The Implementation of Mass-Vaccination against SARS-CoV-2: A Systematic Review of Existing Strategies and Guidelines
Abstract
1. Introduction
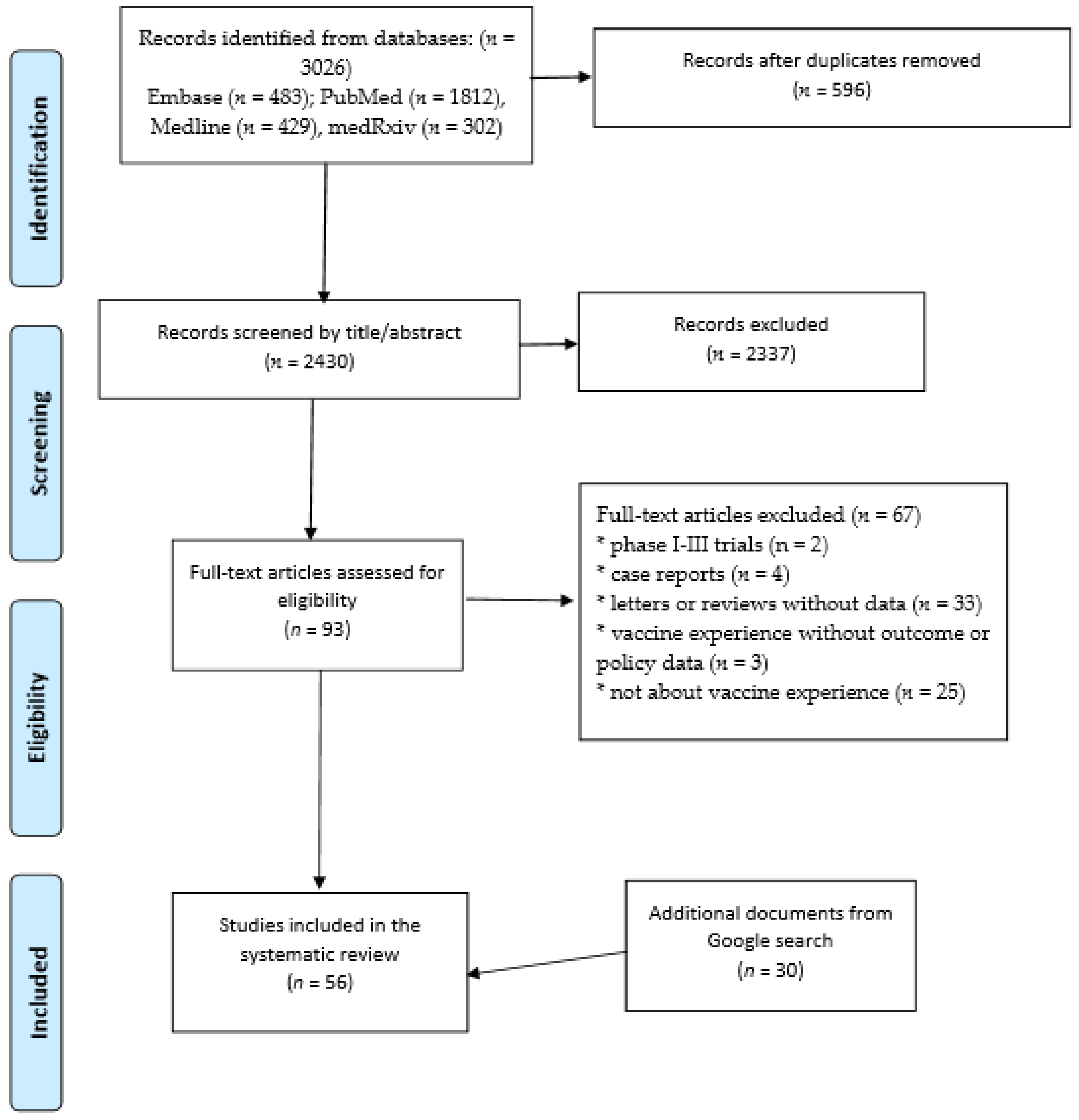
2. Materials and Methods
2.1. Search Strategy—Research Articles
2.2. Search Strategy: Non-Peer Reviewed Literature
2.3. Data extraction and Analysis
2.4. Ethical Issues
3. Results
3.1. National Policy Documents
3.2. Vaccine Deployment
3.3. Vaccination of Priority Groups
3.4. Infrastructure and Staffing for Vaccination
3.5. Peer-Reviewed Research of Vaccination Outcomes
| Setting/Country | Australia | USA | Czech Republic | Qatar | Singapore | Monaco | Canada | Switzerland | EU | UK | South Africa | Lebanon | Cyprus | New Zealand | Ireland |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Date of most recent update | 17 February 2021 | 29 October 2020 | 10 February 2021 | NR | 26 February 2021 | 23 February 2021 | 25 February 2021 | 26 February 2021 | 1 February 2021 | 11 January 2021 | NR | 28 January 2021 | 2020 | 22 December 202 | 24 February 2021 |
| Reference | [11] | [12] | [15] | [16] | [17] | [18] | [19] | [20] | [21] | [22] | [23] | [24] | [25] | [26] | [27] |
| Funding source | Public | Public | Public | Public | Public | Public | Public | Public | Public | Public | Public | Public | Public | Public | Public |
| Pf/B Mod | Pf/B | Private | AZ/Ox | COVAX | NR | ||||||||||
| Vaccine used | Pf/B | NR | NR | NR | Pf/B | No | Pf/B | Pf/B | Pf/B | Pf/B | NR | NR | NR | NR | Pf/B |
| NR | Mod | NR | Mod | Mod | NR | NR | Mod | ||||||||
| AZ/Ox | AZ/Ox | AZ/Ox | AZ/Ox | ||||||||||||
| Vaccination mandatory for target populations? | No | NR | NR | NR | No | No | No | No | No | No | NR | NR | No | ||
| Was an increased delay between the two doses recommended (compared to the frequency in published trials) | NR | NR | NR | NR | Priority 1 | NR | NR | In 2 countries | Yes | Priority 1 | NR | NR | NR | ||
| EQUITABLE ACCESS | Priority 1 | Priority 2 | |||||||||||||
| Priority criteria for vaccination | NR | Priority 1 | |||||||||||||
| Frontline HCW | Priority 1 | Priority 1 | Priority 1 | Priority 1 | Priority 1 | Priority 2 | Priority 1 | Priority 2 | Priority 2 | Priority 1 | Priority 1 | Priority 2 | |||
| Chronic comorbidities | Priority 2 | Priority 2 | Priority 2 | Priority 1 | Priority 1 | Priority 1 | Priority 4 | Priority 4 | |||||||
| Elderly in aged care | Priority 1 | Priority 2 | Priority 1 | Priority 1 | Priority 1 | Priority 1 | Priority 1 | Priority 1 | Priority 1 | ||||||
| Other elderly | Priority 2 | Priority 2 | Priority 1 | Priority 1 | Priority 1 | Priority 1 | Priority 2 | Priority 2 | Priority 1 | Priority 3 | |||||
| Institutionalised | Priority 3 | Priority 2 | Priority 4 | Priority 2 | Priority 5 | ||||||||||
| Indigenous population | Priority 2 | Priority 3 | Priority 1 | ||||||||||||
| Other HCW | Priority 2 | Priority 2 | NR | NR | Priority 1 | NR | Priority 2 | NR | |||||||
| Essential public services | Priority 3 | Priority 2 | NR | NR | NR | Yes | |||||||||
| Children | |||||||||||||||
| Expected date when whole adult population vaccinated | NR | NR | NR | NR | Sep 2021 | NR | NR | NR | NR | NR | Mid 2022 | NR | NR | ||
| Will extra doses be donated to COVAX | Yes | NR | NR | NR | Yes | NR | Yes | NR | Yes | NR | NR | NR | |||
| VACCINATION STRATEGIES | |||||||||||||||
| Location of vaccination | NR | NR | NR | NR | |||||||||||
| Large public venues | Yes | Yes | Drive through | Care home | Yes | Yes | Yes | Yes | Yes | ||||||
| Hospitals | Yes | NR | NR | Yes | NR | Electronic | |||||||||
| Clinics and pharmacies Other | Yes | Yes | NR | Self-reported to clinic, hotline, or email | Yes | Yes | NR | Yes | NR | Yes | |||||
| Vaccination record (electronic/paper) | NR | NR | NR | NR | NR | Paper | Electronic | Electronic | Electronic | Electronic | NR | ||||
| Adverse-effect reporting (automated electronic, electronic self-report, GP self-report) | Active surveillance via electronic prompts | NR | NR | NR | Self-reported to local doctor | NR | NR | NR | 5 countries with self-reported electronic system | Review of electronic health records | NR | Self-reported electronic system | NR | 9 extra freezer procured | NR |
| COORDINATION | |||||||||||||||
| Cold chain infrastructure | NR | Extra freezer procured | NR | NR | NR | NR | NR | NR | Extra freezer procured | NR | NR | 1 freezer procured | Freezer available | NR | |
| PLANNING | Yes | ||||||||||||||
| Increased labour requirements | NR | NR | NR | NR | Yes | NR | |||||||||
| Non-medical staff employed for vaccination | Yes | Yes | Yes | Yes | Yes | Yes | |||||||||
| Training provided to staff | Yes | Yes | NR | NR | Yes | Yes | NR | Yes | NR | ||||||
| COMMUNITY ENGAGEMENT | |||||||||||||||
| Are the following employed? | NR | NR | NR | NR | NR | NR | |||||||||
| Community strategies | Yes | Yes | Yes | ||||||||||||
| Social media | Yes | Yes | Yes | ||||||||||||
| Media campaigns | Yes | Yes | NR | NR | Yes | Yes | NR | Yes | NR | ||||||
| Other | Qatar | Monaco | Celebrities | South Africa | New Zealand | ||||||||||
| Strategies to reduce misinformation | NR | NR | NR | NR | NR | 23 February 2021 | NR | NR | Monitor media, online fact checker | NR | NR | Rumour tracking team | NR | 22 December 202 | NR |
| Country/Reference | Peer Reviewed (Yes/No) | Number Vaccinated in Study | Age Groups Immunised | Female (%) | Vaccines Used (% *) | Outcomes |
|---|---|---|---|---|---|---|
| Studies reporting demographic details | ||||||
| USA Gharpure et al. [40] | Yes | 713,909 (LTCF residents) 582,104 (HCW) | NR | NR | NR | Estimated 77.8% residents and 37.5% staff in 11,460 care facilities, vaccinated by Jan 17 (at least one dose) |
| USA Painter et al. [41] | Yes | 12,928,749 | <0.1% <18 70.9% 18–64 29% ≥65 | 63 | Pf/B Mod | |
| Studies reporting adverse events | ||||||
| Brazil Pagotta et al. [53] | No | 683 (HCW) | 99.3% 18–60 0.7% >60 | 68 | Gamaleya | Adverse effects reported by 71.3% in a survey. Most common—injection site pain, myalgia, fever |
| India Jayadevan et al. [49] | No | 5396 | NR | NR | BB (95) AZ/Ox (3.3) Pf/B (0.8) Sinopharm (0.8) | Adverse effects reported by 65.9% in a survey, highest in those 20–39 years of age and females. Most common adverse effects—lethargy, myalgia, fever |
| USA CDC [42] | Yes | 4,041,396 | NR | NR | Mod | Anaphylaxis reported in 0.0002% (n = 10), serious adverse effects in 0.03% (n = 1266) |
| USA CDC [43] | Yes | 1,893,360 | NR | NR | Pf/B | Anaphylaxis reported in 0.001% (n = 21), serious adverse effects in 0.2% (n = 4393) |
| USA Gee et al. [44] | Yes | 13,794,904 | 0.2% 0–17 90.3% 18–64 6.2% ≥65 | 61 | Pf/B Mod | Adverse effects reported in 0.05% (n = 6994) through a passive national surveillance system |
| USA McMurray et al. [45] | No | 31,029 | NR | NR | Pf/B Mod | Review of electronic medical records found a 2.1 to 1500 times reduced frequency of adverse events reported when events were obtained from self-reported health interactions compared to active solicitation in trials or in post-marketing surveillance |
| Studies reporting vaccine acceptance | ||||||
| Saudi Arabia Barry et al. [50] | No | 352 (HCW) | 91.9% 20–50 8.1% >50 | 57 | Pf/B | Factors associated with not enrolling for vaccine: female, younger age, use of social media, foreign national. Percentage not yet registered for vaccine: 66.7% (n = 706) |
| UK Martin et al. [51] | No | 12,278 (HCW) | 18.7% <30 72% 30–60 9.3% >60 | 76 | Pf/B AZ/Ox | Factors associated with lower vaccine uptake: ethnic minority, younger age, female, lower socio-economic status |
| UK Kim [52] | No | 66,994 | NR | NR | NR | Factors associated with vaccine uptake—pre-pandemic income, education. Least likely to take up vaccination—Black Hispanics |
| USA Pamplona, Sullivan, Kotanko [46] | Yes | 115 (HCW) | NR | NR | NR | Factors associated with not being vaccinated in a dialysis ward (26.8%, n = 42)—past COVID-19, pregnancy, absence. 3.8% (n = 6) declined vaccine |
| USA Schradering et al. [47] | Yes | 1136 (HCW) | 98.6% 22–64 1.4% ≥65 | 59 | NR | 14% (n = 195) HCWs refused vaccination—usually for concern about adverse effects. Ethnic group most likely to decline vaccine: with non-Hispanic black HCWs |
| Studies reporting efficacy (excluding Phase 2 and 3 trials of vaccine efficacy) | ||||||
| Israel Abu Jabal et al. [30] | Yes | 514 | 2.1% <30 79.4% 30–59 18.5% ≥60 | 63 | Pf/B | Immunogenicity post vaccination similar by ethnicity and sex, but decrease with age. Increased immunogenicity in previous COVID-19 cases |
| Israel Amit et al. [31] | Yes | 4081 | NR | NR | Pf/B | In vaccinated HCW, 0.54% (n = 22) developed COVID-19 within 10 days of vaccination |
| Israel Amit et al. [32] | Yes | NR | NR | NR | NR | 30% and 75% reduction in SARS-CoV-2 cases in vaccinated HCW vs. unvaccinated HCW 14 and 28 days, respectively after vaccination |
| Israel Aran [33] | No | NR | NR | NR | Pf/B | 72% reduction in cases and 83% reduction in hospitalisation by modelling |
| Israel Chodick et al. [34] | No | 503,875 | Mean 59.7 | 52 | Pf/B | Cases of COVID-19 infection 24 days post vaccination: 0.84% (n = 3098) 51% vaccine effectiveness calculated after 1st dose |
| Israel Dagan et al. [35] | Yes | 596,618 | 72% <60 28% ≥60 | 50 | Pf/B | 92% reduction in COVID-19 cases, 87% reduction in hospitalisation, 72% reduction in deaths: at 7 days after second dose |
| Israel De Leon et al. [36] | No | NR | NR | NR | >50% estimated vaccine effectiveness by modelling | |
| Israel Levine et al. [37] | No | 1755 COVID-19 cases after vaccination | NR | NR | NR | Four-fold reduction in SARS-CoV-2 viral load for people developing infections 12–28 days after first dose |
| Israel Petter et al. [38] | No | NR | NR | NR | NR | 1.6 to 20x reduction in overall SARS-CoV-2 viral load by vaccinating the community |
| Israel Rossman et al. [39] | No | NR | NR | NR | Pf/B | 49% drop in cases, 36% drop in hospitalisations: 1.5 months after vaccine initiation |
| Scotland Vasileious et al. [54] | Yes | 1,137,775 | 34.8% 18–64 65.2% ≥65 | 61 | Pf/B | Estimated efficacy of single dose 85% (Pfizer) and 94% (AstraZeneca) at 28–34 days post vaccination 81% reduction in hospitalisation in those over 80 |
| UK Hall et al. [55] | Yes | 20,641 | 16.1% <25 76% 25–64 7.9% ≥65 | 85 | Pf/B (94) AZ/Ox (6) | Vaccine effectiveness at 21 days: 72% after 1 dose, 86% after 2 doses 67% receiving vaccine had previous COVID-19 |
| USA Bradley, Grundberg, Selvarangan [48] | No | 188 (HCW) | NR | NR | NR | HCW with previous documented SARS-COV-2 infection have higher IgG titres after COVID-19 vaccination |
4. Discussion
- -
- Vaccinate health care workers, elderly, and those with chronic comorbidities as a priority.
- -
- Utilise mass vaccination hubs such as sporting venues, for maximal scale and efficiency.
- -
- Consider novel delivery techniques such as drive-through clinics for the second dose, where this is feasible and culturally acceptable.
- -
- Consider use of automated electronic surveys for monitoring side-effects among all recipients.
- -
- Engage the community to increase vaccination awareness and acceptance including by
- ○
- Including and training volunteers in the vaccination effort.
- ○
- Using social media and media campaigns to raise awareness.
- ○
- Having a means to monitor mis-information.
- ○
- Utilising high profile "champions", such as political leaders and social celebrities. Support COVAX and global commitments to deliver vaccines to marginalised and vulnerable populations, because everyone deserves to be protected and the global population cannot be safe until we are all vaccinated.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- John Hopkins University. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University; John Hopkins University: Baltimore, MD, USA, 2021; Available online: https://coronavirus.jhu.edu/map.html (accessed on 1 March 2021).
- Pierce, M.; Hope, H.; Ford, T.; Hatch, S.; Hotopf, M.; Kontopantelis, E.; John, A.; Webb, R.T.; Wessely, S.; McManus, S.; et al. Mental health before and during the COVID-19 pandemic: A longitudinal probability sample survey of the UK population. Lancet Psychiatry 2020, 7, 883–892. [Google Scholar] [CrossRef]
- Wadhera, R.; Wadhera, P.; Gaba, P.; Figueroa, J.F.; Maddox, K.E.J.; Yeh, R.W.; Shen, C. Variation in COVID-19 Hospitalizations and Deaths Across New York City Boroughs. JAMA 2020, 323, 2192–2195. [Google Scholar] [CrossRef] [PubMed]
- Covid-19-will-become-endemic-but-with-decreased-potency-over-time,-scientists-believe. BMJ 2021, 372, n492.
- World Health Organisation. The COVID-19 Candidate Vaccine Landscape and Tracke; World Health Organisation: Geneva, Switzerland, 2021. [Google Scholar]
- Paltiel, A.; Schwartz, J.; Zheng, A.; Walensky, R.P. Clinical outcomes of A COVID-19 vaccine: Implementation over efficacy. Health Aff. 2020, 40, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.; Wong, E.; Huang, J.; Cheung, A.; Law, K.; Chong, M.; Ng, R.W.Y.; Lai, C.K.C.; Boon, S.S.; Lau, J.T.F.; et al. Acceptance of the COVID-19 vaccine based on the health belief model: A population-based survey in Hong Kong. Vaccine 2021, 39, 1148–1156. [Google Scholar] [CrossRef]
- Our World in Data. Coronavirus (COVID-19) Vaccinations. University of Oxford. 2021. Available online: https://ourworldindata.org/covid-vaccinations (accessed on 1 March 2021).
- Rosen, B.; Waitzberg, R.; Israeli, A. Israel’s rapid rollout of vaccinations for COVID-19. Isr. J. Health Policy Res. 2021, 10, 6. [Google Scholar] [CrossRef]
- World Health Organisation. Framework for Decision-Making: Implementation of Mass Vaccination Campaigns in the Context of COVID-19; World Health Organisation: Geneva, Switzerland, 2020. [Google Scholar]
- Australian Government. Australian COVID-19 Vaccination Policy; Australian Government: Canberra, Australia, 2021.
- Centers for Disease Control and Prevention. COVID-19 Vaccination Program Interim Playbook for Jurisdiction Operations; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2021. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- The World Bank. Countries and Economies; The World Bank: Washington, DC, USA, 2021. Available online: https://data.worldbank.org/country (accessed on 1 March 2021).
- Vnitra, M. How Does the Prioritization System Works? Ministerstvo vnitra: Prague, Czech Republic, 2021. Available online: https://covid.gov.cz/en/situations/register-vaccination/how-does-prioritization-system-works (accessed on 1 March 2021).
- Ministry of Public Health. Coronavirus 2019 (COVID-19); Ministry of Public Health: Doha, Qatar, 2021. Available online: https://covid19.moph.gov.qa/EN/Covid19-Vaccine/Pages/FAQ.aspx (accessed on 1 March 2021).
- Government of Singapore. COVID-19 Vaccination; Government of Singapore: Singapore, 2021. Available online: https://www.gov.sg/faq (accessed on 1 March 2021).
- Gouvernement Princier. Vaccination COVID-19; Gouvernement Princier: Monaco, 2021. Available online: https://covid19.mc/en/mesures/tout-sur-les-vaccins/ (accessed on 1 March 2021).
- Government of Canada. Vaccines and Treatments for COVID-19: Vaccine Rollout. 2021. Available online: https://www.canada.ca/en/public-health/services/diseases/2019-novel-coronavirus-infection/prevention-risks/covid-19-vaccine-treatment/vaccine-rollout.html (accessed on 1 March 2021).
- FOPH Federal Office of Public Health. Coronavirus: Vaccination. 2021. Available online: https://www.bag.admin.ch/bag/en/home/krankheiten/ausbrueche-epidemien-pandemien/aktuelle-ausbrueche-epidemien/novel-cov/impfen.html (accessed on 1 March 2021).
- European Centre for Disease Prevention and Control. Overview of the Implementation of COVID-19 Vaccination Strategies and Vaccine Deployment Plans in the EU/EEA; European Centre for Disease Prevention and Control: Stockholm, Sweden, 2021. [Google Scholar]
- Department of Health and Social Care. UK COVID-19 Vaccines Delivery Plan; Department of Health and Social Care: London, UK, 2021.
- South African Government. COVID-19/Novel Coronavirus. South African Government, 2021. Available online: https://www.gov.za/covid-19/vaccine/vaccine# (accessed on 1 March 2021).
- Ministry of Public Health. Lebanon National Deployment and Vaccination Plan for COVID-19 Vaccines; Ministry of Public Health: Beirut, Lebanon, 2021.
- Ministry of Health. National Vaccination Plan for COVID 19; Minstry of Health: Nicosia, Cyprus, 2021.
- Ministry of Health. COVID-19: Vaccine Strategy and Planning; Ministry of Health: Auckland, New Zealand, 2021. Available online: https://www.health.govt.nz/our-work/diseases-and-conditions/covid-19-novel-coronavirus/covid-19-vaccines/covid-19-vaccine-strategy-and-planning (accessed on 1 March 2021).
- Health Service Executive. Rollout of COVID-19 Vaccines in Ireland; Health Service Executive: Dublin, Ireland, 2021; Available online: https://www2.hse.ie/screening-and-vaccinations/covid-19-vaccine/rollout-covid-19-vaccines-ireland.html (accessed on 1 March 2021).
- datrix.be. Covid Vaccinations Belgium. 2021. Available online: https://covid-vaccinatie.be/en (accessed on 1 March 2021).
- Gov.UK. Coronavirus (COVID-19) in the UK. 2021. Available online: https://coronavirus.data.gov.uk/details/vaccinations (accessed on 1 March 2021).
- Abu Jabal, K.; Ben-Amram, H.; Beiruti, K.; Batheesh, Y.; Sussan, C.; Zarka, S.; Edelstein, M. Impact of age, ethnicity, sex and prior infection status on immunogenicity following a single dose of the BNT162b2 mRNA COVID-19 vaccine: Real-world evidence from healthcare workers, Israel, December 2020 to January 2021. Euro Surveill. 2021, 26, 2100096. [Google Scholar] [CrossRef]
- Amit, S.; Beni, S.A.; Biber, A.; Grinberg, A.; Leshem, E.; Regev-Yochay, G. Post-Vaccination COVID-19 among Healthcare Workers, Israel. Emerg. Infect. Dis. 2021, 27. [Google Scholar] [CrossRef]
- Amit, S.; Regev-Yochay, G.; Afek, A.; Kreiss, Y.; Leshem, E. Early rate reductions of SARS-CoV-2 infection and COVID-19 in BNT162b2 vaccine recipients. Lancet 2021, 397, 875–877. [Google Scholar] [CrossRef]
- Aran, D. Estimating real-world COVID-19 vaccine effectiveness in Israel using aggregated counts. medRxiv 2021. [Google Scholar] [CrossRef]
- Chodick, G.; Tene, L.; Patalon, T.; Gazit, S.; Tov, A.B.; Cohen, D.; Muhsen, K. The effectiveness of the first dose of BNT162b2 vaccine in reducing SARS-CoV-2 infection 13–24 days after immunization: Real-world evidence. medRxiv 2021. [Google Scholar] [CrossRef]
- Dagan, N.; Barda, N.; Kepten, E.; Miron, O.; Perchik, S.; Katz, M.A.; Hernán, M.A.; Lipsitch, M. BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting. N. Engl. J. Med. 2021. [Google Scholar] [CrossRef]
- De-Leon, H.; Calderon-Margalit, R.; Pederiva, F.; Ashkenazy, Y.; Gazit, D. First indication of the effect of COVID-19 vaccinations on the course of the outbreak in Israel. medRxiv 2021. [Google Scholar] [CrossRef]
- Levine-Tiefenbrun, M.; Yelin, I.; Katz, R.; Herzel, E.; Golan, Z.; Schreiber, L.; Wolf, T.; Nadler, V.; Ben-Tov, A.; Kuint, J.; et al. Decreased SARS-CoV-2 viral load following vaccination. medRxiv 2021. [Google Scholar] [CrossRef]
- Petter, E.; Mor, O.; Zuckerman, N.; Oz-Levi, D.; Younger, A.; Aran, D.; Erlich, Y. Initial real world evidence for lower viral load of individuals who have been vaccinated by BNT162b2. medRxiv 2021. [Google Scholar] [CrossRef]
- Rossman, H.; Shilo, S.; Meir, T.; Gorfine, M.; Shalit, U.; Segal, E.; Patterns of COVID-19 pandemic dynamics following deployment of a broad national immunization program. medRxiv 2021. Available online: http://medrxiv.org/content/early/2021/02/09/2021.02.08.21251325.abstract (accessed on 2 March 2021).
- Gharpure, R.; Guo, A.; Bishnoi, C.K.; Patel, U.; Gifford, D.; Tippins, A.; Jaffe, A.; Shulman, E.; Stone, N.; Mungai, E.; et al. Early COVID-19 First-Dose Vaccination Coverage Among Residents and Staff Members of Skilled Nursing Facilities Participating in the Pharmacy Partnership for Long-Term Care Program—United States, December 2020–January 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 178–182. [Google Scholar] [CrossRef]
- Painter, E.M.; Ussery, E.N.; Patel, A.; Hughes, M.M.; Zell, E.R.; Moulia, D.L.; Scharf, L.G.; Lynch, M.; Ritchey, M.D.; Toblin, R.L.; et al. Demographic Characteristics of Persons Vaccinated During the First Month of the COVID-19 Vaccination Program—United States, December 14, 2020–January 14, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 174–177. [Google Scholar] [CrossRef] [PubMed]
- CDC Covid-Response Team, Food Drug Administration. Allergic Reactions Including Anaphylaxis After Receipt of the First Dose of Moderna COVID-19 Vaccine—United States, December 21, 2020–January 10, 2021. MMWR Morb. Mortal. Wkly. Rep 2021, 70, 125–129. [Google Scholar] [CrossRef] [PubMed]
- CDC Covid-Response Team, Food Drug Administration. Allergic Reactions Including Anaphylaxis After Receipt of the First Dose of Pfizer-BioNTech COVID-19 Vaccine— United States, December 14–23, 2020. MMWR Morb. Mortal. Wkly. Rep 2021, 70, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Gee, J.; Marquez, P.; Su, J.; Calvert, G.M.; Liu, R.; Myers, T.; Nair, N.; Martin, S.; Clark, T.; Markowitz, L.; et al. First Month of COVID-19 Vaccine Safety Monitoring—United States, December 14, 2020–January 13, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 283–288. [Google Scholar] [CrossRef]
- McMurry, R.; Lenehan, P.J.; Awasthi, S.; Silvert, E.; Puranik, A.; Pawlowski, C.; Venkatakrishnan, A.J.; Anand, P.; Agarwal, V.; O’Horo, J.C.; et al. Real-time analysis of a mass vaccination effort confirms the safety of FDA-authorized mRNA vaccines for COVID-19 from Moderna and Pfizer/BioNtech. medRxiv 2021. [Google Scholar] [CrossRef]
- Pamplona, G.; Sullivan, T.; Kotanko, P. COVID-19 vaccination acceptance and hesitancy in dialysis staff: First results from New York City. Kidney Int. Rep. 2021. [Google Scholar] [CrossRef]
- Schrading, W.A.; Trent, S.A.; Paxton, J.H.; Rodriguez, R.M.; Swanson, M.B.; Mohr, N.M.; Talan, D.A.; Project COVERED Emergency Department Network. Vaccination Rates and Acceptance of SARS-CoV-2 Vaccination among US Emergency Department Health Care Personnel. Acad. Emerg. Med 2021. [Google Scholar] [CrossRef]
- Bradley, T.; Grundberg, E.; Selvarangan, R. Antibody responses boosted in seropositive healthcare workers after single dose of SARS-CoV-2 mRNA vaccine. medRxiv 2021. [Google Scholar]
- Jayadevan, R.; Shenoy, R.; Ts, A. Survey of symptoms following COVID-19 vaccination in India. medRxiv 2021. [Google Scholar] [CrossRef]
- Barry, M.; Temsah, M.-H.; Aljamaan, F.; Saddik, B.; Al-Eyadhy, A.; Alenezi, S.; Alamro, N.; Alhuzaimi, A.N.; Alhaboob, A.; Alhasan, K.; et al. COVID-19 vaccine uptake among healthcare workers in the fourth country to authorize BNT162b2 during the first month of rollout. medRxiv 2021. [Google Scholar] [CrossRef]
- Martin, C.A.; Marshall, C.; Patel, P.; Goss, C.; Jenkins, D.R.; Ellwood, C.; Barton, L.; Price, A.; Brunskill, N.J.; Khunti, K.; et al. Association of demographic and occupational factors with SARS-CoV-2 vaccine uptake in a multi-ethnic UK healthcare workforce: A rapid real-world analysis. medRxiv 2021. [Google Scholar] [CrossRef]
- Kim, D. Associations of Race/Ethnicity and Other Demographic and Socioeconomic Factors with Vaccination during the COVID-19 Pandemic in the United States. medRxiv 2021. [Google Scholar] [CrossRef]
- Pagotto, V.; Ferloni, A.; Soriano, M.M.; Díaz, M.; Golde, M.B.; González, M.I.; Asprea, V.; Staneloni, I.; Vidal, G.; Silveira, M.; et al. Active surveillance of the SPUTNIK V vaccine in health workers. medRxiv 2021. [Google Scholar] [CrossRef]
- Vasileiou, E.; Simpson, C.R.; Robertson, C.; Shi, T.; Kerr, S.; Agrawal, U.; Akbari, A.; Bedston, S.; Beggs, J.; Bradley, D.; et al. Effectiveness of First Dose of COVID-19 Vaccines Against Hospital Admissions in Scotland: National Prospective Cohort Study of 5.4 Million People. SSRN Electron. J. 2021. [Google Scholar] [CrossRef]
- Effectiveness of BNT162b2 mRNA vaccine against infection and COVID-19 vaccine coverage in healthcare workers in England, multicentre prospective cohort study (the SIREN study). Lancet Prepr. 2021. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3790399 (accessed on 2 March 2021).
- World Health Organisation. WHO SAGE Roadmap for Prioritizing Uses of COVID-19 Vaccines in the Context of Limited Supply; World Health Organisation: Geneva, Switzerland, 2021. [Google Scholar]
- Borbone, N.; Piccialli, G.; Roviello, G.N.; Oliviero, G. Nucleoside analogs and nucleoside precursors as drugs in the fight against SARS-CoV-2 and other coronaviruses. Molecules 2021, 26, 986. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Yin, X.; Meng, X.; Chan, J.; Ye, Z.; Riva, L.; Pache, L.; Chan, C.C.-Y.; Lai, P.-M.; Chan, C.C.-S.; et al. Clofazimine broadly inhibits coronaviruses including SARS-CoV-2. Nature 2021. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasan, T.; Beardsley, J.; Marais, B.J.; Nguyen, T.A.; Fox, G.J. The Implementation of Mass-Vaccination against SARS-CoV-2: A Systematic Review of Existing Strategies and Guidelines. Vaccines 2021, 9, 326. https://doi.org/10.3390/vaccines9040326
Hasan T, Beardsley J, Marais BJ, Nguyen TA, Fox GJ. The Implementation of Mass-Vaccination against SARS-CoV-2: A Systematic Review of Existing Strategies and Guidelines. Vaccines. 2021; 9(4):326. https://doi.org/10.3390/vaccines9040326
Chicago/Turabian StyleHasan, Tasnim, Justin Beardsley, Ben J. Marais, Thu Anh Nguyen, and Greg J. Fox. 2021. "The Implementation of Mass-Vaccination against SARS-CoV-2: A Systematic Review of Existing Strategies and Guidelines" Vaccines 9, no. 4: 326. https://doi.org/10.3390/vaccines9040326
APA StyleHasan, T., Beardsley, J., Marais, B. J., Nguyen, T. A., & Fox, G. J. (2021). The Implementation of Mass-Vaccination against SARS-CoV-2: A Systematic Review of Existing Strategies and Guidelines. Vaccines, 9(4), 326. https://doi.org/10.3390/vaccines9040326







