Hydrogen Selective SiCH Inorganic–Organic Hybrid/γ-Al2O3 Composite Membranes
Abstract
1. Introduction
2. Experimental Procedures
2.1. Preparation of the Supported Mesoporous γ-Al2O3 Membrane
2.2. Modification of a Supported γ-Al2O3 Membrane with AHPCS-Derived Organic–Inorganic SiCH Hybrid
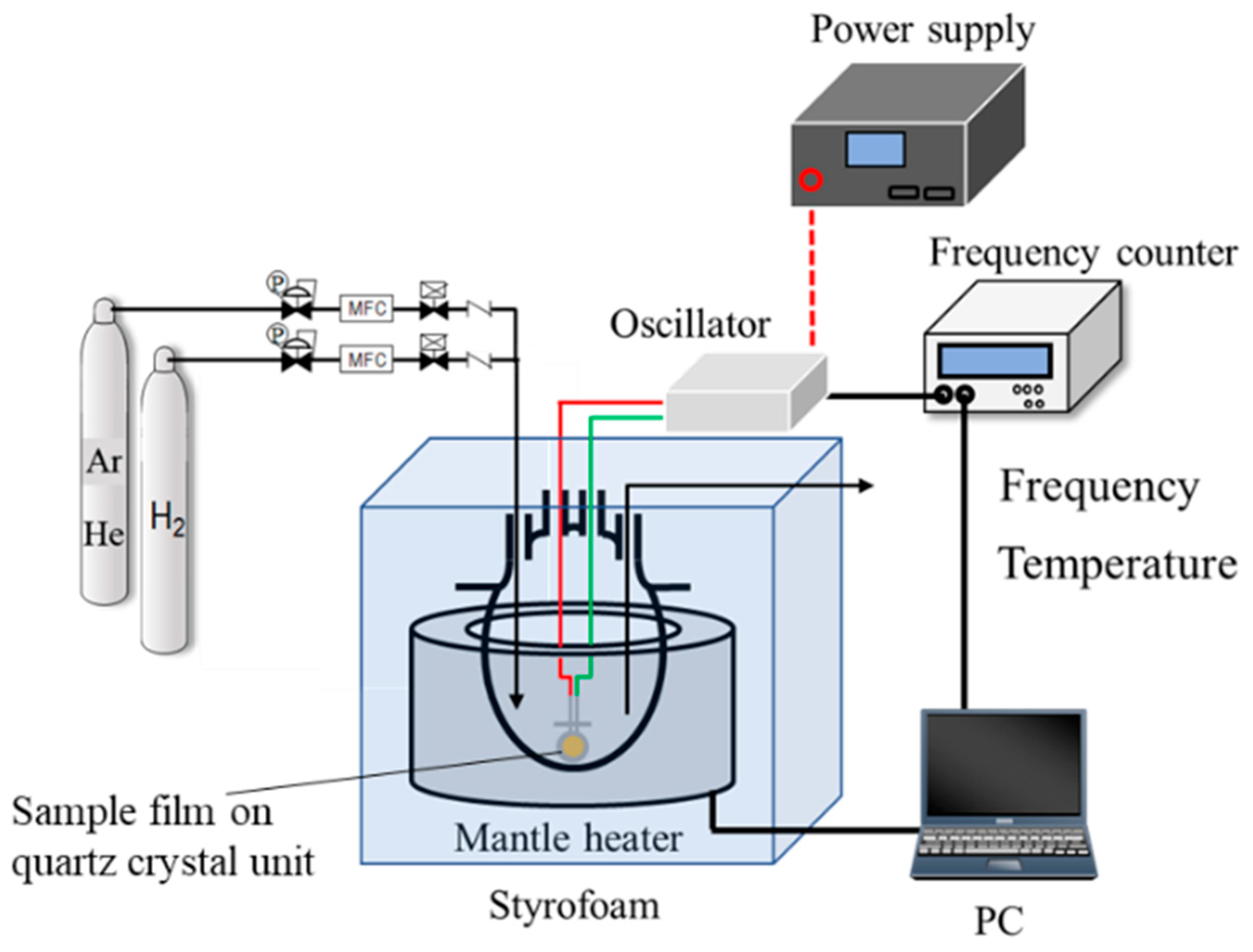
2.3. Characterizations
3. Results and Discussion
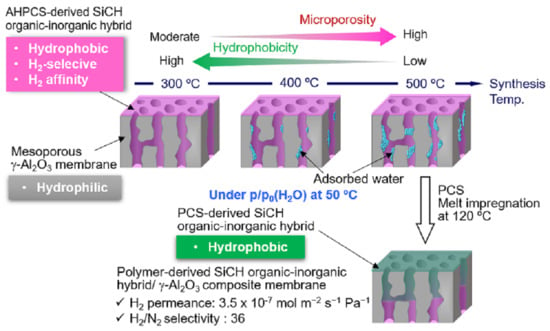
3.1. Heat Treatment Temperatures Selected for Highly Cross-Linked SiCH Organic–Inorganic Hybrid Synthesis
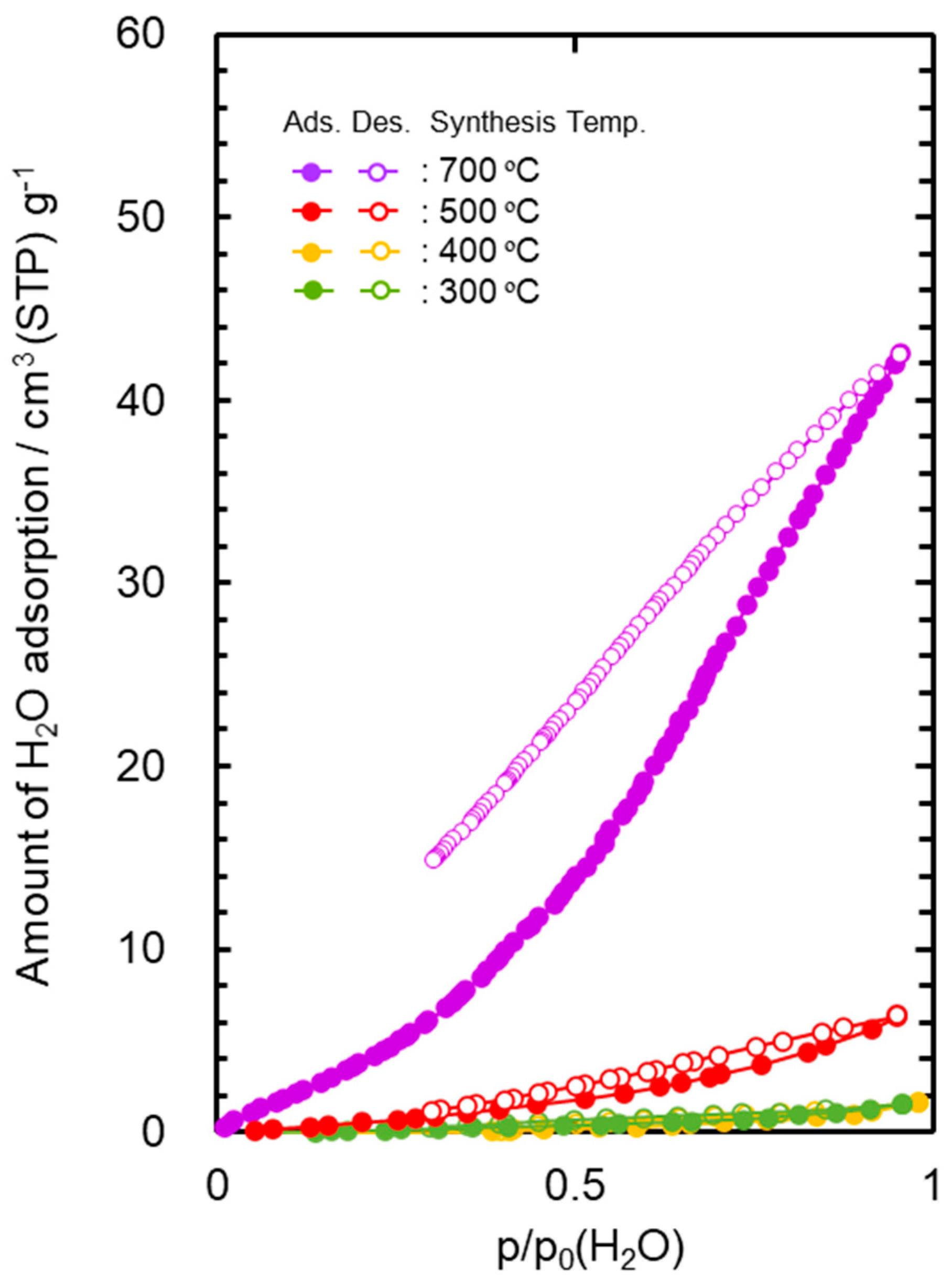
3.2. Hydrophobicity
3.3. Properties of SiCH Hybrid/γ-Al2O3 Composite Membrane
- (1)
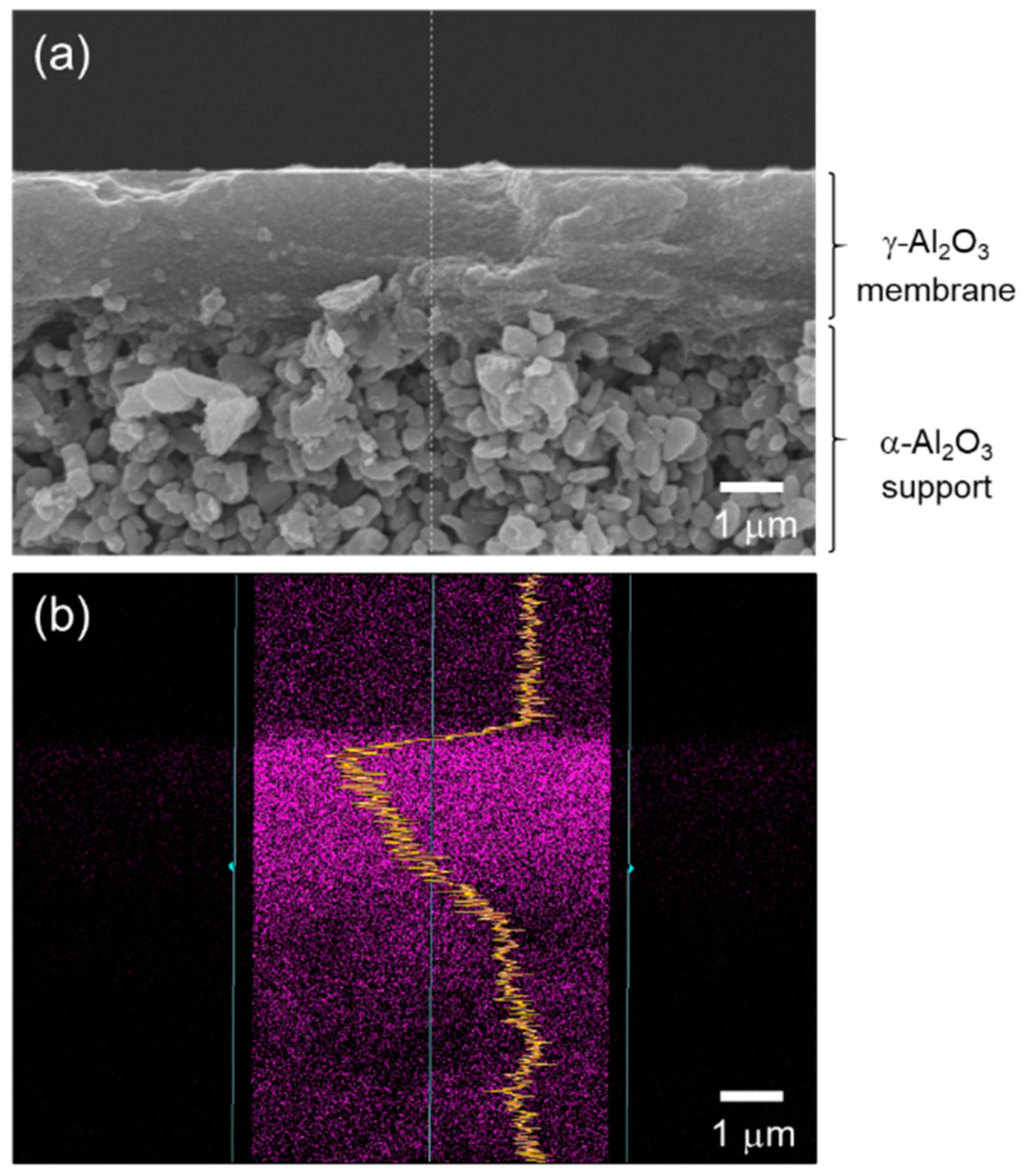
- Structure of the composite membrane
- (2)
- Gas permeation behaviors under dry condition
- (3)
- Gas permeation behaviors under the wet condition
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| A | membrane area, m2 |
| Ae | electrode area, m2 |
| Ea | activation energy, kJ mol−1 |
| Df | measured frequency shift, Hz |
| f0 | frequency of quartz crystal prior to a mass change, Hz |
| Δm | mass change, g |
| Mi | molecular weights of gas-i, kg mol−1 |
| p/p0 (H2O) | relative pressure of water vapor, dimensionless |
| pH | pressures of gas feed side, Pa |
| pL | pressures of gas permeate side, Pa |
| Qi | permeance of gas-i, mol m−2 s−1 Pa−1 |
| QK,i | Knudsen permeance of gas-i, mol m−2 s−1 Pa−1 |
| V | permeate molar flow rate, mol s−1 |
| Va(H2O) | maximum amount of water adsorption, cm3(STP) g−1 |
Greek Symbols
| α | permselectivity, dimensionless |
| ρq | shear modulus of quartz, g m−1 s−2 |
| μq | density of quartz, g cm−3 |
Abbreviations
| AHPCS | allyl-hydro-polycarbosilane |
| BTESE | 1,2-bis(triethoxysilyl)ethane |
| BTESM | bis(triethoxysilyl)methane |
| CMS | carbon molecular sieve |
| EA | ethanoanthracene |
| MOF | metal organic framework |
| PCS | polycarbosilane |
| PDCs | Polymer-Derived Ceramics |
| PDS | polydimethylsilane |
| PEC | photoelectrochemical |
| PFDA | 1H,1H,2H,2H-perfluorodecyl acrylate |
| PIM | polymers of intrinsic microporosity |
| QCM | quartz-crystal microbalance |
| SF | separation factor |
| STP | standard temperature and pressure (273.15 K and 101.30 kPa) |
| THF | tetrahydrofuran |
| TB | 6H,12H-5,11-methanodibenzo[b,f][1,5]diazocine (Tröger’s base) |
| Trip | triptycene |
| ZIF | zeolite imidazolate framework |
References
- Ogden, J.M.; Williams, R.H.; Larson, E.D. Societal lifecycle costs of cars with alternative fuels/engines. Energy Policy 2004, 32, 7–27. [Google Scholar] [CrossRef]
- Maeda, K.; Domen, K. New non-oxide photocatalysts designed for overall water splitting under visible light. J. Phys. Chem. C 2007, 111, 7851–7861. [Google Scholar] [CrossRef]
- Wang, Q.; Hisatomi, T.; Jia, Q.; Tokudome, H.; Zhong, M.; Wang, C.; Pan, Z.; Takata, T.; Nakabayashi, M.; Shibata, N. Scalable water splitting on particulate photocatalyst sheets with a solar-to-hydrogen energy conversion efficiency exceeding 1%. Nat. Mater. 2016, 15, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Goto, Y.; Hisatomi, T.; Wang, Q.; Higashi, T.; Ishikiriyama, K.; Maeda, T.; Sakata, Y.; Okunaka, S.; Tokudome, H.; Katayama, M.; et al. A Particulate Photocatalyst Water-Splitting Panel for Large-Scale Solar Hydrogen Generation. Joule 2018, 2, 509–520. [Google Scholar] [CrossRef]
- Yamada, T.; Domen, K. Development of Sunlight Driven Water Splitting Devices towards Future Artificial Photosynthetic Industry. ChemEngineering 2018, 2, 36. [Google Scholar] [CrossRef]
- Development of Basic Chemical Processes for Carbon Dioxide as Raw Material. Available online: www.nedo.go.jp/activities/EV_00296.html (accessed on 2 August 2020).
- Yagyu, S.; Matsui, H.; Matsuda, T.; Yasumoto, H. Studies of Explosive Characteristics of Hydrogen (1st Report); RIIS-RR-18-1; Ministry of Labour, the Research Institute of Industrial Safety, 1969. Available online: www.jniosh.johas.go.jp/publication/doc/rr/RR-18-1.pdf (accessed on 2 August 2020).
- Tanaka, K.; Sakata, Y. Present and Future Prospects of Hydrogen Production Process Constructed by the Combination of Photocatalytic H2O Splitting and Membrane Separation Process. Membrane 2011, 36, 113–121. [Google Scholar] [CrossRef]
- Oyama, S.T.; Stagg-Williams, S.M. Inorganic, Polymeric and Composite Membranes: Structure, Function and Other Correlations; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Mise, Y.; Ahn, S.J.; Takagaki, A.; Kikuchi, R.; Oyama, S.T. Fabrication and evaluation of trimethylmethoxysilane (TMMOS)-derived membranes for gas separation. Membranes 2019, 9, 123. [Google Scholar] [CrossRef]
- Kato, H.; Lundin, S.-T.B.; Ahn, S.-J.; Takagaki, A.; Kikuchi, R.; Oyama, S.T. Gas Separation Silica Membranes Prepared by Chemical Vapor Deposition of Methyl-Substituted Silanes. Membranes 2019, 9, 144. [Google Scholar] [CrossRef]
- Ted Oyama, S.; Aono, H.; Takagaki, A.; Sugawara, T.; Kikuchi, R. Synthesis of silica membranes by chemical vapor deposition using a dimethyldimethoxysilane precursor. Membranes 2020, 10, 50. [Google Scholar] [CrossRef]
- Yun, S.; Ted Oyama, S. Correlations in palladium membranes for hydrogen separation: A review. J. Membr. Sci. 2011, 375, 28–45. [Google Scholar] [CrossRef]
- Liguori, S.; Iulianelli, A.; Dalena, F.; Pinacci, P.; Drago, F.; Broglia, M.; Huang, Y.; Basile, A. Performance and long-term stability of Pd/PSS and Pd/Al2O3 membranes for hydrogen separation. Membranes 2014, 4, 143–162. [Google Scholar] [CrossRef] [PubMed]
- Alique, D.; Martinez-Diaz, D.; Sanz, R.; Calles, J.A. Review of supported Pd-based membranes preparation by electroless plating for ultra-pure hydrogen production. Membranes 2018, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Castro-Dominguez, B.; Leelachaikul, P.; Takagaki, A.; Sugawara, T.; Kikuchi, R.; Oyama, S.T. Supported perfluorotributylamine liquid membrane for H2/O2 separation. J. Memb. Sci. 2013, 448, 262–269. [Google Scholar] [CrossRef]
- Leelachaikul, P.; Castro-Dominguez, B.; Takagaki, A.; Sugawara, T.; Kikuchi, R.; Oyama, S.T. Perfluorooctanol-based liquid membranes for H2/O2 separation. Sep. Purif. Technol. 2014, 122, 431–439. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Takagaki, A.; Sugawara, T.; Kikuchi, R.; Oyama, S.T. Supported fluorocarbon liquid membranes for hydrogen/oxygen separation. J. Membr. Sci. 2016, 520, 272–280. [Google Scholar] [CrossRef]
- Burneau, A.; Lepage, J.; Maurice, G. Porous silica-water interactions. I. Structural and dimensional changes induced by water adsorption. J. Non-Cryst. Solids 1997, 217, 1–10. [Google Scholar] [CrossRef]
- Tsuru, T.; Hino, T.; Yoshioka, T.; Asaeda, M. Permporometry characterization of microporous ceramic membranes. J. Membr. Sci. 2001, 186, 257–265. [Google Scholar] [CrossRef]
- Díaz, U.; Corma, A. Organic-Inorganic Hybrid Materials: Multi-Functional Solids for Multi-Step Reaction Processes. Chem.-A Eur. J. 2018, 24, 3944–3958. [Google Scholar] [CrossRef]
- Mir, S.H.; Nagahara, L.A.; Thundat, T.; Mokarian-Tabari, P.; Furukawa, H.; Khosla, A. Review—Organic-Inorganic Hybrid Functional Materials: An Integrated Platform for Applied Technologies. J. Electrochem. Soc. 2018, 165, B3137–B3156. [Google Scholar] [CrossRef]
- Bill, J.; Aldinger, F. Precursor-derived Covalent Ceramics. Adv. Mater. 1995, 7, 775–787. [Google Scholar] [CrossRef]
- Riedel, R. Advanced Ceramics from Inorganic Polymers. In Materials Science and Technology; Brook, R.J., Ed.; Wiley-VCH: Weinheim, Germany, 1996; Volume 17B, pp. 1–50. [Google Scholar]
- Judeinstein, P.; Sanchez, C. Hybrid organic–inorganic materials: A land of multidisciplinarity. J. Mater. Chem. 1996, 6, 511–525. [Google Scholar] [CrossRef]
- Yajima, S.; Hayashi, J.; Omori, M. Continuous silicon carbide fiber of high tensile strength. Chem. Lett. 1975, 4, 931–934. [Google Scholar] [CrossRef]
- Yajima, S.; Okamura, K.; Hayashi, J.; Omori, M. Synthesis of Continuous SiC Fibers with High Tensile Strength. J. Am. Ceram. Soc. 1976, 59, 324–327. [Google Scholar] [CrossRef]
- Colombo, P.; Mera, G.; Riedel, R.; Sorarù, G.D. Polymer-derived ceramics: 40 Years of research and innovation in advanced ceramics. J. Am. Ceram. Soc. 2010, 93, 1805–1837. [Google Scholar] [CrossRef]
- Hotza, D.; Di Luccio, M.; Wilhelm, M.; Iwamoto, Y.; Bernard, S.; Diniz da Costa, J.C. Silicon carbide filters and porous membranes: A review of processing, properties, performance and application. J. Membr. Sci. 2020, 610, 118193. [Google Scholar] [CrossRef]
- Lee, L.-L.; Tsai, D.-S. A Hydrogen-Permselective Silicon Oxycarbide Membrane Derived from Polydimethylsilane. J. Am. Ceram. Soc. 1999, 82, 2796–2800. [Google Scholar] [CrossRef]
- Shelekhin, A.B.; Grosgogeat, E.J.; Hwang, S.T. Gas separation properties of a new polymer/inorganic composite membrane. J. Membr. Sci. 1992, 66, 129–141. [Google Scholar] [CrossRef]
- Kusakabe, K.; Yan Li, Z.; Maeda, H.; Morooka, S. Preparation of supported composite membrane by pyrolysis of polycarbosilane for gas separation at high temperature. J. Membr. Sci. 1995, 103, 175–180. [Google Scholar] [CrossRef]
- Li, Z.; Kusakabe, K.; Morooka, S. Preparation of thermostable amorphous Si-C-O membrane and its application to gas separation at elevated temperature. J. Membr. Sci. 1996, 118, 159–168. [Google Scholar] [CrossRef]
- Li, Z.; Kusakabe, K.; Morooka, S. Pore Structure and Permeance of Amorphous Si-C-O Membranes with High Durability at Elevated Temperature. Sep. Sci. Technol. 1997, 32, 1233–1254. [Google Scholar] [CrossRef]
- Suda, H.; Yamauchi, H.; Uchimaru, Y.; Fujiwara, I.; Haraya, K. Preparation and gas permeation properties of silicon carbide-based inorganic membranes for hydrogen separation. Desalination 2006, 193, 252–255. [Google Scholar] [CrossRef]
- Nagano, T.; Sato, K.; Saitoh, T.; Iwamoto, Y. Gas permeation properties of amorphous SiC membranes synthesized from polycarbosilane without oxygen-curing process. J. Ceram. Soc. Jpn. 2006, 114, 533–538. [Google Scholar] [CrossRef]
- Takeyama, A.; Sugimoto, M.; Yoshikawa, M. Gas permeation property of SiC membrane using curing of polymer precursor film by electron beam irradiation in helium atmosphere. Mater. Trans. 2011, 52, 1276–1280. [Google Scholar] [CrossRef]
- Elyassi, B.; Deng, W.; Sahimi, M.; Tsotsis, T.T. On the use of porous and nonporous fillers in the fabrication of silicon carbide membranes. Ind. Eng. Chem. Res. 2013, 52, 10269–10275. [Google Scholar] [CrossRef]
- Dabir, S.; Deng, W.; Sahimi, M.; Tsotsis, T. Fabrication of silicon carbide membranes on highly permeable supports. J. Membr. Sci. 2017, 537, 239–247. [Google Scholar] [CrossRef]
- Ciora, R.J.; Fayyaz, B.; Liu, P.K.T.; Suwanmethanond, V.; Mallada, R.; Sahimi, M.; Tsotsis, T.T. Preparation and reactive applications of nanoporous silicon carbide membranes. Chem. Eng. Sci. 2004, 59, 4957–4965. [Google Scholar] [CrossRef]
- Sandra, F.; Ballestero, A.; NGuyen, V.L.; Tsampas, M.N.; Vernoux, P.; Balan, C.; Iwamoto, Y.; Demirci, U.B.; Miele, P.; Bernard, S. Silicon carbide-based membranes with high soot particle filtration efficiency, durability and catalytic activity for CO/HC oxidation and soot combustion. J. Membr. Sci. 2016, 501, 79–92. [Google Scholar] [CrossRef]
- Wang, Q.; Yokoji, M.; Nagasawa, H.; Yu, L.; Kanezashi, M.; Tsuru, T. Microstructure evolution and enhanced permeation of SiC membranes derived from allylhydridopolycarbosilane. J. Membr. Sci. 2020, 118392. [Google Scholar] [CrossRef]
- Interrante, L.V.; Shen, Q. Polycarbosilanes. In Silicon-Containing Polymers; Jones, R.G., Ando, W., Chojnowski, J., Eds.; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2000; pp. 247–321. [Google Scholar]
- Matsumoto, K. Polycarbosilanes. In Encyclopedia of Polymer Science and Technology; Mark, H.F., Ed.; John Wiley & Sons: New York, NY, USA, 2003; Volume 7, pp. 426–438. [Google Scholar]
- Matsumoto, K.; Endo, T. Synthesis and ring-opening polymerization of functional silacyclobutane derivatives and their application to lithium ion batteries. Macromol. Symp. 2015, 349, 21–28. [Google Scholar] [CrossRef]
- Yajima, S.; Shishido, T.; Okamira, K. SiC Bodies Sintered with Three-Dimensional Cross-Linked Polycarbosilane. Am. Ceram. Soc. Bull. 1977, 56, 1060–1063. [Google Scholar]
- Kim, Y.-W.; Lee, J.-G. Effect of polycarbosilane addition on mechanical properties of hot-pressed silicon carbide. J. Mater. Sci. 1992, 27, 4746–4750. [Google Scholar] [CrossRef]
- Czubarow, P.; Seyferth, D. Application of poly(methylsilane) and Nicalon® polycarbosilane precursors as binders for metal/ceramic powders in preparation of functionally graded materials. J. Mater. Sci. 1997, 32, 2121–2130. [Google Scholar] [CrossRef]
- Sawai, Y.; Iwamoto, Y.; Okuzaki, S.; Yasutomi, Y.; Kikuta, K.; Hirano, S. Synthesis of Silicon Carbide Ceramics Using Chemically Modified Polycarbosilanes as a Compaction Binder. J. Am. Ceram. Soc. 1999, 82, 2121–2125. [Google Scholar] [CrossRef]
- Kubo, M.; Kojima, M.; Mano, R.; Daiko, Y.; Honda, S.; Iwamoto, Y. A hydrostable mesoporous γ-Al2O3 membrane modified with Si–C–H organic-inorganic hybrid derived from polycarbosilane. J. Membr. Sci. 2020, 598, 117799. [Google Scholar] [CrossRef]
- Breck, D.W. Zeolite Molecular Sieves: Structure, Chemistry, and Use; John Wiley & Sons: New York, NY, USA, 1974; ISBN 978-0471099857. [Google Scholar]
- Buttry, D.A.; Ward, M.D. Measurement of Interfaclal Processes at Electrode Surfaces with the Electrochemical Quartz Crystal Microbalance. Chem. Rev. 1992, 92, 1355–1379. [Google Scholar] [CrossRef]
- Sauerbrey, G. Verwendung von Schwingquarzen zur Wägung dünner Schichten und zur Mikrowägung. Zeitschrift Für Physik 1959, 155, 206–222. [Google Scholar] [CrossRef]
- Sreeja, R.; Swaminathan, B.; Painuly, A.; Sebastian, T.V.; Packirisamy, S. Allylhydridopolycarbosilane (AHPCS) as matrix resin for C/SiC ceramic matrix composites. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2010, 168, 204–207. [Google Scholar] [CrossRef]
- Kaur, S.; Riedel, R.; Ionescu, E. Pressureless fabrication of dense monolithic SiC ceramics from a polycarbosilane. J. Eur. Ceram. Soc. 2014, 34, 3571–3578. [Google Scholar] [CrossRef]
- Wen, Q.; Xu, Y.; Xu, B.; Fasel, C.; Guillon, O.; Buntkowsky, G.; Yu, Z.; Riedel, R.; Ionescu, E. Single-source-precursor synthesis of dense SiC/HfCxN1-x-based ultrahigh-temperature ceramic nanocomposites. Nanoscale 2014, 6, 13678–13689. [Google Scholar] [CrossRef]
- Proust, V.; Bechelany, M.C.; Ghisleni, R.; Beaufort, M.F.; Miele, P.; Bernard, S. Polymer-derived Si-C-Ti systems: From titanium nanoparticle-filled polycarbosilanes to dense monolithic multi-phase components with high hardness. J. Eur. Ceram. Soc. 2016, 36, 3671–3679. [Google Scholar] [CrossRef]
- Schmidt, M.; Durif, C.; Acosta, E.D.; Salameh, C.; Plaisantin, H.; Miele, P.; Backov, R.; Machado, R.; Gervais, C.; Alauzun, J.G.; et al. Molecular-Level Processing of Si-(B)-C Materials with Tailored Nano/Microstructures. Chem.-A Eur. J. 2017, 23, 17103–17117. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S.; Cançado, L.G.; Jorio, A.; Saito, R. Studying disorder in graphite-based systems by Raman spectroscopy. Phys. Chem. Chem. Phys. 2007, 9, 1276–1291. [Google Scholar] [CrossRef] [PubMed]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemienieska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Lowell, S.; Shields, J.E.; Thomas, M.A.; Thommes, M. Characterization of Porous Solids and Powders; Springer: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Lee, H.R.; Kanezashi, M.; Shimomura, Y.; Yoshioka, T.; Tsuru, T. Evaluation and fabrication of pore-size-tuned silica membranes with tetraethoxydimethyl disiloxane for gas separation. AIChE J. 2011, 57, 2755–2765. [Google Scholar] [CrossRef]
- Yoshioka, T.; Kanezashi, M.; Tsuru, T. Micropore size estimation on gas separation membranes: A study in experimental and molecular dynamics. AIChE J. 2013, 59, 2179–2194. [Google Scholar] [CrossRef]
- Lee, D.; Zhang, L.; Oyama, S.T.; Niu, S.; Saraf, R.F. Synthesis, characterization, and gas permeation properties of a hydrogen permeable silica membrane supported on porous alumina. J. Membr. Sci. 2004, 231, 117–126. [Google Scholar] [CrossRef]
- Gu, Y.; Oyama, S.T. High Molecular Permeance in a Poreless Ceramic Membrane. Adv. Mater. 2007, 19, 1636–1640. [Google Scholar] [CrossRef]
- Ahn, S.J.; Yun, G.N.; Takagaki, A.; Kikuchi, R.; Oyama, S.T. Synthesis and characterization of hydrogen selective silica membranes prepared by chemical vapor deposition of vinyltriethoxysilane. J. Membr. Sci. 2018, 550, 1–8. [Google Scholar] [CrossRef]
- Ren, X.; Tsuru, T. Organosilica-based membranes in gas and liquid-phase separation. Membranes 2019, 9, 107. [Google Scholar] [CrossRef]
- Li, L.; Wang, C.; Wang, N.; Cao, Y.; Wang, T. The preparation and gas separation properties of zeolite/carbon hybrid membranes. J. Mater. Sci. 2015, 50, 2561–2570. [Google Scholar] [CrossRef]
- Li, L.; Xu, R.; Song, C.; Zhang, B.; Liu, Q.; Wang, T. A review on the progress in nanoparticle/C hybrid CMS membranes for gas separation. Membranes 2018, 8, 134. [Google Scholar] [CrossRef] [PubMed]
- Zeynali, R.; Ghasemzadeh, K.; Sarand, A.B.; Kheiri, F.; Basile, A. Performance evaluation of graphene oxide (GO) nanocomposite membrane for hydrogen separation: Effect of dip coating sol concentration. Sep. Purif. Technol. 2018, 200, 169–176. [Google Scholar] [CrossRef]
- Nouri, M.; Ghasemzadeh, K.; Iulianelli, A. Theoretical evaluation of graphene membrane performance for hydrogen separation using molecular dynamic simulation. Membranes 2019, 9, 110. [Google Scholar] [CrossRef]
- Li, Y.; Lin, L.; Tu, M.; Nian, P.; Howarth, A.J.; Farha, O.K.; Qiu, J.; Zhang, X. Growth of ZnO self-converted 2D nanosheet zeolitic imidazolate framework membranes by an ammonia-assisted strategy. Nano Res. 2018, 11, 1850–1860. [Google Scholar] [CrossRef]
- Fang, M.; Montoro, C.; Semsarilar, M. Metal and covalent organic frameworks for membrane applications. Membranes 2020, 10, 107. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Song, Z.; Zhang, X.; Huang, Y.; Li, S.; Mao, Y.; Ploehn, H.J.; Bao, Y.; Yu, M. Ultrathin, molecular-sieving graphene oxide membranes for selective hydrogen separation. Science 2013, 342, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Lin, H. Engineering sub-nanometer channels in two-dimensional materials for membrane gas separation. Membranes 2018, 8, 100. [Google Scholar] [CrossRef]
- Lasseuguette, E.; Malpass-Evans, R.; Carta, M.; McKeown, N.B.; Ferrari, M.C. Temperature and pressure dependence of gas permeation in a microporous Tröger’s base polymer. Membranes 2018, 8, 132. [Google Scholar] [CrossRef]
- Malpass-Evans, R.; Rose, I.; Fuoco, A.; Bernardo, P.; Clarizia, G.; McKeown, N.B.; Jansen, J.C.; Carta, M. Effect of bridgehead methyl substituents on the gas permeability of Tröger’s-base derived polymers of intrinsic microporosity. Membranes 2020, 10, 62. [Google Scholar] [CrossRef]

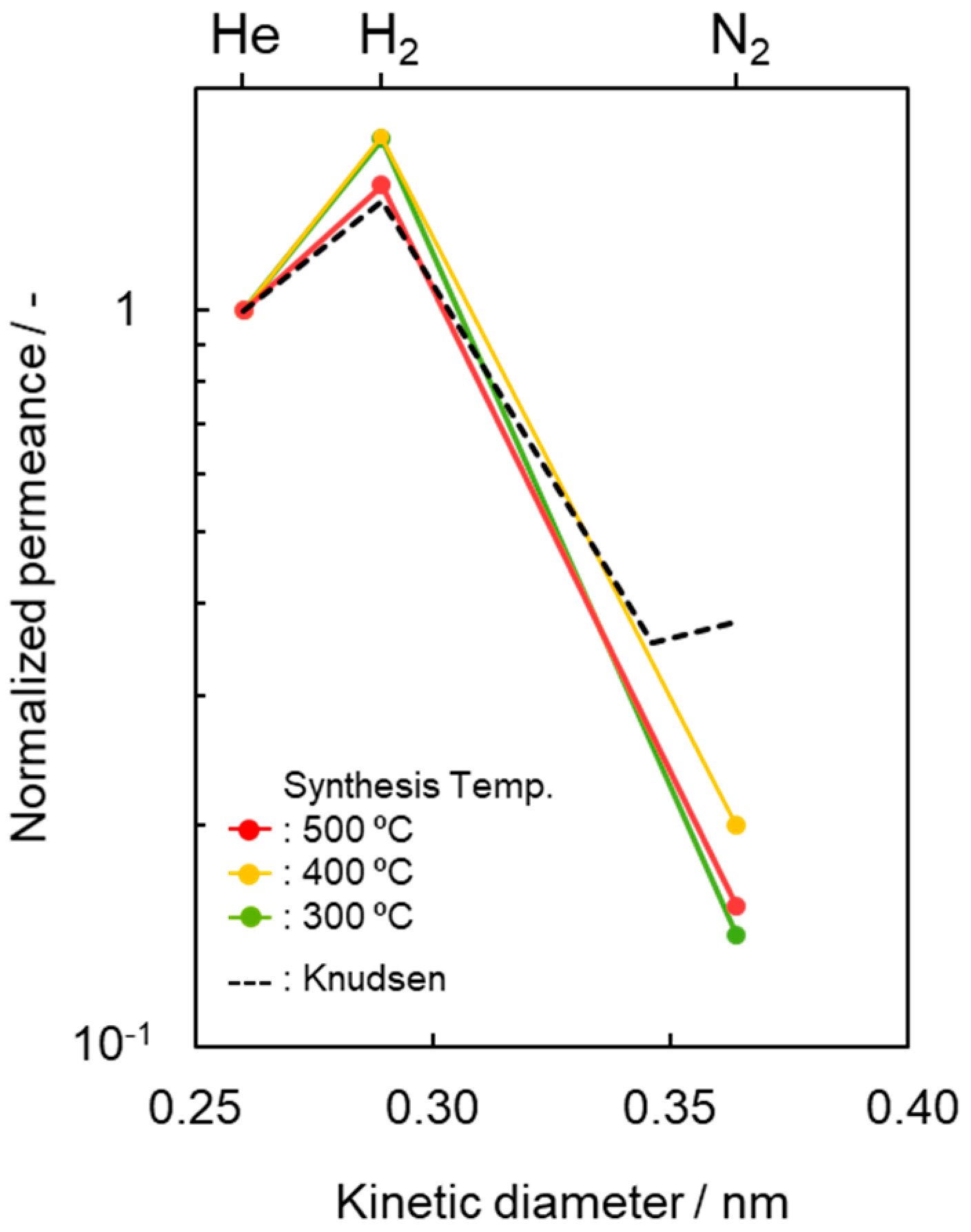
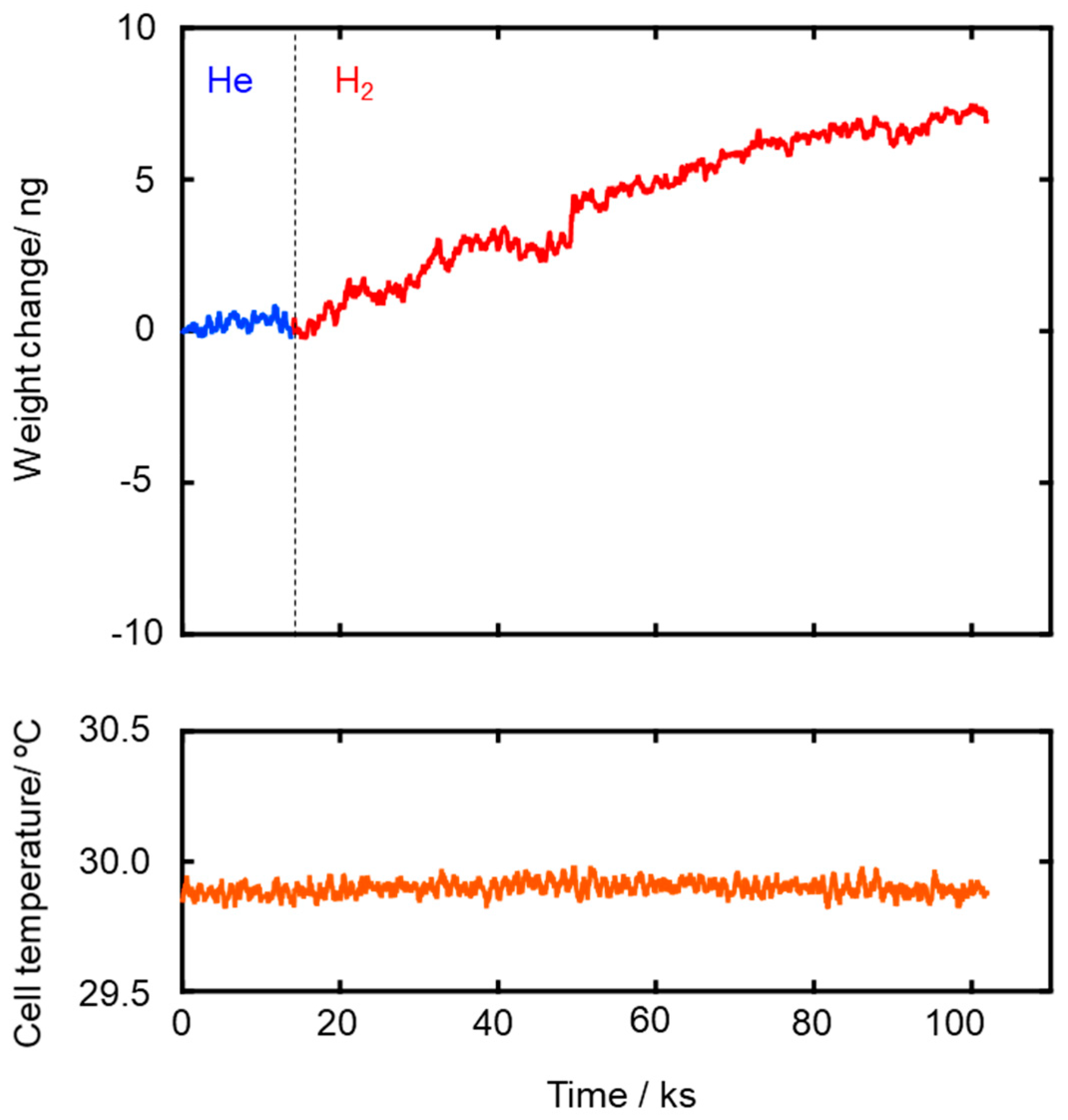
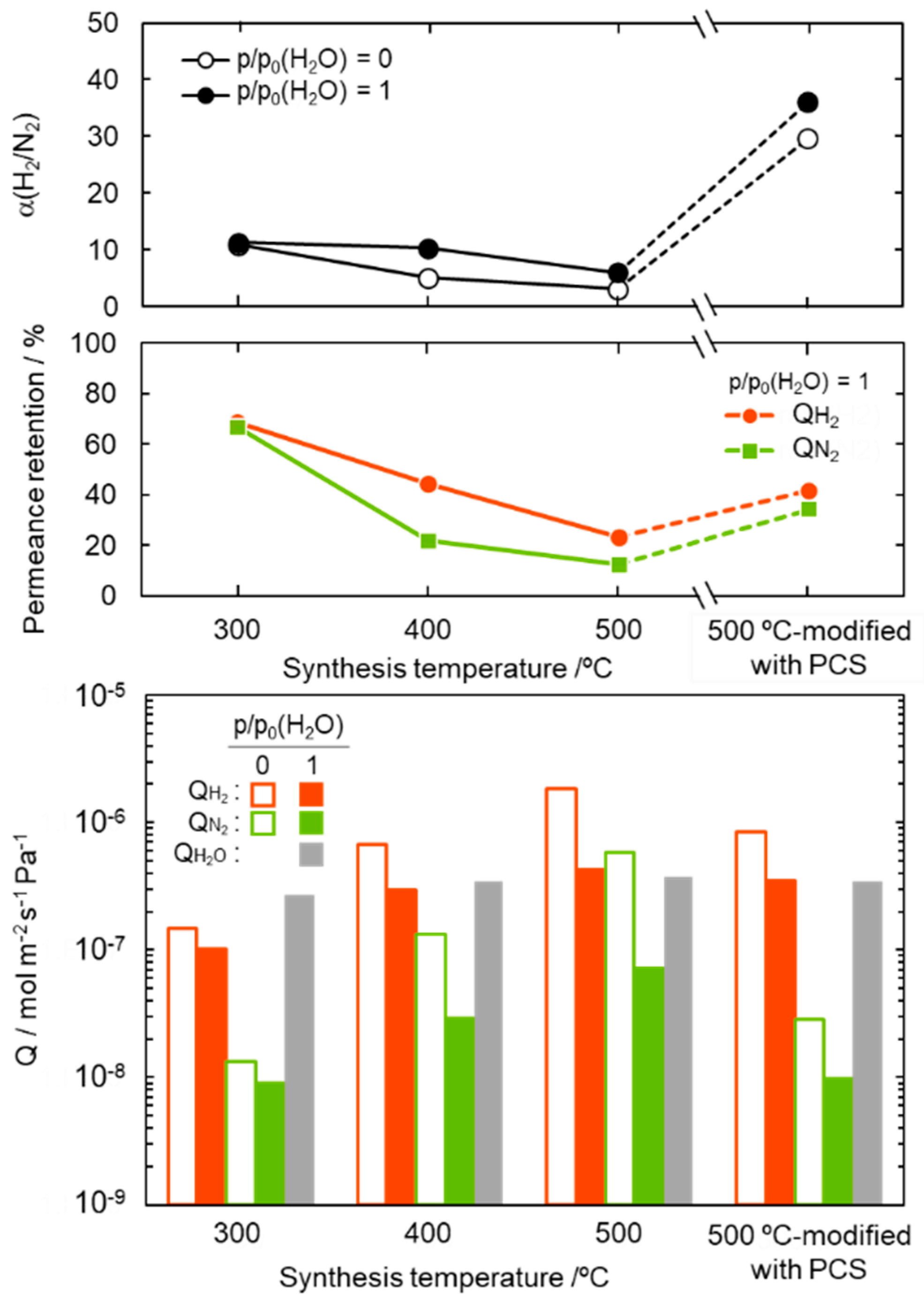
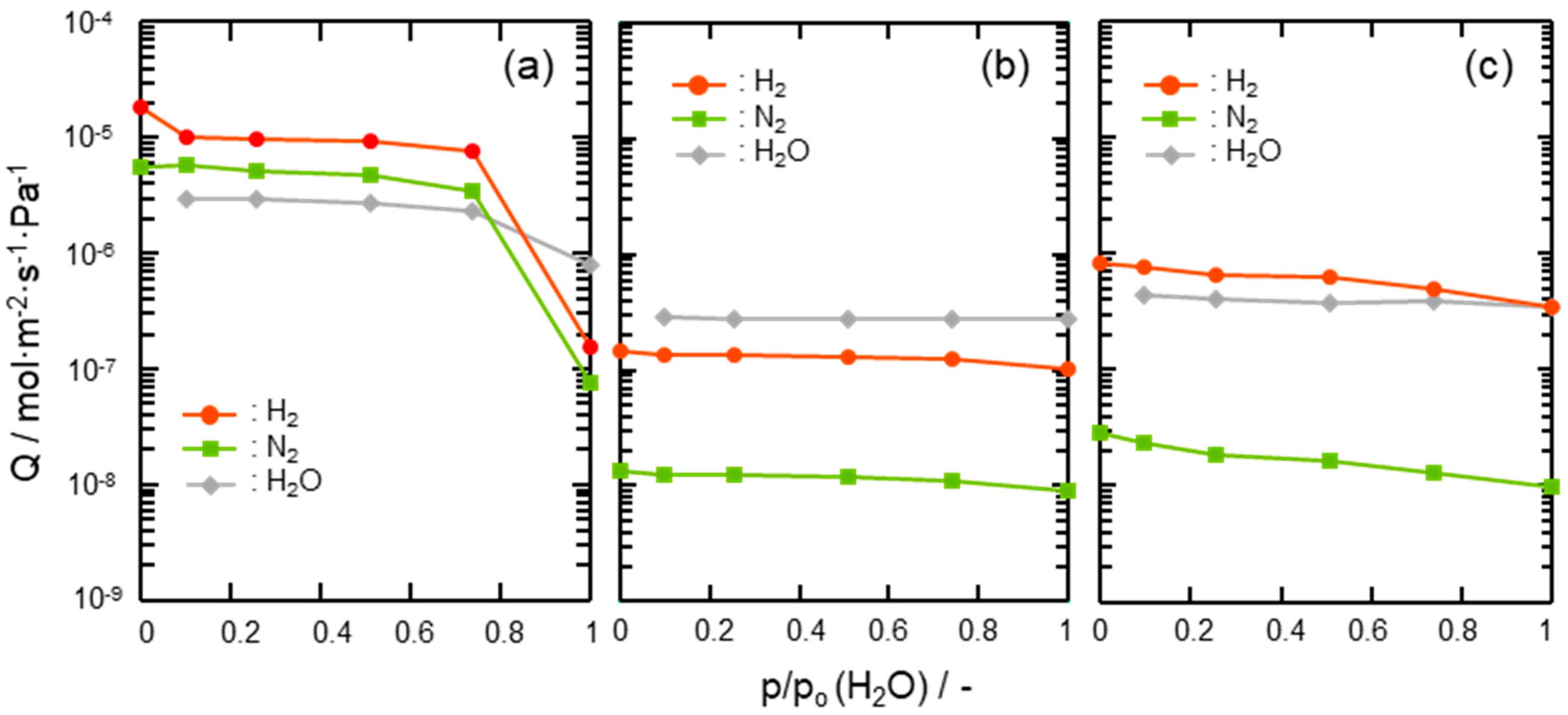

| SiCH Hybrid/γ-Al2O3 Composite Membrane Synthesis Temp. | Activation Energy (Ea)/kJ mol−1 | |
|---|---|---|
| He | H2 | |
| 300 °C | 20.3 | 17.0 |
| 400 °C | 9.8 | 8.6 |
| 500 °C | 1.4 | 0.5 |
| Feed Side | Permeate Side | Separation Factor (SF) | ||||
|---|---|---|---|---|---|---|
| Flow Rate, VF/× 10−5 mol s−1 | Flow Rate, V/× 10−5 mol s−1 | Flux, J/× 10−5 mol m−2 s−1 | ||||
| VF, H2 | VF, N2 | VH2 | VN2 | JH2 | JN2 | |
| 14.7 | 7.4 | 2.2 | 0.043 | 2971.8 | 56.6 | 26 |
| No. | Membranes | Temp./°C | H2 Permeance | H2/X Selectivity | Ref. | |
|---|---|---|---|---|---|---|
| /mol m−2 s−1 Pa−1 | α(H2/X) | X | ||||
| #01 | 500 °C modified with PCS, Dry | 50 | 8.4 × 10−7 | 29.6 | N2 | This study |
| Wet, p/p0(H2O) = 1.0 | 50 | 3.5 × 10−7 | 36.1 | N2 | ||
| #02 | SiO2-based organic–inorganic hybrid (BTESE) | 40 | 7.66 × 10−7 | −20 | N2 | [68] |
| #03 | SiO2-based organic–inorganic hybrid (BTESM) | 50 | 1.79 × 10−6 | 12 | N2 | |
| #04 | zeolite-β/CMS | 30 | 2.64 × 10−9 | 97 | N2 | [69,70] |
| 30 | 1.95 × 10−8 | 67.3 | N2 | |||
| #05 | zeolite-Y/CMS | 30 | 2.77 × 10−9 | 100.2 | N2 | |
| 30 | 1.96 × 10−8 | 61 | N2 | |||
| #06 | PFDA-based liquid membrane | 30 | 2.4 × 10−9 | 10 | O2 | [18] |
| #07 | graphene nanosheet | RT | 5.9 × 10−7 | 16.5 | N2 | [71,72] |
| #08 | ZIF nanosheet membrane | 30 | 2.04 × 10−7 | 66.55 | N2 | [73,74] |
| #09 | ultrathin graphene oxide | 20 | −1 × 10−7 | −900 | N2 | [75,76] |
| #10 | microporous polymer (PIM-EA(H2)-TB) | 30 | (1.7 × 10−12) * | 22 | N2 | [77] |
| #11 | microporous polymer (PIM-Trip(Me2)-TB) | 25 | (1.8 × 10−12) * | 21.4 | N2 | [78] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubo, M.; Mano, R.; Kojima, M.; Naniwa, K.; Daiko, Y.; Honda, S.; Ionescu, E.; Bernard, S.; Riedel, R.; Iwamoto, Y. Hydrogen Selective SiCH Inorganic–Organic Hybrid/γ-Al2O3 Composite Membranes. Membranes 2020, 10, 258. https://doi.org/10.3390/membranes10100258
Kubo M, Mano R, Kojima M, Naniwa K, Daiko Y, Honda S, Ionescu E, Bernard S, Riedel R, Iwamoto Y. Hydrogen Selective SiCH Inorganic–Organic Hybrid/γ-Al2O3 Composite Membranes. Membranes. 2020; 10(10):258. https://doi.org/10.3390/membranes10100258
Chicago/Turabian StyleKubo, Miwako, Ryota Mano, Misako Kojima, Kenichi Naniwa, Yusuke Daiko, Sawao Honda, Emanuel Ionescu, Samuel Bernard, Ralf Riedel, and Yuji Iwamoto. 2020. "Hydrogen Selective SiCH Inorganic–Organic Hybrid/γ-Al2O3 Composite Membranes" Membranes 10, no. 10: 258. https://doi.org/10.3390/membranes10100258
APA StyleKubo, M., Mano, R., Kojima, M., Naniwa, K., Daiko, Y., Honda, S., Ionescu, E., Bernard, S., Riedel, R., & Iwamoto, Y. (2020). Hydrogen Selective SiCH Inorganic–Organic Hybrid/γ-Al2O3 Composite Membranes. Membranes, 10(10), 258. https://doi.org/10.3390/membranes10100258