Processing Ceramic Proton Conductor Membranes for Use in Steam Electrolysis
Abstract
1. Introduction
2. Materials and Methods
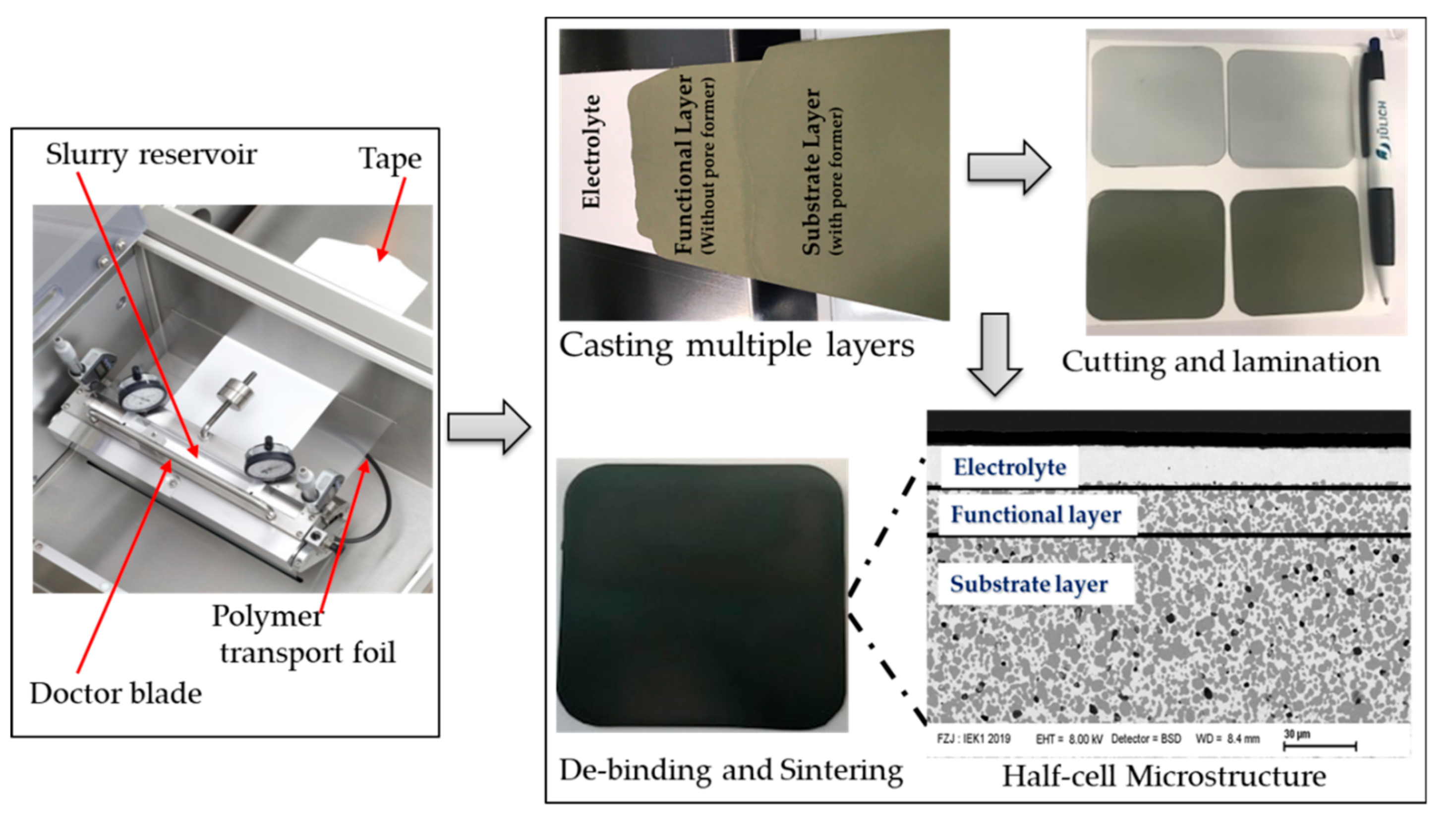
2.1. Fabrication of Electrode-Supported Half-Cells by Sequential Tape-Casting
2.2. Complete Cell Fabrication and Steam Electrolysis Measurements
2.3. Microstructural Observation and Characterization
3. Results and Discussions
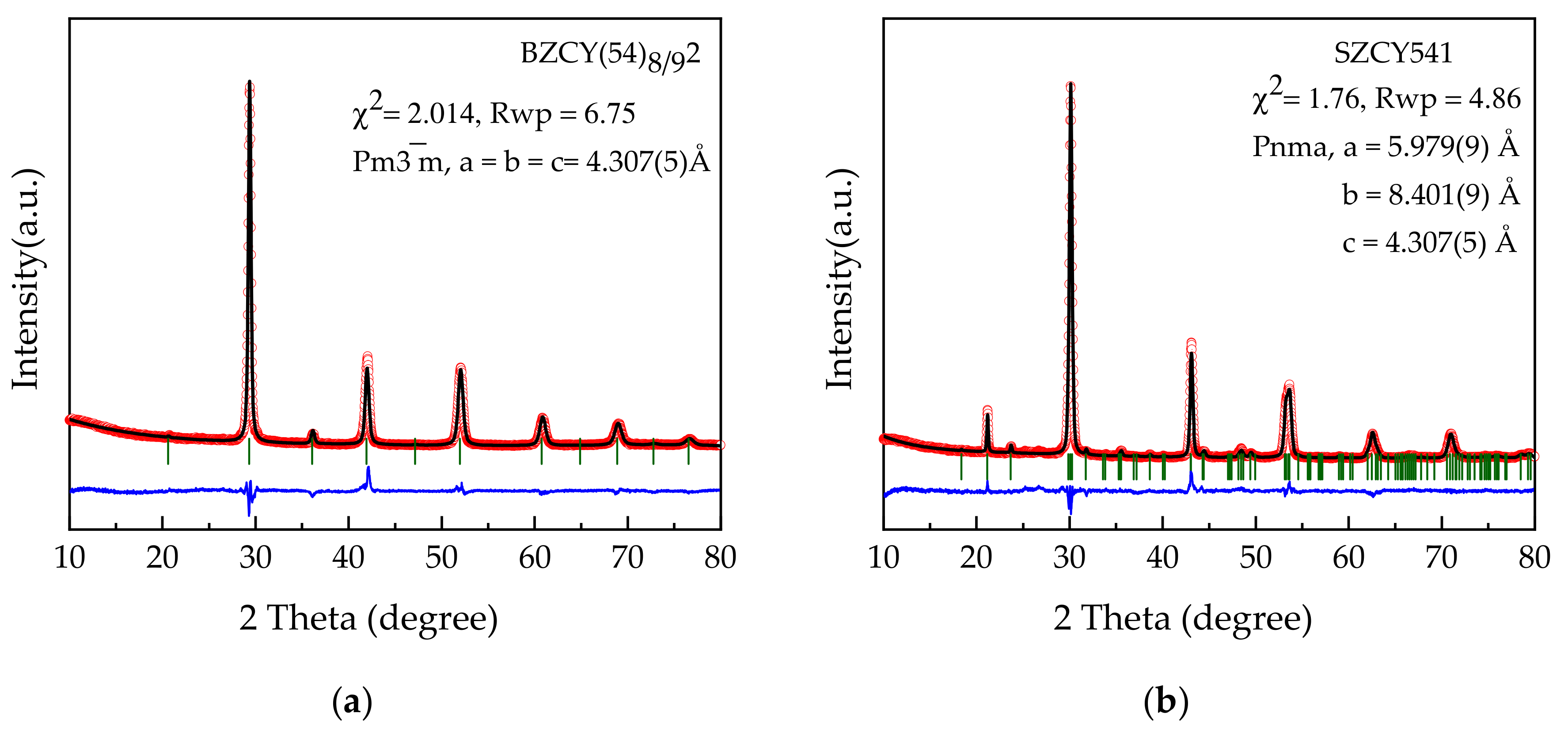
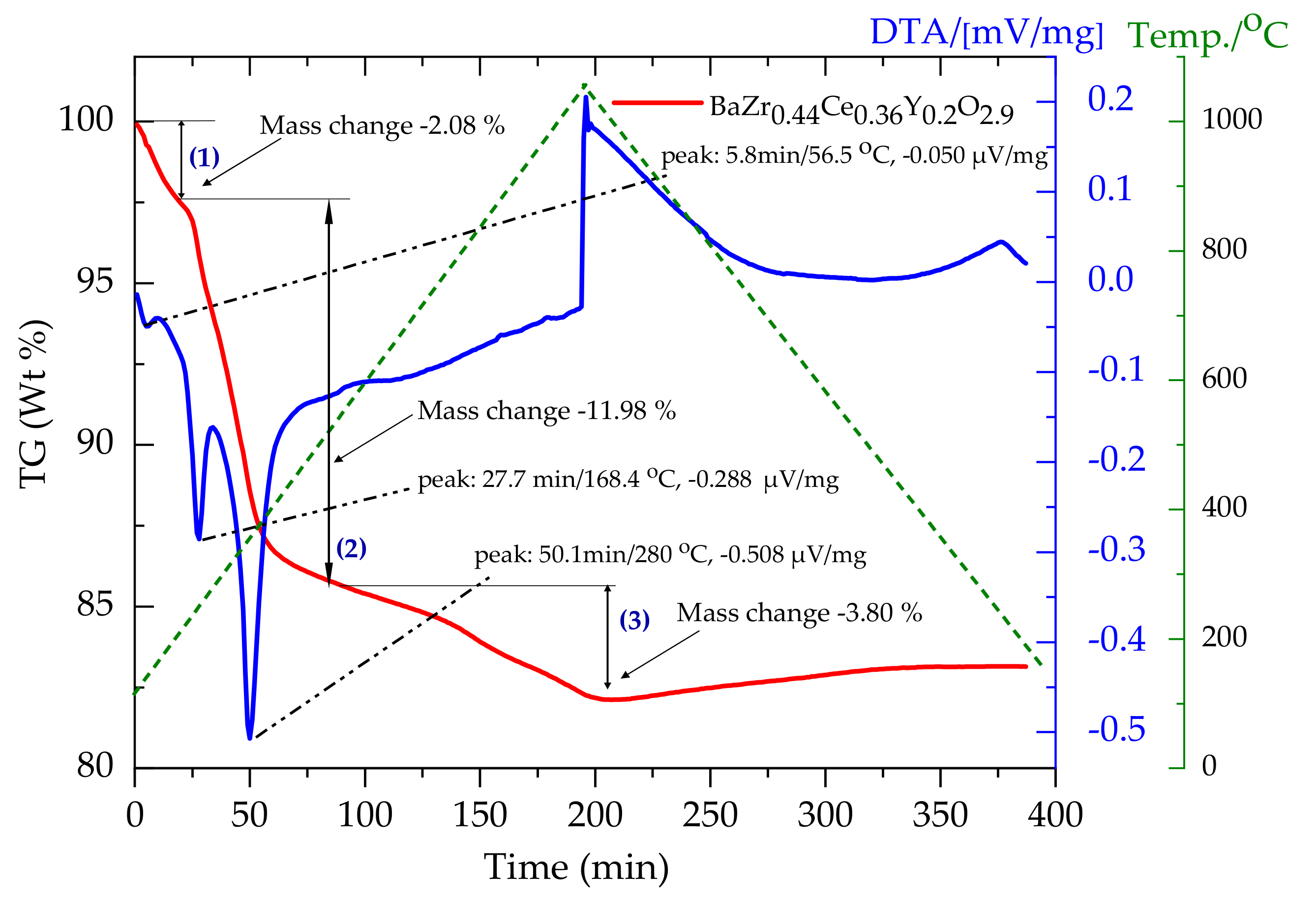
3.1. Phase and Structural Characterization of Powders
3.2. Sequential Tape Casting and Characterization
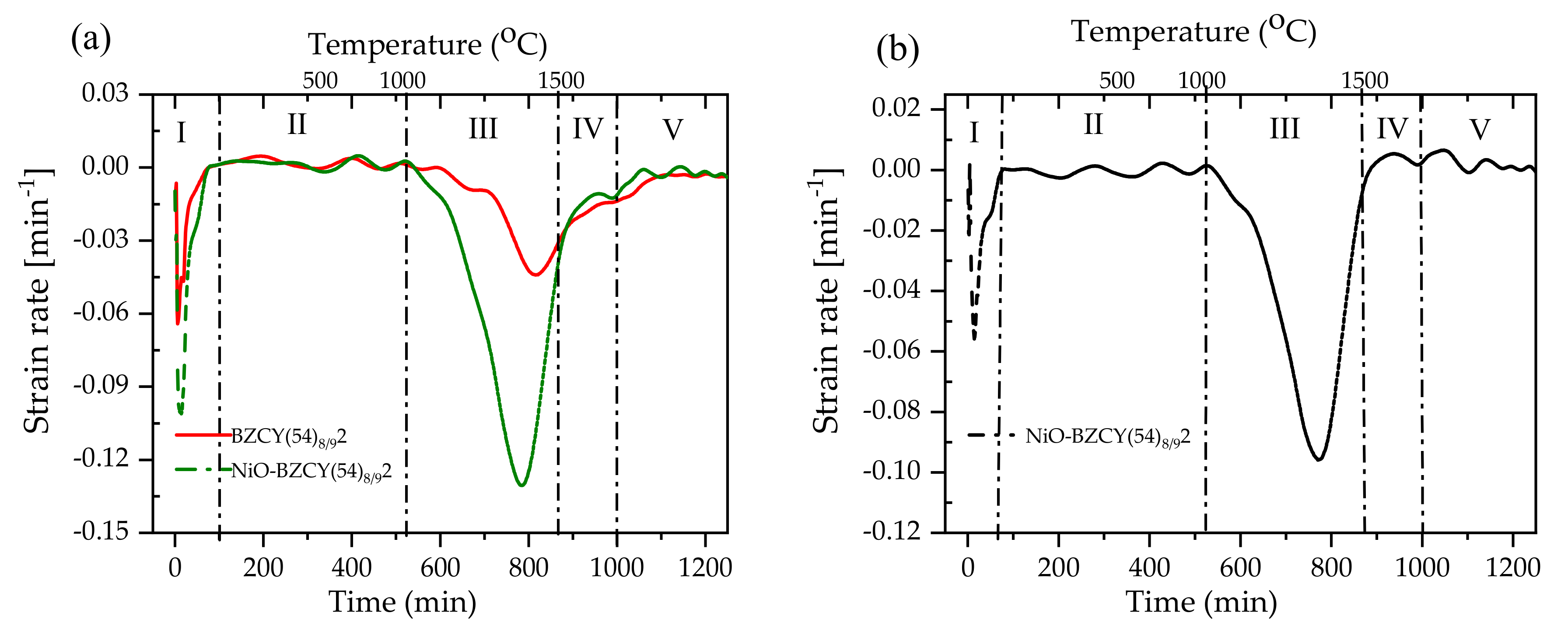
3.3. Sinterability and Bending Behavior of the Half-Cell
3.4. Microstructure and Surface Morphology
3.5. Single-Cell Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Iwahara, H.; Uchida, H.; Maeda, N. High temperature fuel and steam electrolysis cells using proton conductive solid electrolytes. J. Power Sources 1982, 7, 293–301. [Google Scholar] [CrossRef]
- Duan, C.; Kee, R.; Zhu, H.; Sullivan, N.; Zhu, L.; Bian, L.; Jennings, D.; O’Hayre, R. Highly efficient reversible protonic ceramic electrochemical cells for power generation and fuel production. Nat. Energy 2019, 4, 230–240. [Google Scholar] [CrossRef]
- Leonard, K.; Okuyama, Y.; Takamura, Y.; Lee, Y.-S.; Miyazaki, K.; Ivanova, M.E.; Meulenberg, W.A.; Matsumoto, H. Efficient intermediate-temperature steam electrolysis with Y: SrZrO3–SrCeO3 and Y: BaZrO3–BaCeO3 proton conducting perovskites. J. Mater. Chem. A 2018, 6, 19113–19124. [Google Scholar] [CrossRef]
- Choi, S.; Davenport, T.C.; Haile, S.M. Protonic ceramic electrochemical cells for hydrogen production and electricity generation: Exceptional reversibility, stability, and demonstrated faradaic efficiency. Energy Environ. Sci. 2019, 12, 206–215. [Google Scholar] [CrossRef]
- Vøllestad, E.; Strandbakke, R.; Tarach, M.; Catalán-Martínez, D.; Fontaine, M.-L.; Beeaff, D.; Clark, D.R.; Serra, J.M.; Norby, T. Mixed proton and electron conducting double perovskite anodes for stable and efficient tubular proton ceramic electrolysers. Nat. Mater. 2019, 18, 752–759. [Google Scholar] [CrossRef]
- Kreuer, K.D. Proton-Conducting Oxides. Annu. Rev. Mater. Res. 2003, 33, 333–359. [Google Scholar] [CrossRef]
- Leonard, K.; Lee, Y.-S.; Okuyama, Y.; Miyazaki, K.; Matsumoto, H. Influence of dopant levels on the hydration properties of SZCY and BZCY proton conducting ceramics for hydrogen production. Int. J. Hydrog. Energy 2017, 42, 3926–3937. [Google Scholar] [CrossRef]
- Lei, L.; Zhang, J.; Yuan, Z.; Liu, J.; Ni, M.; Chen, F. Progress Report on Proton Conducting Solid Oxide Electrolysis Cells. Adv. Funct. Mater. 2019, 29, 1–17. [Google Scholar] [CrossRef]
- Medvedev, D. Trends in research and development of protonic ceramic electrolysis cells. Int. J. Hydrog. Energy 2019, 44, 26711–26740. [Google Scholar] [CrossRef]
- Grover, C.W. Co-Ionic Conduction in Protonic Ceramics of the Solid Solution, BaCe(x)Zr(y−x)Y(1−y)O3−δ Part I: Fabrication and Microstructure, Adances in Ceramic Synthesis and Characterisation, Processing and Specific Application; Costas Sikalidis, Ed.; In Tech: Rijeka, Croatia, 2018; pp. 479–499. ISBN 978-953-307-505-1. [Google Scholar]
- Fujisaki, T.; Staykov, A.T.; Jing, Y.; Leonard, K.; Aluru, N.R.; Matsumoto, H. Understanding the effect of Ce and Zr on chemical expansion in yttrium doped strontium cerate and zirconate by high temperature X-ray analysis and density functional theory. Solid State Ion. 2019, 333, 1–8. [Google Scholar] [CrossRef]
- Strandbakke, R.; Cherepanov, V.A.; Zuev, A.Y.; Tsvetkov, D.S.; Argirusis, C.; Sourkouni, G.; Prünte, S.; Norby, T. Gd- and Pr-based double perovskite cobaltites as oxygen electrodes for proton ceramic fuel cells and electrolyser cells. Solid State Ion. 2015, 278, 120–132. [Google Scholar] [CrossRef]
- Leonard, K.; Druce, J.; Thoréton, V.; Kilner, J.A.; Matsumoto, H. Exploring mixed proton/electron conducting air electrode materials in protonic electrolysis cell. Solid State Ion. 2018, 319, 218–222. [Google Scholar] [CrossRef]
- Poetzsch, D.; Merkle, R.; Maier, J. Proton conductivity in mixed-conducting BSFZ perovskite from thermogravimetric relaxation. Phys. Chem. Chem. Phys. 2014, 16, 16446–16453. [Google Scholar] [CrossRef] [PubMed]
- Tellez, H.; Druce, J.; Cooper, S.J.; Kilner, J. Double perovskite cathodes for proton-conducting ceramic fuel cells: Are they triple mixed ionic electronic conductors? Sci. Technol. Adv. Mater. 2017, 18, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, A.; Ricote, S.; Sullivan, N.P. Characterization of ionic transport through BaCe 0.2 Zr 0.7 Y 0.1 O 3−δ membranes in galvanic and electrolytic operation. Int. J. Hydrog. Energy 2015, 40, 9278–9286. [Google Scholar] [CrossRef]
- Matsumoto, H.; Sakai, T.; Okuyama, Y. Proton-conducting oxide and applications to hydrogen energy devices. Pure Appl. Chem. 2012, 85, 427–435. [Google Scholar] [CrossRef]
- Gan, Y.; Zhang, J.; Li, Y.; Li, S.; Xie, K.; Irvine, J.T.S. Composite Oxygen Electrode Based on LSCM for Steam Electrolysis in a Proton Conducting Solid Oxide Electrolyzer. J. Electrochem. Soc. 2012, 159, F763–F767. [Google Scholar] [CrossRef]
- Kim, J.; Jun, A.; Gwon, O.; Yoo, S.; Liu, M.; Shin, J.; Lim, T.-H.; Kim, G. Hybrid-solid oxide electrolysis cell: A new strategy for efficient hydrogen production. Nano Energy 2018, 44, 121–126. [Google Scholar] [CrossRef]
- Wu, W.; Ding, H.; Zhang, Y.; Ding, Y.; Katiyar, P.; Majumdar, P.K.; He, T.; Ding, D. 3D Self-Architectured Steam Electrode Enabled Efficient and Durable Hydrogen Production in a Proton-Conducting Solid Oxide Electrolysis Cell at Temperatures Lower Than 600 °C. Adv. Sci. 2018, 5, 1800360. [Google Scholar] [CrossRef]
- Schafbauer, W.; Schulze-Küppers, F.; Baumann, S.; Meulenberg, W.A.; Menzler, N.H.; Buchkremer, H.P.; Stöver, D. Tape Casting as a Multi Purpose Shaping Technology for Different Applications in Energy Issues. Mater. Sci. Forum 2012, 706, 1035–1040. [Google Scholar] [CrossRef]
- Costa, R.; Hafsaoui, J.; De Oliveira, A.P.A.; Grosjean, A.; Caruel, M.; Chesnaud, A.; Thorel, A. Tape casting of proton conducting ceramic material. J. Appl. Electrochem. 2008, 39, 485–495. [Google Scholar] [CrossRef]
- Menzler, N.H.; Malzbender, J.; Schoderböck, P.; Kauert, R.; Buchkremer, H.P. Sequential Tape Casting of Anode-Supported Solid Oxide Fuel Cells. Fuel Cells 2014, 14, 537. [Google Scholar] [CrossRef]
- Fontaine, M.-L.; Larring, Y.; Smith, J.; Raeder, H.; Andersen, Ø.S.; Einarsrud, M.A.; Wiik, K.; Bredesen, R. Shaping of advanced asymmetric structures of proton conducting ceramic materials for SOFC and membrane-based process applications. J. Eur. Ceram. Soc. 2009, 29, 931–935. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, R.; Wang, S.; Wen, T. Fabrication of a large area cathode-supported thin electrolyte film for solid oxide fuel cells via tape casting and co-sintering techniques. Electrochem. Commun. 2009, 11, 842–845. [Google Scholar] [CrossRef]
- Marrony, M.; Ancelin, M.; Lefèvre, G.; Dailly, J. Elaboration of intermediate size planar proton conducting solid oxide cell by wet chemical routes: A way to industrialization. Solid State Ion. 2015, 275, 97–100. [Google Scholar] [CrossRef]
- Dailly, J.; Marrony, M. BCY-based proton conducting ceramic cell: 1000 h of long term testing in fuel cell application. J. Power Sources 2013, 240, 323–327. [Google Scholar] [CrossRef]
- Marrony, M.; Dailly, J. Advanced Proton Conducting Ceramic Cell as Energy Storage Device. ECS Meet. Abstr. 2017, 164, 988. [Google Scholar] [CrossRef]
- Bae, K.; Noh, H.-S.; Jang, D.Y.; Kim, M.; Kim, H.J.; Hong, J.; Lee, J.-H.; Kim, B.-K.; Son, J.-W.; Shim, J.H. Fabrication of NiO-Y:BaZrO3 Composite Anode for Thin Film-Protonic Ceramic Fuel Cells using Tape-Casting. J. Korean Ceram. Soc. 2015, 52, 320–324. [Google Scholar] [CrossRef]
- Orui, H.; Nozawa, K.; Chiba, R.; Komatsu, T.; Watanabe, K.; Sugita, S.; Arai, H.; Arakawa, M. Development of Anode Supported Solid Oxide Fuel Cells using LaNi(Fe)O3 for Cathodes. ECS Trans. 2019, 7, 255–261. [Google Scholar] [CrossRef]
- Deibert, W.; Ivanova, M.E.; Meulenberg, W.A.; Vaßen, R.; Guillon, O. Preparation and sintering behaviour of La5.4WO12-δ asymmetric membranes with optimised microstructure for hydrogen separation. J. Memb. Sci. 2015, 492, 439–451. [Google Scholar] [CrossRef]
- Ivanova, M.E.; Escolástico, S.; Balaguer, M.; Palisaitis, J.; Sohn, Y.J.; Meulenberg, W.A.; Guillon, O.; Mayer, J.; Serra, J.M. Hydrogen separation through tailored dual phase membranes with nominal composition BaCe0.8Eu0.2O3−δ:Ce0.8Y0.2O2-δ at intermediate temperatures. Sci. Rep. 2016, 6, 34773. [Google Scholar] [CrossRef] [PubMed]
- Fedeli, P.; Drago, F.; Schulze-Kuppers, F.; Baumann, S. Asymmetric LSCF Membranes Utilizing Commercial Powders. Materials. 2020, 13, 614. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Küppers, F.; Baumann, S.; Meulenberg, W.A.; Stöver, D.; Buchkremer, H.-P. Manufacturing and performance of advanced supported Ba0.5Sr0.5Co0.8Fe0.2O3−δ (BSCF) oxygen transport membranes. J. Membr. Sci. 2013, 433, 121–125. [Google Scholar] [CrossRef]
- Meulenberg, W.A.; Schulze-Küppers, F.; Deibert, W.; Van Gestel, T.; Baumann, S. Ceramic Membranes: Materials—Components—Potential Applications. Chem. Biol. Eng. Rev. 2019, 6, 198–208. [Google Scholar] [CrossRef]
- Takamura, Y.; Leonard, K.; Luo, A.; Martin, L.W.; Matsumoto, H. Platinum nanoparticle induced nanoionic effects on electrical conduction in strontium cerate and zirconate. J. Solid State Electrochem. 2019, 23, 953–963. [Google Scholar] [CrossRef]
- Gaudon, M.; Menzler, N.H.; Djurado, E.; Buchkremer, H.P. YSZ electrolyte of anode-supported SOFCs prepared from sub micron YSZ powders. J. Mater. Sci. 2005, 40, 3735–3743. [Google Scholar] [CrossRef]
- Ivanova, M.E.; Deibert, W.; Marcano, D.; Escolástico, S.; Mauer, G.; Meulenberg, W.; Bram, M.; Serra, J.; Vaßen, R.; Guillon, O. Lanthanum tungstate membranes for H2 extraction and CO2 utilization: Fabrication strategies based on sequential tape casting and plasma-spray physical vapor deposition. Sep. Purif. Technol. 2019, 219, 100–112. [Google Scholar] [CrossRef]
- Natividad, S.L.; Marotto, V.R.; Walker, L.S.; Pham, D.; Pinc, W.; Corral, E.L. Tape Casting Thin, Continuous, Homogenous, and Flexible Tapes of ZrB2. J. Am. Ceram. Soc. 2011, 94, 2749–2753. [Google Scholar] [CrossRef]
- Lim, K.Y.; Kim, D.H.; Paik, U.; Kim, S.H. Effect of the molecular weight of poly(ethylene glycol) on the plasticization of green sheets composed of ultrafine BaTiO3 particles and poly(vinyl butyral). Mater. Res. Bull. 2003, 38, 1021–1032. [Google Scholar] [CrossRef]
- Shende, R.V.; Lombardo, S.J. Determination of Binder Decomposition Kinetics for Specifying Heating Parameters in Binder Burnout Cycles. J. Am. Ceram. Soc. 2004, 85, 780–786. [Google Scholar] [CrossRef]
- Ren, L.; Luo, X.; Zhou, H. The tape casting process for manufacturing low-temperature co-fired ceramic green sheets: A review. J. Am. Ceram. Soc. 2018, 101, 3874–3889. [Google Scholar] [CrossRef]
- Babilo, P.; Uda, T.; Haile, S.M. Processing of yttrium-doped barium zirconate for high proton conductivity. J. Mater. Res. 2007, 22, 1322–1330. [Google Scholar] [CrossRef]
- Kaiser, A.; Prasad, A.; Foghmoes, S.P.V.; Ramousse, S.; Bonanos, N.; Esposito, V. Sintering process optimization for multi-layer CGO membranes by in situ techniques. J. Eur. Ceram. Soc. 2013, 33, 549–556. [Google Scholar] [CrossRef]
- Green, D.J.; Guillon, O.; Rödel, J. Constrained sintering: A delicate balance of scales. J. Eur. Ceram. Soc. 2008, 28, 1451–1466. [Google Scholar] [CrossRef]
- Cai, P.Z.; Green, D.J.; Messing, G.L. Constrained Densification of Alumina/Zirconia Hybrid Laminates, II: Viscoelastic Stress Computation. J. Am. Ceram. Soc. 2005, 80, 1940–1948. [Google Scholar] [CrossRef]
- Cai, P.Z.; Green, D.J.; Messing, G.L. Constrained Densification of Alumina/Zirconia Hybrid Laminates, I: Experimental Observations of Processing Defects. J. Am. Ceram. Soc. 2005, 80, 1929–1939. [Google Scholar] [CrossRef]
- Ravi, D.; Green, D.J. Sintering stresses and distortion produced by density differences in bi-layer structures. J. Eur. Ceram. Soc. 2006, 26, 17–25. [Google Scholar] [CrossRef]
- Yoo, Y.; Lim, N. Performance and stability of proton conducting solid oxide fuel cells based on yttrium-doped barium cerate-zirconate thin-film electrolyte. J. Power Sources 2013, 229, 48–57. [Google Scholar] [CrossRef]
- Tong, J.; Clark, D.; Bernau, L.; Subramaniyan, A.; O’Hayre, R. Proton-conducting yttrium-doped barium cerate ceramics synthesized by a cost-effective solid-state reactive sintering method. Solid State Ion. 2010, 181, 1486–1498. [Google Scholar] [CrossRef]
- An, H.; Lee, H.W.; Kim, B.K.; Son, J.W.; Yoon, K.J.; Kim, H.; Shin, D.; Ji, H.I.; Lee, J.H. A 5 × 5 cm2 protonic ceramic fuel cell with a power density of 1.3 W cm−2 at 600 °C. Nat. Energy 2018, 3, 870–875. [Google Scholar] [CrossRef]
- Azimova, M.A.; McIntosh, S. On the reversibility of anode supported proton conducting solid oxide cells. Solid State Ion. 2011, 203, 57–61. [Google Scholar] [CrossRef]
- Taniguchi, N.; Hatoh, K.; Niikura, J.; Gamo, T.; Iwahara, H. Proton conductive properties of gadolinium-doped barium cerates at high temperatures. Solid State Ion. 1992, 998–1003. [Google Scholar] [CrossRef]
- Maeda, S.; Shiratori, Y.; Kurashina, D.; Fujisaki, T.; Leonard, K.; Matsumoto, H. Electrochemical reforming of methane using SrZr0.5Ce0.4Y0.1O3-δ proton-conductor cell combined with paper-structured catalyst. Int. J. Hydrog. Energy 2020, 45, 4026–4034. [Google Scholar] [CrossRef]
- Wang, Z.; Mori, M.; Araki, T. Steam electrolysis performance of intermediate-temperature solid oxide electrolysis cell and efficiency of hydrogen production system at 300 Nm3 h−1. Int. J. Hydrog. Energy 2010, 35, 4451–4458. [Google Scholar] [CrossRef]
- Han, D.; Noda, Y.; Onishi, T.; Hatada, N.; Majima, M.; Uda, T. Transport properties of acceptor-doped barium zirconate by electromotive force measurements. Int. J. Hydrog. Energy 2016, 41, 14897–14908. [Google Scholar] [CrossRef]
- Ishihara, T.; Kannou, T. Intermediate temperature steam electrolysis using LaGaO3-based electrolyte. Solid State Ion. 2011, 192, 642–644. [Google Scholar] [CrossRef]







| Powder | Particle Diameter | Specific Surface Area Aspec (m2/g) | Calcination Temperature | ||
|---|---|---|---|---|---|
| d10 | d50 | d90 | |||
| BZCY(54)8/92 purchased | 0.37 | 0.72 | 1.93 | 5.1 | 1200 °C |
| SZCY541 purchased | 0.50 | 0.69 | 0.95 | 3.1 | 1200 °C |
| BZCY(54)8/92 in house | 0.41 | 0.65 | 1.12 | 1.9 | 1300 °C |
| SZCY541 in house | 0.50 | 0.78 | 1.25 | 1.8 | 1300 °C |
| Layer | Blade Gap [μm] | Casting Speed [mm/s] | Drying Time [h] | Green Thickness [μm] | End Fired Thickness [μm] | Investigation |
|---|---|---|---|---|---|---|
| Electrolyte | ||||||
| Electrolyte 1 | 100 | 5 | 6 | 25 | ~20 | TG/Shrinkage |
| Electrolyte 2 | 75 | 5 | 5 | 18.8 | ~15 | Half-cell |
| Electrolyte 3 | 55 | 5 | 5 | 13.4 | ~10 | Half-cell |
| Substrate and functional layer (CFL) | ||||||
| CFL | 40 | 10 | 5 | 10 | ~8 | Half-cell |
| Substrate | 1200 | 2.5 | ~10–12 | 502 | ~390–400 | Half-cell |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leonard, K.; Deibert, W.; Ivanova, M.E.; Meulenberg, W.A.; Ishihara, T.; Matsumoto, H. Processing Ceramic Proton Conductor Membranes for Use in Steam Electrolysis. Membranes 2020, 10, 339. https://doi.org/10.3390/membranes10110339
Leonard K, Deibert W, Ivanova ME, Meulenberg WA, Ishihara T, Matsumoto H. Processing Ceramic Proton Conductor Membranes for Use in Steam Electrolysis. Membranes. 2020; 10(11):339. https://doi.org/10.3390/membranes10110339
Chicago/Turabian StyleLeonard, Kwati, Wendelin Deibert, Mariya E. Ivanova, Wilhelm A. Meulenberg, Tatsumi Ishihara, and Hiroshige Matsumoto. 2020. "Processing Ceramic Proton Conductor Membranes for Use in Steam Electrolysis" Membranes 10, no. 11: 339. https://doi.org/10.3390/membranes10110339
APA StyleLeonard, K., Deibert, W., Ivanova, M. E., Meulenberg, W. A., Ishihara, T., & Matsumoto, H. (2020). Processing Ceramic Proton Conductor Membranes for Use in Steam Electrolysis. Membranes, 10(11), 339. https://doi.org/10.3390/membranes10110339







