Early Investigations and Recent Advances in Intraperitoneal Immunotherapy for Peritoneal Metastasis
Abstract
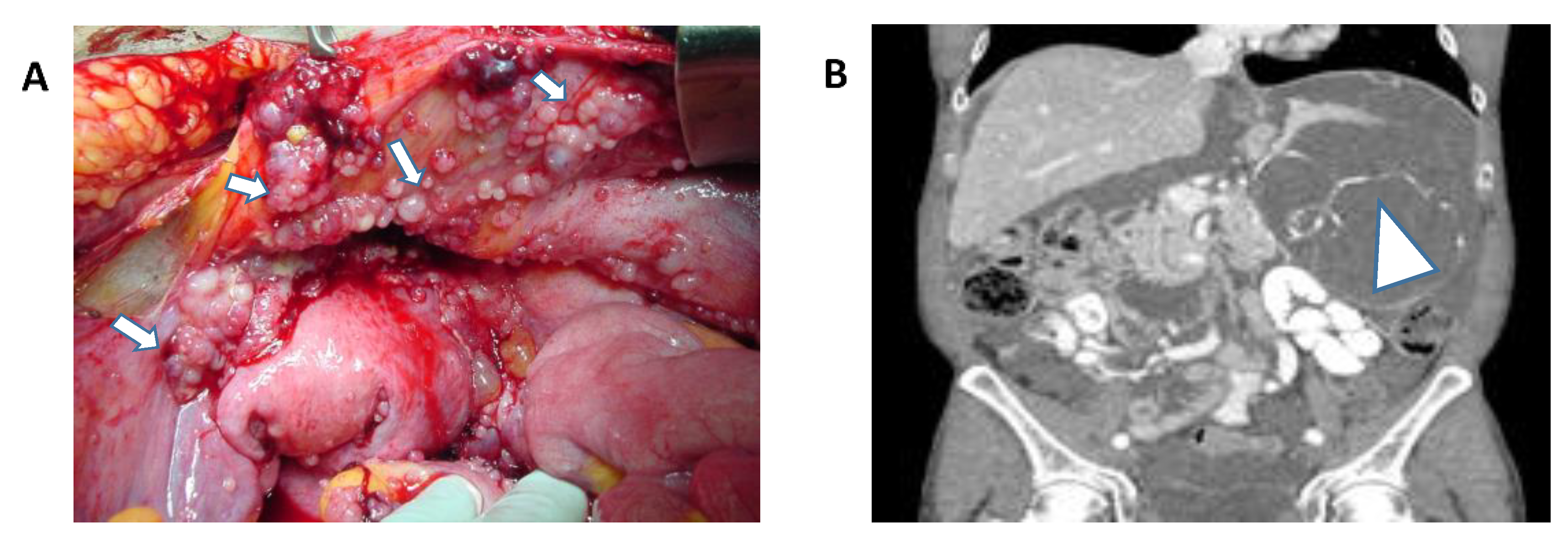
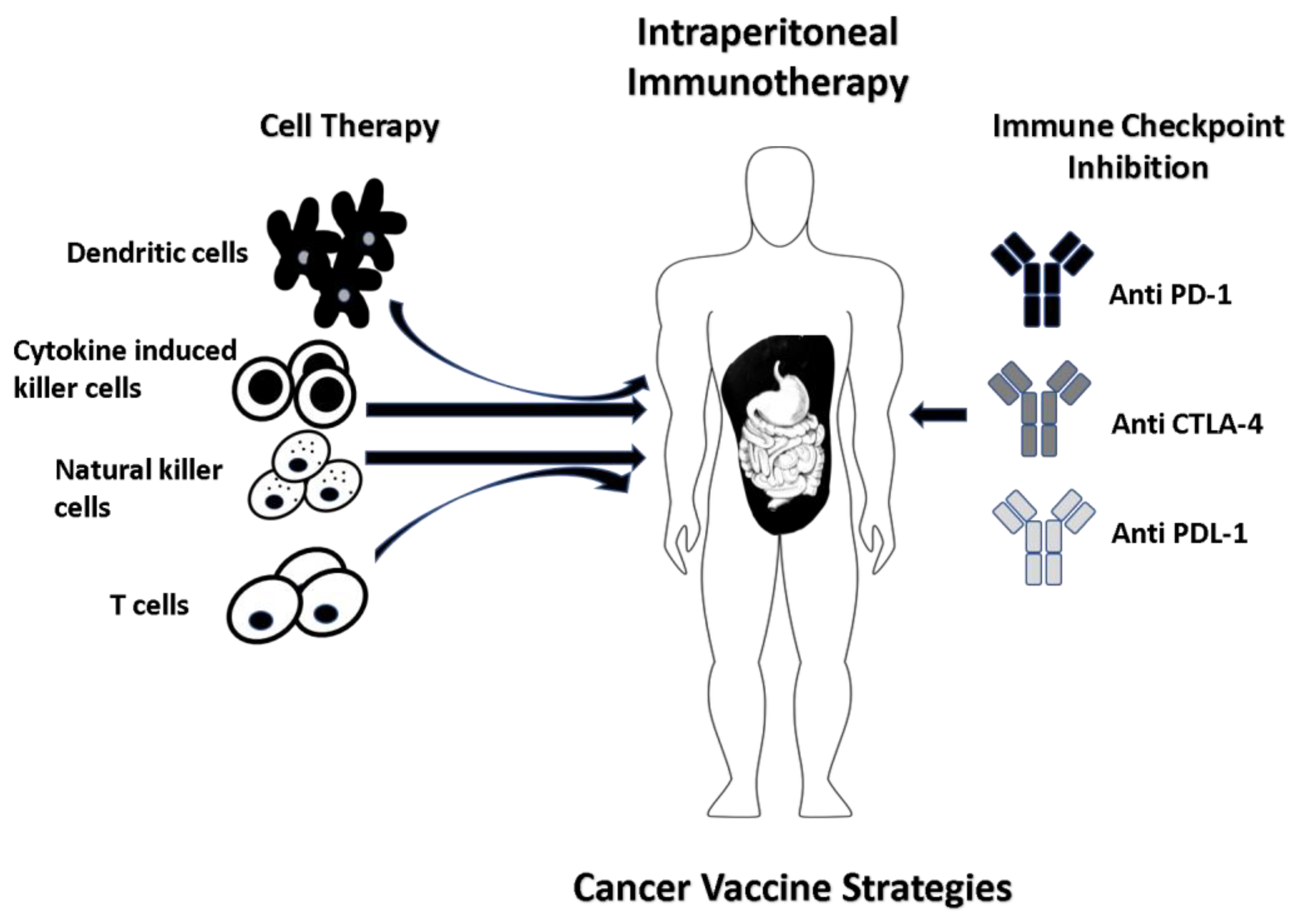
1. Introduction
2. Immunotherapeutic Breakthroughs in the Modern Era
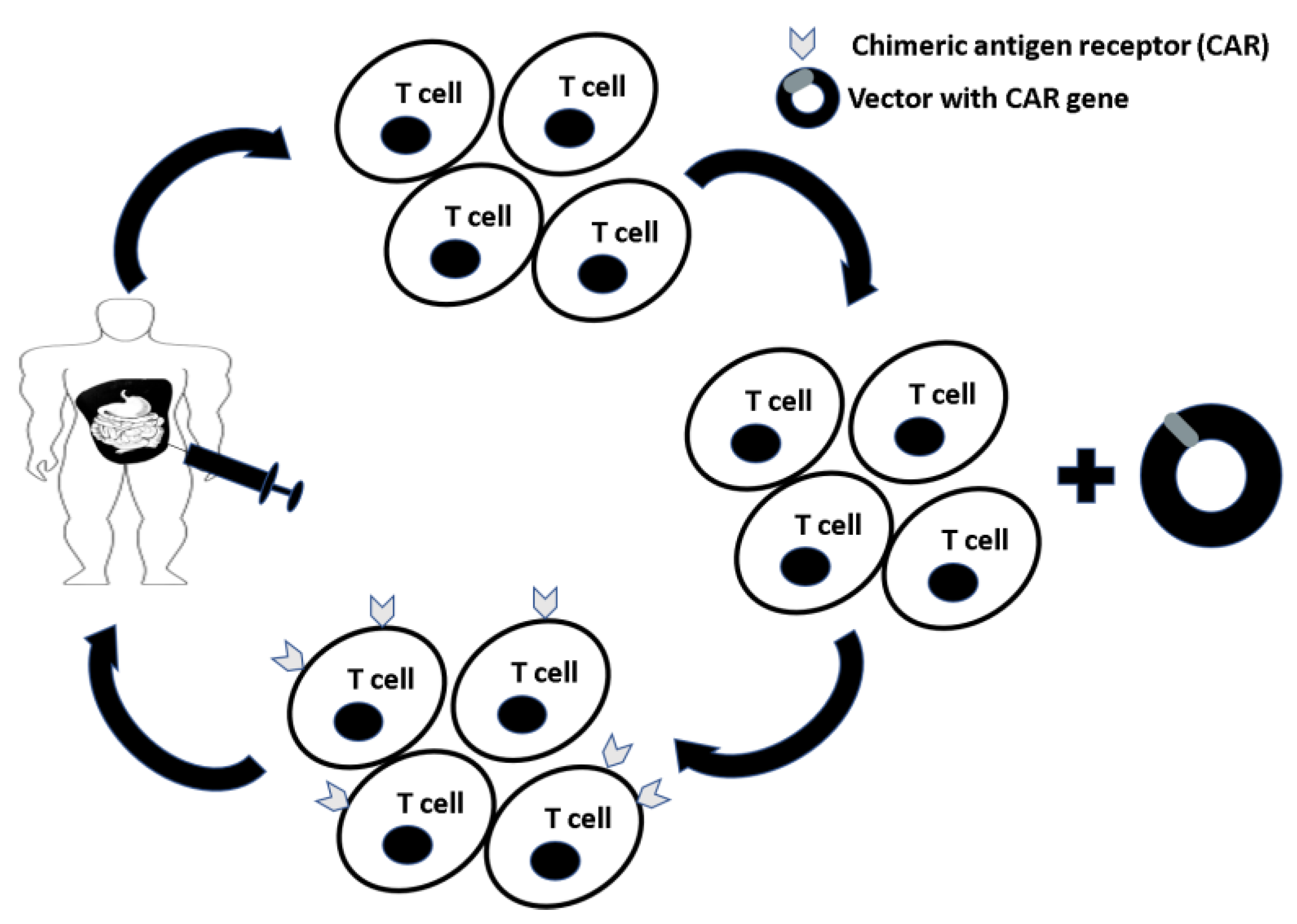
3. CAR-T Cell Immunotherapy
4. CAR-T Cell Investigations for Peritoneal Metastasis
5. Cancer Vaccines for Peritoneal Metastasis
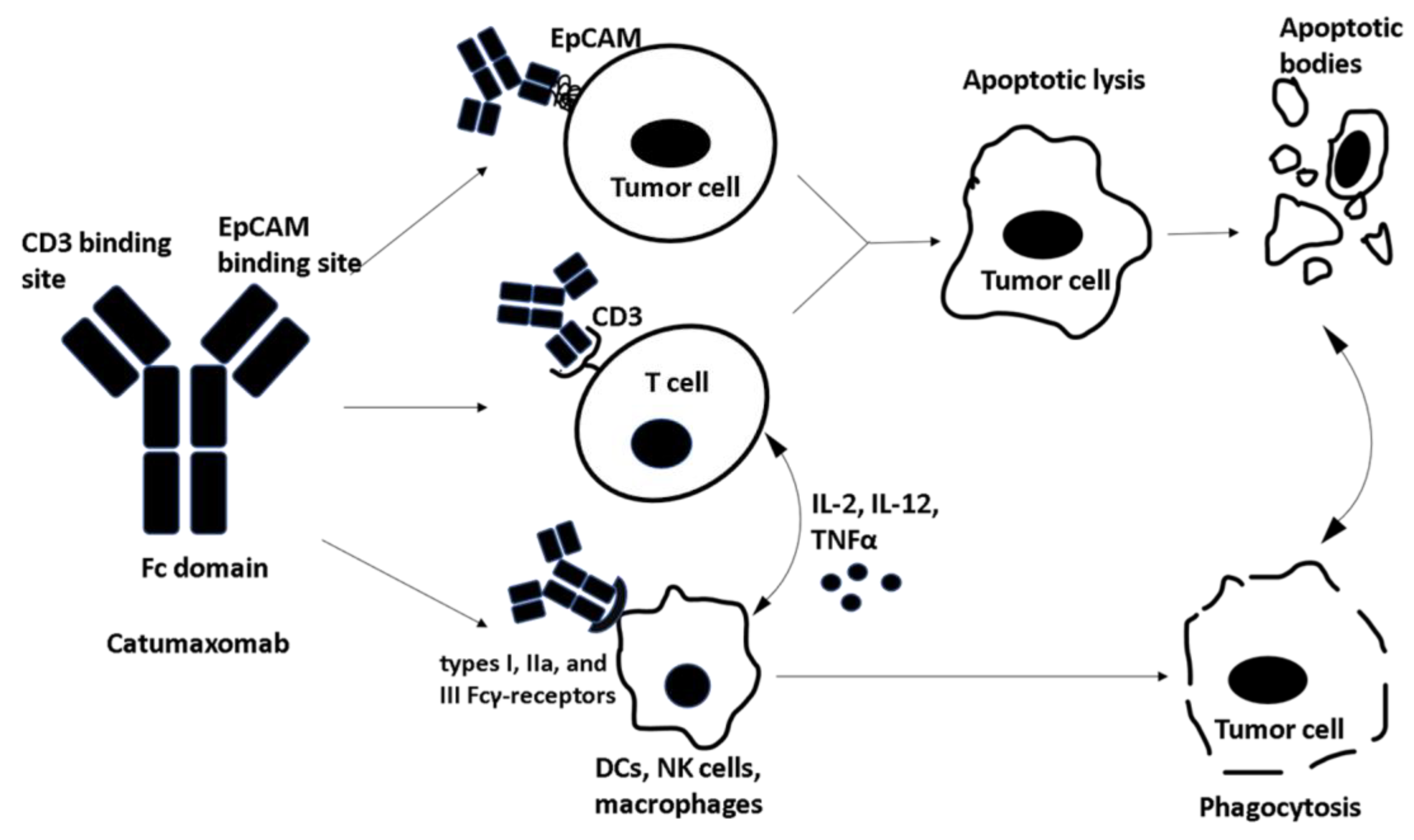
6. Catumaxomab
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Morano, W.F.; Aggarwal, A.; Love, P.; Richard, S.D.; Esquivel, J.; Bowne, W.B. Intraperitoneal immunotherapy: Historical perspectives and modern therapy. Cancer Gene Ther. 2016, 23, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Shariat-Madar, B.; Jayakrishnan, T.T.; Gamblin, T.C.; Turaga, K.K. Surgical management of bowel obstruction in patients with peritoneal carcinomatosis. J. Surg. Oncol. 2014, 110, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.L.; LeDuc, R.J. Small intestinal obstruction from peritoneal carcinomatosis. Am. J. Surg. 1973, 125, 316–317. [Google Scholar] [CrossRef]
- Chu, D.Z.; Lang, N.P.; Thompson, C.; Osteen, P.K.; Westbrook, K.C. Peritoneal carcinomatosis in nongynecologic malignancy. A prospective study of prognostic factors. Cancer 1989, 63, 364–367. [Google Scholar] [CrossRef]
- Kerscher, A.G.; Chua, T.C.; Gasser, M.; Maeder, U.; Kunzmann, V.; Isbert, C.; Germer, C.T.; Pelz, J.O. Impact of peritoneal carcinomatosis in the disease history of colorectal cancer management: A longitudinal experience of 2406 patients over two decades. Br. J. Cancer 2013, 108, 1432–1439. [Google Scholar] [CrossRef] [PubMed]
- Aranha, G.V.; Folk, F.A.; Greenlee, H.B. Surgical palliation of small bowel obstruction due to metastatic carcinoma. Am. Surg. 1981, 47, 99–102. [Google Scholar] [PubMed]
- Glehen, O.; Kwiatkowski, F.; Sugarbaker, P.H.; Elias, D.; Levine, E.A.; De Simone, M.; Barone, R.; Yonemura, Y.; Cavaliere, F.; Quenet, F.; et al. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: A multi-institutional study. J. Clin. Oncol. 2004, 22, 3284–3292. [Google Scholar] [CrossRef] [PubMed]
- Dawson, L.E.; Russell, A.H.; Tong, D.; Wisbeck, W.M. Adenocarcinoma of the sigmoid colon: Sites of initial dissemination and clinical patterns of recurrence following surgery alone. J. Surg. Oncol. 1983, 22, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.T. Surgical resection of tumor bulk in the primary treatment of ovarian carcinoma. Natl. Cancer Inst. Monogr. 1975, 42, 101–104. [Google Scholar] [PubMed]
- Zimm, S.; Cleary, S.M.; Lucas, W.E.; Weiss, R.J.; Markman, M.; Andrews, P.A.; Schiefer, M.A.; Kim, S.; Horton, C.; Howell, S.B. Phase i/pharmacokinetic study of intraperitoneal cisplatin and etoposide. Cancer Res. 1987, 47, 1712–1716. [Google Scholar] [PubMed]
- Sugarbaker, P.H.; Gianola, F.J.; Speyer, J.C.; Wesley, R.; Barofsky, I.; Meyers, C.E. Prospective, randomized trial of intravenous versus intraperitoneal 5-fluorouracil in patients with advanced primary colon or rectal cancer. Surgery 1985, 98, 414–422. [Google Scholar] [PubMed]
- Dedrick, R.L.; Myers, C.E.; Bungay, P.M.; DeVita, V.T., Jr. Pharmacokinetic rationale for peritoneal drug administration in the treatment of ovarian cancer. Cancer Treat. Rep. 1978, 62, 1–11. [Google Scholar] [PubMed]
- Verwaal, V.J.; van Ruth, S.; de Bree, E.; van Sloothen, G.W.; van Tinteren, H.; Boot, H.; Zoetmulder, F.A. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J. Clin. Oncol. 2003, 21, 3737–3743. [Google Scholar] [CrossRef] [PubMed]
- Verwaal, V.J.; Bruin, S.; Boot, H.; van Slooten, G.; van Tinteren, H. 8-year follow-up of randomized trial: Cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann. Surg. Oncol. 2008, 15, 2426–2432. [Google Scholar] [CrossRef] [PubMed]
- Bloemendaal, A.L.; Verwaal, V.J.; van Ruth, S.; Boot, H.; Zoetmulder, F.A. Conventional surgery and systemic chemotherapy for peritoneal carcinomatosis of colorectal origin: A prospective study. Eur. J. Surg. Oncol. 2005, 31, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Van Oudheusden, T.R.; Nienhuijs, S.W.; Luyer, M.D.; Nieuwenhuijzen, G.A.; Lemmens, V.E.; Rutten, H.J.; de Hingh, I.H. Incidence and treatment of recurrent disease after cytoreductive surgery and intraperitoneal chemotherapy for peritoneally metastasized colorectal cancer: A systematic review. Eur. J. Surg. Oncol. 2015, 41, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Miner, T.J.; Shia, J.; Jaques, D.P.; Klimstra, D.S.; Brennan, M.F.; Coit, D.G. Long-term survival following treatment of pseudomyxoma peritonei: An analysis of surgical therapy. Ann. Surg. 2005, 241, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Whang, I.; Chung, H.Y.; Averbach, A.; Sugarbaker, P.H. Indications for early postoperative intraperitoneal chemotherapy of advanced gastric cancer: Results of a prospective randomized trial. World J. Surg. 2001, 25, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Stutman, O. Immunodepression and malignancy. Adv. Cancer Res. 1975, 22, 261–422. [Google Scholar] [PubMed]
- Siegel, B.V. Tumor immunity. An overview. Am. J. Pathol. 1978, 93, 515–524. [Google Scholar] [PubMed]
- Lewis, J.J.; Houghton, A.N. Definition of tumor antigens suitable for vaccine construction. Semin. Cancer Biol. 1995, 6, 321–327. [Google Scholar] [CrossRef]
- Carswell, E.A.; Old, L.J.; Kassel, R.L.; Green, S.; Fiore, N.; Williamson, B. An endotoxin-induced serum factor that causes necrosis of tumors. Proc. Natl. Acad. Sci. USA 1975, 72, 3666–3670. [Google Scholar] [CrossRef] [PubMed]
- Old, L.J.; Clarke, D.A.; Benacerraf, B. Effect of bacillus calmette-guerin infection on transplanted tumours in the mouse. Nature 1959, 184 (Suppl. 5), 291–292. [Google Scholar] [CrossRef]
- Boon, T.; Kellermann, O. Rejection by syngeneic mice of cell variants obtained by mutagenesis of a malignant teratocarcinoma cell line. Proc. Natl. Acad. Sci. USA 1977, 74, 272–275. [Google Scholar] [CrossRef] [PubMed]
- Naftzger, C.; Takechi, Y.; Kohda, H.; Hara, I.; Vijayasaradhi, S.; Houghton, A.N. Immune response to a differentiation antigen induced by altered antigen: A study of tumor rejection and autoimmunity. Proc. Natl. Acad. Sci. USA 1996, 93, 14809–14814. [Google Scholar] [CrossRef] [PubMed]
- Vijayasaradhi, S.; Bouchard, B.; Houghton, A.N. The melanoma antigen gp75 is the human homologue of the mouse b (brown) locus gene product. J. Exp. Med. 1990, 171, 1375–1380. [Google Scholar] [CrossRef] [PubMed]
- Dyall, R.; Bowne, W.B.; Weber, L.W.; LeMaoult, J.; Szabo, P.; Moroi, Y.; Piskun, G.; Lewis, J.J.; Houghton, A.N.; Nikolic-Zugic, J. Heteroclitic immunization induces tumor immunity. J. Exp. Med. 1998, 188, 1553–1561. [Google Scholar] [CrossRef] [PubMed]
- Bowne, W.B.; Srinivasan, R.; Wolchok, J.D.; Hawkins, W.G.; Blachere, N.E.; Dyall, R.; Lewis, J.J.; Houghton, A.N. Coupling and uncoupling of tumor immunity and autoimmunity. J. Exp. Med. 1999, 190, 1717–1722. [Google Scholar] [CrossRef] [PubMed]
- Weber, L.W.; Bowne, W.B.; Wolchok, J.D.; Srinivasan, R.; Qin, J.; Moroi, Y.; Clynes, R.; Song, P.; Lewis, J.J.; Houghton, A.N. Tumor immunity and autoimmunity induced by immunization with homologous DNA. J. Clin. Investig. 1998, 102, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Bowne, W.B.; Wolchok, J.D.; Hawkins, W.G.; Srinivasan, R.; Gregor, P.; Blachere, N.E.; Moroi, Y.; Engelhorn, M.E.; Houghton, A.N.; Lewis, J.J. Injection of DNA encoding granulocyte-macrophage colony-stimulating factor recruits dendritic cells for immune adjuvant effects. Cytokines Cell Mol. Ther. 1999, 5, 217–225. [Google Scholar] [PubMed]
- Hawkins, W.G.; Gold, J.S.; Dyall, R.; Wolchok, J.D.; Hoos, A.; Bowne, W.B.; Srinivasan, R.; Houghton, A.N.; Lewis, J.J. Immunization with DNA coding for gp100 results in cd4 t-cell independent antitumor immunity. Surgery 2000, 128, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Houghton, A.N.; Gold, J.S.; Blachere, N.E. Immunity against cancer: Lessons learned from melanoma. Curr. Opin. Immunol. 2001, 13, 134–140. [Google Scholar] [CrossRef]
- Gold, J.S.; Ferrone, C.R.; Guevara-Patino, J.A.; Hawkins, W.G.; Dyall, R.; Engelhorn, M.E.; Wolchok, J.D.; Lewis, J.J.; Houghton, A.N. A single heteroclitic epitope determines cancer immunity after xenogeneic DNA immunization against a tumor differentiation antigen. J. Immunol. 2003, 170, 5188–5194. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M.; Cohn, Z.A. Pillars article: Identification of a novel cell type in peripheral lymphoid organs of mice. I. Morphology, quantitation, tissue distribution. J. Immunol. 2007, 178, 5–25. [Google Scholar] [PubMed]
- Steinman, R.M.; Nussenzweig, M.C. Dendritic cells: Features and functions. Immunol. Rev. 1980, 53, 127–147. [Google Scholar] [CrossRef] [PubMed]
- Dudley, M.E.; Wunderlich, J.R.; Robbins, P.F.; Yang, J.C.; Hwu, P.; Schwartzentruber, D.J.; Topalian, S.L.; Sherry, R.; Restifo, N.P.; Hubicki, A.M.; et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 2002, 298, 850–854. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A.; Lotze, M.T.; Muul, L.M.; Leitman, S.; Chang, A.E.; Ettinghausen, S.E.; Matory, Y.L.; Skibber, J.M.; Shiloni, E.; Vetto, J.T.; et al. Observations on the systemic administration of autologous lymphokine-activated killer cells and recombinant interleukin-2 to patients with metastatic cancer. N. Engl. J. Med. 1985, 313, 1485–1492. [Google Scholar] [CrossRef] [PubMed]
- Morgan, R.A.; Dudley, M.E.; Wunderlich, J.R.; Hughes, M.S.; Yang, J.C.; Sherry, R.M.; Royal, R.E.; Topalian, S.L.; Kammula, U.S.; Restifo, N.P.; et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science 2006, 314, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Russo, P. Systemic therapy for renal cell carcinoma. J. Urol. 2000, 163, 408–417. [Google Scholar] [CrossRef]
- Gross, J.A.; St John, T.; Allison, J.P. The murine homologue of the t lymphocyte antigen cd28. Molecular cloning and cell surface expression. J. Immunol. 1990, 144, 3201–3210. [Google Scholar] [PubMed]
- Krummel, M.F.; Allison, J.P. Pillars article: Cd28 and ctla-4 have opposing effects on the response of t cells to stimulation. J. Immunol. 2011, 187, 3459–3465. [Google Scholar] [PubMed]
- Freeman, G.J.; Long, A.J.; Iwai, Y.; Bourque, K.; Chernova, T.; Nishimura, H.; Fitz, L.J.; Malenkovich, N.; Okazaki, T.; Byrne, M.C.; et al. Engagement of the pd-1 immunoinhibitory receptor by a novel b7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 2000, 192, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Latchman, Y.; Wood, C.R.; Chernova, T.; Chaudhary, D.; Borde, M.; Chernova, I.; Iwai, Y.; Long, A.J.; Brown, J.A.; Nunes, R.; et al. Pd-l2 is a second ligand for pd-1 and inhibits t cell activation. Nat. Immunol. 2001, 2, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Curtis, S.A.; Cohen, J.V.; Kluger, H.M. Evolving immunotherapy approaches for renal cell carcinoma. Curr. Oncol. Rep. 2016, 18, 57. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.A.; June, C.H. The principles of engineering immune cells to treat cancer. Cell 2017, 168, 724–740. [Google Scholar] [CrossRef] [PubMed]
- Chmielewski, M.; Hombach, A.A.; Abken, H. Antigen-specific t-cell activation independently of the mhc: Chimeric antigen receptor-redirected t cells. Front. Immunol. 2013, 4, 371. [Google Scholar] [CrossRef] [PubMed]
- Kershaw, M.H.; Westwood, J.A.; Darcy, P.K. Gene-engineered t cells for cancer therapy. Nat. Rev. Cancer 2013, 13, 525–541. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.; Schussler-Lenz, M.; Bondanza, A.; Buchholz, C.J. Clinical development of car t cells-challenges and opportunities in translating innovative treatment concepts. EMBO Mol. Med. 2017, 9, 1183–1197. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.C.; Burga, R.A.; McCormack, E.; Wang, L.J.; Mooring, W.; Point, G.R.; Khare, P.D.; Thorn, M.; Ma, Q.; Stainken, B.F.; et al. Phase i hepatic immunotherapy for metastases study of intra-arterial chimeric antigen receptor-modified t-cell therapy for cea+ liver metastases. Clin. Cancer Res. 2015, 21, 3149–3159. [Google Scholar] [CrossRef] [PubMed]
- Parkhurst, M.R.; Yang, J.C.; Langan, R.C.; Dudley, M.E.; Nathan, D.A.; Feldman, S.A.; Davis, J.L.; Morgan, R.A.; Merino, M.J.; Sherry, R.M.; et al. T cells targeting carcinoembryonic antigen can mediate regression of metastatic colorectal cancer but induce severe transient colitis. Mol. Ther. 2011, 19, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Hasegawa, K.; Matsushita, H.; Fujieda, N.; Sato, S.; Miyagi, E.; Kakimi, K.; Fujiwara, K. Expression of multiple immune checkpoint molecules on t cells in malignant ascites from epithelial ovarian carcinoma. Oncol. Lett. 2018, 15, 6457–6468. [Google Scholar] [CrossRef] [PubMed]
- Abiko, K.; Mandai, M.; Hamanishi, J.; Yoshioka, Y.; Matsumura, N.; Baba, T.; Yamaguchi, K.; Murakami, R.; Yamamoto, A.; Kharma, B.; et al. Pd-l1 on tumor cells is induced in ascites and promotes peritoneal dissemination of ovarian cancer through ctl dysfunction. Clin. Cancer Res. 2013, 19, 1363–1374. [Google Scholar] [CrossRef] [PubMed]
- Bayne, L.J.; Beatty, G.L.; Jhala, N.; Clark, C.E.; Rhim, A.D.; Stanger, B.Z.; Vonderheide, R.H. Tumor-derived granulocyte-macrophage colony-stimulating factor regulates myeloid inflammation and t cell immunity in pancreatic cancer. Cancer Cell 2012, 21, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Dilek, N.; Vuillefroy de Silly, R.; Blancho, G.; Vanhove, B. Myeloid-derived suppressor cells: Mechanisms of action and recent advances in their role in transplant tolerance. Front. Immunol. 2012, 3, 208. [Google Scholar] [CrossRef] [PubMed]
- Pylayeva-Gupta, Y.; Lee, K.E.; Hajdu, C.H.; Miller, G.; Bar-Sagi, D. Oncogenic kras-induced gm-csf production promotes the development of pancreatic neoplasia. Cancer Cell 2012, 21, 836–847. [Google Scholar] [CrossRef] [PubMed]
- Ledermann, J.A.; Canevari, S.; Thigpen, T. Targeting the folate receptor: Diagnostic and therapeutic approaches to personalize cancer treatments. Ann. Oncol. 2015, 26, 2034–2043. [Google Scholar] [CrossRef] [PubMed]
- Song, D.G.; Ye, Q.; Carpenito, C.; Poussin, M.; Wang, L.P.; Ji, C.; Figini, M.; June, C.H.; Coukos, G.; Powell, D.J., Jr. In vivo persistence, tumor localization, and antitumor activity of car-engineered t cells is enhanced by costimulatory signaling through cd137 (4-1bb). Cancer Res. 2011, 71, 4617–4627. [Google Scholar] [CrossRef] [PubMed]
- Kawalekar, O.U.; O’Connor, R.S.; Fraietta, J.A.; Guo, L.; McGettigan, S.E.; Posey, A.D., Jr.; Patel, P.R.; Guedan, S.; Scholler, J.; Keith, B.; et al. Distinct signaling of coreceptors regulates specific metabolism pathways and impacts memory development in car t cells. Immunity 2016, 44, 712. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.C.; Point, G.R.; Cunetta, M.; Thorn, M.; Guha, P.; Espat, N.J.; Boutros, C.; Hanna, N.; Junghans, R.P. Regional car-t cell infusions for peritoneal carcinomatosis are superior to systemic delivery. Cancer Gene Ther. 2016, 23, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Liu, C.; Li, G.; Li, J.; Lv, X.; Shi, H.; Liu, J.; Liu, S.; Yan, P.; Wang, S.; et al. Antitumor effects and persistence of a novel her2 car t cells directed to gastric cancer in preclinical models. Am. J. Cancer Res. 2018, 8, 106–119. [Google Scholar] [PubMed]
- Hong, H.; Brown, C.E.; Ostberg, J.R.; Priceman, S.J.; Chang, W.C.; Weng, L.; Lin, P.; Wakabayashi, M.T.; Jensen, M.C.; Forman, S.J. L1 cell adhesion molecule-specific chimeric antigen receptor-redirected human t cells exhibit specific and efficient antitumor activity against human ovarian cancer in mice. PLoS ONE 2016, 11, e0146885. [Google Scholar] [CrossRef] [PubMed]
- Daponte, A.; Kostopoulou, E.; Kollia, P.; Papamichali, R.; Vanakara, P.; Hadjichristodoulou, C.; Nakou, M.; Samara, S.; Koukoulis, G.; Messinis, I.E. L1 (cam) (cd171) in ovarian serous neoplasms. Eur. J. Gynaecol. Oncol. 2008, 29, 26–30. [Google Scholar] [PubMed]
- Koneru, M.; Purdon, T.J.; Spriggs, D.; Koneru, S.; Brentjens, R.J. Il-12 secreting tumor-targeted chimeric antigen receptor t cells eradicate ovarian tumors in vivo. Oncoimmunology 2015, 4, e994446. [Google Scholar] [CrossRef] [PubMed]
- Koneru, M.; O'Cearbhaill, R.; Pendharkar, S.; Spriggs, D.R.; Brentjens, R.J. A phase i clinical trial of adoptive t cell therapy using il-12 secreting muc-16(ecto) directed chimeric antigen receptors for recurrent ovarian cancer. J. Transl. Med. 2015, 13, 102. [Google Scholar] [CrossRef] [PubMed]
- Ai, Y.Q.; Cai, K.; Hu, J.H.; Jiang, L.W.; Gao, Y.R.; Zhao, H.; Jia, S.C. The clinical effects of dendritic cell vaccines combined with cytokine-induced killer cells intraperitoneal injected on patients with malignant ascites. Int. J. Clin. Exp. Med. 2014, 7, 4272–4281. [Google Scholar] [PubMed]
- Geller, M.A.; Knorr, D.A.; Hermanson, D.A.; Pribyl, L.; Bendzick, L.; McCullar, V.; Miller, J.S.; Kaufman, D.S. Intraperitoneal delivery of human natural killer cells for treatment of ovarian cancer in a mouse xenograft model. Cytotherapy 2013, 15, 1297–1306. [Google Scholar] [CrossRef] [PubMed]
- Denman, C.J.; Denyukov, V.V.; Somanchi, S.S.; Phatarpekar, P.V.; Kopp, L.M.; Johnson, J.L.; Singh, H.; Hurton, L.; Maiti, S.N.; Huls, M.H.; et al. Membrane-bound il-21 promotoes sustained ex vivo proliferation of human natural killer cells. PLoS ONE 2012, 7, e30264. [Google Scholar] [CrossRef] [PubMed]
- Oyer, J.L.; Pandey, V.; Igarashi, R.Y.; Somanchi, S.S.; Zakari, A.; Solh, M.; Lee, D.A.; Altomare, D.A.; Copik, A.J. Natural killer cells stimulated with pm21 particles expand and biodistribute in vivo: Clinical implications for cancer treatment. Cytotherapy 2016, 18, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Lauer, U.M.; Schell, M.; Beil, J.; Berchtold, S.; Koppenhofer, U.; Glatzle, J.; Konigsrainer, A.; Mohle, R.; Nann, D.; Fend, F.; et al. Phase i study of oncolytic vaccinia virus gl-onc1 in patients with peritoneal carcinomatosis. Clin. Cancer Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Alkayyal, A.A.; Tai, L.H.; Kennedy, M.A.; de Souza, C.T.; Zhang, J.; Lefebvre, C.; Sahi, S.; Ananth, A.A.; Mahmoud, A.B.; Makrigiannis, A.P.; et al. Nk-cell recruitment is necessary for eradication of peritoneal carcinomatosis with an il12-expressing maraba virus cellular vaccine. Cancer Immunol. Res. 2017, 5, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Luo, M.; Wei, X.W.; Ma, C.C.; Yang, Y.H.; Shao, B.; Liu, Y.T.; Liu, T.; Ren, J.; Liu, L.; et al. A folate receptor-targeted lipoplex delivering interleukin-15 gene for colon cancer immunotherapy. Oncotarget 2016, 7, 52207–52217. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Li, W.; Yoshiya, S.; Xu, Y.; Hata, M.; El-Darawish, Y.; Markova, T.; Yamanishi, K.; Yamanishi, H.; Tahara, H.; et al. Augmentation of immune checkpoint cancer immunotherapy with il18. Clin. Cancer Res. 2016, 22, 2969–2980. [Google Scholar] [CrossRef] [PubMed]
- Berinstein, N.L.; Karkada, M.; Oza, A.M.; Odunsi, K.; Villella, J.A.; Nemunaitis, J.J.; Morse, M.A.; Pejovic, T.; Bentley, J.; Buyse, M.; et al. Survivin-targeted immunotherapy drives robust polyfunctional T cell generation and differentiation in advanced ovarian cancer patients. Oncoimmunology 2015, 4, e1026529. [Google Scholar] [CrossRef] [PubMed]
- Gujar, S.A.; Marcato, P.; Pan, D.; Lee, P.W. Reovirus virotherapy overrides tumor antigen presentation evasion and promotes protective antitumor immunity. Mol. Cancer Ther. 2010, 9, 2924–2933. [Google Scholar] [CrossRef] [PubMed]
- Dobrzanski, M.J.; Rewers-Felkins, K.A.; Quinlin, I.S.; Samad, K.A.; Phillips, C.A.; Robinson, W.; Dobrzanski, D.J.; Wright, S.E. Autologous muc1-specific th1 effector cell immunotherapy induces differential levels of systemic treg cell subpopulations that result in increased ovarian cancer patient survival. Clin. Immunol. 2009, 133, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Chianese-Bullock, K.A.; Irvin, W.P., Jr.; Petroni, G.R.; Murphy, C.; Smolkin, M.; Olson, W.C.; Coleman, E.; Boerner, S.A.; Nail, C.J.; Neese, P.Y.; et al. A multipeptide vaccine is safe and elicits t-cell responses in participants with advanced stage ovarian cancer. J. Immunother. 2008, 31, 420–430. [Google Scholar] [CrossRef] [PubMed]
- Grosso, J.F.; Jure-Kunkel, M.N. Ctla-4 blockade in tumor models: An overview of preclinical and translational research. Cancer Immunity 2013, 13, 5. [Google Scholar] [PubMed]
- Deng, J.; Wang, L.; Chen, H.; Li, L.; Ma, Y.; Ni, J.; Li, Y. The role of tumour-associated muc1 in epithelial ovarian cancer metastasis and progression. Cancer Metastasis Rev. 2013, 32, 535–551. [Google Scholar] [CrossRef] [PubMed]
- Sangisetty, S.L.; Miner, T.J. Malignant ascites: A review of prognostic factors, pathophysiology and therapeutic measures. World J. Gastrointest Surg. 2012, 4, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Gujar, S.; Dielschneider, R.; Clements, D.; Helson, E.; Shmulevitz, M.; Marcato, P.; Pan, D.; Pan, L.Z.; Ahn, D.G.; Alawadhi, A.; et al. Multifaceted therapeutic targeting of ovarian peritoneal carcinomatosis through virus-induced immunomodulation. Mol. Ther. 2013, 21, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Clements, D.; Helson, E.; Gujar, S.A.; Lee, P.W. Reovirus in cancer therapy: An evidence-based review. Oncolytic virother. 2014, 3, 69–82. [Google Scholar] [PubMed]
- Kelemen, L.E. The role of folate receptor alpha in cancer development, progression and treatment: Cause, consequence or innocent bystander? Int. J. Cancer 2006, 119, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Berger, M.; Arina, A.; Schreiber, K.; Hyjek, E.; Schietinger, A.; Binder, D.C.; Butz, E.; Krausz, T.; Rowley, D.A.; et al. Safety and immunologic effects of il-15 administration in nonhuman primates. Blood 2009, 114, 2417–2426. [Google Scholar] [CrossRef] [PubMed]
- Chelius, D.; Ruf, P.; Gruber, P.; Ploscher, M.; Liedtke, R.; Gansberger, E.; Hess, J.; Wasiliu, M.; Lindhofer, H. Structural and functional characterization of the trifunctional antibody catumaxomab. mAbs 2010, 2, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Seimetz, D. Novel monoclonal antibodies for cancer treatment: The trifunctional antibody catumaxomab (removab). J. Cancer 2011, 2, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Heiss, M.M.; Murawa, P.; Koralewski, P.; Kutarska, E.; Kolesnik, O.O.; Ivanchenko, V.V.; Dudnichenko, A.S.; Aleknaviciene, B.; Razbadauskas, A.; Gore, M.; et al. The trifunctional antibody catumaxomab for the treatment of malignant ascites due to epithelial cancer: Results of a prospective randomized phase II/III trial. Int. J. Cancer 2010, 127, 2209–2221. [Google Scholar] [CrossRef] [PubMed]
- Heiss, M.M.; Ströhlein, M.A.; Jäger, M.; Kimmig, R.; Burges, A.; Schoberth, A.; Jauch, K.W.; Schildberg, F.W.; Lindhofer, H. Immunotherapy of malignant ascites with trifunctional antibodies. Tumor Immunol. 2005, 117, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Bokemeyer, C.; Stein, A.; Ridwelski, K.; Atanackovic, D.; Arnold, D.; Woll, E.; Ulrich, A.; Fischer, R.; Kruger, C.; Schuhmacher, C. A phase II study of catumaxomab administered intra- and postoperatively as part of a multimodal approach in primarily resectable gastric cancer. Gastric Cancer 2015, 18, 833–842. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Borlak, J.; Langer, F.; Spanel, R.; Schondorfer, G.; Dittrich, C. Immune-mediated liver injury of the cancer therapeutic antibody catumaxomab targeting epcam, cd3 and fcgamma receptors. Oncotarget 2016, 7, 28059–28074. [Google Scholar] [CrossRef] [PubMed]
- Wimberger, P.; Gilet, H.; Gonschior, A.K.; Heiss, M.M.; Moehler, M.; Oskay-Oezcelik, G.; Al-Batran, S.E.; Schmalfeldt, B.; Schmittel, A.; Schulze, E.; et al. Deterioration in quality of life (qol) in patients with malignant ascites: Results from a phase II/III study comparing paracentesis plus catumaxomab with paracentesis alone. Ann. Oncol. 2012, 23, 1979–1985. [Google Scholar] [CrossRef] [PubMed]
- Strohlein, M.A.; Siegel, R.; Jager, M.; Lindhofer, H.; Jauch, K.W.; Heiss, M.M. Induction of anti-tumor immunity by trifunctional antibodies in patients with peritoneal carcinomatosis. J. Exp. Clin. Cancer Res. 2009, 28, 18. [Google Scholar] [CrossRef] [PubMed]
- Burges, A.; Wimberger, P.; Kumper, C.; Gorbounova, V.; Sommer, H.; Schmalfeldt, B.; Pfisterer, J.; Lichinitser, M.; Makhson, A.; Moiseyenko, V.; et al. Effective relief of malignant ascites in patients with advanced ovarian cancer by a trifunctional anti-epcam x anti-cd3 antibody: A phase I/II study. Clin. Cancer Res. 2007, 13, 3899–3905. [Google Scholar] [CrossRef] [PubMed]
- Ruf, P.; Gires, O.; Jager, M.; Fellinger, K.; Atz, J.; Lindhofer, H. Characterisation of the new epcam-specific antibody ho-3: Implications for trifunctional antibody immunotherapy of cancer. Br. J. Cancer 2007, 97, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Mackey, J.R.; Venner, P.M. Malignant ascites: Demographics, therapeutic effacy and predictors of survival. Can. J. Oncol. 1996, 6, 474–480. [Google Scholar] [PubMed]
- Sadeghi, B.; Arvieux, C.; Glehen, O.; Beaujard, A.C.; Rivoire, M.; Baulieux, J.; Fontaumard, E.; Brachet, A.; Caillot, J.L.; Faure, J.L.; et al. Peritoneal carcinomatosis from non-gynecologic malignancies: Results of the evocape i multicentric prospective study. Cancer 2000, 88, 358–363. [Google Scholar] [CrossRef]
- Trivedi, A.; Stienen, S.; Zhu, M.; Li, H.; Yuraszeck, T.; Gibbs, J.; Heath, T.; Loberg, R.; Kasichayanula, S. Clinical pharmacology and translational aspects of bispecific antibodies. Clin. Transl. Sci. 2017, 10, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Goéré, D.; Gras-Chaput, N.; Aupérin, A.; Flament, C.; Mariette, C.; Glehen, O.; Zitvogel, L.; Elias, D. Treatment of gastric peritoneal carcinomatosis by combining complete surgical resection of lesions and intraperitoneal immunotherapy using catumaxomab. BMC Cancer 2014, 14, 148. [Google Scholar] [CrossRef] [PubMed]
- Mau-Sorensen, M.; Dittrich, C.; Dienstmann, R.; Lassen, U.; Buchler, W.; Martinius, H.; Tabernero, J. A phase i trial of intravenous catumaxomab: A bispecific monoclonal antibody targeting epcam and the t cell coreceptor cd3. Cancer Chemother. Pharmacol. 2015, 75, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
| Cancer Type | Treatment | Target | Model | Author (Year) |
|---|---|---|---|---|
| Gastric and Ovarian | chA21-4-1BBz CAR-T cells | HER2 | Murine | Han et al. [60] (2018) |
| Ovarian cancer | CE7+R TCM CAR-T cells | L1-CAM | Murine | Hong et al. [61] (2016) Daponte et al. [62] (2008) |
| Colorectal cancer | Anti CEA CAR-T cells with anti Gr1/GITR and anti PD-L1 | CEA, Gr1 and PD-L1 | Murine | Katz et al. [59] (2016) |
| Ovarian cancer | Anti MUC16 CAR-T cells | MUC16 | Human | Koneru et al. [63,64] (2015) |
| Breast and gastric cancer | Anti CEA CAR-T cells | CEA | Human | NCT02349724 (2015) |
| Ovarian, Breast and Colorectal cancer | Anti FRα CAR-T cells | FRα | Murine | Song et al. [57] (2011) |
| Cancer Type | Treatment | Target | Model | Author (Year) |
|---|---|---|---|---|
| Ovarian cancer, peritoneal carcinomatosis | GL-ONC1 | Malignant ascites | Human | Lauer et al. [69] (2018) |
| Colon cancer | MG1-IL12-ICV | CD69 and IP10 | Murine | Alkayyal et al. [70] (2017) |
| Colon cancer | FRα targeted lipoplex delivering IL-15 gene. | FRα | Murine | Liang et al. [71] (2016) |
| Colon and breast | Anti PD-L1 and CTLA-4 in combination with IL-18 | PD-L1 and CTLA-4 | Murine | Ma et al. [72] (2016) |
| Chronic myelogenous leukemia | NK cells stimulated by IL-21 | NKs | Murine | Oyer et al. [68] (2016) |
| Ovarian cancer, peritoneal metastasis | Survivac vaccine | Survivin | Human | Berinstein et al. [73] (2015) |
| Colon, ovarian, gastric, pancreatic cancer | Dendritic cell vaccine+CIKs | Tumor inducing cytokines, CD4+CD25+Tregs | Human | Ai et al. [65] (2014) |
| Ovarian cancer | Reovirus based anti-cancer therapy | Gr 1.1+, CD11b+MDSCs, FOXP3+Tregs, CD3+cells. | Human, Murine | Gujar et al. [74] (2013) |
| Ovarian cancer | IP delivered human NKs | Murine | Geller et al. [66] (2013) | |
| Ovarian cancer | Anti MUC1 T cells | MUC1 | Human | Dobrzanski et al. [75] (2009) |
| Ovarian cancer | Multipeptide vaccine | MAGE-A1, FBP, Her-2/neu | Human | Chianene-Bullock et al. [76] (2008) |
| Cancer Type | Treatment | Target | Model | Author (Year) |
|---|---|---|---|---|
| Gastric cancer | Intra and postoperatively administered Catumaxomab | EpCAM | Human | Bokemeyer et al. [88] (2015) |
| Colorectal cancer | Catumaxomab | EpCAM | Human | Borlak et al. [89] (2015) |
| Ovarian cancer | Catumaxomab | EpCAM | Human | Wimberger et al. [90] (2012) |
| Ovarian, pancreatic, colon, gastric, breast | Catumaxomab+paracentesis | EpCAM | Human | Heiss et al. [86] (2010) |
| Gastric, ovarian, PM ααom unknown primary | Catumaxomab | EpCAM | Human | StrÖhlein et al. [91] (2009) |
| Ovarian cancer | Catumaxomab | EpCAM | Human | Burges et al. [92] (2007) |
| Colon cancer | Catumaxomab | EpCAM | Murine | Ruf et al. [93] (2007) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thadi, A.; Khalili, M.; Morano, W.F.; Richard, S.D.; Katz, S.C.; Bowne, W.B. Early Investigations and Recent Advances in Intraperitoneal Immunotherapy for Peritoneal Metastasis. Vaccines 2018, 6, 54. https://doi.org/10.3390/vaccines6030054
Thadi A, Khalili M, Morano WF, Richard SD, Katz SC, Bowne WB. Early Investigations and Recent Advances in Intraperitoneal Immunotherapy for Peritoneal Metastasis. Vaccines. 2018; 6(3):54. https://doi.org/10.3390/vaccines6030054
Chicago/Turabian StyleThadi, Anusha, Marian Khalili, William F. Morano, Scott D. Richard, Steven C. Katz, and Wilbur B. Bowne. 2018. "Early Investigations and Recent Advances in Intraperitoneal Immunotherapy for Peritoneal Metastasis" Vaccines 6, no. 3: 54. https://doi.org/10.3390/vaccines6030054
APA StyleThadi, A., Khalili, M., Morano, W. F., Richard, S. D., Katz, S. C., & Bowne, W. B. (2018). Early Investigations and Recent Advances in Intraperitoneal Immunotherapy for Peritoneal Metastasis. Vaccines, 6(3), 54. https://doi.org/10.3390/vaccines6030054





