Humoral Immunity to Varicella Zoster Virus in Patients with Systemic Lupus Erythematosus and Rheumatoid Arthritis Compared to Healthy Controls
Abstract
1. Introduction
2. Methods and Study Design
Biostatistical Analysis
3. Results
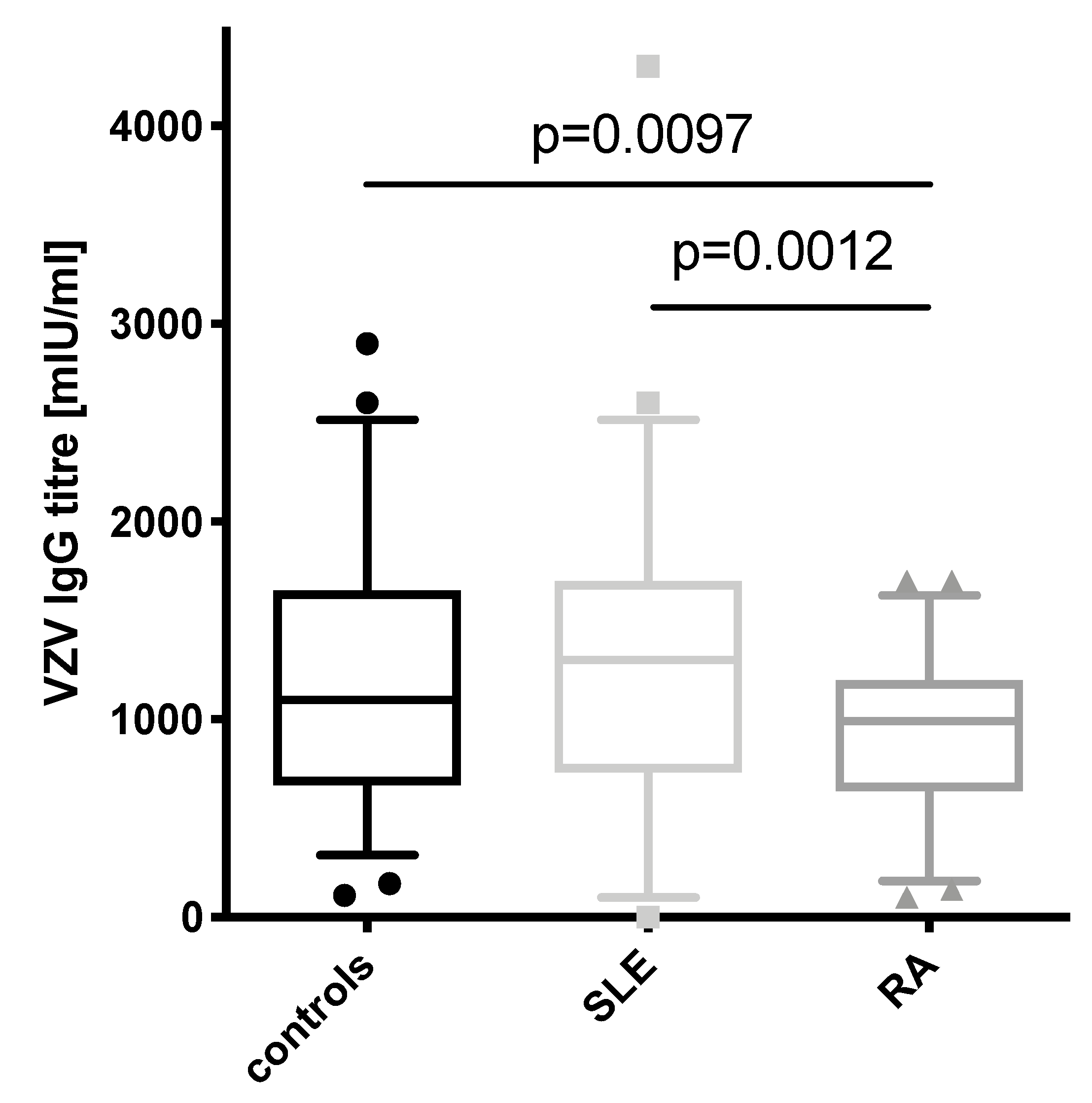
3.1. VZV IgG Concentrations in SLE Patients Are Comparable to Healthy Controls but Higher Than in RA Patients
3.2. Young SLE Patients Show the Highest VZV Titers
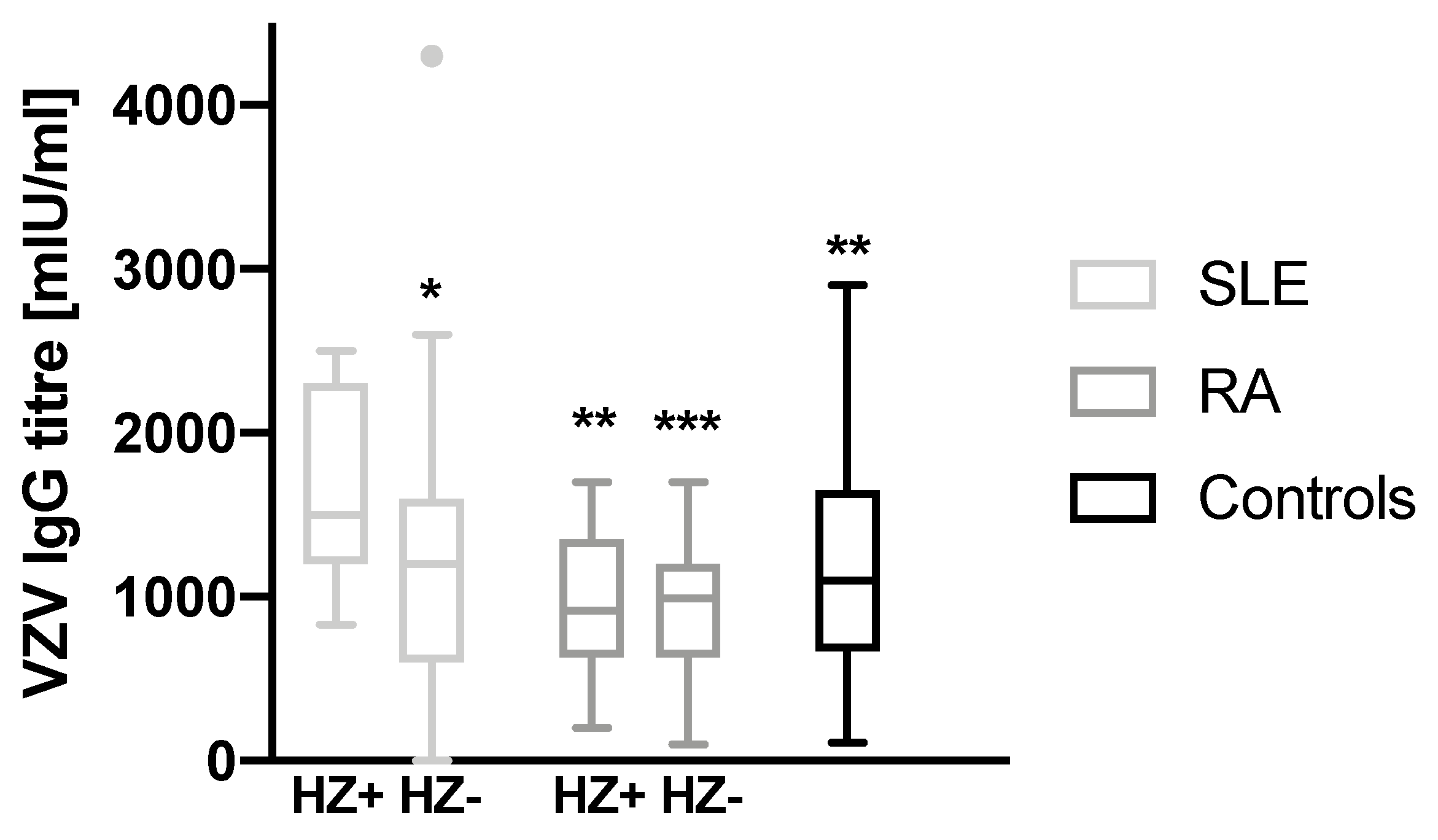
3.3. History of Herpes Zoster in SLE Patients Is Associated with Higher Antibody Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thomas, S.L.; Hall, A.J. What Does Epidemiology Tell Us About Risk Factors for Herpes Zoster? Lancet Infect. Dis. 2004, 4, 26–33. [Google Scholar] [CrossRef]
- Tseng, H.F.; Bruxvoort, K.; Ackerson, B.; Luo, Y.; Tanenbaum, H.; Tian, Y.; Zheng, C.; Cheung, B.; Patterson, B.J.; Van Oorschot, D.; et al. The epidemiology of herpes zoster in immunocompetent, unvaccinated adults >/= 50 years old: Incidence, complications, hospitalization, mortality, and recurrence. J. Infect. Dis. 2020, 222, 798–806. [Google Scholar] [CrossRef]
- Oxman, M.N.; Levin, M.J.; Johnson, G.R.; Schmader, K.E.; Straus, S.E.; Gelb, L.D.; Arbeit, R.D.; Simberkoff, M.S.; Gershon, A.A.; Davis, L.E.; et al. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N. Engl. J. Med. 2005, 352, 2271–2284. [Google Scholar] [CrossRef]
- Forbes, H.J.; Bhaskaran, K.; Thomas, S.L.; Smeeth, L.; Clayton, T.; Langan, S.M. Quantification of risk factors for herpes zoster: Population based case-control study. Br. Med. J. 2014, 348, g2911. [Google Scholar] [CrossRef]
- Veetil, B.M.A.; Myasoedova, E.; Matteson, E.L.; Gabriel, S.E.; Green, A.B.; Crowson, C.S. Incidence and time trends of herpes zoster in rheumatoid arthritis: A population-based cohort study. Arthritis Care Res. 2013, 65, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Hata, A.; Kuniyoshi, M.; Ohkusa, Y. Risk of herpes zoster in patients with underlying diseases: A retrospective hospital-based cohort study. Infection 2011, 39, 537–544. [Google Scholar] [CrossRef]
- Rondaan, C.; De Haan, A.; Horst, G.; Hempel, J.C.; Van Leer, C.; Bos, N.A.; Van Assen, S.; Bijl, M.; Westra, J. Altered cellular and humoral immunity to varicella-zoster virus in patients with autoimmune diseases. Arthritis Rheumatol. 2014, 66, 3122–3128. [Google Scholar] [CrossRef]
- Tan, E.M.; Cohen, A.S.; Fries, J.F.; Masi, A.T.; McShane, D.J.; Rothfield, N.F.; Schaller, J.G.; Talal, N.; Winchester, R.J. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheumatol. 1982, 25, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D. 2010 rheumatoid arthritis classification criteria: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Ann. Rheum. Dis. 2010, 69, 1580–1588. [Google Scholar] [CrossRef] [PubMed]
- Smitten, A.L.; Choi, H.K.; Hochberg, M.C.; Suissa, S.; Simon, T.A.; Testa, M.A.; Chan, K.A. The risk of herpes zoster in patients with rheumatoid arthritis in the United States and the United Kingdom. Arthritis Rheumatol. 2007, 57, 1431–1438. [Google Scholar] [CrossRef]
- Kawai, K.; Yawn, B.P. Risk factors for herpes zoster: A systematic review and meta-analysis. Mayo Clin. Proc. 2017, 92, 1806–1821. [Google Scholar] [CrossRef]
- Pappas, D.A.; Hooper, M.M.; Kremer, J.M.; Reed, G.; Shan, Y.; Wenkert, D.; Greenberg, J.D.; Curtis, J.R. Herpes zoster reactivation in patients with rheumatoid arthritis: Analysis of disease characteristics and disease-modifying antirheumatic drugs. Arthritis Care Res. 2015, 67, 1671–1678. [Google Scholar] [CrossRef] [PubMed]
- Liao, T.-L.; Chen, Y.-M.; Liu, H.-J.; Chen, D.-Y. Risk and severity of herpes zoster in patients with rheumatoid arthritis receiving different immunosuppressive medications: A case-control study in Asia. Br. Med. J. Open 2017, 7, e014032. [Google Scholar] [CrossRef] [PubMed]
- Bechman, K.; Subesinghe, S.; Norton, S.; Atzeni, F.; Galli, M.; Cope, A.P.; Winthrop, K.L.; Galloway, J.B. A Systematic review and meta-analysis of infection risk with small molecule jak inhibitors in rheumatoid arthritis. Rheumatology 2019, 58, 1755–1766. [Google Scholar] [CrossRef] [PubMed]
- Firestein, G.; Budd, R.; Sherine, E.G.; Iain, B.M.; O′Dell, J. Firestein & Kelley′s Textbook of Rheumatology-E-Book; Elsevier: London, UK, 2020. [Google Scholar]
- Blaese, R.; Grayson, J.; Steinberg, A.D. Increased immunoglobulin-secreting cells in the blood of patients with active systemic lupus erythematosus. Am. J. Med. 1980, 69, 345–3450. [Google Scholar] [CrossRef]
- Krasselt, M.; Baerwald, C. Sex, symptom severity, and quality of life in rheumatology. Clin. Rev. Allergy Immunol. 2017, 26, 1214–1221. [Google Scholar] [CrossRef]
- Levin, M.J.; Oxman, M.N.; Zhang, J.H.; Johnson, G.R.; Stanley, H.; Hayward, A.R.; Caulfield, M.J.; Irwin, M.R.; Smith, J.G.; Clair, J.; et al. Varicella-zoster virus-specific immune responses in elderly recipients of a herpes zoster vaccine. J. Infect. Dis. 2008, 197, 825–835. [Google Scholar] [CrossRef]
- Levin, M.J.; Smith, J.G.; Kaufhold, R.M.; Barber, D.; Hayward, A.R.; Chan, C.Y.; Chan, I.S.; Li, D.J.; Wang, W.; Keller, P.M.A. Decline in varicella-zoster virus (Vzv)-Specific cell-mediated immunity with increasing age and boosting with a high-dose Vzv vaccine. J. Infect. Dis. 2003, 188, 1336–1344. [Google Scholar] [CrossRef]
- Rondaan, C.; Van Leer, C.C.; Van Assen, S.; Bootsma, H.; De Leeuw, K.; Arends, S.; Bos, N.A.; Westra, J. Longitudinal analysis of varicella-zoster virus-specific antibodies in systemic lupus erythematosus: No association with subclinical viral reactivations or lupus disease activity. Lupus 2018, 27, 1271–1278. [Google Scholar] [CrossRef]
- Nagasawa, K.; Yamauchi, Y.; Tada, Y.; Kusaba, T.; Niho, Y.; Yoshikawa, H. High incidence of herpes zoster in patients with systemic Lupus erythematosus: An immunological analysis. Ann. Rheumatol. Dis. 1990, 49, 630–633. [Google Scholar] [CrossRef]
- Fauci, A.S.; Moutsopoulos, H.M. Polyclonally yriggered B cells in the peripheral blood and bone marrow of normal individuals and in patients with systemic Lupus erythematosus and primary Sjogren’s syndrome. Arthritis Rheumatol. 1981, 24, 577–5783. [Google Scholar] [CrossRef]
- Cuadrado, M.J.; Calatayud, I.; Urquizu-Padilla, M.; Wijetilleka, S.; Kiani-Alikhan, S.; Karim, M.Y. Immunoglobulin abnormalities are frequent in patients with Lupus nephritis. BMC Rheumatol. 2019, 3, 30. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.B.; Rönnelid, J.; Gunnarsson, I.; Karlsson, G.; Berg, L.; Lundberg, I. Increased serum levels of immunoglobulins, C-reactive protein, type 1 and type 2 cytokines in patients with mixed connective tissue disease. J. Autoimmun. 1998, 11, 503–508. [Google Scholar] [CrossRef]
- Kay, R.A.; Wood, K.J.; Bernstein, R.M.; Holt, P.J.; Pumphrey, R.S. An igg subclass imbalance in connective tissue disease. Ann. Rheumatol. Dis. 1988, 47, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Draborg, A.H.; Duus, K.; Houen, G. Epstein-barr virus and systemic Lupus erythematosus. Clin. Dev. Immunol. 2012, 2012, 370516. [Google Scholar] [CrossRef]
- Kelly, C.; Sykes, H. Rheumatoid arthritis, malignancy, and paraproteins. Ann. Rheum. Dis. 1990, 49, 657–659. [Google Scholar] [CrossRef]
- Scarsi, M.; Paolini, L.; Ricotta, D.; Pedrini, A.; Piantoni, S.; Caimi, L.; Tincani, A.; Airò, P. Abatacept reduces levels of switched memory B cells, autoantibodies, and immunoglobulins in patients with rheumatoid arthritis. J. Rheumatol. 2014, 41, 666–672. [Google Scholar] [CrossRef]
- Krasselt, M.; Baerwald, C.; Liebert, U.G.; Seifert, O. Humoral immunity to varicella zoster virus is altered in patients with rheumatoid arthritis. Clin. Rheumatol. 2019, 38, 2493–2500. [Google Scholar] [CrossRef]
- Arvin, A.M. Humoral and cellular immunity to varicella-zoster virus: An overview. J. Infect. Dis. 2008, 197, S58–S60. [Google Scholar] [CrossRef]
- Harpaz, R. How little we know herpes zoster. J. Infect. Dis. 2020, 222, 708–711. [Google Scholar] [CrossRef]
- Kraaij, T.; Arends, E.J.; Van Dam, L.S.; Kamerling, S.W.A.; Van Daele, P.L.A.; Bredewold, O.W.; Ray, A.; Bakker, J.A.; Scherer, H.U.; Huizinga, T.J.W.; et al. Long-Term effects of combined B-cell immunomodulation with rituximab and belimumab in severe, refractory systemic Lupus erythematosus: 2-year results. Nephrol. Dial. Transpl. 2020. online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Reichelt, M.; Zerboni, L.; Arvin, A.M. Mechanisms of varicella-zoster virus neuropathogenesis in human dorsal root ganglia. J. Virol. 2008, 82, 3971–3983. [Google Scholar] [CrossRef] [PubMed]
- Korn, T.; Oukka, M. A baffling association between malaria resistance and the risk of multiple sclerosis. N. Engl. J. Med. 2017, 376, 1680–1681. [Google Scholar] [CrossRef]
- Weyand, C.M.; Fujii, H.; Shao, L.; Goronzy, J.J. Rejuvenating the immune system in rheumatoid arthritis. Nat. Rev. Rheumatol. 2009, 5, 583–588. [Google Scholar] [CrossRef]
- Asada, H. Vzv-specific cell-mediated immunity, but not humoral immunity, correlates inversely with the incidence of herpes zoster and the severity of skin symptoms and zoster-associated pain: The Shez study. Vaccine 2019, 37, 6776–6781. [Google Scholar] [CrossRef] [PubMed]
- Asada, H.; Nagayama, K.; Okazaki, A.; Mori, Y.; Okuno, Y.; Takao, Y.; Miyazaki, Y.; Onishi, F.; Okeda, M.; Yano, S.; et al. An inverse correlation of Vzv skin-test reaction, but not antibody, with severity of herpes zoster skin symptoms and zoster-associated pain. J. Derm. Sci. 2013, 69, 243–249. [Google Scholar] [CrossRef]
- Imoto, K.; Okazaki, A.; Onishi, F.; Miyazaki, Y.; Okeda, M.; Yano, S.; Takao, Y.; Gomi, Y.; Ishikawa, T.; Okuno, Y.; et al. Vzv skin-test reaction, but not antibody, is an important predictive factor for postherpetic neuralgia. J. Derm. Sci. 2015, 79, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P.B.; Gabriel, E.E.; Miao, X.; Li, X.; Su, S.-C.; Parrino, J.; Chan, I.S.F. Fold rise in antibody titers by measured by glycoprotein-based enzyme-linked immunosorbent assay is an excellent correlate of protection for a herpes zoster vaccine, demonstrated via the vaccine efficacy curve. J. Infect. Dis. 2014, 210, 1573–1581. [Google Scholar] [CrossRef]
- Mok, C.C. Herpes zoster vaccination in systemic lupus erythematosus: The current status. Hum. Vaccin. Immunother. 2019, 15, 45–48. [Google Scholar] [CrossRef]
- Krasselt, M.; Ivanov, J.-P.; Baerwald, C.; Seifert, O. Low vaccination rates among patients with rheumatoid arthritis in a german outpatient clinic. Rheumatol Int. 2017, 37, 229–237. [Google Scholar] [CrossRef]
- Krasselt, M.; Baerwald, C.; Seifert, O. Insufficient vaccination rates in patients with systemic Lupus erythematosus in a german outpatient clinic. Z. Rheumatol. 2018, 77, 727–734. [Google Scholar] [CrossRef]
- Schmedt, N.; Schiffner-Rohe, J.; Sprenger, R.; Walker, J.; Von Eiff, C.; Häckl, D. Pneumococcal vaccination rates in immunocompromised patients—A cohort study based on claims data from more than 200.000 patients in Germany. PLoS ONE 2019, 14, e0220848. [Google Scholar] [CrossRef] [PubMed]
- Chehab, G.; Richter, J.G.; Brinks, R.; Fischer-Betz, R.; Winkler-Rohlfing, B.; Schneider, M. Vaccination coverage in systemic Lupus erythematosus—A cross-sectional analysis of the German long-term study (Lula Cohort). Rheumatology 2018, 57, 1439–1447. [Google Scholar] [CrossRef]
- Furer, V.; Rondaan, C.; Heijstek, M.W.; Agmon-Levin, N.; Van Assen, S.; Bijl, M.; Breedveld, F.C.; D’Amelio, R.; Dougados, M.; Kapetanovic, M.C.; et al. 2019 update of eular recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Ann. Rheumatol. Dis. 2020, 79, 39–52. [Google Scholar] [CrossRef]
- Singh, J.A.; Furst, D.E.; Bharat, A.; Curtis, J.R.; Kavanaugh, A.F.; Kremer, J.M.; Moreland, L.W.; O’Dell, J.; Winthrop, K.L.; Beukelman, T.; et al. 2012 update of the 2008 American College of Rheumatology recommendations for the use of disease-modifying antirheumatic drugs and biologic agents in the treatment of rheumatoid arthritis. Arthritis Care Res. 2012, 64, 625–639. [Google Scholar] [CrossRef]
- RKI. StäNdige Impfkommission: Empfehlungen Der StäNdigen Impfkommission; Robert Koch-Institut: Berlin, Germany, 2020; pp. 1–68. [Google Scholar]
- Cunningham, A.L.; Lal, H.; Kovac, M.; Chlibek, R.; Hwang, S.-J.; Díez-Domingo, J.; Godeaux, O.; Levin, M.J.; McElhaney, J.E.; Puig-Barberà, J.; et al. Efficacy of the herpes zoster subunit vaccine in adults 70 years of age or older. N. Engl. J. Med. 2016, 375, 1019–1032. [Google Scholar] [CrossRef]
- Lal, H.; Cunningham, A.L.; Godeaux, O.; Chlibek, R.; Diez-Domingo, J.; Hwang, S.-J.; Levin, M.J.; McElhaney, J.E.; Poder, J.P.-B.A.; Puig-Barberà, J.; et al. Efficacy of an adjuvanted herpes zoster subunit vaccine in older adults. N. Engl. J. Med. 2015, 372, 2087–2096. [Google Scholar] [CrossRef]
- Berkowitz, E.M.; Moyle, G.; Stellbrink, H.J.; Schurmann, D.; Kegg, S.; Stoll, M.; El Idrissi, M.; Oostvogels, L.; Heineman, T.C.; Zoster-015 HZ/su Study Group. Safety and immunogenicity of an adjuvanted herpes zoster subunit candidate vaccine in hiv-infected adults: A phase 1/2a randomized, placebo-controlled study. J. Infect. Dis. 2015, 211, 1279–1287. [Google Scholar] [CrossRef]
- Bastidas, A.; de la Serna, J.; El Idrissi, M.; Oostvogels, L.; Quittet, P.; Lopez-Jimenez, J.; Vural, F.; Pohlreich, D.; Zuckerman, T.; Issa, N.C. Effect of recombinant zoster vaccine on incidence of herpes zoster after autologous stem cell transplantation: A randomized clinical trial. J. Am. Med. Assoc. 2019, 322, 123–133. [Google Scholar] [CrossRef]
- Stadtmauer, E.A.; Sullivan, K.M.; Marty, F.M.; Dadwal, S.S.; Papanicolaou, G.A.; Shea, T.C.; Mossad, S.B.; Andreadis, C.; Young, J.A.; Buadi, F.K. A phase 1/2 study of an adjuvanted varicella-zoster virus subunit vaccine in autologous hematopoietic cell transplant recipients. Blood 2014, 124, 2921–2929. [Google Scholar] [CrossRef]
- Vink, P. Immunogenicity and safety of a candidate subunit adjuvanted herpes zoster vaccine (Hz/Su) in adults post renal transplant: A phase iii randomized clinical trial. Open Forum Infect. Dis. 2017, 4, S417–S418. [Google Scholar] [CrossRef][Green Version]
- Wagner, N.; Assmus, F.; Arendt, G.; Baum, E.; Baumann, U.; Bogdan, C.; Burchard, G.; Foll, D.; Garbe, E.; Hecht, J.; et al. Impfen bei Immundefizienz. Bundesgesundheitsbl. 2019, 62, 494–515. [Google Scholar] [CrossRef] [PubMed]
- Mok, C.C.; Chan, K.H.; Ho, L.Y.; Fung, Y.F.; Fung, W.F.; Woo, P.C.Y. Safety and immune response of a live-attenuated herpes zoster vaccine in patients with systemic lupus erythematosus: A randomised placebo-controlled trial. Ann. Rheumatol. Dis. 2019, 78, 1663–1668. [Google Scholar] [CrossRef]
- Zhang, J.; Xie, F.; Delzell, E.; Chen, L.; Winthrop, K.L.; Lewis, J.D.; Saag, K.G.; Baddley, J.W.; Curtis, J.R. Association between vaccination for herpes zoster and risk of herpes zoster infection among older patients with selected immune-mediated diseases. J. Am. Med. Assoc. 2012, 308, 43–49. [Google Scholar] [CrossRef]
- Zhang, J.; Delzell, E.; Xie, F.; Baddley, J.W.; Spettell, C.; Mcmahan, R.M.; Fernandes, J.; Chen, L.; Winthrop, K.; Curtis, J.R. The use, safety, and effectiveness of herpes zoster vaccination in individuals with inflammatory and autoimmune diseases: A longitudinal observational study. Arthritis Res. Ther. 2011, 13, R174. [Google Scholar] [CrossRef]
- Tavares-Da-Silva, F.; Co, M.M.; Dessart, C.; Hervé, C.; López-Fauqued, M.; Mahaux, O.; Van Holle, L.; Stegmann, J.-U. Review of the initial post-marketing safety surveillance for the recombinant zoster vaccine. Vaccine 2020, 38, 3489–3500. [Google Scholar] [CrossRef]
- Dagnew, A.F.; Rausch, D.; Hervé, C.; Zahaf, T.; Levin, M.J.; Schuind, A. Efficacy and serious adverse events profile of the adjuvanted recombinant zoster vaccine in adults with pre-existing potential immune-mediated diseases: A pooled post hoc analysis on two parallel randomized trials. Rheumatology 2021, 60, 1226–1233. [Google Scholar] [CrossRef]
- Acharya, S.; Raza, S.; Pattanaik, D.; Howard, A. Safety of adjuvanted herpes zoster subunit vaccine (Hz/Su, Shingrix) among patients with autoimmune inflammatory diseases. In Arthritis & Rheumatology; Wiley: Hoboken, NJ, USA, 2019; Volume 71. [Google Scholar]
- Källmark, H.; Gullstrand, B.; Nagel, J.; Einarsson, J.; Jönsson, G.; Kahn, F.; Kahn, R.; Bangtsson, A.; Bergström, T.; Kapetanovic, M. Immunogenicity of adjuvanted herpes zoster subunit vaccine in rheumatoid arthritis patients treated with janus kinase inhibitors and controls: Preliminary results. In Arthritis & Rheumatology; Wiley: Hoboken, NJ, USA, 2020; Volume 72. [Google Scholar]
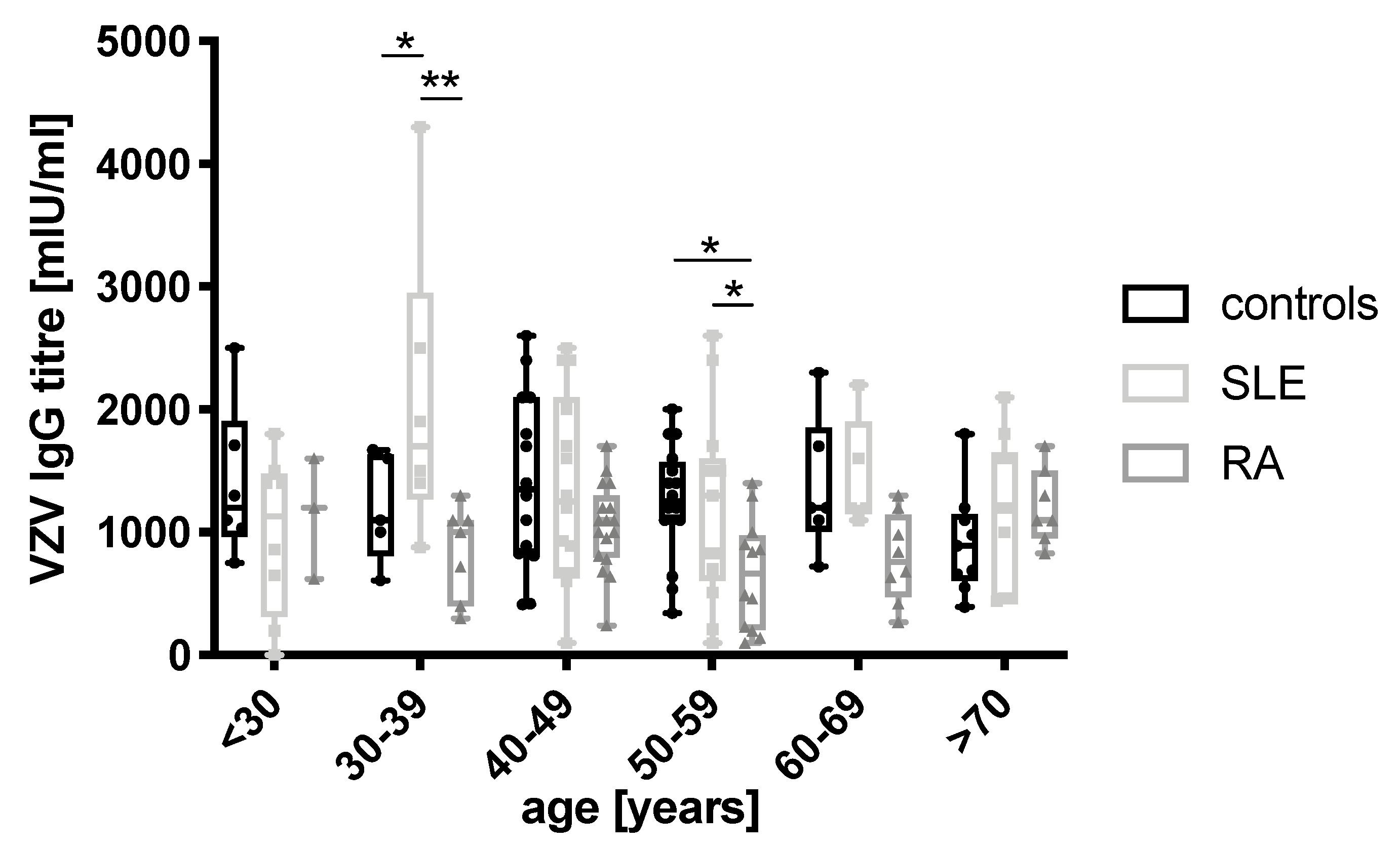
| SLE Patients N = 56 | RA Patients N = 54 | Controls N = 56 | |
|---|---|---|---|
| Mean age, years | 49.8 ± 15.9 | 51.3 ± 13.4 | 51.9 ± 14.8 |
| Female, n (%) | 54 (96.4) | 47 (87) | 53 (94.6) |
| Subjects with positive HZ history, n (%) | 17 (30.4) | 14 (25.9) | n.a. |
| Mean age at HZ, years | 33.5 ± 12.4 | 36.7 ± 12.4 | n.a. |
| Mean time from disease onset to HZ, years | 6.8 ± 5.9 | 15.2 ± 10.2 | n.a. |
| Characteristics | SLE | RA |
|---|---|---|
| Medication, n (%) | ||
| All immunosuppressives | 38 (67.9) | 50 (92.6) |
| Prednisolone | 44 (78.6) | 44 (81.5) |
| Hydroxychloroquine | 13 (23.2) | 0 (0) |
| Azathioprine | 18 (32.1) | 1 (1.9) |
| Mycofenolat-mofetil | 17 (30.4) | 0 (0) |
| Cyclophosphamide | 1 (1.8) | 0 (0) |
| Methotrexate | 1 (1.8) | 33 (61.1) |
| Leflunomide | 0 (0) | 5 (9.3) |
| Sulfasalazine | 0 (0) | 1 (1.9) |
| bDMARDs, n (%) | ||
| Adalimumab | 0 (0) | 12 (22.2) |
| Etanercept | 0 (0) | 6 (11.1) |
| Abatacept | 0 (0) | 4 (7.4) |
| Rituximab | 1 (1.8) | 2 (3.7) |
| Certolizumab | 0 (0) | 2 (3.7) |
| tsDMARDs, n (%) | ||
| Tofacitinib | 0 (0) | 2 (3.7) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krasselt, M.; Baerwald, C.; Liebert, U.G.; Seifert, O. Humoral Immunity to Varicella Zoster Virus in Patients with Systemic Lupus Erythematosus and Rheumatoid Arthritis Compared to Healthy Controls. Vaccines 2021, 9, 325. https://doi.org/10.3390/vaccines9040325
Krasselt M, Baerwald C, Liebert UG, Seifert O. Humoral Immunity to Varicella Zoster Virus in Patients with Systemic Lupus Erythematosus and Rheumatoid Arthritis Compared to Healthy Controls. Vaccines. 2021; 9(4):325. https://doi.org/10.3390/vaccines9040325
Chicago/Turabian StyleKrasselt, Marco, Christoph Baerwald, Uwe G. Liebert, and Olga Seifert. 2021. "Humoral Immunity to Varicella Zoster Virus in Patients with Systemic Lupus Erythematosus and Rheumatoid Arthritis Compared to Healthy Controls" Vaccines 9, no. 4: 325. https://doi.org/10.3390/vaccines9040325
APA StyleKrasselt, M., Baerwald, C., Liebert, U. G., & Seifert, O. (2021). Humoral Immunity to Varicella Zoster Virus in Patients with Systemic Lupus Erythematosus and Rheumatoid Arthritis Compared to Healthy Controls. Vaccines, 9(4), 325. https://doi.org/10.3390/vaccines9040325






