Impact of Maternal Immunity on Congenital Cytomegalovirus Birth Prevalence and Infant Outcomes: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
3. Results
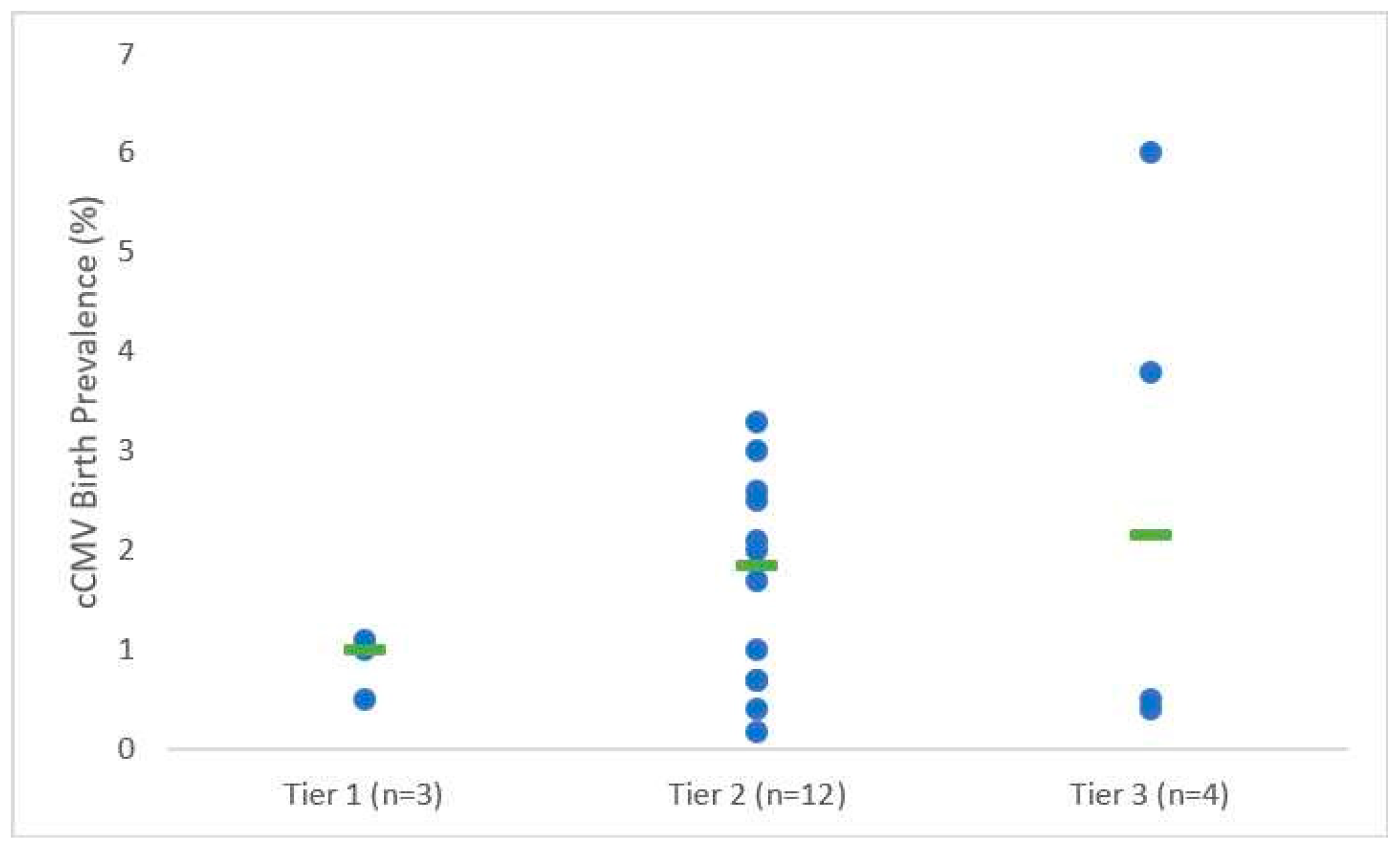
3.1. cCMV Birth Prevalence and Transmission Rates among Highly Seroprevalent Maternal Populations
3.1.1. Tier 1 Studies
3.1.2. Tier 2 Studies
3.1.3. Tier 3 Studies
3.2. Sequelae of cCMV Infection Among Infants Born to CMV Seropositive Mothers in Highly Seroprevalent Populations
3.3. Hearing Loss among cCMV-Infected Infants Born to CMV Seropositive Mothers in Highly Seroprevalent Populations
3.4. Prematurity Among cCMV-Infected Infants Born to CMV Seropositive Mothers in Highly Seroprevalent Populations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Permar, S.R.; Schleiss, M.R.; Plotkin, S.A. Advancing Our Understanding of Protective Maternal Immunity as a Guide for Development of Vaccines to Reduce Congenital Cytomegalovirus Infections. J. Virol. 2018, 92, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhang, X.; Bialek, S.; Cannon, M.J. Attribution of congenital cytomegalovirus infection to primary versus non-primary maternal infection. Clin. Infect. Dis 2011, 52, e11–e13. [Google Scholar] [CrossRef] [PubMed]
- Giannattasio, A.; Di Costanzo, P.; De Matteis, A.; Milite, P.; De Martino, D.; Bucci, L.; Augurio, M.R.; Bravaccio, C.; Ferrara, T.; Capasso, L.; et al. Outcomes of congenital cytomegalovirus disease following maternal primary and non-primary infection. J. Clin. Virol. 2017, 96, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Van Zuylen, W.J.; Hamilton, S.T.; Naing, Z.; Hall, B.; Shand, A.; Rawlinson, W.D. Congenital cytomegalovirus infection: Clinical presentation, epidemiology, diagnosis and prevention. Obstet Med. 2014, 7, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Bialas, K.M.; Permar, S.R. The March towards a Vaccine for Congenital CMV: Rationale and Models. PLoS Pathog. 2016, 12, e1005355. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kenneson, A.; Cannon, M.J. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev. Med. Virol. 2007, 17, 253–276. [Google Scholar] [CrossRef] [PubMed]
- Fowler, K.B.; Boppana, S.B. Congenital cytomegalovirus infection. Semin. Perinatol. 2018, 42, 149–154. [Google Scholar] [CrossRef]
- Britt, W.J. Congenital Human Cytomegalovirus Infection and the Engima of Maternal Immunity. J. Virol. 2017, 91, 1–7. [Google Scholar] [CrossRef]
- Marsico, C.; Kimberlin, D.W. Congenital Cytomegalovirus infection: Advances and challenges in diagnosis, prevention and treatment. Ital. J. Pediatr. 2017, 43, 38. [Google Scholar] [CrossRef]
- Boppana, S.B.; Ross, S.A.; Fowler, K.B. Congenital Cytomegalovirus Infection: Clinical Outcome. Clin. Infect. Dis. 2013, 57, S178–S181. [Google Scholar] [CrossRef]
- Schleiss, M.R.; Permar, S.R.; Plotkin, S.A. Progress toward Development of a Vaccine against Congenital Cytomegalovirus Infection. Clin. Vaccine Immunol. 2017, 24, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Fowler, K.B.; Stagno, S.; Pass, R.F.; Britt, W.J.; Boll, T.J.; Alford, C.A. The Outcome of Congenital Cytomegalovirus Infection in Relation to Maternal Antibody Status. N. Engl. J. Med. 1992, 326, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.A.; Arora, N.; Novak, Z.; Fowler, K.B.; Britt, W.J.; Boppana, S.B. Cytomegalovirus reinfections in healthy seroimmune women. J. Infect. Dis. 2010, 201, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, A.Y.; Mussi-Pinhata, M.M.; Boppana, S.B.; Novak, Z.; Wagatsuma, V.M.; Oliveira, P.D.F.; Duarte, G.; Britt, W.J. Human cytomegalovirus reinfection is associated with intrauterine transmission in a highly cytomegalovirus-immune maternal population. Am. J. Obstet. Gynecol. 2010, 202, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Simonazzi, G.; Curti, A.; Cervi, F.; Gabrielli, L.; Contoli, M.; Capretti, M.G.; Rizzo, N.; Guerra, B.; Farina, A.; Lazzarotto, T. Perinatal Outcomes of Non-Primary Maternal Cytomegalovirus Infection: A 15-Year Experience. Fetal Diagn. Ther. 2018, 43, 138–142. [Google Scholar] [CrossRef]
- Davis, N.L.; King, C.C.; Kourtis, A.P. Cytomegalovirus infection in pregnancy. Birth Defects Res. 2017, 109, 336–346. [Google Scholar] [CrossRef]
- Johnson, J.; Anderson, B. Cytomegalovirus: Should We Screen Pregnant Women for Primary Infection? Am. J. Perinatol. 2013, 30, 121–124. [Google Scholar] [PubMed]
- Stagno, S.; Pass, R.F.; Cloud, G.; Britt, W.J.; Henderson, R.E.; Walton, P.D.; Veren, D.A.; Page, F.; Alford, C.A. Primary cytomegalovirus infection in pregnancy. Incidence, transmission to fetus, and clinical outcome. JAMA 1986, 256, 1904–1908. [Google Scholar] [CrossRef]
- Mussi-Pinhata, M.M.; Yamamoto, A.Y.; Aragon, D.C.; Duarte, G.; Fowler, K.B.; Boppana, S.; Britt, W.J. Seroconversion for Cytomegalovirus Infection During Pregnancy and Fetal Infection in a Highly Seropositive Population: “The BraCHS Study”. J. Infect. Dis. 2018, 218, 1200–1204. [Google Scholar] [CrossRef]
- Leruez-Ville, M.; Magny, J.F.; Couderc, S.; Pichon, C.; Parodi, M.; Bussieres, L.; Guilleminot, T.; Ghout, I.; Ville, Y. Risk factors for congenital cytomegalovirus infection following primary and non-primary maternal infection: A prospective neonatal screening study using polymerase chain reaction in saliva. Clin. Infect. Dis. 2017, 65, 398–404. [Google Scholar] [CrossRef]
- Hughes, B.L.; Gyamfi-Bannerman, C. Diagnosis and antenatal management of congenital cytomegalovirus infection. Am. J. Obstet. Gynecol. 2016, 214, B5–B11. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Executive Summary. In Vaccines for the 21st Century: A Tool for Decisionmaking; Stratton, K.R., Durch, J.S., Lawrence, R.S., Eds.; The National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Lanzieri, T.M.; Dollard, S.C.; Bialek, S.R.; Grosse, S.D. Systematic review of the birth prevalence of congenital cytomegalovirus infection in developing countries. Int. J. Infect. Dis. 2014, 22, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [PubMed]
- Revello, M.G.; Gerna, G. Diagnosis and Management of Human Cytomegalovirus Infection in the Mother, Fetus, and Newborn Infant. Clin. Microbiol. Rev. 2002, 15, 680–715. [Google Scholar] [CrossRef]
- Yamamoto, A.Y.; Mussi-Pinhata, M.M.; Isaac, M.D.L.; Amaral, F.R.; Carvalheiro, C.G.; Aragon, D.C.; Manfredi, A.K.D.S.; Boppana, S.B.; Britt, W.J. Congenital cytomegalovirus infection as a cause of sensorineural hearing loss in a highly immune population. Pediatr. Infect. Dis. J. 2011, 30, 1043–1046. [Google Scholar] [CrossRef]
- Mussi-Pinhata, M.M.; Yamamoto, A.Y.; Brito, R.M.M.; Isaac, M.D.L.; Oliveira, P.F.D.C.E.; Boppana, S.; Britt, W.J.; Britto, R.M.M. Birth prevalence and natural history of congenital cytomegalovirus infection in a highly seroimmune population. Clin. Infect. Dis. 2009, 49, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Arellano-Galindo, J.; Villanueva-García, D.; Cruz-Ramirez, J.L.; Yalaupari-Mejìa, J.P.; Uribe-Gutiérrez, G.; Velazquez-Guadarrama, N.; Nava-Frias, M.; Munoz-Hernández, O.; Mejía-Arangure, J.M. Detection and gB genotyping of CMV in Mexican preterm infants in the context of maternal seropositivity. J. Infect. Dev. Ctries 2014, 8, 758–767. [Google Scholar] [CrossRef][Green Version]
- Mussi-Pinhata, M.M.; Pinto, P.C.G.; Jorge, S.M.; Yamamoto, A.Y.; Figueiredo, L.T.M. Congenital cytomegalovirus infection in preterm and full-term newborn infants from a population with a high seroprevalence rate. Pediatr. Infect. Dis. J. 2001, 20, 188–192. [Google Scholar]
- Wang, S.; Wang, T.; Zhang, W.; Liu, X.; Wang, X.; Wang, H.; He, X.; Zhang, S.; Xu, S.; Yu, Y.; et al. Cohort study on maternal cytomegalovirus seroprevalence and prevalence and clinical manifestations of congenital infection in China. Medicine 2017, 96, e6007. [Google Scholar] [CrossRef]
- Barbi, M.; Binda, S.; Caroppo, S.; Calvario, A.; Germinario, C.A.; Bozzi, A.; Tanzi, M.L.; Veronesi, L.; Mura, I.; Piana, A.; et al. Multicity Italian Study of Congenital Cytomegalovirus Infection. Pediatr. Infect. Dis. J. 2006, 25, 156–159. [Google Scholar] [CrossRef][Green Version]
- Kaneko, M.; Sameshima, H.; Minematsu, T.; Kusumoto, K.; Yamauchi, A.; Ikenoue, T. Maternal IgG avidity, IgM and ultrasound abnormalities: Combined method to detect congenital cytomegalovirus infection with sequelae. J. Perinatol. 2013, 33, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Madrid, L.; Varo, R.; Maculuve, S.; Nhampossa, T.; Muñoz-Almagro, C.; Calderón, E.J.; Esteva, C.; Carrilho, C.; Ismail, M.; Vieites, B.; et al. Congenital cytomegalovirus, parvovirus and enterovirus infection in Mozambican newborns at birth: A cross-sectional survey. PLoS ONE 2018, 13, e0194186. [Google Scholar] [CrossRef] [PubMed]
- Ziyaeyan, M.; Alborzi, A.; Abbasian, A.; Kalani, M.; Moravej, A.; Nasiri, J.; Amiri, A.; Hashemi, N.; Sefiddashti, F. Detection of HCMV DNA in placenta, amniotic fluid and fetuses of seropositive women by nested PCR. Eur. J. Pediatr. 2007, 166, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Fowler, K.B.; Stagno, S.; Pas, R.F. Maternal Immunity and Prevention of Congenital Cytomegalovirus Infection. Obstet. Gynecol. Surv. 2003, 58, 642–643. [Google Scholar] [CrossRef][Green Version]
- Stagno, S.; Dworsky, M.E.; Torres, J.; Mesa, T.; Hirsh, T. Prevalence and importance of congenital cytomegalovirus infection in three different populations. J. Pediatr. 1982, 101, 897–900. [Google Scholar] [CrossRef]
- Alkhawaja, S.; Ismaeel, A.; Botta, G.; Senok, A.C. The prevalence of congenital and perinatal cytomegalovirus infections among newborns of seropositive mothers. J. Infect. Dev. Ctries. 2012, 6, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Dar, L.; Pati, S.K.; Patro, A.R.K.; Deorari, A.K.; Rai, S.; Kant, S.; Broor, S.; Fowler, K.B.; Britt, W.J.; Boppana, S.B. Congenital cytomegalovirus infection in a highly seropositive semi-urban population in India. Pediatr. Infect. Dis. J. 2008, 27, 841–843. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, Y.; Halle, D.; Eidelman, A.; Reich, D.; Dayan, D.; Rudensky, B.; Raveh, D.; Branski, D.; Kaplan, M.; Shefer, V.; et al. Urine polymerase chain reaction as a screening tool for the detection of congenital cytomegalovirus infection. Arch. Dis. Child. Fetal Neonatal Ed. 2003, 88, F371–F374. [Google Scholar] [CrossRef]
- Viswanathan, R.; Bafna, S.; Mergu, R.; Deshpande, G.; Gunjikar, R.; Gaikwad, S.; Mullick, J. Direct Saliva Real Time Polymerase Chain Reaction Assay Shows Low Birth Prevalence of Congenital CMV Infection in Urban Western India. Pediatr. Infect. Dis. J. 2018, 38, e65–e68. [Google Scholar] [CrossRef]
- Karimian, P.; Yaghini, O.; Azadani, H.N.; Mohammadizadeh, M.; Arabzadeh, S.A.M.; Adibi, A.; Rahimi, H. Prevalence, Characteristics, and One-Year Follow-Up of Congenital Cytomegalovirus Infection in Isfahan City, Iran. Interdiscip. Perspect. Infect. Dis. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Mwaanza, N.; Chilukutu, L.; Tembo, J.; Kabwe, M.; Musonda, K.; Kapasa, M.; Chabala, C.; Sinyangwe, S.; Mwaba, P.; Zumla, A.; et al. High rates of congenital cytomegalovirus infection linked with maternal HIV infection among neonatal admissions at a large referral center in sub-Saharan Africa. Clin. Infect. Dis. 2014, 58, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Tshabalala, D.; Newman, H.; Businge, C.; Mabunda, S.A.; Kemp, W.; Beja, P. Prevalence and determinants of congenital cytomegalovirus infection at a rural South African central hospital in the Eastern Cape. S. Afr. J. Infect. Dis. 2018, 33, 89–92. [Google Scholar]
- Tabatabaee, M.; Tayyebi, D. Seroepidemiologic study of human cytomegalovirus in pregnant women in Valiasr Hospital of Kazeroon, Fars, Iran. J. Matern. Neonatal Med. 2009, 22, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Bates, M.; Brantsaeter, A.B. Human Cytomeglovirus (CMV) in Africa: A neglected but important pathogen. J. Virus Erad. 2016, 2, 136–142. [Google Scholar] [PubMed]
- Prince, H.E.; Lapé-Nixon, M. Role of Cytomegalovirus (CMV) IgG Avidity Testing in Diagnosing Primary CMV Infection during Pregnancy. Clin. Vaccine Immunol. 2014, 21, 1377–1384. [Google Scholar] [CrossRef]
- Ross, S.A.; Ahmed, A.; Palmer, A.L.; Michaels, M.G.; Sanchez, P.J.; Bernstein, D.I.; Tolan, R.W.; Novak, Z.; Chowdhury, N.; Fowler, K.B.; et al. Detection of Congenital Cytomegalovirus Infection by Real-Time Polymerase Chain Reaction Analysis of Saliva or Urine Specimens. J. Infect. Dis. 2014, 210, 1415–1418. [Google Scholar] [CrossRef]
- Ornoy, A.; Diavcitrin, O. Fetal effects of primary and secondary cytomegalovirus infection in pregnancy. Reprod. Toxicol. 2006, 21, 399–409. [Google Scholar] [CrossRef]
- Yinon, Y.; Farine, D.; Yudin, M.H. Screening, Diagnosis, and Management of Cytomegalovirus Infection in Pregnancy. Obstet. Gynecol. Surv. 2010, 65, 736–743. [Google Scholar] [CrossRef]
- Enders, G.; Daiminger, A.; Bader, U.; Exler, S.; Enders, M. Intrauterine transmission and clinical outcome of 248 pregnancies with primary cytomegalovirus infection in relation to gestational age. J. Clin. Virol. 2011, 52, 244–246. [Google Scholar] [CrossRef]
- Boppana, S.B.; Ross, S.A.; Shimamura, M.; Palmer, A.L.; Ahmed, A.; Michaels, M.G.; Sánchez, P.J.; Bernstein, D.I.; Tolan, R.W.; Novak, Z.; et al. Saliva polymerase-chain-reaction assay for cytomegalovirus screening in newborns. N. Engl. J. Med. 2011, 364, 2111–2118. [Google Scholar] [CrossRef]
- Boppana, S.B.; Ross, S.A.; Novak, Z.; Shimamura, M.; Tolan, R.W.; Palmer, A.L.; Ahmed, A.; Michaels, M.G.; Sánchez, P.J.; Bernstein, D.I.; et al. Dried blood spot real-time polymerase chain reaction assays to screen newborns for congenital cytomegalovirus infection. JAMA 2010, 303, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
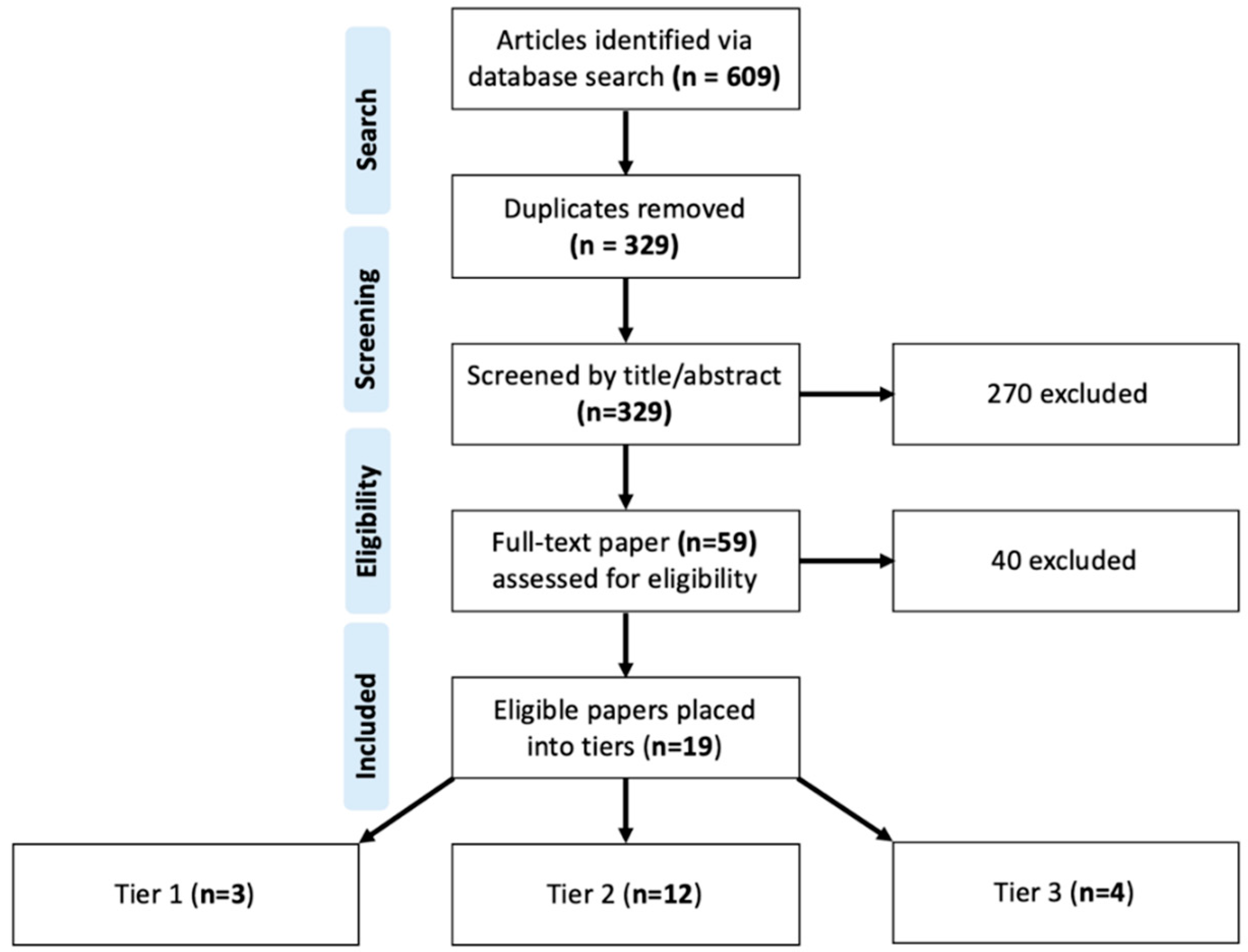
| Study | Region | Mothers (N) | Maternal Seroprevalence (%) | IgG Avidity Testing (n/N, %) | Testing Method | ||||
|---|---|---|---|---|---|---|---|---|---|
| Low | High | Undetermined | Mothers | Infants | |||||
| Tier 1 | Mussi-Pinhata et al. (2018) [19] | Brazil | 1952 A | 98.1 | 5/36 B | 1915/1952 (98.1) | N/A | Serum Ab | Saliva PCR |
| Yamamoto et al. (2011) [26] | Brazil | 452 C | 96.7 | 1/44 (2.2) D | 39/44 (88.6) D | 3/44 (6.8) D | Serum Ab | Saliva or Urine PCR, Culture (Cx) | |
| Mussi-Pinhata et al. (2009) [27] | Brazil | 7848 E | 95.7 F | 1/42 (2.4) D | 41/42 (97.6) D | N/A | Serum Ab | Saliva/Urine PCR and Saliva/Urine Cx | |
| Tier 2 | Arellano-Galindo et al. (2014) [28] | Mexico | 375 G | 97.3 | Serum Ab | Dried Blood PCR, Saliva PCR, Cx | |||
| Yamamoto et al. (2001) [29] | Brazil | 452 | 95.7 | Serum Ab | Urine PCR, Cx | ||||
| Wang S et al. (2017) [30] | China | 5020 H | 96.2 F | Dried Blood Ab | Dried Blood PCR, Saliva PCR | ||||
| Barbi et al. (2006) [31] | Italy | 9032 | 80.0 F | Serum Ab | Dried Blood PCR, Urine Cx | ||||
| Kaneko et al. (2013) [32] | Japan | 1163 | 83.3 | Serum Ab | Urine PCR | ||||
| Madrid et al. (2018) [33] | Mozambique | 118 I | 100 | Serum Ab | Dried Blood PCR | ||||
| Ziyaeyan et al. (2007) [34] | Iran | 92 | 98.6 | Serum Ab | Blood PCR | ||||
| Fowler et al. (2003) [35] | USA | 2857 | 100 | Serum Ab | Saliva Rapid Cx, Saliva/Urine Cx | ||||
| Stagno et al. (1982) [36] | Chile | 197 J | 98 | Serum Ab | Urine Cx | ||||
| Alkhawaja et al. (2012) [37] | Bahrain | 83 K | 100 | Serum Ab | Saliva/Urine PCR, Saliva/Urine Cx | ||||
| Dar et al. (2008) [38] | India | 423 | 99 | Serum Ab | Saliva PCR, Urine PCR | ||||
| Schlesinger et al. (2003) [39] | Israel | 1939 L | 82.6 | Dried Blood Ab | Urine PCR, Cx | ||||
| Tier 3 | Viswanathan et al. (2018) [40] | India | 750 | 99 | Serum Ab | Blood PCR, Saliva PCR, Urine PCR | |||
| Karimian et al. (2016) [41] | Iran | Not reported | 97.6 M | Not tested | Urine PCR | ||||
| Mwaanza et al. (2014) [42] | Sub-Saharan Africa | 395 | 87.8 N | Not tested | Saliva PCR, Serum PCR, Urine PCR, Serum IgM | ||||
| Tshabalala et al. (2018) [43] | South Africa | 302 | 87.8 N | Not tested | Saliva PCR | ||||
| Study | cCMV Rate (n/N, %) | Asymptomatic Infants (n, %) | Symptomatic Infants (n, %) | Symptoms (n) | Hearing Loss Rate (Type; n/N, %) | |||
|---|---|---|---|---|---|---|---|---|
| At Birth | After Birth | |||||||
| Tier 1 | Mussi-Pinhata et al. (2018) [19] | Low: 1/36 (2.8) A | High: 8/1685 (0.47) A | 9 (100) | 0 (0) | None | Not reported | Not reported |
| Yamamoto et al. (2011) [26] | 121/12195 (1.0) | 109 (90.1) | 12 (9.9) | Small for gestational age (8), Abnormal CT findings (3), Death (1) | Not reported | SNHL: 10/102 (9.8) | ||
| Mussi-Pinhata et al. (2009) [27] | 87/8047 (1.1) E | 80 (92.0) | 7 (8.0) | Hepatosplenomegaly (4), Jaundice (4), Petechiae (4), Thrombocytopenia (4), Abnormal CT findings (3), Elevated CSF protein (3), Elevated alanine/aspartate aminotransferase (3), Purpura (2), Seizures (1) | Not reported | SNHL: 3 bilateral, 2 unilateral; 5/58 (8.6) | ||
| Tier 2 | Arellano-Galindo et al. (2014) [28] | 7/277 (2.5) G | 7 (100) | 0 (0) | None | Not reported | Not reported | |
| Yamamoto et al. (2001) [29] | 9/452 (2.0) | 5 (55.6) | 4 (44.4) | Hepatosplenomegaly (3), Intrauterine growth restriction (2), Hypertonia (1), Microcephaly (1), Petechiae (1), Subcortical and periventricular hypodensity of white matter (1) | Not reported | Not reported | ||
| Wang S et al. (2017) [30] | 75/10933 (0.69) H | 71 (94.7) | 4 (5.3) | Hearing loss (2), Jaundice (2) | Bilateral: 2/75 (2.7) | Not reported | ||
| Barbi et al. (2006) [31] | 16/9032 (0.18) | 14 (87.5) | 2 (12.5) | Elevated direct bilirubin (2), Elevated alanine aminotransferase (1), Cerebral calcifications (1) | Transient unilateral: 2/16 (12.5) | Not reported | ||
| Kaneko et al. (2013) [32] | 5/1163 (0.43) | 3 (60) | 2 (40) | Cerebral palsy (1), Intellectual disability (1) | Bilateral: 2/5 (40.0) | Not reported | ||
| Madrid et al. (2018) [33] | 3/115 (2.6) I | 3 (100) | 0 (0) | None | Not reported | Not reported | ||
| Ziyaeyan et al. (2007) [34] | 3/92 (3.3) | 3 (100) | 0 (0) | None | Not reported | Not reported | ||
| Fowler et al. (2003) [35] | 29/2857 (1.0) | Not reported | Not reported | Not reported | Not reported | Not reported | ||
| Stagno et al. (1982) [36] | 2/118 (1.7) J | 2 (100) | 0 (0) | None | Not reported | Not reported | ||
| Alkhawaja et al. (2012) [37] | 3/100 (3.0) K | Not reported | Not reported | Not reported | Not reported | Not reported | ||
| Dar et al. (2008) [38] | 9/423 (2.1) | 8 (88.9) | 1 (11.1) | Hepatosplenomegaly (1) | 0/9 (0) | Unilateral: 1/9 (11.1) | ||
| Schlesinger et al. (2003) [39] | 14/2000 (0.70) L | 12 (85.7) | 2 (14.3) | Hepatosplenomegaly (1), Microcephaly (1), Petechiae (1), Periventricular calcifications (1), Small for gestational age (1), Thrombocytopenia (1), Transient mild hepatitis (1) | 0/10 (0) | Not reported | ||
| Tier 3 | Viswanathan et al. (2018) [40] | 3/750 (0.40) | 2 (66.7) | 1 (33.3) | Lethargy (1), Low birth weight (1), Preterm (1), Respiratory distress (1), Sepsis (1) | Not reported | Not reported | |
| Karimian et al. (2016) [41] | 8/1617 (0.49) | 5 (62.5) | 3 (37.5) | Jaundice (3), Respiratory distress (3), Suspected sepsis (3) | Mild: 1/8 (12.5) | Not reported | ||
| Mwaanza et al. (2014) [42] | 15/395 (3.8) | 9 (60.0) | 6 (40.0) | Jaundice (6), Death (4), Petechiae (1), Respiratory distress (1) | Not reported | Not reported | ||
| Tshabalala et al. (2018) [43] | 18/302 (6.0) | Not reported | Not reported | Not reported | Not reported | Not reported | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coppola, T.; Mangold, J.F.; Cantrell, S.; Permar, S.R. Impact of Maternal Immunity on Congenital Cytomegalovirus Birth Prevalence and Infant Outcomes: A Systematic Review. Vaccines 2019, 7, 129. https://doi.org/10.3390/vaccines7040129
Coppola T, Mangold JF, Cantrell S, Permar SR. Impact of Maternal Immunity on Congenital Cytomegalovirus Birth Prevalence and Infant Outcomes: A Systematic Review. Vaccines. 2019; 7(4):129. https://doi.org/10.3390/vaccines7040129
Chicago/Turabian StyleCoppola, Tiziana, Jesse F. Mangold, Sarah Cantrell, and Sallie R. Permar. 2019. "Impact of Maternal Immunity on Congenital Cytomegalovirus Birth Prevalence and Infant Outcomes: A Systematic Review" Vaccines 7, no. 4: 129. https://doi.org/10.3390/vaccines7040129
APA StyleCoppola, T., Mangold, J. F., Cantrell, S., & Permar, S. R. (2019). Impact of Maternal Immunity on Congenital Cytomegalovirus Birth Prevalence and Infant Outcomes: A Systematic Review. Vaccines, 7(4), 129. https://doi.org/10.3390/vaccines7040129




