Generation and Characterization of Universal Live-Attenuated Influenza Vaccine Candidates Containing Multiple M2e Epitopes
Abstract
1. Introduction
2. Materials and Methods
2.1. Viruses, Cells, Proteins, Antibodies
2.2. Generation of Recombinant LAIV Viruses
2.3. In Vitro Studies
2.4. Replication of LAIV Viruses in Mouse Respiratory Tract
2.5. Direct Protection against Heterologous and Heterosubtypic Challenge
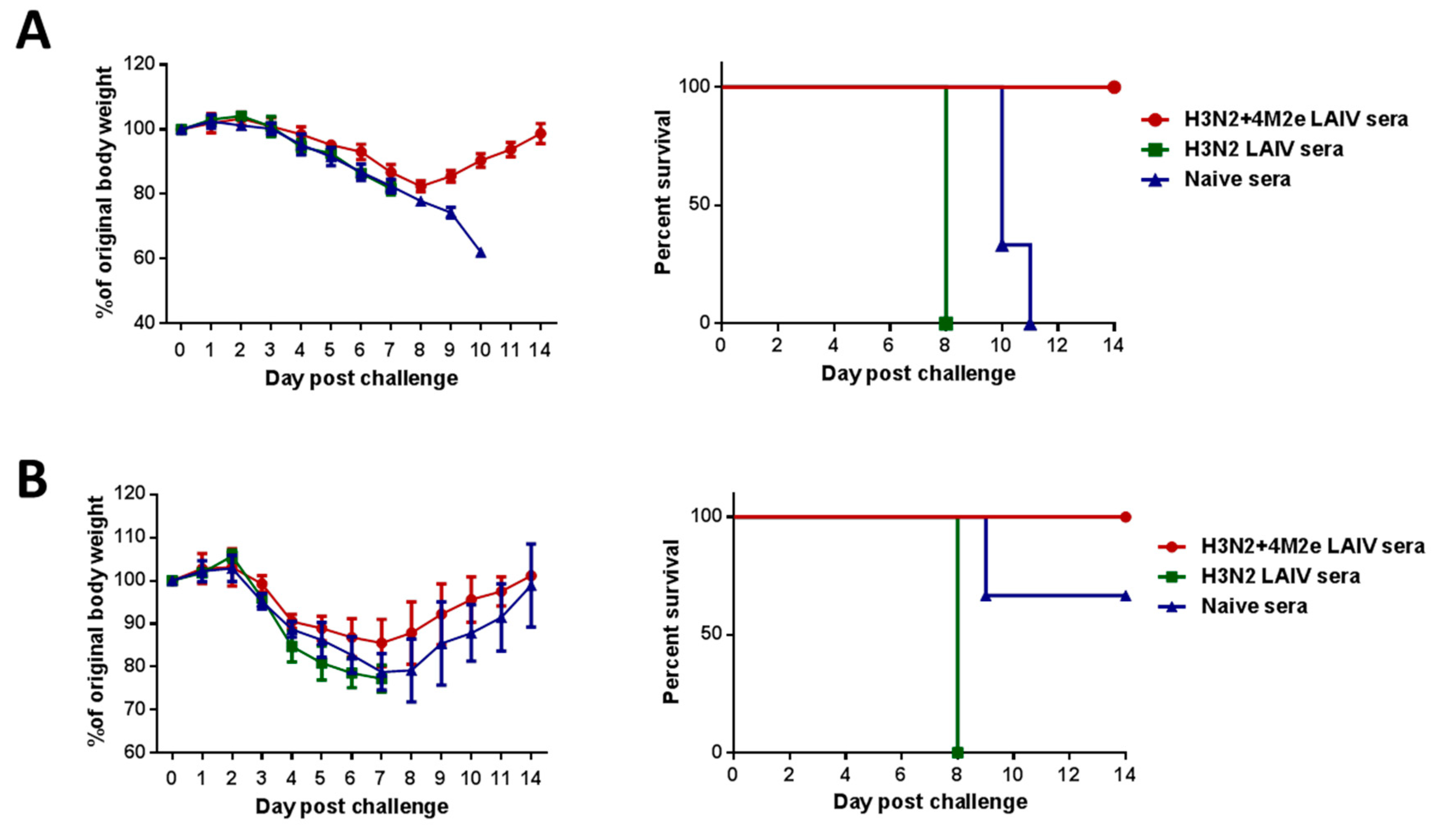
2.6. Indirect Protection (In Vivo Protection Assay of Immune Sera)
2.7. Comparing Immunogenicity and Cross-Protection of Homologous and Heterologous Prime-Boost Vaccination Regimens
2.8. Hemagglutination Inhibition (HAI) Assay
2.9. Ethical Statement
2.10. Statistical Analysis
3. Results
3.1. Expression of M2e Epitopes by LAIV+4M2e Chimeric Vaccines
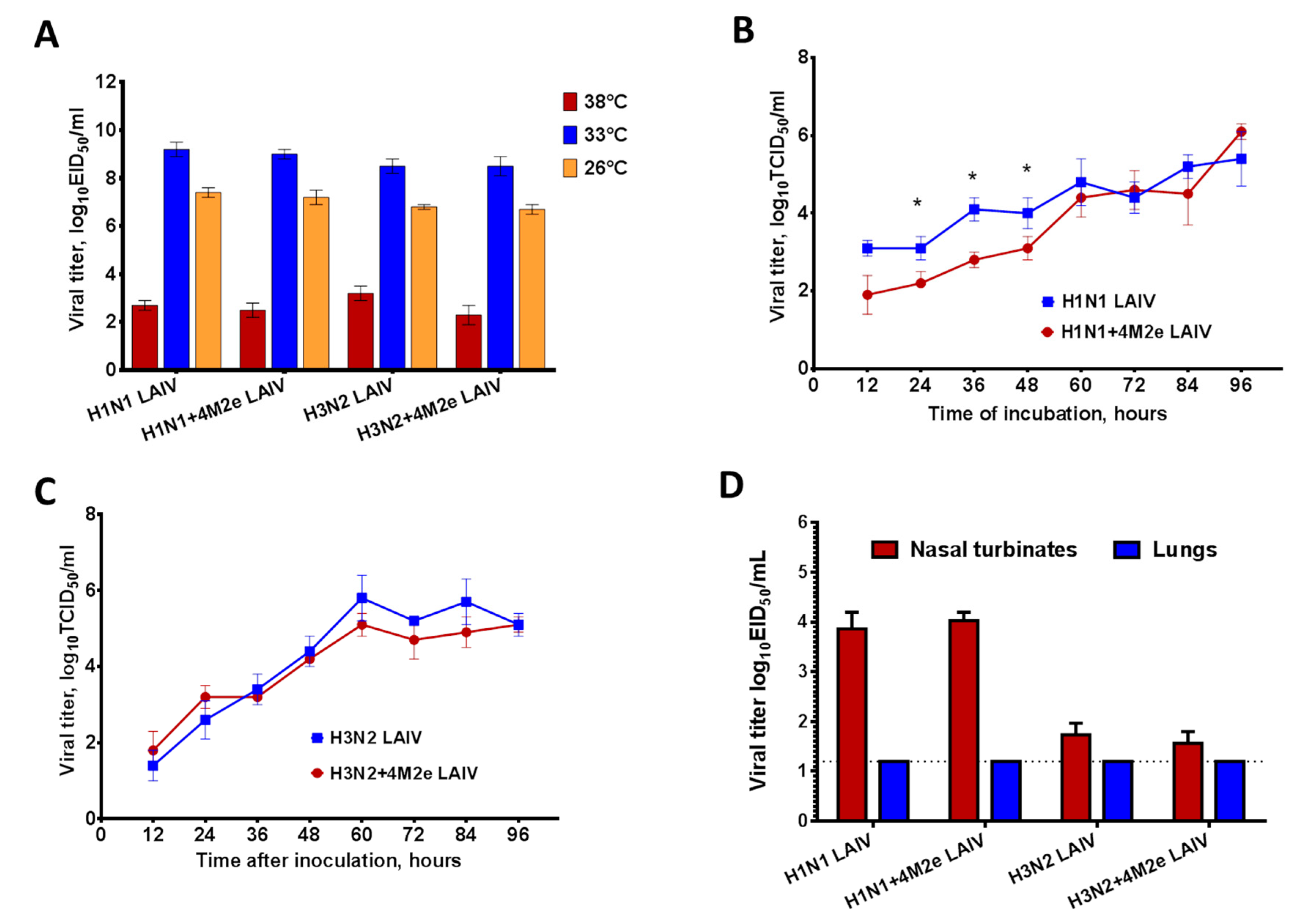
3.2. Replication of Chimeric LAIV+4M2e and Their Classical LAIV Counterparts In Vitro and In Vivo
3.3. Immunogenicity of the Recombinant LAIV+4M2e Viruses
3.4. Protection against Heterologous and Heterosubtypic Viruses
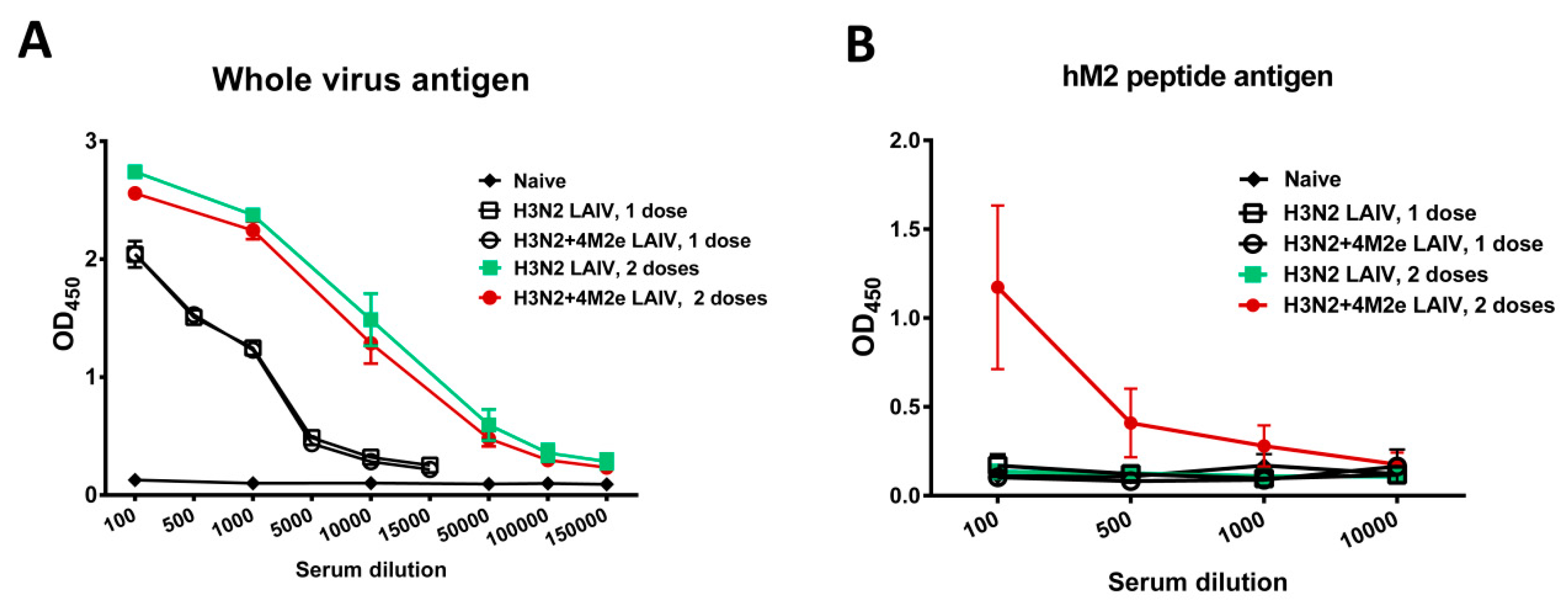
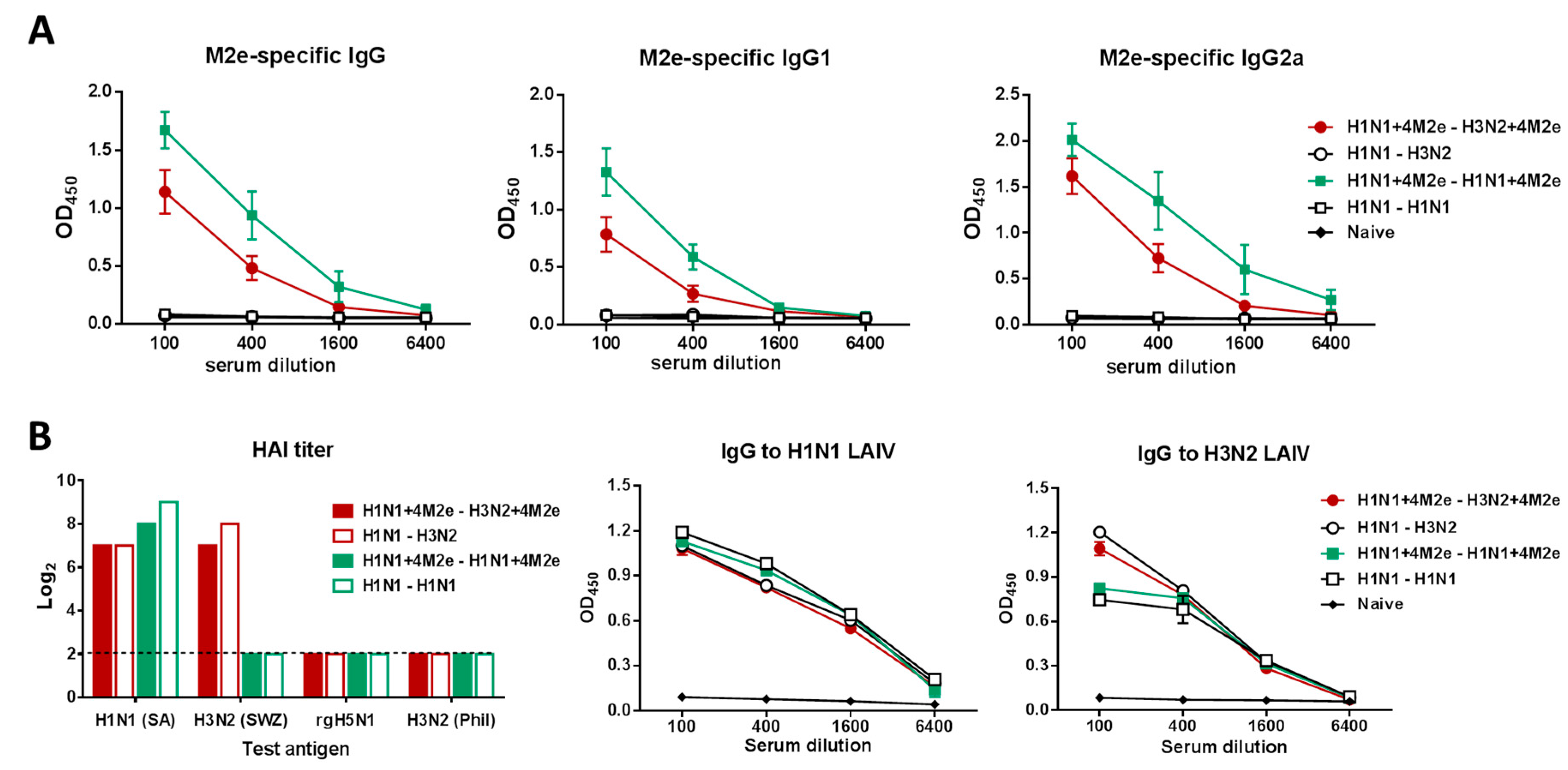
3.5. Assessment of Antibody Immune Responses After Homologous and Heterologous Prime-Boost Immunization with LAIV+4M2e Universal Vaccine Candidates
3.6. Long-Term Cross-Protection Afforded by Homologous and Heterologous Prime-Boost Immunization with LAIV+4M2e Vaccine Candidates
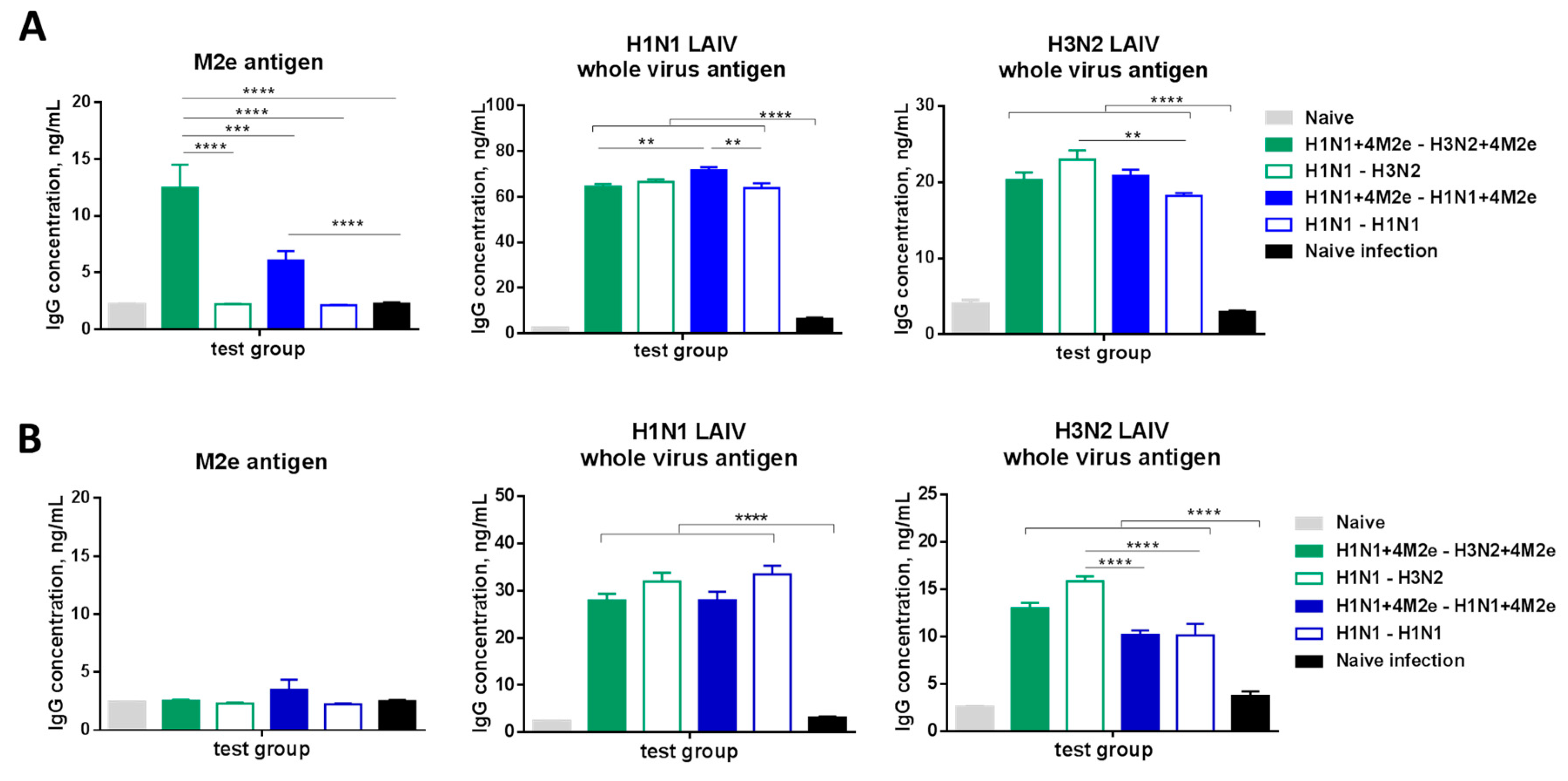
3.7. Antibody Immune Responses in Immunized Mice after Challenge with Heterologous Influenza Virus
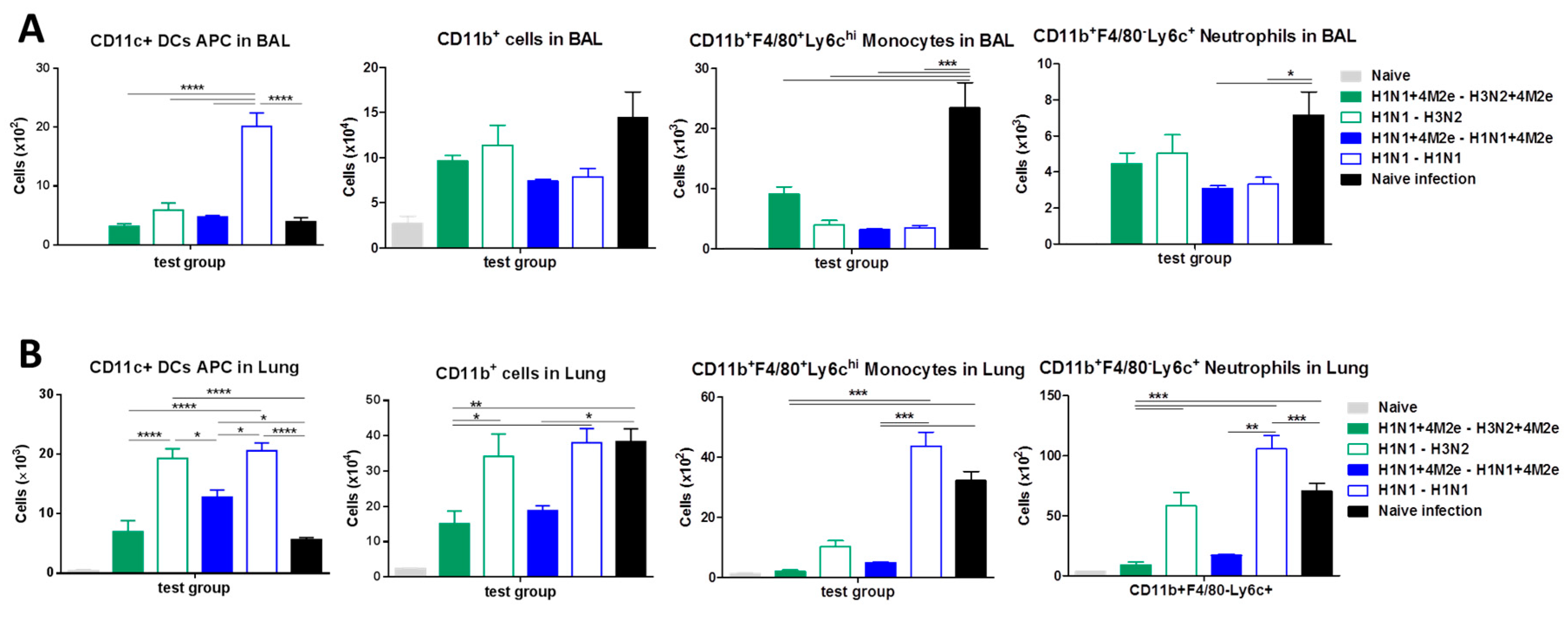
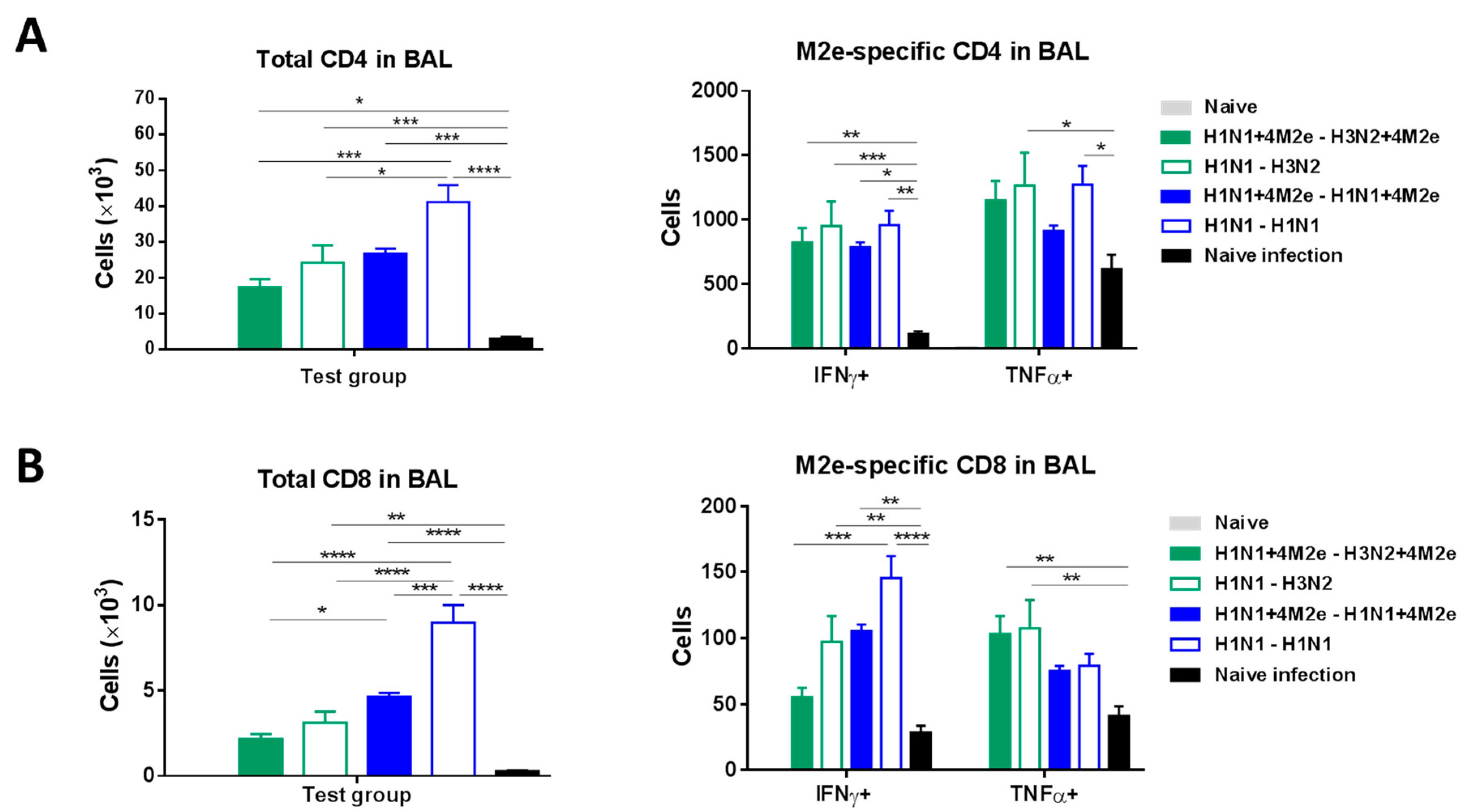
3.8. Potential Contribution of Innate Immune Cells and T-Cell Responses to Protection in Immunized Mice after Challenge with Heterologous Influenza Virus
4. Discussions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Iuliano, A.D.; Roguski, K.M.; Chang, H.H.; Muscatello, D.J.; Palekar, R.; Tempia, S.; Cohen, C.; Gran, J.M.; Schanzer, D.; Cowling, B.J.; et al. Estimates of Global Seasonal Influenza-Associated Respiratory Mortality: A Modelling Study. Lancet 2018, 391, 1285–1300. [Google Scholar] [CrossRef]
- Webster, R.G.; Govorkova, E.A. Continuing Challenges in Influenza. Ann. N. Y. Acad. Sci. 2014, 1323, 115–139. [Google Scholar] [CrossRef]
- Sautto, G.A.; Kirchenbaum, G.A.; Ross, T.M. Towards a Universal Influenza Vaccine: Different Approaches for One Goal. Virol. J. 2018, 15, 1–12. [Google Scholar] [CrossRef]
- Epstein, S.L. Universal Influenza Vaccines: Progress in Achieving Broad Cross-Protection In Vivo. Am. J. Epidemiol. 2018, 187, 2603–2614. [Google Scholar] [CrossRef]
- Scorza, F.B.; Tsvetnitsky, V.; Donnelly, J.J. Universal Influenza Vaccines: Shifting to Better Vaccines. Vaccine 2016, 34, 2926–2933. [Google Scholar] [CrossRef] [PubMed]
- Black, R.A.; Rota, P.A.; Gorodkova, N.; Klenk, H.-D.; Kendal, A.P. Antibody Response to the M2 Protein of Influenza a Virus Expressed in Insect Cells. J. Gen. Virol. 1993, 74, 143–146. [Google Scholar] [CrossRef]
- Mezhenskaya, D.; Isakova-Sivak, I.; Rudenko, L. M2e- Based Universal Influenza Vaccines: A Historical Overview and New Approaches to Development. J. Biomed. Sci. 2019, 26, 1–15. [Google Scholar] [CrossRef]
- Feng, J.; Zhang, M.; Mozdzanowska, K.; Zharikova, D.; Hoff, H.; Wunner, W.; Couch, R.B.; Gerhard, W. Influenza A Virus Infection Engenders a Poor Antibody Response Against the Ectodomain of Matrix Protein 2. Virol. J. 2006, 3, 102. [Google Scholar] [CrossRef]
- Hutchinson, E.C.; Charles, P.D.; Hester, S.S.; Thomas, B.; Trudgian, D.C.; Martínez-Alonso, M.; Fodor, E.J.B. Conserved and Host-Specific Features of Influenza Virion Architecture. Nat. Commun. 2014, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zebedee, S.L.; Lamb, R.A. Influenza A Virus M2 Protein: Monoclonal Antibody Restriction of Virus Growth and Detection of M2 in Virions. J. Virol. 1988, 62, 2762–2772. [Google Scholar] [CrossRef]
- Zhong, W.; Reed, C.; Blair, P.J.; Katz, J.M.; Hancock, K.; Aranio, D.; Bai, Y.; Browning, P.; Branch, A.; Steward-Clark, E.; et al. Serum Antibody Response to Matrix Protein 2 Following Natural Infection with 2009 Pandemic Influenza A(H1N1) Virus in Humans. J. Infect. Dis. 2013, 209, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-C.; Lee, J.-S.; Kwon, Y.-M.; O, E.; Lee, Y.-J.; Choi, J.-G.; Wang, B.-Z.; Compans, R.W.; Kang, S.-M. Multiple Heterologous M2 Extracellular Domains Presented on Virus-Like Particles Confer Broader and Stronger M2 Immunity Than Live Influenza A Virus Infection. Antivir. Res. 2013, 99, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-C.; Song, J.-M.; Eunju, O.; Kwon, Y.-M.; Lee, Y.-J.; Compans, R.W.; Kang, S.-M. Virus-like Particles Containing Multiple M2 Extracellular Domains Confer Improved Cross-protection Against Various Subtypes of Influenza Virus. Mol. Ther. 2013, 21, 485–492. [Google Scholar] [CrossRef]
- Kolpe, A.; Schepens, B.; Fiers, W.; Saelens, X. M2- Based Influenza Vaccines: Recent Advances and Clinical Potential. Expert Rev. Vaccines 2016, 16, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Kwon, Y.-M.; Lee, Y.-T.; Kim, M.-C.; Hwang, H.S.; Ko, E.-J.; Lee, Y.; Montemagno, C.; Kang, S.-M. Virus-Like Particles Are a Superior Platform for Presenting M2e Epitopes to Prime Humoral and Cellular Immunity against Influenza Virus. Vaccines 2018, 6, 66. [Google Scholar] [CrossRef]
- Kirsteina, A.; Akopjana, I.; Bogans, J.; Lieknina, I.; Jansons, J.; Skrastina, D.; Kazaka, T.; Tars, K.; Isakova-Sivak, I.; Mezhenskaya, D.; et al. Construction and Immunogenicity of a Novel Multivalent Vaccine Prototype Based on Conserved Influenza Virus Antigens. Vaccines 2020, 8, 197. [Google Scholar] [CrossRef]
- Mohn, K.G.-I.; Smith, I.; Sjursen, H.; Cox, R.J. Immune Responses After Live Attenuated Influenza Vaccination. Hum. Vaccines Immunother. 2018, 14, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Korenkov, D.; Isakova-Sivak, I.; Rudenko, L. Basics of CD8 T-cell immune responses after influenza infection and vaccination with inactivated or live attenuated influenza vaccine. Expert Rev. Vaccines 2018, 17, 977–987. [Google Scholar] [CrossRef]
- Isakova-Sivak, I.; Matyushenko, V.; Kotomina, T.; Kiseleva, I.V.; Krutikova, E.; Donina, S.A.; Rekstin, A.; Larionova, N.; Mezhenskaya, D.; Sivak, K.V.; et al. Sequential Immunization with Universal Live Attenuated Influenza Vaccine Candidates Protects Ferrets against a High-Dose Heterologous Virus Challenge. Vaccines 2019, 7, 61. [Google Scholar] [CrossRef]
- Liu, W.-C.; Nachbagauer, R.; Stadlbauer, D.; Solórzano, A.; Berlanda-Scorza, F.; García-Sastre, A.; Palese, P.; Krammer, F.; Albrecht, R.A. Sequential Immunization With Live-Attenuated Chimeric Hemagglutinin-Based Vaccines Confers Heterosubtypic Immunity Against Influenza A Viruses in a Preclinical Ferret Model. Front. Immunol. 2019, 10, 756. [Google Scholar] [CrossRef]
- Nachbagauer, R.; Krammer, F.; Albrecht, R.A. A Live-Attenuated Prime, Inactivated Boost Vaccination Strategy with Chimeric Hemagglutinin-Based Universal Influenza Virus Vaccines Provides Protection in Ferrets: A Confirmatory Study. Vaccines 2018, 6, 47. [Google Scholar] [CrossRef]
- Kim, M.-C.; Lee, Y.-N.; Kim, Y.-J.; Choi, H.-J.; Kim, K.-H.; Lee, Y.-J.; Kang, S.-M. Immunogenicity and Efficacy of Replication-Competent Recombinant Influenza Virus Carrying Multimeric M2 Extracellular Domains in a Chimeric Hemagglutinin Conjugate. Antivir. Res. 2017, 148, 43–52. [Google Scholar] [CrossRef]
- Adler-Moore, J.P.; Ernst, W.; Kim, H.; Ward, N.; Chiang, S.M.; Do, T.; Fujii, G. Monomeric M2e Antigen in VesiVax® Liposomes Stimulates Protection Against Type a Strains of Influenza Comparable to Liposomes With Multimeric Forms of M2e. J. Liposome Res. 2017, 27, 210–220. [Google Scholar] [CrossRef]
- Holsinger, L.J.; Alams, R. Influenza Virus M2 Integral Membrane Protein Is a Homotetramer Stabilized by Formation of Disulfide Bonds. Virology 1991, 183, 32–43. [Google Scholar] [CrossRef]
- Isakova-Sivak, I.; Chen, L.-M.; Matsuoka, Y.; Voeten, J.T.M.; Kiseleva, I.; Heldens, J.G.; Bosch, H.V.D.; Klimov, A.; Rudenko, L.; Cox, N.J.; et al. Genetic Bases of the Temperature-Sensitive Phenotype of a Master Donor Virus Used in Live Attenuated Influenza Vaccines: A/Leningrad/134/17/57 (H2N2). Virology 2011, 412, 297–305. [Google Scholar] [CrossRef]
- Reed, L.; Muench, H. A Simple Method of Estimating Fifty per Cent Endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Lee, Y.-N.; Lee, Y.-T.; Kim, M.-C.; Hwang, H.S.; Lee, J.S.; Kim, K.-H.; Kang, S.-M. Fc Receptor Is Not Required for Inducing Antibodies but Plays a Critical Role in Conferring Protection After Influenza M2 Vaccination. Immunology 2014, 143, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Lee, Y.-T.; Park, S.; Jung, Y.-J.; Lee, Y.; Ko, E.-J.; Kim, Y.-J.; Li, X.; Kang, S.-M. Neuraminidase Expressing Virus-Like Particle Vaccine Provides Effective Cross Protection Against Influenza Virus. Virology 2019, 535, 179–188. [Google Scholar] [CrossRef]
- Hawksworth, A.; Jayachander, M.; Hester, S.; Mohammed, S.; Hutchinson, E.C. Proteomics as a Tool for Live Attenuated Influenza Vaccine Characterisation. Vaccine 2020, 38, 868–877. [Google Scholar] [CrossRef]
- Kurosaki, T.; Kometani, K.; Ise, W. Memory B Cells. Nat. Rev. Immunol. 2015, 15, 149–159. [Google Scholar] [CrossRef]
- Braciale, T.J.; Sun, J.; Kim, T.S. Regulating the Adaptive Immune Response to Respiratory Virus Infection. Nat. Rev. Immunol. 2012, 12, 295–305. [Google Scholar] [CrossRef]
- Fiers, W.; De Filette, M.; El Bakkouri, K.; Schepens, B.; Roose, K.; Schotsaert, M.; Birkett, A.; Saelens, X. M2e- Based Universal Influenza A Vaccine. Vaccine 2009, 27, 6280–6283. [Google Scholar] [CrossRef]
- Deng, L.; Cho, K.J.; Fiers, W.; Saelens, X. M2e-Based Universal Influenza A Vaccines. Vaccines 2015, 3, 105–136. [Google Scholar] [CrossRef]
- Neirynck, S.; DeRoo, T.; Saelens, X.; Vanlandschoot, P.; Jou, W.M.; Fiers, W. A Universal Influenza A Vaccine Based on the Extracellular Domain of the M2 Protein. Nat. Med. 1999, 5, 1157–1163. [Google Scholar] [CrossRef]
- Sun, W.; Zheng, A.; Miller, R.; Krammer, F.; Palese, P. An Inactivated Influenza Virus Vaccine Approach to Targeting the Conserved Hemagglutinin Stalk and M2e Domains. Vaccines 2019, 7, 117. [Google Scholar] [CrossRef]
- Thomas, P.G.; Brown, S.A.; Yue, W.; So, J.; Webby, R.J.; Doherty, P.C. An Unexpected Antibody Response to an Engineered Influenza Virus Modifies cd8+ T Cell Responses. Proc. Natl. Acad. Sci. USA 2006, 103, 2764–2769. [Google Scholar] [CrossRef]
- Lu, X.; Edwards, L.E.; Desheva, Y.; Nguyen, D.C.; Rekstin, A.R.; Stephenson, I.; Szretter, K.J.; Cox, N.J.; Rudenko, L.; Klimov, A.; et al. Cross-Protective Immunity in Mice Induced by Live-Attenuated or Inactivated Vaccines Against Highly Pathogenic Influenza A (H5N1) Viruses. Vaccine 2006, 24, 6588–6593. [Google Scholar] [CrossRef]
- Korenkov, D.A.; Laurie, K.L.; Reading, P.C.; Carolan, L.A.; Chan, K.F.; Isakova-Sivak, I.I.; Smolonogina, T.A.; Subbarao, K.; Barr, I.; Villanueva, J.; et al. Safety, Immunogenicity and Protection of A(H3N2) Live Attenuated Influenza Vaccines Containing Wild-Type Nucleoprotein in a Ferret Model. Infect. Genet. Evol. 2018, 64, 95–104. [Google Scholar] [CrossRef]
- Lam, J.H.; Baumgarth, N. The Multifaceted B Cell Response to Influenza Virus. J. Immunol. 2019, 202, 351–359. [Google Scholar] [CrossRef]
- Taylor, H.P.; Dimmock, N.J. Mechanism of Neutralization of Influenza Virus by Secretory IgA Is Different from That of Monomeric IgA or IgG. J. Exp. Med. 1985, 161, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Biondo, C.; Lentini, G.; Beninati, C.; Teti, G. The Dual Role of Innate Immunity During Influenza. Biomed. J. 2019, 42, 8–18. [Google Scholar] [CrossRef]
- Wu, T.; Guan, J.; Handel, A.; Tscharke, D.C.; Sidney, J.; Sette, A.; Wakim, L.M.; Sng, X.Y.X.; Thomas, P.G.; Croft, N.P.; et al. Quantification of Epitope Abundance Reveals the Effect of Direct and Cross-Presentation on Influenza CTL Responses. Nat. Commun. 2019, 10, 1–14. [Google Scholar] [CrossRef]
- Schotsaert, M.; Ibañez, L.I.; Fiers, W.; Saelens, X. Controlling Influenza by Cytotoxic T-Cells: Calling for Help from Destroyers. J. Biomed. Biotechnol. 2010, 2010, 1–13. [Google Scholar] [CrossRef][Green Version]
- Di Mario, G.; Garulli, B.; Sciaraffia, E.; Facchini, M.; Donatelli, I.; Castrucci, M.R. A Heat-Inactivated H7N3 Vaccine Induces Cross-Reactive Cellular Immunity in HLA-A2.1 Transgenic Mice. Virol. J. 2016, 13, 56. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, D.G.; Omokanye, A.; Schön, K.; Wenzel, U.A.; Bernasconi, V.; Bemark, M.; Kolpe, A.; El Bakkouri, K.; Ysenbaert, T.; Deng, L.; et al. M2e-Tetramer-Specific Memory CD4 T Cells Are Broadly Protective Against Influenza Infection. Mucosal Immunol. 2017, 11, 273–289. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotomina, T.; Isakova-Sivak, I.; Kim, K.-H.; Park, B.R.; Jung, Y.-J.; Lee, Y.; Mezhenskaya, D.; Matyushenko, V.; Kang, S.-M.; Rudenko, L. Generation and Characterization of Universal Live-Attenuated Influenza Vaccine Candidates Containing Multiple M2e Epitopes. Vaccines 2020, 8, 648. https://doi.org/10.3390/vaccines8040648
Kotomina T, Isakova-Sivak I, Kim K-H, Park BR, Jung Y-J, Lee Y, Mezhenskaya D, Matyushenko V, Kang S-M, Rudenko L. Generation and Characterization of Universal Live-Attenuated Influenza Vaccine Candidates Containing Multiple M2e Epitopes. Vaccines. 2020; 8(4):648. https://doi.org/10.3390/vaccines8040648
Chicago/Turabian StyleKotomina, Tatiana, Irina Isakova-Sivak, Ki-Hye Kim, Bo Ryoung Park, Yu-Jin Jung, Youri Lee, Daria Mezhenskaya, Victoria Matyushenko, Sang-Moo Kang, and Larisa Rudenko. 2020. "Generation and Characterization of Universal Live-Attenuated Influenza Vaccine Candidates Containing Multiple M2e Epitopes" Vaccines 8, no. 4: 648. https://doi.org/10.3390/vaccines8040648
APA StyleKotomina, T., Isakova-Sivak, I., Kim, K.-H., Park, B. R., Jung, Y.-J., Lee, Y., Mezhenskaya, D., Matyushenko, V., Kang, S.-M., & Rudenko, L. (2020). Generation and Characterization of Universal Live-Attenuated Influenza Vaccine Candidates Containing Multiple M2e Epitopes. Vaccines, 8(4), 648. https://doi.org/10.3390/vaccines8040648




