Differential Antibody Response against Conformational and Linear Epitopes of the L1 Proteins from Human Papillomavirus Types 16/18 Is Observed in Vaccinated Women or with Uterine Cervical Lesions
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Production and Purification of VLPs from HPV16/18
2.3. Coomassie Staining and Western Blot Analysis of L1 from HPV16/18 Expressed in Baculovirus
2.4. Identification of VLPs from HPV16/18 by Transmission Electron Microscopy
2.5. Detection of Anti-VLPs and Anti-L1 Antibodies from HPV16/18 by ELISA and Validation of the Assays
2.6. Statistical Analysis
3. Results
3.1. Population Characteristics
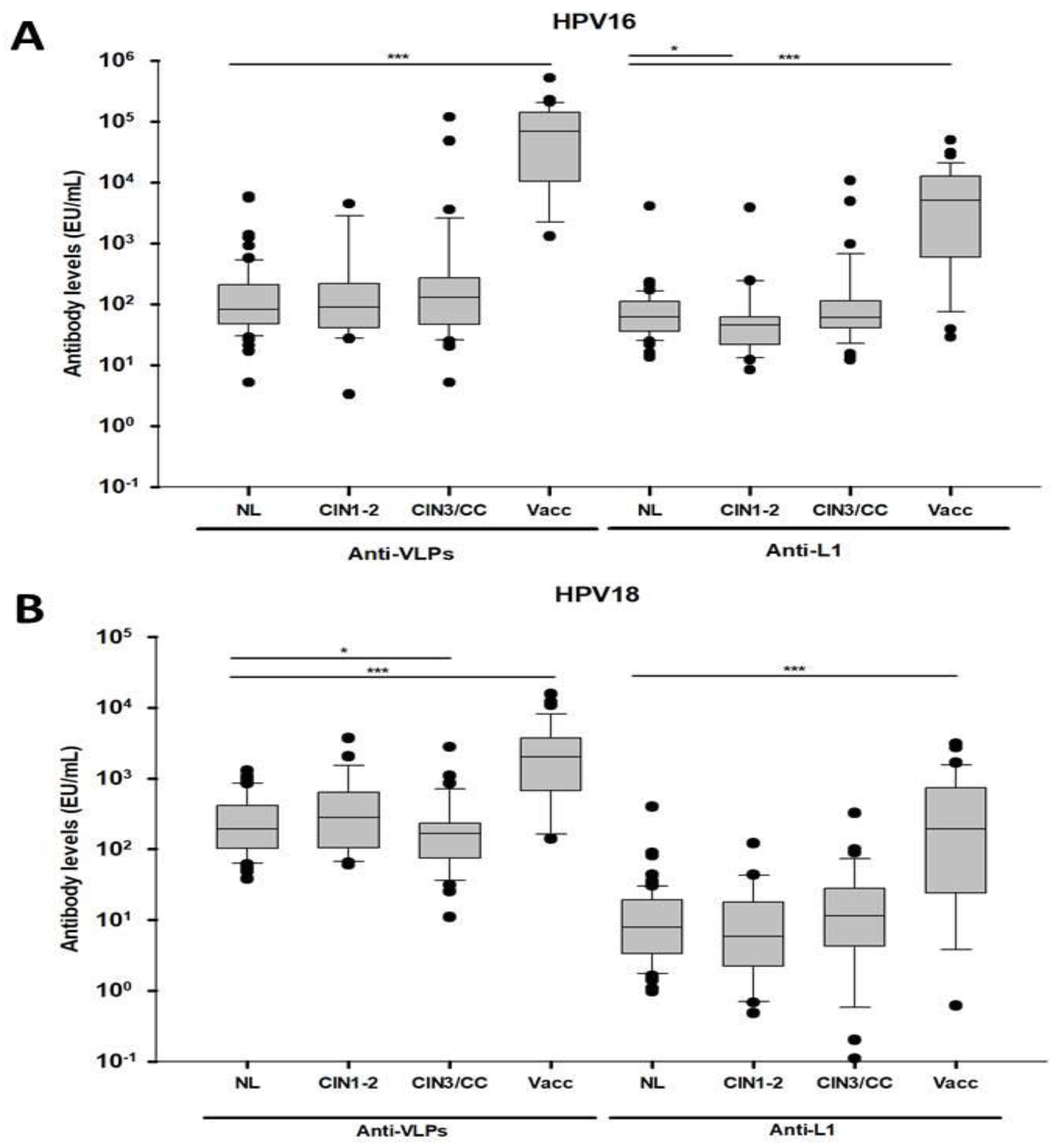
3.2. Differential Antibody Response Against Conformational and Linear L1 Epitopes from HPV 16 and 18 in Women with CIN3/CC and Vaccinated Women
3.3. Differential Association of Antibodies against L1 from HPV16/18 with the Presence of CIN3/CC and HPV Vaccination
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef]
- zur Hausen, H. Papillomaviruses in the causation of human cancers—A brief historical account. Virology 2009, 384, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Campos-Romero, A.; Anderson, K.S.; Longatto-Filho, A.; Luna-Ruiz Esparza, M.A.; Moran-Portela, D.J.; Castro-Menendez, J.A.; Moreno-Camacho, J.L.; Calva-Espinosa, D.Y.; Acosta-Alfaro, M.A.; Meynard-Mejia, F.A.; et al. The burden of 14 hr-HPV genotypes in women attending routine cervical cancer screening in 20 states of Mexico: A cross-sectional study. Sci. Rep. 2019, 9, 10094. [Google Scholar] [CrossRef]
- Roldao, A.; Mellado, M.C.; Castilho, L.R.; Carrondo, M.J.; Alves, P.M. Virus-like particles in vaccine development. Expert Rev. Vaccines 2010, 9, 1149–1176. [Google Scholar] [CrossRef]
- Lowy, D.R.; Schiller, J.T. Prophylactic human papillomavirus vaccines. J. Clin. Investig. 2006, 116, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Stanley, M.; Lowy, D.R.; Frazer, I. Chapter 12: Prophylactic HPV vaccines: Underlying mechanisms. Vaccine 2006, 24 (Suppl. 3), S106–S113. [Google Scholar] [CrossRef] [PubMed]
- Yaddanapudi, K.; Mitchell, R.A.; Eaton, J.W. Cancer vaccines: Looking to the future. Oncoimmunology 2013, 2, e23403. [Google Scholar] [CrossRef]
- Opalka, D.; Lachman, C.E.; MacMullen, S.A.; Jansen, K.U.; Smith, J.F.; Chirmule, N.; Esser, M.T. Simultaneous quantitation of antibodies to neutralizing epitopes on virus-like particles for human papillomavirus types 6, 11, 16, and 18 by a multiplexed luminex assay. Clin. Diagn. Lab. Immunol. 2003, 10, 108–115. [Google Scholar] [CrossRef]
- Dessy, F.J.; Giannini, S.L.; Bougelet, C.A.; Kemp, T.J.; David, M.P.; Poncelet, S.M.; Pinto, L.A.; Wettendorff, M.A. Correlation between direct ELISA, single epitope-based inhibition ELISA and pseudovirion-based neutralization assay for measuring anti-HPV-16 and anti-HPV-18 antibody response after vaccination with the AS04-adjuvanted HPV-16/18 cervical cancer vaccine. Hum. Vaccines 2008, 4, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Harper, D.M.; Franco, E.L.; Wheeler, C.; Ferris, D.G.; Jenkins, D.; Schuind, A.; Zahaf, T.; Innis, B.; Naud, P.; De Carvalho, N.S.; et al. Efficacy of a bivalent L1 virus-like particle vaccine in prevention of infection with human papillomavirus types 16 and 18 in young women: A randomised controlled trial. Lancet 2004, 364, 1757–1765. [Google Scholar] [CrossRef]
- Naud, P.S.; Roteli-Martins, C.M.; De Carvalho, N.S.; Teixeira, J.C.; de Borba, P.C.; Sanchez, N.; Zahaf, T.; Catteau, G.; Geeraerts, B.; Descamps, D. Sustained efficacy, immunogenicity, and safety of the HPV-16/18 AS04-adjuvanted vaccine: Final analysis of a long-term follow-up study up to 9.4 years post-vaccination. Hum. Vaccines Immunother. 2014, 10, 2147–2162. [Google Scholar] [CrossRef]
- Leung, T.F.; Liu, A.P.; Lim, F.S.; Thollot, F.; Oh, H.M.L.; Lee, B.W.; Rombo, L.; Tan, N.C.; Rouzier, R.; De Simoni, S.; et al. Comparative immunogenicity and safety of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine and 4vHPV vaccine administered according to two- or three-dose schedules in girls aged 9-14years: Results to month 36 from a randomized trial. Vaccine 2018, 36, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, C.; Di Bonito, P.; Grasso, F.; Mochi, S.; Accardi, L.; Dona, M.G.; Branca, M.; Costa, S.; Mariani, L.; Agarossi, A.; et al. Clinical and epidemiological correlates of antibody response to human papillomaviruses (HPVs) as measured by a novel ELISA based on denatured recombinant HPV16 late (L) and early (E) antigens. Infect. Agent. Cancer 2008, 3, 9. [Google Scholar] [CrossRef] [PubMed]
- Mehlhorn, G.; Hautmann, S.K.; Koch, M.C.; Strehl, J.D.; Hartmann, A.; Hilfrich, R.; Beckmann, M.W.; Griesser, H. HPV16-L1-specific antibody response is associated with clinical remission of high-risk HPV-positive early dysplastic lesions. Anticancer Res. 2014, 34, 5127–5132. [Google Scholar] [PubMed]
- Jin, Y.; Choi, J.W.; Kim, H.J.; Eddouzi, J.; Kim, S.C.; Ju, W.; Kim, Y.H.; Kim, H.J. Profiling of serum antibodies against human papillomavirus antigens in Korean women with cervical intraepithelial neoplasia and cervical cancer. Cancer Med. 2018, 7, 5655–5664. [Google Scholar] [CrossRef]
- Gutierrez-Xicotencatl, L.; Salazar-Pina, D.A.; Pedroza-Saavedra, A.; Chihu-Amparan, L.; Rodriguez-Ocampo, A.N.; Maldonado-Gama, M.; Esquivel-Guadarrama, F.R. Humoral Immune Response Against Human Papillomavirus as Source of Biomarkers for the Prediction and Detection of Cervical Cancer. Viral Immunol. 2016, 29, 83–94. [Google Scholar] [CrossRef]
- Rocha-Zavaleta, L.; Ambrosio, J.P.; Mora-Garcia Mde, L.; Cruz-Talonia, F.; Hernandez-Montes, J.; Weiss-Steider, B.; Ortiz-Navarrete, V.; Monroy-Garcia, A. Detection of antibodies against a human papillomavirus (HPV) type 16 peptide that differentiate high-risk from low-risk HPV-associated low-grade squamous intraepithelial lesions. J. Gen. Virol. 2004, 85, 2643–2650. [Google Scholar] [CrossRef]
- Urquiza, M.; Sanchez, R.; Amaya, J.; Leon, S.; Acosta, J.; Patarroyo, M.A.; Camargo, M.; Patarroyo, M.E. Specificity of L1 peptides versus virus-like particles for detection of human papillomavirus-positive cervical lesions in females attending Engativa Hospital, Bogota, Colombia. J. Clin. Microbiol. 2008, 46, 3714–3720. [Google Scholar] [CrossRef]
- Ewaisha, R.; Panicker, G.; Maranian, P.; Unger, E.R.; Anderson, K.S. Serum Immune Profiling for Early Detection of Cervical Disease. Theranostics 2017, 7, 3814–3823. [Google Scholar] [CrossRef]
- Xu, Y.F.; Zhang, Y.Q.; Xu, X.M.; Song, G.X. Papillomavirus virus-like particles as vehicles for the delivery of epitopes or genes. Arch Virol. 2006, 151, 2133–2148. [Google Scholar] [CrossRef]
- Park, J.S.; Park, D.C.; Kim, C.J.; Ahn, H.K.; Um, S.J.; Park, S.N.; Kim, S.J.; Namkoong, S.E. HPV-16-related proteins as the serologic markers in cervical neoplasia. Gynecol. Oncol. 1998, 69, 47–55. [Google Scholar] [CrossRef]
- Touze, A.; de Sanjose, S.; Coursaget, P.; Almirall, M.R.; Palacio, V.; Meijer, C.J.; Kornegay, J.; Bosch, F.X. Prevalence of anti-human papillomavirus type 16, 18, 31, and 58 virus-like particles in women in the general population and in prostitutes. J. Clin. Microbiol. 2001, 39, 4344–4348. [Google Scholar] [CrossRef]
- Combita, A.L.; Bravo, M.M.; Touze, A.; Orozco, O.; Coursaget, P. Serologic response to human oncogenic papillomavirus types 16, 18, 31, 33, 39, 58 and 59 virus-like particles in colombian women with invasive cervical cancer. Int. J. Cancer 2002, 97, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Heim, K.; Widschwendter, A.; Pirschner, G.; Wieland, U.; Awerkiew, S.; Christensen, N.D.; Bergant, A.; Marth, C.; Hopfl, R. Antibodies to human papillomavirus 16 L1 virus-like particles as an independent prognostic marker in cervical cancer. Am. J. Obstet. Gynecol. 2002, 186, 705–711. [Google Scholar] [CrossRef]
- Skiba, D.; Mehlhorn, G.; Fasching, P.A.; Beckmann, M.W.; Ackermann, S. Prognostic significance of serum antibodies to HPV-16 L1 virus-like particles in patients with invasive cervical cancer. Anticancer Res. 2006, 26, 4921–4926. [Google Scholar] [PubMed]
- Pedroza-Saavedra, A.; Cruz, A.; Esquivel, F.; De La Torre, F.; Berumen, J.; Gariglio, P.; Gutierrez, L. High prevalence of serum antibodies to Ras and type 16 E4 proteins of human papillomavirus in patients with precancerous lesions of the uterine cervix. Arch. Virol. 2000, 145, 603–623. [Google Scholar] [CrossRef] [PubMed]
- Muller, M.; Zhou, J.; Reed, T.D.; Rittmuller, C.; Burger, A.; Gabelsberger, J.; Braspenning, J.; Gissmann, L. Chimeric papillomavirus-like particles. Virology 1997, 234, 93–111. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.P.; White, W.I.; Palmer-Hill, F.; Koenig, S.; Suzich, J.A. Quantitative disassembly and reassembly of human papillomavirus type 11 viruslike particles in vitro. J. Virol. 1998, 72, 32–41. [Google Scholar] [CrossRef]
- Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 1979, 76, 4350–4354. [Google Scholar] [CrossRef]
- Ludmerer, S.W.; Benincasa, D.; Mark, G.E., 3rd; Christensen, N.D. A neutralizing epitope of human papillomavirus type 11 is principally described by a continuous set of residues which overlap a distinct linear, surface-exposed epitope. J. Virol. 1997, 71, 3834–3839. [Google Scholar] [CrossRef]
- Joura, E.A.; Kjaer, S.K.; Wheeler, C.M.; Sigurdsson, K.; Iversen, O.E.; Hernandez-Avila, M.; Perez, G.; Brown, D.R.; Koutsky, L.A.; Tay, E.H.; et al. HPV antibody levels and clinical efficacy following administration of a prophylactic quadrivalent HPV vaccine. Vaccine 2008, 26, 6844–6851. [Google Scholar] [CrossRef]
- Villa, L.L.; Costa, R.L.; Petta, C.A.; Andrade, R.P.; Paavonen, J.; Iversen, O.E.; Olsson, S.E.; Hoye, J.; Steinwall, M.; Riis-Johannessen, G.; et al. High sustained efficacy of a prophylactic quadrivalent human papillomavirus types 6/11/16/18 L1 virus-like particle vaccine through 5 years of follow-up. Br. J. Cancer 2006, 95, 1459–1466. [Google Scholar] [CrossRef]
- Wang, S.S.; Schiffman, M.; Shields, T.S.; Herrero, R.; Hildesheim, A.; Bratti, M.C.; Sherman, M.E.; Rodriguez, A.C.; Castle, P.E.; Morales, J.; et al. Seroprevalence of human papillomavirus-16, -18, -31, and -45 in a population-based cohort of 10000 women in Costa Rica. Br. J. Cancer 2003, 89, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
- Petrosky, E.Y.; Hariri, S.; Markowitz, L.E.; Panicker, G.; Unger, E.R.; Dunne, E.F. Is vaccine type seropositivity a marker for human papillomavirus vaccination? National Health and Nutrition Examination Survey, 2003–2010. Int. J. Infect. Dis. 2015, 33, 137–141. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coseo, S.E.; Porras, C.; Dodd, L.E.; Hildesheim, A.; Rodriguez, A.C.; Schiffman, M.; Herrero, R.; Wacholder, S.; Gonzalez, P.; Sherman, M.E.; et al. Evaluation of the polyclonal ELISA HPV serology assay as a biomarker for human papillomavirus exposure. Sex. Transm. Dis. 2011, 38, 976–982. [Google Scholar] [CrossRef]
- Goncalves, A.K.; Machado, P.R.; de Souza, L.C.; Costa, A.P.; Gimenes, F.; Consolaro, M.L.; Crispim, J.O.; Eleuterio, J., Jr.; Giraldo, P.C. Detection of immunoglobulin IgA and IgG against human papilloma virus. Viral. Immunol. 2014, 27, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Dillner, L.; Heino, P.; Moreno-Lopez, J.; Dillner, J. Antigenic and immunogenic epitopes shared by human papillomavirus type 16 and bovine, canine, and avian papillomaviruses. J. Virol. 1991, 65, 6862–6871. [Google Scholar] [CrossRef] [PubMed]
- Godi, A.; Bissett, S.L.; Miller, E.; Beddows, S. Relationship between Humoral Immune Responses against HPV16, HPV18, HPV31 and HPV45 in 12-15 Year Old Girls Receiving Cervarix(R) or Gardasil(R) Vaccine. PLoS ONE 2015, 10, e0140926. [Google Scholar] [CrossRef] [PubMed]
- Schiller, J.T.; Lowy, D.R. Immunogenicity testing in human papillomavirus virus-like-particle vaccine trials. J. Infect. Dis. 2009, 200, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Van Doornum, G.J.; Korse, C.M.; Buning-Kager, J.C.; Bonfrer, J.M.; Horenblas, S.; Taal, B.G.; Dillner, J. Reactivity to human papillomavirus type 16 L1 virus-like particles in sera from patients with genital cancer and patients with carcinomas at five different extragenital sites. Br. J. Cancer 2003, 88, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Achour, M.; Zeghal, D.; Kochbati, L.; Kahla, S.; Zouari, F.; Maalej, M.; Oueslati, R. Antibody response for L1, E6 and E7 HPV 16 and HPV 18 antigens in Tunisian women with cervical cancer and controls. J. Immunoass. Immunochem. 2008, 29, 266–280. [Google Scholar] [CrossRef] [PubMed]
- Skjeldestad, F.E.; Mehta, V.; Sings, H.L.; Ovreness, T.; Turpin, J.; Su, L.; Boerckel, P.; Roberts, C.; Bryan, J.; Jansen, K.U.; et al. Seroprevalence and genital DNA prevalence of HPV types 6, 11, 16 and 18 in a cohort of young Norwegian women: Study design and cohort characteristics. Acta Obstet. Gynecol. Scand. 2008, 87, 81–88. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | No Lesion | Lesions | Vaccinated | ||||
|---|---|---|---|---|---|---|---|
| Demographic | n = 68 | % | n = 62 | % | n = 36 | % | |
| Age | |||||||
| 18–27 years | 8 | 11.8 | 4 | 6.5 | 28 | 77.8 | |
| 28–37 years | 19 | 27.9 | 13 | 21.0 | 5 | 13.9 | |
| ≥38 years | 41 | 60.3 | 45 | 72.5 | 3 | 8.3 | |
| Total | 68 | 62 | 36 | ||||
| Marital status | |||||||
| Single | 1 | 1.5 | 5 | 8.0 | 28 | 77.8 | |
| Married or steady partner | 63 | 92.6 | 52 | 84.0 | 8 | 22.2 | |
| Widow, Divorced or separated | 4 | 5.9 | 5 | 8.0 | 0 | 0.0 | |
| Total | 68 | 62 | 36 | ||||
| Educational level | |||||||
| Basic | 52 | 76.5 | 28 | 77.8 | 0 | 0.0 | |
| ≥High School | 11 | 16.2 | 2 | 5.5 | 36 | 100.0 | |
| No education | 5 | 7.3 | 6 | 16.7 | 0 | 0.0 | |
| Missing | - | - | 26 | - | - | - | |
| Total | 68 | 62 | 36 | ||||
| Smoking | |||||||
| No | 59 | 86.8 | 32 | 88.9 | 21 | 58.3 | |
| Yes | 9 | 13.2 | 4 | 11.1 | 15 | 41.7 | |
| Missing | - | - | 26 | - | - | - | |
| Total | 68 | 62 | 36 | ||||
| Sexual Behavior | |||||||
| Time of sexual activity 1 | |||||||
| ≤7 years | 6 | 8.8 | 4 | 6.4 | 26 | 72.2 | |
| 8–17 years | 19 | 27.9 | 12 | 19.4 | 7 | 19.4 | |
| 18–27 years | 25 | 36.8 | 30 | 48.4 | 1 | 2.8 | |
| ≥28 years | 18 | 26.5 | 16 | 25.8 | 2 | 5.6 | |
| Total | 68 | 62 | 36 | ||||
| Number of sexual partners last year | |||||||
| 0–1 | 63 | 92.7 | 18 | 29.0 | 18 | 50.0 | |
| ≥2 | 5 | 7.3 | 44 | 71.0 | 18 | 50.0 | |
| Total | 68 | 62 | 36 | ||||
| Diagnostic (Pap or histopathology) | |||||||
| Normal | 68 | 100 | - | - | - | - | |
| CIN1 | - | - | 15 | 24.2 | - | - | |
| CIN2 | - | - | 8 | 12.9 | - | - | |
| CIN3 | - | - | 10 | 16.1 | - | - | |
| CC | - | - | 29 | 46.8 | - | - | |
| Total | 68 | 62 | |||||
| HPV STATUS | |||||||
| Negative | 42 | 61.8 | 15 | 24.2 | - | - | |
| Positive | 26 | 38.2 | 47 | 75.8 | - | - | |
| HPV genotypes | |||||||
| HPV16 | 6 | 23.1 | 39 | 82.9 | - | - | |
| HR-HPV | 17 | 65.4 | 8 | 17.1 | - | - | |
| LR-HPV | 3 | 11.5 | 0 | 0.0 | - | - | |
| Anti-VLPs Seropositivity | |||||||||||
| Variable | Total | HPV16 (n = 49) | HPV18 (n = 25) | ||||||||
| n | n | (%) | OR | (CI 95%) | p | n | (%) | OR | (CI 95%) | p | |
| Population Group | |||||||||||
| Normal Adult | 68 | 5 | (7.4) | 1.00 | 0 | (−) | |||||
| CIN1-2 | 23 | 2 | (8.7) | 1.20 | (0.21–6.65) | 0.835 | 2 | (8.7) | 1.00 3 | ||
| CIN3/CC | 39 | 6 | (15.4) | 2.29 | (0.65–8.07) | 0.197 | 1 | (2.6) | 0.27 | (0.23–3.23) | 0.305 |
| Vaccinated | 36 | 36 | (100.0) | 2.11 × 108 | 0.000 | 22 | (61.1) | 16.50 | (3.33–81.53) | 0.001 | |
| No. sexual partners last year | |||||||||||
| 0–1 | 99 | 23 | (23.2) | 1.00 | 14 | (14.1) | 1.00 | ||||
| ≥2 | 67 | 26 | (38.8) | 2.09 | (1.06–4.12) | 0.032 | 11 | (16.4) | 1.19 | (0.50–2.81) | 0.688 |
| Smoking 1 | |||||||||||
| No | 112 | 28 | (25.0) | 1.00 | 14 | (12.5) | 1.00 | ||||
| Yes | 28 | 16 | (57.1) | 3.99 | (1.68–9.47) | 0.002 | 10 | (35.7) | 3.88 | (1.49–10.10) | 0.005 |
| Anti-L1 Seropositivity | |||||||||||
| Variable | Total | HPV16 (n = 40) | HPV18 (n = 32) | ||||||||
| n | n | (%) | OR | (CI 95%) | p | n | (%) | OR | (CI 95%) | p | |
| Population Group 2 | |||||||||||
| Normal Adult | 68 | 1 | (1.5) | 1.00 | 3 | (4.4) | 1.00 | ||||
| CIN1-2 | 23 | 1 | (4.4) | 3.04 | (0.18–50.75) | 0.438 | 1 | (4.4) | 0.98 | (0.97–9.96) | 0.990 |
| CIN3/CC | 39 | 6 | (15.4) | 12.18 | (1.40–105.38) | 0.023 | 3 | (7.7) | 1.80 | (0.34–9.41) | 0.483 |
| Vaccinated | 36 | 32 | (88.9) | 536.0 | (57.55–4991.7) | 0.000 | 25 | (69.4) | 49.2 | (12.67–191.3) | 0.000 |
| No. sexual partners last year | |||||||||||
| 0–1 | 99 | 18 | (18.2) | 1.00 | 16 | (16.1) | 1.00 | ||||
| ≥2 | 67 | 22 | (32.8) | 2.20 | (1.06–4.52) | 0.032 | 16 | (23.9) | 1.62 | (0.74–3.53) | 0.219 |
| Smoking 1 | |||||||||||
| No | 112 | 20 | (17.9) | 1.00 | 17 | (15.2) | 1.00 | ||||
| Yes | 28 | 15 | (53.6) | 5.30 | (2.18–12.87) | 0.000 | 13 | (46.4) | 4.84 | (1.96–11.96) | 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pedroza-Saavedra, A.; Rodriguez-Ocampo, A.N.; Salazar-Piña, A.; Perez-Morales, A.C.; Chihu-Amparan, L.; Maldonado-Gama, M.; Cruz-Valdez, A.; Esquivel-Guadarrama, F.; Gutierrez-Xicotencatl, L. Differential Antibody Response against Conformational and Linear Epitopes of the L1 Proteins from Human Papillomavirus Types 16/18 Is Observed in Vaccinated Women or with Uterine Cervical Lesions. Vaccines 2021, 9, 442. https://doi.org/10.3390/vaccines9050442
Pedroza-Saavedra A, Rodriguez-Ocampo AN, Salazar-Piña A, Perez-Morales AC, Chihu-Amparan L, Maldonado-Gama M, Cruz-Valdez A, Esquivel-Guadarrama F, Gutierrez-Xicotencatl L. Differential Antibody Response against Conformational and Linear Epitopes of the L1 Proteins from Human Papillomavirus Types 16/18 Is Observed in Vaccinated Women or with Uterine Cervical Lesions. Vaccines. 2021; 9(5):442. https://doi.org/10.3390/vaccines9050442
Chicago/Turabian StylePedroza-Saavedra, Adolfo, Angelica Nallelhy Rodriguez-Ocampo, Azucena Salazar-Piña, Aislinn Citlali Perez-Morales, Lilia Chihu-Amparan, Minerva Maldonado-Gama, Aurelio Cruz-Valdez, Fernando Esquivel-Guadarrama, and Lourdes Gutierrez-Xicotencatl. 2021. "Differential Antibody Response against Conformational and Linear Epitopes of the L1 Proteins from Human Papillomavirus Types 16/18 Is Observed in Vaccinated Women or with Uterine Cervical Lesions" Vaccines 9, no. 5: 442. https://doi.org/10.3390/vaccines9050442
APA StylePedroza-Saavedra, A., Rodriguez-Ocampo, A. N., Salazar-Piña, A., Perez-Morales, A. C., Chihu-Amparan, L., Maldonado-Gama, M., Cruz-Valdez, A., Esquivel-Guadarrama, F., & Gutierrez-Xicotencatl, L. (2021). Differential Antibody Response against Conformational and Linear Epitopes of the L1 Proteins from Human Papillomavirus Types 16/18 Is Observed in Vaccinated Women or with Uterine Cervical Lesions. Vaccines, 9(5), 442. https://doi.org/10.3390/vaccines9050442






