Clinical Characteristics of Hospitalized COVID-19 Patients Who Received at Least One Dose of COVID-19 Vaccine
Abstract
:1. Introduction
2. Material and Methods
2.1. Data Extraction
2.2. Statistical Analysis
3. Results
3.1. Demographic Characteristics
3.2. Clinical Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rzymski, P.; Borkowski, L.; Drąg, M.; Flisiak, R.; Jemielity, J.; Krajewski, J.; Mastalerz-Migas, A.; Matyja, A.; Pyrć, K.; Simon, K.; et al. The Strategies to Support the COVID-19 Vaccination with Evidence-Based Communication and Tackling Misinformation. Vaccines 2021, 9, 109. [Google Scholar] [CrossRef] [PubMed]
- Kashte, S.; Gulbake, A.; El-Amin Iii, S.F.; Gupta, A. COVID-19 vaccines: Rapid development, implications, challenges and future prospects. Hum. Cell 2021, 34, 711–733. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Sadoff, J.; Le Gars, M.; Shukarev, G.; Heerwegh, D.; Truyers, C.; de Groot, A.M.; Stoop, J.; Tete, S.; Van Damme, W.; Leroux-Roels, I.; et al. Interim Results of a Phase 1-2a Trial of Ad26.COV2.S Covid-19 Vaccine. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2020, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Zeyland, J.; Poniedziałek, B.; Małecka, I.; Wysocki, J. The Perception and Attitudes toward COVID-19 Vaccines: A Cross-Sectional Study in Poland. Vaccines 2021, 9, 382. [Google Scholar] [CrossRef] [PubMed]
- Forman, R.; Shah, S.; Jeurissen, P.; Jit, M.; Mossialos, E. COVID-19 vaccine challenges: What have we learned so far and what remains to be done? Health Policy 2021, 125, 553–567. [Google Scholar] [CrossRef]
- Chen, J.; Lu, H. New challenges to fighting COVID-19: Virus variants, potential vaccines, and development of antivirals. Biosci. Trends 2021, 15, 126–128. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Perek, B.; Flisiak, R. Thrombotic Thrombocytopenia after COVID-19 Vaccination: In Search of the Underlying Mechanism. Vaccines 2021, 9, 559. [Google Scholar] [CrossRef] [PubMed]
- Walsh, E.E.; Frenck, R.W., Jr.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R.; et al. Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.R.; Belij-Rammerstorfer, S.; Dold, C.; Ewer, K.J.; Folegatti, P.M.; Gilbride, C.; Halkerston, R.; Hill, J.; Jenkin, D.; Stockdale, L.; et al. Phase 1/2 trial of SARS-CoV-2 vaccine ChAdOx1 nCoV-19 with a booster dose induces multifunctional antibody responses. Nat. Med. 2021, 27, 279–288. [Google Scholar] [CrossRef]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J.; et al. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Andrée, M.; Moskorz, W.; Drexler, I.; Walotka, L.; Grothmann, R.; Ptok, J.; Hillebrandt, J.; Ritchie, A.; Rabl, D.; et al. Age-dependent immune response to the Biontech/Pfizer BNT162b2 COVID-19 vaccination. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2021. [Google Scholar] [CrossRef]
- Monin, L.; Laing, A.G.; Muñoz-Ruiz, M.; McKenzie, D.R.; Del Molino Del Barrio, I.; Alaguthurai, T.; Domingo-Vila, C.; Hayday, T.S.; Graham, C.; Seow, J.; et al. Safety and immunogenicity of one versus two doses of the COVID-19 vaccine BNT162b2 for patients with cancer: Interim analysis of a prospective observational study. Lancet Oncol. 2021. [Google Scholar] [CrossRef]
- Boyarsky, B.J.; Werbel, W.A.; Avery, R.K.; Tobian, A.A.R.; Massie, A.B.; Segev, D.L.; Garonzik-Wang, J.M. Immunogenicity of a Single Dose of SARS-CoV-2 Messenger RNA Vaccine in Solid Organ Transplant Recipients. JAMA 2021, 325, 1784–1786. [Google Scholar] [CrossRef]
- Agha, M.; Blake, M.; Chilleo, C.; Wells, A.; Haidar, G. Suboptimal response to COVID-19 mRNA vaccines in hematologic malignancies patients. medRxiv 2021. [Google Scholar] [CrossRef]
- Lauer, S.A.; Grantz, K.H.; Bi, Q.; Jones, F.K.; Zheng, Q.; Meredith, H.R.; Azman, A.S.; Reich, N.G.; Lessler, J. The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application. Ann. Intern. Med. 2020, 172, 577–582. [Google Scholar] [CrossRef] [Green Version]
- Flisiak, R.; Jaroszewicz, J.; Rogalska, M.; Łapiński, T.; Berkan-Kawińska, A.; Bolewska, B.; Tudrujek-Zdunek, M.; Kozielewicz, D.; Rorat, M.; Leszczyński, P.; et al. Tocilizumab Improves the Prognosis of COVID-19 in Patients with High IL-6. J. Clin. Med. 2021, 10, 1583. [Google Scholar] [CrossRef]
- Flisiak, R.; Horban, A.; Jaroszewicz, J.; Kozielewicz, D.; Pawłowska, M.; Parczewski, M.; Piekarska, A.; Simon, K.; Tomasiewicz, K.; Zarębska-Michaluk, D. Management of SARS-CoV-2 infection: Recommendations of the Polish Association of Epidemiologists and Infectiologists as of 31 March 2020. Pol. Arch. Intern. Med. 2020, 130, 352–357. [Google Scholar] [CrossRef] [Green Version]
- Leung, C. The Incubation Period of COVID-19: Current Understanding and Modeling Technique. Adv. Exp. Med. Biol. 2021, 1318, 81–90. [Google Scholar] [CrossRef]
- CDC COVID-19 Vaccine Breakthrough Case Investigations Team. COVID-19 Vaccine Breakthrough Infections Reported to CDC—United States, 1 January–30 April 2021. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70, 792–793. [Google Scholar] [CrossRef]
- Pritchard, E.; Matthews, P.C.; Stoesser, N.; Eyre, D.W.; Gethings, O.; Vihta, K.D.; Jones, J.; House, T.; VanSteenHouse, H.; Bell, I.; et al. Impact of Vaccination on New SARS-CoV-2 Infections in the United Kingdom. Nat. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.; Reed, A.B.; Ponzo, S.; Yassaee, A.; Aral, M.; Plans, D.; Labrique, A.; Mohan, D. Population risk factors for severe disease and mortality in COVID-19: A global systematic review and meta-analysis. PLoS ONE 2021, 16, e0247461. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, A.L.V.; Turner, N.; Omer, I.; Walker, M.K.; Beaulieu, R.M.; Sheikh, M.; Spires, S.S.; Fiske, C.T.; Dare, R.; Goorha, S.; et al. Risk Factors for Mortality and Progression to Severe COVID-19 Disease in the Southeast United States (US): A Report from the SEUS Study Group. Infect. Control Hosp. Epidemiol. 2021, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Crooke, S.N.; Ovsyannikova, I.G.; Poland, G.A.; Kennedy, R.B. Immunosenescence and human vaccine immune responses. Immun. Ageing 2019, 16, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahase, E. Covid vaccine: Prioritise over 16s living with immunosuppressed adults, says JCVI. BMJ 2021, 372, n852. [Google Scholar] [CrossRef]
- Charkiewicz, R.; Nikliński, J.; Biecek, P.; Kiśluk, J.; Pancewicz, S.; Moniuszko-Malinowska, A.M.; Flisiak, R.; Krętowski, A.J.; Dzięcioł, J.; Moniuszko, M.; et al. The first SARS-CoV-2 genetic variants of concern (VOC) in Poland: The concept of a comprehensive approach to monitoring and surveillance of emerging variants. Adv. Med Sci. 2021, 66, 237–245. [Google Scholar] [CrossRef]
- Hacisuleyman, E.; Hale, C.; Saito, Y.; Blachere, N.E.; Bergh, M.; Conlon, E.G.; Schaefer-Babajew, D.J.; DaSilva, J.; Muecksch, F.; Gaebler, C.; et al. Vaccine Breakthrough Infections with SARS-CoV-2 Variants. N. Engl. J. Med. 2021, 384, 2212–2218. [Google Scholar] [CrossRef]
- Zhou, D.; Dejnirattisai, W.; Supasa, P.; Liu, C.; Mentzer, A.J.; Ginn, H.M.; Zhao, Y.; Duyvesteyn, H.M.E.; Tuekprakhon, A.; Nutalai, R.; et al. Evidence of escape of SARS-CoV-2 variant B.1.351 from natural and vaccine-induced sera. Cell 2021, 184, 2348–2361.e6. [Google Scholar] [CrossRef]
- McEwen, A.E.; Cohen, S.; Bryson-Cahn, C.; Liu, C.; Pergam, S.A.; Lynch, J.; Schippers, A.; Strand, K.; Whimbey, E.; Mani, N.S.; et al. Variants of concern are overrepresented among post-vaccination breakthrough infections of SARS-CoV-2 in Washington State. medRxiv 2021. [Google Scholar] [CrossRef]
- Geers, D.; Shamier, M.C.; Bogers, S.; den Hartog, G.; Gommers, L.; Nieuwkoop, N.N.; Schmitz, K.S.; Rijsbergen, L.C.; van Osch, J.A.T.; Dijkhuizen, E.; et al. SARS-CoV-2 variants of concern partially escape humoral but not T-cell responses in COVID-19 convalescent donors and vaccinees. Sci. Immunol. 2021, 6, eabj1750. [Google Scholar] [CrossRef]
- SARS-CoV-2 w Polsce. Mapa Wariantów (Map of Variants). Available online: https://sarswpolsce.pl/?page_id=7 (accessed on 22 June 2021).
- Haas, E.J.; Angulo, F.J.; McLaughlin, J.M.; Anis, E.; Singer, S.R.; Khan, F.; Brooks, N.; Smaja, M.; Mircus, G.; Pan, K.; et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data. Lancet 2021, 397, 1819–1829. [Google Scholar] [CrossRef]
- Iacobucci, G. Covid-19: Single vaccine dose is 33% effective against variant from India, data show. BMJ 2021, 373, n1346. [Google Scholar] [CrossRef]
- Madhi, S.A.; Baillie, V.; Cutland, C.L.; Voysey, M.; Koen, A.L.; Fairlie, L.; Padayachee, S.D.; Dheda, K.; Barnabas, S.L.; Bhorat, Q.E.; et al. Efficacy of the ChAdOx1 nCoV-19 Covid-19 Vaccine against the B.1.351 Variant. N. Engl. J. Med. 2021, 384, 1885–1898. [Google Scholar] [CrossRef] [PubMed]
- Abu-Raddad, L.J.; Chemaitelly, H.; Butt, A.A. Effectiveness of the BNT162b2 Covid-19 Vaccine against the B.1.1.7 and B.1.351 Variants. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
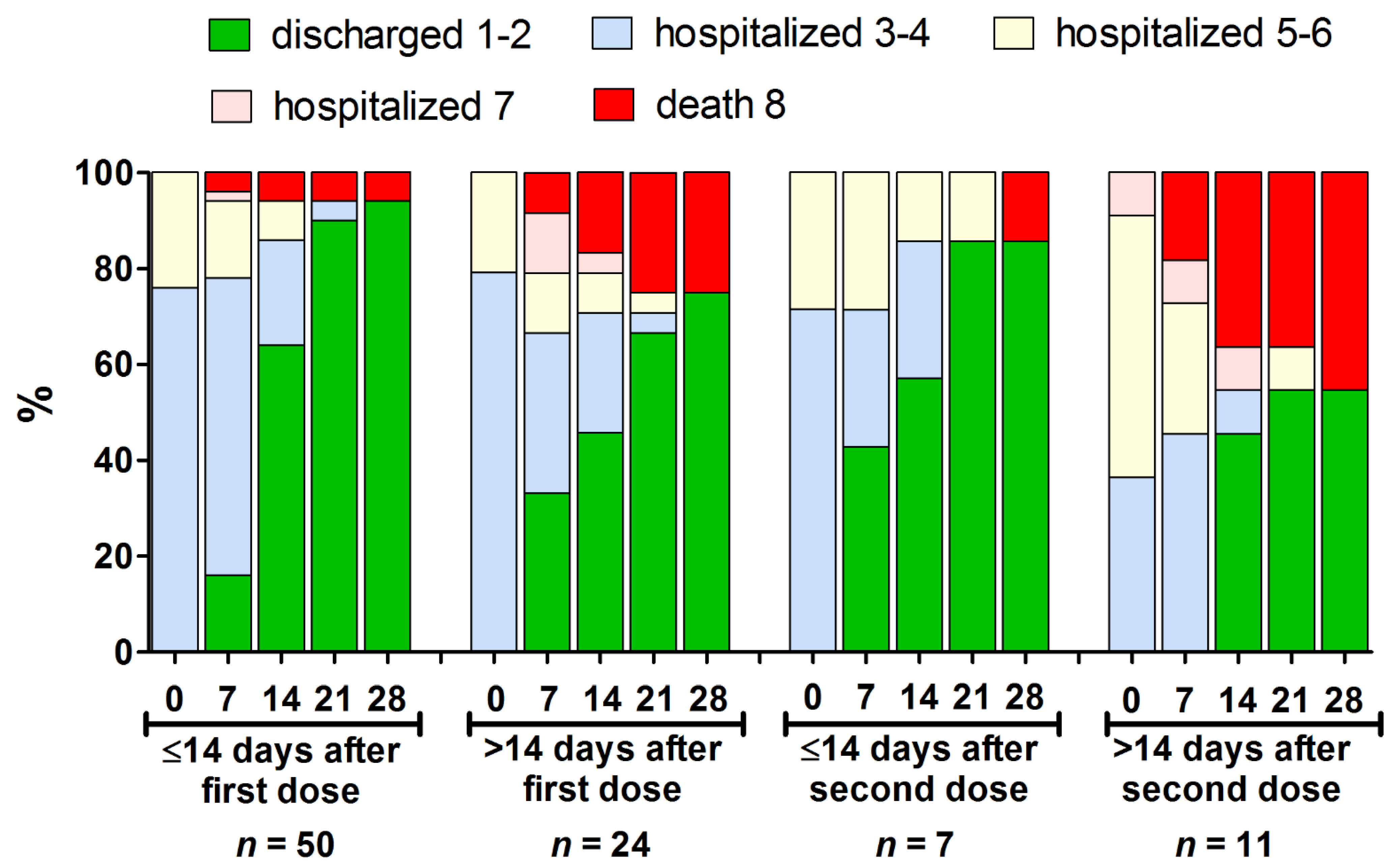
| Parameter | All (n = 92) | ≤14 Days after 1st Dose (n = 50) | >14 Days after 1st Dose (n = 24) | ≤14 Days after 2nd Dose (n = 7) | >14 Days after 2nd Dose (n = 11) |
|---|---|---|---|---|---|
| Age Mean ± SD, years Range, years >70 years, n (%) | 68.3 ± 11.9 32–93 40 (43.5) | 65.5 ± 11.5 a 32–89 16 (32.0) | 69.9 ± 10.2 ab 46–89 12 (50.0) | 73.4 ± 6.9 ab 67–85 4 (57.1) | 78.5 ± 14.3 b 52–93 8 (72.7) |
| Gender Male, n (%) Female, n (%) | 58 (63.0) 34 (37.0) | a 31 (62.0) 19 (38.0) | a 15 (62.5) 9 (37.5) | a 4 (57.1) 3 (42.9) | a 8 (72.7) 3 (27.3) |
| BMI Mean ± SD, kg/m2 Underweight Normal (18.5–24.9), n (%) Overweight (25.0–29.9), n (%) Obesity (≥30.0), n (%) | 28.7 ± 5.4 1 (1.1) 23 (25.0) 36 (39.1) 32 (34.8) | 28.6 ± 4.5 a 1 (2.0) 8 (16.0) 21 (42.0) 20 (40.0) | 29.5 ± 7.3 a 0 (0.0) 8 (33.3) 8(33.3) 8 (33.3) | 28.7 ± 3.8 a 0 (0.0) 2 (28.6) 3 (42.9) 2 (28.6) | 27.4 ± 6.3 a 0 (0.0) 5 (45.5) 4 (36.4) 2 (18.2) |
| Chronic comorbidities, n (%) | 82 (89.1) | 41 (82.0) a | 23 (95.8) a | 7 (100.0) a | 11 (100.0) a |
| Immunosuppression, n (%) | 7 (7.6) | 0 (0.0) | 3 (12.5) | 1 (14.3) | 3 (27.3) |
| Vaccine BNT162b2, n (%) mRNA-1273, n (%) AZD1222, n (%) | 51 (55.4) 13 (14.1) 28 (30.4) | a 22 (44.0) 9 (18.0) 19 (38.0) | a 12 (50.0) 3 (12.5) 9 (37.5) | b 6 (85.7) 1 (14.3) 0 (0.0) | b 11 (100.0) 0 (0,0) 0 (0.0) |
| Confirmed vaccine non-responders, n (%) | 4 (4.4) | - | 2 (8.3) | 1 (14.3) | 1 (9.1) |
| Parameter | Group | |||
|---|---|---|---|---|
| ≤14 Days after 1st Dose (n = 50) | >14 Days after 1st Dose (n = 24) | ≤14 Days after 2nd Dose (n = 7) | >14 Days after 2nd Dose (n = 11) | |
| Baseline WHO, median (IQR) | 4 (4-4) a | 4 (4-4) a | 4 (4-5) a | 5 (4-6) a |
| Fever > 38 °C, % | 62.1 a | 62.5 a | 57.1 a | 36.4 a |
| SpO2 < 90%, % | 30.0 a | 54.2 a | 28.6 a | 54.5 a |
| Lung involvement, % | 30 (20–40) a | 30 (25–47.5) a | 30 (10–50) a | 24 (15–70) a |
| WBC * (×103/µL), median (IQR) | 6.2 (5.1–8.2) a | 7.2 (5.1–9.3) a | 9.3 (5.7–12.9) a | 7.1 (5.4–13.3) a |
| ALC nadir * (×103/µL, median (IQR) | 1.0 (0.8–1.3) a | 0.9 (0.6–2.0) a | 1.3 (0.9–2.5) a | 0.7 (0.6–1.0) a |
| IL-6 (pg/mL), median (IQR) | 63.3 (37.1–139.1) a | 47.8 (23.6–138.1) a | 35.6 (17.4–72.6) a | 112.5 (73.0–208.7) a |
| CRP (mg/L), median (IQR) | 62.8 (39.1–143.9) a | 76.8 (45.1–200.5) a | 82.0 (3.2–108.0) a | 113.0 (73.0–208.7) a |
| PCT (ng/mL), median (IQR) | 0.09 (0.05–0.14) a | 0.11 (0.05–0.31) a | 0.11 (0.05–0.16) a | 0.09 (0.05–0.18) a |
| Fatal outcome, n (%) | 3 (6.0) a | 6 (25) b | 1 (14.3) ab | 5 (45.5) b |
| Patients | Group | All | |||
|---|---|---|---|---|---|
| ≤14 Days after 1st Dose | >14 Days after 1st Dose | ≤14 Days after 2nd Dose | >14 Days after 2nd Dose | ||
| Hospitalized | 50 | 24 | 7 | 11 | 92 |
| % of all hospitalized (n = 7552) | 0.66 | 0.32 | 0.09 | 0.15 | 1.22 |
| Fatal cases | 3 | 6 | 1 | 5 | 15 |
| % of all fatal cases (n = 1413) | 0.21 | 0.42 | 0.07 | 0.35 | 1.06 |
| Parameter | Group | |||
|---|---|---|---|---|
| ≤14 Days after 1st Dose (n = 3) | >14 Days after 1st Dose (n = 6) | ≤14 Days after 2nd Dose (n = 1) | >14 Days after 2nd Dose (n = 5) | |
| Age, mean ± SD, years >70 years, n (%) | 71.7 ± 3.2 2 (66.6) | 77.0 ± 8.7 5 (83.3) | 74 1 (100) | 77.4 ± 14.3 4 (80.0) |
| Gender Male, n (%) / Female, n (%) | 1 (33.3)/2 (66.6) | 3 (50.0)/3 (50.0) | 1 (100.0)/0 (0.0) | 4 (80.0)/1 (20.0) |
| BMI, mean ± SD, kg/m2 Normal (18.5–24.9), n (%) Overweight (25.0–29.9), n (%) Obesity (≥30.0), n (%) | 31.7 ± 3.2 0 (0) 1 (33.3) 2 (66.6) | 24.7 ± 3.4 4 (66.6) 2 (33.3) 0 (0) | 21.7 1 (100) 0 (0) 0 (0) | 24.5 ± 0.75 3 (60.0) 2 (40.0) 0 (0) |
| Chronic comorbidities, n (%) CVD, n (%) Diabetes, n (%) | 100 (100) 100 (100) 100 (100) | 5 (83.3) 4 (66.6) 1 (16.6) | 100 (100) 0 (0) 0 (0) | 100 (100) 4 (80) 3 (60) |
| Immunosuppression, n (%) | 0 (0) | 0 (0) | 1 (100) | 2 (40) |
| Confirmed vaccine non-responders, n (%) | - | 1 (16.7) | 1 (100) | 1 (20.0) |
| Baseline WHO, median (IQR) | 4 (4-4) | 4 (4-5) | 3 | 6 (5-6) |
| Fever >38 °C, % | 2 (66.6) | 3 (50.0) | 1 (100.0) | 0 (0.0) |
| SpO2 <90%, % | 2 (66.6) | 4 (66.6) | 0 (0) | 5 (100.0) |
| Lung involvement, % | 40 (30–40) | 55 (40–60) | 60 | 70 (50–70) |
| WBC (×103/µL),median (IQR) | 6.0 (5.9–11.9) | 7.3 (3.5–15.4) | 19.5 * | 8.8 (7.1–14.5) |
| ALC nadir (×103/µL), median (IQR) | 1 (0.4–1.4) | 0.6 (0.3–2.0) | 15.0 * | 0.6 (0.5–0.7) |
| IL-6 (pg/mL),median (IQR) | n.a. | 138 (134–142) | 40.8 | 122 (103–228) |
| CRP (mg/L), median (IQR) | 207 (61–226) | 136 (43–238) | 82 | 125 (22–199) |
| PCT (ng/mL), median (IQR) | 0.3 (0.06–0.6) | 0.1 (0.1–0.6) | 0.12 | 0.2 (0.2–0.2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rzymski, P.; Pazgan-Simon, M.; Simon, K.; Łapiński, T.; Zarębska-Michaluk, D.; Szczepańska, B.; Chojnicki, M.; Mozer-Lisewska, I.; Flisiak, R. Clinical Characteristics of Hospitalized COVID-19 Patients Who Received at Least One Dose of COVID-19 Vaccine. Vaccines 2021, 9, 781. https://doi.org/10.3390/vaccines9070781
Rzymski P, Pazgan-Simon M, Simon K, Łapiński T, Zarębska-Michaluk D, Szczepańska B, Chojnicki M, Mozer-Lisewska I, Flisiak R. Clinical Characteristics of Hospitalized COVID-19 Patients Who Received at Least One Dose of COVID-19 Vaccine. Vaccines. 2021; 9(7):781. https://doi.org/10.3390/vaccines9070781
Chicago/Turabian StyleRzymski, Piotr, Monika Pazgan-Simon, Krzysztof Simon, Tadeusz Łapiński, Dorota Zarębska-Michaluk, Barbara Szczepańska, Michał Chojnicki, Iwona Mozer-Lisewska, and Robert Flisiak. 2021. "Clinical Characteristics of Hospitalized COVID-19 Patients Who Received at Least One Dose of COVID-19 Vaccine" Vaccines 9, no. 7: 781. https://doi.org/10.3390/vaccines9070781
APA StyleRzymski, P., Pazgan-Simon, M., Simon, K., Łapiński, T., Zarębska-Michaluk, D., Szczepańska, B., Chojnicki, M., Mozer-Lisewska, I., & Flisiak, R. (2021). Clinical Characteristics of Hospitalized COVID-19 Patients Who Received at Least One Dose of COVID-19 Vaccine. Vaccines, 9(7), 781. https://doi.org/10.3390/vaccines9070781









