Immuno-Oncotherapeutic Approaches in Advanced Hepatocellular Carcinoma
Abstract
1. Introduction
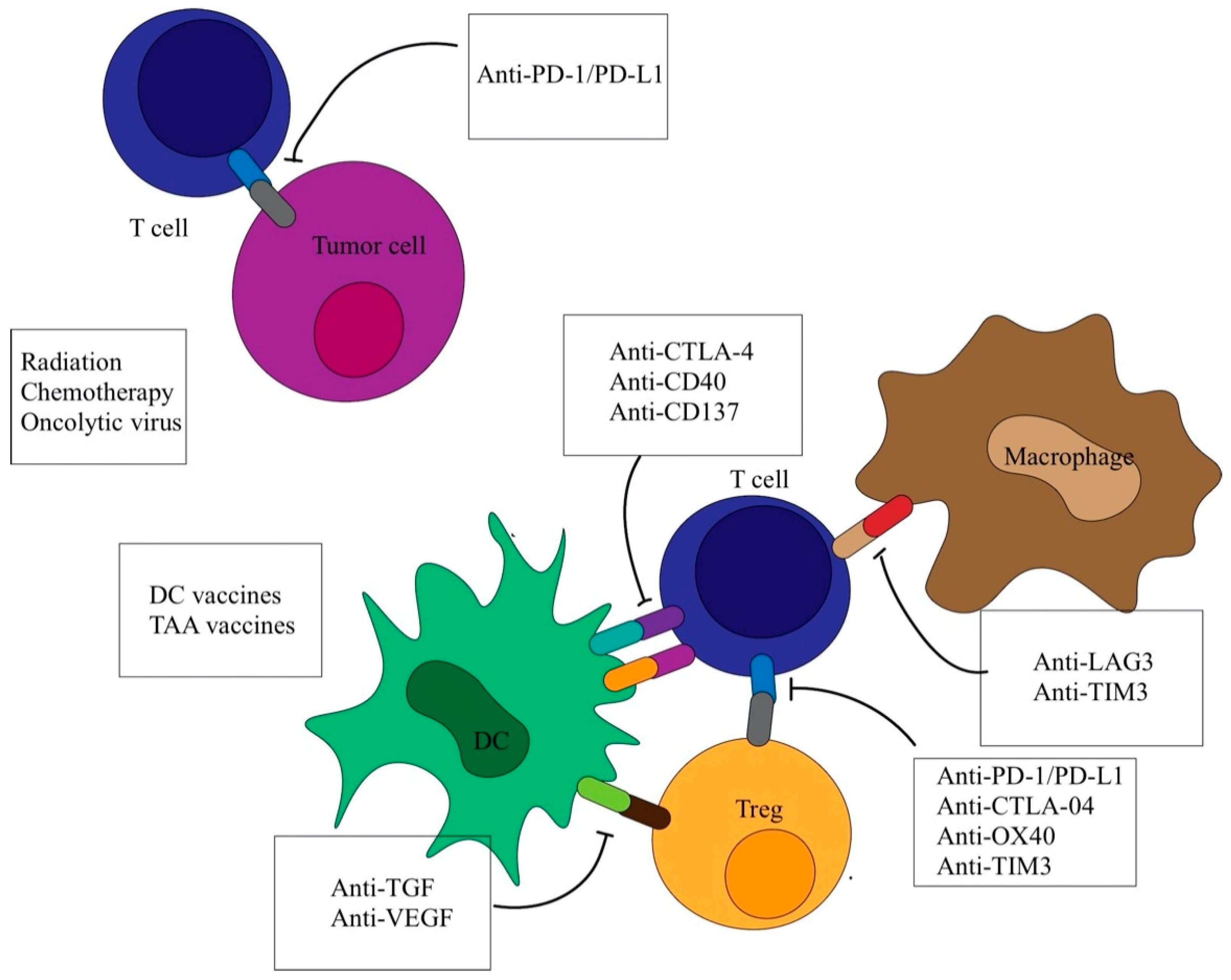
2. Indirect Immunotherapy: Immune Checkpoint Inhibitors
2.1. PD-1/PD-L1 Inhibitors
2.2. CTLA-4 Inhibitors
2.3. Combination Therapy with Immune Checkpoint Inhibitors
3. Indirect Immunotherapy: Cancer Vaccines
3.1. Antigen Peptide Vaccines
3.2. Dendritic Cells (DC) Vaccines
3.3. Oncolytic Virus Vaccines (OVs)
4. Direct Immunotherapy: Adoptive Cell Therapy
4.1. Chimeric Antigen Receptor T (CAR-T) Cells
4.2. Cytokine-Induced Killer (CIK) Cells
4.3. Natural Killer (NK) Cells
4.4. Tumor-Infiltrating Lymphocytes (TILs)
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gco.Iarc.Fr. Fact Sheets by the Global Cancer Observatory. 2018. Available online: https://gco.iarc.fr/ (accessed on 18 September 2018).
- Loffler, M.W.; Mohr, C.; Bichmann, L.; Freudenmann, L.K.; Walzer, M.; Schroeder, C.M.; Trautwein, N.; Hilke, F.J.; Zinser, R.S.; Muhlenbruch, L.; et al. Multi-omics discovery of exome-derived neoantigens in hepatocellular carcinoma. Genome Med. 2019, 11, 28. [Google Scholar] [CrossRef]
- Granata, V.; Fusco, R.; Amato, D.M.; Albino, V.; Patrone, R.; Izzo, F.; Petrillo, A. Beyond the vascular profile: Conventional DWI, IVIM and kurtosis in the assessment of hepatocellular carcinoma. Eur. Rev. Med. Pharm. Sci. 2020, 24, 7284–7293. [Google Scholar] [CrossRef]
- Xu, X.; Wang, Y.; Xue, F.; Guan, E.; Tian, F.; Xu, J.; Zhang, H. BST2 Promotes Tumor Growth via Multiple Pathways in Hepatocellular Carcinoma. Cancer Invest. 2020, 38, 329–337. [Google Scholar] [CrossRef]
- Restifo, N.P.; Kawakami, Y.; Marincola, F.; Shamamian, P.; Taggarse, A.; Esquivel, F.; Rosenberg, S.A. Molecular mechanisms used by tumors to escape immune recognition: Immunogenetherapy and the cell biology of major histocompatibility complex class I. J. Immunother. Emphas. Tumor Immunol. Off. J. Soc. Biol. Ther. 1993, 14, 182–190. [Google Scholar] [CrossRef]
- Fujiwara, K.; Higashi, T.; Nouso, K.; Nakatsukasa, H.; Kobayashi, Y.; Uemura, M.; Nakamura, S.; Sato, S.; Hanafusa, T.; Yumoto, Y.; et al. Decreased expression of B7 costimulatory molecules and major histocompatibility complex class-I in human hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2004, 19, 1121–1127. [Google Scholar] [CrossRef]
- Tatsumi, T.; Takehara, T.; Katayama, K.; Mochizuki, K.; Yamamoto, M.; Kanto, T.; Sasaki, Y.; Kasahara, A.; Hayashi, N. Expression of costimulatory molecules B7-1 (CD80) and B7-2 (CD86) on human hepatocellular carcinoma. Hepatology 1997, 25, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Sun, Q.; Zhang, X. PD-1 and its ligands are important immune checkpoints in cancer. Oncotarget 2017, 8, 2171–2186. [Google Scholar] [CrossRef] [PubMed]
- Yasuoka, H.; Asai, A.; Ohama, H.; Tsuchimoto, Y.; Fukunishi, S.; Higuchi, K. Increased both PD-L1 and PD-L2 expressions on monocytes of patients with hepatocellular carcinoma was associated with a poor prognosis. Sci. Rep. 2020, 10, 10377. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.M.; Chen, H.L.; Hu, R.H.; Lee, P.H. Harnessing immunotherapy for liver recipients with hepatocellular carcinoma: A review from a transplant oncology perspective. Ther. Adv. Med. Oncol. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Greten, T.F.; Wang, X.W.; Korangy, F. Current concepts of immune based treatments for patients with HCC: From basic science to novel treatment approaches. Gut 2015, 64, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Maleki Vareki, S.; Garrigos, C.; Duran, I. Biomarkers of response to PD-1/PD-L1 inhibition. Crit. Rev. Oncol. Hematol. 2017, 116, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Sznol, M.; Chen, L. Antagonist antibodies to PD-1 and B7-H1 (PD-L1) in the treatment of advanced human cancer. Clin. Cancer Res. 2013, 19, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef] [PubMed]
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.; Kim, T.Y.; Choo, S.P.; Trojan, J.; Welling, T.H.; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Gong, J.; Chehrazi-Raffle, A.; Reddi, S.; Salgia, R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: A comprehensive review of registration trials and future considerations. J. Immunother. Cancer 2018, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Yau, T.; Park, J.W.; Finn, R.S.; Cheng, A.L.; Mathurin, P.; Edeline, J.; Kudo, M.; Han, K.H.; Harding, J.J.; Merle, P.; et al. LBA38_PR-CheckMate 459: A randomized, multi-center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Ann. Oncol. 2019, 30, v874–v875. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Edeline, J.; Cattan, S.; Ogasawara, S.; Palmer, D.H.; Verslype, C.; Zagonel, V.; Fartoux, L.; Vogel, A.; et al. Updated efficacy and safety of KEYNOTE-224: A phase II study of pembrolizumab (Pembro) in patients with advanced hepatocellular carcinoma (HCC). J. Clin. Oncol. 2020, 38. [Google Scholar] [CrossRef]
- Finn, R.S.; Ryoo, B.Y.; Merle, P.; Kudo, M.; Bouattour, M.; Lim, H.Y.; Breder, V.; Edeline, J.; Chao, Y.; Ogasawara, S.; et al. Pembrolizumab As second-line therapy in patients with advanced hepatocellular carcinoma in KEYNOTE-240: A randomized, double-blind, phase III trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2020, 38, 193–202. [Google Scholar] [CrossRef]
- Lee, K.-M.; Chuang, E.; Griffin, M.; Khattri, R.; Hong, D.K.; Zhang, W.; Straus, D.; Samelson, L.E.; Thompson, C.B.; Bluestone, J.A. Molecular basis of T cell inactivation by CTLA-4. Science 1998, 282, 2263. [Google Scholar] [CrossRef]
- Sangro, B.; Matilla, A.M.; Santoro, A.; Melero, I.; Gracian, A.C.; Acosta, M.R.; Choo, S.P.; El-Khoureiry, A.; Kuromatsu, R.; El-Rayes, B.; et al. Efficacy and hepatic safety of nivolumab treatment in patients with Child-Pugh B disease and advanced hepatocellular carcinoma in CheckMate 040. J. Hepatol. 2019, 70, e619. [Google Scholar] [CrossRef]
- Duffy, A.G.; Ma, C.; Ulahannan, S.V.; Rahma, O.E.; Makarova-Rusher, O.; Cao, L.; Yu, Y.; Kleiner, D.E.; Trepel, J.; Lee, M.J.; et al. Phase I and preliminary phase II study of TRC105 in combination with sorafenib in hepatocellular carcinoma. Clin. Cancer Res. 2017, 23, 4633–4641. [Google Scholar] [CrossRef] [PubMed]
- Kelley, R.K.; Sangro, B.; Harris, W.P.; Ikeda, M.; Okusaka, T.; Kang, Y.-K.; Qin, S.; Tai, W.M.D.; Lim, H.Y.; Yau, T.; et al. Efficacy, tolerability, and biologic activity of a novel regimen of tremelimumab (T) in combination with durvalumab (D) for patients (pts) with advanced hepatocellular carcinoma (aHCC). J. Clin. Oncol. 2020, 38, 4508. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Paz-Ares, L.; Bernabe Caro, R.; Zurawski, B.; Kim, S.-W.; Carcereny Costa, E.; Park, K.; Alexandru, A.; Lupinacci, L.; de la Mora Jimenez, E.; et al. Nivolumab plus Ipilimumab in Advanced Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2019, 381, 2020–2031. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Tannir, N.M.; McDermott, D.F.; Arén Frontera, O.; Melichar, B.; Choueiri, T.K.; Plimack, E.R.; Barthélémy, P.; Porta, C.; George, S.; et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2018, 378, 1277–1290. [Google Scholar] [CrossRef] [PubMed]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.-J.; Rutkowski, P.; Lao, C.D.; Cowey, C.L.; Schadendorf, D.; Wagstaff, J.; Dummer, R.; et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N. Engl. J. Med. 2019, 381, 1535–1546. [Google Scholar] [CrossRef]
- Johnson, P.J.; Qin, S.; Park, J.W.; Poon, R.T.; Raoul, J.L.; Philip, P.A.; Hsu, C.H.; Hu, T.H.; Heo, J.; Xu, J.; et al. Brivanib versus sorafenib as first-line therapy in patients with unresectable, advanced hepatocellular carcinoma: Results from the randomized phase III BRISK-FL study. J. Clin. Oncol. 2013, 31, 3517–3524. [Google Scholar] [CrossRef]
- Zhu, A.X.; Kudo, M.; Assenat, E.; Cattan, S.; Kang, Y.K.; Lim, H.Y.; Poon, R.T.; Blanc, J.F.; Vogel, A.; Chen, C.L.; et al. Effect of everolimus on survival in advanced hepatocellular carcinoma after failure of sorafenib: The EVOLVE-1 randomized clinical trial. JAMA 2014, 312, 57–67. [Google Scholar] [CrossRef]
- Cainap, C.; Qin, S.; Huang, W.T.; Chung, I.J.; Pan, H.; Cheng, Y.; Kudo, M.; Kang, Y.K.; Chen, P.J.; Toh, H.C.; et al. Phase III trial of linifanib versus sorafenib in patients with advanced hepatocellular carcinoma (HCC). J. Clin. Oncol. 2013, 31, 249. [Google Scholar] [CrossRef]
- Kang, Y.K.; Yau, T.; Park, J.W.; Lim, H.Y.; Lee, T.Y.; Obi, S.; Chan, S.L.; Qin, S.; Kim, R.D.; Casey, M.; et al. Randomized phase II study of axitinib versus placebo plus best supportive care in second-line treatment of advanced hepatocellular carcinoma. Ann. Oncol. 2015, 26, 2457–2463. [Google Scholar] [CrossRef]
- Santoro, A.; Rimassa, L.; Borbath, I.; Daniele, B.; Salvagni, S.; Van Laethem, J.L.; Van Vlierberghe, H.; Trojan, J.; Kolligs, F.T.; Weiss, A.; et al. Tivantinib for second-line treatment of advanced hepatocellular carcinoma: A randomised, placebo-controlled phase 2 study. Lancet Oncol. 2013, 14, 55–63. [Google Scholar] [CrossRef]
- Motz, G.T.; Santoro, S.P.; Wang, L.P.; Garrabrant, T.; Lastra, R.R.; Hagemann, I.S.; Lal, P.; Feldman, M.D.; Benencia, F.; Coukos, G. Tumor endothelium FasL establishes a selective immune barrier promoting tolerance in tumors. Nat. Med. 2014, 20, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Roland, C.L.; Dineen, S.P.; Lynn, K.D.; Sullivan, L.A.; Dellinger, M.T.; Sadegh, L.; Sullivan, J.P.; Shames, D.S.; Brekken, R.A. Inhibition of vascular endothelial growth factor reduces angiogenesis and modulates immune cell infiltration of orthotopic breast cancer xenografts. Mol. Cancer Ther. 2009, 8, 1761–1771. [Google Scholar] [CrossRef] [PubMed]
- Voron, T.; Colussi, O.; Marcheteau, E.; Pernot, S.; Nizard, M.; Pointet, A.-L.; Latreche, S.; Bergaya, S.; Benhamouda, N.; Tanchot, C.; et al. VEGF-A modulates expression of inhibitory checkpoints on CD8+ T cells in tumors. J. Exp. Med. 2015, 212, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Wallin, J.J.; Bendell, J.C.; Funke, R.; Sznol, M.; Korski, K.; Jones, S.; Hernandez, G.; Mier, J.; He, X.; Hodi, F.S.; et al. Atezolizumab in combination with bevacizumab enhances antigen-specific T-cell migration in metastatic renal cell carcinoma. Nat. Commun. 2016, 7, 12624. [Google Scholar] [CrossRef]
- Hegde, P.S.; Wallin, J.J.; Mancao, C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin. Cancer Biol. 2018, 52, 117–124. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- He, A.R.; Yau, T.; Hsu, C.; Kang, Y.K.; Kim, T.Y.; Santoro, A.; Sangro, B.; Melero, I.; Kudo, M.; Hou, M.M.; et al. Nivolumab (NIVO)+ipilimumab (IPI) combination therapy in patients (pts) with advanced hepatocellular carcinoma (aHCC): Subgroup analyses from CheckMate 040. J. Clin. Oncol. 2020, 38. [Google Scholar] [CrossRef]
- Yau, T.; Kang, Y.K.; Kim, T.Y.; El-Khoueiry, A.B.; Santoro, A.; Sangro, B.; Melero, I.; Kudo, M.; Hou, M.M.; Matilla, A.; et al. Nivolumab (NIVO)+ipilimumab (IPI) combination therapy in patients (pts) with advanced hepatocellular carcinoma (aHCC): Results from Check-Mate 040. J. Clin. Oncol. 2019, 37. [Google Scholar] [CrossRef]
- Bei, R.; Mizejewski, G.J. Alpha fetoprotein is more than a hepatocellular cancer biomarker: From spontaneous immune response in cancer patients to the development of an AFP-based cancer vaccine. Curr. Mol. Med. 2011, 11, 564–581. [Google Scholar] [CrossRef]
- Galluzzi, L.; Senovilla, L.; Vacchelli, E.; Eggermont, A.; Fridman, W.H.; Galon, J.; Sautes-Fridman, C.; Tartour, E.; Zitvogel, L.; Kroemer, G. Trial watch: Dendritic cell-based interventions for cancer therapy. Oncoimmunology 2012, 1, 1111–1134. [Google Scholar] [CrossRef]
- Kirkwood, J.M.; Butterfield, L.H.; Tarhini, A.A.; Zarour, H.; Kalinski, P.; Ferrone, S. Immunotherapy of cancer in 2012. CA Cancer J. Clin. 2012, 62, 309–335. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, N.; Yoshikawa, T.; Fujinami, N.; Saito, K.; Mizuno, S.; Sawada, Y.; Endo, I.; Nakatsura, T. Immunological efficacy of glypican-3 peptide vaccine in patients with advanced hepatocellular carcinoma. Oncoimmunology 2017, 6, e1346764. [Google Scholar] [CrossRef] [PubMed]
- Tada, F.; Abe, M.; Hirooka, M.; Ikeda, Y.; Hiasa, Y.; Lee, Y.; Jung, N.C.; Lee, W.B.; Lee, H.S.; Bae, Y.S.; et al. Phase I/II study of immunotherapy using tumor antigen-pulsed dendritic cells in patients with hepatocellular carcinoma. Int. J. Oncol. 2012, 41, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.R.; Kratzke, R.A. Oncolytic virus therapy for cancer: The first wave of translational clinical trials. Transl. Res. 2013, 161, 355–364. [Google Scholar] [CrossRef]
- Bell, J.; McFadden, G. Viruses for tumor therapy. Cell Host. Microbe. 2014, 15, 260–265. [Google Scholar] [CrossRef]
- Schneider, W.M.; Chevillotte, M.D.; Rice, C.M. Interferon-stimulated genes: A complex web of host defenses. Annu. Rev. Immunol. 2014, 32, 513–545. [Google Scholar] [CrossRef]
- Yu, F.; Wang, X.; Guo, Z.S.; Bartlett, D.L.; Gottschalk, S.M.; Song, X.T. T-cell engager-armed oncolytic vaccinia virus significantly enhances antitumor therapy. Mol. Ther. 2014, 22, 102–111. [Google Scholar] [CrossRef]
- Russell, S.J.; Peng, K.W. Oncolytic Virotherapy: A Contest between Apples and Oranges. Mol. Ther. 2017, 25, 1107–1116. [Google Scholar] [CrossRef]
- Burke, S.; Shergold, A.; Elder, M.J.; Whitworth, J.; Cheng, X.; Jin, H.; Wilkinson, R.W.; Harper, J.; Carroll, D.K. Oncolytic Newcastle disease virus activation of the innate immune response and priming of antitumor adaptive responses in vitro. Cancer Immunol. Immunother. 2020, 69, 1015–1027. [Google Scholar] [CrossRef]
- Maroun, J.; Munoz-Alia, M.; Ammayappan, A.; Schulze, A.; Peng, K.W.; Russell, S. Designing and building oncolytic viruses. Future Virol. 2017, 12, 193–213. [Google Scholar] [CrossRef]
- Jung, M.Y.; Offord, C.P.; Ennis, M.K.; Kemler, I.; Neuhauser, C.; Dingli, D. In Vivo Estimation of Oncolytic Virus Populations within Tumors. Cancer Res. 2018, 78, 5992–6000. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.; Reid, T.; Ruo, L.; Breitbach, C.J.; Rose, S.; Bloomston, M.; Cho, M.; Lim, H.Y.; Chung, H.C.; Kim, C.W.; et al. Randomized dose-finding clinical trial of oncolytic immunotherapeutic vaccinia JX-594 in liver cancer. Nat. Med. 2013, 19, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.Y.; Jeong, S.N.; Kang, D.H.; Heo, J. Evolutionary cancer-favoring engineered vaccinia virus for metastatic hepatocellular carcinoma. Oncotarget 2017, 8, 71489–71499. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Xiang, Y.; Sheng, J.; Zhang, D.; Yao, X.; Yang, Y.; Zhang, X. Immunotherapy for Hepatocellular Carcinoma: Current Advances and Future Expectations. J Immunol Res. 2018, 2018. [Google Scholar] [CrossRef]
- Da Silva, J.L.; Dos Santos, A.L.S.; Nunes, N.C.C.; de Moraes Lino da Silva, F.; Ferreira, C.G.M.; de Melo, A.C. Cancer immunotherapy: The art of targeting the tumor immune microenvironment. Cancer Chemother. Pharmacol. 2019, 84, 227–240. [Google Scholar] [CrossRef]
- Atilla, E.; Kilic, P.; Gurman, G. Cellular therapies: Day by day, all the way. Transfus. Apher. Sci. 2018, 57, 187–196. [Google Scholar] [CrossRef]
- Noyan, F.; Zimmermann, K.; Hardtke-Wolenski, M.; Knoefel, A.; Schulde, E.; Geffers, R.; Hust, M.; Huehn, J.; Galla, M.; Morgan, M.; et al. Prevention of Allograft Rejection by Use of Regulatory T Cells With an MHC-Specific Chimeric Antigen Receptor. Am. J. Transplant. 2017, 17, 917–930. [Google Scholar] [CrossRef]
- Prieto, J.; Melero, I.; Sangro, B. Immunological landscape and immunotherapy of hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 681–700. [Google Scholar] [CrossRef]
- Wallstabe, L.; Gottlich, C.; Nelke, L.C.; Kuhnemundt, J.; Schwarz, T.; Nerreter, T.; Einsele, H.; Walles, H.; Dandekar, G.; Nietzer, S.L.; et al. ROR1-CAR T cells are effective against lung and breast cancer in advanced microphysiologic 3D tumor models. JCI Insight 2019, 4, 126345. [Google Scholar] [CrossRef]
- Forsberg, E.M.V.; Lindberg, M.F.; Jespersen, H.; Alsen, S.; Bagge, R.O.; Donia, M.; Svane, I.M.; Nilsson, O.; Ny, L.; Nilsson, L.M.; et al. HER2 CAR-T cells eradicate uveal melanoma and T-cell therapy-resistant human melanoma in IL2 transgenic NOD/SCID IL2 receptor knockout mice. Cancer Res. 2019, 79, 899–904. [Google Scholar] [CrossRef]
- Reiser, J.W. Regulation of CD8+T Cell Differentiation by Cytokines and T-Box Transcription Factors. Available online: https://archive.hshsl.umaryland.edu/handle/10713/9610 (accessed on 20 June 2020).
- Fraietta, J.A.; Lacey, S.F.; Orlando, E.J.; Pruteanu-Malinici, I.; Gohil, M.; Lundh, S.; Boesteanu, A.C.; Wang, Y.; O’Connor, R.S.; Hwang, W.T.; et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat. Med. 2018, 24, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.T.; Yuan, J.H.; Zhu, T.T.; Li, Y.Y.; Cheng, X.Y. Long noncoding RNA glypican 3 (GPC3) antisense transcript 1 promotes hepatocellular carcinoma progression via epigenetically activating GPC3. FEBS J. 2016, 283, 3739–3754. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Jiang, X.; Chen, S.; Lai, Y.; Wei, X.; Li, B.; Lin, S.; Wang, S.; Wu, Q.; Liang, Q.; et al. Anti-GPC3-CAR T cells suppress the growth of tumor cells in patient- derived xenografts of hepatocellular carcinoma. Front Immunol. 2016, 7, 690. [Google Scholar] [CrossRef]
- Wu, X.; Luo, H.; Shi, B.; Di, S.; Sun, R.; Su, J.; Liu, Y.; Li, H.; Jiang, H.; Li, Z. Combined antitumor effects of sorafenib and GPC3-CAR T cells in mouse models of hepatocellular carcinoma. Mol. Ther. 2019, 27, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Morse, M.A.; Sun, W.; Kim, R.; He, A.R.; Abada, P.B.; Mynderse, M.; Finn, R.S. The role of angiogenesis in hepatocellular carcinoma. Clin. Cancer Res. 2019, 25, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Batra, S.A.; Rathi, P.; Guo, L.; Courtney, A.N.; Fleurence, J.; Balzeau, J.; Shaik, R.S.; Nguyen, T.P.; Wu, M.F.; Bulsara, S.; et al. Glypican-3-specific CAR T cells coexpressing IL15 and IL21 have superior expansion and antitumor activity against hepatocellular carcinoma. Cancer Immunol. Res. 2020, 8, 309–320. [Google Scholar] [CrossRef]
- Berraondo, P.; Sanmamed, M.F.; Ochoa, M.C.; Etxeberria, I.; Aznar, M.A.; Perez-Gracia, J.L.; Rodriguez-Ruiz, M.E.; Ponz-Sarvise, M.; Castanon, E.; Melero, I. Cytokines in clinical cancer immunotherapy. Br. J. Cancer 2019, 120, 6–15. [Google Scholar] [CrossRef]
- Chen, C.; Li, K.; Jiang, H.; Song, F.; Gao, H.; Pan, X.; Shi, B.; Bi, Y.; Wang, H.; Li, Z. Development of T cells carrying two complementary chimeric antigen receptors against glypican-3 and asialoglycoprotein receptor 1 for the treatment of hepatocellular carcinoma. Cancer Immunol. Immunother. 2017, 66, 475–489. [Google Scholar] [CrossRef]
- Wang, H.; Liu, A.; Bo, W.; Feng, X.; Hu, Y.; Tian, L.; Zhang, H.; Tang, X. Adjuvant immunotherapy with autologous cytokine-induced killer cells for hepatocellular carcinoma patients after curative resection, a systematic review and meta-analysis. Dig. Liver. Dis. 2016, 48, 1275–1282. [Google Scholar] [CrossRef]
- Meng, Y.; Yu, Z.; Wu, Y.; Du, T.; Chen, S.; Meng, F.; Su, N.; Ma, Y.; Li, X.; Sun, S.; et al. Cell-based immunotherapy with cytokine-induced killer (CIK) cells: From preparation and testing to clinical application. Hum. Vaccin. Immunother. 2017, 13, 1–9. [Google Scholar] [CrossRef]
- Xu, L.; Wang, J.; Kim, Y.; Shuang, Z.Y.; Zhang, Y.J.; Lao, X.M.; Li, Y.Q.; Chen, M.S.; Pawlik, T.M.; Xia, J.C.; et al. A randomized controlled trial on patients with or without adjuvant autologous cytokine-induced killer cells after curative resection for hepatocellular carcinoma. Oncoimmunology 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Veluchamy, J.P.; Kok, N.; van der Vliet, H.J.; Verheul, H.M.W.; de Gruijl, T.D.; Spanholtz, J. The Rise of allogeneic natural killer cells as a platform for cancer immunotherapy: Recent innovations and future developments. Front Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, H.; Brenner, M.K. Immunotherapy against cancer-related viruses. Cell Res. 2017, 27, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Shubina, I.; Bliumenberg, A.G.; Volkov, S.M.; Demidov, L.V.; Kiselevskii, M.V. Adoptive immunotherapy of malignancies. Vestn. Ross. Akad. Med. Nauk. 2007, 11, 9–15. [Google Scholar]
- Wen, S.Y.; Wang, X.H.; Lin, L.; Guan, W.; Wang, S.Q. Preparation and property analysis of a hepatocyte targeting pH-sensitive liposome. World J. Gastroenterol. 2004, 10, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Galun, D.; Srdic-Rajic, T.; Bogdanovic, A.; Loncar, Z.; Zuvela, M. Targeted therapy and personalized medicine in hepatocellular carcinoma: Drug resistance, mechanisms, and treatment strategies. J. Hepatocell. Carcinoma. 2017, 4, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.X.; Chauhan, V.P.; Posada, J.; Ng, M.R.; Wu, M.W.; Adstamongkonkul, P.; Huang, P.; Lindeman, N.; Langer, R.; Jain, R.K. Blocking CXCR4 alleviates desmoplasia, increases T-lymphocyte infiltration, and improves immunotherapy in metastatic breast cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 4558–4566. [Google Scholar] [CrossRef]
- Martin, J.D.; Seano, G.; Jain, R.K. Normalizing function of tumor vessels: Progress, opportunities, and challenges. Annu. Rev. Physiol. 2019, 81, 505–534. [Google Scholar] [CrossRef]
- Wickstrom, S.; Lovgren, T. Expansion of tumor-infiltrating lymphocytes from melanoma tumors. Methods Mol. Biol. 2019, 1913, 105–118. [Google Scholar] [CrossRef]
| Immunotherapeutic Strategies for HCC | ||
|---|---|---|
| Direct | ||
| ACT * | CIK cells * | CIK -valproate |
| DC-CIK with TACE * | ||
| TLS | HCC | |
| NK cells * | NK with K562-mb1541BBL | |
| Sorafenib | ||
| NKG2D * | ||
| CAR-T * | Targeting GPC3 * | |
| (Generation 1–4) | Targeting GPC3 and ASGR1 * | |
| Indirect | ||
| Oncolytic viruses | CVV, JX-594 | |
| HCC vaccines | Cell vaccines | HCC cells with GM-CSF * |
| Antigen Peptide Vaccines | AFP *, GPC3, SSX-2 | |
| DC vaccines | Nifuroxazide | |
| DC-loaded TCL * | ||
| Immune checkpoint inhibitors | CTLA-4 Inhibitors * | Tremelimumab Ipilimumab |
| PD-1 Inhibitors * | Nivolumab | |
| Pembrolizumab | ||
| Pidilizumab | ||
| PD-L1 inhibitors * | Atezolizumab | |
| Durvalumab | ||
| NCT Number | Status | Conditions | Interventions | Phases |
|---|---|---|---|---|
| PD-1/PD-L1 | ||||
| NCT03419481 | Recruiting | Hepatocellular Carcinoma | Pembrolizumab | Phase 2 |
| NCT01658878 | Active, not recruiting | Hepatocellular Carcinoma | Nivolumab + Sorafenib | Phase 1/2 |
| NCT02940496 | Active, not recruiting | Hepatocellular Carcinoma | Pembrolizumab in combination with various drugs | Phase 1/2 |
| NCT03857815 | Recruiting | Hepatocellular Carcinoma | Anti-PD-1 + radiation | Phase 2 |
| NCT03914352 | Recruiting | Hepatocellular Carcinoma | Anti-PD-1 + TACE | n/a |
| NCT03722875 | Recruiting | Hepatocellular Carcinoma | SHR-1210 (anti-PD-1) + apatinib | n/a |
| NCT03966209 | Recruiting | Hepatocellular Carcinoma | JS001(PD-1 inhibitor) | Phase 1 |
| NCT03732547 | Recruiting | Hepatocellular Carcinoma | Anti-PD-1 | Phase 2 |
| NCT03071094 | Active, not recruiting | Hepatocellular Carcinoma | Nivolumab + Pexastimogene Devacirepvec (Pexa Vec) | Phase 1/2 |
| NCT03463876 | Active, not recruiting | Hepatocellular Carcinoma | SHR 1210 + apatinib | Phase 2 |
| NCT02989922 | Unknown | Hepatocellular Carcinoma, Non-Resectable | SHR-1210 | Phase 2 |
| NCT03412773 | Active, not recruiting | Hepatocellular Carcinoma | BGB-A317 (Tislelizumab, anti-PD-1) + Sorafenib | Phase 3 |
| NCT03419897 | Active, not recruiting | Hepatocellular Carcinoma | BGB-A317 | Phase 2 |
| NCT03785210 | Recruiting | Various solid cancers including hepatocellular carcinoma | Nivolumab in combination with various drugs | Phase 2 |
| NCT03655613 | Recruiting | Hepatocellular Carcinoma, Renal Cell Carcinoma | Nivolumab in combination with APL-501 or APL-101 | Phase 1/2 |
| NCT03605706 | Recruiting | Hepatocellular Carcinoma | SHR-1210 + FOLFOX4 | Phase 3 |
| NCT02423343 | Active, not recruiting | Various solid cancers including hepatocellular carcinoma | Nivolumab + Galunisertib | Phase 1/2 |
| NCT03259867 | Active, not recruiting | Various solid cancers including hepatocellular carcinoma | Anti-PD-1 | Phase 2 |
| NCT03973112 | Recruiting | Hepatocellular Carcinoma | HLX10 (anti-PD-1) + HLX04 | Phase 2 |
| NCT03645980 | Recruiting | Hepatocellular Carcinoma | DKN-01 + Sorafenib | Phase 1/2 |
| NCT03222076 | Recruiting | Hepatocellular Carcinoma | Nivolumab + Ipilimumab | Phase 2 |
| NCT02702401 | Active, not recruiting | Hepatocellular Carcinoma | Pembrolizumab | Phase 3 |
| NCT03316872 | Recruiting | Hepatocellular Carcinoma | Pembrolizumab + radiation | Phase 2 |
| NCT02886897 | Recruiting | Various solid cancers including hepatocellular carcinoma | Anti-PD-1 + D-CIK | Phase 1/2 |
| NCT03867084 | Unknown | Hepatocellular Carcinoma | Pembrolizumab | Phase 3 |
| NCT02795429 | Active, not recruiting | Hepatocellular Carcinoma | PDR001 (anti-PD-1) + INC280 | Phase 1/2 |
| NCT03099564 | Recruiting | Hepatocellular Carcinoma | Pembrolizumab | Phase 1 |
| NCT03949231 | Active, not recruiting | Hepatocellular Carcinoma | Toripalimab | Phase 3 |
| NCT03062358 | Active, not recruiting | Hepatocellular Carcinoma | Pembrolizumab | Phase 3 |
| NCT03474640 | Recruiting | Various solid cancers including hepatocellular carcinoma | Toripalimab | Phase 1 |
| NCT02947165 | Recruiting | Various solid cancers including hepatocellular carcinoma | PDR001 + NIS793 (anti-TGF) | Phase 1 |
| NCT03836352 | Recruiting | Various solid cancers including hepatocellular carcinoma | Pembrolizumab in combination with various drugs | Phase 2 |
| NCT03170960 | Recruiting | Various solid cancers including hepatocellular carcinoma | Atezolizumab + Cabozantinib | Phase 1/2 |
| NCT03941873 | Recruiting | Various solid cancers including hepatocellular carcinoma | Tislelizumab + Sitravatinib | Phase 1/2 |
| NCT03638141 | Recruiting | Hepatocellular Carcinoma | Durvalumab and Tremelimumab | Phase 2 |
| NCT02658019 | Active, not recruiting | Hepatocellular Carcinoma | Pembrolizumab | Phase 2 |
| NCT02702414 | Active, not recruiting | Hepatocellular Carcinoma | Pembrolizumab | Phase 2 |
| NCT03539822 | Recruiting | Various GI cancers including hepatocellular carcinoma | Durvalumab + Cabozantinib | Phase 1 |
| NCT03228667 | Recruiting | Various solid cancers including hepatocellular carcinoma | Pembrolizumab, Nivolumab, Atezolizumab, Avelumab, and ALT-803 | Phase 2 |
| NCT03713593 | Active, not recruiting | Hepatocellular Carcinoma | Pembrolizumab + Lenvatinib | Phase 3 |
| NCT03829501 | Recruiting | Various solid cancers including hepatocellular carcinoma | Atezolizumab + KY1044 | Phase 1/2 |
| NCT03563170 | Active, not recruiting | Hepatocellular Carcinoma | Avelumab in combination with various drugs | Phase 1/2 |
| NCT04246177 | Recruiting | Hepatocellular Carcinoma | Pembrolizumab with lenvatinib with TACE | Phase 3 |
| NCT04170556 | Recruiting | Hepatocellular Carcinoma | Regorafenib followed by pembrolizumab | Phase 1/2 |
| CTLA-4 | ||||
| NCT02821754 | Recruiting | Liver and biliary tract cancer | Tremelimumab and durvalumab | Phase 2 |
| NCT04430452 | Not yet recruiting | Hepatocellular Carcinoma | Radiation followed by durvalumab with or without tremelimumab | |
| Vaccines | ||||
| NCT03674073 | Recruiting | Hepatocellular Carcinoma | Neoantigen vaccine | Phase 1 |
| NCT02232490 | Recruiting | Hepatocellular Carcinoma | hepcortespenlisimut-L | Phase 3 |
| NCT03086564 | Unknown | Hepatocellular Carcinoma | HBV expressing DCs | Phase 1/2 |
| NCT02432963 | Active, not recruiting | Various solid cancers including hepatocellular carcinoma | Modified Vaccinia Virus Expressing p53 + Pembrolizumab | Phase 1 |
| NCT04317248 | Not yet recruiting | Hepatocellular Carcinoma | Dendritic Cell Vaccine | Phase 2 |
| NCT04251117 | Recruiting | HCC | GNOS-PV02 (Peptide Vaccine) | Phase 1/2 |
| NCT04248569 | Recruiting | Hepatocellular Carcinoma | Peptide Vaccine | Phase 1 |
| NCT03311334 | Recruiting | Various solid cancers including hepatocellular carcinoma | DSP-7888 (Peptide Vaccine) | Phase 1/2 |
| NCT01266707 | Unknown status | Hepatocellular Carcinoma | Peptide Vaccine | Phase 1 |
| NCT00610389 | Unknown status | Various solid cancers including hepatocellular carcinoma | Dendritic Cell Vaccine | Phase 2 |
| Oncolytic viruses | ||||
| NCT02562755 | Active, not recruiting | Hepatocellular Carcinoma | Pexastimogene Devacirepvec (Pexa Vec) + Sorafenib | Phase 3 |
| NCT01628640 | Active, not recruiting | Various solid cancers including hepatocellular carcinoma | Recombinant VSV-expressing Interferon-beta | Phase 1 |
| NCT03647163 | Recruiting | Various solid cancers including hepatocellular carcinoma | VSV-IFNÎ2-NIS + Pembrolizumab | Phase 1 |
| NCT03313596 | Recruiting | Hepatocellular Carcinoma | ADV-Tk | Phase 3 |
| NCT02293850 | Recruiting | Hepatocellular Carcinoma | Telomelysin (adenovirus) | Phase 1 |
| ACT | ||||
| NCT03175679 | Unknown | Hepatocellular Carcinoma | iNKT cells | Phase 1 |
| NCT03175705 | Unknown | Hepatocellular Carcinoma | HCC antigens-specific CD8+ T lymphocytes | Phase 1 |
| NCT03441100 | Recruiting | Various solid cancers including hepatocellular carcinoma | IMA202 (TCR-engineered T cells) | Phase 1 |
| NCT03980288 | Recruiting | Hepatocellular Carcinoma | CAR-GPC3 T Cells | Phase 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, R.; Eshrat, F.; Al-Jumayli, M.; Saeed, A.; Saeed, A. Immuno-Oncotherapeutic Approaches in Advanced Hepatocellular Carcinoma. Vaccines 2020, 8, 447. https://doi.org/10.3390/vaccines8030447
Park R, Eshrat F, Al-Jumayli M, Saeed A, Saeed A. Immuno-Oncotherapeutic Approaches in Advanced Hepatocellular Carcinoma. Vaccines. 2020; 8(3):447. https://doi.org/10.3390/vaccines8030447
Chicago/Turabian StylePark, Robin, Fariha Eshrat, Mohammed Al-Jumayli, Azhar Saeed, and Anwaar Saeed. 2020. "Immuno-Oncotherapeutic Approaches in Advanced Hepatocellular Carcinoma" Vaccines 8, no. 3: 447. https://doi.org/10.3390/vaccines8030447
APA StylePark, R., Eshrat, F., Al-Jumayli, M., Saeed, A., & Saeed, A. (2020). Immuno-Oncotherapeutic Approaches in Advanced Hepatocellular Carcinoma. Vaccines, 8(3), 447. https://doi.org/10.3390/vaccines8030447




