Next Generation Cancer Vaccines—Make It Personal!
Abstract
1. Introduction
2. Tumour Antigens
2.1. Tumour Associated Antigens
2.2. Tumour Specific Antigens
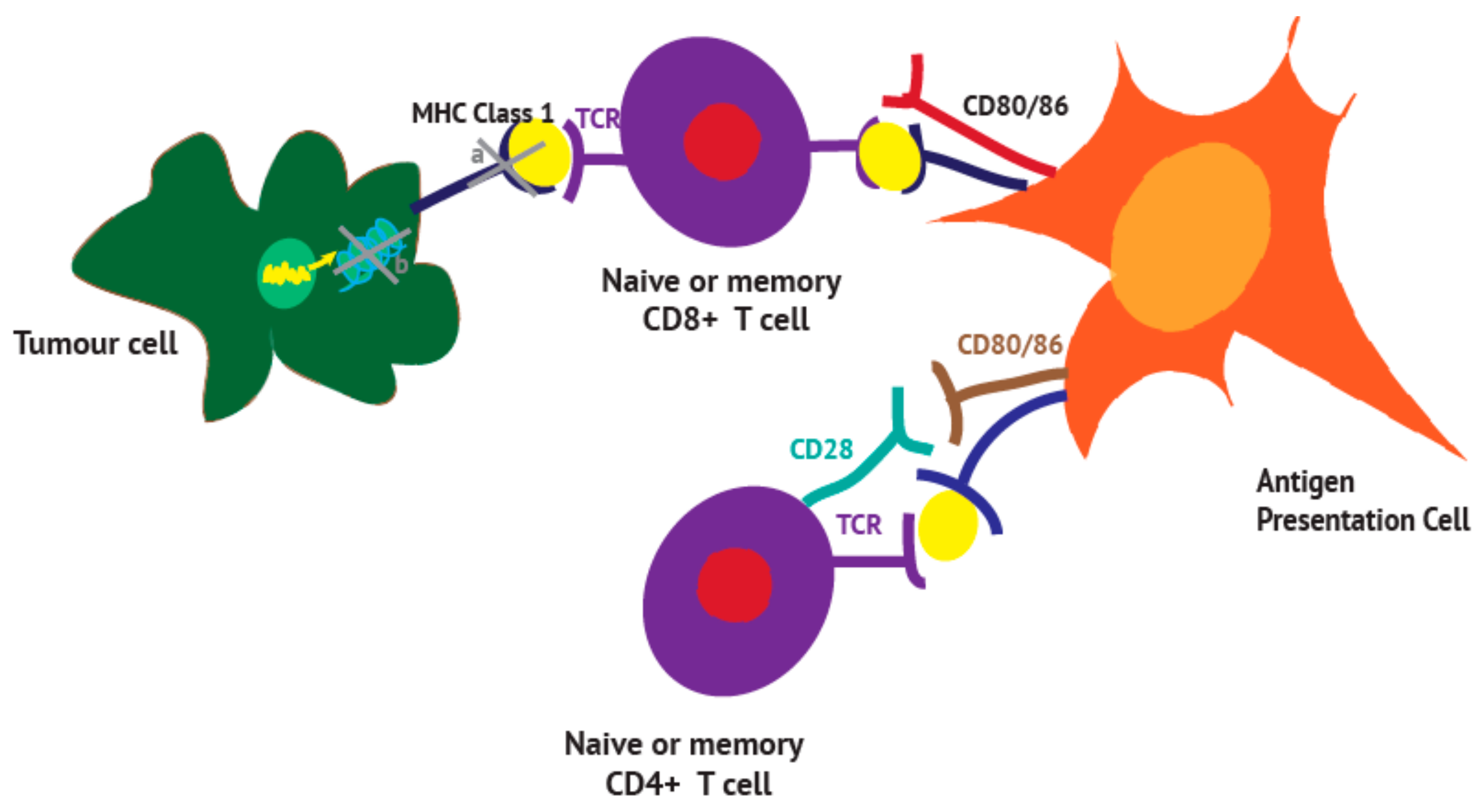
3. Antigen Presentation
4. Cancer Vaccines
4.1. Ex Vivo Approaches
4.2. In Vivo Approaches
5. Personalised Cancer Vaccines
6. Challenges
6.1. Choosing the Right Antigen—Improving Bioinformatics
6.2. Choosing the Right Combination
6.3. Choosing the Right Time—Adjuvant vs. Palliative?
6.4. Tumour Evolution and Loss of Antigen
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, D.S.; Mellman, I. Oncology meets immunology: The cancer-immunity cycle. Immunity 2013, 39, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Allison, J.P. Immune checkpoint targeting in cancer therapy: Toward combination strategies with curative potential. Cell 2015, 161, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Allison, J.P. The future of immune checkpoint therapy. Science 2015, 348, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.R.; Maute, R.L.; Dulken, B.W.; Hutter, G.; George, B.M.; McCracken, M.N.; Gupta, R.; Tsai, J.M.; Sinha, R.; Corey, D.; et al. Pd-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity. Nature 2017, 545, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, T.; Honjo, T. The pd-1-pd-l pathway in immunological tolerance. Trends Immunol. 2006, 27, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Keir, M.E.; Butte, M.J.; Freeman, G.J.; Sharpe, A.H. Pd-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 2008, 26, 677–704. [Google Scholar] [CrossRef] [PubMed]
- Umansky, V.; Blattner, C.; Gebhardt, C.; Utikal, J. The role of myeloid-derived suppressor cells (mdsc) in cancer progression. Vaccines 2016, 4, 36. [Google Scholar] [CrossRef] [PubMed]
- Noman, M.Z.; Desantis, G.; Janji, B.; Hasmim, M.; Karray, S.; Dessen, P.; Bronte, V.; Chouaib, S. Pd-l1 is a novel direct target of hif-1alpha, and its blockade under hypoxia enhanced mdsc-mediated t cell activation. J. Exp. Med. 2014, 211, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Balkwill, F.R. The chemokine system and cancer. J. Pathol. 2012, 226, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Flies, D.B. Molecular mechanisms of t cell co-stimulation and co-inhibition. Nat. Rev. Immunol. 2013, 13, 227–242. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Tumeh, P.C.; Harview, C.L.; Yearley, J.H.; Shintaku, I.P.; Taylor, E.J.; Robert, L.; Chmielowski, B.; Spasic, M.; Henry, G.; Ciobanu, V.; et al. Pd-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 2014, 515, 568–571. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-pd-l1 antibody mpdl3280a in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Hegde, P.S.; Karanikas, V.; Evers, S. The where, the when, and the how of immune monitoring for cancer immunotherapies in the era of checkpoint inhibition. Clin. Cancer Res. 2016, 22, 1865–1874. [Google Scholar] [CrossRef] [PubMed]
- Schietinger, A.; Philip, M.; Schreiber, H. Specificity in cancer immunotherapy. Semin. Immunol. 2008, 20, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Ott, P.A.; Wu, C.J. Towards personalized, tumour-specific, therapeutic vaccines for cancer. Nat. Rev. Immunol. 2018, 18, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Thaxton, J.E.; Li, Z. To affinity and beyond: Harnessing the t cell receptor for cancer immunotherapy. Hum. Vacc. Immunother. 2014, 10, 3313–3321. [Google Scholar] [CrossRef] [PubMed]
- Theobald, M.; Biggs, J.; Hernandez, J.; Lustgarten, J.; Labadie, C.; Sherman, L.A. Tolerance to p53 by a2.1-restricted cytotoxic t lymphocytes. J. Exp. Med. 1997, 185, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Stern, P.L.; Harrop, R. 5t4 oncofoetal antigen: An attractive target for immune intervention in cancer. Cancer Immunol. Immunother. 2017, 66, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Melief, C.J.M.; van Hall, T.; Arens, R.; Ossendorp, F.; van der Burg, S.H. Therapeutic cancer vaccines. J. Clin. Investig. 2015, 125, 3401–3412. [Google Scholar] [CrossRef] [PubMed]
- Kenter, G.G.; Welters, M.J.; Valentijn, A.R.; Lowik, M.J.; Berends-van der Meer, D.M.; Vloon, A.P.; Essahsah, F.; Fathers, L.M.; Offringa, R.; Drijfhout, J.W.; et al. Vaccination against hpv-16 oncoproteins for vulvar intraepithelial neoplasia. N. Engl. J. Med. 2009, 361, 1838–1847. [Google Scholar] [CrossRef] [PubMed]
- Herrero, R.; Gonzalez, P.; Markowitz, L.E. Present status of human papillomavirus vaccine development and implementation. Lancet Oncol. 2015, 16, e206–e216. [Google Scholar] [CrossRef]
- Batich, K.A.; Reap, E.A.; Archer, G.E.; Sanchez-Perez, L.; Nair, S.K.; Schmittling, R.J.; Norberg, P.; Xie, W.; Herndon, J.E., 2nd; Healy, P.; et al. Long-term survival in glioblastoma with cytomegalovirus pp65-targeted vaccination. Clin. Cancer Res. 2017, 23, 1898–1909. [Google Scholar] [CrossRef] [PubMed]
- Obeid, J.; Hu, Y.N.; Slingluff, C.L. Vaccines, adjuvants, and dendritic cell activators-current status and future challenges. Semin. Oncol. 2015, 42, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Ott, P.A.; Hu, Z.; Keskin, D.B.; Shukla, S.A.; Sun, J.; Bozym, D.J.; Zhang, W.; Luoma, A.; Giobbie-Hurder, A.; Peter, L.; et al. An immunogenic personal neoantigen vaccine for patients with melanoma. Nature 2017, 547, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Baumgaertner, P.; Costa Nunes, C.; Cachot, A.; Maby-El Hajjami, H.; Cagnon, L.; Braun, M.; Derre, L.; Rivals, J.P.; Rimoldi, D.; Gnjatic, S.; et al. Vaccination of stage iii/iv melanoma patients with long ny-eso-1 peptide and cpg-b elicits robust cd8(+) and cd4(+) t-cell responses with multiple specificities including a novel dr7-restricted epitope. Oncoimmunology 2016, 5, e1216290. [Google Scholar] [CrossRef] [PubMed]
- Rooney, M.S.; Shukla, S.A.; Wu, C.J.; Getz, G.; Hacohen, N. Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell 2015, 160, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.D.; Warren, R.L.; Gibb, E.A.; Martin, S.D.; Spinelli, J.J.; Nelson, B.H.; Holt, R.A. Neo-antigens predicted by tumor genome meta-analysis correlate with increased patient survival. Genome Res. 2014, 24, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Giannakis, M.; Mu, X.J.; Shukla, S.A.; Qian, Z.R.; Cohen, O.; Nishihara, R.; Bahl, S.; Cao, Y.; Amin-Mansour, A.; Yamauchi, M.; et al. Genomic correlates of immune-cell infiltrates in colorectal carcinoma. Cell Rep. 2016, 17, 1206. [Google Scholar] [CrossRef] [PubMed]
- Gubin, M.M.; Zhang, X.; Schuster, H.; Caron, E.; Ward, J.P.; Noguchi, T.; Ivanova, Y.; Hundal, J.; Arthur, C.D.; Krebber, W.J.; et al. Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature 2014, 515, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Yadav, M.; Jhunjhunwala, S.; Phung, Q.T.; Lupardus, P.; Tanguay, J.; Bumbaca, S.; Franci, C.; Cheung, T.K.; Fritsche, J.; Weinschenk, T.; et al. Predicting immunogenic tumour mutations by combining mass spectrometry and exome sequencing. Nature 2014, 515, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Mittendorf, E.A.; Ardavanis, A.; Symanowski, J.; Murray, J.L.; Shumway, N.M.; Litton, J.K.; Hale, D.F.; Perez, S.A.; Anastasopoulou, E.A.; Pistamaltzian, N.F.; et al. Primary analysis of a prospective, randomized, single-blinded phase ii trial evaluating the her2 peptide ae37 vaccine in breast cancer patients to prevent recurrence. Ann. Oncol. 2016, 27, 1241–1248. [Google Scholar] [CrossRef] [PubMed]
- Lilleby, W.; Gaudernack, G.; Brunsvig, P.F.; Vlatkovic, L.; Schulz, M.; Mills, K.; Hole, K.H.; Inderberg, E.M. Phase i/iia clinical trial of a novel htert peptide vaccine in men with metastatic hormone-naive prostate cancer. Cancer Immunol. Immun. 2017, 66, 891–901. [Google Scholar] [CrossRef] [PubMed]
- Kantoff, P.W.; Higano, C.S.; Shore, N.D.; Berger, E.R.; Small, E.J.; Penson, D.F.; Redfern, C.H.; Ferrari, A.C.; Dreicer, R.; Sims, R.B.; et al. Sipuleucel-t immunotherapy for castration-resistant prostate cancer. N. Engl. J. Med. 2010, 363, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Tiriveedhi, V.; Tucker, N.; Herndon, J.; Li, L.J.; Sturmoski, M.; Ellis, M.; Ma, C.; Naughton, M.; Lockhart, A.C.; Gao, F.; et al. Safety and preliminary evidence of biologic efficacy of a mammaglobin-a DNA vaccine in patients with stable metastatic breast cancer. Clin. Cancer Res. 2014, 20, 5964–5975. [Google Scholar] [CrossRef] [PubMed]
- Tarhini, A.A.; Leng, S.; Moschos, S.J.; Yin, Y.; Sander, C.; Lin, Y.; Gooding, W.E.; Kirkwood, J.M. Safety and immunogenicity of vaccination with mart-1 (26-35, 27 l), gp100 (209-217, 210 m), and tyrosinase (368-376, 370d) in adjuvant with pf-3512676 and gm-csf in metastatic melanoma. J. Immunother. 2012, 35, 359–366. [Google Scholar] [CrossRef]
- Vansteenkiste, J.F.; Cho, B.C.; Vanakesa, T.; De Pas, T.; Zielinski, M.; Kim, M.S.; Jassem, J.; Yoshimura, M.; Dahabreh, J.; Nakayama, H.; et al. Efficacy of the mage-a3 cancer immunotherapeutic as adjuvant therapy in patients with resected mage-a3-positive non-small-cell lung cancer (magrit): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016, 17, 822–835. [Google Scholar] [CrossRef]
- Guo, C.; Manjili, M.H.; Subjeck, J.R.; Sarkar, D.; Fisher, P.B.; Wang, X.Y. Therapeutic cancer vaccines: Past, present, and future. Adv. Cancer Res. 2013, 119, 421–475. [Google Scholar] [PubMed]
- Antonilli, M.; Rahimi, H.; Visconti, V.; Napoletano, C.; Ruscito, I.; Zizzari, I.G.; Caponeto, S.; Barchiesi, G.; Iadarola, R.; Pierelli, L.; et al. Triple peptide vaccination as consolidation treatment in women affected by ovarian and breast cancer: Clinical and immunological data of a phase i/ii clinical trial. Int. J. Oncol. 2016, 48, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhu, H.; Liu, Y.; Musetti, S.; Huang, L. Braf peptide vaccine facilitates therapy of murine braf-mutant melanoma. Cancer Immunol. Immunother. 2018, 67, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Chaft, J.E.; Litvak, A.; Arcila, M.E.; Patel, P.; D’Angelo, S.P.; Krug, L.M.; Rusch, V.; Mattson, A.; Coeshott, C.; Park, B.; et al. Phase ii study of the gi-4000 kras vaccine after curative therapy in patients with stage i-iii lung adenocarcinoma harboring a kras g12c, g12d, or g12v mutation. Clin. Lung Cancer 2014, 15, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Park, H.W. Germs, hosts, and the origin of frank macfarlane burnet’s concept of “self” and “tolerance”, 1936–1949. J. Hist. Med. Allied Sci. 2006, 61, 492–534. [Google Scholar] [CrossRef] [PubMed]
- Zitvogel, L.; Kroemer, G. Anticancer immunochemotherapy using adjuvants with direct cytotoxic effects. J. Clin. Investig. 2009, 119, 2127–2130. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.D.; Martin, S.; Golab, J.; Agostinis, P. Danger signalling during cancer cell death: Origins, plasticity and regulation. Cell Death Differ. 2014, 21, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, T.A.; Choi, J.; Green, D.R. Armed response: How dying cells influence t-cell functions. Immunol. Rev. 2011, 241, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Andersen, B.M.; Ohlfest, J.R. Increasing the efficacy of tumor cell vaccines by enhancing cross priming. Cancer Lett. 2012, 325, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Lasek, W.; Zagozdzon, R.; Jakobisiak, M. Interleukin 12: Still a promising candidate for tumor immunotherapy? Cancer Immunol. Immunother. 2014, 63, 419–435. [Google Scholar] [CrossRef] [PubMed]
- Kreiter, S.; Vormehr, M.; van de Roemer, N.; Diken, M.; Lower, M.; Diekmann, J.; Boegel, S.; Schrors, B.; Vascotto, F.; Castle, J.C.; et al. Mutant mhc class ii epitopes drive therapeutic immune responses to cancer. Nature 2015, 520, 692–696. [Google Scholar] [CrossRef] [PubMed]
- Kumai, T.; Lee, S.; Cho, H.I.; Sultan, H.; Kobayashi, H.; Harabuchi, Y.; Celis, E. Optimization of peptide vaccines to induce robust antitumor cd4 t-cell responses. Cancer Immunol. Res. 2017, 5, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Godet, Y.; Fabre, E.; Dosset, M.; Lamuraglia, M.; Levionnois, E.; Ravel, P.; Benhamouda, N.; Cazes, A.; Le Pimpec-Barthes, F.; Gaugler, B.; et al. Analysis of spontaneous tumor-specific cd4 t-cell immunity in lung cancer using promiscuous hla-dr telomerase-derived epitopes: Potential synergistic effect with chemotherapy response. Clin. Cancer Res. 2012, 18, 2943–2953. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, E.F. The toxins of william b. Coley and the treatment of bone and soft-tissue sarcomas. Iowa Orthop. J. 2006, 26, 154–158. [Google Scholar] [PubMed]
- Kantoff, P.W.; Schuetz, T.J.; Blumenstein, B.A.; Glode, L.M.; Bilhartz, D.L.; Wyand, M.; Manson, K.; Panicali, D.L.; Laus, R.; Schlom, J.; et al. Overall survival analysis of a phase ii randomized controlled trial of a poxviral-based psa-targeted immunotherapy in metastatic castration-resistant prostate cancer. J. Clin. Oncol. 2010, 28, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Morse, M.A.; Chaudhry, A.; Gabitzsch, E.S.; Hobeika, A.C.; Osada, T.; Clay, T.M.; Amalfitano, A.; Burnett, B.K.; Devi, G.R.; Hsu, D.S.; et al. Novel adenoviral vector induces t-cell responses despite anti-adenoviral neutralizing antibodies in colorectal cancer patients. Cancer Immunol. Immunother. 2013, 62, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Berinstein, N.L.; Karkada, M.; Oza, A.M.; Odunsi, K.; Villella, J.A.; Nemunaitis, J.J.; Morse, M.A.; Pejovic, T.; Bentley, J.; Buyse, M.; et al. Survivin-targeted immunotherapy drives robust polyfunctional t cell generation and differentiation in advanced ovarian cancer patients. Oncoimmunology 2015, 4, e1026529. [Google Scholar] [CrossRef] [PubMed]
- McNeel, D.G.; Dunphy, E.J.; Davies, J.G.; Frye, T.P.; Johnson, L.E.; Staab, M.J.; Horvath, D.L.; Straus, J.; Alberti, D.; Marnocha, R.; et al. Safety and immunological efficacy of a DNA vaccine encoding prostatic acid phosphatase in patients with stage d0 prostate cancer. J. Clin. Oncol. 2009, 27, 4047–4054. [Google Scholar] [CrossRef] [PubMed]
- Neninger Vinageras, E.; de la Torre, A.; Osorio Rodriguez, M.; Catala Ferrer, M.; Bravo, I.; Mendoza del Pino, M.; Abreu Abreu, D.; Acosta Brooks, S.; Rives, R.; del Castillo Carrillo, C.; et al. Phase ii randomized controlled trial of an epidermal growth factor vaccine in advanced non-small-cell lung cancer. J. Clin. Oncol. 2008, 26, 1452–1458. [Google Scholar] [CrossRef] [PubMed]
- Quoix, E.; Lena, H.; Losonczy, G.; Forget, F.; Chouaid, C.; Papai, Z.; Gervais, R.; Ottensmeier, C.; Szczesna, A.; Kazarnowicz, A.; et al. Tg4010 immunotherapy and first-line chemotherapy for advanced non-small-cell lung cancer (time): Results from the phase 2b part of a randomised, double-blind, placebo-controlled, phase 2b/3 trial. Lancet Oncol. 2016, 17, 212–223. [Google Scholar] [CrossRef]
- Chiappori, A.A.; Soliman, H.; Janssen, W.E.; Antonia, S.J.; Gabrilovich, D.I. Ingn-225: A dendritic cell-based p53 vaccine (ad.P53-dc) in small cell lung cancer: Observed association between immune response and enhanced chemotherapy effect. Expert Opin. Biol. Ther. 2010, 10, 983–991. [Google Scholar] [CrossRef] [PubMed]
- Sahin, U.; Derhovanessian, E.; Miller, M.; Kloke, B.P.; Simon, P.; Lower, M.; Bukur, V.; Tadmor, A.D.; Luxemburger, U.; Schrors, B.; et al. Personalized rna mutanome vaccines mobilize poly-specific therapeutic immunity against cancer. Nature 2017, 547, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Brun, J.L.; Dalstein, V.; Leveque, J.; Mathevet, P.; Raulic, P.; Baldauf, J.J.; Scholl, S.; Huynh, B.; Douvier, S.; Riethmuller, D.; et al. Regression of high-grade cervical intraepithelial neoplasia with tg4001 targeted immunotherapy. Am. J. Obstet. Gynecol. 2011, 204, 169.e1–169.e8. [Google Scholar] [CrossRef] [PubMed]
- Slingluff, C.L., Jr.; Petroni, G.R.; Chianese-Bullock, K.A.; Smolkin, M.E.; Ross, M.I.; Haas, N.B.; von Mehren, M.; Grosh, W.W. Randomized multicenter trial of the effects of melanoma-associated helper peptides and cyclophosphamide on the immunogenicity of a multipeptide melanoma vaccine. J. Clin. Oncol. 2011, 29, 2924–2932. [Google Scholar] [CrossRef] [PubMed]
- Santos, P.M.; Butterfield, L.H. Dendritic cell-based cancer vaccines. J. Immunol. 2018, 200, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Bol, K.F.; Schreibelt, G.; Gerritsen, W.R.; de Vries, I.J.; Figdor, C.G. Dendritic cell-based immunotherapy: State of the art and beyond. Clin. Cancer Res. 2016, 22, 1897–1906. [Google Scholar] [CrossRef] [PubMed]
- Schadendorf, D.; Ugurel, S.; Schuler-Thurner, B.; Nestle, F.O.; Enk, A.; Brocker, E.B.; Grabbe, S.; Rittgen, W.; Edler, L.; Sucker, A.; et al. Dacarbazine (dtic) versus vaccination with autologous peptide-pulsed dendritic cells (dc) in first-line treatment of patients with metastatic melanoma: A randomized phase iii trial of the dc study group of the decog. Ann. Oncol. 2006, 17, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Carreno, B.M.; Magrini, V.; Becker-Hapak, M.; Kaabinejadian, S.; Hundal, J.; Petti, A.A.; Ly, A.; Lie, W.R.; Hildebrand, W.H.; Mardis, E.R.; et al. Cancer immunotherapy. A dendritic cell vaccine increases the breadth and diversity of melanoma neoantigen-specific t cells. Science 2015, 348, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Sonpavde, G.; Di Lorenzo, G.; Higano, C.S.; Kantoff, P.W.; Madan, R.; Shore, N.D. The role of sipuleucel-t in therapy for castration-resistant prostate cancer: A critical analysis of the literature. Eur. Urol. 2012, 61, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.L.; Haynes, L.; Parker, C.; Iversen, P. Interdisciplinary critique of sipuleucel-t as immunotherapy in castration-resistant prostate cancer. J. Natl. Cancer Inst. 2012, 104, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, D.A.; Batich, K.A.; Gunn, M.D.; Huang, M.N.; Sanchez-Perez, L.; Nair, S.K.; Congdon, K.L.; Reap, E.A.; Archer, G.E.; Desjardins, A.; et al. Tetanus toxoid and ccl3 improve dendritic cell vaccines in mice and glioblastoma patients. Nature 2015, 519, 366–369. [Google Scholar] [CrossRef] [PubMed]
- Wilgenhof, S.; Corthals, J.; Heirman, C.; van Baren, N.; Lucas, S.; Kvistborg, P.; Thielemans, K.; Neyns, B. Phase ii study of autologous monocyte-derived mrna electroporated dendritic cells (trimixdc-mel) plus ipilimumab in patients with pretreated advanced melanoma. J. Clin. Oncol. 2016, 34, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- De Vries, I.J.; Krooshoop, D.J.; Scharenborg, N.M.; Lesterhuis, W.J.; Diepstra, J.H.; Van Muijen, G.N.; Strijk, S.P.; Ruers, T.J.; Boerman, O.C.; Oyen, W.J.; et al. Effective migration of antigen-pulsed dendritic cells to lymph nodes in melanoma patients is determined by their maturation state. Cancer Res. 2003, 63, 12–17. [Google Scholar] [PubMed]
- De Vries, I.J.; Lesterhuis, W.J.; Scharenborg, N.M.; Engelen, L.P.; Ruiter, D.J.; Gerritsen, M.J.; Croockewit, S.; Britten, C.M.; Torensma, R.; Adema, G.J.; et al. Maturation of dendritic cells is a prerequisite for inducing immune responses in advanced melanoma patients. Clin. Cancer Res. 2003, 9, 5091–5100. [Google Scholar] [PubMed]
- Barratt-Boyes, S.M.; Zimmer, M.I.; Harshyne, L.A.; Meyer, E.M.; Watkins, S.C.; Capuano, S., 3rd; Murphey-Corb, M.; Falo, L.D., Jr.; Donnenberg, A.D. Maturation and trafficking of monocyte-derived dendritic cells in monkeys: Implications for dendritic cell-based vaccines. J. Immunol. 2000, 164, 2487–2495. [Google Scholar] [CrossRef] [PubMed]
- Anguille, S.; Smits, E.L.; Bryant, C.; Van Acker, H.H.; Goossens, H.; Lion, E.; Fromm, P.D.; Hart, D.N.; Van Tendeloo, V.F.; Berneman, Z.N. Dendritic cells as pharmacological tools for cancer immunotherapy. Pharmacol. Rev. 2015, 67, 731–753. [Google Scholar] [CrossRef] [PubMed]
- Anguille, S.; Smits, E.L.; Lion, E.; van Tendeloo, V.F.; Berneman, Z.N. Clinical use of dendritic cells for cancer therapy. Lancet Oncol. 2014, 15, e257–e267. [Google Scholar] [CrossRef]
- Hirayama, M.; Nishimura, Y. The present status and future prospects of peptide-based cancer vaccines. Int. Immunol. 2016, 28, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Marin-Acevedo, J.A.; Soyano, A.E.; Dholaria, B.; Knutson, K.L.; Lou, Y. Cancer immunotherapy beyond immune checkpoint inhibitors. J. Hematol. Oncol. 2018, 11, 8. [Google Scholar] [CrossRef] [PubMed]
- Khong, H.; Overwijk, W.W. Adjuvants for peptide-based cancer vaccines. J. Immunother. Cancer 2016, 4, 56. [Google Scholar] [CrossRef] [PubMed]
- Duraiswamy, J.; Freeman, G.J.; Coukos, G. Dual blockade of pd-1 and ctla-4 combined with tumor vaccine effectively restores t-cell rejection function in tumors--response. Cancer Res. 2014, 74, 633–634, discussion 635. [Google Scholar] [CrossRef] [PubMed]
- Wada, S.; Jackson, C.M.; Yoshimura, K.; Yen, H.R.; Getnet, D.; Harris, T.J.; Goldberg, M.V.; Bruno, T.C.; Grosso, J.F.; Durham, N.; et al. Sequencing ctla-4 blockade with cell-based immunotherapy for prostate cancer. J. Transl. Med. 2013, 11, 89. [Google Scholar] [CrossRef] [PubMed]
- Curran, M.A.; Montalvo, W.; Yagita, H.; Allison, J.P. Pd-1 and ctla-4 combination blockade expands infiltrating t cells and reduces regulatory t and myeloid cells within b16 melanoma tumors. Proc. Natl. Acad. Sci. USA 2010, 107, 4275–4280. [Google Scholar] [CrossRef] [PubMed]
- Jinushi, M.; Nakazaki, Y.; Dougan, M.; Carrasco, D.R.; Mihm, M.; Dranoff, G. Mfg-e8-mediated uptake of apoptotic cells by apcs links the pro- and antiinflammatory activities of gm-csf. J. Clin. Investig. 2007, 117, 1902–1913. [Google Scholar] [CrossRef] [PubMed]
- Pardi, N.; Hogan, M.J.; Porter, F.W.; Weissman, D. Mrna vaccines—A new era in vaccinology. Nat. Rev. Drug Discov. 2018, 17, 261–279. [Google Scholar] [CrossRef] [PubMed]
- Rini, B.I.; Stenzl, A.; Zdrojowy, R.; Kogan, M.; Shkolnik, M.; Oudard, S.; Weikert, S.; Bracarda, S.; Crabb, S.J.; Bedke, J.; et al. Ima901, a multipeptide cancer vaccine, plus sunitinib versus sunitinib alone, as first-line therapy for advanced or metastatic renal cell carcinoma (imprint): A multicentre, open-label, randomised, controlled, phase 3 trial. Lancet Oncol. 2016, 17, 1599–1611. [Google Scholar] [CrossRef]
- Walter, S.; Weinschenk, T.; Stenzl, A.; Zdrojowy, R.; Pluzanska, A.; Szczylik, C.; Staehler, M.; Brugger, W.; Dietrich, P.Y.; Mendrzyk, R.; et al. Multipeptide immune response to cancer vaccine ima901 after single-dose cyclophosphamide associates with longer patient survival. Nat. Med. 2012, 18, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Butts, C.; Socinski, M.A.; Mitchell, P.L.; Thatcher, N.; Havel, L.; Krzakowski, M.; Nawrocki, S.; Ciuleanu, T.E.; Bosquee, L.; Trigo, J.M.; et al. Tecemotide (l-blp25) versus placebo after chemoradiotherapy for stage iii non-small-cell lung cancer (start): A randomised, double-blind, phase 3 trial. Lancet Oncol. 2014, 15, 59–68. [Google Scholar] [CrossRef]
- Elia, A.R.; Cappello, P.; Puppo, M.; Fraone, T.; Vanni, C.; Eva, A.; Musso, T.; Novelli, F.; Varesio, L.; Giovarelli, M. Human dendritic cells differentiated in hypoxia down-modulate antigen uptake and change their chemokine expression profile. J. Leukoc. Biol. 2008, 84, 1472–1482. [Google Scholar] [CrossRef] [PubMed]
- Degregorio, M.; Degregorio, M.; Wurz, G.T.; Wurz, G.T.; Gutierrez, A.; Gutierrez, A.; Wolf, M. L-blp25 vaccine plus letrozole for breast cancer: Is translation possible? Oncoimmunology 2012, 1, 1422–1424. [Google Scholar] [CrossRef] [PubMed]
- Wurz, G.T.; Kao, C.J.; Wolf, M.; DeGregorio, M.W. Tecemotide: An antigen-specific cancer immunotherapy. Hum. Vacc. Immunother. 2014, 10, 3383–3393. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Wang, J.; Xu, Y.; Jiang, F.; Xu, L. L-blp25 as a peptide vaccine therapy in non-small cell lung cancer: A review. J. Thorac. Dis. 2014, 6, 1513–1520. [Google Scholar] [PubMed]
- Van Poelgeest, M.I.; Welters, M.J.; Vermeij, R.; Stynenbosch, L.F.; Loof, N.M.; Berends-van der Meer, D.M.; Lowik, M.J.; Hamming, I.L.; van Esch, E.M.; Hellebrekers, B.W.; et al. Vaccination against oncoproteins of hpv16 for noninvasive vulvar/vaginal lesions: Lesion clearance is related to the strength of the t-cell response. Clin. Cancer Res. 2016, 22, 2342–2350. [Google Scholar] [CrossRef] [PubMed]
- Welters, M.J.; Kenter, G.G.; Piersma, S.J.; Vloon, A.P.; Lowik, M.J.; Berends-van der Meer, D.M.; Drijfhout, J.W.; Valentijn, A.R.; Wafelman, A.R.; Oostendorp, J.; et al. Induction of tumor-specific cd4+ and cd8+ t-cell immunity in cervical cancer patients by a human papillomavirus type 16 e6 and e7 long peptides vaccine. Clin. Cancer Res. 2008, 14, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.K. Neoepitopes of cancers: Looking back, looking ahead. Cancer Immunol. Res. 2015, 3, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.W.; Hajjar, J.; Hwu, P.; Naing, A. Targeting the indoleamine 2,3-dioxygenase pathway in cancer. J. Immunother. Cancer 2015, 3, 51. [Google Scholar] [CrossRef] [PubMed]
- Braun, D.; Longman, R.S.; Albert, M.L. A two-step induction of indoleamine 2,3 dioxygenase (ido) activity during dendritic-cell maturation. Blood 2005, 106, 2375–2381. [Google Scholar] [CrossRef] [PubMed]
- Motz, G.T.; Coukos, G. Deciphering and reversing tumor immune suppression. Immunity 2013, 39, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Bouzin, C.; Brouet, A.; De Vriese, J.; Dewever, J.; Feron, O. Effects of vascular endothelial growth factor on the lymphocyte-endothelium interactions: Identification of caveolin-1 and nitric oxide as control points of endothelial cell anergy. J. Immunol. 2007, 178, 1505–1511. [Google Scholar] [CrossRef] [PubMed]
- Gulley, J.L.; Madan, R.A.; Schlom, J. Impact of tumour volume on the potential efficacy of therapeutic vaccines. Curr. Oncol. 2011, 18, e150–e157. [Google Scholar] [CrossRef] [PubMed]
- Bol, K.F.; Aarntzen, E.H.; Hout, F.E.; Schreibelt, G.; Creemers, J.H.; Lesterhuis, W.J.; Gerritsen, W.R.; Grunhagen, D.J.; Verhoef, C.; Punt, C.J.; et al. Favorable overall survival in stage iii melanoma patients after adjuvant dendritic cell vaccination. Oncoimmunology 2016, 5, e1057673. [Google Scholar] [CrossRef] [PubMed]
- Meissner, M.; Reichert, T.E.; Kunkel, M.; Gooding, W.; Whiteside, T.L.; Ferrone, S.; Seliger, B. Defects in the human leukocyte antigen class i antigen processing machinery in head and neck squamous cell carcinoma: Association with clinical outcome. Clin Cancer Res 2005, 11, 2552–2560. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.M.; Jordanova, E.S.; Kenter, G.G.; Ferrone, S.; Fleuren, G.J. Association of antigen processing machinery and hla class i defects with clinicopathological outcome in cervical carcinoma. Cancer Immunol. Immunother. 2008, 57, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Atkins, D.; Ferrone, S.; Schmahl, G.E.; Storkel, S.; Seliger, B. Down-regulation of hla class i antigen processing molecules: An immune escape mechanism of renal cell carcinoma? J. Urol. 2004, 171, 885–889. [Google Scholar] [CrossRef] [PubMed]
- Seliger, B.; Ritz, U.; Abele, R.; Bock, M.; Tampe, R.; Sutter, G.; Drexler, I.; Huber, C.; Ferrone, S. Immune escape of melanoma: First evidence of structural alterations in two distinct components of the mhc class i antigen processing pathway. Cancer Res. 2001, 61, 8647–8650. [Google Scholar] [PubMed]
- Seliger, B.; Stoehr, R.; Handke, D.; Mueller, A.; Ferrone, S.; Wullich, B.; Tannapfel, A.; Hofstaedter, F.; Hartmann, A. Association of hla class i antigen abnormalities with disease progression and early recurrence in prostate cancer. Cancer Immunol. Immunother. 2010, 59, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, S.C.; Peters, H.L.; Taguchi, A.; Katayama, H.; Wang, H.; Momin, A.; Jolly, M.K.; Celiktas, M.; Rodriguez-Canales, J.; Liu, H.; et al. Immunoproteasome deficiency is a feature of non-small cell lung cancer with a mesenchymal phenotype and is associated with a poor outcome. Proc. Natl. Acad. Sci. USA 2016, 113, E1555–E1564. [Google Scholar] [CrossRef] [PubMed]
- Reeves, E.; James, E. Antigen processing and immune regulation in the response to tumours. Immunology 2017, 150, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.M.; Jordanova, E.S.; Corver, W.E.; van Wezel, T.; Uh, H.W.; Kenter, G.G.; Fleuren, G.J. Single nucleotide polymorphisms in antigen processing machinery component erap1 significantly associate with clinical outcome in cervical carcinoma. Gene Chromosome Cancer 2009, 48, 410–418. [Google Scholar] [CrossRef] [PubMed]
| Type of Tumour Antigen | Examples (Ref.) | |
|---|---|---|
| Tumour associated | Overexpression | HER2 [32] TERT [33] |
| Tissue differentiation | PSA [34] Mammaglobin-A [35] Tyrosinase [36] | |
| Cancer-germline | MAGE [37] BAGE [38] NY-ESO-1 [26] | |
| Oncofetal | CEA [39] MUC-1 [39] TPBG [16] | |
| Tumour specific | Oncogenic viral | HPV [21] HBV [20] HHV-8 [30] |
| Neoantigens | BRAF V600E [40] KRAS G12D [41] | |
| Clinical Trials.gov Identifier; Phase | Tumour Type; Setting | Intervention | Mode of Action | Results from Previous Vaccine Trials |
|---|---|---|---|---|
| NCT03328026; Phase 1/2 | Breast cancer; palliative | SV-BR-1-GM id., pembrolizumab, ipilimumab, cyclophosphamide, interferon | GM-CSF secreting whole cell vaccine, anti-PD-1, anti-CTLA-4, chemotherapy, cytokine | Clinical responses seen in monotherapy with SV-BR-1-GM (phase 1 NCT03066947) |
| NCT02826434; Phase 1 | TNBC; adjuvant | PVX-410 im., durvalumab | Peptide vaccine, anti-PD-L1 | – |
| NCT03199040; Phase 1 | TNBC; adjuvant | Neoantigen DNA vaccine im. alone or plus durvalumab | DNA vaccine, anti-PD-L1 | – |
| NCT03362060; Phase 1 | TNBC; palliative | PVX-410 sc. alone or plus pembrolizumab | Peptide vaccine, anti-PD-1 | – |
| NCT02451982; Phase 1/2 | Pancreatic cancer; neoadjuvant and adjuvant | GVAX id., cyclophosphamide alone or plus nivolumab | GM-CSF secreting whole tumour cell vaccine, chemotherapy, anti-PD-1 | Palliative phase 2 study (ECLIPSE) with GVAX and cyclophosphamide and CRS-207 showed no OS compared to standard of care (ASCO GI abstract 2017) |
| NCT03050814; Phase 2 | Colorectal cancer; palliative | Standard of care alone or plus ad-CEA vaccine sc. and avelumab | Adenovirus vector vaccine expressing CEA, anti-PD-L1 | Ad-CEA induced T cell mediated immune response measured by IFNγ Elispot (phase 1) [53] |
| NCT03152565; Phase 1/2 | Colorectal cancer; palliative | ADC id., avelumab | Autologous dendritic cell vaccine, anti-PD-L1 | – |
| NCT03029403; Phase 2 | Ovarian, tubal, peritoneal; palliative | DPX survivac sc., cyclophosphamide, pembrolizumab | Survivin targeting peptide vaccine, chemotherapy, anti-PD-1 | DPX induced CD8+ T-cell responses, measured by IFNγ Elispot (phase 1) [54] |
| NCT02499835; Phase 1 | Prostate cancer; palliative | pTVG-HP id., pembrolizumab | Plasmid DNA vaccine encoding prostatic acid phosphatase, anti-PD-1 | pTVG-HP induced CD8+ T-cell responses, measured by IFNγ Elispot (phase 1) [55] |
| NCT02933255; Phase 1/2 | Prostate cancer; metastatic and localized | PROSTVAC sc., nivolumab | Poxvirus expressing PSA vaccine, anti-PD-1 | PROSTVAC alone no difference in OS (phase 3, ASCO abstract 2018) |
| NCT02808143; Phase 1 | Non-muscle-invasive bladder cancer; recurrent | BCG, pembrolizumab intravesically | BCG, anti-PD-1 | – |
| NCT03164772; Phase 1/2 | NSCLC; palliative | BI 1361849 id., durvalumab alone or plus tremelimumab | mRNA vaccine, anti-PDL-1, anti-CTLA-4 | – |
| NCT02879760; Phase 1/2 | NSCLC; palliative | Ad-MAGEA3 im., MG1-MAGEA3 iv., pembrolizumab | Adenovirus vaccine expressing MAGEA3, Maraba virus expressing MAGEA3, anti-PD-1 | – |
| NCT03380871; Phase 1 | NSCLC; palliative | NEO-PV-01 sc., pembrolizumab, carboplatin, pemetrexed | Personalized cancer vaccine, anti-PD-1, chemotherapy | – |
| NCT02955290; Phase 1/2 | NSCLC; palliative | CIMAvax im., nivolumab | Peptide vaccine containing recombinant human EGF, anti-PD-1 | Phase 2 study of CIMAvax showed increased OS for patients with good anti-EGF antibody response [56] |
| NCT02823990; Phase 2 | NSCLC; palliative | TG4010 sc., nivolumab | Ankara-virus vaccine expressing MUC1and IL-2, anti-PD-1 | Phase 2 of first line chemo with TG4010 or placebo showed improved PFS for the vaccine arm [57] |
| NCT02439450; Phase 1/2 | NSCLC; palliative | Viagenpumatucel-L id., nivolumab | gp96-Ig secreting lung cancer cells, anti-PD-1 | – |
| NCT03406715; Phase 2 | SCLC; palliative | Ad.p53-DC id., nivolumab, ipilimumab | Autologous dendritc cell based p53 vaccine, anti-PD-1, anti-CTLA-4 | Ad.p53-DC induced immune-cell responses, measured by IFNγ Elispot (phase 1) [58] |
| NCT02775292; Phase 1 | Solid tumours; palliative | NY-ESO-1 TCR iv., NY-ESO-1 DC id., nivolumab, cyclophosphamide, fludarabine | Gene modified T cells, peptide-pulsed dendritic cells, anti-PD-1, chemotherapy | – |
| NCT03289962; Phase 1 | Solid tumours; palliative | RO7198457 iv., atezolizumab | Personalized RNA mutanome vaccine, anti-PD-L1 | RO7198457 induced T cell mediated immune response measured by IFNγ Elispot (phase 1) [59] |
| NCT03311334; Phase 1 | Solid tumours; palliative | DSP-7888 id., nivolumab or atezolizumab | WT1 protein-derived peptide vaccine, anti-PD-1 or anti-PD-L1 | – |
| NCT03162224; Phase 1/2 | Head and neck cancer; palliative | MEDI0457 im., durvalumab | HPV DNA vaccine, anti-PD-L1 | – |
| NCT03260023; Phase 1/2 | HPV-16 positive cancer; palliative | TG4001 sc., avelumab | Modified vaccinia of Ankara-virus expressing HPV 16 and IL-2, anti PD-L1 | Clinical responses seen in patients with HPV-16 related cervical intraepithelial neoplasia after TG4001 injections (phase 2) [60] |
| NCT03047928; Phase 1/2 | Melanoma; palliative | PD-L1/IDO vaccine sc., nivolumab | Peptide based vaccine, anti-PD-1 | – |
| NCT02385669; Phase 1/2 | Melanoma; neoadjuvant, adjuvant, palliative | 6MHP, ipilimumab | Melanoma-associated helper peptide vaccine, anti-CTLA-4 | 6MHP decreased CD8+ T-cell responses, measured by IFNγ Elispot (phase 1) [61] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terbuch, A.; Lopez, J. Next Generation Cancer Vaccines—Make It Personal! Vaccines 2018, 6, 52. https://doi.org/10.3390/vaccines6030052
Terbuch A, Lopez J. Next Generation Cancer Vaccines—Make It Personal! Vaccines. 2018; 6(3):52. https://doi.org/10.3390/vaccines6030052
Chicago/Turabian StyleTerbuch, Angelika, and Juanita Lopez. 2018. "Next Generation Cancer Vaccines—Make It Personal!" Vaccines 6, no. 3: 52. https://doi.org/10.3390/vaccines6030052
APA StyleTerbuch, A., & Lopez, J. (2018). Next Generation Cancer Vaccines—Make It Personal! Vaccines, 6(3), 52. https://doi.org/10.3390/vaccines6030052





