Evaluation of SARS-CoV-2 Spike Protein Antibody Titers in Cord Blood after COVID-19 Vaccination during Pregnancy in Polish Healthcare Workers: Preliminary Results
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Characteristics
2.2. Laboratory Methods
2.3. Collection of Variables
2.4. Statistical Analysis
3. Results
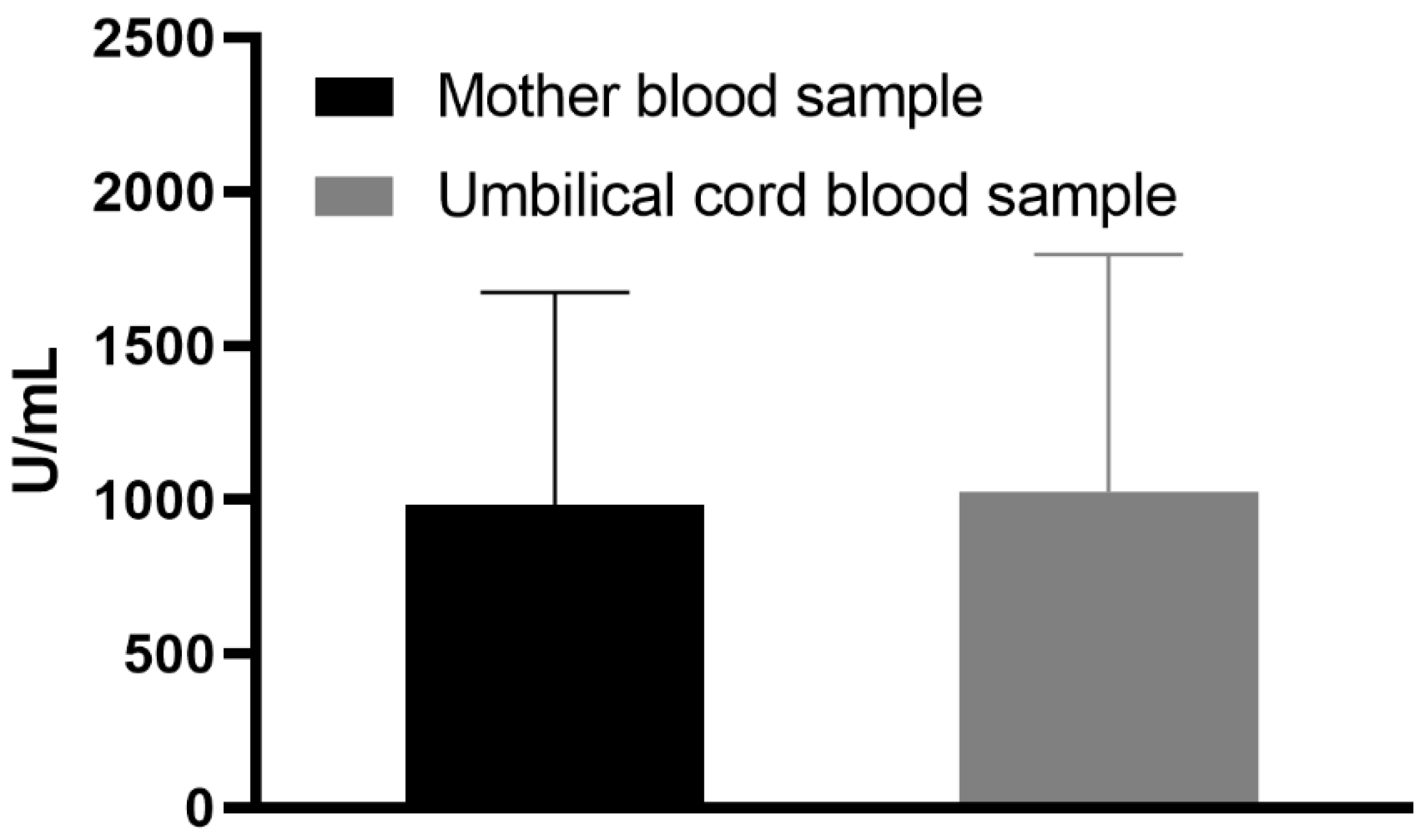
3.1. Main Characteristics of the Studied Population
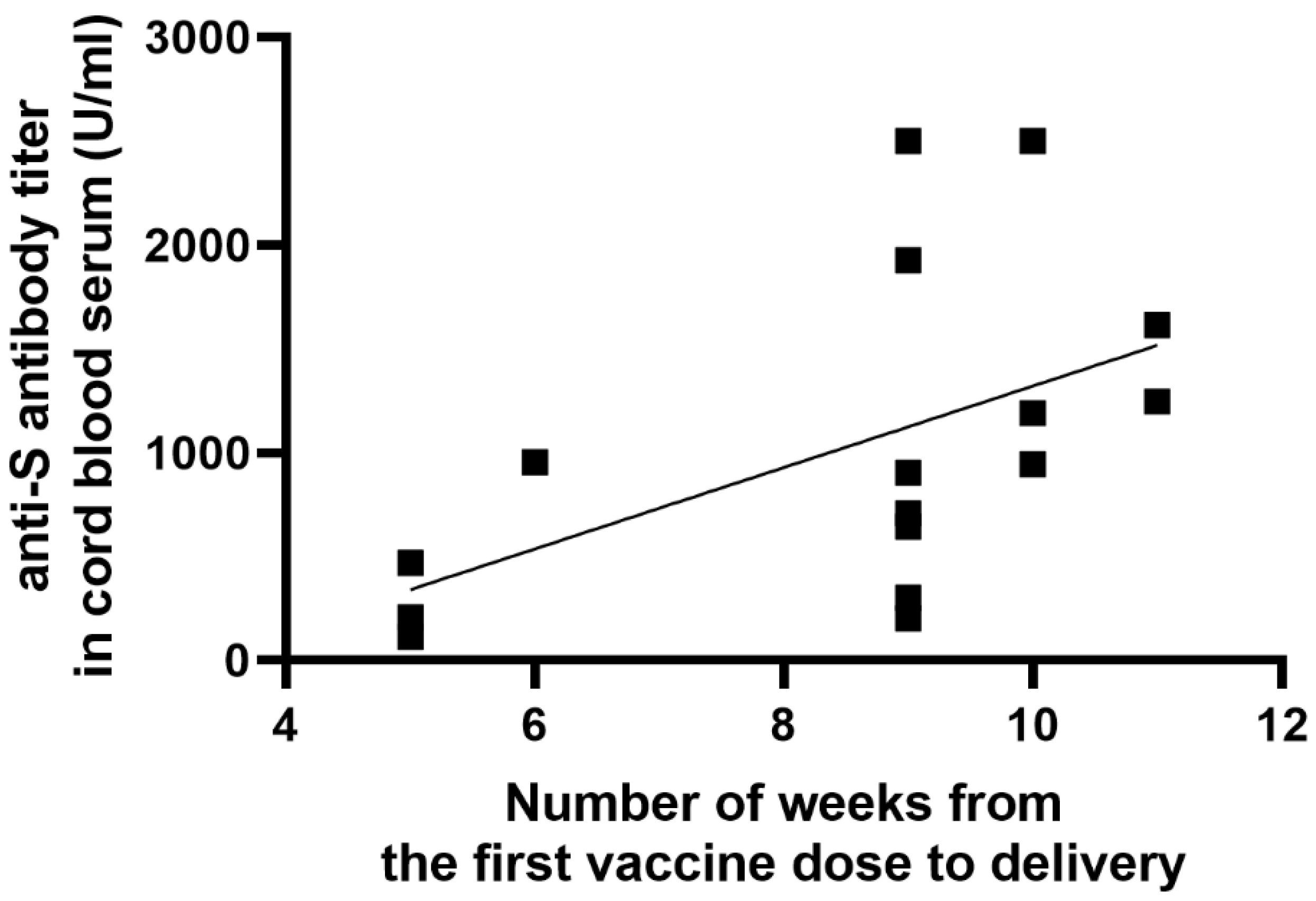
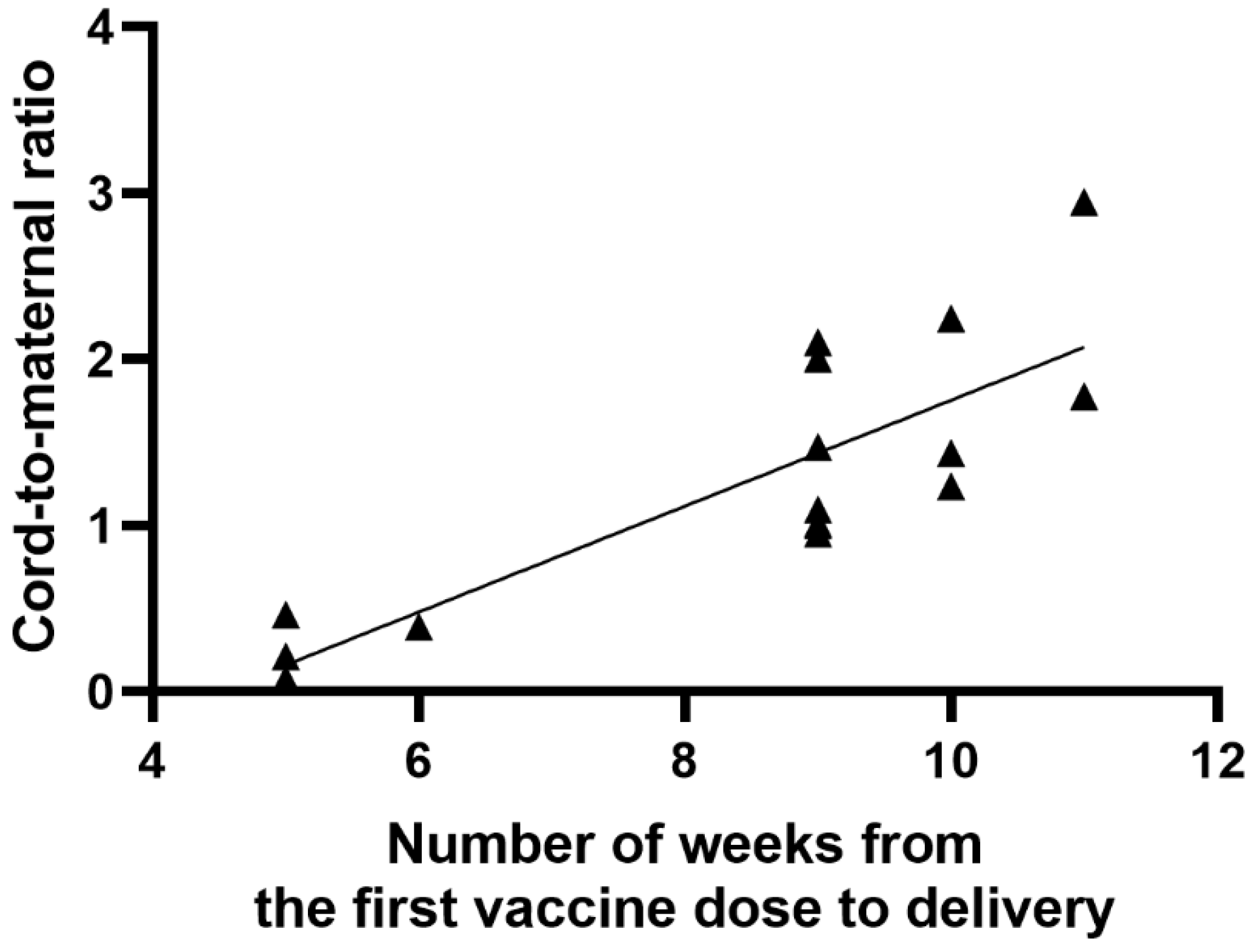
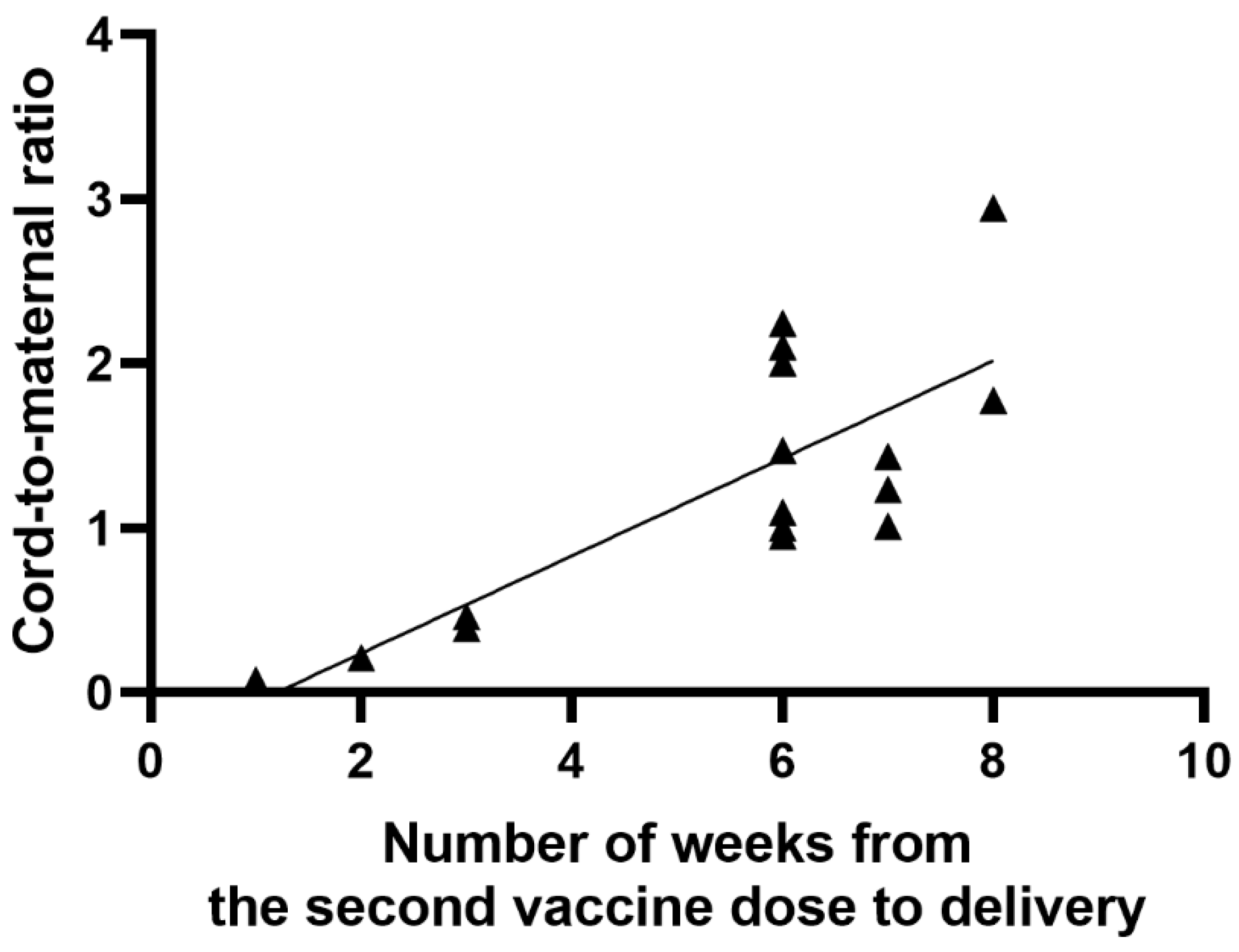
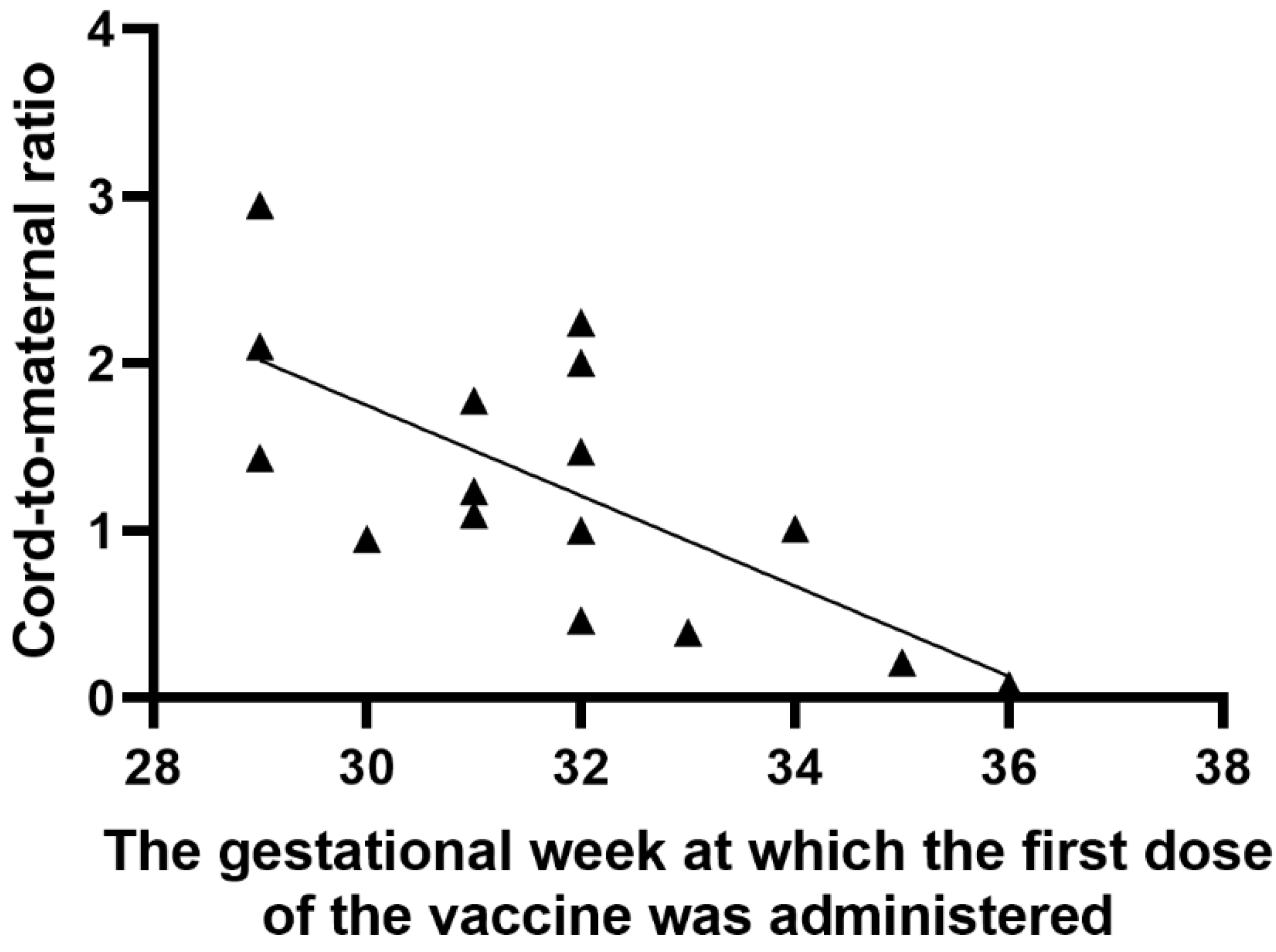
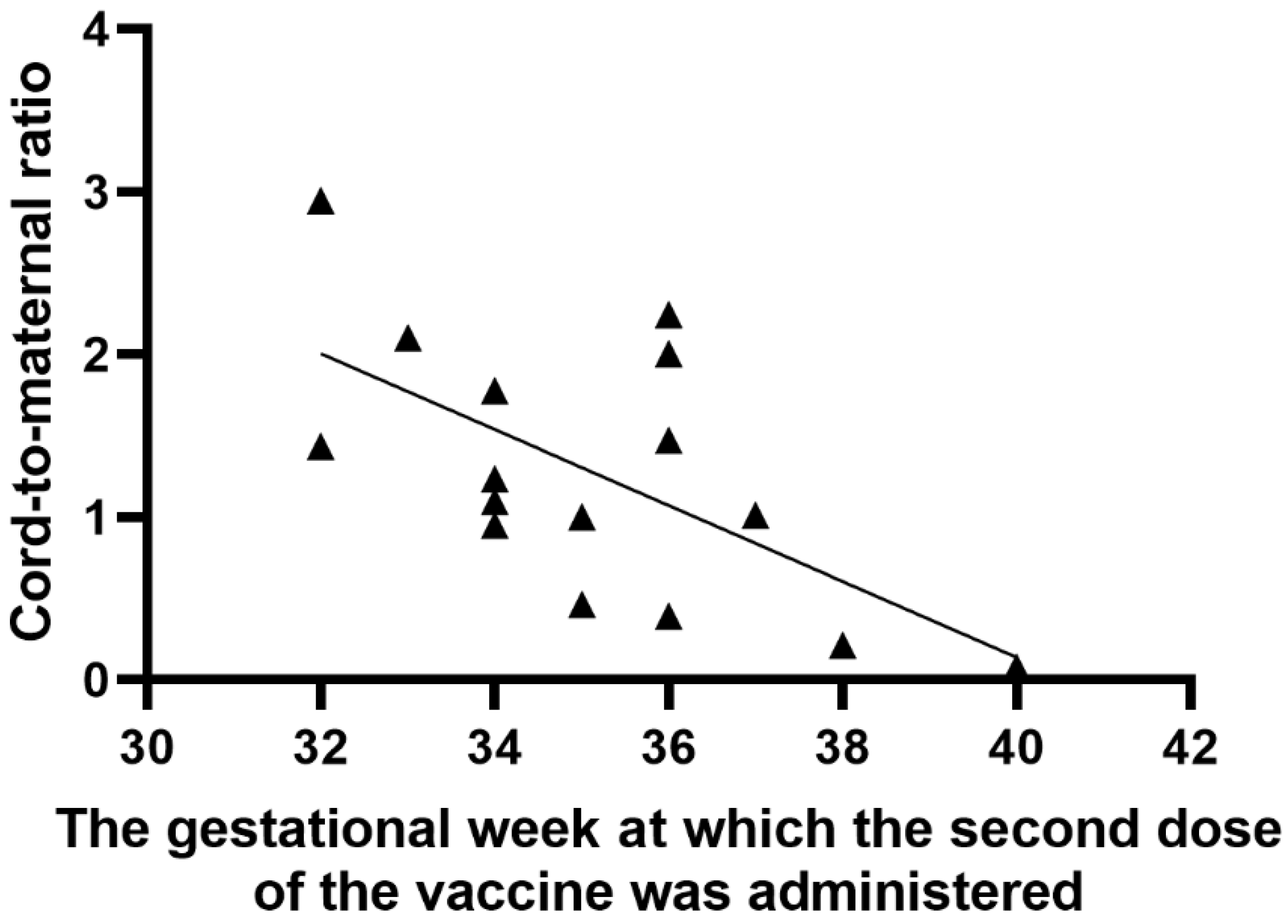
3.2. Characteristics of Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Masmejan, S.; Pomar, L.; Lepigeon, K.; Favre, G.; Baud, D.; Rieder, W. COVID-19 et grossesse [COVID-19 and pregnancy]. Rev. Med. Suisse 2020, 16, 944–946. [Google Scholar] [PubMed]
- Rasmussen, S.A.; Kelley, C.F.; Horton, J.P.; Jamieson, D.J. Coronavirus disease 2019 (COVID-19) vaccines and pregnancy: What obstetricians need to know. Obstet. Gynecol. 2021, 137, 408–414. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Procedure No. EMEA/H/C/005735/0000. Assessment Report: COVID-19 mRNA Vaccine (Nucleoside-Modified). Available online: https://www.ema.europa.eu/en/documents/assessment-report/comirnaty-epar-public-assessment-report_en.pdf (accessed on 6 April 2021).
- Stafford, I.A.; Parchem, J.G.; Sibai, B.M. The coronavirus disease 2019 vaccine in pregnancy: Risks, benefits, and recommendations. Am. J. Obstet. Gynecol. 2021, 224, 484–495. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 (Coronavirus Disease): People with Certain Medical Conditions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html (accessed on 6 April 2021).
- COVID-19 (Coronavirus Disease): Information about COVID-19 Vaccines for People Who Are Pregnant or Breastfeeding. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/recommendations/pregnancy.html (accessed on 6 April 2021).
- Royal College of Obstetricians and Gynaecologists. Updated Advice on COVID-19 Vaccination in Pregnancy and Women Who Are Breastfeeding. Available online: https://www.rcog.org.uk/en/news/updated-advice-on-covid-19-vaccination-in-pregnancy-and-women-who-are-breastfeeding/ (accessed on 6 April 2021).
- Roberts, J.N.; Gruber, M.F. Regulatory considerations in the clinical development of vaccines indicated for use during pregnancy. Vaccine 2015, 33, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Abu Raya, B.; Edwards, K.M.; Scheifele, D.W.; Halperin, S.A. Pertussis and influenza immunisation during pregnancy: A landscape review. Lancet Infect. Dis. 2017, 17, e209–e222. [Google Scholar] [CrossRef]
- Letko, M.; Marzi, A.; Munster, V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat. Microbiol. 2020, 5, 562–569. [Google Scholar] [CrossRef] [PubMed]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zhong, L.; Deng, J.; Peng, J.; Dan, H.; Zeng, X.; Li, T.; Chen, Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef] [PubMed]
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D.; et al. COVID-19 vaccine BNT162b1 elicits human antibody and Th1 T cell responses. Nature 2020, 586, 594–599. [Google Scholar] [CrossRef] [PubMed]
- L’Huillier, A.G.; Meyer, B.; Andrey, D.O.; Arm-Vernez, I.; Baggio, S.; Didierlaurent, A.; Eberhardt, C.S.; Eckerle, I.; Grasset-Salomon, C.; Huttner, A.; et al. Antibody persistence in the first 6 months following SARS-CoV-2 infection among hospital workers: A prospective longitudinal study. Clin. Microbiol. Infect. 2021, 27, 784.e1–784.e8. [Google Scholar]
- Higgins, V.; Fabros, A.; Kulasingam, V. Quantitative measurement of anti-SARS-CoV-2 antibodies: Analytical and clinical evaluation. J. Clin. Microbiol. 2021, 59. [Google Scholar] [CrossRef] [PubMed]
- The American College of Obstetricians and Gynecologists. Vaccinating Pregnant and Lactating Patients against COVID-19. 2020. Available online: https://www.acog.org/en/clinical/clinical-guidance/practice-advisory/articles/2020/12/vaccinating-Pregnant-and-Lactating-Patients-Against-COVID-19 (accessed on 3 January 2021).
- Society for Maternal-Fetal Medicine (SMFM) Statement. SARS-Co-V-2 Vaccination in Pregnancy. Available online: https://s3.amazonaws.com/cdn.smfm.org/media/2591/SMFM_Vaccine_Statement_12-1-20 (accessed on 19 January 2021).
- Klein, S.L.; Creisher, P.S.; Burd, I. COVID-19 vaccine testing in pregnant females is necessary. J. Clin. Investig. 2021, 131, e147553. [Google Scholar] [CrossRef] [PubMed]
- Esposito, S.; Bosis, S.; Morlacchi, L.; Baggi, E.; Sabatini, C.; Principi, N. Can infants be protected by means of maternal vaccination? Clin. Microbiol. Infect. 2012, 18, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Edlow, A.G.; Li, J.Z.; Collier, A.-R.Y.; Atyeo, C.; James, K.E.; Boatin, A.A.; Gray, K.J.; Bordt, E.A.; Shook, L.L.; Yonker, L.M.; et al. Assessment of maternal and neonatal SARS-CoV-2 viral load, transplacental antibody transfer, and placental pathology in pregnancies during the COVID-19 pandemic. JAMA Netw. Open 2020, 3, e2030455. [Google Scholar] [CrossRef] [PubMed]
- Vilajeliu, A.; Goncé, A.; López, M.; Costa, J.; Rocamora, L.; Ríos, J.; Teixidó, I.; Bayas, J.M. Combined tetanus-diphtheria and pertussis vaccine during pregnancy: Transfer of maternal pertussis antibodies to the newborn. Vaccine 2015, 33, 1056–1062. [Google Scholar] [CrossRef] [PubMed]
- Gray, K.J.; Bordt, E.A.; Atyeo, C.; Deriso, E.; Akinwunmi, B.; Young, N.; Baez, A.M.; Shook, L.L.; Cvrk, D.; James, K.; et al. Coronavirus disease 2019 vaccine response in pregnant and lactating women: A cohort study. Am. J. Obstet. Gynecol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Omer, S.B.; Clark, D.R.; Madhi, S.A.; Tapia, M.D.; Nunes, M.C.; Cutland, C.L.; Simões, E.A.F.; Aqil, A.R.; Katz, J.; Tielsch, J.M.; et al. Efficacy, duration of protection, birth outcomes, and infant growth associated with influenza trials vaccination in pregnancy: A pooled analysis of three randomized controlled. Lancet Respir. Med. 2020, 8, 597–608. [Google Scholar] [CrossRef]
- Shimabukuro, T.T.; Kim, S.Y.; Myers, T.R.; Moro, P.L.; Oduyebo, T.; Panagiotakopoulos, L.; Marquez, P.L.; Olson, C.K.; Liu, R.; Chang, K.T.; et al. Preliminary findings of mRNA Covid-19 vaccine safety in pregnant persons. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
| Variable | Included in the Analysis n = 16 |
|---|---|
| Age of mothers (years) | 31.2 * (±2.2 **) Min 28 Max 35 |
| Parity | 11 (69% ***) |
| ≥1 | |
| BMI | 23.78 * (±4.51 **) Min 17.84 Max 34.95 |
| Weeks of gestation at the first dose of COVID-19 vaccination (weeks) | 31.75 * (±2.05 **) Min 29 Max 36 |
| Weeks of gestation at the second dose of COVID-19 vaccination (weeks) | 35.13 * (±2.13 **) Min 32 Max 40 |
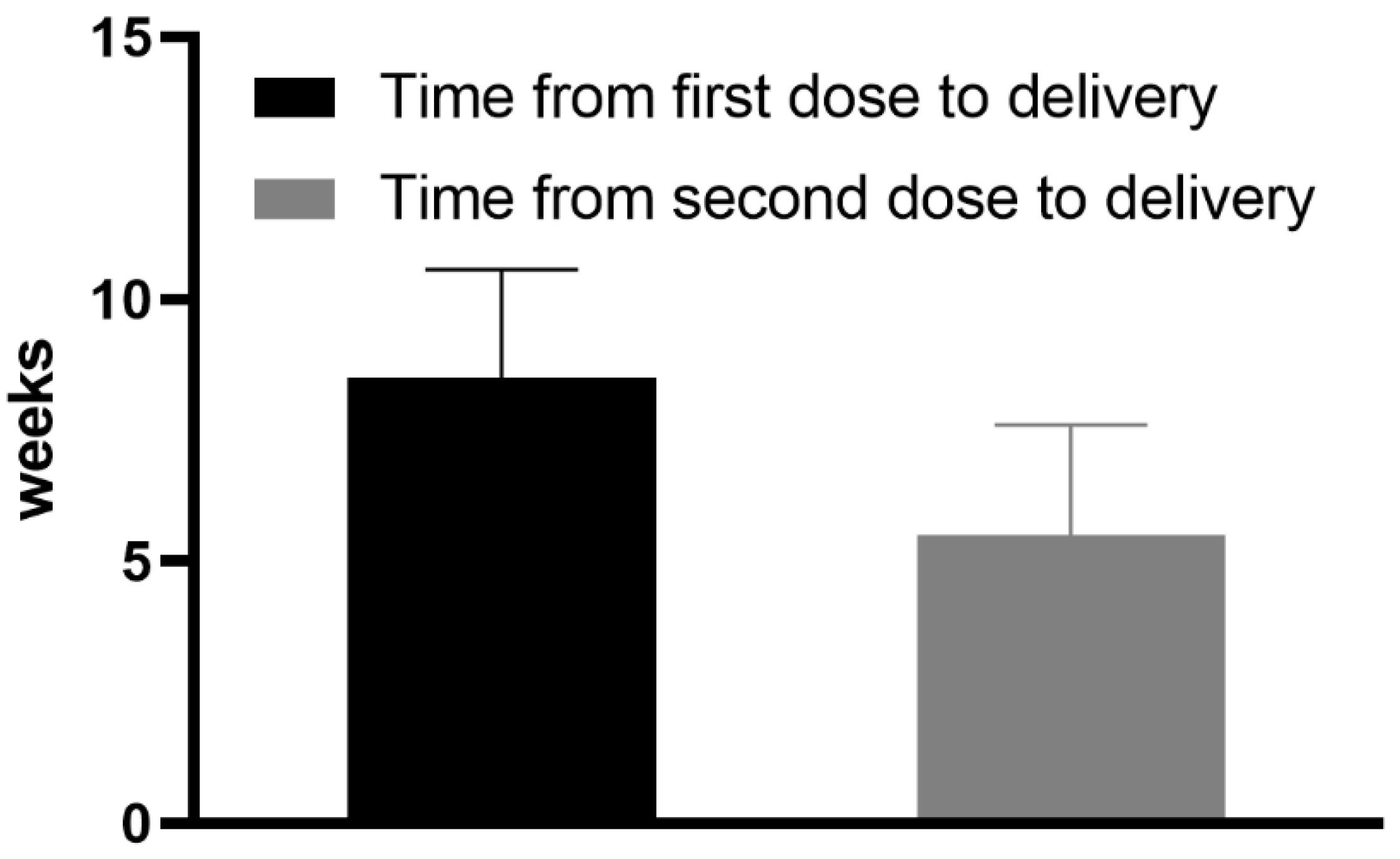
| Interval between the second dose of COVID-19 vaccination and collection of blood samples (day of delivery) (weeks) | 5.5 * (±2.1 **) Min 1 Max 8 |
| Interval between the first dose of COVID-19 vaccination and the collection of blood samples (day of delivery) (weeks) | 8.5 * (±2.07 **) Min 5 Max 11 |
| Weeks of gestation at delivery (weeks) | 39.69 * (±1.01 **) Min 38 Max 41 |
| Sex of newborn | |
| Male | 8 (50% ***) |
| Female | 8 (50% ***) |
| Weight of newborn (g) | 3353 * (±495 **) Min 2270 Max 4000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zdanowski, W.; Waśniewski, T. Evaluation of SARS-CoV-2 Spike Protein Antibody Titers in Cord Blood after COVID-19 Vaccination during Pregnancy in Polish Healthcare Workers: Preliminary Results. Vaccines 2021, 9, 675. https://doi.org/10.3390/vaccines9060675
Zdanowski W, Waśniewski T. Evaluation of SARS-CoV-2 Spike Protein Antibody Titers in Cord Blood after COVID-19 Vaccination during Pregnancy in Polish Healthcare Workers: Preliminary Results. Vaccines. 2021; 9(6):675. https://doi.org/10.3390/vaccines9060675
Chicago/Turabian StyleZdanowski, Wojciech, and Tomasz Waśniewski. 2021. "Evaluation of SARS-CoV-2 Spike Protein Antibody Titers in Cord Blood after COVID-19 Vaccination during Pregnancy in Polish Healthcare Workers: Preliminary Results" Vaccines 9, no. 6: 675. https://doi.org/10.3390/vaccines9060675
APA StyleZdanowski, W., & Waśniewski, T. (2021). Evaluation of SARS-CoV-2 Spike Protein Antibody Titers in Cord Blood after COVID-19 Vaccination during Pregnancy in Polish Healthcare Workers: Preliminary Results. Vaccines, 9(6), 675. https://doi.org/10.3390/vaccines9060675






