Peptide-Based Vaccines: Foot-and-Mouth Disease Virus, a Paradigm in Animal Health
Abstract
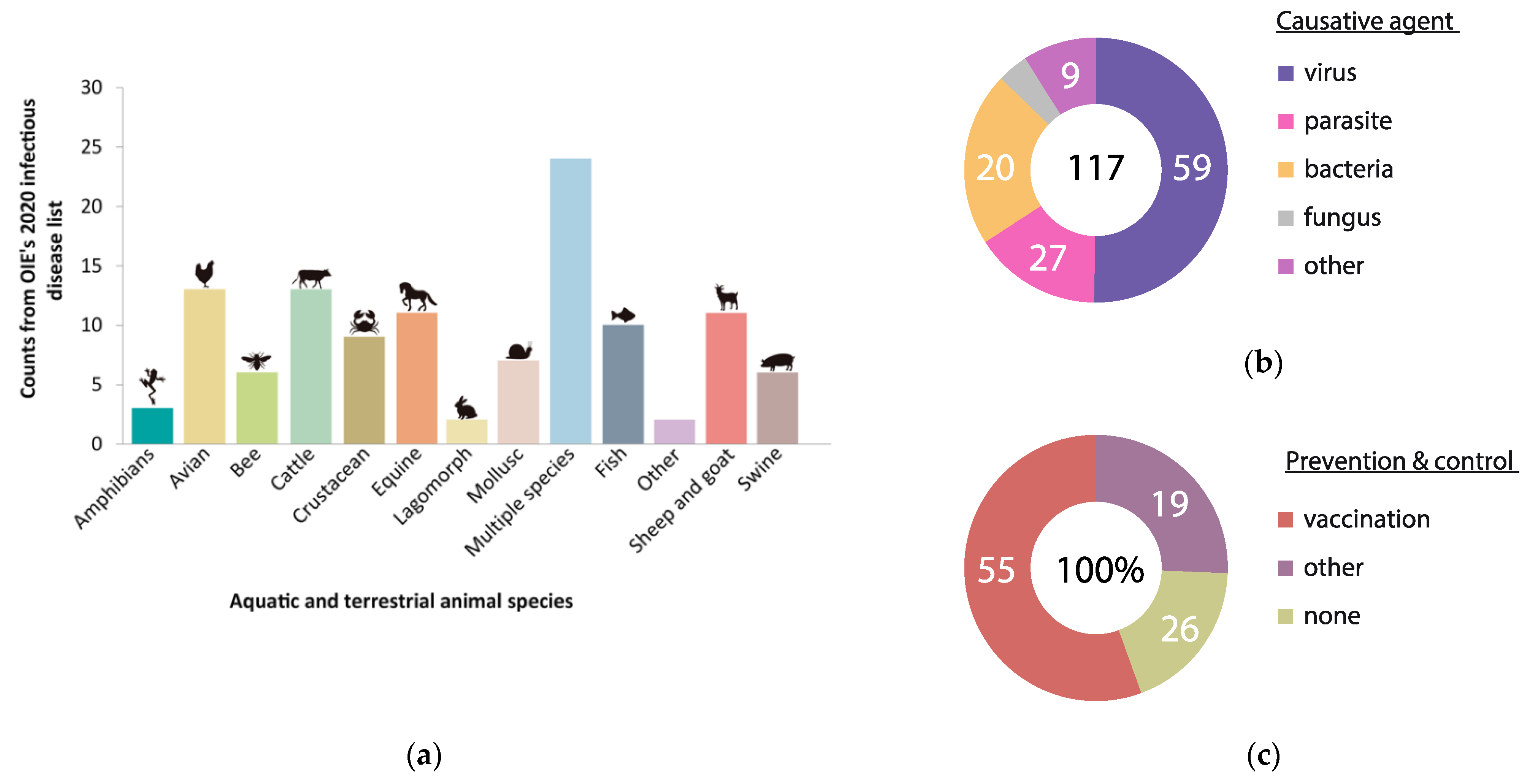
1. Introduction
Current State of FMD Vaccines
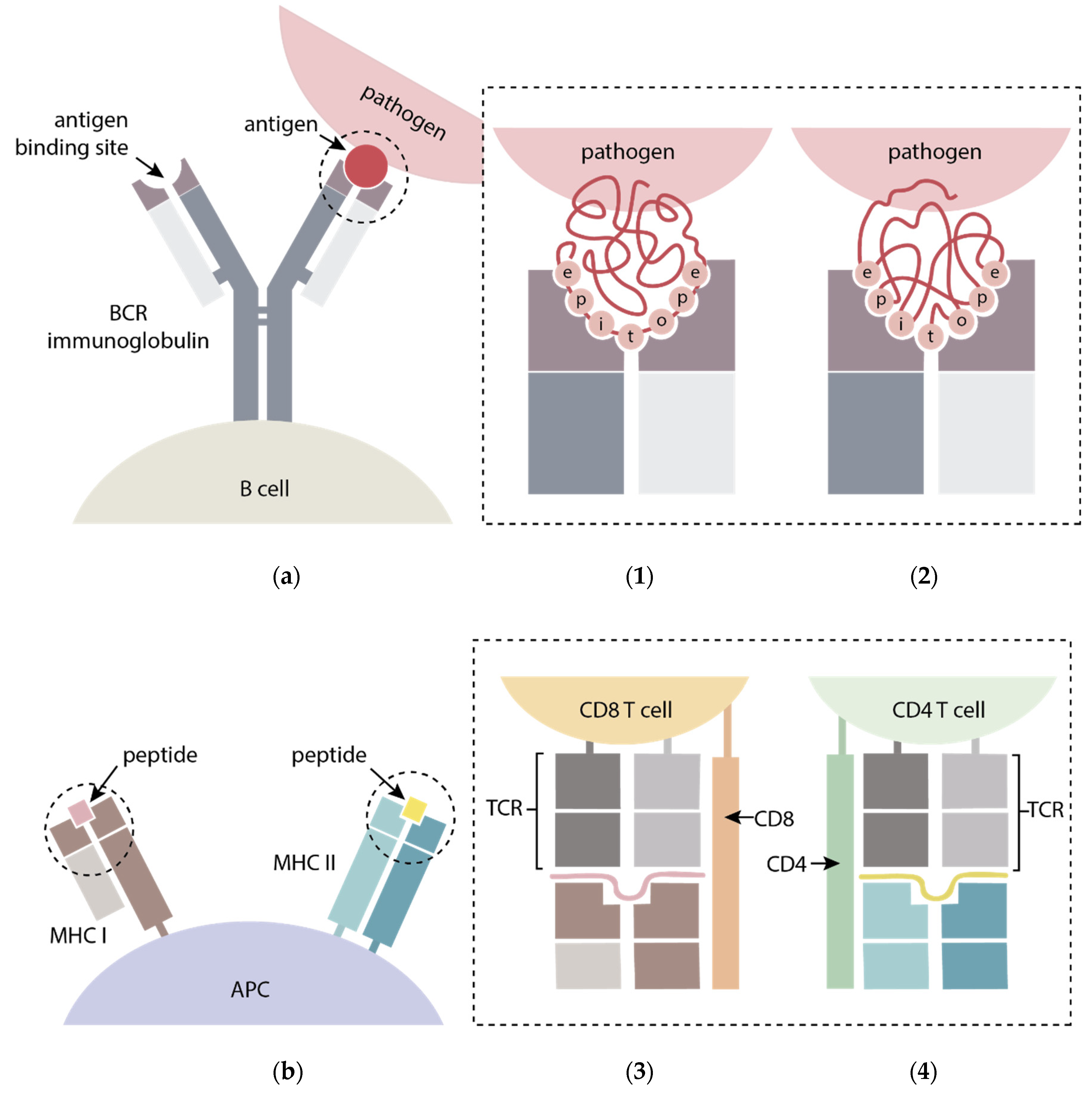
2. Overview of Epitope-Based Vaccines
3. FMDV-Specific Immune Response
4. FMDV Antigenic Structure
4.1. B-Cell Epitopes
4.2. T-Cell Epitopes
4.3. CTL Epitopes
5. Reproducing FMDV Antigenic Sites by Synthetic Peptides
5.1. Discontinuous Antigenic Sites
5.2. Continuous Antigenic Sites
6. Next-Generation Peptide Vaccines: Multimerization Approaches
7. FMDV Dendrimer Peptide Vaccines
8. Extending the Dendrimer Vaccine Approach to Classical Swine Fever
9. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Saker, L.; Lee, K.; Cannito, B.; Gilmore, A.; Campbell-Lendrum, D.H. Globalization and Infectious Diseases: A Review of the Linkages; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Mackenzie, J.S.; Jeggo, M. The One Health Approach-Why Is It So Important? Trop. Med. Infect. Dis. 2019, 4, 88. [Google Scholar] [CrossRef]
- Bonbon, E. A framework for national official assurance systems with reference to World Organisation for Animal Health standards. Rev. Sci. Tech. 2020, 39, 193–200. [Google Scholar] [CrossRef]
- Caceres, P.; Tizzani, P.; Ntsama, F.; Mora, R. The World Organisation for Animal Health: Notification of animal diseases. Rev. Sci. Tech. 2020, 39, 289–297. [Google Scholar] [CrossRef]
- Diaz-San Segundo, F.; Medina, G.N.; Stenfeldt, C.; Arzt, J.; de Los Santos, T. Foot-and-mouth disease vaccines. Vet. Microbiol. 2017, 206, 102–112. [Google Scholar] [CrossRef]
- Lombard, M.; Füssel, A.E. Antigen and vaccine banks: Technical requirements and the role of the european antigen bank in emergency foot and mouth disease vaccination. Rev. Sci. Tech. 2007, 26, 117–134. [Google Scholar] [CrossRef]
- Blanco, E.; Andreu, D.; Sobrino, F. Peptide Vaccines Against Foot-and-mouth Disease. In Foot-and-Mouth Disease Virus. Current Research and Emerging Trends; Sobrino, F., Domingo, E., Eds.; Caister Academic Press: Norfolk, UK, 2017; pp. 317–332. [Google Scholar] [CrossRef]
- de Los Santos, T.; Diaz-San Segundo, F.; Rodriguez, L.L. The need for improved vaccines against foot-and-mouth disease. Curr. Opin. Virol. 2018, 29, 16–25. [Google Scholar] [CrossRef]
- Doel, T.R. FMD vaccines. Virus Res. 2003, 91, 81–99. [Google Scholar] [CrossRef]
- Capstick, P.B.; Garland, A.J.; Masters, R.C.; Chapman, W.G. Some functional and morphological alterations occurring during and after the adaptation of BHK 21 clone 13 cells to suspension culture. Exp. Cell Res. 1966, 44, 119–128. [Google Scholar] [CrossRef]
- Capstick, P.B.; Telling, R.C.; Chapman, W.G.; Stewart, D.L. Growth of a cloned strain of hamster kidney cells in suspended cultures and their susceptibility to the virus of foot-and-mouth disease. Nature 1962, 195, 1163–1164. [Google Scholar] [CrossRef] [PubMed]
- Mowat, G.N.; Brooksby, J.B.; Pay, T.W. Use of BHK 21 cells in the preparation of mouse attenuated live foot-and-mouth disease vaccines for the immunization of cattle. Nature 1962, 196, 655–656. [Google Scholar] [CrossRef]
- Bahnemann, H.G. The inactivation of foot-and-mouth disease virus by ethylenimine and propylenimine. Zent. Vet. Reihe B 1972, 20, 356–360. [Google Scholar] [CrossRef]
- Cao, Y.; Lu, Z.; Liu, Z. Foot-and-mouth disease vaccines: Progress and problems. Expert Rev. Vaccines 2016, 15, 783–789. [Google Scholar] [CrossRef]
- Barteling, S.J. Modern inactivated Foot-and-mouth disease (FMD) vaccines: Historical background and key elements in production and use. In Foot-and-Mouth Disease: Current Perspectives; Sobrino, F., Domingo, E., Eds.; CRC Press: Boca Raton, FL, USA, 2004; ISBN 1-904933-00-9. [Google Scholar]
- Cox, S.J.; Aggarwal, N.; Statham, R.J.; Barnett, P.V. Longevity of antibody and cytokine responses following vaccination with high potency emergency FMD vaccines. Vaccine 2003, 21, 1336–1347. [Google Scholar] [CrossRef]
- Parida, S. Vaccination against foot-and-mouth disease virus: Strategies and effectiveness. Expert Rev. Vaccines 2009, 8, 347–365. [Google Scholar] [CrossRef]
- Rodriguez, L.L.; Gay, C.G. Development of vaccines toward the global control and eradication of foot-and-mouth disease. Expert Rev. Vaccines 2011, 10, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Alexandersen, S.; Zhang, Z.; Donaldson, A.I. Aspects of the persistence of foot-and-mouth disease virus in animals--the carrier problem. Microbes Infect. 2002, 4, 1099–1110. [Google Scholar] [CrossRef]
- Kitching, P.; Hammond, J.; Jeggo, M.; Charleston, B.; Paton, D.; Rodriguez, L.; Heckert, R. Global FMD control—Is it an option? Vaccine 2007, 25, 5660–5664. [Google Scholar] [CrossRef] [PubMed]
- Sobrino, F.; Domingo, E. Foot-and-mouth disease in Europe. FMD is economically the most important disease of farm animals. Its re-emergence in Europe is likely to have consequences that go beyond severe alterations of livestock production and trade. EMBO Rep. 2001, 2, 459–461. [Google Scholar] [CrossRef]
- Geale, D.W.; Barnett, P.V.; Clarke, G.W.; Davis, J.; Kasari, T.R. A Review of OIE Country Status Recovery Using Vaccinate-to-Live Versus Vaccinate-to-Die Foot-and-Mouth Disease Response Policies II: Waiting Periods After Emergency Vaccination in FMD Free Countries. Transbound. Emerg. Dis. 2015, 62, 388–406. [Google Scholar] [CrossRef]
- Ritmahan, W.; Kesmir, C.; Vroomans, R.M.A. Revealing factors determining immunodominant responses against dominant epitopes. Immunogenetics 2020, 72, 109–118. [Google Scholar] [CrossRef]
- Kwong, P.D.; DeKosky, B.J.; Ulmer, J.B. Antibody-guided structure-based vaccines. Semin. Immunol. 2020, 50, 101428. [Google Scholar] [CrossRef] [PubMed]
- Paul, W.E. Fundamental Immunology, 7th ed.; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; ISBN 9781451117837. [Google Scholar]
- Stern, L.J.; Wiley, D.C. Antigenic peptide binding by class I and class II histocompatibility proteins. Structure 1994, 2, 245–251. [Google Scholar] [CrossRef]
- Van Regenmortel, M.H. What is a B-cell epitope? Methods Mol. Biol. 2009, 524, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Trincado, J.L.; Gomez-Perosanz, M.; Reche, P.A. Fundamentals and Methods for T- and B-Cell Epitope Prediction. J. Immunol. Res. 2017, 2017, 2680160. [Google Scholar] [CrossRef]
- Sette, A.; Fikes, J. Epitope-based vaccines: An update on epitope identification, vaccine design and delivery. Curr. Opin. Immunol. 2003, 15, 461–470. [Google Scholar] [CrossRef]
- Gershoni, J.M.; Roitburd-Berman, A.; Siman-Tov, D.D.; Tarnovitski Freund, N.; Weiss, Y. Epitope mapping: The first step in developing epitope-based vaccines. Biodrugs Clin. Immunother. Biopharm. Gene Ther. 2007, 21, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, D.C. Molecular approaches to the study of B cell epitopes. Int. Rev. Immunol. 1991, 7, 149–164. [Google Scholar] [CrossRef]
- Cason, J. Strategies for mapping and imitating viral B-cell epitopes. J. Virol. Methods 1994, 49, 209–219. [Google Scholar] [CrossRef]
- Potocnakova, L.; Bhide, M.; Pulzova, L.B. An Introduction to B-Cell Epitope Mapping and In Silico Epitope Prediction. J. Immunol. Res. 2016, 2016, 6760830. [Google Scholar] [CrossRef]
- Di Marco, M.; Peper, J.K.; Rammensee, H.G. Identification of Immunogenic Epitopes by MS/MS. Cancer J. 2017, 23, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Pinilla, C.; Appel, J.R.; Judkowski, V.; Houghten, R.A. Identification of B cell and T cell epitopes using synthetic peptide combinatorial libraries. Curr. Protoc. Immunol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Vanniasinkam, T.; Barton, M.D.; Das, T.P.; Heuzenroeder, M.W. B-Cell Epitope Mapping Using a Library of Overlapping Synthetic Peptides in an Enzyme-Linked Immunosorbent Assay. Methods Mol. Biol. 2018, 1785, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Rosa, D.S.; Ribeiro, S.P.; Cunha-Neto, E. CD4+ T cell epitope discovery and rational vaccine design. Arch. Immunol. Ther. Exp. 2010, 58, 121–130. [Google Scholar] [CrossRef]
- Sidney, J.; Peters, B.; Sette, A. Epitope prediction and identification- adaptive T cell responses in humans. Semin. Immunol. 2020, 50, 101418. [Google Scholar] [CrossRef] [PubMed]
- Patarroyo, M.E.; Patarroyo, M.A. Emerging rules for subunit-based, multiantigenic, multistage chemically synthesized vaccines. Acc. Chem. Res. 2008, 41, 377–386. [Google Scholar] [CrossRef]
- Uttenthal, A.; Parida, S.; Rasmussen, T.B.; Paton, D.J.; Haas, B.; Dundon, W.G. Strategies for differentiating infection in vaccinated animals (DIVA) for foot-and-mouth disease, classical swine fever and avian influenza. Expert Rev. Vaccines 2010, 9, 73–87. [Google Scholar] [CrossRef]
- Matthias, D.M.; Robertson, J.; Garrison, M.M.; Newland, S.; Nelson, C. Freezing temperatures in the vaccine cold chain: A systematic literature review. Vaccine 2007, 25, 3980–3986. [Google Scholar] [CrossRef]
- Domingo, E. Genetic variation and quasi-species. Curr. Opin. Genet. Dev. 1992, 2, 61–63. [Google Scholar] [CrossRef]
- Kyte, J.A.; Gaudernack, G.; Dueland, S.; Trachsel, S.; Julsrud, L.; Aamdal, S. Telomerase peptide vaccination combined with temozolomide: A clinical trial in stage IV melanoma patients. Clin. Cancer Res. 2011, 17, 4568–4580. [Google Scholar] [CrossRef] [PubMed]
- Lennerz, V.; Gross, S.; Gallerani, E.; Sessa, C.; Mach, N.; Boehm, S.; Hess, D.; von Boehmer, L.; Knuth, A.; Ochsenbein, A.F.; et al. Immunologic response to the survivin-derived multi-epitope vaccine EMD640744 in patients with advanced solid tumors. Cancer Immunol. Immunother. 2014, 63, 381–394. [Google Scholar] [CrossRef]
- Zhang, L. Multi-epitope vaccines: A promising strategy against tumors and viral infections. Cell. Mol. Immunol. 2018, 15, 182–184. [Google Scholar] [CrossRef]
- Tsuji, M.; Zavala, F. Peptide-based subunit vaccines against pre-erythrocytic stages of malaria parasites. Mol. Immunol. 2001, 38, 433–442. [Google Scholar] [CrossRef]
- Kunwar, P.; Hawkins, N.; Dinges, W.L.; Liu, Y.; Gabriel, E.E.; Swan, D.A.; Stevens, C.E.; Maenza, J.; Collier, A.C.; Mullins, J.I.; et al. Superior control of HIV-1 replication by CD8+ T cells targeting conserved epitopes: Implications for HIV vaccine design. PLoS ONE 2013, 8, e64405. [Google Scholar] [CrossRef] [PubMed]
- McCullough, K.C.; Sobrino, F. Immunology of Foot-and-mouth disease. In Foot-and-Mouth Disease: Current Perspective; Sobrino, F., Domingo, E., Eds.; CRC Press: Boca Raton, FL, USA, 2004; ISBN 1-904933-00-9. [Google Scholar]
- McCullough, K.C.; De Simone, F.; Brocchi, E.; Capucci, L.; Crowther, J.R.; Kihm, U. Protective immune response against foot-and-mouth disease. J. Virol. 1992, 66, 1835–1840. [Google Scholar] [CrossRef]
- McCullough, K.C.; Summerfield, A. Basic concepts of immune response and defense development. ILAR J. 2005, 46, 230–240. [Google Scholar] [CrossRef] [PubMed]
- McCullough, K.C.; Pullen, L.; Parkinson, D. The immune response against foot-and-mouth disease virus: Influence of the T lymphocyte growth factors IL-1 and IL-2 on the murine humoral response in vivo. Immunol. Lett. 1992, 31, 41–46. [Google Scholar] [CrossRef]
- Halabi, G.; McCullough, K.C. Influence of antigen presentation and exogenous cytokine activity during in vitro primary immunizations employed for the generation of monoclonal antibodies. J. Immunol. Methods 1995, 186, 205–216. [Google Scholar] [CrossRef]
- McCullough, K.C.; Parkinson, D.; Crowther, J.R. Opsonization-enhanced phagocytosis of foot-and-mouth disease virus. Immunology 1988, 65, 187–191. [Google Scholar]
- Baxt, B.; Mason, P.W. Foot-and-mouth disease virus undergoes restricted replication in macrophage cell cultures following Fc receptor-mediated adsorption. Virology 1995, 207, 503–509. [Google Scholar] [CrossRef]
- McCullough, K.C.; Crowther, J.R.; Butcher, R.N.; Carpenter, W.C.; Brocchi, E.; Capucci, L.; De Simone, F. Immune protection against foot-and-mouth disease virus studied using virus-neutralizing and non-neutralizing concentrations of monoclonal antibodies. Immunology 1986, 58, 421–428. [Google Scholar]
- McCullough, K.C.; Ruggli, N.; Summerfield, A. Dendritic cells—At the front-line of pathogen attack. Vet. Immunol. Immunopathol. 2009, 128, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Summerfield, A.; McCullough, K.C. The porcine dendritic cell family. Dev. Comp. Immunol. 2009, 33, 299–309. [Google Scholar] [CrossRef]
- Guzman, E.; Taylor, G.; Charleston, B.; Ellis, S.A. Induction of a cross-reactive CD8(+) T cell response following foot-and-mouth disease virus vaccination. J. Virol. 2010, 84, 12375–12384. [Google Scholar] [CrossRef]
- Sáiz, J.C.; Rodríguez, A.; González, M.; Alonso, F.; Sobrino, F. Heterotypic lymphoproliferative response in pigs vaccinated with foot-and-mouth disease virus. Involvement of isolated capsid proteins. J. Gen. Virol. 1992, 73 Pt 10, 2601–2607. [Google Scholar] [CrossRef]
- Sobrino, F.; Sáiz, M.; Jiménez-Clavero, M.A.; Núñez, J.I.; Rosas, M.F.; Baranowski, E.; Ley, V. Foot-and-mouth disease virus: A long known virus, but a current threat. Vet. Res. 2001, 32, 1–30. [Google Scholar] [CrossRef]
- Summerfield, A.; Guzylack-Piriou, L.; Harwood, L.; McCullough, K.C. Innate immune responses against foot-and-mouth disease virus: Current understanding and future directions. Vet. Immunol. Immunopathol. 2009, 128, 205–210. [Google Scholar] [CrossRef]
- Fry, E.E.; Stuart, D.I.; Rowlands, D.J. The structure of foot-and-mouth disease virus. Curr. Top. Microbiol. Immunol. 2005, 288, 71–101. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Shao, J.; Chen, D.; Chang, Y.; Chang, H.; Zhang, Y. Identification of three linear B cell epitopes against non-structural protein 3ABC of FMDV using monoclonal antibodies. Appl. Microbiol. Biotechnol. 2019, 103, 8075–8086. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, J.P.; Compaired, D.; Trotta, M.; Perez, M.; Trono, K.; Fondevila, N. Validation of an r3AB1-FMDV-NSP ELISA to distinguish between cattle infected and vaccinated with foot-and-mouth disease virus. J. Virol. Methods 2011, 178, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Asfor, A.S.; Upadhyaya, S.; Knowles, N.J.; King, D.P.; Paton, D.J.; Mahapatra, M. Novel antibody binding determinants on the capsid surface of serotype O foot-and-mouth disease virus. J. Gen. Virol. 2014, 95, 1104–1116. [Google Scholar] [CrossRef] [PubMed]
- Acharya, R.; Fry, E.; Stuart, D.; Fox, G.; Rowlands, D.; Brown, F. The three-dimensional structure of foot-and-mouth disease virus at 2.9 A resolution. Nature 1989, 337, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Curry, S.; Fry, E.; Blakemore, W.; Abu-Ghazaleh, R.; Jackson, T.; King, A.; Lea, S.; Newman, J.; Rowlands, D.; Stuart, D. Perturbations in the surface structure of A22 Iraq foot-and-mouth disease virus accompanying coupled changes in host cell specificity and antigenicity. Structure 1996, 4, 135–145. [Google Scholar] [CrossRef]
- Lea, S.; Hernández, J.; Blakemore, W.; Brocchi, E.; Curry, S.; Domingo, E.; Fry, E.; Abu-Ghazaleh, R.; King, A.; Newman, J.; et al. The structure and antigenicity of a type C foot-and-mouth disease virus. Structure 1994, 2, 123–139. [Google Scholar] [CrossRef]
- Wild, T.F.; Burroughs, J.N.; Brown, F. Surface structure of foot-and-mouth disease virus. J. Gen. Virol. 1969, 4, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Niepmann, M. Porcine polypyrimidine tract-binding protein stimulates translation initiation at the internal ribosome entry site of foot-and-mouth-disease virus. FEBS Lett. 1996, 388, 39–42. [Google Scholar] [CrossRef]
- Fox, G.; Parry, N.R.; Barnett, P.V.; McGinn, B.; Rowlands, D.J.; Brown, F. The cell attachment site on foot-and-mouth disease virus includes the amino acid sequence RGD (arginine-glycine-aspartic acid). J. Gen. Virol. 1989, 70 Pt 3, 625–637. [Google Scholar] [CrossRef]
- Mason, P.W.; Piccone, M.E.; McKenna, T.S.; Chinsangaram, J.; Grubman, M.J. Evaluation of a live-attenuated foot-and-mouth disease virus as a vaccine candidate. Virology 1997, 227, 96–102. [Google Scholar] [CrossRef]
- Strohmaier, K.; Franze, R.; Adam, K.H. Location and characterization of the antigenic portion of the FMDV immunizing protein. J. Gen. Virol. 1982, 59, 295–306. [Google Scholar] [CrossRef]
- Rowlands, D.J.; Clarke, B.E.; Carroll, A.R.; Brown, F.; Nicholson, B.H.; Bittle, J.L.; Houghten, R.A.; Lerner, R.A. Chemical basis of antigenic variation in foot-and-mouth disease virus. Nature 1983, 306, 694–697. [Google Scholar] [CrossRef]
- Thomas, A.A.; Woortmeijer, R.J.; Puijk, W.; Barteling, S.J. Antigenic sites on foot-and-mouth disease virus type A10. J. Virol. 1988, 62, 2782–2789. [Google Scholar] [CrossRef]
- Parry, N.; Fox, G.; Rowlands, D.; Brown, F.; Fry, E.; Acharya, R.; Logan, D.; Stuart, D. Structural and serological evidence for a novel mechanism of antigenic variation in foot-and-mouth disease virus. Nature 1990, 347, 569–572. [Google Scholar] [CrossRef]
- Kitson, J.D.; McCahon, D.; Belsham, G.J. Sequence analysis of monoclonal antibody resistant mutants of type O foot and mouth disease virus: Evidence for the involvement of the three surface exposed capsid proteins in four antigenic sites. Virology 1990, 179, 26–34. [Google Scholar] [CrossRef]
- Kleid, D.G.; Yansura, D.; Small, B.; Dowbenko, D.; Moore, D.M.; Grubman, M.J.; McKercher, P.D.; Morgan, D.O.; Robertson, B.H.; Bachrach, H.L. Cloned viral protein vaccine for foot-and-mouth disease: Responses in cattle and swine. Science 1981, 214, 1125–1129. [Google Scholar] [CrossRef] [PubMed]
- Bittle, J.L.; Houghten, R.A.; Alexander, H.; Shinnick, T.M.; Sutcliffe, J.G.; Lerner, R.A.; Rowlands, D.J.; Brown, F. Protection against foot-and-mouth disease by immunization with a chemically synthesized peptide predicted from the viral nucleotide sequence. Nature 1982, 298, 30–33. [Google Scholar] [CrossRef]
- Clarke, B.E.; Brown, A.L.; Currey, K.M.; Newton, S.E.; Rowlands, D.J.; Carroll, A.R. Potential secondary and tertiary structure in the genomic RNA of foot and mouth disease virus. Nucleic Acids Res. 1987, 15, 7067–7079. [Google Scholar] [CrossRef][Green Version]
- Zhang, Q.; Yang, Y.Q.; Zhang, Z.Y.; Li, L.; Yan, W.Y.; Jiang, W.J.; Xin, A.G.; Lei, C.X.; Zheng, Z.X. Immunogenicity of a recombinant fusion protein of tandem repeat epitopes of foot-and-mouth disease virus type Asia 1 for guinea pigs. Acta Virol. 2002, 46, 1–9. [Google Scholar] [PubMed]
- Taboga, O.; Tami, C.; Carrillo, E.; Núñez, J.I.; Rodríguez, A.; Saíz, J.C.; Blanco, E.; Valero, M.L.; Roig, X.; Camarero, J.A.; et al. A large-scale evaluation of peptide vaccines against foot-and-mouth disease: Lack of solid protection in cattle and isolation of escape mutants. J. Virol. 1997, 71, 2606–2614. [Google Scholar] [CrossRef]
- Knowles, N.J.; Samuel, A.R. Molecular epidemiology of foot-and-mouth disease virus. Virus Res. 2003, 91, 65–80. [Google Scholar] [CrossRef]
- Beck, E.; Forss, S.; Strebel, K.; Cattaneo, R.; Feil, G. Structure of the FMDV translation initiation site and of the structural proteins. Nucleic Acids Res. 1983, 11, 7873–7885. [Google Scholar] [CrossRef]
- Dopazo, J.; Sobrino, F.; Palma, E.L.; Domingo, E.; Moya, A. Gene encoding capsid protein VP1 of foot-and-mouth disease virus: A quasispecies model of molecular evolution. Proc. Natl. Acad. Sci. USA 1988, 85, 6811–6815. [Google Scholar] [CrossRef]
- Collen, T.; Dimarchi, R.; Doel, T.R. A T cell epitope in VP1 of foot-and-mouth disease virus is immunodominant for vaccinated cattle. J. Immunol. 1991, 146, 749–755. [Google Scholar]
- Rodríguez, A.; Sáiz, J.C.; Novella, I.S.; Andreu, D.; Sobrino, F. Antigenic specificity of porcine T cell response against foot-and-mouth disease virus structural proteins: Identification of T helper epitopes in VP1. Virology 1994, 205, 24–33. [Google Scholar] [CrossRef]
- Carrillo, C.; Tulman, E.R.; Delhon, G.; Lu, Z.; Carreno, A.; Vagnozzi, A.; Kutish, G.F.; Rock, D.L. Comparative genomics of foot-and-mouth disease virus. J. Virol. 2005, 79, 6487–6504. [Google Scholar] [CrossRef]
- van Lierop, M.J.; van Noort, J.M.; Wagenaar, J.P.; Rutten, V.P.; Langeveld, J.; Meloen, R.H.; Hensen, E.J. T cell-stimulatory fragments of foot-and-mouth disease virus released by mild treatment with cathepsin D. J. Gen. Virol. 1994, 75 Pt 11, 2937–2946. [Google Scholar] [CrossRef]
- Blanco, E.; McCullough, K.; Summerfield, A.; Fiorini, J.; Andreu, D.; Chiva, C.; Borrás, E.; Barnett, P.; Sobrino, F. Interspecies major histocompatibility complex-restricted Th cell epitope on foot-and-mouth disease virus capsid protein VP4. J. Virol. 2000, 74, 4902–4907. [Google Scholar] [CrossRef]
- Gerner, W.; Denyer, M.S.; Takamatsu, H.H.; Wileman, T.E.; Wiesmüller, K.H.; Pfaff, E.; Saalmüller, A. Identification of novel foot-and-mouth disease virus specific T-cell epitopes in c/c and d/d haplotype miniature swine. Virus Res. 2006, 121, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Weber, C.A.; Mehta, P.J.; Ardito, M.; Moise, L.; Martin, B.; De Groot, A.S. T cell epitope: Friend or foe? Immunogenicity of biologics in context. Adv. Drug Deliv. Rev. 2009, 61, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Ganges, L.; Borrego, B.; Fernández-Pacheco, P.; Revilla, C.; Fernández-Borges, N.; Domínguez, J.; Sobrino, F.; Rodriguez, F. DNA immunization of pigs with foot-and-mouth disease virus minigenes: From partial protection to disease exacerbation. Virus Res. 2011, 157, 121–125. [Google Scholar] [CrossRef]
- Blanco, E.; Garcia-Briones, M.; Sanz-Parra, A.; Gomes, P.; De Oliveira, E.; Valero, M.L.; Andreu, D.; Ley, V.; Sobrino, F. Identification of T-cell epitopes in nonstructural proteins of foot-and-mouth disease virus. J. Virol. 2001, 75, 3164–3174. [Google Scholar] [CrossRef] [PubMed]
- Blanco, E.; Guerra, B.; de la Torre, B.G.; Defaus, S.; Dekker, A.; Andreu, D.; Sobrino, F. Full protection of swine against foot-and-mouth disease by a bivalent B-cell epitope dendrimer peptide. Antivir. Res. 2016, 129, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Cubillos, C.; de la Torre, B.G.; Bárcena, J.; Andreu, D.; Sobrino, F.; Blanco, E. Inclusion of a specific T cell epitope increases the protection conferred against foot-and-mouth disease virus in pigs by a linear peptide containing an immunodominant B cell site. Virol. J. 2012, 9, 66. [Google Scholar] [CrossRef]
- Cubillos, C.; de la Torre, B.G.; Jakab, A.; Clementi, G.; Borrás, E.; Bárcena, J.; Andreu, D.; Sobrino, F.; Blanco, E. Enhanced mucosal immunoglobulin A response and solid protection against foot-and-mouth disease virus challenge induced by a novel dendrimeric peptide. J. Virol. 2008, 82, 7223–7230. [Google Scholar] [CrossRef] [PubMed]
- Monsó, M.; de la Torre, B.G.; Blanco, E.; Moreno, N.; Andreu, D. Influence of conjugation chemistry and B epitope orientation on the immune response of branched peptide antigens. Bioconjug. Chem. 2013, 24, 578–585. [Google Scholar] [CrossRef]
- Collen, T.; Baron, J.; Childerstone, A.; Corteyn, A.; Doel, T.R.; Flint, M.; Garcia-Valcarcel, M.; Parkhouse, R.M.; Ryan, M.D. Heterotypic recognition of recombinant FMDV proteins by bovine T-cells: The polymerase (P3Dpol) as an immunodominant T-cell immunogen. Virus Res. 1998, 56, 125–133. [Google Scholar] [CrossRef]
- Foster, M.; Cook, A.; Cedillo, L.; Parkhouse, R.M. Serological and cellular immune responses to non-structural proteins in animals infected with FMDV. Vet. Q. 1998, 20 (Suppl. 2), S28–S30. [Google Scholar] [CrossRef]
- García-Briones, M.M.; Russell, G.C.; Oliver, R.A.; Tami, C.; Taboga, O.; Carrillo, E.; Palma, E.L.; Sobrino, F.; Glass, E.J. Association of bovine DRB3 alleles with immune response to FMDV peptides and protection against viral challenge. Vaccine 2000, 19, 1167–1171. [Google Scholar] [CrossRef]
- Cañas-Arranz, R.; Forner, M.; Defaus, S.; Rodríguez-Pulido, M.; de León, P.; Torres, E.; Bustos, M.J.; Borrego, B.; Sáiz, M.; Blanco, E.; et al. A bivalent B-cell epitope dendrimer peptide can confer long-lasting immunity in swine against foot-and-mouth disease. Transbound. Emerg. Dis. 2020, 67, 1614–1622. [Google Scholar] [CrossRef] [PubMed]
- Patch, J.R.; Pedersen, L.E.; Toka, F.N.; Moraes, M.; Grubman, M.J.; Nielsen, M.; Jungersen, G.; Buus, S.; Golde, W.T. Induction of foot-and-mouth disease virus-specific cytotoxic T cell killing by vaccination. Clin. Vaccine Immunol. 2011, 18, 280–288. [Google Scholar] [CrossRef]
- Barfoed, A.M.; Rodriguez, F.; Therrien, D.; Borrego, B.; Sobrino, F.; Kamstrup, S. DNA immunization with 2C FMDV non-structural protein reveals the presence of an immunodominant CD8+, CTL epitope for Balb/c mice. Antivir. Res. 2006, 72, 178–189. [Google Scholar] [CrossRef]
- Gao, F.S.; Feng, L.; Zhang, Q.; Yan, R.Q.; Li, Y.G.; Li, X.S. Immunogenicity of two FMDV nonameric peptides encapsulated in liposomes in mice and the protective efficacy in guinea pigs. PLoS ONE 2013, 8, e68658. [Google Scholar] [CrossRef]
- Gao, F.S.; Feng, L.; Jiang, P.; Li, Z.B.; Gao, H.; Zhai, X.X.; Zhang, Z.H.; Hu, X. Expression, purification, crystallization and preliminary X-ray diffraction analysis of swine leukocyte antigen 2 complexed with a CTL epitope AS64 derived from Asia1 serotype of foot-and-mouth disease virus. BMC Vet. Res. 2018, 14, 407. [Google Scholar] [CrossRef]
- Gao, F.S.; Zhai, X.X.; Jiang, P.; Zhang, Q.; Gao, H.; Li, Z.B.; Han, Y.; Yang, J.; Zhang, Z.H. Identification of two novel foot-and-mouth disease virus cytotoxic T lymphocyte epitopes that can bind six SLA-I proteins. Gene 2018, 653, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Pandya, M.; Rasmussen, M.; Hansen, A.; Nielsen, M.; Buus, S.; Golde, W.; Barlow, J. A modern approach for epitope prediction: Identification of foot-and-mouth disease virus peptides binding bovine leukocyte antigen (BoLA) class I molecules. Immunogenetics 2015, 67, 691–703. [Google Scholar] [CrossRef] [PubMed]
- Svitek, N.; Awino, E.; Nene, V.; Steinaa, L. BoLA-6*01301 and BoLA-6*01302, two allelic variants of the A18 haplotype, present the same epitope from the Tp1 antigen of Theileria parva. Vet. Immunol. Immunopathol. 2015, 167, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, L.E.; Patch, J.R.; Kenney, M.; Glabman, R.A.; Nielsen, M.; Jungersen, G.; Buus, S.; Golde, W.T. Expanding specificity of class I restricted CD8(+) T cells for viral epitopes following multiple inoculations of swine with a human adenovirus vectored foot-and-mouth disease virus (FMDV) vaccine. Vet. Immunol. Immunopathol. 2016, 181, 59–67. [Google Scholar] [CrossRef]
- Pedersen, L.E.; Harndahl, M.; Nielsen, M.; Patch, J.R.; Jungersen, G.; Buus, S.; Golde, W.T. Identification of peptides from foot-and-mouth disease virus structural proteins bound by class I swine leukocyte antigen (SLA) alleles, SLA-1*0401 and SLA-2*0401. Anim. Genet. 2013, 44, 251–258. [Google Scholar] [CrossRef]
- Ning, S.; Wang, Z.B.; Qi, P.; Xiao, J.; Wang, X.J. Crystallization of SLA-2*04:02:02 complexed with a CTL epitope derived from FMDV. Res. Vet. Sci. 2020, 128, 90–98. [Google Scholar] [CrossRef]
- Laver, W.G.; Air, G.M.; Webster, R.G.; Smith-Gill, S.J. Epitopes on protein antigens: Misconceptions and realities. Cell 1990, 61, 553–556. [Google Scholar] [CrossRef]
- Mateu, M.G.; Escarmís, C.; Domingo, E. Mutational analysis of discontinuous epitopes of foot-and-mouth disease virus using an unprocessed capsid protomer precursor. Virus Res. 1998, 53, 27–37. [Google Scholar] [CrossRef]
- Hill, T.A.; Shepherd, N.E.; Diness, F.; Fairlie, D.P. Constraining cyclic peptides to mimic protein structure motifs. Angew. Chem. 2014, 53, 13020–13041. [Google Scholar] [CrossRef]
- Borràs, E.; Giralt, E.; Andreu, D. A Rationally Designed Synthetic Peptide Mimic of a Discontinuous Viral Antigenic Site Elicits Neutralizing Antibodies. J. Am. Chem. Soc. 1999, 121, 11932–11933. [Google Scholar] [CrossRef]
- Villén, J.; Rodríguez-Mias, R.A.; Núñez, J.I.; Giralt, E.; Sobrino, F.; Andreu, D. Rational dissection of binding surfaces for mimicking of discontinuous antigenic sites. Chem. Biol. 2006, 13, 815–823. [Google Scholar] [CrossRef]
- Trabi, M.; Craik, D.J. Circular proteins--no end in sight. Trends Biochem. Sci. 2002, 27, 132–138. [Google Scholar] [CrossRef]
- Valero, M.L.; Camarero, J.A.; Haack, T.; Mateu, M.G.; Domingo, E.; Giralt, E.; Andreu, D. Native-like cyclic peptide models of a viral antigenic site: Finding a balance between rigidity and flexibility. J. Mol. Recognit. 2000, 13, 5–13. [Google Scholar] [CrossRef]
- Villén, J.; de Oliveira, E.; Núñez, J.I.; Molina, N.; Sobrino, F.; Andreu, D. Towards a multi-site synthetic vaccine to foot-and-mouth disease: Addition of discontinuous site peptide mimic increases the neutralization response in immunized animals. Vaccine 2004, 22, 3523–3529. [Google Scholar] [CrossRef] [PubMed]
- Villén, J.; Borràs, E.; Schaaper, W.M.; Meloen, R.H.; Dávila, M.; Domingo, E.; Giralt, E.; Andreu, D. Functional mimicry of a discontinuous antigenic site by a designed synthetic peptide. ChemBioBhem 2002, 3, 175–182. [Google Scholar] [CrossRef]
- Pfaff, E.; Mussgay, M.; Böhm, H.O.; Schulz, G.E.; Schaller, H. Antibodies against a preselected peptide recognize and neutralize foot and mouth disease virus. EMBO J. 1982, 1, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Doel, T.R.; Doel, C.M.; Staple, R.F.; DiMarchi, R. Cross-reactive and serotype-specific antibodies against foot-and-mouth disease virus generated by different regions of the same synthetic peptide. J. Virol. 1992, 66, 2187–2194. [Google Scholar] [CrossRef]
- DiMarchi, R.; Brooke, G.; Gale, C.; Cracknell, V.; Doel, T.; Mowat, N. Protection of cattle against foot-and-mouth disease by a synthetic peptide. Science 1986, 232, 639–641. [Google Scholar] [CrossRef]
- Morgan, D.O.; Moore, D.M. Protection of cattle and swine against foot-and-mouth disease, using biosynthetic peptide vaccines. Am. J. Vet. Res. 1990, 51, 40–45. [Google Scholar]
- Gras-Masse, H.; Ameisen, J.C.; Boutillon, C.; Rouaix, F.; Bossus, M.; Deprez, B.; Neyrinck, J.L.; Capron, A.; Tartar, A. Synthetic vaccines and HIV-1 hypervariability: A “mixotope” approach. Pept. Res. 1992, 5, 211–216. [Google Scholar] [PubMed]
- Oliveira, E.; Jiménez-Clavero, M.A.; Núñez, J.I.; Sobrino, F.; Andreu, D. Analysis of the immune response against mixotope peptide libraries from a main antigenic site of foot-and-mouth disease virus. Vaccine 2005, 23, 2647–2657. [Google Scholar] [CrossRef] [PubMed]
- Doel, T.R.; Gale, C.; Brooke, G.; DiMarchi, R. Immunization against foot-and-mouth disease with synthetic peptides representing the C-terminal region of VP1. J. Gen. Virol. 1988, 69 Pt 9, 2403–2406. [Google Scholar] [CrossRef]
- Barteling, S.J. Possibilities and limitations of synthetic peptide vaccines. Adv. Biotechnol. Process. 1988, 10, 25–60. [Google Scholar]
- Collen, T. Foot-and-mouth disease virus (Aphthovirus): Viral T-cell epitopes. In Cell-Mediated Immunity in Ruminants; Goddeeris, B.L., Morrison, W., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 173–197. ISBN 9780849349522. [Google Scholar]
- Glass, E.J.; Millar, P. Bovine T cells preferentially recognize non-viral spacer epitopes in a putative FMDV vaccinal peptide. Vaccine 1995, 13, 225–229. [Google Scholar] [CrossRef]
- Tami, C.; Taboga, O.; Berinstein, A.; Núñez, J.I.; Palma, E.L.; Domingo, E.; Sobrino, F.; Carrillo, E. Evidence of the coevolution of antigenicity and host cell tropism of foot-and-mouth disease virus in vivo. J. Virol. 2003, 77, 1219–1226. [Google Scholar] [CrossRef]
- Lee, H.B.; Piao, D.C.; Lee, J.Y.; Choi, J.Y.; Bok, J.D.; Cho, C.S.; Kang, S.K.; Choi, Y.J. Artificially designed recombinant protein composed of multiple epitopes of foot-and-mouth disease virus as a vaccine candidate. Microb. Cell Factories 2017, 16, 33. [Google Scholar] [CrossRef]
- Merrifield, R.B.; Juvvadi, P.; Andreu, D.; Ubach, J.; Boman, A.; Boman, H.G. Retro and retroenantio analogs of cecropin-melittin hybrids. Proc. Natl. Acad. Sci. USA 1995, 92, 3449–3453. [Google Scholar] [CrossRef] [PubMed]
- Briand, J.P.; Benkirane, N.; Guichard, G.; Newman, J.F.; Van Regenmortel, M.H.; Brown, F.; Muller, S. A retro-inverso peptide corresponding to the GH loop of foot-and-mouth disease virus elicits high levels of long-lasting protective neutralizing antibodies. Proc. Natl. Acad. Sci. USA 1997, 94, 12545–12550. [Google Scholar] [CrossRef]
- Tuchscherer, G.; Mutter, M. Templates in protein de novo design. J. Biotechnol. 1995, 41, 197–210. [Google Scholar] [CrossRef]
- Chen, Y.S.; Hung, Y.C.; Lin, W.H.; Huang, G.S. Assessment of gold nanoparticles as a size-dependent vaccine carrier for enhancing the antibody response against synthetic foot-and-mouth disease virus peptide. Nanotechnology 2010, 21, 195101. [Google Scholar] [CrossRef]
- Chang, C.Y.; Huang, C.C.; Deng, M.C.; Huang, Y.L.; Lin, Y.J.; Liu, H.M.; Lin, Y.L.; Wang, F.I. Antigenic mimicking with cysteine-based cyclized peptides reveals a previously unknown antigenic determinant on E2 glycoprotein of classical swine fever virus. Virus Res. 2012, 163, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.Y.; Hussein, W.M.; Jia, Z.; Ziora, Z.M.; McMillan, N.A.; Monteiro, M.J.; Toth, I.; Skwarczynski, M. Self-adjuvanting polymer-peptide conjugates as therapeutic vaccine candidates against cervical cancer. Biomacromolecules 2013, 14, 2798–2806. [Google Scholar] [CrossRef] [PubMed]
- Tam, J.P. Synthetic peptide vaccine design: Synthesis and properties of a high-density multiple antigenic peptide system. Proc. Natl. Acad. Sci. USA 1988, 85, 5409–5413. [Google Scholar] [CrossRef] [PubMed]
- Tam, J.P.; Spetzler, J.C. Chemoselective approaches to the preparation of peptide dendrimers and branched artificial proteins using unprotected peptides as building blocks. Biomed. Pept. Proteins Nucleic Acids 1995, 1, 123–132. [Google Scholar] [CrossRef]
- Pessi, A.; Bianchi, E.; Chiappinelli, L.; Bonelli, F.; Tougne, C.; Lambert, P.H.; Del Giudice, G. Multiple antigen peptides (MAPs) as candidate vaccines against malaria. Parassitologia 1991, 33, 79–84. [Google Scholar] [PubMed]
- Mahajan, B.; Berzofsky, J.A.; Boykins, R.A.; Majam, V.; Zheng, H.; Chattopadhyay, R.; de la Vega, P.; Moch, J.K.; Haynes, J.D.; Belyakov, I.M.; et al. Multiple antigen peptide vaccines against Plasmodium falciparum malaria. Infect. Immun. 2010, 78, 4613–4624. [Google Scholar] [CrossRef]
- Heegaard, P.M.; Boas, U.; Sorensen, N.S. Dendrimers for vaccine and immunostimulatory uses. A review. Bioconjug. Chem. 2010, 21, 405–418. [Google Scholar] [CrossRef]
- Kowalczyk, W.; de la Torre, B.G.; Andreu, D. Strategies and limitations in dendrimeric immunogen synthesis. The influenza virus M2e epitope as a case study. Bioconjug. Chem. 2010, 21, 102–110. [Google Scholar] [CrossRef]
- Kowalczyk, W.; Monsó, M.; de la Torre, B.G.; Andreu, D. Synthesis of multiple antigenic peptides (MAPs)-strategies and limitations. J. Pept. Sci. 2011, 17, 247–251. [Google Scholar] [CrossRef]
- Forner, M.; Defaus, S.; Andreu, D. Peptide-Based Multiepitopic Vaccine Platforms via Click Reactions. J. Org. Chem. 2020, 85, 1626–1634. [Google Scholar] [CrossRef] [PubMed]
- Monsó, M.; Kowalczyk, W.; Andreu, D.; de la Torre, B.G. Reverse thioether ligation route to multimeric peptide antigens. Org. Biomol. Chem. 2012, 10, 3116–3121. [Google Scholar] [CrossRef]
- Blanco, E.; Cubillos, C.; Moreno, N.; Bárcena, J.; de la Torre, B.G.; Andreu, D.; Sobrino, F. B epitope multiplicity and B/T epitope orientation influence immunogenicity of foot-and-mouth disease peptide vaccines. Clin. Dev. Immunol. 2013, 2013, 475960. [Google Scholar] [CrossRef] [PubMed]
- Soria, I.; Quattrocchi, V.; Langellotti, C.; Gammella, M.; Digiacomo, S.; Garcia de la Torre, B.; Andreu, D.; Montoya, M.; Sobrino, F.; Blanco, E.; et al. Dendrimeric peptides can confer protection against foot-and-mouth disease virus in cattle. PLoS ONE 2017, 12, e0185184. [Google Scholar] [CrossRef] [PubMed]
- Soria, I.; Quattrocchi, V.; Langellotti, C.; Pérez-Filgueira, M.; Pega, J.; Gnazzo, V.; Romera, S.; Schammas, J.; Bucafusco, D.; Di Giacomo, S.; et al. Immune Response and Partial Protection against Heterologous Foot-and-Mouth Disease Virus Induced by Dendrimer Peptides in Cattle. J. Immunol. Res. 2018, 2018, 3497401. [Google Scholar] [CrossRef]
- Cañas-Arranz, R.; Forner, M.; Defaus, S.; de León, P.; Bustos, M.J.; Torres, E.; Sobrino, F.; Andreu, D.; Blanco, E. A Single Dose of Dendrimer B(2)T Peptide Vaccine Partially Protects Pigs against Foot-and-Mouth Disease Virus Infection. Vaccines 2020, 8, 19. [Google Scholar] [CrossRef]
- Defaus, S.; Forner, M.; Cañas-Arranz, R.; de León, P.; Bustos, M.J.; Rodríguez-Pulido, M.; Blanco, E.; Sobrino, F.; Andreu, D. Designing Functionally Versatile, Highly Immunogenic Peptide-Based Multiepitopic Vaccines against Foot-and-Mouth Disease Virus. Vaccines 2020, 8, 406. [Google Scholar] [CrossRef]
- Cañas-Arranz, R.; de León, P.; Forner, M.; Defaus, S.; Bustos, M.J.; Torres, E.; Andreu, D.; Blanco, E.; Sobrino, F. Immunogenicity of a Dendrimer B(2)T Peptide Harboring a T-Cell Epitope From FMDV Non-structural Protein 3D. Front. Vet. Sci. 2020, 7, 498. [Google Scholar] [CrossRef]
- de León, P.; Cañas-Arranz, R.; Defaus, S.; Torres, E.; Forner, M.; Bustos, M.J.; Revilla, C.; Dominguez, J.; Andreu, D.; Blanco, E.; et al. Swine T-Cells and Specific Antibodies Evoked by Peptide Dendrimers Displaying Different FMDV T-Cell Epitopes. Front. Immunol. 2021, 11. [Google Scholar] [CrossRef]
- de León, P.; Cañas-Arranz, R.; Saez, Y.; Forner, M.; Defaus, S.; Cuadra, D.; Bustos, M.J.; Torres, E.; Andreu, D.; Blanco, E.; et al. Association of Porcine Swine Leukocyte Antigen (SLA) Haplotypes with B- and T-Cell Immune Response to Foot-and-Mouth Disease Virus (FMDV) Peptides. Vaccines 2020, 8, 513. [Google Scholar] [CrossRef]
- Ji, W.; Guo, Z.; Ding, N.Z.; He, C.Q. Studying classical swine fever virus: Making the best of a bad virus. Virus Res. 2015, 197, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.N.; Qi, Y.; Ying, J.; Chen, X.; Chen, Y.H. Candidate peptide-vaccine induced potent protection against CSFV and identified a principal sequential neutralizing determinant on E2. Vaccine 2006, 24, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Lin, F.; Mallory, M.; Clavijo, A. Deletions of structural glycoprotein E2 of classical swine fever virus strain alfort/187 resolve a linear epitope of monoclonal antibody WH303 and the minimal N-terminal domain essential for binding immunoglobulin G antibodies of a pig hyperimmune serum. J. Virol. 2000, 74, 11619–11625. [Google Scholar] [CrossRef] [PubMed]
- Monsó, M.; Tarradas, J.; de la Torre, B.G.; Sobrino, F.; Ganges, L.; Andreu, D. Peptide vaccine candidates against classical swine fever virus: T cell and neutralizing antibody responses of dendrimers displaying E2 and NS2-3 epitopes. J. Pept. Sci. 2011, 17, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Tarradas, J.; Monsó, M.; Muñoz, M.; Rosell, R.; Fraile, L.; Frías, M.T.; Domingo, M.; Andreu, D.; Sobrino, F.; Ganges, L. Partial protection against classical swine fever virus elicited by dendrimeric vaccine-candidate peptides in domestic pigs. Vaccine 2011, 29, 4422–4429. [Google Scholar] [CrossRef]
- Dong, X.N.; Chen, Y.H. Spying the neutralizing epitopes on E2 N-terminal by candidate epitope-vaccines against classical swine fever virus. Vaccine 2006, 24, 4029–4034. [Google Scholar] [CrossRef]
- Dong, X.N.; Chen, Y.H. Candidate peptide-vaccines induced immunity against CSFV and identified sequential neutralizing determinants in antigenic domain A of glycoprotein E2. Vaccine 2006, 24, 1906–1913. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.N.; Wei, K.; Liu, Z.Q.; Chen, Y.H. Candidate peptide vaccine induced protection against classical swine fever virus. Vaccine 2002, 21, 167–173. [Google Scholar] [CrossRef]
- Liu, S.; Tu, C.; Wang, C.; Yu, X.; Wu, J.; Guo, S.; Shao, M.; Gong, Q.; Zhu, Q.; Kong, X. The protective immune response induced by B cell epitope of classical swine fever virus glycoprotein E2. J. Virol. Methods 2006, 134, 125–129. [Google Scholar] [CrossRef]
- Bohórquez, J.A.; Defaus, S.; Muñoz-González, S.; Perez-Simó, M.; Rosell, R.; Fraile, L.; Sobrino, F.; Andreu, D.; Ganges, L. A bivalent dendrimeric peptide bearing a T-cell epitope from foot-and-mouth disease virus protein 3A improves humoral response against classical swine fever virus. Virus Res. 2017, 238, 8–12. [Google Scholar] [CrossRef]
- Li, G.X.; Zhou, Y.J.; Yu, H.; Li, L.; Wang, Y.X.; Tong, W.; Hou, J.W.; Xu, Y.Z.; Zhu, J.P.; Xu, A.T.; et al. A novel dendrimeric peptide induces high level neutralizing antibodies against classical swine fever virus in rabbits. Vet. Microbiol. 2012, 156, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Sadler, K.; Tam, J.P. Peptide dendrimers: Applications and synthesis. J. Biotechnol. 2002, 90, 195–229. [Google Scholar] [CrossRef]
- Purcell, A.W.; McCluskey, J.; Rossjohn, J. More than one reason to rethink the use of peptides in vaccine design. Nat. Rev. Drug Discov. 2007, 6, 404–414. [Google Scholar] [CrossRef]
- Li, W.; Joshi, M.D.; Singhania, S.; Ramsey, K.H.; Murthy, A.K. Peptide Vaccine: Progress and Challenges. Vaccines 2014, 2, 515–536. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, D. Medicinal Chemistry and Methodological Advances in the Development of Peptide-Based Vaccines. J. Med. Chem. 2020, 63, 14184–14196. [Google Scholar] [CrossRef]
- Combadière, B.; Beaujean, M.; Chaudesaigues, C.; Vieillard, V. Peptide-Based Vaccination for Antibody Responses Against HIV. Vaccines 2019, 7, 105. [Google Scholar] [CrossRef]
- Lamonaca, V.; Missale, G.; Urbani, S.; Pilli, M.; Boni, C.; Mori, C.; Sette, A.; Massari, M.; Southwood, S.; Bertoni, R.; et al. Conserved hepatitis C virus sequences are highly immunogenic for CD4(+) T cells: Implications for vaccine development. Hepatology 1999, 30, 1088–1098. [Google Scholar] [CrossRef]


| Type of Vaccine | 2005 | 2019 |
|---|---|---|
| Live attenuated vaccine | 402 | 444 |
| Inactivated vaccine | 285 | 329 |
| Conjugated vaccine | 6 | 6 |
| Recombinant vector vaccine | 6 | 13 |
| Subunit vaccine | 1 | 4 |
| DNA vaccine | - | 2 |
| Pros | Cons |
|---|---|
| Absence of infectious agent | Laborious identification of antigenic epitopes |
| No risk of mutation or reversion | Emulating 3D structures |
| Chemical stability | T-cell epitopes insufficiently specified |
| DIVA capability | Low immunogenicity |
| Simple handling, storage and transport | |
| Easy to manufacture |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forner, M.; Cañas-Arranz, R.; Defaus, S.; de León, P.; Rodríguez-Pulido, M.; Ganges, L.; Blanco, E.; Sobrino, F.; Andreu, D. Peptide-Based Vaccines: Foot-and-Mouth Disease Virus, a Paradigm in Animal Health. Vaccines 2021, 9, 477. https://doi.org/10.3390/vaccines9050477
Forner M, Cañas-Arranz R, Defaus S, de León P, Rodríguez-Pulido M, Ganges L, Blanco E, Sobrino F, Andreu D. Peptide-Based Vaccines: Foot-and-Mouth Disease Virus, a Paradigm in Animal Health. Vaccines. 2021; 9(5):477. https://doi.org/10.3390/vaccines9050477
Chicago/Turabian StyleForner, Mar, Rodrigo Cañas-Arranz, Sira Defaus, Patricia de León, Miguel Rodríguez-Pulido, Llilianne Ganges, Esther Blanco, Francisco Sobrino, and David Andreu. 2021. "Peptide-Based Vaccines: Foot-and-Mouth Disease Virus, a Paradigm in Animal Health" Vaccines 9, no. 5: 477. https://doi.org/10.3390/vaccines9050477
APA StyleForner, M., Cañas-Arranz, R., Defaus, S., de León, P., Rodríguez-Pulido, M., Ganges, L., Blanco, E., Sobrino, F., & Andreu, D. (2021). Peptide-Based Vaccines: Foot-and-Mouth Disease Virus, a Paradigm in Animal Health. Vaccines, 9(5), 477. https://doi.org/10.3390/vaccines9050477









