Recombinant Live Attenuated Influenza Vaccine Viruses Carrying Conserved T Cell Epitopes of Human Adenoviruses Induce Functional Cytotoxic T Cell Responses and Protect Mice against both Infections
Abstract
1. Introduction
2. Materials and Methods
2.1. Viruses and Peptides
2.1.1. Viruses
2.1.2. Peptides
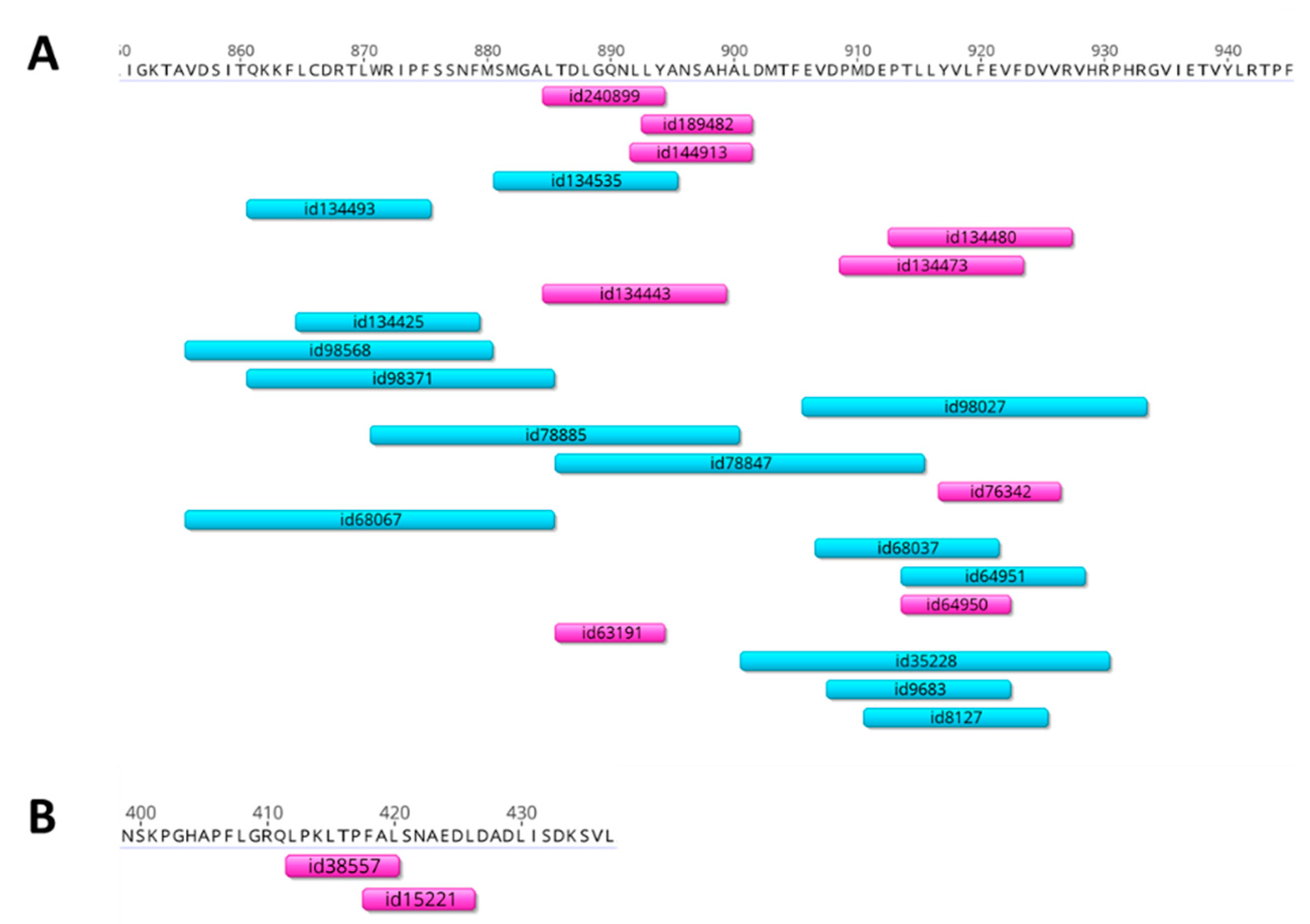
2.2. Selection of Adenovirus T-Cell Epitopes for Insertion into the LAIV Genome
2.3. Rescue of the Recombinant LAIV Viruses
2.4. In Vitro Studies
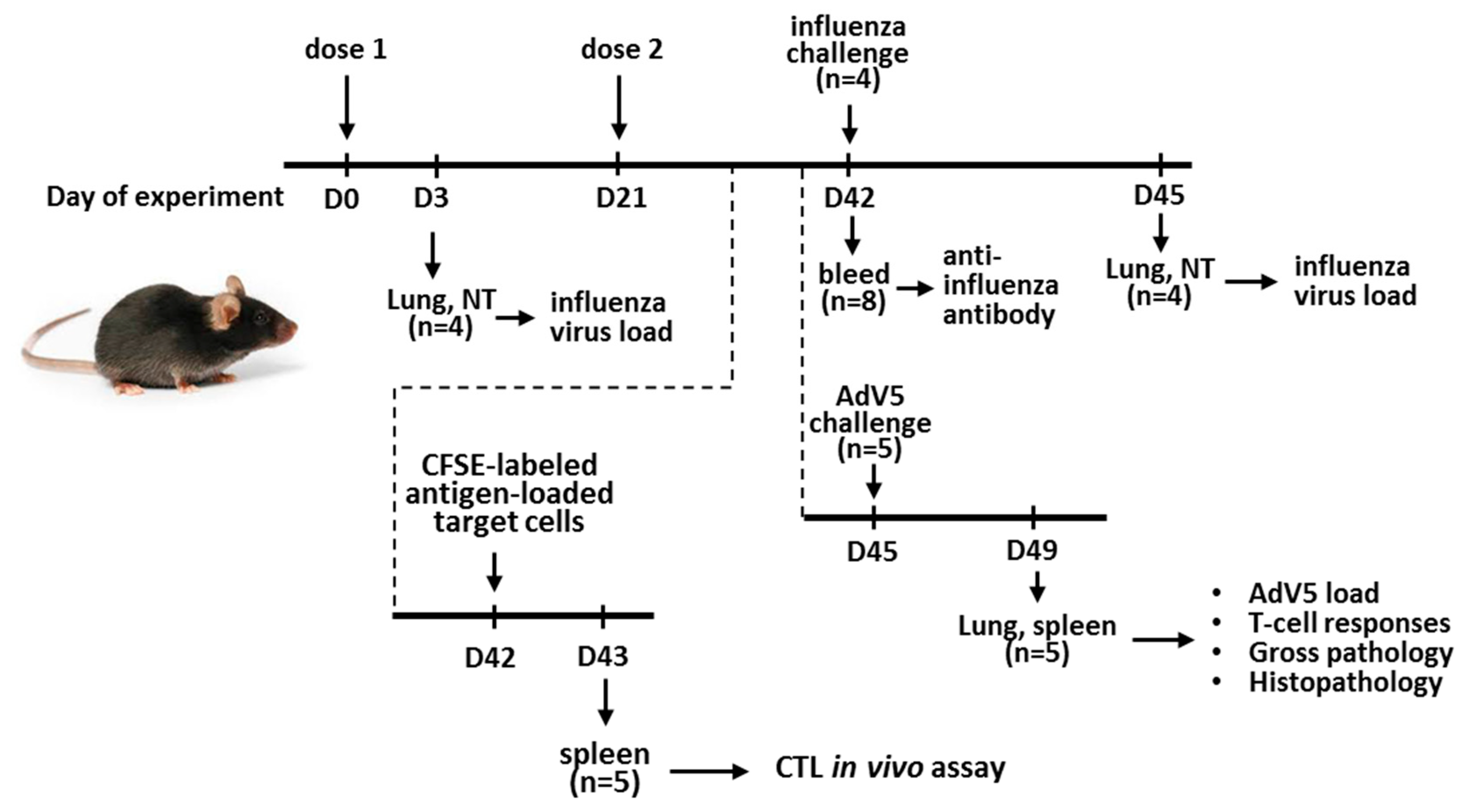
2.5. Mouse Immunization and Protection Studies
2.5.1. Ethical Statement
2.5.2. Mouse Immunization and LAIV Virus Replication in the Mouse Respiratory Tract
2.5.3. Protection against the Influenza Virus
2.5.4. Protection against the Adenovirus
2.6. Assessment of Immune Responses to the Recombinant Vaccines
2.6.1. Antibody Immune Responses
2.6.2. Cell-Mediated Immunity
2.6.3. CTL in vivo Assay
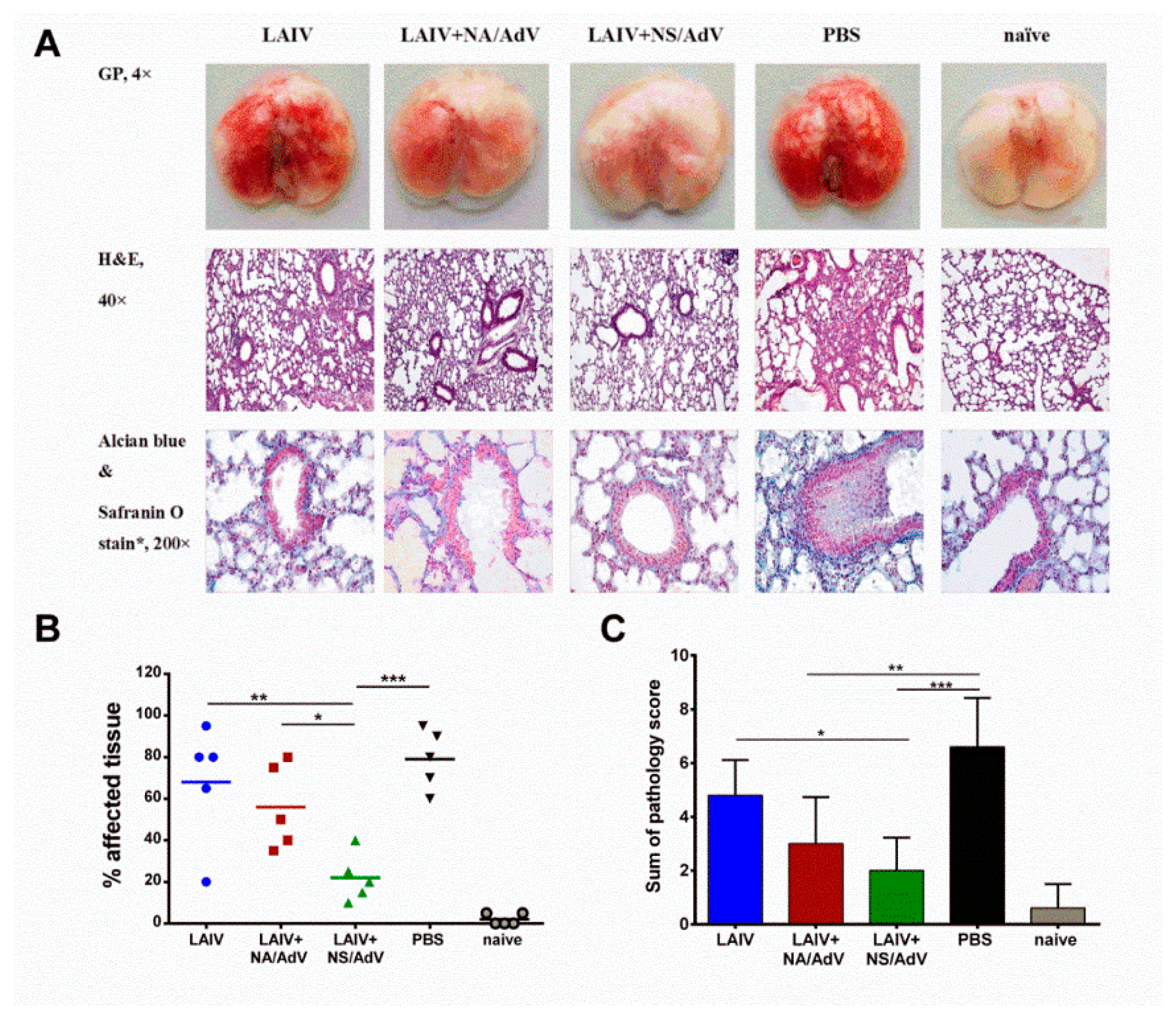
2.7. Histopathological Studies
2.8. Statistical Analyses
3. Results
3.1. Selection of Adenoviral T-Cell Epitopes for Designing the Recombinant LAIV-AdV Vaccines
3.2. Generation of Recombinant LAIV-AdV Viruses
3.3. Assessment of LAIV-AdV Virus Replication in vitro and in vivo
3.4. Anti-influenza Antibody Immune Responses and Protection
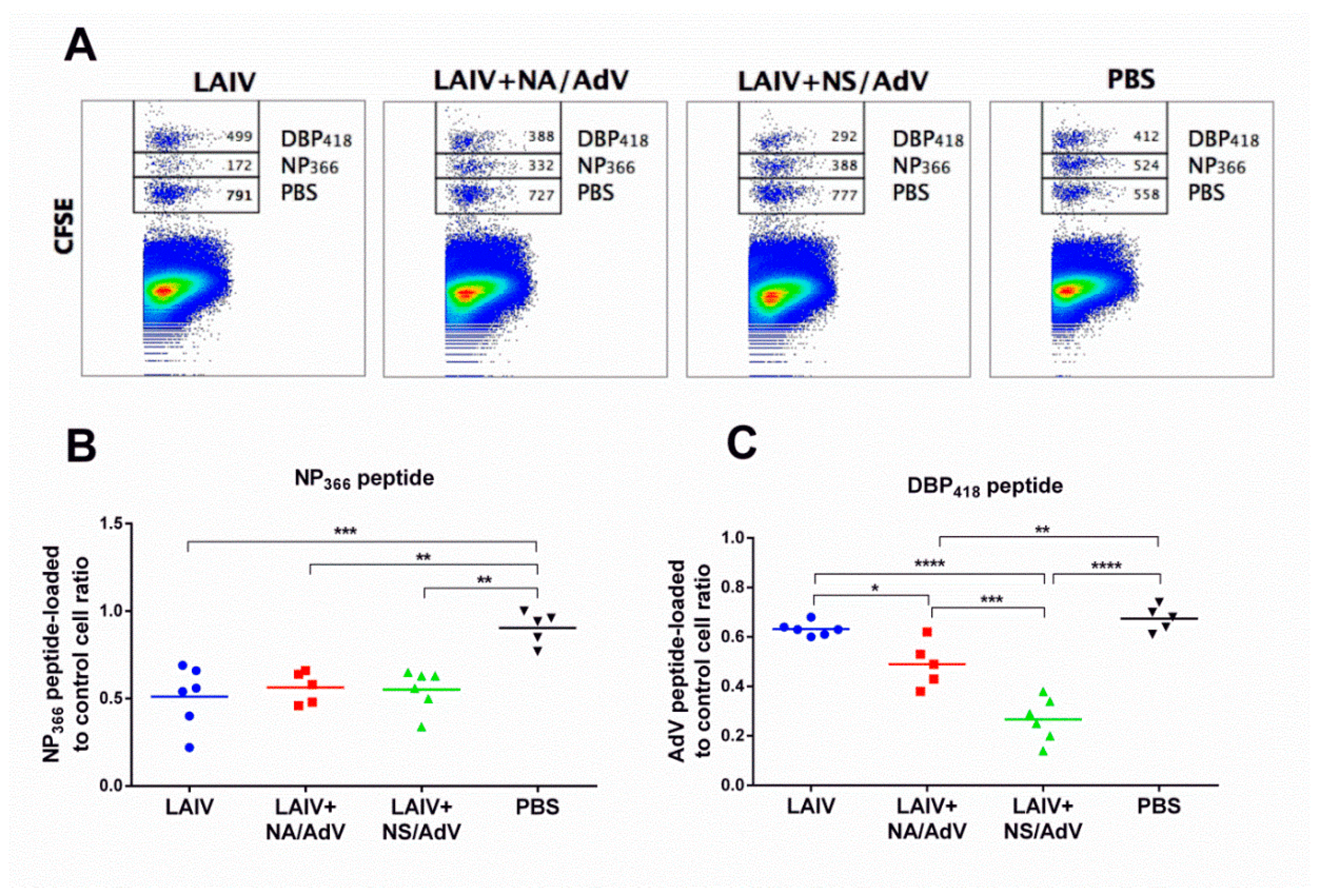
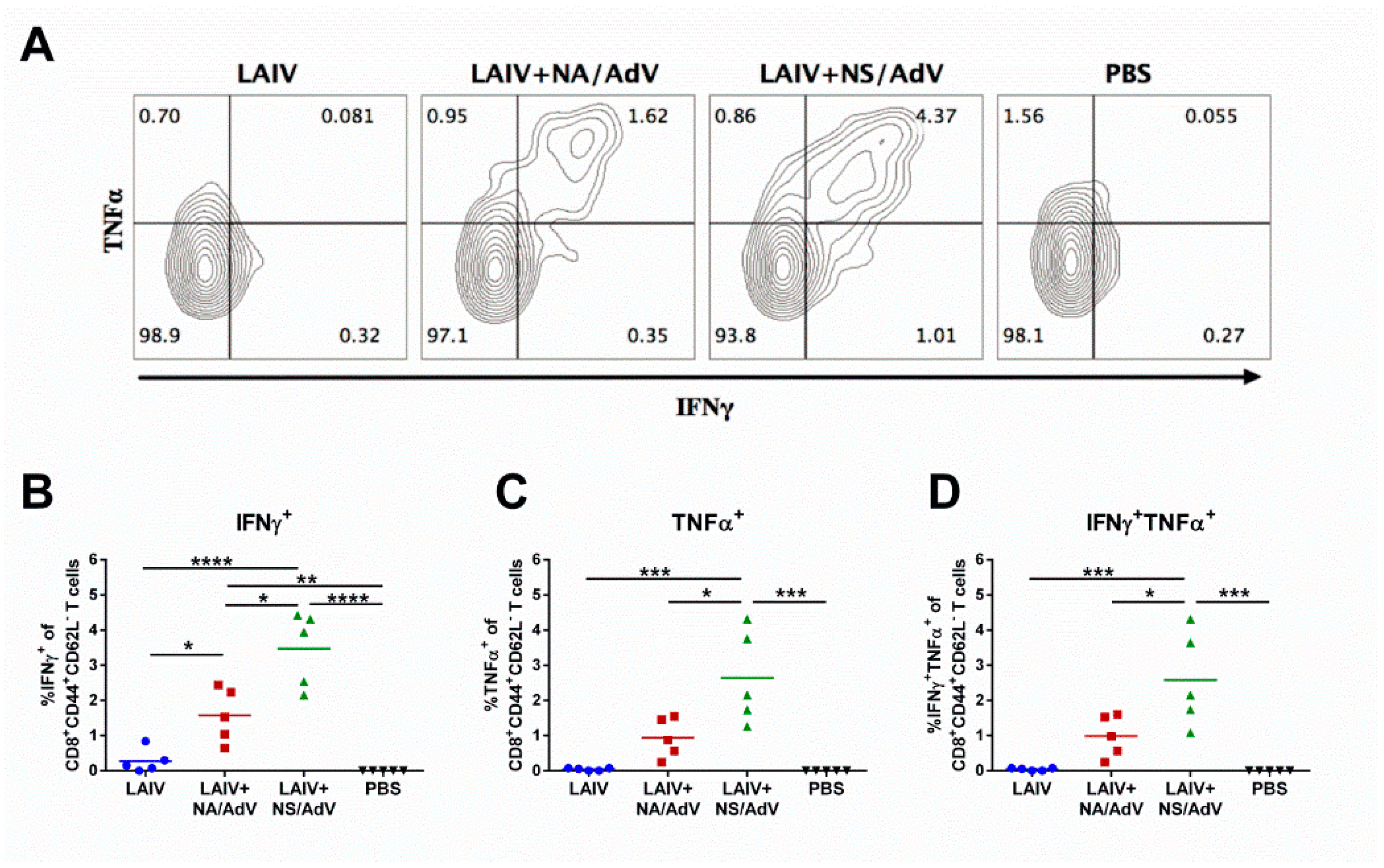
3.5. Functional Cytotoxic T-Cell Responses to Influenza and Adenovirus CTL Epitopes
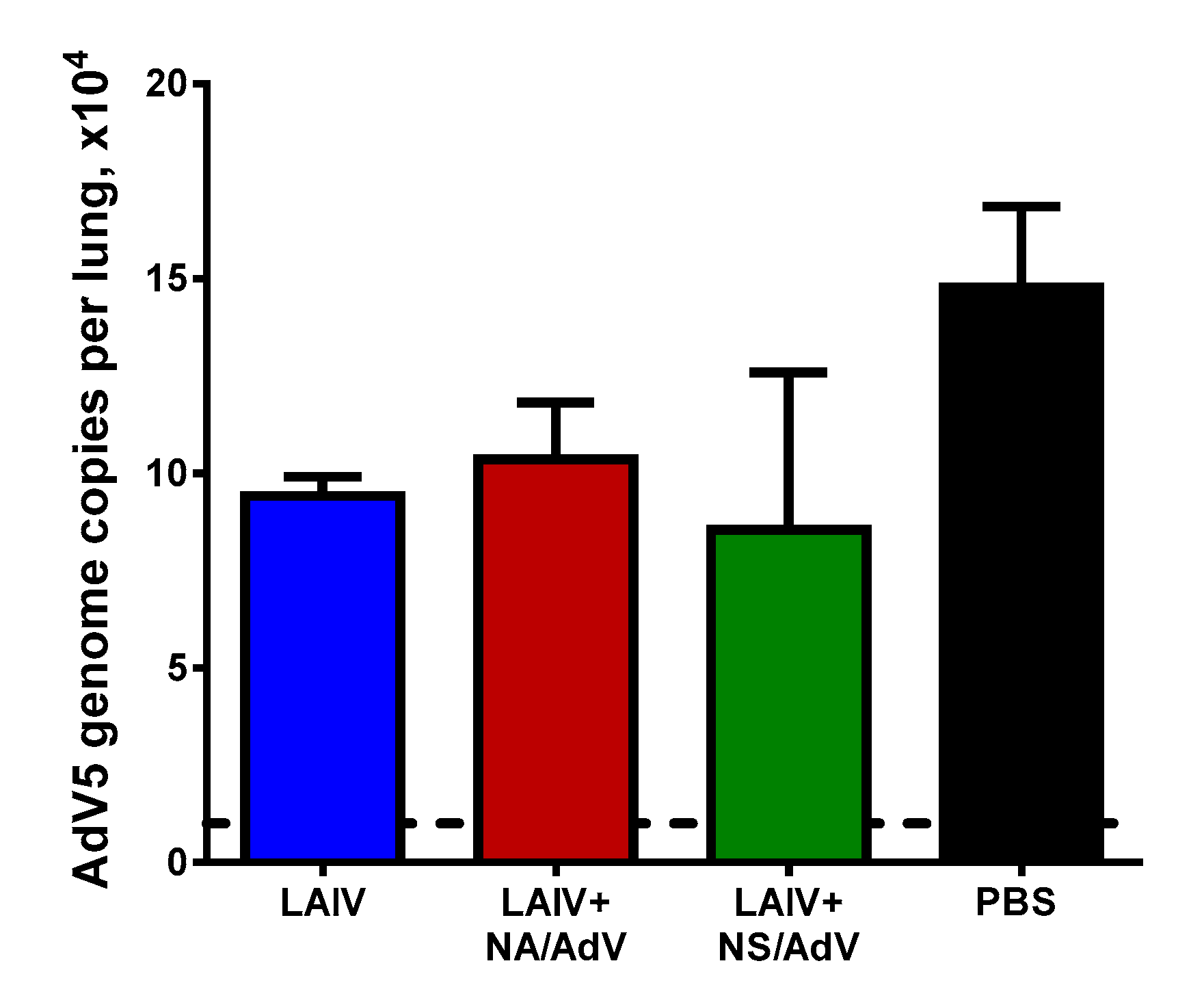
3.6. Protection against Wild-Type AdV5 Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Acronym list and glossary
| AdV | adenovirus |
| ARVI | acute respiratory viral infection |
| ATCC | American Type Culture Collection |
| AUC | area under the curve |
| BSA | bovine serum albumin |
| Ca | cold-adapted |
| CFSE | carboxyfluorescein succinimidyl ester dye |
| CPE | cytopathic effect |
| Ct | cycle of threshold |
| CTL | cytotoxic T lymphocyte |
| DBP | DNA-binding protein |
| DMEM | Dulbecco’s Modified Eagle Medium |
| EID50 | 50% egg infective dose |
| ELISA | enzyme-linked immunosorbent assay |
| ELISPOT | enzyme-linked immunospot assay |
| FBS | fetal bovine serum |
| GP | gross pathology |
| hAdV | human adenovirus |
| HAI | hemagglutination inhibition assay |
| H&E | hematoxylin and eosin |
| HLA | human leucocyte antigen |
| hMPV | human metapneumovirus |
| HPLC | high-performance liquid chromatography |
| IEDB | immune epitope database |
| IFNγ | interferon gamma |
| ICS | intracellular cytokine staining |
| i.n. | intranasal |
| i.v. | intravenous |
| LAIV | live attenuated influenza vaccine |
| LD50 | 50% mouse lethal dose |
| Len/17 | A/Leningrad/134/17/57 (H2N2) virus |
| MHC | major histocompatibility complex |
| NA | neuraminidase |
| NP | nucleoprotein |
| NS | non-structural protein |
| NT | nasal turbinates |
| OD | optical density |
| P2A | self-cleaving site from porcine teschovirus-1 |
| PBS | phosphate-buffered saline |
| PCR | polymerase chain reaction |
| PMA | phorbol myristate acetate |
| PR8 | A/PR/8/34 (H1N1) virus |
| RSV | respiratory syncytial virus |
| SH/13 | PR8-based reassortant virus bearing HA and NA from A/Shanghai/2/2013 (H7N9) |
| Tcm | central memory T cells |
| Tem | effector memory T cells |
| TNFα | tumor necrosis factor alpha |
| Ts | temperature sensitive |
References
- GBD 2016 Lower Respiratory Infections Collaborators. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet. Infect. Dis. 2018, 18, 1191–1210. [Google Scholar] [CrossRef]
- Zhao, H.; Green, H.; Lackenby, A.; Donati, M.; Ellis, J.; Thompson, C.; Bermingham, A.; Field, J.; Sebastianpillai, P.; Zambon, M.; et al. A new laboratory-based surveillance system (Respiratory DataMart System) for influenza and other respiratory viruses in England: Results and experience from 2009 to 2012. Euro Surveill. Bull. Eur. Sur Les Mal. Transm. Eur. Commun. Dis. Bull. 2014, 19. [Google Scholar] [CrossRef]
- Taylor, S.; Lopez, P.; Weckx, L.; Borja-Tabora, C.; Ulloa-Gutierrez, R.; Lazcano-Ponce, E.; Kerdpanich, A.; Angel Rodriguez Weber, M.; Mascarenas de Los Santos, A.; Tinoco, J.C.; et al. Respiratory viruses and influenza-like illness: Epidemiology and outcomes in children aged 6 months to 10 years in a multi-country population sample. J. Infect. 2017, 74, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Khanal, S.; Ghimire, P.; Dhamoon, A.S. The Repertoire of Adenovirus in Human Disease: The Innocuous to the Deadly. Biomedicines 2018, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Hage, E.; Gerd Liebert, U.; Bergs, S.; Ganzenmueller, T.; Heim, A. Human mastadenovirus type 70: A novel, multiple recombinant species D mastadenovirus isolated from diarrhoeal faeces of a haematopoietic stem cell transplantation recipient. J. Gen. Virol. 2015, 96, 2734–2742. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, S.; Gonzalez, G.; Harada, S.; Oosako, H.; Hanaoka, N.; Hinokuma, R.; Fujimoto, T. Recombinant type Human mastadenovirus D85 associated with epidemic keratoconjunctivitis since 2015 in Japan. J. Med Virol. 2018, 90, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P., 3rd; Kajon, A.E. Adenovirus: Epidemiology, Global Spread of Novel Serotypes, and Advances in Treatment and Prevention. Semin. Respir. Crit. Care Med. 2016, 37, 586–602. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Tian, X. Vaccine development for human mastadenovirus. J. Thorac. Dis. 2018, 10, S2280–S2294. [Google Scholar] [CrossRef]
- Liu, M.A. Immunologic basis of vaccine vectors. Immunity 2010, 33, 504–515. [Google Scholar] [CrossRef]
- Isakova-Sivak, I.; Tretiak, T.; Rudenko, L. Cold-adapted influenza viruses as a promising platform for viral-vector vaccines. Expert Rev. Vaccines 2016, 15, 1241–1243. [Google Scholar] [CrossRef][Green Version]
- Yang, P.; Li, T.; Liu, N.; Gu, H.; Han, L.; Zhang, P.; Li, Z.; Wang, Z.; Zhang, S.; Wang, X. Recombinant influenza virus carrying human adenovirus epitopes elicits protective immunity in mice. Antivir. Res. 2015, 121, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Gao, Y.; Zhou, S.; Sun, F.; Zhao, Z.; Wang, K.; Zhao, L.; Zhang, P.; Wang, Z.; Zhang, S.; et al. Bivalent vaccine platform based on ca influenza virus vaccine elicits protective immunity against human adenoviruses. Antivir. Res. 2018, 153, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Kotomina, T.; Isakova-Sivak, I.; Matyushenko, V.; Kim, K.H.; Lee, Y.; Jung, Y.J.; Kang, S.M.; Rudenko, L. Recombinant live attenuated influenza vaccine viruses carrying CD8 T-cell epitopes of respiratory syncytial virus protect mice against both pathogens without inflammatory disease. Antivir. Res. 2019, 168, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Kotomina, T.; Korenkov, D.; Matyushenko, V.; Prokopenko, P.; Rudenko, L.; Isakova-Sivak, I. Live attenuated influenza vaccine viral vector induces functional cytotoxic T-cell immune response against foreign CD8+ T-cell epitopes inserted into NA and NS1 genes using the 2A self-cleavage site. Hum. Vaccines Immunother. 2018, 14, 2964–2970. [Google Scholar] [CrossRef]
- Stepanova, E.A.; Kotomina, T.S.; Matyushenko, V.A.; Smolonogina, T.A.; Shapovalova, V.S.; Rudenko, L.G.; Isakova-Sivak, I.N. Amino Acid Substitutions N123D and N149D in Hemagglutinin Molecule Enhance Immunigenicity of Live Attenuated Influenza H7N9 Vaccine Strain in Experiment. Bull. Exp. Biol. Med. 2019, 166, 631–636. [Google Scholar] [CrossRef]
- Nasukawa, T.; Uchiyama, J.; Taharaguchi, S.; Ota, S.; Ujihara, T.; Matsuzaki, S.; Murakami, H.; Mizukami, K.; Sakaguchi, M. Virus purification by CsCl density gradient using general centrifugation. Arch. Virol. 2017, 162, 3523–3528. [Google Scholar] [CrossRef]
- Uchino, J.; Curiel, D.T.; Ugai, H. Species D human adenovirus type 9 exhibits better virus-spread ability for antitumor efficacy among alternative serotypes. PLoS ONE 2014, 9, e87342. [Google Scholar] [CrossRef]
- Dou, Y.; Li, Y.; Ma, C.; Zhu, H.; Du, J.; Liu, H.; Liu, Q.; Chen, R.; Tan, Y. Rapid diagnosis of human adenovirus B, C and E in the respiratory tract using multiplex quantitative polymerase chain reaction. Mol. Med. Rep. 2018, 18, 2889–2897. [Google Scholar] [CrossRef]
- Isakova-Sivak, I.; Chen, L.M.; Matsuoka, Y.; Voeten, J.T.; Kiseleva, I.; Heldens, J.G.; den Bosch, H.; Klimov, A.; Rudenko, L.; Cox, N.J.; et al. Genetic bases of the temperature-sensitive phenotype of a master donor virus used in live attenuated influenza vaccines: A/Leningrad/134/17/57 (H2N2). Virology 2011, 412, 297–305. [Google Scholar] [CrossRef]
- Ginsberg, H.S.; Moldawer, L.L.; Sehgal, P.B.; Redington, M.; Kilian, P.L.; Chanock, R.M.; Prince, G.A. A mouse model for investigating the molecular pathogenesis of adenovirus pneumonia. Proc. Natl. Acad. Sci. USA 1991, 88, 1651–1655. [Google Scholar] [CrossRef]
- Tas, J. The Alcian blue and combined Alcian blue--Safranin O staining of glycosaminoglycans studied in a model system and in mast cells. Histochem. J. 1977, 9, 205–230. [Google Scholar] [CrossRef] [PubMed]
- Pearse, A. Histochemistry. Theoretical and Applied; Churchill Livingstone: London, UK, 1968. [Google Scholar]
- Ginsberg, H.S.; Horswood, R.L.; Chanock, R.M.; Prince, G.A. Role of early genes in pathogenesis of adenovirus pneumonia. Proc. Natl. Acad. Sci. USA 1990, 87, 6191–6195. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, H.S.; Moldawer, L.L.; Prince, G.A. Role of the type 5 adenovirus gene encoding the early region 1B 55-kDa protein in pulmonary pathogenesis. Proc. Natl. Acad. Sci. USA 1999, 96, 10409–10411. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.H.; Wang, Y.C.; Jin, W.J.; Zhao, B.B.; Chen, C.F.; Yang, J.; Wang, J.F.; Guo, Y.Y.; Liu, J.J.; Zhang, D.; et al. Structure-based high-throughput epitope analysis of hexon proteins in B and C species human adenoviruses (HAdVs). PLoS ONE 2012, 7, e32938. [Google Scholar] [CrossRef]
- Davison, A.J.; Benko, M.; Harrach, B. Genetic content and evolution of adenoviruses. J. Gen. Virol. 2003, 84, 2895–2908. [Google Scholar] [CrossRef]
- Crawford-Miksza, L.; Schnurr, D.P. Analysis of 15 adenovirus hexon proteins reveals the location and structure of seven hypervariable regions containing serotype-specific residues. J. Virol. 1996, 70, 1836–1844. [Google Scholar] [CrossRef]
- Leen, A.M.; Christin, A.; Khalil, M.; Weiss, H.; Gee, A.P.; Brenner, M.K.; Heslop, H.E.; Rooney, C.M.; Bollard, C.M. Identification of hexon-specific CD4 and CD8 T-cell epitopes for vaccine and immunotherapy. J. Virol. 2008, 82, 546–554. [Google Scholar] [CrossRef]
- Tischer, S.; Geyeregger, R.; Kwoczek, J.; Heim, A.; Figueiredo, C.; Blasczyk, R.; Maecker-Kolhoff, B.; Eiz-Vesper, B. Discovery of immunodominant T-cell epitopes reveals penton protein as a second immunodominant target in human adenovirus infection. J. Transl. Med. 2016, 14, 286. [Google Scholar] [CrossRef]
- Keib, A.; Gunther, P.S.; Faist, B.; Halenius, A.; Busch, D.H.; Neuenhahn, M.; Jahn, G.; Dennehy, K.M. Presentation of a Conserved Adenoviral Epitope on HLA-C*0702 Allows Evasion of Natural Killer but Not T Cell Responses. Viral Immunol. 2017, 30, 149–156. [Google Scholar] [CrossRef]
- Gerdemann, U.; Keirnan, J.M.; Katari, U.L.; Yanagisawa, R.; Christin, A.S.; Huye, L.E.; Perna, S.K.; Ennamuri, S.; Gottschalk, S.; Brenner, M.K.; et al. Rapidly generated multivirus-specific cytotoxic T lymphocytes for the prophylaxis and treatment of viral infections. Mol. Ther. J. Am. Soc. Gene Ther. 2012, 20, 1622–1632. [Google Scholar] [CrossRef]
- Leen, A.M.; Sili, U.; Vanin, E.F.; Jewell, A.M.; Xie, W.; Vignali, D.; Piedra, P.A.; Brenner, M.K.; Rooney, C.M. Conserved CTL epitopes on the adenovirus hexon protein expand subgroup cross-reactive and subgroup-specific CD8+ T cells. Blood 2004, 104, 2432–2440. [Google Scholar] [CrossRef]
- Dasari, V.; Schuessler, A.; Smith, C.; Wong, Y.; Miles, J.J.; Smyth, M.J.; Ambalathingal, G.; Francis, R.; Campbell, S.; Chambers, D.; et al. Prophylactic and therapeutic adenoviral vector-based multivirus-specific T-cell immunotherapy for transplant patients. Mol. Ther. Methods Clin. Dev. 2016, 3, 16058. [Google Scholar] [CrossRef] [PubMed]
- Leen, A.M.; Sili, U.; Savoldo, B.; Jewell, A.M.; Piedra, P.A.; Brenner, M.K.; Rooney, C.M. Fiber-modified adenoviruses generate subgroup cross-reactive, adenovirus-specific cytotoxic T lymphocytes for therapeutic applications. Blood 2004, 103, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Lugthart, G.; Albon, S.J.; Ricciardelli, I.; Kester, M.G.; Meij, P.; Lankester, A.C.; Amrolia, P.J. Simultaneous generation of multivirus-specific and regulatory T cells for adoptive immunotherapy. J. Immunother. 2012, 35, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Chakupurakal, G.; Onion, D.; Bonney, S.; Cobbold, M.; Mautner, V.; Moss, P. HLA-peptide multimer selection of adenovirus-specific T cells for adoptive T-cell therapy. J. Immunother. 2013, 36, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Imahashi, N.; Nishida, T.; Ito, Y.; Kawada, J.; Nakazawa, Y.; Toji, S.; Suzuki, S.; Terakura, S.; Kato, T.; Murata, M.; et al. Identification of a novel HLA-A*24:02-restricted adenovirus serotype 11-specific CD8+ T-cell epitope for adoptive immunotherapy. Mol. Immunol. 2013, 56, 399–405. [Google Scholar] [CrossRef]
- Joshi, A.; Zhao, B.; Romanowski, C.; Rosen, D.; Flomenberg, P. Comparison of human memory CD8 T cell responses to adenoviral early and late proteins in peripheral blood and lymphoid tissue. PLoS ONE 2011, 6, e20068. [Google Scholar] [CrossRef][Green Version]
- Tang, J.; Olive, M.; Champagne, K.; Flomenberg, N.; Eisenlohr, L.; Hsu, S.; Flomenberg, P. Adenovirus hexon T-cell epitope is recognized by most adults and is restricted by HLA DP4, the most common class II allele. Gene Ther. 2004, 11, 1408–1415. [Google Scholar] [CrossRef]
- McKelvey, T.; Tang, A.; Bett, A.J.; Casimiro, D.R.; Chastain, M. T-cell response to adenovirus hexon and DNA-binding protein in mice. Gene Ther. 2004, 11, 791–796. [Google Scholar] [CrossRef][Green Version]
- Rollier, C.S.; Hill, A.V.S.; Reyes-Sandoval, A. Influence of adenovirus and MVA vaccines on the breadth and hierarchy of T cell responses. Vaccine 2016, 34, 4470–4474. [Google Scholar] [CrossRef]
- Schirmbeck, R.; Reimann, J.; Kochanek, S.; Kreppel, F. The immunogenicity of adenovirus vectors limits the multispecificity of CD8 T-cell responses to vector-encoded transgenic antigens. Mol. Ther. J. Am. Soc. Gene Ther. 2008, 16, 1609–1616. [Google Scholar] [CrossRef] [PubMed]
- Schone, D.; Hrycak, C.P.; Windmann, S.; Lapuente, D.; Dittmer, U.; Tenbusch, M.; Bayer, W. Immunodominance of Adenovirus-Derived CD8(+) T Cell Epitopes Interferes with the Induction of Transgene-Specific Immunity in Adenovirus-Based Immunization. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Price, G.E.; Soboleski, M.R.; Lo, C.Y.; Misplon, J.A.; Quirion, M.R.; Houser, K.V.; Pearce, M.B.; Pappas, C.; Tumpey, T.M.; Epstein, S.L. Single-dose mucosal immunization with a candidate universal influenza vaccine provides rapid protection from virulent H5N1, H3N2 and H1N1 viruses. PLoS ONE 2010, 5, e13162. [Google Scholar] [CrossRef] [PubMed]
- Talon, J.; Salvatore, M.; O’Neill, R.E.; Nakaya, Y.; Zheng, H.; Muster, T.; Garcia-Sastre, A.; Palese, P. Influenza A and B viruses expressing altered NS1 proteins: A vaccine approach. Proc. Natl. Acad. Sci. USA 2000, 97, 4309–4314. [Google Scholar] [CrossRef]
- Steel, J.; Lowen, A.C.; Pena, L.; Angel, M.; Solorzano, A.; Albrecht, R.; Perez, D.R.; Garcia-Sastre, A.; Palese, P. Live attenuated influenza viruses containing NS1 truncations as vaccine candidates against H5N1 highly pathogenic avian influenza. J. Virol. 2009, 83, 1742–1753. [Google Scholar] [CrossRef]
- Zhou, B.; Li, Y.; Belser, J.A.; Pearce, M.B.; Schmolke, M.; Subba, A.X.; Shi, Z.; Zaki, S.R.; Blau, D.M.; Garcia-Sastre, A.; et al. NS-based live attenuated H1N1 pandemic vaccines protect mice and ferrets. Vaccine 2010, 28, 8015–8025. [Google Scholar] [CrossRef]
- Ferko, B.; Stasakova, J.; Sereinig, S.; Romanova, J.; Katinger, D.; Niebler, B.; Katinger, H.; Egorov, A. Hyperattenuated recombinant influenza A virus nonstructural-protein-encoding vectors induce human immunodeficiency virus type 1 Nef-specific systemic and mucosal immune responses in mice. J. Virol. 2001, 75, 8899–8908. [Google Scholar] [CrossRef][Green Version]
- Isakova-Sivak, I.; Korenkov, D.; Smolonogina, T.; Tretiak, T.; Donina, S.; Rekstin, A.; Naykhin, A.; Shcherbik, S.; Pearce, N.; Chen, L.M.; et al. Comparative studies of infectivity, immunogenicity and cross-protective efficacy of live attenuated influenza vaccines containing nucleoprotein from cold-adapted or wild-type influenza virus in a mouse model. Virology 2017, 500, 209–217. [Google Scholar] [CrossRef]
- Sun, J.; Braciale, T.J. Role of T cell immunity in recovery from influenza virus infection. Curr. Opin. Virol. 2013, 3, 425–429. [Google Scholar] [CrossRef]
- Isakova-Sivak, I.; Rudenko, L. Safety, immunogenicity and infectivity of new live attenuated influenza vaccines. Expert Rev. Vaccines 2015, 14, 1313–1329. [Google Scholar] [CrossRef]
- Liu, M.A. Gene-based vaccines: Recent developments. Curr. Opin. Mol. 2010, 12, 86–93. [Google Scholar]
- Rolland, M.; Nickle, D.C.; Mullins, J.I. HIV-1 group M conserved elements vaccine. PLoS Pathog. 2007, 3, e157. [Google Scholar] [CrossRef] [PubMed]
- Wold, W.S.; Toth, K. Adenovirus vectors for gene therapy, vaccination and cancer gene therapy. Curr. Gene Ther. 2013, 13, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Thacker, E.E.; Timares, L.; Matthews, Q.L. Strategies to overcome host immunity to adenovirus vectors in vaccine development. Expert Rev. Vaccines 2009, 8, 761–777. [Google Scholar] [CrossRef] [PubMed]
- Haye, K.; Burmakina, S.; Moran, T.; Garcia-Sastre, A.; Fernandez-Sesma, A. The NS1 protein of a human influenza virus inhibits type I interferon production and the induction of antiviral responses in primary human dendritic and respiratory epithelial cells. J. Virol. 2009, 83, 6849–6862. [Google Scholar] [CrossRef] [PubMed]
- Vasilyev, K.; Yukhneva, M.; Shurygina, A.-P.; Stukova, M.; Egorov, A. Enhancement of the immunogenicity of influenza A virus by the inhibition of immunosuppressive function of NS1 protein. Proteins 2018, 5, 48–58. [Google Scholar] [CrossRef]
- Korenkov, D.; Isakova-Sivak, I.; Rudenko, L. Basics of CD8 T-cell immune responses after influenza infection and vaccination with inactivated or live attenuated influenza vaccine. Expert Rev. Vaccines 2018, 17, 977–987. [Google Scholar] [CrossRef]
- Roberts, A.D.; Ely, K.H.; Woodland, D.L. Differential contributions of central and effector memory T cells to recall responses. J. Exp. Med. 2005, 202, 123–133. [Google Scholar] [CrossRef]
- Dong, W.; Bhide, Y.; Sicca, F.; Meijerhof, T.; Guilfoyle, K.; Engelhardt, O.G.; Boon, L.; de Haan, C.A.M.; Carnell, G.; Temperton, N.; et al. Cross-Protective Immune Responses Induced by Sequential Influenza Virus Infection and by Sequential Vaccination with Inactivated Influenza Vaccines. Front. Immunol. 2018, 9, 2312. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isakova-Sivak, I.; Matyushenko, V.; Stepanova, E.; Matushkina, A.; Kotomina, T.; Mezhenskaya, D.; Prokopenko, P.; Kudryavtsev, I.; Kopeykin, P.; Sivak, K.; et al. Recombinant Live Attenuated Influenza Vaccine Viruses Carrying Conserved T Cell Epitopes of Human Adenoviruses Induce Functional Cytotoxic T Cell Responses and Protect Mice against both Infections. Vaccines 2020, 8, 196. https://doi.org/10.3390/vaccines8020196
Isakova-Sivak I, Matyushenko V, Stepanova E, Matushkina A, Kotomina T, Mezhenskaya D, Prokopenko P, Kudryavtsev I, Kopeykin P, Sivak K, et al. Recombinant Live Attenuated Influenza Vaccine Viruses Carrying Conserved T Cell Epitopes of Human Adenoviruses Induce Functional Cytotoxic T Cell Responses and Protect Mice against both Infections. Vaccines. 2020; 8(2):196. https://doi.org/10.3390/vaccines8020196
Chicago/Turabian StyleIsakova-Sivak, Irina, Victoria Matyushenko, Ekaterina Stepanova, Anastasia Matushkina, Tatiana Kotomina, Daria Mezhenskaya, Polina Prokopenko, Igor Kudryavtsev, Pavel Kopeykin, Konstantin Sivak, and et al. 2020. "Recombinant Live Attenuated Influenza Vaccine Viruses Carrying Conserved T Cell Epitopes of Human Adenoviruses Induce Functional Cytotoxic T Cell Responses and Protect Mice against both Infections" Vaccines 8, no. 2: 196. https://doi.org/10.3390/vaccines8020196
APA StyleIsakova-Sivak, I., Matyushenko, V., Stepanova, E., Matushkina, A., Kotomina, T., Mezhenskaya, D., Prokopenko, P., Kudryavtsev, I., Kopeykin, P., Sivak, K., & Rudenko, L. (2020). Recombinant Live Attenuated Influenza Vaccine Viruses Carrying Conserved T Cell Epitopes of Human Adenoviruses Induce Functional Cytotoxic T Cell Responses and Protect Mice against both Infections. Vaccines, 8(2), 196. https://doi.org/10.3390/vaccines8020196





