Impact of Inflammatory Immune Dysfunction in Psoriasis Patients at Risk for COVID-19
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Isolation of Plasma Samples
2.3. ELISA Cytokines Measurement
2.4. Detection of IgG Antibodies to COVID-19
2.5. Determination of Cytokines by Cytometric Bead Array (CBA)
2.6. Statistical Analysis
3. Results
3.1. Positivity of IgG Antibodies to COVID-19 in Psoriasis Patients
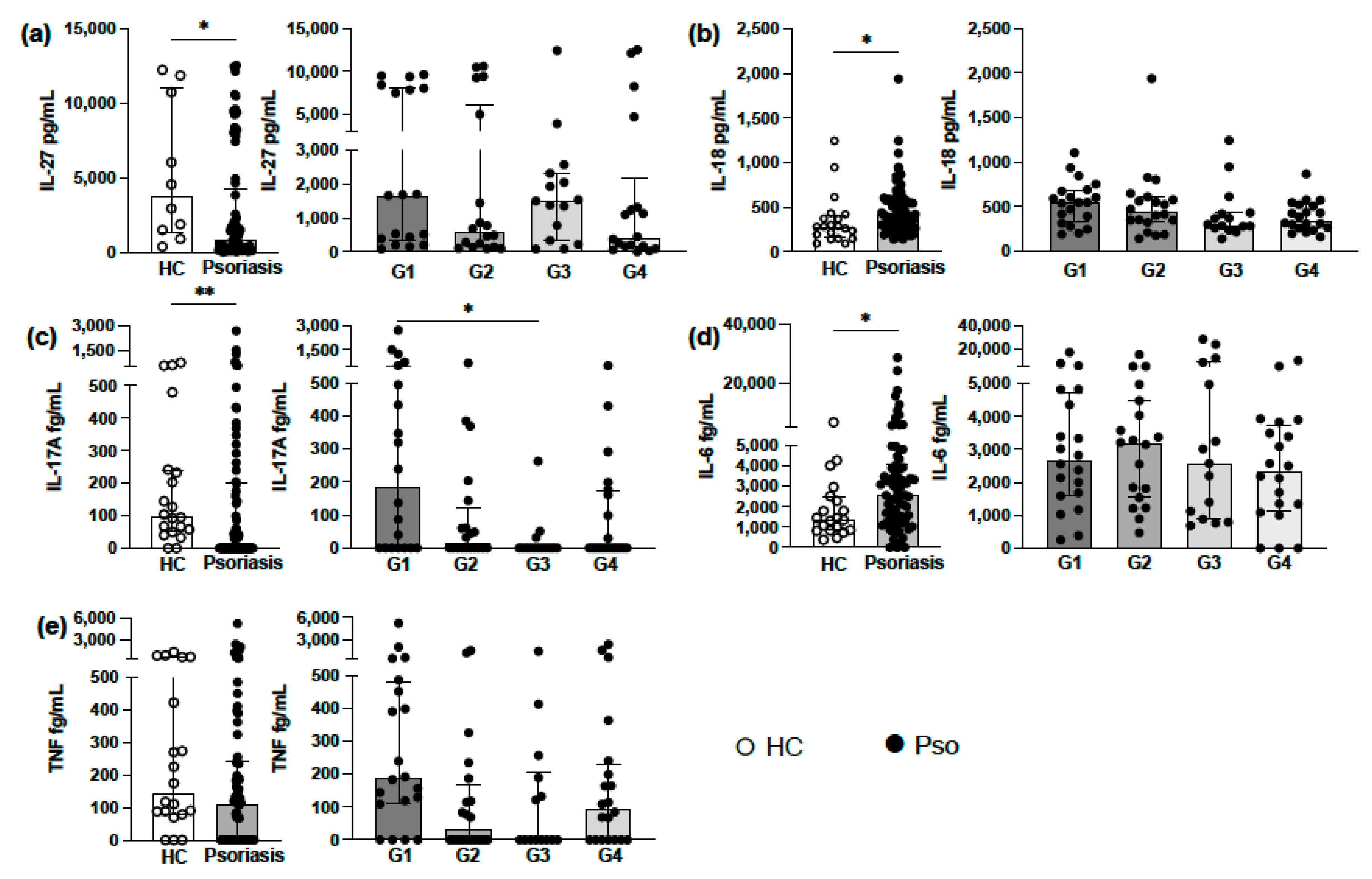
3.2. Analysis of Circulating Levels of Proinflammatory Cytokines
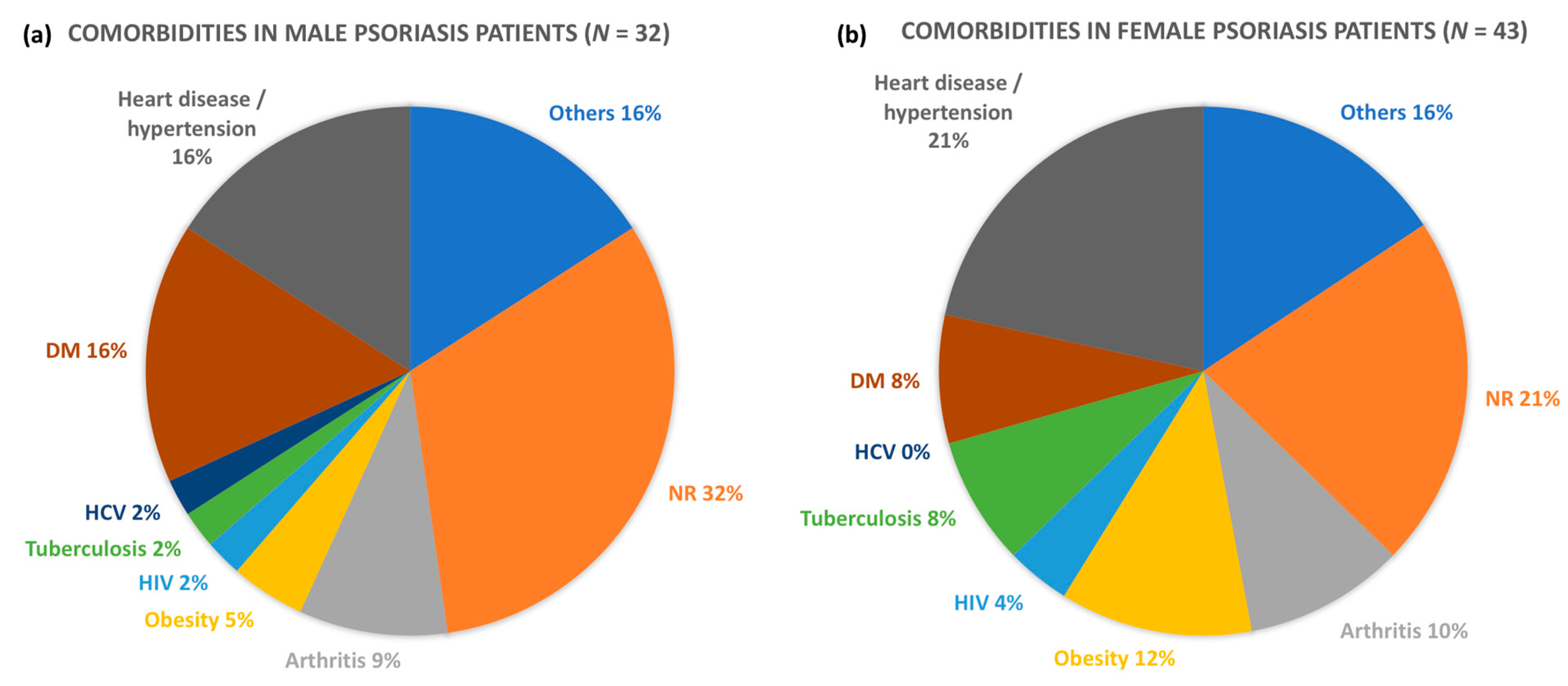
3.3. Comorbidities in Psoriasis Patients
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rendon, A.; Schäkel, K. Psoriasis pathogenesis and treatment. Int. J. Mol. Sci. 2019, 20, 1475. [Google Scholar] [CrossRef]
- Wolk, K.; Witte, K.; Witte, E.; Raftery, M.; Kokolakis, G.; Philipp, S.; Schonrich, G.; Warszawska, K.; Kirsch, S.; Prosch, S.; et al. IL-29 is produced by T(H)17 cells and mediates the cutaneous antiviral competence in psoriasis. Sci. Transl. Med. 2013, 5, 204ra129. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, J.E.; Yan, B.Y.; Chan, T.C.; Krueger, J.G. Discovery of the IL-23/IL-17 signaling pathway and the treatment of psoriasis. J. Immunol. 2018, 201, 1605–1613. [Google Scholar] [CrossRef] [PubMed]
- Nestle, F.O.; Kaplan, D.H.; Barker, J. Psoriasis. N. Engl. J. Med. 2009, 361, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Lew, W.; Bowcock, A.M.; Krueger, J.G. Psoriasis vulgaris: Cutaneous lymphoid tissue supports T-cell activation and ‘Type 1’ inflammatory gene expression. Trends Immunol. 2004, 25, 295–305. [Google Scholar] [CrossRef]
- Shibata, S.; Tada, Y.; Kanda, N.; Nashiro, K.; Kamata, M.; Karakawa, M.; Miyagaki, T.; Kai, H.; Saeki, H.; Shirakata, Y.; et al. Possible roles of IL-27 in the pathogenesis of psoriasis. J. Investig. Dermatol. 2010, 130, 1034–1039. [Google Scholar] [CrossRef]
- Handfield, C.; Kwock, J.; MacLeod, A.S. Innate antiviral immunity in the skin. Trends Immunol. 2018, 39, 328–340. [Google Scholar] [CrossRef]
- Radzikowska, U.; Ding, M.; Tan, G.; Zhakparov, D.; Peng, Y.; Wawrzyniak, P.; Wang, M.; Li, S.; Morita, H.; Altunbulakli, C.; et al. Distribution of ACE2, CD147, CD26, and other SARS-CoV-2 associated molecules in tissues and immune cells in health and in asthma, COPD, obesity, hypertension, and COVID-19 risk factors. Allergy 2020, 75, 2829–2845. [Google Scholar] [CrossRef]
- Li, B.; Huang, L.; Lv, P.; Li, X.; Liu, G.; Chen, Y.; Wang, Z.; Qian, X.; Shen, Y.; Li, Y.; et al. The role of Th17 cells in psoriasis. Immunol. Res. 2020, 68, 296–309. [Google Scholar] [CrossRef]
- Carl, J.W.; Bai, X.-F. IL27: Its roles in the induction and inhibition of inflammation. Int. J. Clin. Exp. Pathol. 2008, 1, 117–123. [Google Scholar]
- Kwock, J.T.; Handfield, C.; Suwanpradid, J.; Hoang, P.; McFadden, M.J.; Labagnara, K.F.; Floyd, L.; Shannon, J.; Uppala, R.; Sarkar, M.K.; et al. IL-27 signaling activates skin cells to induce innate antiviral proteins and protects against Zika virus infection. Sci. Adv. 2020, 6, eaay3245. [Google Scholar] [CrossRef] [PubMed]
- Wittmann, M.; Doble, R.; Bachmann, M.; Pfeilschifter, J.; Werfel, T.; Mühl, H. IL-27 regulates IL-18 binding protein in skin resident cells. PLoS ONE 2012, 7, e38751. [Google Scholar] [CrossRef]
- Orfali, R.L.; Sato, M.N.; Takaoka, R.; Duarte, A.J.S.; Rivitti, E.A.; Aoki, V. Atopic dermatitis in adults: Evaluation of peripheral blood mononuclear cells proliferation response to staphylococcus aureus enterotoxins A and B and analysis of interleukin-18 secretion. Dermatitis 2008, 19, 218–238. [Google Scholar] [CrossRef]
- Armstrong, A.W.; Read, C. Pathophysiology, clinical presentation, and treatment of psoriasis: A review. JAMA 2020, 323, 1945–1960. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Chang, C.; Lu, Q. The inflammatory response in psoriasis: A comprehensive review. Clin. Rev. Allergy Immunol. 2016, 50, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Inquérito Sorológico para SARS-COV-2. Evolução Da Prevalência Da Infecção No Município De Sao Paulo. Available online: https://www.prefeitura.sp.gov.br/cidade/secretarias/upload/saude/27_8_2020_PPT_COLETIVA_PREFEITO_%20fase%2004.pdf (accessed on 18 April 2021).
- Takian, A.; Kiani, M.M.; Khanjankhani, K. COVID-19 and the need to prioritize health equity and social determinants of health. Int. J. Public Health 2020, 65, 521–523. [Google Scholar] [CrossRef]
- Mahil, S.K.; Dand, N.; Mason, K.J.; Yiu, Z.Z.; Tsakok, T.; Meynell, F.; Coker, B.; McAteer, H.; Moorhead, L.; MacKenzie, T.; et al. Factors associated with adverse COVID-19 outcomes in patients with psoriasis—Insights from a global registry–based study. J. Allergy Clin. Immunol. 2021, 147, 60–71. [Google Scholar] [CrossRef]
- Wang, C.; Rademaker, M.; Baker, C.; Foley, P. COVID-19 and the use of immunomodulatory and biologic agents for severe cutaneous disease: An Australian/New Zealand consensus statement. Australas. J. Dermatol. 2020, 61, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Elmas, Ö.F.; Demirbaş, A.; Kutlu, Ö.; Bağcıer, F.; Metin, M.S.; Özyurt, K.; Akdeniz, N.; Atasoy, M.; Türsen, Ü.; Lotti, T. Psoriasis and COVID-19: A narrative review with treatment considerations. Dermatol. Ther. 2020, 33, 13858. [Google Scholar] [CrossRef]
- Rodriguez-Villa Lario, A.; Vega-Diez, D.; Gonzalez-Canete, M.; Polo-Rodriguez, I.; Piteiro-Bermejo, A.B.; Herrero-Fernandez, M.; Arevalo-Serrano, J.; Trasobares-Marugan, L.; Medina-Montalvo, S. Patient s perspective: Psychological burden of the COVID-19 pandemic in 146 psoriatic patients treated with biological drugs and small molecules in real clinical practice. J. Dermatolog. Treat. 2020, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lima, X.; Cueva, M.; Lopes, E.; Alora, M. Severe COVID-19 outcomes in patients with psoriasis. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e776–e778. [Google Scholar] [CrossRef]
- Ricardo, J.W.; Lipner, S.R. Considerations for safety in the use of systemic medications for psoriasis and atopic dermatitis during the COVID-19 pandemic. Dermatol. Ther. 2020, 33, 13687. [Google Scholar] [CrossRef] [PubMed]
- Gisondi, P.; Facheris, P.; Dapavo, P.; Piaserico, S.; Conti, A.; Naldi, L.; Cazzaniga, S.; Malagoli, P.; Costanzo, A. The impact of the COVID-19 pandemic on patients with chronic plaque psoriasis being treated with biological therapy: The Northern Italy experience. Br. J. Dermatol. 2020, 183, 373–374. [Google Scholar] [CrossRef]
- Georgakopoulos, J.R.; Mufti, A.; Vender, R.; Yeung, J. Treatment discontinuation and rate of disease transmission in psoriasis patients receiving biologic therapy during the COVID-19 pandemic: A Canadian multicenter retrospective study. J. Am. Acad. Dermatol. 2020, 83, 1212–1214. [Google Scholar] [CrossRef]
- Cardoso, P.R.G.; Lima, E.V.D.A.; Lima, M.M.D.A.; Rêgo, M.J.B.D.M.; Marques, C.D.L.; Pitta, I.D.R.; Duarte, A.L.B.P.; Pitta, M.G.D.R. Clinical and cytokine profile evaluation in Northeast Brazilian psoriasis plaque-type patients. Eur. Cytokine Netw. 2016, 27, 1–5. [Google Scholar] [CrossRef]
- Yang, B.; Suwanpradid, J.; Sanchez-Lagunes, R.; Choi, H.W.; Hoang, P.; Wang, D.; Abraham, S.N.; MacLeod, A.S. IL-27 facilitates skin wound healing through induction of epidermal proliferation and host defense. J. Investig. Dermatol. 2017, 137, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Forouzandeh, M.; Besen, J.; Keane, R.W.; Vaccari, J.P.D.R. The inflammasome signaling proteins ASC and IL-18 as biomarkers of psoriasis. Front. Pharmacol. 2020, 11, 1238. [Google Scholar] [CrossRef]
- Morrow, K.N.; Coopersmith, C.M.; Ford, M.L. IL-17, IL-27, and IL-33: A novel axis linked to immunological dysfunction during sepsis. Front. Immunol. 2019, 10, 1982. [Google Scholar] [CrossRef] [PubMed]
- Owczarczyk-Saczonek, A.; Placek, W. Compounds of psoriasis with obesity and overweight. Postępy Hig. I Med. Dosw. 2017, 71, 761–772. [Google Scholar] [CrossRef]
- Watanabe, M.; Risi, R.; Tuccinardi, D.; Baquero, C.J.; Manfrini, S.; Gnessi, L. Obesity and SARS-CoV-2: A population to safeguard. Diabetes Metab. Res. Rev. 2020, 36, e3325. [Google Scholar] [CrossRef]
| Groups | Gender/n | Age (Years)/Mean ± SD | PASI/Mean ± SD | Seropositive COVID-19 (IgG)/n (Total) | Treatment Option/n |
|---|---|---|---|---|---|
| G1 | M/8 F/9 | 20–80/53.73 ± 13.66 | 0–24/3.744 ± 3.662 | 5/20 | mmunobiologics
|
| G2 | M/11 F/9 | 8/20 | Methotrexate: 20/20 | ||
| G3 | M/4 F/11 | 3/15 | Topical treatment: 15/15 | ||
| G4 | M/9 F/11 | 8/20 | Acitretin: 20/20 | ||
| HC | M/5 F/15 | 24–49/32.75 ± 6.10 | NA | None | NA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yendo, T.M.; Sato, M.N.; Branco, A.C.C.C.; Pietrobon, A.J.; Teixeira, F.M.E.; Ramos, Y.Á.L.; Alberca, R.W.; Valêncio, C.G.; Arruda, V.N.; Romiti, R.; et al. Impact of Inflammatory Immune Dysfunction in Psoriasis Patients at Risk for COVID-19. Vaccines 2021, 9, 478. https://doi.org/10.3390/vaccines9050478
Yendo TM, Sato MN, Branco ACCC, Pietrobon AJ, Teixeira FME, Ramos YÁL, Alberca RW, Valêncio CG, Arruda VN, Romiti R, et al. Impact of Inflammatory Immune Dysfunction in Psoriasis Patients at Risk for COVID-19. Vaccines. 2021; 9(5):478. https://doi.org/10.3390/vaccines9050478
Chicago/Turabian StyleYendo, Tatiana Mina, Maria Notomi Sato, Anna Cláudia Calvielli Castelo Branco, Anna Julia Pietrobon, Franciane Mouradian Emidio Teixeira, Yasmim Álefe Leuzzi Ramos, Ricardo Wesley Alberca, Cesar Giudice Valêncio, Vivian Nunes Arruda, Ricardo Romiti, and et al. 2021. "Impact of Inflammatory Immune Dysfunction in Psoriasis Patients at Risk for COVID-19" Vaccines 9, no. 5: 478. https://doi.org/10.3390/vaccines9050478
APA StyleYendo, T. M., Sato, M. N., Branco, A. C. C. C., Pietrobon, A. J., Teixeira, F. M. E., Ramos, Y. Á. L., Alberca, R. W., Valêncio, C. G., Arruda, V. N., Romiti, R., Arnone, M., Hirayama, A. L. d. S., Duarte, A. J. d. S., Aoki, V., & Orfali, R. L. (2021). Impact of Inflammatory Immune Dysfunction in Psoriasis Patients at Risk for COVID-19. Vaccines, 9(5), 478. https://doi.org/10.3390/vaccines9050478








