Propylthiouracil-Induced Antineutrophil Cytoplasmic Antibody-Associated Vasculitis after COVID-19 Vaccination
Abstract
:1. Introduction
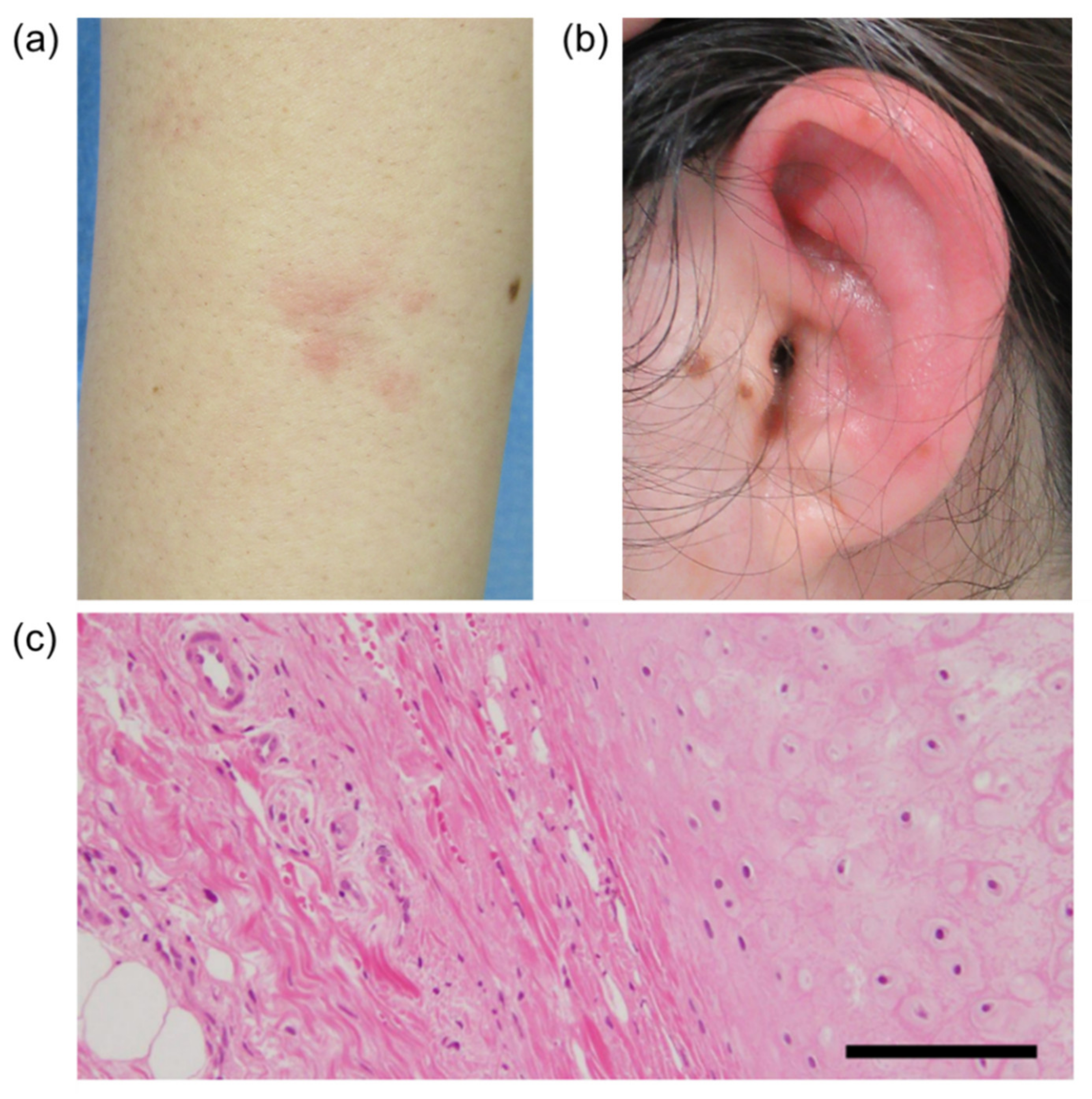
2. Case
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coronavirus Resource Center. Johns Hopkins University and Medicine. Available online: https://coronavirus.jhu.edu/ (accessed on 22 June 2021).
- Lamb, Y.N. BNT162b2 mRNA COVID-19 Vaccine: First Approval. Drugs 2021, 81, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Schultz, N.H.; Sørvoll, I.H.; Michelsen, A.E.; Munthe, L.A.; Lund-Johansen, F.; Ahlen, M.T.; Wiedmann, M.; Aamodt, A.H.; Skattør, T.H.; Tjønnfjord, G.E.; et al. Thrombosis and thrombocytopenia after ChAdOx1 nCoV-19 vaccination. N. Engl. J. Med. 2021, 384, 2124–2130. [Google Scholar] [CrossRef] [PubMed]
- Tarawneh, O.; Tarawneh, H. Immune thrombocytopenia in a 22-year-old post Covid-19 vaccine. Am. J. Hematol. 2021, 96, E133–E134. [Google Scholar] [CrossRef]
- Lee, E.J.; Cines, D.B.; Gernsheimer, T.; Kessler, C.; Michel, M.; Tarantino, M.D.; Semple, J.W.; Arnold, D.M.; Godeau, B.; Lambert, M.P.; et al. Thrombocytopenia following Pfizer and Moderna SARS-CoV-2 vaccination. Am. J. Hematol. 2021, 96, 534–537. [Google Scholar] [CrossRef]
- Fueyo-Rodriguez, O.; Valente-Acosta, B.; Jimenez-Soto, R.; Neme-Yunes, Y.; Inclán-Alarcón, S.I.; Trejo-Gonzalez, R.; García-Salcido, M.Á. Secondary immune thrombocytopenia supposedly attributable to COVID-19 vaccination. BMJ Case Rep. 2021, 14, e242220. [Google Scholar] [CrossRef]
- Idogun, P.O.; Ward, M.C.; Teklie, Y.; Wiese-Rometsch, W.; Baker, J. Newly diagnosed idiopathic thrombocytopenia post COVID-19 vaccine Administration. Cureus 2021, 13, e14853. [Google Scholar]
- Geetha, D.; Jefferson, J.A. ANCA-associated vasculitis: Core curriculum 2020. Am. J. Kidney Dis. 2020, 75, 124–137. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Zhao, M.H. Review article: Drug-induced anti-neutrophil cytoplasmic antibody-associated vasculitis. Nephrology 2009, 14, 33–41. [Google Scholar] [CrossRef]
- Chen, M.; Gao, Y.; Guo, X.H.; Zhao, M.H. Propylthiouracil-induced antineutrophil cytoplasmic antibody-associated vasculitis. Nat. Rev. Nephrol. 2012, 8, 476–483. [Google Scholar] [CrossRef]
- McAdam, L.P.; O’Hanlan, M.A.; Bluestone, R.; Pearson, C.M. Relapsing polychondritis: Prospective study of 23 patients and a review of the literature. Medicine 1976, 55, 193–215. [Google Scholar] [CrossRef]
- Papo, T.; Piette, J.C.; Le, T.H.D.; Godeau, P.; Meyer, O.; Kahn, M.F.; Bourgeois, P. Antineutrophil cytoplasmic antibodies in polychondritis. Ann. Rheum. Dis. 1993, 52, 384–385. [Google Scholar] [CrossRef] [Green Version]
- Xuan, Y.Y.; Li, T.F.; Zhang, L.; Liu, S.Y. ANCA positive relapsing polychondritis, Graves disease, and suspected moyamoya disease: A case report. Medicine 2017, 96, e9378. [Google Scholar] [CrossRef]
- Michet, C.J., Jr.; McKenna, C.H.; Luthra, H.S.; O’Fallon, W.M. Relapsing polychondritis. Survival and predictive role of early disease manifestations. Ann. Intern. Med. 1986, 104, 74–78. [Google Scholar] [CrossRef]
- Trentham, D.E.; Le, C.H. Relapsing polychondritis. Ann. Intern. Med. 1998, 129, 114–122. [Google Scholar] [CrossRef]
- Yates, M.; Watts, R.A.; Bajema, I.M.; Cid, M.C.; Crestani, B.; Hauser, T.; Hellmich, B.; Holle, J.U.; Laudien, M.; Little, M.A.; et al. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann. Rheum. Dis. 2016, 75, 1583–1594. [Google Scholar] [CrossRef] [Green Version]
- Allena, N.; Patel, J.; Nader, G.; Patel, M.; Medvedovsky, B. A rare case of SARS-CoV-2-induced microscopic polyangiitis. Cureus 2021, 13, e15259. [Google Scholar]
- Izci Duran, T.; Turkmen, E.; Dilek, M.; Sayarlioglu, H.; Arik, N. ANCA-associated vasculitis after COVID-19. Rheumatol. Int. 2021, 41, 1523–1529. [Google Scholar] [CrossRef] [PubMed]
- Gulati, K.; Prendecki, M.; Clarke, C.; Willicombe, M.; McAdoo, S. COVID-19 Reinfection in a patient receiving immunosuppressive treatment for antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheumatol. 2021, 73, 1091–1092. [Google Scholar] [CrossRef] [PubMed]
- Misra, D.P.; Thomas, K.N.; Gasparyan, A.Y.; Zimba, O. Mechanisms of thrombosis in ANCA-associated vasculitis. Clin. Rheumatol. 2021, 9, 1–9. [Google Scholar]
- Gapud, E.J.; Kronbichler, A.; Gauckler, P.; Geetha, D. Immunotherapy for ANCA-associated vasculitis during the COVID-19 pandemic. Eur. J. Rheumatol. 2020, 7, S121–S128. [Google Scholar] [CrossRef] [PubMed]
- Hernández, A.F.; Calina, D.; Poulas, K.; Docea, A.O.; Tsatsakis, A.M. Safety of COVID-19 vaccines administered in the EU: Should we be concerned? Toxicol. Rep. 2021, 8, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Segal, Y.; Shoenfeld, Y. Vaccine-induced autoimmunity: The role of molecular mimicry and immune crossreaction. Cell. Mol. Immunol. 2018, 15, 586–594. [Google Scholar] [CrossRef]
- Vojdani, A.; Vojdani, E.; Kharrazian, D. Reaction of human monoclonal antibodies to SARS-CoV-2 proteins with tissue antigens: Implications for autoimmune diseases. Front. Immunol. 2021, 11, 617089. [Google Scholar] [CrossRef]
- Vojdani, A.; Kharrazian, D. Potential antigenic cross-reactivity between SARS-CoV-2 and human tissue with a possible link to an increase in autoimmune diseases. Clin. Immunol. 2020, 217, 108480. [Google Scholar] [CrossRef]
- Talotta, R. Do COVID-19 RNA-based vaccines put at risk of immune-mediated diseases? In reply to “potential antigenic cross-reactivity between SARS-CoV-2 and human tissue with a possible link to an increase in autoimmune diseases”. Clin. Immunol. 2021, 224, 108665. [Google Scholar] [CrossRef] [PubMed]
- Sera, N.; Ashizawa, K.; Ando, T.; Abe, Y.; Ide, A.; Usa, T.; Tominaga, T.; Ejima, E.; Yokoyama, N.; Eguchi, K. Treatment with propylthiouracil is associated with appearance of antineutrophil cytoplasmic antibodies in some patients with Graves’ disease. Thyroid 2000, 10, 595–599. [Google Scholar] [CrossRef]
- Noh, J.Y.; Yasuda, S.; Sato, S.; Matsumoto, M.; Kunii, Y.; Noguchi, Y.; Mukasa, K.; Ito, K.; Ito, K.; Sugiyama, O.; et al. Clinical characteristics of myeloperoxidase antineutrophil cytoplasmic antibody-associated vasculitis caused by antithyroid drugs. J. Clin. Endocrinol. Metab. 2009, 94, 2806–2811. [Google Scholar] [CrossRef] [Green Version]
| Complete blood counts: | Total bilirubin | 0.5 | mg/dL | ||
| White blood cells | 7490 | /μL | Aspartate transaminase | 12 | U/L |
| Neutrophils | 69.2 | % | Alanine aminotransferase | 13 | U/L |
| Lymphocytes | 21.6 | % | Lactate dehydrogenase | 127 | U/L |
| Red blood cells | 434 × 104 | /μL | Creatinine kinase | 13 | U/L |
| Hemoglobin | 12.7 | g/dL | TSH | 2.46 | μIU/mL |
| Hematocrit | 38.4 | % | Free thyroxine | 0.99 | ng/dL |
| Platelets | 46.7 × 104 | /μL | Immunology: | ||
| Urinalysis: | Rheumatoid factor | negative | |||
| Protein | 22 | mg/dL | Antinuclear antibody | negative | |
| Creatinine | 239.2 | mg/dL | Anti-ds-DNA antibody | negative | |
| Estimated urine protein | 0.1 | g/day | Anti-GBM antibody | negative | |
| Biochemistry: | PR3-ANCA | 28.3 | U/mL | ||
| C-reactive protein | 10.16 | mg/dL | MPO-ANCA | 494 | U/mL |
| Blood urea nitrogen | 9.5 | mg/dL | CH50 | 83 | U/mL |
| Creatinine | 0.62 | mg/dL | IgG | 1366 | mg/dL |
| Total protein | 8.1 | g/dL | IgA | 245 | mg/dL |
| Albumin | 4 | g/dL | IgM | 247 | mg/dL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okuda, S.; Hirooka, Y.; Sugiyama, M. Propylthiouracil-Induced Antineutrophil Cytoplasmic Antibody-Associated Vasculitis after COVID-19 Vaccination. Vaccines 2021, 9, 842. https://doi.org/10.3390/vaccines9080842
Okuda S, Hirooka Y, Sugiyama M. Propylthiouracil-Induced Antineutrophil Cytoplasmic Antibody-Associated Vasculitis after COVID-19 Vaccination. Vaccines. 2021; 9(8):842. https://doi.org/10.3390/vaccines9080842
Chicago/Turabian StyleOkuda, Saki, Yasuaki Hirooka, and Masafumi Sugiyama. 2021. "Propylthiouracil-Induced Antineutrophil Cytoplasmic Antibody-Associated Vasculitis after COVID-19 Vaccination" Vaccines 9, no. 8: 842. https://doi.org/10.3390/vaccines9080842
APA StyleOkuda, S., Hirooka, Y., & Sugiyama, M. (2021). Propylthiouracil-Induced Antineutrophil Cytoplasmic Antibody-Associated Vasculitis after COVID-19 Vaccination. Vaccines, 9(8), 842. https://doi.org/10.3390/vaccines9080842






