaters: A critical review. Bioresour. Technol. 2018, 247, 1038–1046. [Google Scholar] [CrossRef]
- Tian, J.Y.; Ernst, M.; Cui, F.Y.; Jekel, M. Effect of particle size and concentration on the synergistic UF membrane fouling by particles and NOM fractions. J. Membr. Sci. 2013, 446, 1–9. [Google Scholar] [CrossRef]
- Blandin, G.; Gautier, C.; Toran, S.M.; Monclus, H.; Rodriguez-Roda, I.; Comas, J. Retrofitting membrane bioreactor (MBR) into osmotic membrane bioreactor (OMBR): A pilot scale study. Chem. Eng. J. 2019, 339, 268–277. [Google Scholar] [CrossRef]
- Oliver, T.I.; Rania, A.H.; Joo, H.T. Membrane fouling control in membrane bioreactors (MBRs) using granular materials. Bioresour. Technol. 2017, 240, 9–24. [Google Scholar]
- Lin, H.J.; Zhang, M.J.; Wang, F.Y.; Meng, F.; Liao, B.-Q.; Hong, H.; Chen, J.; Gao, W. A critical review of extracellular polymeric substances (EPSs) in membrane bioreactors: Characteristics, roles in membrane fouling and control strategies. J. Membr. Sci. 2014, 460, 110–125. [Google Scholar] [CrossRef]
- Zhou, J.E.; Wang, Y.Q.; Zhang, X.Z. Electrokinetic properties of zirconia/cordierite microfiltration membrane and its influence on permeate flux. J. Chin. Ceram. Soc. 2009, 37, 299–303. [Google Scholar]
- Wu, N.P.; Kong, X.Y.; Fang, S. Study on the application of MBR technology in the recycling of micro-polluted surface water. Memb. Sci. Technol. 2016, 36, 103–108. [Google Scholar]
- Noor, S.A.M.; Zainura, Z.N.; Mohd, A.A.H.; Gustaf, O. Application of membrane bioreactor technology in treating high strength industrial wastewater: A performance review. Desalination 2012, 305, 1–11. [Google Scholar]
- Qiu, H.Y.; Xiao, T.H.; Hu, N.E. The microporous membrane with different pore sizes was used to study the treatment of micro-polluted water in MBR. Technol. Water Treat. 2018, 44, 94–98. [Google Scholar]
- Aslam, M.; Lee, P.H.; Kim, J. Analysis of membrane fouling with porous membrane filters by microbial suspensions for autotrophic nitrogen transformations. Sep. Purif. Technol. 2015, 146, 284–293. [Google Scholar] [CrossRef]
- Kang, S.; Hoekem, V. Effect of membrane surface properties during the fast evaluation of cell attachment. Sep. Sci. Technol. 2006, 41, 1475–1487. [Google Scholar] [CrossRef]
- Fang, H.P.; Shi, X.L. Pore fouling of microfiltration membranes by activated sludge. Membr. Sci. 2005, 264, 161–166. [Google Scholar] [CrossRef]
- Choijh, Y. Effect of membrane type and material on performance of a submerged membrane bioreactor. Chemosphere 2008, 71, 853–859. [Google Scholar]
- Hale, O.; Recep, K.D.; Mustafa, E.E.; Cumali, K.; Spanjers, H.; Lier, J.B.V. A review of anaerobic membrane bioreactors for municipal wastewater treatment: Integration options, limitations and expectations. Sep. Purif. Technol. 2013, 118, 89–104. [Google Scholar]
- Pawel, K.; Lance, L.; Simos, M.; Evina, K. Membrane bioreactors -a review on recent developments in energy reduction, fouling control, novel configurations, LCA and market prospects. J. Membr. Sci. 2017, 527, 207–227. [Google Scholar]
- Lin, H.J.; Peng, W.; Zhang, M.J.; Chen, J.R.; Hong, H.C.; Zhang, Y. A review on anaerobic membrane bioreactors: Applications, membrane fouling and future perspectives. Desalination 2013, 314, 169–188. [Google Scholar] [CrossRef]
- Arabi, S.; Nakhla, G. Impact of protein/carbohydrate ratio in the feed wastewater on the membrane fouling in membrane bioreactors. J. Membr. Sci. 2008, 324, 142–150. [Google Scholar] [CrossRef]
- Chen, J.R.; Zhang, M.J.; Li, F.Q.; Qian, L.; Lin, H.G.; Yang, L.N.; Wu, X.L.; Zhou, X.L.; He, Y.M.; Liao, B.Q. Membrane fouling in a membrane bioreactor: High filtration resistance of gel layer and its underlying mechanism. Water Res. 2016, 102, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.J.; Guo, W.S.; Huu, H.N.; Zhang, H.W.; Wang, J.; Li, J.X.; Xia, S.Q.; Wu, Y. Biofouling and control approaches in membrane bioreactors. Bioresour. Technol. 2016, 221, 656–665. [Google Scholar] [CrossRef]
- Vanysacker, L.; Declerck, P.; Bilad, M.R. Biofouling on microfiltration membranes in MBRs: Role of membrane type and microbial community. J. Membr. Sci. 2014, 453, 394–401. [Google Scholar] [CrossRef]
- Zhang, H.F.; Sun, B.S.; Zhao, X.H. Effects of soluble microbial product on the performance of submerged membrane bioreactor. Environ. Sci. 2008, 29, 82–86. [Google Scholar]
- Dukwoo, J.; Youngo, K.; Saeedreza, H.; Yoo, K.; Hoek, E.M.V.; Jeonghwan, K. Biologically induced mineralization in anaerobic membrane bioreactors: Assessment of membrane scaling mechanisms in a long-term pilot study. J. Membr. Sci. 2017, 543, 342–350. [Google Scholar]
- Ji, L.; Zhou, J.T.; Zhang, X.H. Influence of influent composition on membrane fouling in membrane bioreactors. Environ. Sci. 2007, 28, 18–23. [Google Scholar]
- Hwang, B.K.; Lee, W.N.; Yeon, K.M.; Park, C.H.; Chang, I.S.; Drews, A.; Kraume, M. Correlating TMP increases with microbial characteristics in the bio-cake on the membrane surface in a membrane bioreactor. Environ. Sci. Technol. 2008, 42, 3963–3968. [Google Scholar] [CrossRef]
- Lin, H.J.; Lu, X.F.; Duan, W. Filtration characteristics and mechanism of membrane fouling in a membrane bioreactor for municipal wastewater treatment. Environ. Sci. 2006, 27, 2511–2517. [Google Scholar]
- Deng, L.J.; Guo, W.S.; Huu, H.N.; Du, B.; Wei, Q.; Ngoc, H.T.; Nguyen, C.N.; Chen, S.S.; Li, J.X. Effects of hydraulic retention time and bioflocculant addition on membrane fouling in a sponge-submerged membrane bioreactor. Bioresour. Technol. 2016, 210, 11–17. [Google Scholar] [CrossRef]
- Yu, Q.H.; Chi, L.N.; Zhou, W.L. Overview of forward osmosis membrane separation technology: Research and its application to water treatment. Environ. Sci. Technol. 2010, 33, 117–122. [Google Scholar]
- Park, S.; Yeon, K.M.; Moon, S.; Kim, J.O. Enhancement of operating flux in a membrane bio-reactor coupled with a mechanical sieve unit. Chemosphere 2018, 191, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Wang, Y.; Huang, X. Experimental study on economic aeration of integrated membrane bioreactor. Membr. Sci. Technol. 2004, 24, 11–15. [Google Scholar]
- Huang, B.C.; Guan, Y.F.; Chen, W.; Yu, H.Q. Membrane fouling characteristics and mitigation in a coagulation–assisted microfiltration process for municipal wastewater pretreatment. Water Res. 2017, 123, 216–223. [Google Scholar] [CrossRef]
- Besha, A.T.; Gebreyohannes, A.Y.; Ashu, T.R. Removal of emerging micropollutants by activated sludge process and membrane bioreactors and the effects of micropollutants on membrane fouling: A review. J. Environ. Chem. Eng. 2017, 5, 2395–2414. [Google Scholar] [CrossRef]
- Wu, H.Y. Study on the Mechanism of Microbial Products in MBR on Membrane Fouling. Master’s Thesis, Chinese Research Academy of Environmental Sciences, Beijing, China, 2012. [Google Scholar]
- Chin, H.N.; Zainura, Z.N.; Noor, S.A.M. Green technology in wastewater treatment technologies: Integration of membrane bioreactor with various wastewater treatment systems. Chem. Eng. J. 2016, 283, 582–594. [Google Scholar]
- Long, X.Y.; Long, T.R.; Tang, R. SRT on components and surface characters of extracellula polymeric substances. China Water Wastewater 2008, 24, 1–6. [Google Scholar]
- Zhao, J.; Xu, G.T.; Qin, Z. Composing of extracellular polymeric substances and its effect on sludge characteristics. Safety Environ. Eng. 2008, 15, 66–69. [Google Scholar]
- Tao, L.J.; Li, X.F.; Wang, X.H. Effects of sludge retention time on sludge characteristics in membrane bioreactors. Chinese J. Environ. Eng. 2012, 6, 719–724. [Google Scholar]
- Recep, K.D.; Aurelie, G.; Barry, H.; Frank, P.Z.; Jules, B.L. Implications of changes in solids retention time on long term evolution of sludge filterability in anaerobic membrane bioreactors treating high strength industrial wastewater. Water Res. 2014, 59, 11–22. [Google Scholar]
- Tian, Y.; Li, H.; Li, L.P. In-situ integration of microbial fuel cell with hollow-fiber membrane bioreactor for wastewater treatment and membrane fouling mitigation. Biosens. Bioelectron. 2015, 64, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Alturki, A.; McDonald, J.; Khan, S.J.; Hai, F.L.; Price, W.E.; Nghiem, L.D. Performance of a novel osmotic membrane bioreactor (OMBR) system: Flux stability and removal of trace organics. Bioresour. Technol. 2012, 113, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Aslam, M.; McCarty, P.L.; Shin, C.; Bae, J.; Kim, J. Low energy single-staged anaerobic fluidized bed ceramic membrane bioreactor (AFCMBR) for wastewater treatment. Bioresour. Technol. 2017, 240, 33–41. [Google Scholar] [CrossRef]
- Jin, Z.; Meng, F.; Gong, H.; Wang, C.; Wang, K. Improved low-carbon-consuming fouling control in long-term membrane- based wastewater pre-concentration: The role of enhanced coagulation process and air backflushing in sustainable wastewater treatment. Membr. Sci. 2017, 529, 252–262. [Google Scholar] [CrossRef]
- Ali, S.M.; Kim, J.E.; Phuntsho, S.; Jang, A.; Choi, J.Y.; Shon, H.K. Forward osmosis system analysis for optimum design and operating conditions. Water Res. 2018, 145, 429–441. [Google Scholar] [CrossRef]
- Kim, S.R.; Lee, K.B.; Kim, J.E.; Won, Y.J.; Yeon, K.M. Macroencapsulation of quorum quenching bacteria by polymeric membrane layer and its application to MBR for biofouling control. J. Membr. Sci. 2015, 473, 109–117. [Google Scholar] [CrossRef]
- Hai, F.I.; Riley, T.; Shawkat, S.; Magram, S.F.; Yamamoto, K. Removal of pathogens by membrane bioreactors: A review of the mechanisms, influencing factors and reduction in chemical disinfectant dosing. Water 2014, 6, 3603–3630. [Google Scholar] [CrossRef]
- Arevalo, J.; Ruiz, L.M.; P erez, J. Effect of temperature on membrane bioreactor performance working with high hydraulic and sludge retention time. Biochem. Eng. J. 2014, 88, 42–49. [Google Scholar] [CrossRef]
- Charfi, A.; Aslam, M.; Lesage, G.; Heran, M.; Kim, J. Macroscopic approach to develop fouling model under GAC fluidization in anaerobic fluidized bed membrane bioreactor. J. Ind. Eng. Chem. 2017, 49, 219–229. [Google Scholar] [CrossRef]
- Liu, Y.L.; Xie, Y.M.; Wang, X.X. fouling analysis and chemical cleaning of reverse osmosis membrane in waste water reuse project of steel plant. Clean. World 2019, 35, 8–10. [Google Scholar]
- Achilli, A.; Cath, T.Y.; Marchand, E.A.; Childress, A.E. The forward osmosis membrane bioreactor: A low fouling alternative to MBR processes. Desalination 2009, 239, 10–21. [Google Scholar] [CrossRef]
- Wang, Z.W.; Wu, Z.C.; Gu, G.W. Study on operation mode of SMBR process for treatment of wastewater. Environ. Eng. 2005, 23, 7–9. [Google Scholar]
- Xie, Y.H.; Zhu, T.; Xu, C.H. Membrane cleaning method in metal membrane bioreactor. Chem. Eng. 2010, 38, 190–193. [Google Scholar]
- Hong, Y.; Xiao, P.; Dong, W. Membrane fouling and chemical cleaning for wastewater reclamation using submerged ultrafiltration membrane. Chin. J. Environ. Eng. 2016, 10, 2495–2500. [Google Scholar]
- Guo, W.S.; Ngo, H.H.; Li, J.X. A mini-review on membrane fouling. Bioresour. Technol. 2017, 122, 27–34. [Google Scholar] [CrossRef]
- Wang, Z.W.; Wu, Z.C. A review of membrane fouling in MBRs: Characteristics and role of sludge cake formed on membrane surfaces. Sep. Sci. Technol. 2009, 44, 3571–3596. [Google Scholar] [CrossRef]
- Bell, E.A.; Holloway, R.W.; Cath, T.Y. Evaluation of forward osmosis membrane performance and fouling during long-term osmotic membrane bioreactor study. J. Membr. Sci. 2016, 517, 1–13. [Google Scholar] [CrossRef]
- Chen, C.H.; Fu, Y.; Gao, D.W. Membrane biofouling process correlated to the microbial community succession in an A/O MBR. Bioresour. Technol. 2015, 197, 185–192. [Google Scholar] [CrossRef]
- Abdessemed, D. Treatment of primary effluent by coagulation-adsorption-ultrafiltration for reuse. Desalination 2002, 152, 367–373. [Google Scholar] [CrossRef]
- Zinadini, S.; Vatanpour, V.; Zinatizadeh, A.A.; Rahimi, M.; Rahimi, Z.; Kian, M. Preparation and characterization of antifouling graphene oxide/polyethersulfone ultrafiltration membrane: Application in MBR for dairy wastewater treatment. J. Water Process Eng. 2015, 7, 280–294. [Google Scholar] [CrossRef]
- Zhang, J.L.; Cao, Z.P.; Zhang, H.W. Effects of sludge retention time (SRT) on the characteristics of membrane bioreactor (MBR). Environ. Sci. 2008, 29, 2788–2793. [Google Scholar]
- Wang, Z.W.; Ma, J.X.; Tang, C.Y.Y.; Kimura, K.; Wang, Q.Y.; Han, X.M. Membrane cleaning in membrane bioreactors: A review. J. Membr. Sci. 2014, 468, 276–307. [Google Scholar] [CrossRef]
- Fu, C.; Yue, X.D.; Shi, X.Q.; Ng, K.K.; Ng, H.Y. Membrane fouling between a membrane bioreactor and a moving bed membrane bioreactor: Effects of solids retention time. Chem. Eng. J. 2017, 309, 397–408. [Google Scholar] [CrossRef]
- Li, S.F.; Cui, C.W.; Huang, J. Effect of extracellular polymeric substances on membrane fouling of membrane bioreactor. J. Harbin Inst. Technol. 2007, 39, 266–269. [Google Scholar]
- Shen, L.G.; Lei, Q.; Chen, J.R.; Hong, H.C.; He, Y.M.; Lin, H.J. Membrane fouling in a submerged membrane bioreactor: Impacts of floc size. Chem. Eng. J. 2015, 269, 328–334. [Google Scholar] [CrossRef]
- Hu, Y.S.; Wang, X.C.; Yu, Z.Z.; Ngo, H.H.; Sun, Q.Y.; Zhang, Q.H. New insight into fouling behavior and foulants accumulation property of cake sludge in a full-scale membrane bioreactor. J. Membr. Sci. 2016, 510, 10–17. [Google Scholar] [CrossRef]
- Wang, L.L.; Song, W.C. Study on chemical cleaning technology of membrane bioreactor. Water Sci. Eng. Technol. 2018, 1, 68–70. [Google Scholar]
- Oh, K.S.; Poh, P.E.; Chong, M.N.; Chan, E.S.; Lau, E.V.; Saint, C.P. Bathroom greywater recycling using polyelectrolyte-complex bilayer membrane: Advanced study of membrane structure and treatment efficiency. Carbohydr. Polym. 2016, 148, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Li, S.F.; Gao, Y. Effect of powdered activated carbon on the sludge mixed liquor characteristics and membrane fouling of MBR. Environ. Sci. 2012, 32, 508–514. [Google Scholar]
- Samira, A.O.; Alireza, K.; Mahdie, S.; Yasin, O.; Vahid, V. A review on the applications of ultrasonic technology in membrane bioreactors. Ultrason. Sonochem. 2019, 58, 104633. [Google Scholar]
- Laqbaqbi, M.; García-Payo, M.C.; Khayet, M.; EI Kharraz, J.; Chaouch, M. Application of direct contact membrane distillation for textile wastewater treatment and fouling study. Sep. Purif. Technol. 2019, 209, 815–825. [Google Scholar] [CrossRef]
- Tian, Y.; Li, Z.N.; Chen, L. Fouling property and interaction energy with EPS membrane in normal sludge and bulking sludge. Environ. Sci. 2013, 33, 1224–1230. [Google Scholar]
- Berkessa, Y.W.; Yan, B.H.; Li, T.F.; Tan, M.; She, Z.L.; Jegatheesan, V.; Jiang, H.Q.; Zhang, Y. Novel anaerobic membrane bioreactor (AnMBR) design for wastewater treatment at long HRT and high solid concentration. Bioresour. Technol. 2018, 250, 281–289. [Google Scholar] [CrossRef]
- Chen, X.G.; Li, G.; Lin, H.B. Operation performance and membrane fouling of a spiral symmetry stream anaerobic membrane bioreactor supplemented with biogas aeration. J. Membr. Sci. 2017, 539, 206–212. [Google Scholar]
- Wang, Z.Z.; Meng, F.G.; He, X.; Zhou, Z.B.; Huang, L.N.; Liang, S. Optimisation and performance of NaClO-assisted maintenance cleaning for fouling control in membrane bioreactors. Water Res. 2014, 53, 1–11. [Google Scholar] [CrossRef]
- Amine, C.; Eunyoung, P.; Muhammad, A.; Kim, J. Particle-sparged anaerobic membrane bioreactor with fluidized polyethylene terephthalate beads for domestic wastewater treatment: Modelling approach and fouling control. Bioresour. Technol. 2018, 258, 263–269. [Google Scholar]
- Zhou, Z.B.; Meng, F.G.; Lu, H.; Li, Y.; Jia, X.S.; He, X. Simultaneous alkali supplementation and fouling mitigation in membrane bioreactors by on-line NaOH backwashing. J. Membr. Sci. 2014, 457, 120–127. [Google Scholar] [CrossRef]
- Judd, S.J. The status of industrial and municipal effluent treatment with membrane bioreactor technology. Chem. Eng. J. 2016, 305, 37–45. [Google Scholar] [CrossRef]
- Ly, Q.V.; Hu, Y.X.; Li, J.X.; Cho, J.; Hur, J. Characteristics and influencing factors of organic fouling in forward osmosis operation for wastewater applications: A comprehensive review. Environ. Int. 2019, 129, 164–184. [Google Scholar] [CrossRef] [PubMed]
- Mezohegyi, G.; Bilad, M.R.; Vankelecom, I.F.J. Direct wastewater up-concentration by submerged aerated and vibrated membranes. Bioresour. Technol. 2012, 118, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Naidu, G.; Jeong, S.; Choi, Y.; Vigneswaran, S. Membrane distillation for wastewater reverse osmosis concentrate treatment with water reuse potential. J. Membr. Sci. 2017, 524, 565–575. [Google Scholar] [CrossRef]
- Xiong, J.Q.; Yu, S.C.; Hu, Y.S.; Yang, Y.; Wang, X.C.C. Applying a dynamic membrane filtration (DMF) process for domestic wastewater preconcentration: Organics recovery and bioenergy production potential analysis. Sci. Total Environ. 2019, 68, 35–43. [Google Scholar] [CrossRef]
- Meng, F.G.; Zhang, H.M.; Yang, F.L. Effectof filamentous bacteriaon membrane foulingin submerged membrane bioreactor. J. Membr Sci. 2006, 272, 161–168. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, P.; Li, J.; Hou, D.Y.; Wang, J.; Liu, H.J. A hybrid process combining homogeneous catalytic ozonation and membrane distillation for wastewater treatment. Chemosphere 2016, 160, 134–140. [Google Scholar] [CrossRef]
- Wang, L. Gas/liquid membrane contact separation process and its membrane material research. Light Ind. Sci. Technol. 2013, 4, 46–60. [Google Scholar]
- Yan, T.; Ye, Y.Y.; Ma, H.M. A critical review on membrane hybrid system for nutrient recovery from wastewater. Chem. Eng. J. 2018, 348, 143–156. [Google Scholar] [CrossRef]
- Wang, Z.; Lv, Y.W.; Wang, S.M. Preparation and characterization of PVDF/CA blend ultrafiltration membrane. Membr. Sci. Technol. 2002, 22, 4–8. [Google Scholar]
- Sun, Q. Preparation and antifouling property of polyacrylonitrile (PAN)-sulfobetaine copolymer ultrafiltration membrane. Master’s Thesis, Tianjin University, Tian Jin, China, 2007. [Google Scholar]
- Li, B.R.; Meng, M.J.; Cui, Y.H.; Wu, Y.L.; Zhang, Y.L.; Dong, H.J.; Zhu, Z.; Feng, Y.H.; Wu, C.D. Changing conventional blending photocatalytic membranes (BPMs): Focus on improving photocatalytic performance of Fe3O4/g-C3N4/PVDF membranes through magnetically induced freezing casting method. Chem. Eng. J. 2019, 365, 405–414. [Google Scholar] [CrossRef]
- Selina, H.; Majid, E. Direct membrane filtration for wastewater treatment and resource recovery: A review. Sci. Total Environ. 2020, 710, 136375. [Google Scholar]
- Ng, D.Y.F.; Wu, B.; Chen, Y.; Dong, Z.; Wang, R. A novel thin film composite hollow fiber osmotic membrane with one-step prepared dual-layer substrate for sludge thickening. J. Membr. Sci. 2019, 575, 98–108. [Google Scholar] [CrossRef]
- Xu, J.; Tran, T.N.; Lin, H.; Dai, N. Removal of disinfection byproducts in forward osmosis for wastewater recycling. J. Membr. Sci. 2018, 564, 352–360. [Google Scholar] [CrossRef]
- Ye, Z.L.; Ghyselbrecht, K.; Monballiu, A.; Pinoy, L.; Meesschaert, B. Fractionating various nutrient ions for resource recovery from swine wastewater using simultaneous anionic and cationic selective-electrodialysis. Water Res. 2019, 160, 424–434. [Google Scholar] [CrossRef]
- Meng, F.; Chae, S.R.; Shin, H.S. Recent advances in membrane bioreactors: configuration development, pollutant elimination, and sludge reduction. Environ. Eng. Sci. 2011, 29, 139–160. [Google Scholar] [CrossRef]
- Zuthi, M.F.R.; Ngo, H.H.; Guo, W.S. A review towards finding a simplified approach for modelling the kinetics of the soluble microbial products (SMP) in an integrated mathematical model of membrane bioreactor (MBR). Int. Biodeterior. Biodegrad. 2013, 85, 466–473. [Google Scholar] [CrossRef]
- Yoon, Y.; Hwang, Y.; Kwon, M.; Jung, Y.; Hwang, T.M.; Kang, J.W. Application of O-3 and O-3/H2O2 as post-treatment processes for color removal in swine wastewater from a membrane filtration system. J. Ind. Eng. Chem. 2014, 20, 2801–2805. [Google Scholar] [CrossRef]
- Zarebska, A.; Nieto, D.R.; Christensen, K.V.; Norddahl, B. Ammonia recovery from agricultural wastes by membrane distillation: Fouling characterization and mechanism. Water Res. 2014, 56, 1–10. [Google Scholar] [CrossRef]
- Wu, W.Z.; Chen, G.E. Research progress in modification of PVDF membranes. J. Shanghai Inst. Technol. (Nat. Sci.) 2013, 13, 118–127. [Google Scholar]
- Bagastyo, A.Y.; Anggrainy, A.D.; Nindita, C.S. Electrodialytic removal of fluoride and calcium ions to recover phosphate from fertilizer industry wastewater. Sustain. Environ. Res. 2017, 27, 230–237. [Google Scholar] [CrossRef]
- Zhao, X.; Xuan, H.; Chen, Y.; He, C. Preparation and characterization of superior antifouling PVDF membrane with extremely ordered and hydrophilic surface layer. J. Membr. Sci. 2015, 494, 48–56. [Google Scholar] [CrossRef]
- Zhang, Y.C.; Yang, Q.; Chang, Q. Low temperature plasma modification and electrokinetic characteristics of PVDF hollow fiber membrane. Technol. Water Treat. 2011, 37, 57–60. [Google Scholar]
- Zhan, W.; Zhang, J.; Zuo, W. Research progress in hydrophilic and photocatalytic modification of organic polymer membrane. Environ. Sci. Technol. 2017, 40, 114–119. [Google Scholar]
- Wang, L.; Wei, J.F.; Chen, Y. Preparation of novel hydrophilic PVDF hollow fiber membrane. J. Tianjin Polytech. Univ. 2013, 32, 7–10. [Google Scholar]
- Cui, K.L.; Meng, S.Q.; Gao, B.J. Adsorption and separation of toxic anions on polysulfone microfiltration membrane surface grafted with tertiary amine monomer DMAEMA and grafted membrane. Polym. Mater. Sci. Eng. 2017, 33, 137–143. [Google Scholar]
- Wang, A.; Wei, J.F.; Zhao, K.Y. Study on UV Grafting Modification of Polypropylene Film. J. Funct. Mater. 2012, 43, 2851–2854. [Google Scholar]
- Zhao, Y.H.; Wee, K.H.; Bai, R. Highly hydrophilic and low-protein-fouling polypropylene membrane prepared by surface modification with sulfobetaine-based zwitterionic polymer through a combined surface polymerization method. J. Membr. Sci. 2010, 362, 326–333. [Google Scholar] [CrossRef]
- Jin, G.; Wei, J.F.; Wang, A. Study of grafting sodium p-styrene sulfonate onto surface of polypropylene hollow fiber membrane. Mod. Chem. Ind. 2018, 38, 98–101. [Google Scholar]
- Wang, L.; Wei, J.F.; Zhao, K.Y. Preparation and characterization of high-hydrophilic polyhydroxy functional PP hollow fiber membrane. Mater. Lett. 2015, 159, 189–192. [Google Scholar] [CrossRef]
- Hu, M.X.; Yang, Q.; Xu, Z.K. Enhancing the hydrophilicity of polypropylene microporous membranes by the grafting of 2-hydroxyethyl methacrylate via a synergistic effect of photoinitiators. J. Membr. Sci. 2006, 285, 196–205. [Google Scholar] [CrossRef]
- Xiong, C.C.; Li, W.X.; Liu, Y.F. Optimization membrane module structure of column type by CFD. CIESC J. 2017, 68, 4341–4350. [Google Scholar]
- Zavala, M.Á.L.; Pérez, L.B.S.; Reynoso-Cuevas, L.; Funamizu, N. Pre-filtration for enhancing direct membrane filtration of graywater from washing machine discharges. Ecol. Eng. 2014, 64, 116–119. [Google Scholar] [CrossRef]
- Xu, X.J. New spiral membrane module and its application in water treatment. Master’s Thesis, Dalian University of Technology, Dalian, China, 2010. [Google Scholar]
- Benvenuti, T.; Krapf, R.S.; Rodrigues, M.A.S. Recovery of nickel and water from nickel electroplating wastewater by electrodialysis. Sep. Purif. Technol. 2014, 129, 106–112. [Google Scholar] [CrossRef]
- Benvenuti, T.; Siqueira Rodrigues, M.A.; Bernardes, A.M.; Zoppas-Ferreira, J. Closing the loop in the electroplating industry by electrodialysis. J. Clean. Prod. 2017, 155, 130–138. [Google Scholar] [CrossRef]
- Merkel, A.; Ashrafi, A.M.; Ondrušek, M. The use of electrodialysis for recovery of sodium hydroxide from the high alkaline solution as a model of mercerization wastewater. J. Water Process Eng. 2017, 20, 123–129. [Google Scholar] [CrossRef]
- Drews, A. Membrane fouling in membrane bioreactors characterisation, contradictions, cause and cures. J. Membr. Sci. 2010, 363, 1–28. [Google Scholar] [CrossRef]
- El-Abbassi, A.; Kiai, H.; Hafidi, A.; Garcia-Payo, M.C.; Khayet, M. Treatment of olive mill wastewater by membrane distillation using polytetrafluoroethylene membranes. Sep. Purif. Technol. 2012, 98, 55–61. [Google Scholar] [CrossRef]
- Zhang, T.; Li, C.X.; Guo, K. Swirling flow enhanced hollow fiber membrane module structure optimization and shell-side hydrodynamics investigation. CIESC J. 2018, 10, 1–19. [Google Scholar]
- Zhang, J.P.; Zhang, H.M.; Yang, F.L. Optimization design of new folding plate module and its application in MBR. Membr. Sci. Technol. 2015, 35, 72–80. [Google Scholar]
- Damtie, M.M.; Kim, B.; Woo, Y.C.; Choi, J.S. Membrane distillation for industrial wastewater treatment: Studying the effects of membrane parameters on the wetting performance. Chemosphere 2018, 206, 793–801. [Google Scholar] [CrossRef] [PubMed]
- El-Abbassi, A.; Hafidi, A.; García-Payo, M.C.; Khayet, M. Concentration of olive mill wastewater by membrane distillation for polyphenols recovery. Desalination 2009, 245, 670–674. [Google Scholar] [CrossRef]
- Li, Y.Y.; Zhao, Y.H.; Yang, J. Influence of adding zeolite on membrane filtration resistance in MBR and its decolorizing effect. China Water Wastewater 2008, 24, 49–51. [Google Scholar]
- Djouadi Belkada, F.; Kitous, O.; Drouiche, N.; Aoudj, S.; Bouchelaghem, O.; Abdi, N.; Grib, H.; Mameri, N. Electrodialysis for fluoride and nitrate removal from synthesized photovoltaic industry wastewater. Sep. Purif. Technol. 2018, 204, 108–115. [Google Scholar] [CrossRef]
- Wu, J.L.; Chen, F.T.; Huang, X. Using inorganic coagulants to control membrane fouling in a submerged membrane bioreactor. Desalination 2006, 197, 124–136. [Google Scholar] [CrossRef]
- Zhang, J.; Padmasiri, S.I.; Fitch, M. Influence of cleaning frequency and membrane history on fouling in an anaerobic membrane bioreactor. Desalination 2007, 207, 153–166. [Google Scholar] [CrossRef]
- Du, X.; Zhang, Z.; Carlson, K.H.; Lee, J.; Tong, T.Z. Membrane fouling and reusability in membrane distillation of shale oil and gas produced water: Effects of membrane surface wettability. J. Membr. Sci. 2018, 567, 199–208. [Google Scholar] [CrossRef]
- Zhang, G.Q.; Li, P.; Qin, X.Q. Ultrasonic control membrane bioreactor membrane fouling. Low Temp. Build. Technol. 2011, 11, 13–14. [Google Scholar]
- El-Abbassi, A.; Hafidi, A.; Khayet, M. Integrated direct contact membrane distillation for olive mill wastewater treatment. Desalination 2013, 323, 31–38. [Google Scholar] [CrossRef]
- Wu, J.L.; Huang, X. Use of ozonation to mitigate fouling in a long-term membrane bioreactor. Bioresour. Technol. 2010, 101, 6019–6027. [Google Scholar] [CrossRef]
- Dai, J.; Deng, Z.J.; Chen, L.W. Research progress on membrane fouling control technology in membrane bioreactor. Water Treat. Technol. 2013, 39, 10–13. [Google Scholar]
- Kim, J.; Kim, K.; Ye, H.; Lee, E.; Shin, C.; McCarty, P.L.; Bae, J. Anaerobic fluidized bed membrane bioreactor for wastewater treatment. Environ. Sci. Technol. 2010, 45, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.P.; Bae, T.H.; Tak, T.M. Fouling control in activated sludge submerged hollow fiber membrane bioreactors. Desalination 2002, 143, 219–228. [Google Scholar] [CrossRef]
- Jiang, T.; Kennedy, M.D.; Guinzbourg, B.F.; Vanrolleghem, P.A.; Schippers, J.C. Optimising the operation of a MBR pilot plant by quantitative analysis of the membrane fouling mechanism. Water. Sci. Treat. 2005, 51, 19–25. [Google Scholar] [CrossRef]
- Fan, Y.B.; Wang, J.S.; Jiang, Z.C. Optimal backwash cycle for membranes in membrane bioreactors. J. Environ. Sci. 1997, 17, 439–444. [Google Scholar]
- Wen, X.; Sui, P.; Huang, X. Exerting ultrasound to control the membrane fouling in filtration of anaerobic activated sludge-mechanism and membrane damage. Water Sci. Technol. 2008, 57, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Porcellin, J.S. Chemical cleaning of potable water membranes: A review. Sep. Purif. Treat. 2010, 71, 137–143. [Google Scholar] [CrossRef]
- Viet, N.D.; Cho, J.; Yoon, Y.; Jang, A. Enhancing the removal efficiency of osmotic membrane bioreactors: A comprehensive review of influencing parameters and hybrid configurations. Chemosphere 2019, 236, 124363. [Google Scholar] [CrossRef]
- Gao, Y.; Fang, Z.; Liang, P.; Huang, X. Direct concentration of municipal wastewater by forward osmosis and membrane fouling behavior. Bioresour. Technol. 2018, 247, 730–735. [Google Scholar] [CrossRef]
- Bae, J.; Shin, C.; Lee, E. Anaerobic treatment of lowstrength wastewater: A comparison between single and staged anaerobic fluidized bed membrane bioreactors. Bioresour. Technol. 2014, 165, 75–80. [Google Scholar] [CrossRef]
- Crone, B.C.; Garland, J.L.; Sorial, G.A.; Vane, L.M. Significance of dissolved methane in effluents of anaerobically treated low strength wastewater and potential for recovery as an energy product: A review. Water Res. 2016, 104, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Parker, W.; Dagnew, M. Long term performance of membranes in an anaerobic membrane bioreactor treating municipal wastewater. Chemosphere 2016, 144, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Krzeminski, P.; Van der Graaf, J.H.; van Lier, J.B. Specific energy consumption of membrane bioreactor (MBR) for wastewater treatment. Water Sci. Technol. 2012, 65, 380–392. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Chen, J.; Wang, F.; Ding, L.; Hong, H. Feasibility evaluation of submerged anaerobic membrane bioreactor for municipal secondary wastewater treatment. Desalination 2011, 280, 120–126. [Google Scholar] [CrossRef]
- Martin, I.; Pidou, M.; Soares, A.; Judd, S.; Jefferson, B. Modelling the energy demands of aerobic and anaerobic membrane bioreactors for wastewater treatment. Environ. Technol. 2011, 32, 921–932. [Google Scholar] [CrossRef]
- Hong, H.; Zhang, M.; He, Y.; Chen, J.R.; Lin, H.J. Fouling mechanisms of gel layer in a submerged membrane bioreactor. Bioresour. Technol. 2014, 166, 295–302. [Google Scholar] [CrossRef]
- Xu, M.; Wen, X.; Huang, X.; Li, Y.S. Membrane fouling control in an anaerobic membrane bioreactor coupled with online ultrasound equipment for digestion of waste activated sludge. Sep. Sci. Technol. 2010, 45, 941–947. [Google Scholar] [CrossRef]
- Sui, P.; Wen, X.; Huang, X. Feasibility of employing ultrasound for on-line membrane fouling control in an anaerobic membrane bioreactor. Desalination 2008, 219, 203–213. [Google Scholar] [CrossRef]
- Ruiz, L.; Perez, J.; Gómez, A.; Letona, A.; Gomez, M.A. Ultrasonic irradiation for ultrafiltration membrane cleaning in MBR systems: Operational conditions and consequences. Water Sci. Technol. 2017, 75, 802–812. [Google Scholar] [CrossRef]
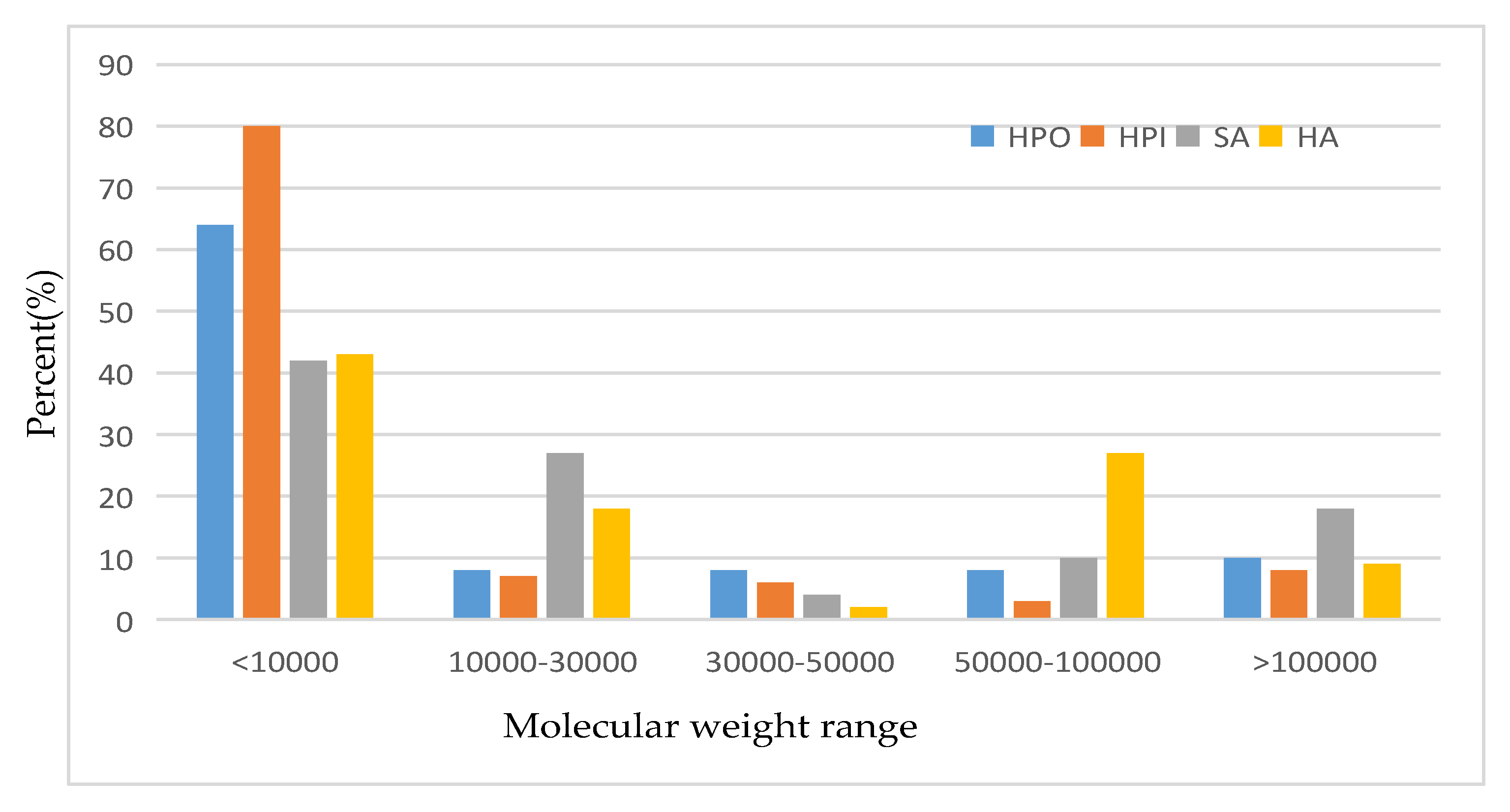
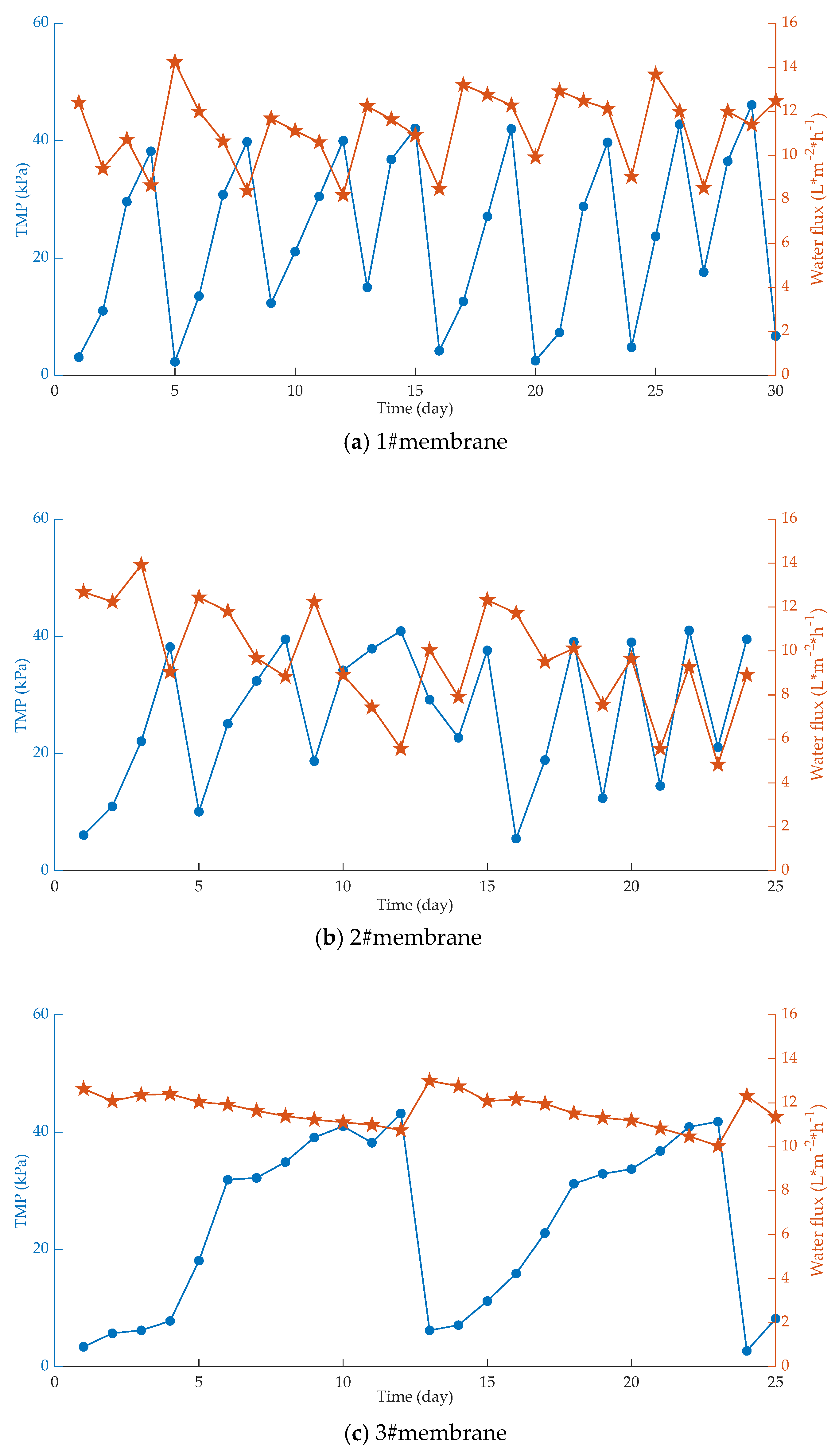
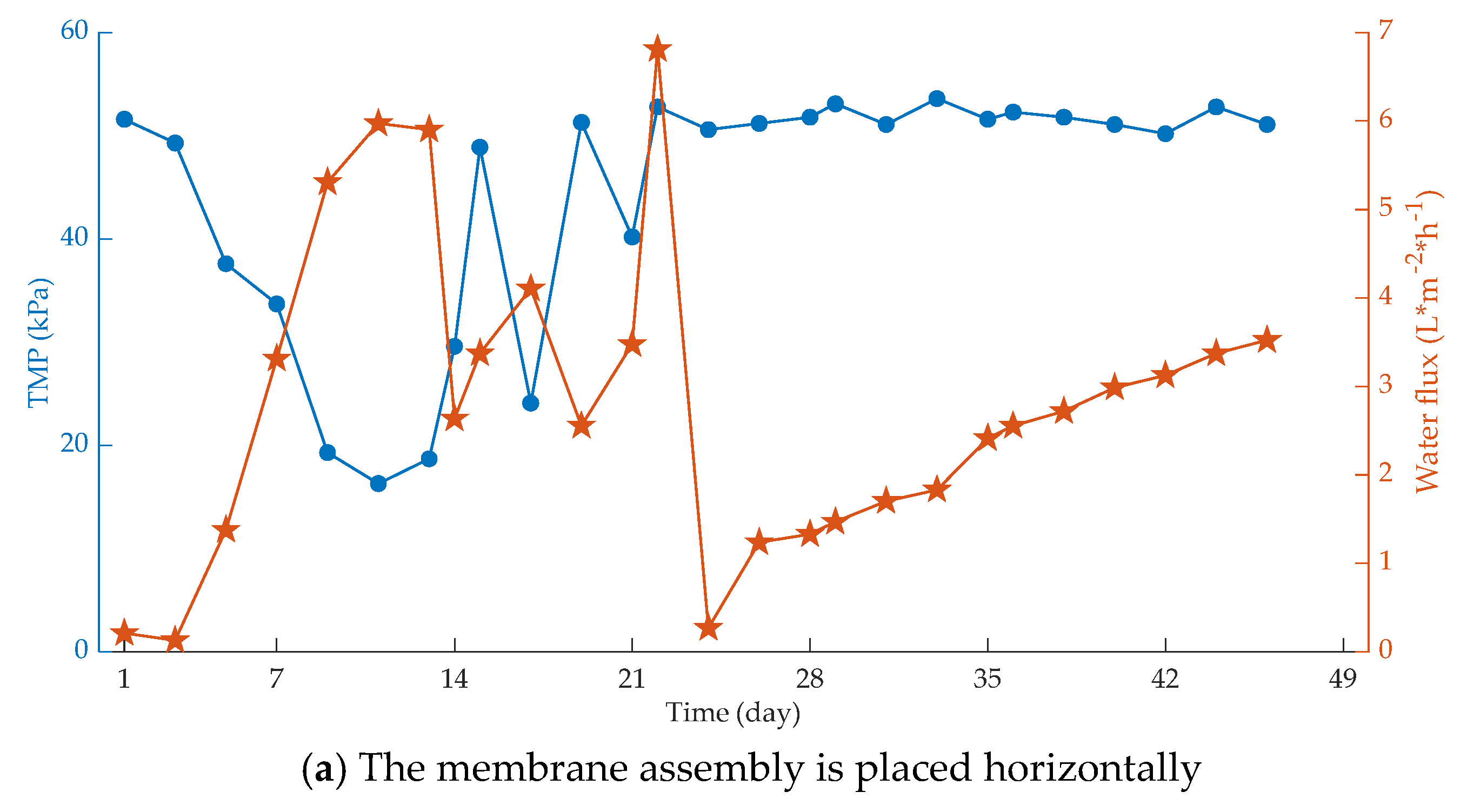
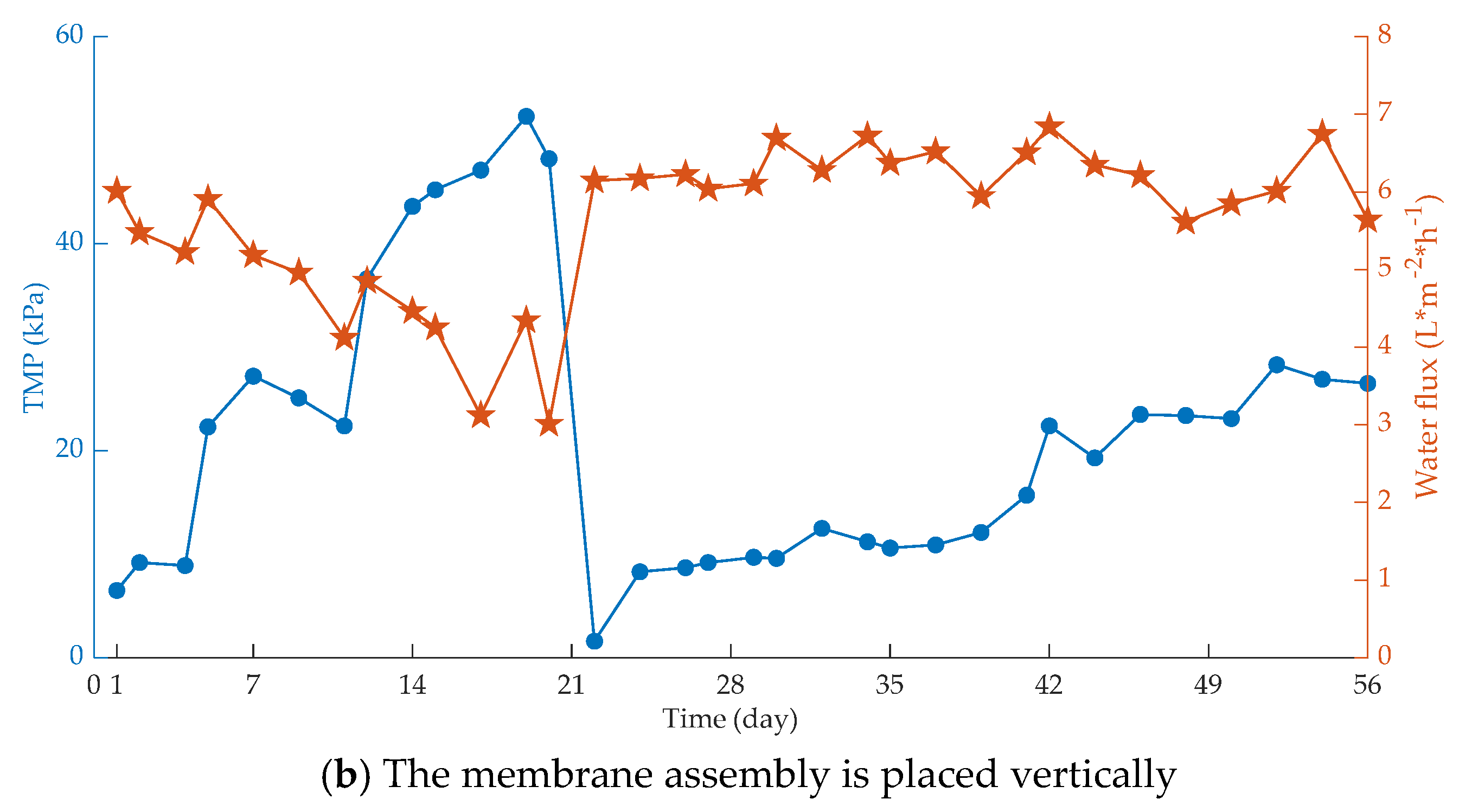
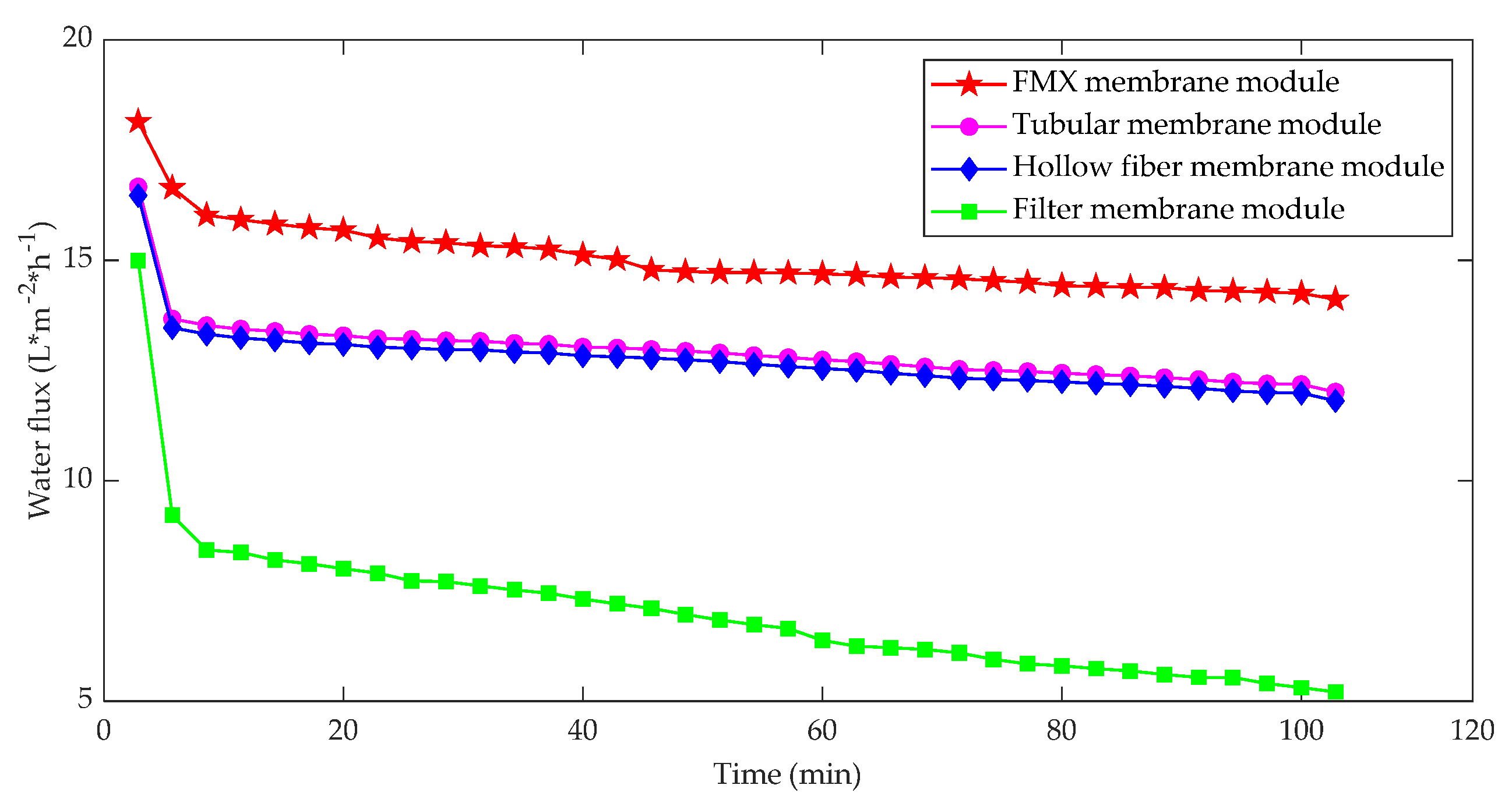
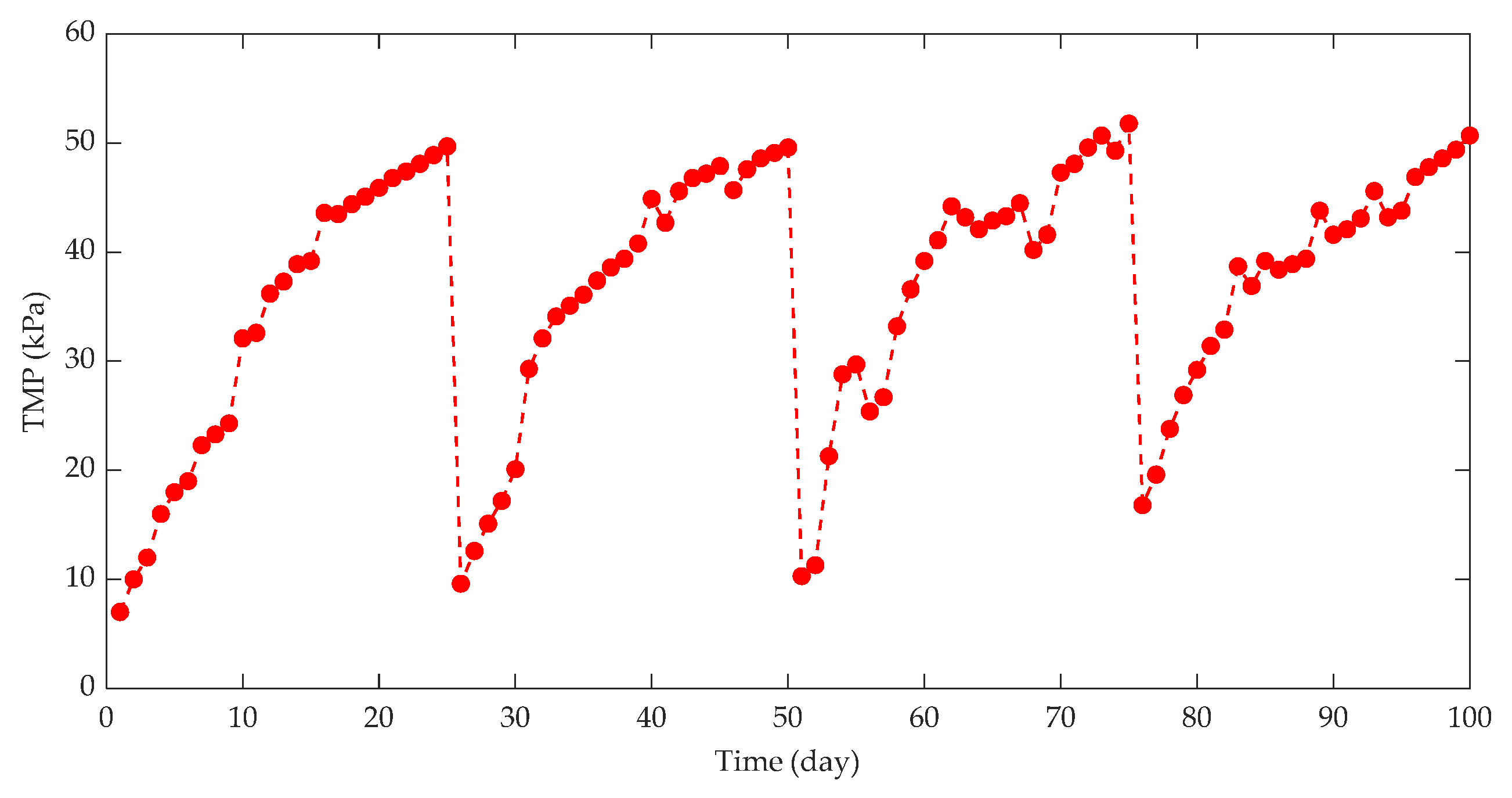
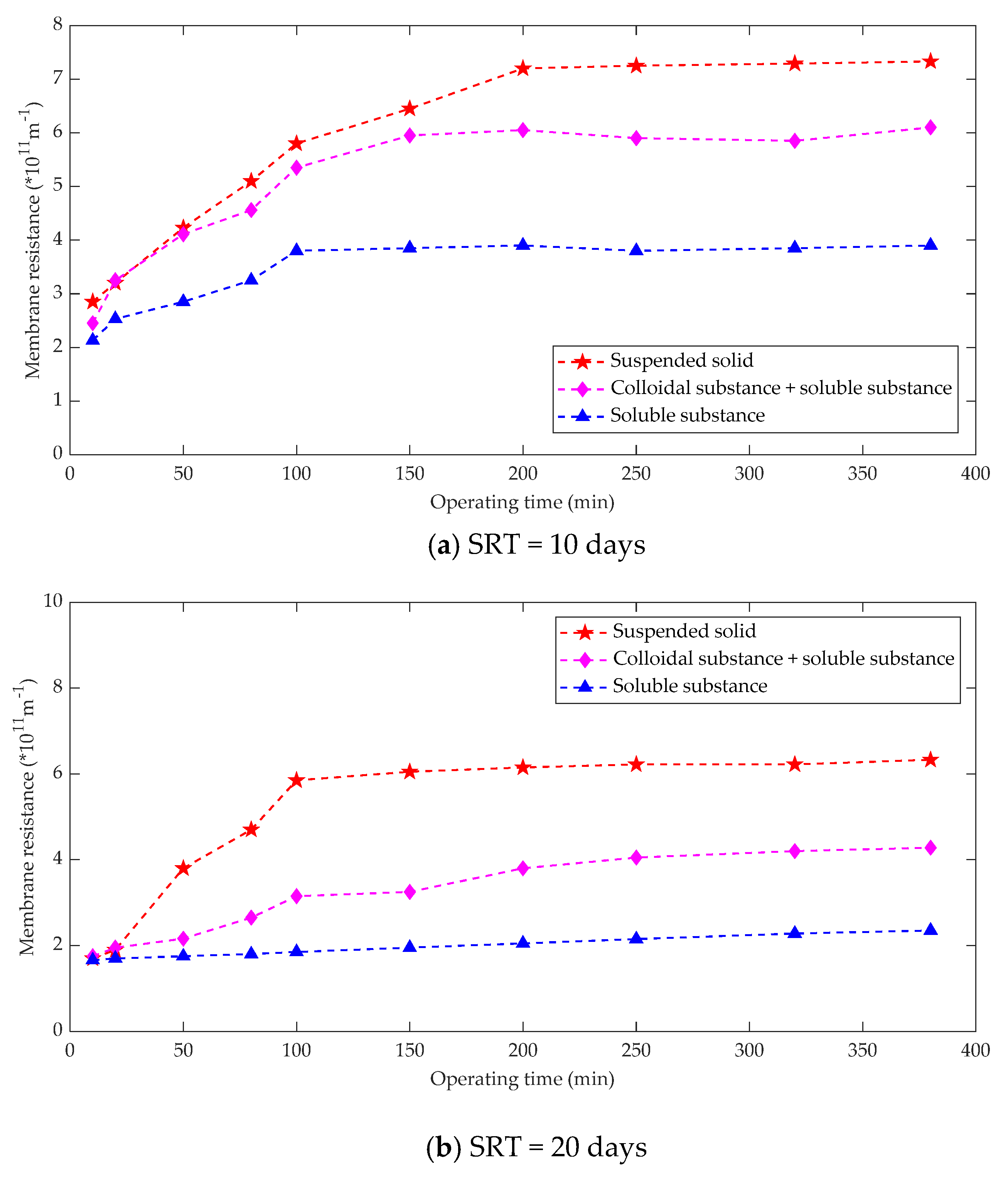
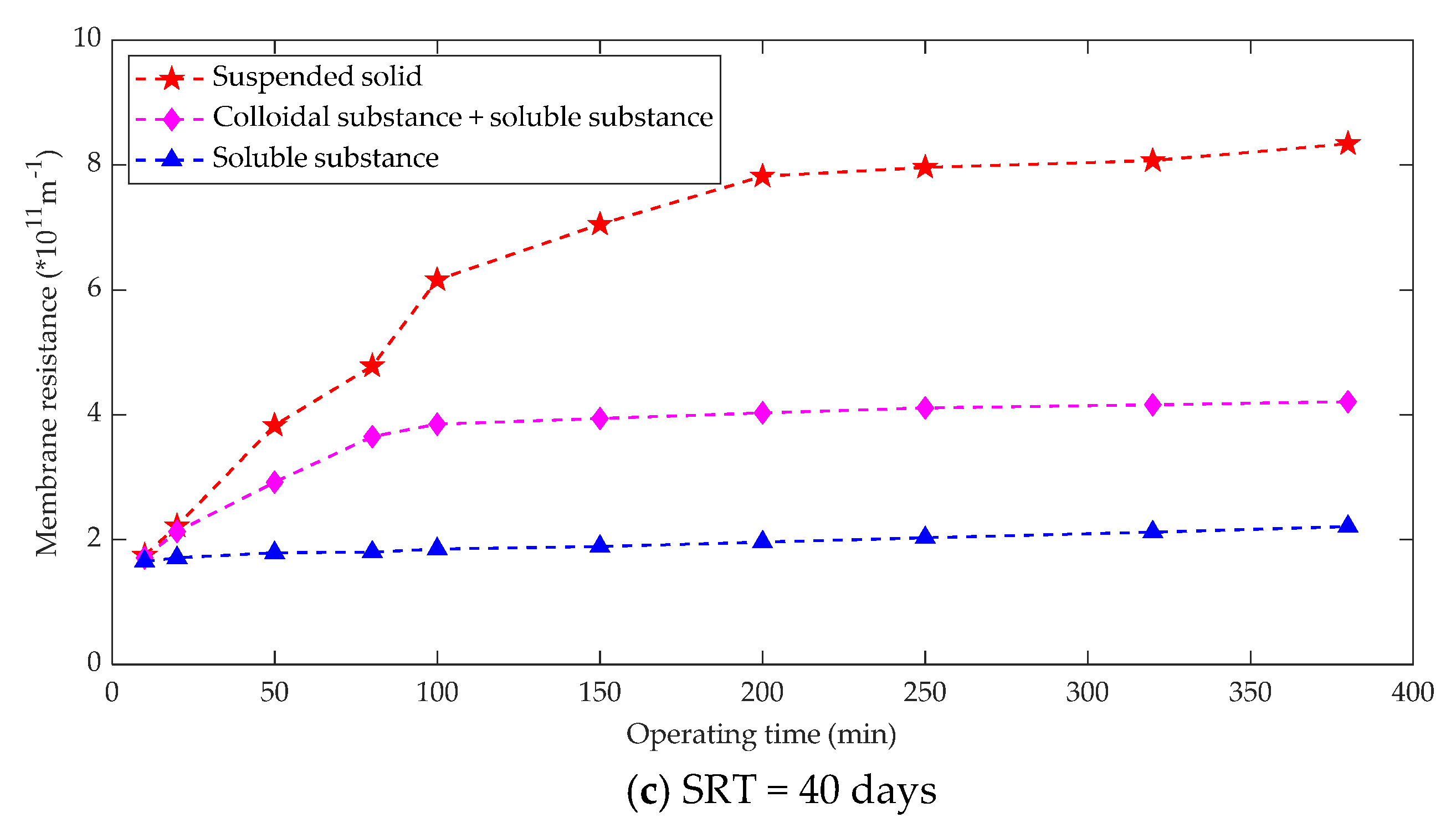
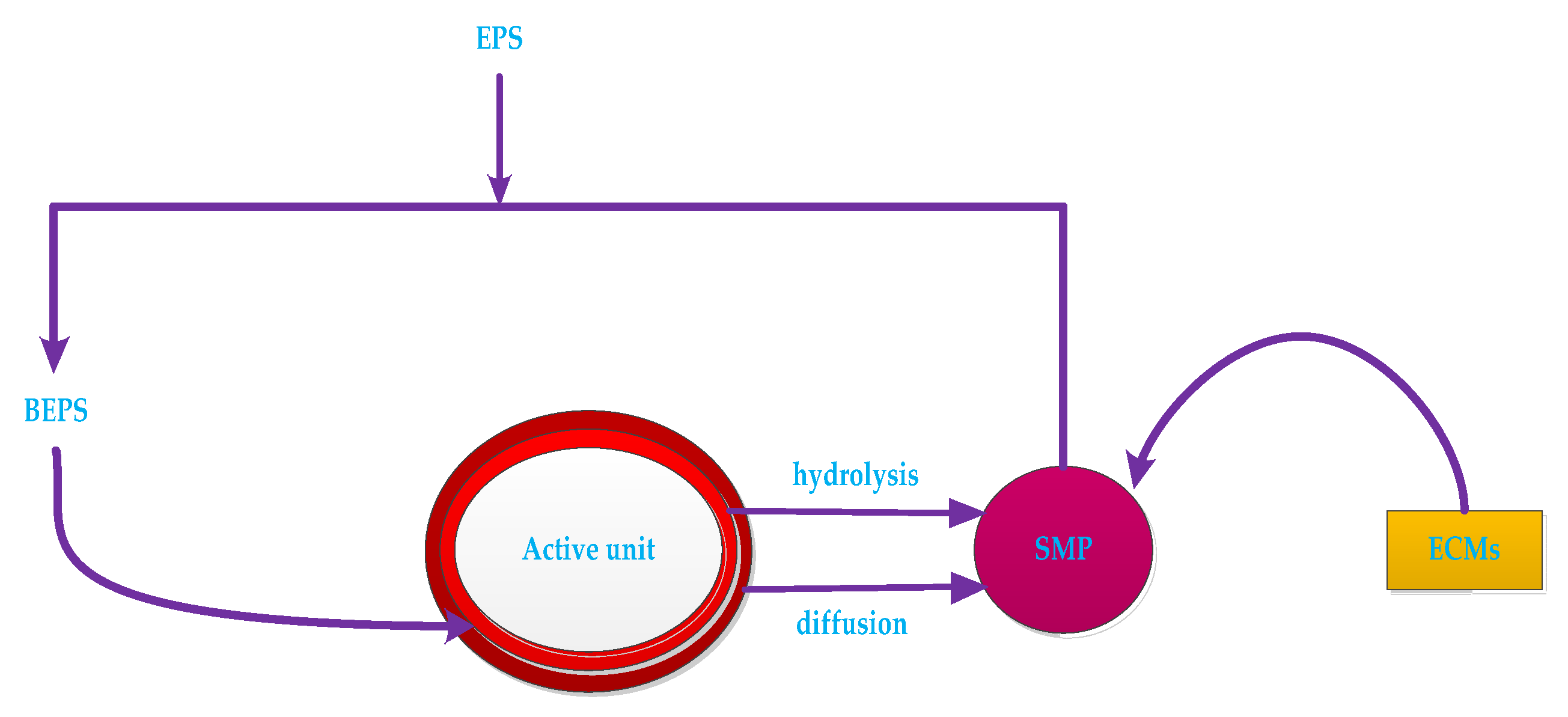
| Fouling Type | Rate of Fouling (Pa.min−1) | Onset of Fouling |
|---|---|---|
| Reversible fouling | 10–100 | 10 min |
| Irreversible fouling (removed by maintenance chemical cleaning) | 1–10 | 1–2 weeks |
| Irreversible fouling (removed by mandatory chemical cleaning) | 0.1–1 | 6–12 months |
| Non-restorable fouling | 0.01–0.1 | A few years |
| Factor | Influence | Type of Wastewater |
|---|---|---|
| Membrane structure properties | The formation of the cake layer can be observed in the organic fouling, and inorganic fouling did not easily cause membrane fouling. | - |
| The protein in the EPS was more than the polysaccharide, and the viscosity of the liquid increased. | Hot white pulp wastewater | |
| Material characteristics | Increased SMP, increased filtration resistance, and deterioration of membrane due to fouling. | Domestic wastewater |
| Supernatant SMP had more protein than polysaccharides, the viscosity increased, and the cake layer was easy to form. | Industrial waste | |
| When SRT increased, SMP and sludge viscosity increased. | Low concentration wastewater | |
| Operating condition | At 30 and 50 d, the activated sludge floc increased, the low fouling rate SRT was too small, the SMP increased, and the fouling accelerated. | Municipal wastewater |
| If it was too large, MLSS, SMP and other microbial products increased. | - | |
| HRT declined, protein substances in SMP increased, and EPS concentration increased. | Low concentration wastewater | |
| HRT decreased, filtration resistance increased, and granular sludge particle size decreased. | Artificial wastewater | |
| Small flocs increased under high temperature conditions, SMP, EPS increase, filter cake layer was easy to form | Evaporator condensate | |
| When the temperature went up, the membrane fouling resistance increased, and the protein content in EPS increased. | Hot pulping press |
| Control Methods | Controlling Factors | Expected Results | Precautions |
|---|---|---|---|
| Modification of membrane material | Improve membrane surface hydrophilicity | Reduce the adsorption of impurities on the membrane surface and membrane pores | The membrane material should be modified according to treatment objectives |
| Optimization of membrane components | Improve membrane surface water conditions | Improve the effect of membrane surface gas flow flushing and decontamination | High mechanical properties for membrane materials |
| Aeration, ultrasound | Remove membrane deposits and improve liquid properties | Gas–liquid flow flushes out membrane deposits to increase activated sludge activity | Excessive aeration or microwave vibration will break up the sludge flocs and increase the fouling of the membrane |
| Add flocculant or adsorbent (PAC), ozone | Improve liquid properties | Improve sludge settling and reduce EPS and SMP in feed liquid | Inorganic flocculants change the pH of the feed, the adsorbent itself may also become a contaminant, and ozone inhibits microbial activity |
| Intermittent suction | Improve film surface detachment properties | Conducive to the membrane surface gas flow flushing with pollutants | Too long stoppage will affect the amount of water produced, too short to achieve the desired results |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, X.; Shi, Y.; Jegatheesan, V.; Haq, I.U. A Review on the Mechanism, Impacts and Control Methods of Membrane Fouling in MBR System. Membranes 2020, 10, 24. https://doi.org/10.3390/membranes10020024
Du X, Shi Y, Jegatheesan V, Haq IU. A Review on the Mechanism, Impacts and Control Methods of Membrane Fouling in MBR System. Membranes. 2020; 10(2):24. https://doi.org/10.3390/membranes10020024
Chicago/Turabian StyleDu, Xianjun, Yaoke Shi, Veeriah Jegatheesan, and Izaz Ul Haq. 2020. "A Review on the Mechanism, Impacts and Control Methods of Membrane Fouling in MBR System" Membranes 10, no. 2: 24. https://doi.org/10.3390/membranes10020024
APA StyleDu, X., Shi, Y., Jegatheesan, V., & Haq, I. U. (2020). A Review on the Mechanism, Impacts and Control Methods of Membrane Fouling in MBR System. Membranes, 10(2), 24. https://doi.org/10.3390/membranes10020024






