Efficacy of Two Chlamydia abortus Subcellular Vaccines in a Pregnant Ewe Challenge Model for Ovine Enzootic Abortion
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Preparation of C. abortus Elementary Bodies (EBs)
2.3. Vaccine Antigen Preparation and Quantification
2.4. Formulation of Vaccine Preparations
2.5. Preparation of C. abortus Challenge Inoculum
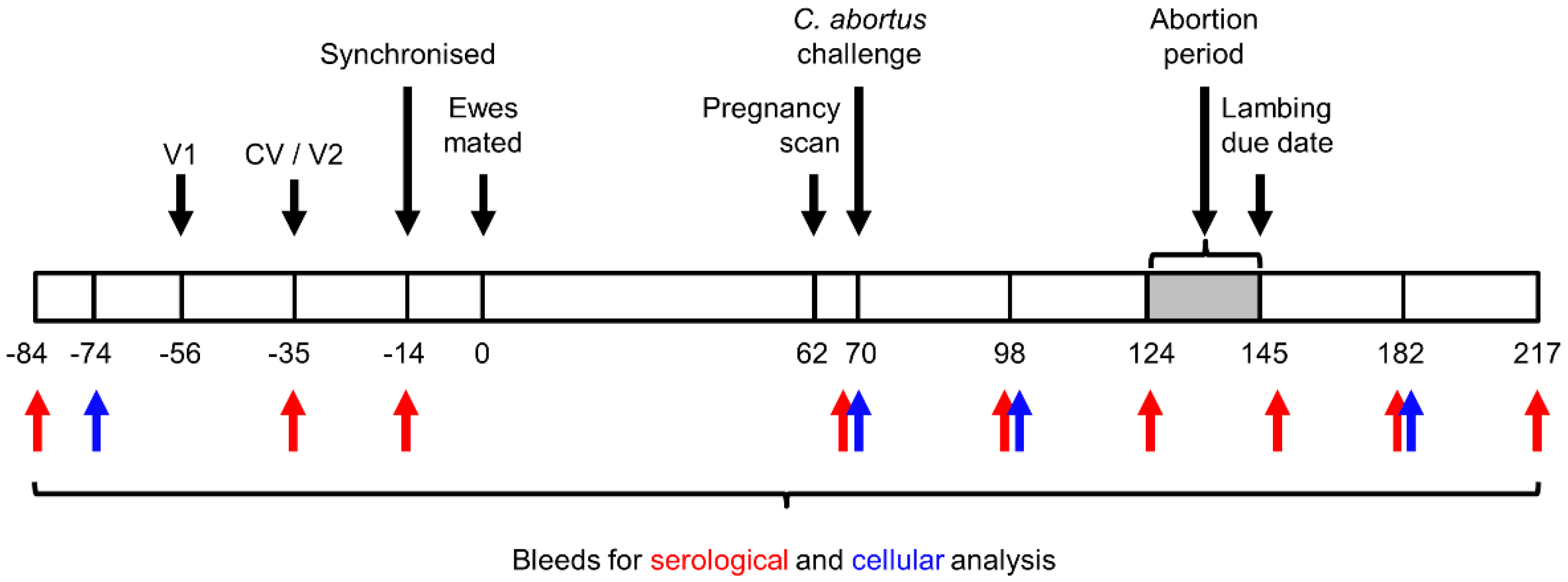
2.6. Experimental Design
2.7. Sample Collection
2.8. Quantitative Real-Time PCR (qPCR)
2.9. PCR-Restriction Fragment Length Polymorphism (RFLP) Analysis
2.10. Histopathological Examination and Immunohistochemical (IHC) Analysis
2.11. Serological Analysis
2.12. Cellular Analysis
2.13. Statistical Analysis
3. Results
3.1. Clinical Outcome of Pregnancy
3.2. Detection of C. abortus Infection
3.3. Histology and Immunohistochemical Analysis
3.4. PCR-RFLP Analysis
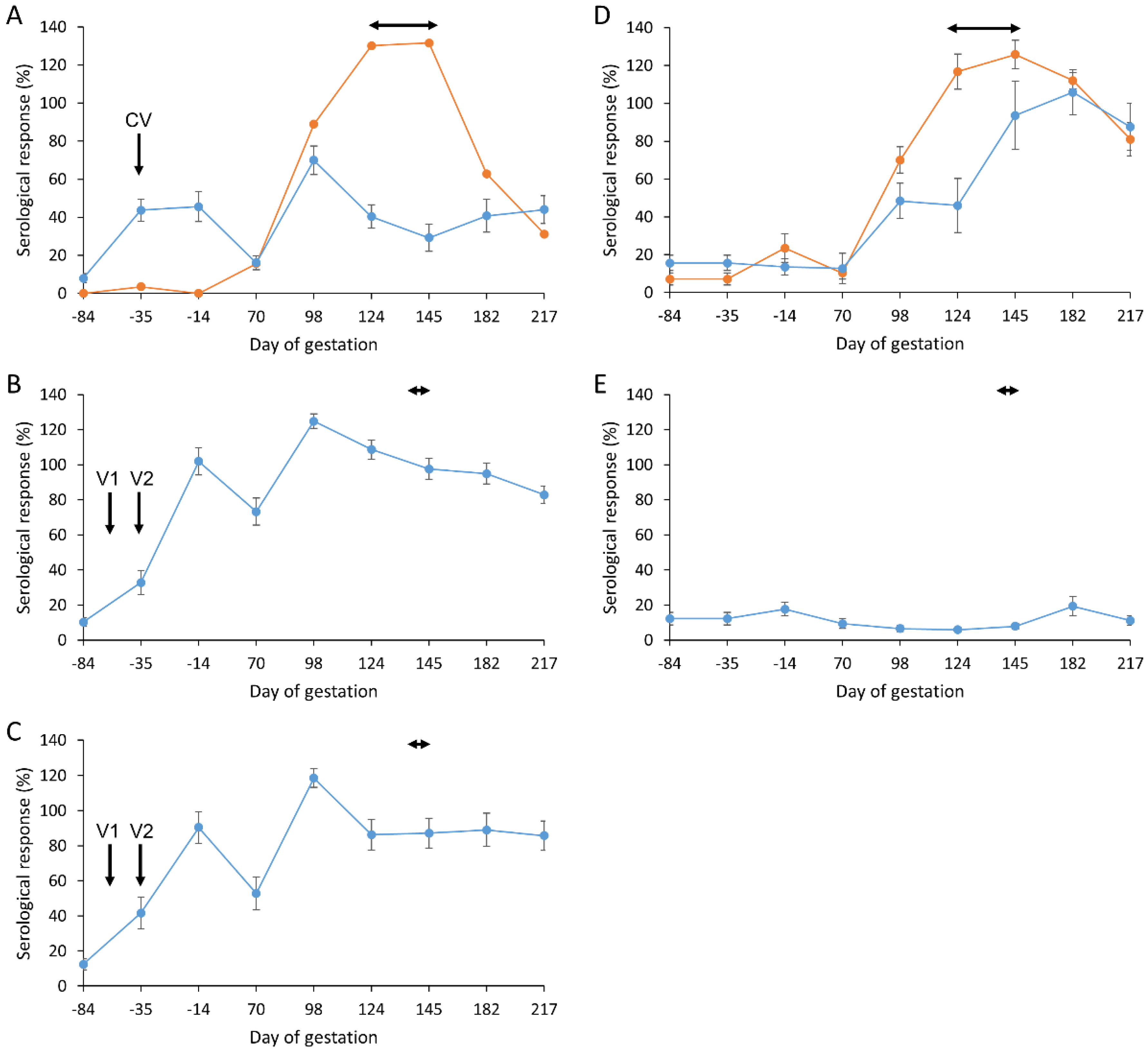
3.5. Antibody Responses
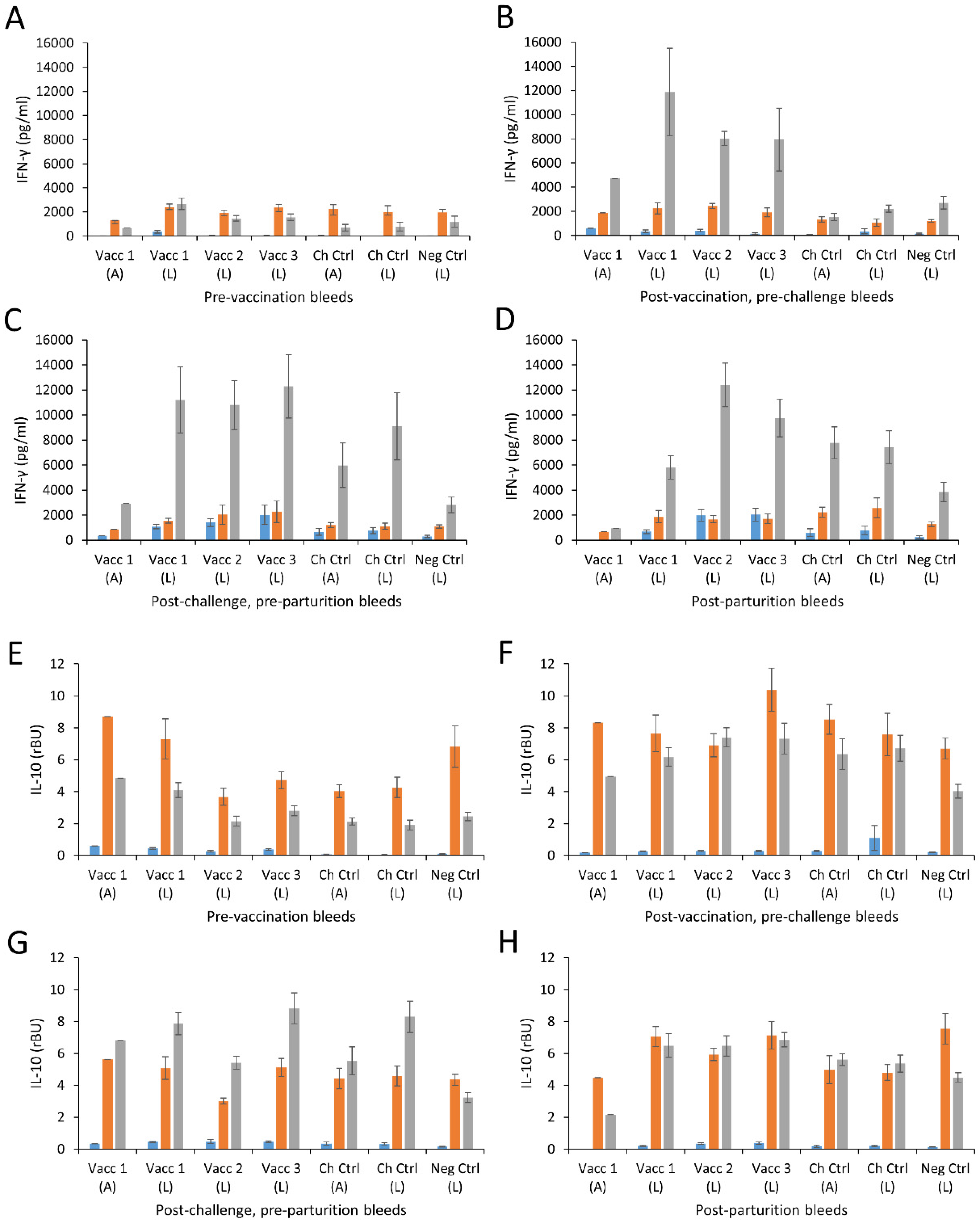
3.6. Cellular Responses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rodolakis, A.; Laroucau, K. Chlamydiaceae and chlamydial infections in sheep or goats. Vet. Microbiol. 2015, 181, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Borel, N.; Polkinghorne, A.; Pospischil, A. A Review on Chlamydial Diseases in Animals: Still a Challenge for Pathologists? Vet. Pathol. 2018, 55, 374–390. [Google Scholar] [CrossRef]
- Schautteet, K.; Vanrompay, D. Chlamydiaceae infections in pig. Vet. Res. 2011, 42, 29. [Google Scholar] [CrossRef]
- Longbottom, D.; Coulter, L.J. Animal chlamydioses and zoonotic implications. J. Comp. Pathol. 2003, 128, 217–244. [Google Scholar] [CrossRef]
- Nietfeld, J.C. Chlamydial infections in small ruminants. Vet. Clin. N. Am. Food Anim. Pract. 2001, 17, 301–314. [Google Scholar] [CrossRef]
- CABI Invasive Species Compendium: Chlamydophila abortus. Available online: https://www.cabi.org/isc/datasheet/89292#toDistributionMaps (accessed on 14 June 2021).
- GB Sheep Disease Surveillance Dashboard. Available online: https://public.tableau.com/app/profile/siu.apha/viz/SheepDashboard_/Overview (accessed on 14 June 2021).
- Giroud, P.; Roger, F.; Dumes, N. Certaines avortements chez la femme peuvent être dus a des agents situés a côté du groupe de la psittacose. Comptes Rendus Hebd. Seances Acad. Sci. 1956, 242, 697–699. [Google Scholar]
- Johnson, F.W.; Matheson, B.A.; Williams, H.; Laing, A.G.; Jandial, V.; Davidson-Lamb, R.; Halliday, G.J.; Hobson, D.; Wong, S.Y.; Hadley, K.M.; et al. Abortion due to infection with Chlamydia psittaci in a sheep farmer’s wife. Br. Med. J. (Clin. Res. Ed.) 1985, 290, 592–594. [Google Scholar] [CrossRef]
- Wong, S.Y.; Gray, E.S.; Buxton, D.; Finlayson, J.; Johnson, F.W. Acute placentitis and spontaneous abortion caused by Chlamydia psittaci of sheep origin: A histological and ultrastructural study. J. Clin. Pathol. 1985, 38, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Pospischil, A.; Thoma, R.; Hilbe, M.; Grest, P.; Zimmermann, D.; Gebbers, J.O. [Abortion in humans caused by Chlamydophila abortus (Chlamydia psittaci serovar 1)]. Schweiz. Arch. Tierheilkd. 2002, 144, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Buxton, D. Potential danger to pregnant women of Chlamydia psittaci from sheep. Vet. Rec. 1986, 118, 510–511. [Google Scholar] [CrossRef]
- Enzootic Abortion of Ewes (Ovine chlamydioses) (Infection with Chlamydia abortus), Chapter 3.8.5. Available online: https://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/3.08.05_ENZ_ABOR.pdf (accessed on 8 March 2021).
- Montbrau, C.; Fontseca, M.; March, R.; Sitja, M.; Benavides, J.; Ortega, N.; Caro, M.R.; Salinas, J. Evaluation of the efficacy of a new commercially available inactivated vaccine against ovine enzootic abortion. Front. Vet. Sci. 2020, 7, 593. [Google Scholar] [CrossRef]
- Jones, G.E.; Jones, K.A.; Machell, J.; Brebner, J.; Anderson, I.E.; How, S. Efficacy trials with tissue-culture grown, inactivated vaccines against chlamydial abortion in sheep. Vaccine 1995, 13, 715–723. [Google Scholar] [CrossRef]
- Caro, M.R.; Ortega, N.; Buendia, A.J.; Gallego, M.C.; Del Rio, L.; Cuello, F.; Salinas, J. Protection conferred by commercially available vaccines against Chlamydophila abortus in a mouse model. Vet. Rec. 2001, 149, 492–493. [Google Scholar] [CrossRef]
- Garcia de la Fuente, J.N.; Gutierrez-Martin, C.B.; Ortega, N.; Rodriguez-Ferri, E.F.; del Rio, M.L.; Gonzalez, O.R.; Salinas, J. Efficacy of different commercial and new inactivated vaccines against ovine enzootic abortion. Vet. Microbiol. 2004, 100, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Rodolakis, A.; Souriau, A. Response of goats to vaccination with temperature-sensitive mutants of Chlamydia psittaci obtained by nitrosoguanidine mutagenesis. Am. J. Vet. Res. 1986, 47, 2627–2631. [Google Scholar] [PubMed]
- Chalmers, W.S.; Simpson, J.; Lee, S.J.; Baxendale, W. Use of a live chlamydial vaccine to prevent ovine enzootic abortion. Vet. Rec. 1997, 141, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Longbottom, D.; Livingstone, M. Vaccination against chlamydial infections of man and animals. Vet. J. 2006, 171, 263–275. [Google Scholar] [CrossRef]
- Caspe, S.G.; Livingstone, M.; Frew, D.; Aitchison, K.; Wattegedera, S.R.; Entrican, G.; Palarea-Albaladejo, J.; McNeilly, T.N.; Milne, E.; Sargison, N.D.; et al. The 1B vaccine strain of Chlamydia abortus produces placental pathology indistinguishable from a wild type infection. PLoS ONE 2020, 15, e0242526. [Google Scholar] [CrossRef]
- Sargison, N.D.; Truyers, I.G.; Howie, F.E.; Thomson, J.R.; Cox, A.L.; Livingstone, M.; Longbottom, D. Identification of the 1B vaccine strain of Chlamydia abortus in aborted placentas during the investigation of toxaemic and systemic disease in sheep. N. Z. Vet. J. 2015, 63, 284–287. [Google Scholar] [CrossRef]
- Wheelhouse, N.; Aitchison, K.; Laroucau, K.; Thomson, J.; Longbottom, D. Evidence of Chlamydophila abortus vaccine strain 1B as a possible cause of ovine enzootic abortion. Vaccine 2010, 28, 5657–5663. [Google Scholar] [CrossRef]
- Longbottom, D.; Sait, M.; Livingstone, M.; Laroucau, K.; Sachse, K.; Harris, S.R.; Thomson, N.R.; Seth-Smith, H.M.B. Genomic evidence that the live Chlamydia abortus vaccine strain 1B is not attenuated and has the potential to cause disease. Vaccine 2018, 36, 3593–3598. [Google Scholar] [CrossRef] [PubMed]
- Laroucau, K.; Aaziz, R.; Vorimore, F.; Menard, M.F.; Longbottom, D.; Denis, G. Abortion storm induced by the live C. abortus vaccine 1B strain in a vaccinated sheep flock, mimicking a natural wild-type infection. Vet. Microbiol. 2018, 225, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Seco, T.; Perez-Sancho, M.; Salinas, J.; Navarro, A.; Diez-Guerrier, A.; Garcia, N.; Pozo, P.; Goyache, J.; Dominguez, L.; Alvarez, J. Effect of preventive Chlamydia abortus vaccination in offspring development in sheep challenged experimentally. Front. Vet. Sci. 2016, 3, 67. [Google Scholar] [CrossRef]
- O’Neill, L.M.; Keane, O.M.; Ross, P.J.; Nally, J.E.; Seshu, J.; Markey, B. Evaluation of protective and immune responses following vaccination with recombinant MIP and CPAF from Chlamydia abortus as novel vaccines for enzootic abortion of ewes. Vaccine 2019, 37, 5428–5438. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Pais, R.; Ohandjo, A.; He, C.; He, Q.; Omosun, Y.; Igietseme, J.U.; Eko, F.O. Comparative evaluation of the protective efficacy of two formulations of a recombinant Chlamydia abortus subunit candidate vaccine in a mouse model. Vaccine 2015, 33, 1865–1872. [Google Scholar] [CrossRef]
- Hechard, C.; Grepinet, O.; Rodolakis, A. Molecular cloning of the Chlamydophila abortus groEL gene and evaluation of its protective efficacy in a murine model by genetic vaccination. J. Med. Microbiol. 2004, 53, 861–868. [Google Scholar] [CrossRef][Green Version]
- Ou, C.; Tian, D.; Ling, Y.; Pan, Q.; He, Q.; Eko, F.O.; He, C. Evaluation of an ompA-based phage-mediated DNA vaccine against Chlamydia abortus in piglets. Int. Immunopharmacol. 2013, 16, 505–510. [Google Scholar] [CrossRef]
- Ling, Y.; Liu, W.; Clark, J.R.; March, J.B.; Yang, J.; He, C. Protection of mice against Chlamydophila abortus infection with a bacteriophage-mediated DNA vaccine expressing the major outer membrane protein. Vet. Immunol. Immunopathol. 2011, 144, 389–395. [Google Scholar] [CrossRef]
- Herring, A.J.; Jones, G.E.; Dunbar, S.M.; Nettleton, P.F.; Fitzgerald, T.A.; Anderson, I.E.; Chapman, S.N.; Wilson, T.M.A. Recombinant vaccines against Chlamydia psittaci—An overview of results using bacterial expression and a new approach using a plant virus ‘overcoat’ system. In Proceedings of the Ninth International Symposium on Human Chlamydial Infection, San Francisco, CA, USA, 21–26 June 1998; Stephens, R.S., Byrne, G.I., Christiansen, G., Clarke, I.N., Grayston, J.T., Rank, R.G., Ridgway, G.L., Saikku, P., Schachter, J., Stamm, W.E., Eds.; International Chlamydia Symposium: San Francisco, CA, USA, 1998; pp. 434–437. [Google Scholar]
- Herring, A.J. The function of the major outer membrane protein (MOMP) in the biology and pathogenicity of Chlamydia. Alpe Adria Microbiol. J. 1996, 5, 223–232. [Google Scholar]
- Tan, T.W.; Herring, A.J.; Anderson, I.E.; Jones, G.E. Protection of sheep against Chlamydia psittaci infection with a subcellular vaccine containing the major outer membrane protein. Infect. Immun. 1990, 58, 3101–3108. [Google Scholar] [CrossRef]
- Batteiger, B.E.; Rank, R.G.; Bavoil, P.M.; Soderberg, L.S. Partial protection against genital reinfection by immunization of guinea-pigs with isolated outer-membrane proteins of the chlamydial agent of guinea-pig inclusion conjunctivitis. J. Gen. Microbiol. 1993, 139, 2965–2972. [Google Scholar] [CrossRef]
- De Sa, C.; Souriau, A.; Bernard, F.; Salinas, J.; Rodolakis, A. An oligomer of the major outer membrane protein of Chlamydia psittaci is recognized by monoclonal antibodies which protect mice from abortion. Infect. Immun. 1995, 63, 4912–4916. [Google Scholar] [CrossRef]
- Pal, S.; Theodor, I.; Peterson, E.M.; de la Maza, L.M. Immunization with an acellular vaccine consisting of the outer membrane complex of Chlamydia trachomatis induces protection against a genital challenge. Infect. Immun. 1997, 65, 3361–3369. [Google Scholar] [CrossRef]
- Igietseme, J.U.; Black, C.M.; Caldwell, H.D. Chlamydia vaccines: Strategies and status. BioDrugs 2002, 16, 19–35. [Google Scholar] [CrossRef]
- McClenaghan, M.; Herring, A.J.; Aitken, I.D. Comparison of Chlamydia psittaci isolates by DNA restriction endonuclease analysis. Infect. Immun. 1984, 45, 384–389. [Google Scholar] [CrossRef]
- Hobson, D.; Johnson, F.W.A.; Byng, R.E. The growth of the ewe abortion chlamydial agent in McCoy cell cultures. J. Comp. Pathol. 1977, 87, 155–159. [Google Scholar] [CrossRef]
- Longbottom, D.; Livingstone, M.; Aitchison, K.D.; Imrie, L.; Manson, E.; Wheelhouse, N.; Inglis, N.F. Proteomic characterisation of the Chlamydia abortus outer membrane complex (COMC) using combined rapid monolithic column liquid chromatography and fast MS/MS scanning. PLoS ONE 2019, 14, e0224070. [Google Scholar] [CrossRef] [PubMed]
- Aucouturier, J.; Dupuis, L.; Ganne, V. Adjuvants designed for veterinary and human vaccines. Vaccine 2001, 19, 2666–2672. [Google Scholar] [CrossRef]
- Longbottom, D.; Livingstone, M.; Maley, S.; van der Zon, A.; Rocchi, M.; Wilson, K.; Wheelhouse, N.; Dagleish, M.; Aitchison, K.; Wattegedera, S.; et al. Intranasal infection with Chlamydia abortus induces dose-dependent latency and abortion in sheep. PLoS ONE 2013, 8, e57950. [Google Scholar] [CrossRef] [PubMed]
- Arif, E.D.; Saeed, N.M.; Rachid, S.K. Isolation and identification of Chlamydia abortus from aborted ewes in Sulaimani Province, Northern Iraq. Pol. J. Microbiol. 2020, 69, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, M.; Wheelhouse, N.; Maley, S.W.; Longbottom, D. Molecular detection of Chlamydophila abortus in post-abortion sheep at oestrus and subsequent lambing. Vet. Microbiol. 2009, 135, 134–141. [Google Scholar] [CrossRef]
- Wilson, K.; Livingstone, M.; Longbottom, D. Comparative evaluation of eight serological assays for diagnosing Chlamydophila abortus infection in sheep. Vet. Microbiol. 2009, 135, 38–45. [Google Scholar] [CrossRef]
- Livingstone, M.; Wheelhouse, N.; Ensor, H.; Rocchi, M.; Maley, S.; Aitchison, K.; Wattegedera, S.; Wilson, K.; Sait, M.; Siarkou, V.; et al. Pathogenic outcome following experimental infection of sheep with Chlamydia abortus variant strains LLG and POS. PLoS ONE 2017, 12, e0177653. [Google Scholar] [CrossRef]
- Laroucau, K.; Vorimore, F.; Sachse, K.; Vretou, E.; Siarkou, V.I.; Willems, H.; Magnino, S.; Rodolakis, A.; Bavoil, P.M. Differential identification of Chlamydophila abortus live vaccine strain 1B and C. abortus field isolates by PCR-RFLP. Vaccine 2010, 28, 5653–5656. [Google Scholar] [CrossRef] [PubMed]
- Wattegedera, S.R.; Livingstone, M.; Maley, S.; Rocchi, M.; Lee, S.; Pang, Y.; Wheelhouse, N.M.; Aitchison, K.; Palarea-Albaladejo, J.; Buxton, D.; et al. Defining immune correlates during latent and active chlamydial infection in sheep. Vet. Res. 2020, 51, 75. [Google Scholar] [CrossRef]
- Dunnett’s Test/Dunnett’s Method: Definition. Available online: https://www.statisticshowto.com/dunnetts-test/ (accessed on 14 June 2021).
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 17 May 2021).
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R Stat. Soc. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Sammin, D.J.; Markey, B.K.; Quinn, P.J.; McElroy, M.C.; Bassett, H.F. Comparison of fetal and maternal inflammatory responses in the ovine placenta after experimental infection with Chlamydophila abortus. J. Comp. Pathol. 2006, 135, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R. The ‘Direct Costs’ of livestock disease: The development of a system of models for the analysis of 30 endemic livestock diseases in Great Britain. J. Agric. Econ. 2003, 54, 55–71. [Google Scholar] [CrossRef]
- Wood, R. Enzootic abortion costs home industry £20m pa. Farmers Wkly. 1992, 117, 60. [Google Scholar]
- Aitken, I.D.; Clarkson, M.J.; Linklater, K. Enzootic abortion of ewes. Vet. Rec. 1990, 126, 136–138. [Google Scholar] [CrossRef]
- Sammin, D.; Markey, B.; Bassett, H.; Buxton, D. The ovine placenta and placentitis—A review. Vet. Microbiol. 2009, 135, 90–97. [Google Scholar] [CrossRef]
- Studdert, M.J. Bedsonia abortion of sheep. II. Pathology and pathogenesis with observations on the normal ovine placenta. Res. Vet. Sci. 1968, 9, 57–64. [Google Scholar] [CrossRef]
- Adamson, S.L.; Lu, Y.; Whiteley, K.J.; Holmyard, D.; Hemberger, M.; Pfarrer, C.; Cross, J.C. Interactions between trophoblast cells and the maternal and fetal circulation in the mouse placenta. Dev. Biol. 2002, 250, 358–373. [Google Scholar] [CrossRef]
- Comparative Placentation. Available online: http://placentation.ucsd.edu/ (accessed on 14 June 2021).
- Wattegedera, S.R.; Doull, L.E.; Goncheva, M.I.; Wheelhouse, N.M.; Watson, D.M.; Pearce, J.; Benavides, J.; Palarea-Albaladejo, J.; McInnes, C.J.; Ballingall, K.; et al. Immunological homeostasis at the ovine placenta may reflect the degree of maternal fetal interaction. Front. Immunol. 2018, 9, 3025. [Google Scholar] [CrossRef]
- Begg, D.J.; Dhungyel, O.; Naddi, A.; Dhand, N.K.; Plain, K.M.; de Silva, K.; Purdie, A.C.; Whittington, R.J. The immunogenicity and tissue reactivity of Mycobacterium avium subsp paratuberculosis inactivated whole cell vaccine is dependent on the adjuvant used. Heliyon 2019, 5, e01911. [Google Scholar] [CrossRef] [PubMed]
- Papp, J.R.; Shewen, P.E. Localization of chronic Chlamydia psittaci infection in the reproductive tract of sheep. J. Infect. Dis. 1996, 174, 1296–1302. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Papp, J.R.; Shewen, P.E.; Gartley, C.J. Abortion and subsequent excretion of chlamydiae from the reproductive tract of sheep during estrus. Infect. Immun. 1994, 62, 3786–3792. [Google Scholar] [CrossRef] [PubMed]
- Wilsmore, A.J.; Izzard, K.A.; Wilsmore, B.C.; Dagnall, G.J. Breeding performance of sheep infected with Chlamydia psittaci (ovis) during their preceding pregnancy. Vet. Rec. 1990, 126, 40–41. [Google Scholar]
- Su, H.; Feilzer, K.; Caldwell, H.D.; Morrison, R.P. Chlamydia trachomatis genital tract infection of antibody-deficient gene knockout mice. Infect. Immun. 1997, 65, 1993–1999. [Google Scholar] [CrossRef]
- Moore, T.; Ananaba, G.A.; Bolier, J.; Bowers, S.; Belay, T.; Eko, F.O.; Igietseme, J.U. Fc receptor regulation of protective immunity against Chlamydia trachomatis. Immunology 2002, 105, 213–221. [Google Scholar] [CrossRef]
- Graham, S.P.; Jones, G.E.; MacLean, M.; Livingstone, M.; Entrican, G. Recombinant ovine interferon gamma inhibits the multiplication of Chlamydia psittaci in ovine cells. J. Comp. Pathol. 1995, 112, 185–195. [Google Scholar] [CrossRef]
- Rocchi, M.S.; Wattegedera, S.; Meridiani, I.; Entrican, G. Protective adaptive immunity to Chlamydophila abortus infection and control of ovine enzootic abortion (OEA). Vet. Microbiol. 2009, 135, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Entrican, G.; Wheelhouse, N.; Wattegedera, S.R.; Longbottom, D. New challenges for vaccination to prevent chlamydial abortion in sheep. Comp. Immunol. Microbiol. Infect. Dis. 2012, 35, 271–276. [Google Scholar] [CrossRef] [PubMed]
| Group | Ewes | Number of Lambs | |||||
|---|---|---|---|---|---|---|---|
| No. Pregnant | No. Lambed (%) | No. Aborted (%) | Mean Gestational Length | Viable | Non-Viable 1 | Dead | |
| 1 | 25 | 24 (96) | 1 (4) | 142 | 41 | 0 | 2 2 |
| 2 | 26 | 26 (100) | 0 (0) | 144 | 47 | 0 | 0 |
| 3 | 24 | 24 (100) | 0 (0) | 144 | 39 | 0 | 0 |
| 4 | 25 | 12 (48) | 13 (52) | 135 | 23 | 5 | 20 |
| 5 | 25 | 25 (100) | 0 (0) | 144 | 43 | 0 | 0 |
| Group | Pregnancy Outcome 1 | No. Ewes | Lesions 2 | mZN 3 | Placental qPCR 4 | Swab qPCR 5 |
|---|---|---|---|---|---|---|
| 1 | Lambed | 24 6 | 3+, 21− | 3+, 21− | 9+, 15− | 90.7 (1.65) |
| Aborted | 1 | 1+ | 1+ | 1+ | - 7 | |
| 2 | Lambed | 26 | 0+, 26− | 3+, 23− | 6+, 20− | 49.13 (1.71) |
| 3 | Lambed | 24 | 3+, 21− | 3+, 21− | 14+, 10− | 73.63 (2.2) |
| 4 | Lambed | 12 | 11+, 1− | 11+, 1− | 12+, 0− | 43651.42 (3.39) |
| Aborted | 13 | 13+, 0− | 13+, 0− | 13+, 0− | 919232.3 (1.78) | |
| 5 | Lambed | 25 | 25− | 25− | 25− | 12.14 (1.19) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Livingstone, M.; Wattegedera, S.R.; Palarea-Albaladejo, J.; Aitchison, K.; Corbett, C.; Sait, M.; Wilson, K.; Chianini, F.; Rocchi, M.S.; Wheelhouse, N.; et al. Efficacy of Two Chlamydia abortus Subcellular Vaccines in a Pregnant Ewe Challenge Model for Ovine Enzootic Abortion. Vaccines 2021, 9, 898. https://doi.org/10.3390/vaccines9080898
Livingstone M, Wattegedera SR, Palarea-Albaladejo J, Aitchison K, Corbett C, Sait M, Wilson K, Chianini F, Rocchi MS, Wheelhouse N, et al. Efficacy of Two Chlamydia abortus Subcellular Vaccines in a Pregnant Ewe Challenge Model for Ovine Enzootic Abortion. Vaccines. 2021; 9(8):898. https://doi.org/10.3390/vaccines9080898
Chicago/Turabian StyleLivingstone, Morag, Sean Ranjan Wattegedera, Javier Palarea-Albaladejo, Kevin Aitchison, Cecilia Corbett, Michelle Sait, Kim Wilson, Francesca Chianini, Mara Silvia Rocchi, Nicholas Wheelhouse, and et al. 2021. "Efficacy of Two Chlamydia abortus Subcellular Vaccines in a Pregnant Ewe Challenge Model for Ovine Enzootic Abortion" Vaccines 9, no. 8: 898. https://doi.org/10.3390/vaccines9080898
APA StyleLivingstone, M., Wattegedera, S. R., Palarea-Albaladejo, J., Aitchison, K., Corbett, C., Sait, M., Wilson, K., Chianini, F., Rocchi, M. S., Wheelhouse, N., Entrican, G., & Longbottom, D. (2021). Efficacy of Two Chlamydia abortus Subcellular Vaccines in a Pregnant Ewe Challenge Model for Ovine Enzootic Abortion. Vaccines, 9(8), 898. https://doi.org/10.3390/vaccines9080898








