1. Introduction
Vaccination is one of the most successful strategy to prevent infectious diseases in the human population, including those caused by emerging viruses [
1]. Among various vaccine types, such as inactivated virus, live attenuated virus, and viral vector-based vaccines, subunit vaccines using proteins or peptides are believed to be much safer as they do not contain any live virus components and/or cause undesirable severe side effects [
2]. However, unlike attenuated vaccines (composed of a virus or bacterium that replicates within the host) or inactivated vaccines (composed of either heat or chemically-inactivated parts of the pathogen), subunit vaccines (that are derived from known pathogen target antigens) are generally much less immunogenic, which can be improved with the addition of appropriate adjuvant(s) to the vaccine [
1].
Adjuvants play an important role in enhancing the potency of subunit vaccines by improving humoral and/or cellular immune responses to the various subunit protein vaccines, decreasing the antigen dosages, and/or reducing immunization regimens [
3,
4]. Aluminum salts (hereinafter alum) are the most widely used adjuvants in licensed human vaccines, such as hepatitis A virus (HAV), HBV, human papilloma virus (HPV), and tetanus [
1]. Alum has strong safety profiles, and generally stimulates humoral immunity through Th2-type immune responses, but it is a poor inducer of Th1-type humoral and cellular immune responses [
1,
5]. Two types of alum adjuvants are commercially available, including aluminum hydroxide and aluminum phosphate adjuvants [
6]. Since alum induces a weak or no Th1 response, an increasing number of licensed human vaccines or those under clinical development have combined alum with other immunostimulatory molecules that induce different immune mechanisms and thus generate synergistic Th1- and Th2-type of responses [
6]. For instance, AS04 adjuvant combines alum and the toll-like receptor 4 (TLR4) agonist monophosphoryl lipid A (MPL), and when used as an combinatorial adjuvant, it improves both humoral and cell-mediated immunity induced by the HBV and HPV vaccines [
1,
7]. Alum has also been combined with other immunostimulatory molecules, such as CpG (TLR9), QS-21, or GLA (TLR4). However, each combination requires that the ratios of the immunostimulant adjuvant to alum, as well as the sequence of mixing, are optimized in order for the adjuvanted vaccine to stimulate optimal protective immune responses [
6].
The rASP-1 recombinant protein adjuvant corresponds to the
Onchocerca volvulus secreted protein
Ov-ASP-1. In previous studies, we have shown that rASP-1 is an immunopotentiating adjuvant that binds and activates innate cells [
8,
9,
10], induces in mice a balanced Th1 (IgG2) and Th2 (IgG1)-associated antibody response, a biased Th1 cytokine response, and/or protection when combined with commercial vaccines (Haemorrhagic fever with renal syndrome, Influenza and Rabies), or with various experimental vaccine antigens such as ovalbumin, HIV-1 polypeptide, and the receptor binding protein domain (RBD) of severe acute respiratory syndrome coronavirus (SARS-CoV) spike (S) protein [
9,
10,
11,
12]. When used with the commercially available trivalent influenza vaccine (IIV3), in addition to protecting mice from lethal H1N1 challenge after one immunization with rASP-1-adjuvnated IIV3 vaccine, rASP-1 also facilitated antigen sparing of the IIV3 dose by 10 to 40-fold [
12].
Middle East respiratory syndrome (MERS) coronavirus (MERS-CoV) is an emerging zoonotic virus that was first reported in 2012 [
13]. The virus first binds to a cellular receptor dipeptidyl peptidase 4 (DPP4) through the receptor-binding domain (RBD) present within the S1 subunit of the S protein, whereas the S2 subunit undergoes conformational changes and subsequently mediates virus and cell membrane fusion, thus enabling MERS-CoV to enter into target cells [
14,
15]. The MERS-CoV RBD protein was identified as an important immunogen and the target for protective immune responses against the virus, which could also be induced by using experimental adjuvanted MERS-CoV RBD vaccines [
16,
17]. In previous studies, we have demonstrated that Montanide ISA51- or MF59-adjuvanted MERS-CoV RBD protein fused with a Fc tag of the human IgG (MERS-RBD-Fc) elicited strong antibody responses with neutralizing activity, and protected immunized transgenic mice against MERS-CoV infection [
18,
19]. In addition, a foldon (Fd) trimeric motif-fused MERS-RBD-Fd protein formulated with MF59 elicited long-term neutralizing antibodies with protective efficacy against MERS-CoV challenge in mice [
20]. These data confirmed the potency of the MERS-RBD protein as a vaccine antigen against MERS-CoV infections.
There has been an increase interest in employing combinatorial adjuvants in a vaccine to improve the immunogenicity and potency of subunit, attenuated and anti-cancer vaccines [
21,
22,
23]. Although alum has been tested for its combinatorial effects with various traditional immunostimulatory adjuvants, such as MPL, CpG, GLA, and QS21 [
1,
4], its combinatorial effect with a protein adjuvant, such as rASP-1 has not been investigated yet. In this study, we evaluated the adjuvanticity of alum (Th2-type stimulator) in combination with our novel rASP-1 protein adjuvant (Th1-type stimulator) using MERS-RBD-Fd (hereinafter RBD) as the model vaccine antigen. Notably, mice immunized with the combinatorial adjuvant system, where rASP-1 and the alum-adjuvanted RBD vaccine were co-administered separately, elicited neutralizing antibody titers that were similar to, or slightly lower than, those induced in mice immunized with either Montanide ISA51-adjuvanted or MF59-adjuvanted MERS CoV-RBD vaccines [
18,
19,
20]. Notably, the functional antibodies induced by the co-administered combinatorial alum and rASP-1 vaccine were significantly and positively associated with an increased frequency of T follicular helper (TfH) and germinal center B (GC B) cells within the draining lymph nodes of the immunized mice.
2. Materials and Methods
2.1. Mice
First, 6–8-week-old female C57BL/6 mice were purchased from Jackson Laboratory (Bar Harbor, ME, USA). All mice were maintained in an AAALAC approved barrier facility at the New York Blood Center (NYBC, New York, NY, USA) and allowed to acclimatize for at least one week in the animal facility prior to use. All protocols involving mice were conducted with the approval of the Institutional Animal Care and Use Committee (IACUC Protocol 255.15) at NYBC.
2.2. Preparation of Recombinant Proteins
Recombinant MERS-RBD-Fd, MERS-RBD-Fc, and MERS-S1 proteins were prepared as previously described [
20]. Briefly, genes encoding residues 377–588 of MERS-CoV S protein and a C-terminal Fd trimeric motif and His
6 tag (MERS-RBD-Fd) or a C-terminal Fc tag of human IgG (MERS-RBD-Fc) were constructed into the hIgG-Fc vector, whereas gene encoding residues 18-725 of MERS-CoV S protein containing a C-terminal His
6 tag (MERS-S1) was inserted into the pJW4303 vector. The sequence-confirmed recombinant plasmids were transfected into 293T cells using the calcium phosphate method, and culture supernatants containing expressed MERS-CoV proteins were collected 72 h post-transfection. The His-tagged (for MERS-RBD-Fd and MERS-S1) and Fc-tagged (for MERS-RBD-Fc) proteins were purified using Ni-NTA Superflow (Qiagen, Hilden, Germany) and Protein A affinity chromatography (GE Healthcare, Chicago, IL, USA), respectively.
Recombinant
Ov-ASP-1 (rASP-1) was expressed using the T7-promoter driven pET28a expression vector (Novagen, Merck KGaA, Darmstadt, Germany), and the sequence-confirmed recombinant plasmid was transformed into
E. coli BL21(DE3). The rASP-1 protein with an N-terminal His
6 tag was expressed after induction with 1 mM IPTG for 5 h. The resulting inclusion bodies were solubilized in 1% SDS and purified with IMAC as described [
12,
24]. The purified protein remained soluble in PBS, pH 7.4 containing 0.1% SDS. The endotoxin level was less than 11.41 EU/mg, as measured by Endosafe cartridge (Charles River, Houston, TX, USA). To make sure that the residual LPS within the 25 µg rASP-1 used per mouse for immunization did not influence the adjuvant specific outcomes in the experiments listed below, the protein was pre-treated with Polymyxin B sulfate (Sigma-Millipore, Burlington, MA, USA) for 60 min at RT before use [
12].
2.3. Mouse Immunization and Sample Collection
C57BL/6 mice were immunized intramuscularly (i.m.) twice, three weeks apart, with MERS-RBD-Fd (5 µg; hereinafter RBD) formulated with or without alum (Alhydrogel
® 250 µg) (InvivoGen, San Diego, CA, USA), rASP-1 (25 µg), or with distinct adjuvant combinations. Inoculums were prepared in a final volume of 100 µL per mouse and 50 µL was injected in the caudal thigh muscle of each hind leg in the appropriate site as outlined in
Table 1. Control mice were injected with PBS in 0.1% SDS, the buffer solution of rASP-1 and is referred to as naive mice hereafter. Complete adsorption of the RBD and rASP-1 proteins by alum was confirmed by SDS-gel electrophoresis of the unbound protein samples after absorption with alum for 30 min on a rotator at RT. Sera samples were collected 7 days post-2nd immunization for analyses of anti-MERS-CoV neutralizing antibody titers, inhibition of MERS-CoV RBD-DPP4 receptor binding, and MERS-CoV RBD-specific antibody responses using MERS-CoV S1 as a target. The draining lymph nodes from each hind leg per mouse were also recovered at day 7 post-2nd immunization for analyses of the various cell profiles within.
2.4. MERS Neutralization Assay
The neutralizing activity of sera from the immunized mice against MERS-CoV infection in vitro was carried out using our established pseudovirus neutralization assay [
17,
20]. Briefly, 293T cells were co-transfected with a plasmid encoding the S protein of MERS-CoV (strain EMC2012) and a plasmid encoding Env-defective, luciferase-expressing HIV-1 genome (pNL4-3.luc.RE). The supernatants containing MERS-CoV S expressing pseudovirus collected 72 h post-transfection were incubated with serially diluted mouse sera at 37 °C for 1 h. The virus-serum mixtures were then added into Huh-7 cells expressing the MERS-CoV receptor DPP4. The cells were refed with fresh medium 24 h later, and after 72 h, lysed using cell lysis buffer (Promega, Madison, WI, USA) before the supernatants were transferred into 96-well luminometer plates. After addition of luciferase substrate (Promega, Madison, WI, USA), the plates were measured for relative luciferase activity using Infinite 200 PRO Luminator (Tecan, Männedorf, Switzerland). Neutralizing activity was calculated using the CalcuSyn computer program [
25] and is expressed as 50% pseudovirus neutralizing antibody titers (NT
50).
2.5. Inhibition of Binding of the Middle East Respiratory Syndrome Coronavirus (MERS-CoV) RBD-Fc Protein to DPP4-Expressing Huh-7 Cells
Sera from immunized mice were tested for their ability to inhibit the binding of the recombinant MERS-RBD-Fc protein to the cell-associated hDPP4 receptor in Huh-7 cells using flow cytometry analysis [
26]. Briefly, cells were incubated with the MERS-RBD-Fc protein (5 µg/mL) in the presence or absence of diluted mouse sera (1:50) for 30 min at room temperature. After three washes and staining with FITC-labeled goat anti-human IgG Fc secondary antibody (1:500, Thermo Fisher Scientific, Waltham, MA, USA) for 30 min at room temperature, the cells were measured for fluorescence in a flow cytometer (BD LSRFortessa 4 system). Mean fluorescence intensity (MFI) values of the FITC channel from cells incubated with MERS-RBD-Fc protein in the absence of diluted sera were treated as 100 percentage binding. Inhibition of binding was calculated as the percentage of reduced binding to hDPP4 receptor in the presence of diluted sera from the different immunization groups versus the maximum binding observed in the absence of sera.
2.6. ELISA
ELISA to measure MERS-CoV RBD-specific antibody responses in immunized mouse sera was performed with MERS-CoV S1 as the target antigen and as previously described with some modifications [
17,
20]. The MERS-CoV S1 subunit of the MERS-CoV S protein contains the RBD region of the virus. Briefly, 96-well ELISA plates were coated with MERS-CoV S1 (1 µg/mL) overnight at 4 °C, blocked with 2% fat-free milk in PBS containing tween-20 (PBST) at 37 °C for 2 h, and then washed with PBST 3 times. The plates were subsequently incubated at 37 °C for 1 h with serially diluted mouse sera, and horseradish peroxidase (HRP)-conjugated anti-mouse IgG (1:5000), IgG1 (1:5000), or IgG2c (1:2000) antibodies (Thermo Fisher Scientific, Waltham, MA, USA). The substrate 3,3′,5,5′-tetramethylbenzidine (Sigma-Aldrich, St. Louis, MO, USA) was added to the plates after additional washes, and the reaction was stopped by the addition of 1 N H
2SO
4. Absorbance at 450 nm was measured using an ELISA plate reader (Tecan, Männedorf, Switzerland). Endpoint titers were calculated as the reciprocal of the highest dilution of sera giving an optical density greater than the mean ± 3 times the standard deviation of sera from naïve mice.
2.7. Profile of Cells in the Lymph Nodes
Draining lymph nodes from each hind leg per mouse were harvested at day 7 post-2nd immunization. The lymph nodes were dissociated into single cell suspensions using a syringe plunger, then passed through a 70 µm cell strainer and resuspended in complete RPMI 1640 media containing 10% fetal bovine serum (R10). Subsequently, 2.5 × 106 cells were washed and resuspended in fresh R10 media in a 96-well cell culture plate for flow cytometry staining. 1.5 × 106 cells were stained with a cocktail of the following fluorescently labeled antibodies: CD45-AF700, CD11b-PE-Cy5, CD11c-BV711, Ly6C-PerCP, CD40-APC, CCR7-PE, CD80-BV650, CD86-BV421, Ly6G-PE-Cy7, and B220-BV605. While 1 × 106 cells were stained with a cocktail of the following fluorescently labeled antibodies: CD45-AF700, CD4-PE-Cy7, CXCR5-BV605, PD-1-BV421, B220-BV650 and GL-7-AF647 (all from Biolegend, San Diego, CA, USA), and CD95-BV510 (BD Biosciences, Dublin, Ireland), in a brilliant violet cell stain buffer (BD Biosciences, Dublin, Ireland) for 20 min in the dark at room temperature. Cells were then washed, resuspended in cell stain buffer (BioLegend, San Diego, CA, USA), and the number of stained cells was acquired using BD LSRFortessa cell analyzer (BD Biosciences, Dublin, Ireland). The data were analyzed using Flowjo Software (Tree Star, Ashland, OR, USA). CD45+CD11c-Ly6C+ cells were identified as monocytes, CD45+CD11c-Ly6C+CD40+ cells were identified as activated monocytes, CD45+CD11c-Ly6C+CCR7+ cells were identified as migratory monocytes, CD45+CD4+ cells were identified as CD4+ T cells, CD45+CD4+CXCR5+PD-1+ cells were identified as TfH cells, CD45+B220+ cells were identified as B cells and CD45+B220+CD95+GL-7+ cells were identified as GC B cells.
2.8. Statistical Analysis
One-way ANOVA test with Tukey’s multiple comparison was used for statistical analysis using GraphPad Prism v6 (GraphPad, San Diego, CA, USA). Spearman correlation was performed to determine the association of the fold increase in the frequency of TfH and GC B cells with neutralizing antibody titers using GraphPad Prism v6 (GraphPad, San Diego, CA, USA). p < 0.05: *, p < 0.01: **, p < 0.001: ***, p < 0.0001: ****. ND: not detectable.
4. Discussion
Adjuvants are essential components in both prophylactic and therapeutic vaccines since they ameliorate antigen-specific protective immune responses [
28]. However, choosing the appropriate adjuvant that can be employed safely and that enhances vaccine efficacy is still elusive and needs to be optimized experimentally first [
29].
Besides a handful of adjuvants such as CPG, Poly I:C, MPLA and MF59, aluminum-based (alum) adjuvants are being used in most of the adjuvanted vaccines for humans globally even today since its inception 85 years ago [
30]. Although beneficial effects of alum as an adjuvant were observed with the DTap, HepB, and HepA vaccines, a biased Th2-type immune response, absence of strong cellular responses, and the induction of adverse reactions were some of the limitations found with the various alum-adjuvanted vaccines [
30]. Therefore, the utilization of a combination of adjuvants in vaccines that can improve the safety and efficacy of vaccines against emerging pathogens is being actively pursued by the research community [
31]. The use of combinatorial adjuvant system is beneficial since they can be tailored to target varied pattern-recognition receptors (PRRs) with each being able to enhance antigen-specific responses (cellular and humoral) in a complementary or synergistic outcome [
32]. For instance, intranasal vaccination with emulsified fine particles like PELC in combination with LD-indolicidin enhanced protective influenza-specific serological immunity in mice [
33]. MPL and CpG combination adjuvants promoted homologous and heterosubtypic cross protection when used with the inactivated split influenza virus vaccine [
34]. The co-administration of alum and a TLR-7 adjuvant enhanced memory B cell response to lymphocytic choriomeningitis virus (LCMV) antigen [
35]. Alum in combination with MPLA-HA-adjuvanted HBsAg increased both the magnitude and the persistence of HBsAg-specific immune responses against hepatitis B virus infection [
36].
The aim of the present study was to explore the synergistic potential of combining the
O. volvulus-derived protein adjuvant, rASP-1 with alum as a novel combinatorial adjuvant system using MERS-RBD-Fd as the model vaccine antigen. We have previously shown that rASP-1 enhances the immune response when co-administered in an aqueous formulation with several bystander vaccine antigens [
9,
10,
11]. Moreover, we have also reported that rASP-1-adjuvanted trivalent influenza vaccine (IIV3) elicits a balanced IgG1/IgG2c response to IIV3 and protects mice following H1N1 virus challenge, potentially via MyD88-independent TLR4 signaling [
8,
12].
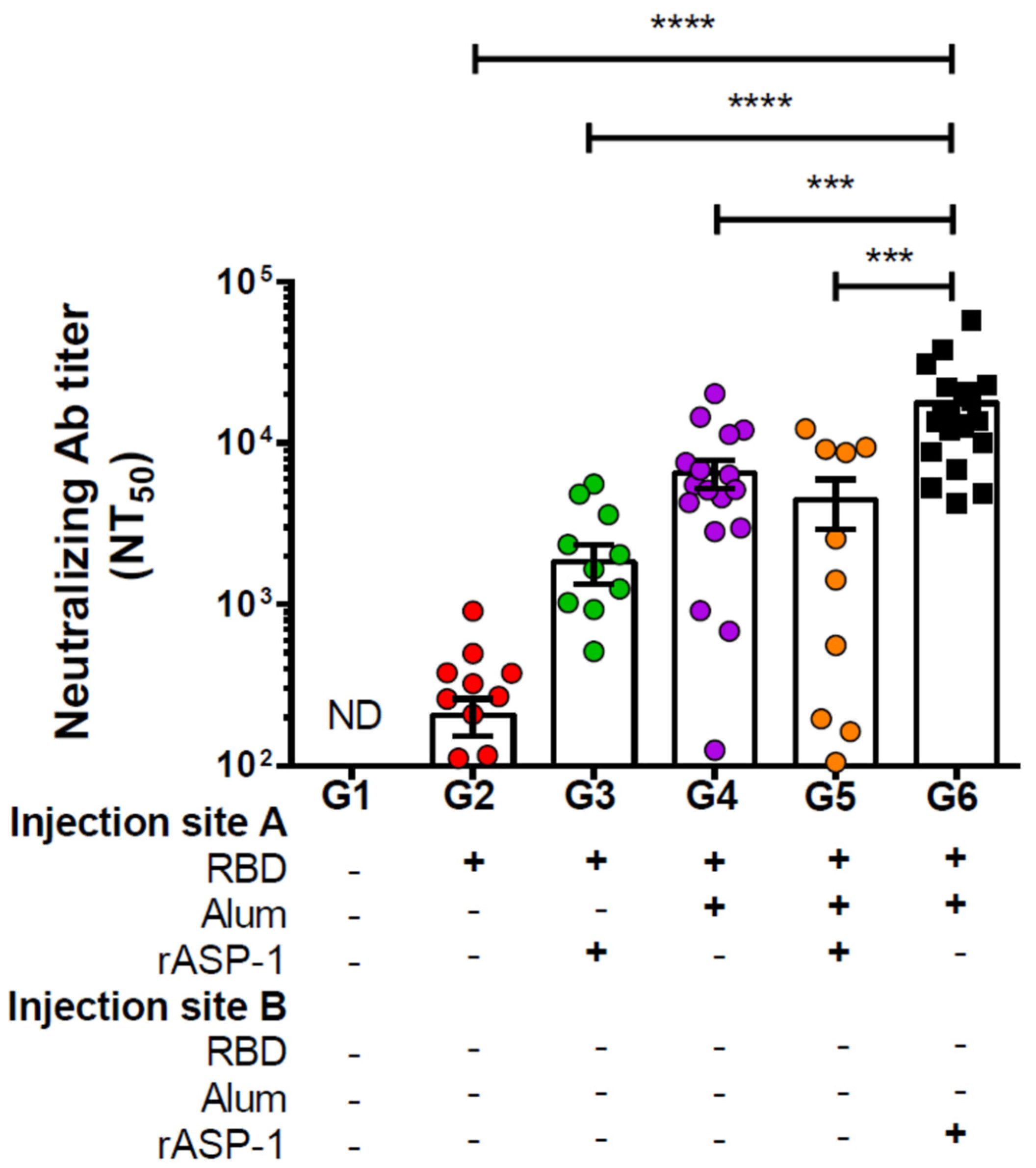
In this study, we have shown that mice immunized with RBD + rASP-1 + alum in a single inoculum elicited neutralizing antibody titers against pseudotyped MERS-CoV that were not significantly different from mice that received either rASP-1-adjuvanted RBD vaccine or alum-adjuvanted RBD vaccine alone (
Figure 1). Notably, when rASP-1 and the alum-adjuvanted RBD vaccine were co-administered in separate sites the vaccine ameliorated the production of neutralizing antibody titers by ~4-fold as compared to the combinatorial adjuvant system administered in a single inoculum (
Figure 1).
We also observed that mice that received two immunizations at three-week intervals of the combinatorial adjuvant system where rASP-1 and the alum-adjuvanted RBD vaccine were co-administered separately elicited neutralizing antibody titers against pseudotyped MERS-CoV infection that were similar to, or slightly lower than, those elicited in mice that received three immunizations of the Montanide ISA51-adjuvanted, or two immunizations of the MF59-adjuvanted MERS CoV-RBD-Fc or MERS-RBD-Fd vaccines [
20]. Another noteworthy observation is that the heightened neutralizing antibody titers induced by the unique experimental combinatorial adjuvant system was achieved using 5 µg of the MERS-RBD-Fd vaccine protein, compared to 10 µg of MERS-RBD-Fc or MERS-RBD-Fd proteins used in previous studies [
20]. This suggests that the rASP-1 in this unique combinatorial adjuvant system enabled also RBD dose sparing and with two immunizations. We have previously reported that rASP-1 also facilitates IIV3 antigen dose sparing up to a 10- or 40-fold decrease, and with a single immunization of the rASP-1-adjuvanted IIV3, mice were still protected from a lethal H1N1 influenza virus challenge [
12].
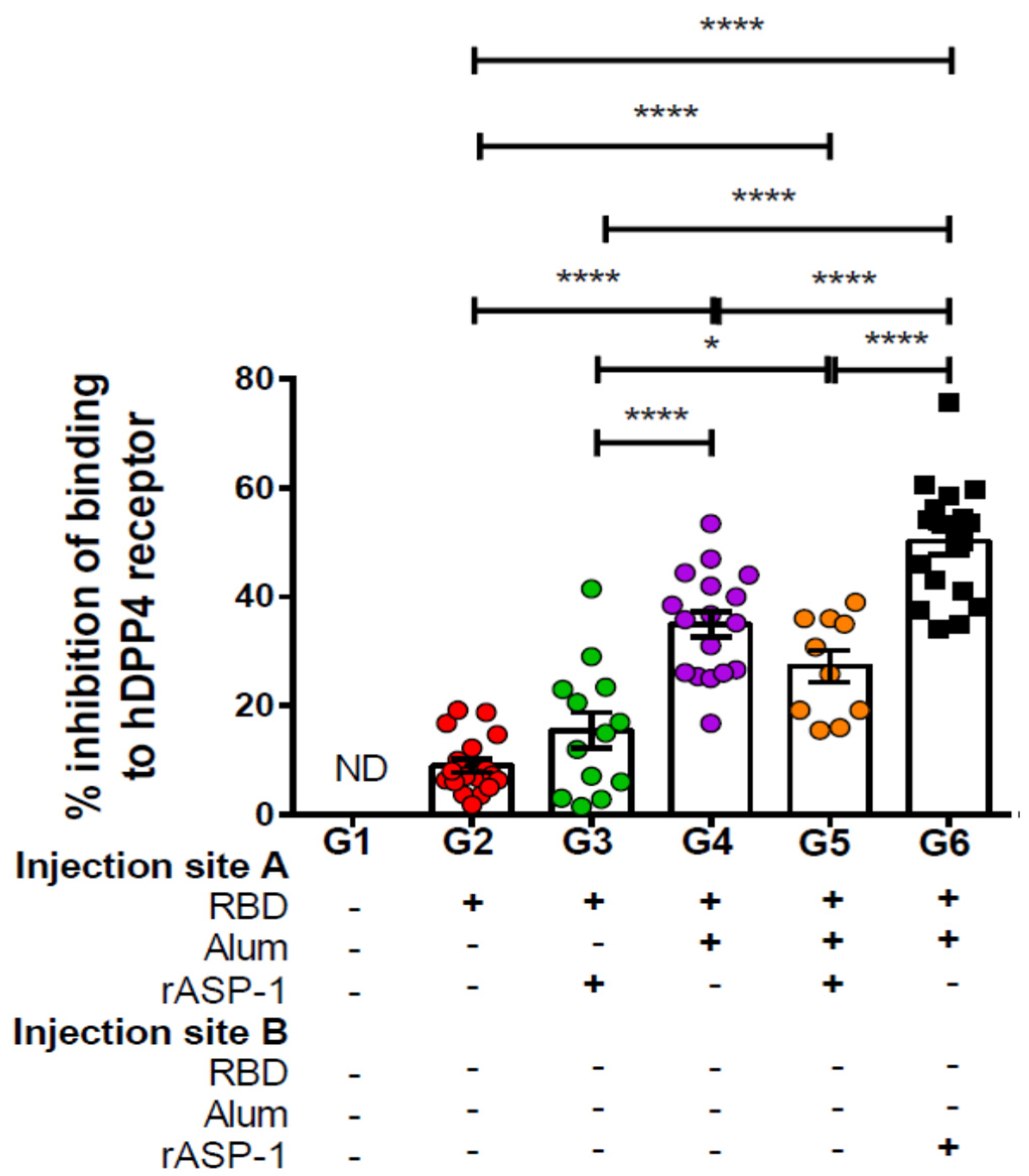
Importantly, the antibodies elicited by the different combinatorial RBD-adjuvanted vaccines were also functional in their ability to inhibit the binding of MERS-RBD to the human DPP4 receptor. Mice that received rASP-1 and the alum-adjuvanted RBD vaccine separately inhibited the binding by ~2-fold more as compared to mice administered with RBD rASP-1 + alum in a single inoculum (
Figure 2). Although mice that received alum-adjuvanted RBD vaccine significantly inhibited binding (35% ± 2.3) compared to rASP-1-adjuvanted RBD vaccine, they were not significantly different compared to the combinatorial adjuvant system administered in a single inoculum (
Figure 2). The inhibition of binding, however, was only enhanced in the combinatorial adjuvant system where rASP-1 and alum-adjuvanted RBD vaccine are co-administered separately. These data collectively suggest that adsorption of rASP-1 to alum in a combinatorial adjuvant system does not enhance the functional antibody responses elicited by alum-adjuvanted RBD vaccine. While rASP-1 when not adsorbed to alum in a combinatorial adjuvant system was able to ameliorate the functional antibody responses. An important concern raised when anti-viral vaccine are developed, especially with the ongoing COVID-19 crisis, is that some vaccine approaches may induce unwantedly adverse side effects due to antibody dependent enhancement (ADE) and thus more severe pathology [
37]. Since ADE is generally correlated to the neutralizing antibody titers, studies have also shown that high neutralizing antibody titer may eliminate the potential induction of ADE [
38,
39]. Previous studies on MERS-CoV have shown that MERS-CoV RBD-inducing neutralizing antibodies are positively correlated with protective immunity against MERS-CoV infection, and serum neutralizing antibody titers against live MERS-CoV infection of 1:40, 1:119, 1:141, or above (equivalent to NT
50 neutralizing antibody titer of about 1:1,000 against pseudotyped MERS-CoV infection) are associated with a protective immune response with no evidence of immunological toxicity or eosinophilic immune enhancement [
18,
19,
20,
40]. In our study, we show immunization of mice with rASP-1 and the alum-adjuvanted MERS-CoV RBD vaccine in separate sites have induced NT
50 neutralizing antibody titers greater than 1:15,000 against pseudotyped MERS-CoV infection. We expect that such high-titer neutralizing may prevent MERS-CoV infection in vivo without causing any adverse effects. However, this will have to be proven experimentally in the future.
In this study, we also observed that immunization with the alum-adjuvanted RBD vaccine elicits an RBD-specific IgG1-biased response, while the rASP-1-adjuvanted RBD vaccine elicits a balanced RBD specific IgG1-IgG2c response (
Figure S1B,C). Notably, in the combinatorial adjuvant system where rASP-1 and the alum-adjuvanted RBD vaccine are co-administered separately the balanced IgG1/IgG2c response (
Figure S1D) was preserved, while mice that received the combinatorial adjuvant system in a single inoculum elicited an IgG1-biased response (
Figure S1B,D). These results suggest that the presence of rASP-1 in an aqueous formulation shifts the dominant IgG1 response elicited by the alum-adjuvanted RBD vaccine to a balanced IgG1-IgG2c response and this was more pronounced when rASP-1 was not adsorbed to alum.
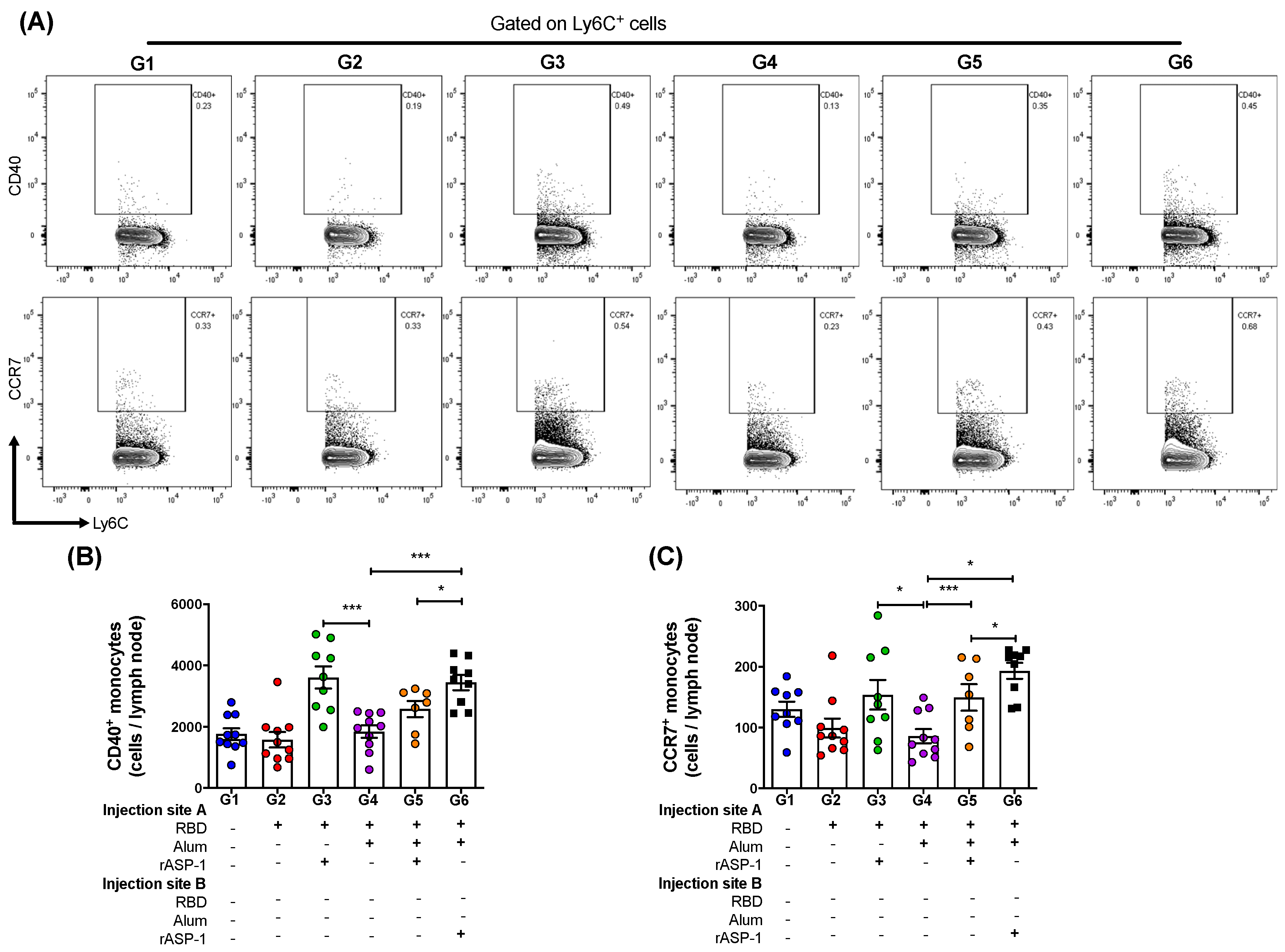
Interestingly, the administration of the combinatorial adjuvant system showed differences in the Ly6C
+ activated monocyte but not in CD11c
+ activated DC subsets (
Figure S3). This may likely be due to the presence of rASP-1 in the vaccine, since we have previously shown that intra-muscular injection rASP-1 alone or the rASP-1-adjuvanted trivalent influenza (IIV3) vaccine elicited an increased recruitment of monocytes than DCs at the site of injection (24 h after injection) as compared to PBS control group or IIV3 alone [
12]. In the present study, the administration of the combinatorial adjuvant system where rASP-1 was completely adsorbed to alum (single inoculum immunization group) significantly reduced the number of CD40
+ (activated) monocytes and CCR7
+ (migratory) monocytes to the draining LN compared to the administration of the combinatorial adjuvant system where rASP-1 was not adsorbed to alum (
Figure 3B,C). Interestingly, the number of CD80
+ monocytes in the LN was similar whether rASP-1 + alum + RBD were administered as a single inoculum or as a co-administered vaccine in two separate sites. However, both of these vaccine formulations as well as the rASP-1 adjuvanted-MERS-RBD vaccine resulted in significantly higher number of recruited CD80
+ monocytes than the alum-adjuvanted RBD vaccine, suggesting that the presence of alum did not significantly alter the number of CD80
+ monocytes recruited by the combinatorial rASP-1 and alum adjuvanted-MERS-RBD vaccines (
Figure S3B).
Moreover, there was a 2-fold increase in the number of the activated monocytes and migratory monocytes recruited to the draining LN in mice that received the rASP-1-adjuvanted RBD vaccine when compared to the alum-adjuvanted RBD vaccine (
Figure 3B,C). The number of migratory monocytes doubled in the draining LN of mice immunized with the combinatorial adjuvant system where RBD + rASP-1 + alum was administered in a single inoculum as compared to the alum-adjuvanted RBD vaccine alone. The number of migratory monocytes further increased in mice that received the combinatorial adjuvant system where rASP-1 and the alum-adjuvanted RBD vaccine were co-administered separately (
Figure 3C). There were no significant differences observed in the number of activated and migratory DCs across all the immunization groups. Collectively, these data suggest that the rASP-1 in the combinatorial adjuvant system may play a significant role in the enhanced recruitment of monocyte subsets. This is supported with the data where the administration of the rASP-1-adjuvanted RBD vaccine also significantly increased the number of activated (CD40
+) and migratory (CCR7
+) monocytes in the draining LN compared to alum-adjuvanted RBD vaccine (
Figure 3B,C). Also, rASP-1 and alum may work in synergy to improve the number of migratory monocytes in the draining LN compared to what alum could do alone.
One of the important events in the generation of an adaptive cellular response is the effective migration of innate cells to the lymph nodes to encounter naïve T cells, a process in which CCR7, a chemokine receptor, is known to play a dominant role [
41]. In addition, the absence of CCR7 has been shown to affect the magnitude of protective responses against viral infections in mouse models [
42,
43]. Therefore, we suggest that rASP-1, when not adsorbed to alum in a vaccine formulation, may improve the effective recruitment of innate cells that lead to the induction of effector adaptive cellular responses.
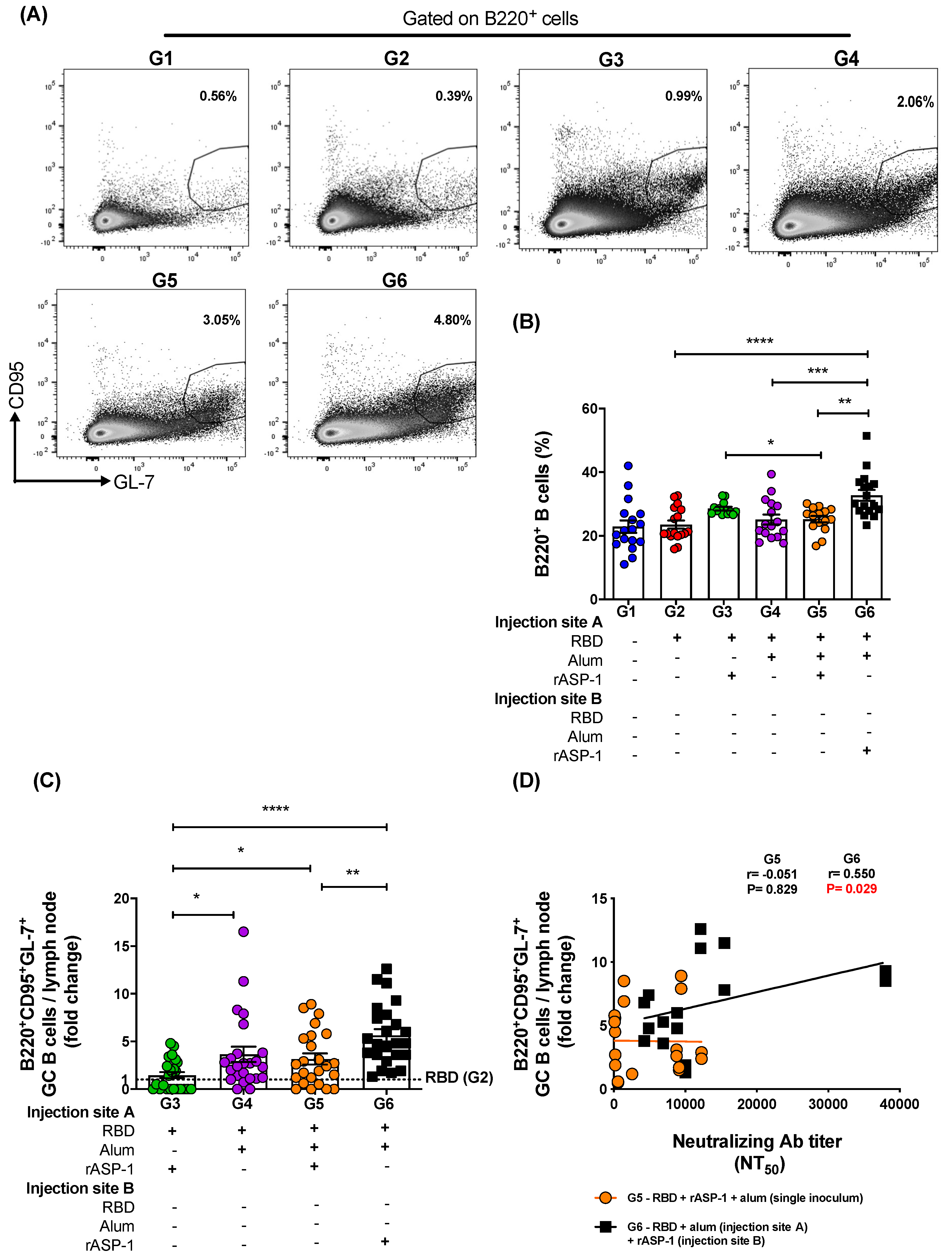
TfH cells can determine humoral immunity that is also derived from GC B cells, and therefore both of these cell types have become an important aspect for rational designs of more effective vaccines, in particular those depending on functional antibodies for their efficacy [
44,
45]. To better understand what contributed to the improved elicitation of functional anti-MERS-CoV neutralizing antibodies, the frequencies of TfH (CD4
+CXCR5
+PD-1
+) cells and GC B (B220
+CD95
+GL-7
+) cells in the draining LN of immunized mice were analyzed. A two-fold increase in both TfH and GC B frequencies were induced when rASP-1 and the alum-adjuvanted RBD vaccine were co-administered in separate sites as compared to the combinatorial adjuvant system where RBD + rASP-1 + alum were administered in a single inoculum (
Figure 4B and
Figure 5C). While no significant difference was observed in the fold increase of the frequency of GC B cells in the LN of mice that were immunized with rASP-1 and the alum-adjuvanted RBD vaccine co-administered separately as compared to the alum-adjuvanted RBD vaccine, a six-fold increase was observed when this was compared to rASP-1-adjuvanted RBD vaccine alone (
Figure 5C). These data suggest that the complete adsorption of rASP-1 to alum diminished not only the ability to induce migratory monocyte, but also the development of cells that are important for mounting an effective humoral response.
Importantly, we found a significant and positive correlation between the neutralizing antibody titers in sera of mice vaccinated with rASP-1 and the alum adjuvanted RBD vaccine separately and the fold increase in the frequency of TfH and GC B cells recruited in the draining LN (
Figure 4C and
Figure 5D). Interestingly, the fold increase in the frequency of TfH cells was also significantly and positively associated with the titers of neutralizing antibodies in mice that were immunized with the combinatorial adjuvant system administered in a single inoculum (RBD + rASP-1 + alum;
Figure 4B), suggesting that the rASP-1 and alum may work in synergy.