Activation-induced Markers Detect Vaccine-Specific CD4+ T Cell Responses Not Measured by Assays Conventionally Used in Clinical Trials
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Vaccine Trial
2.2. Ethics and Regulatory Approval
2.3. Sample Handling
2.4. IFNγ ELISpot
2.5. Intracellular Cytokine Staining (ICS)
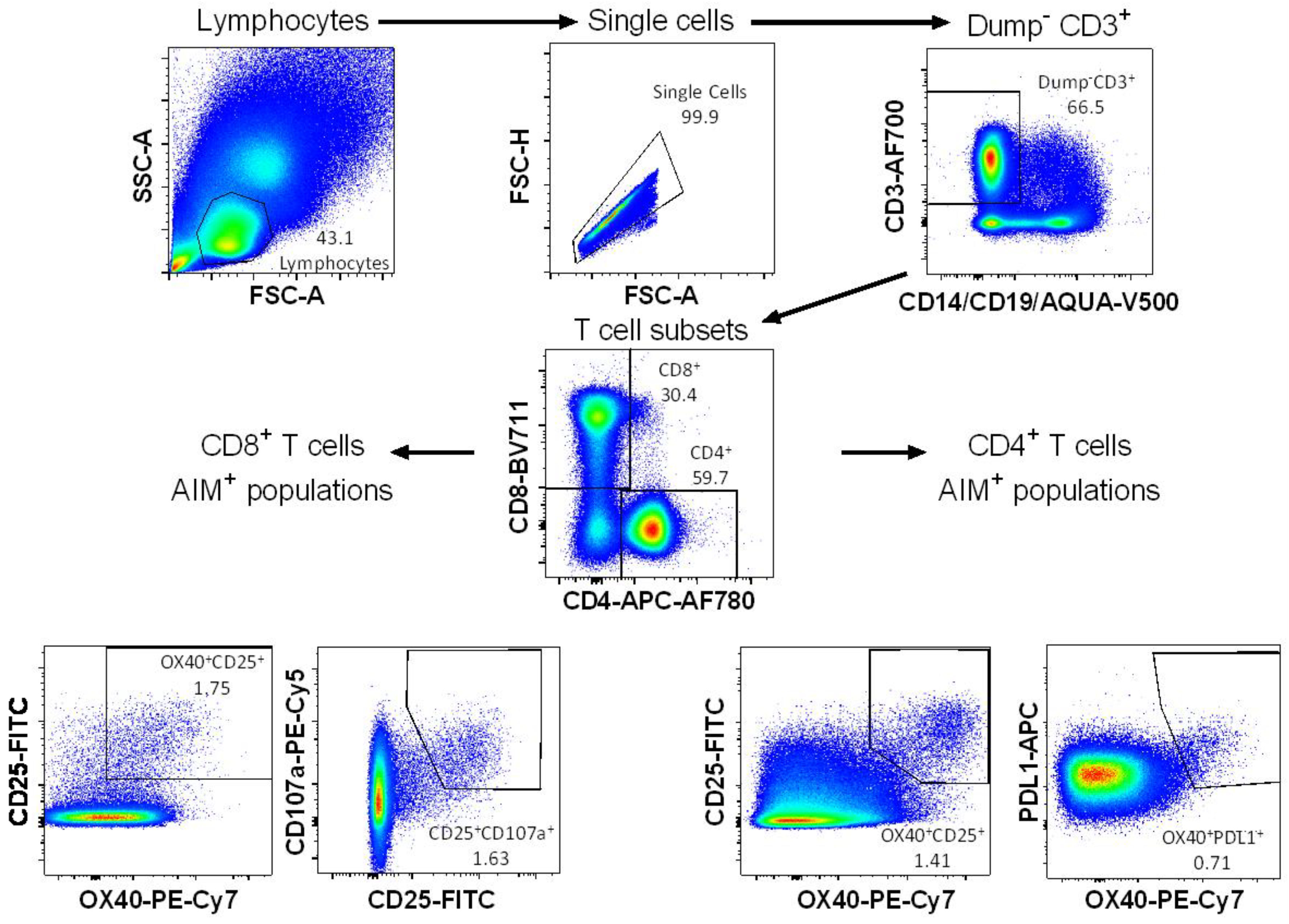
2.6. Activation-Induced Markers (AIM) Assay
2.7. Statistical Analysis
3. Results
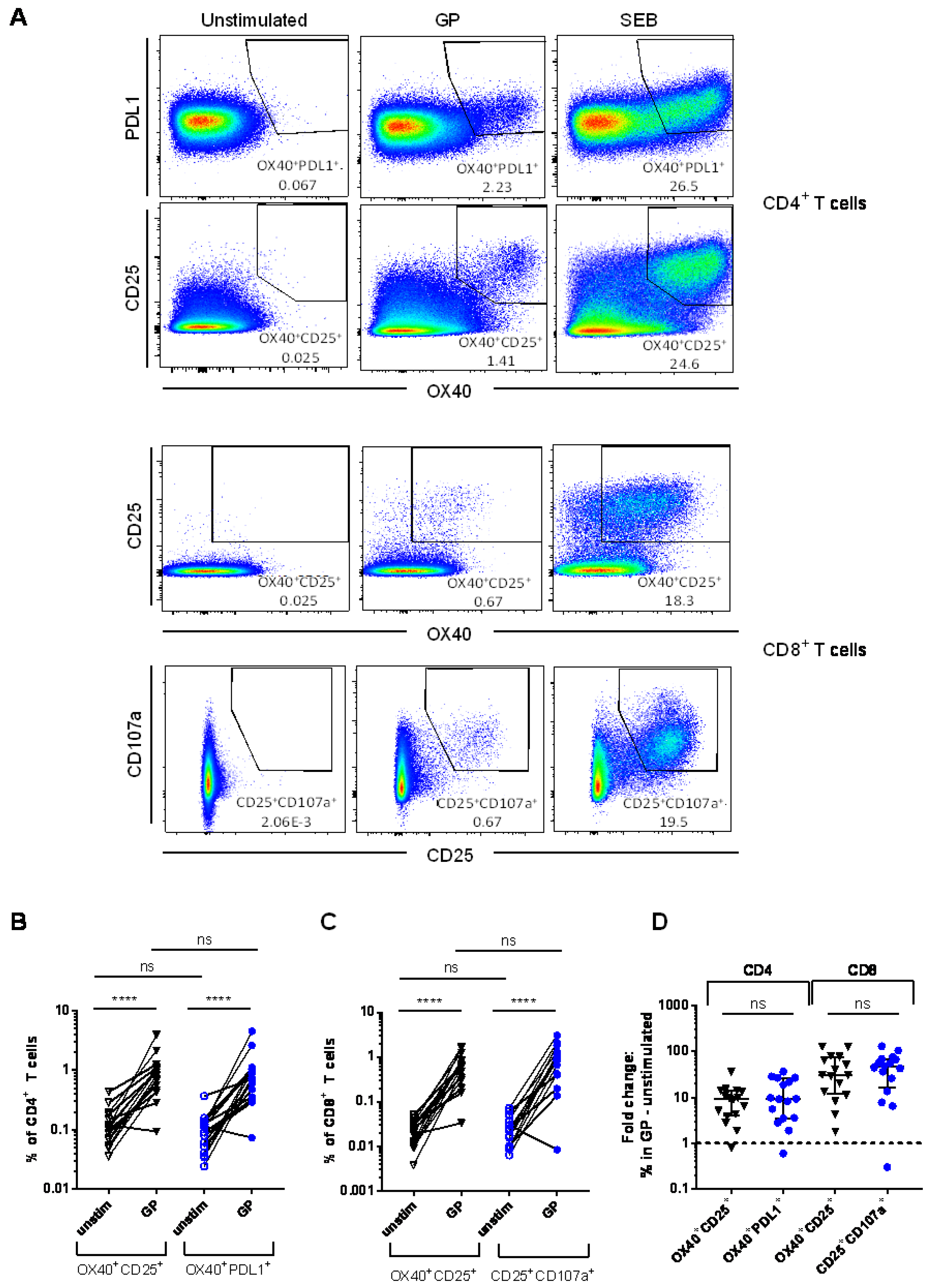
3.1. Detection of Vaccine-Specific T cells Using Activation-Induced Markers
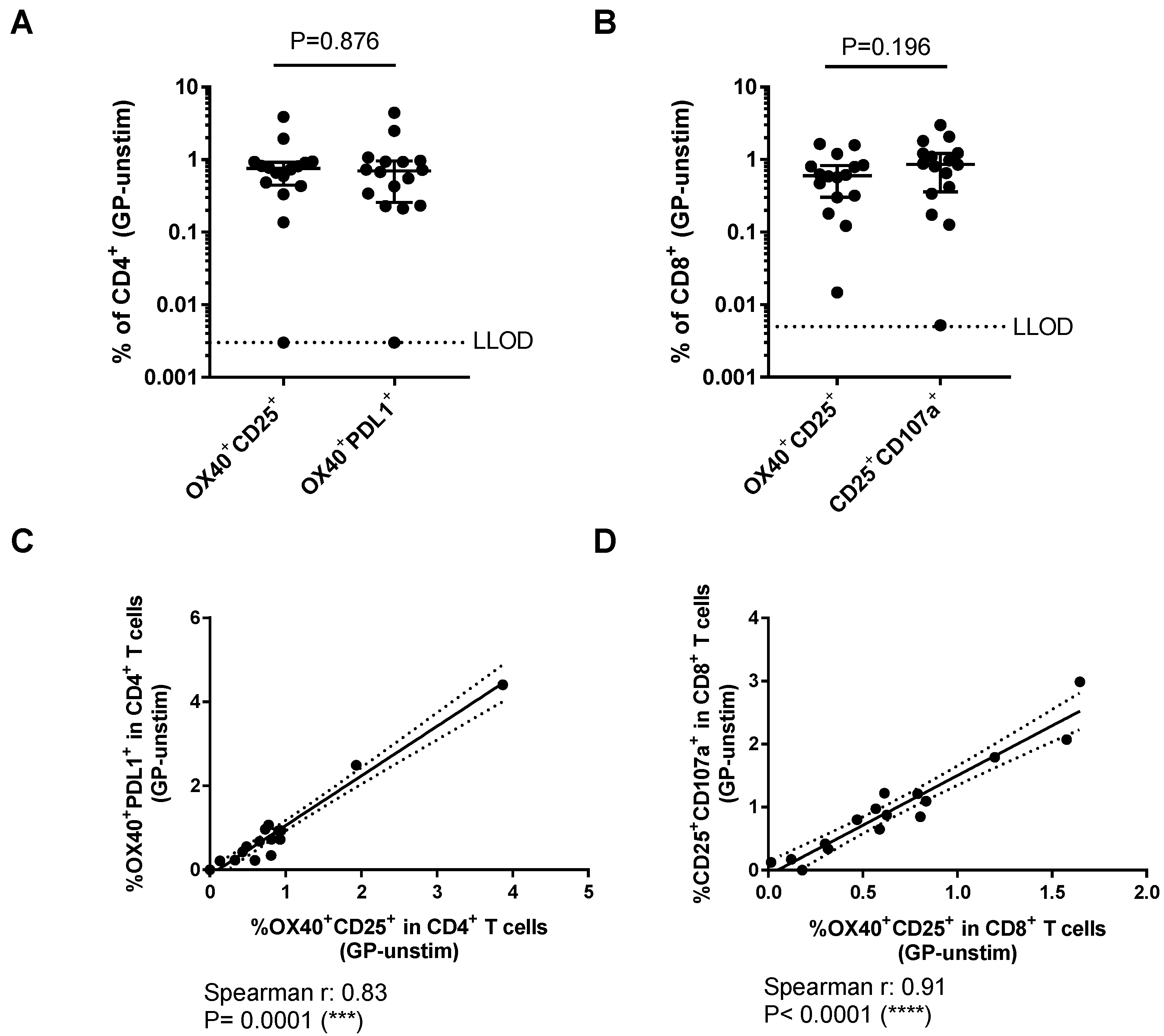
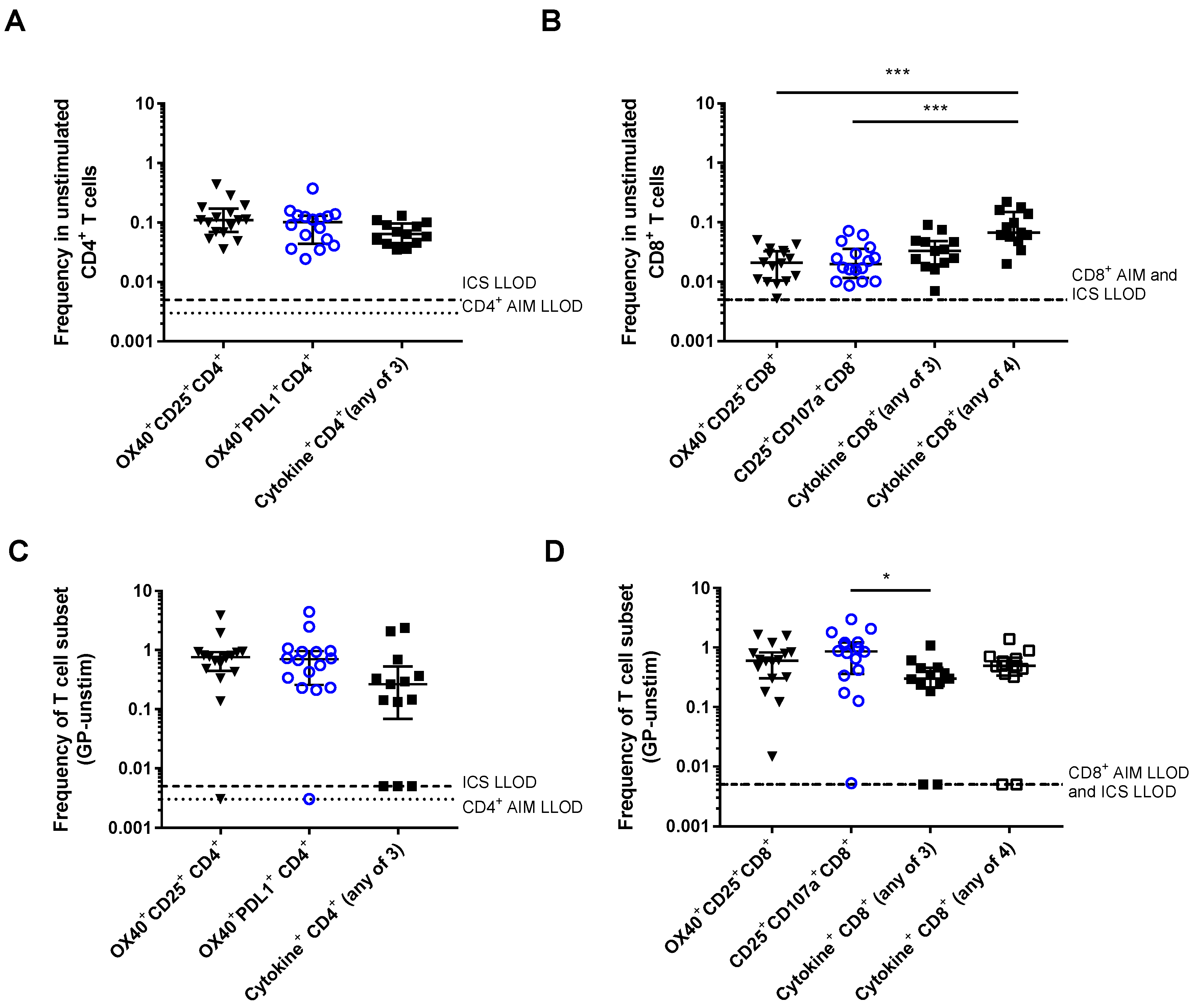
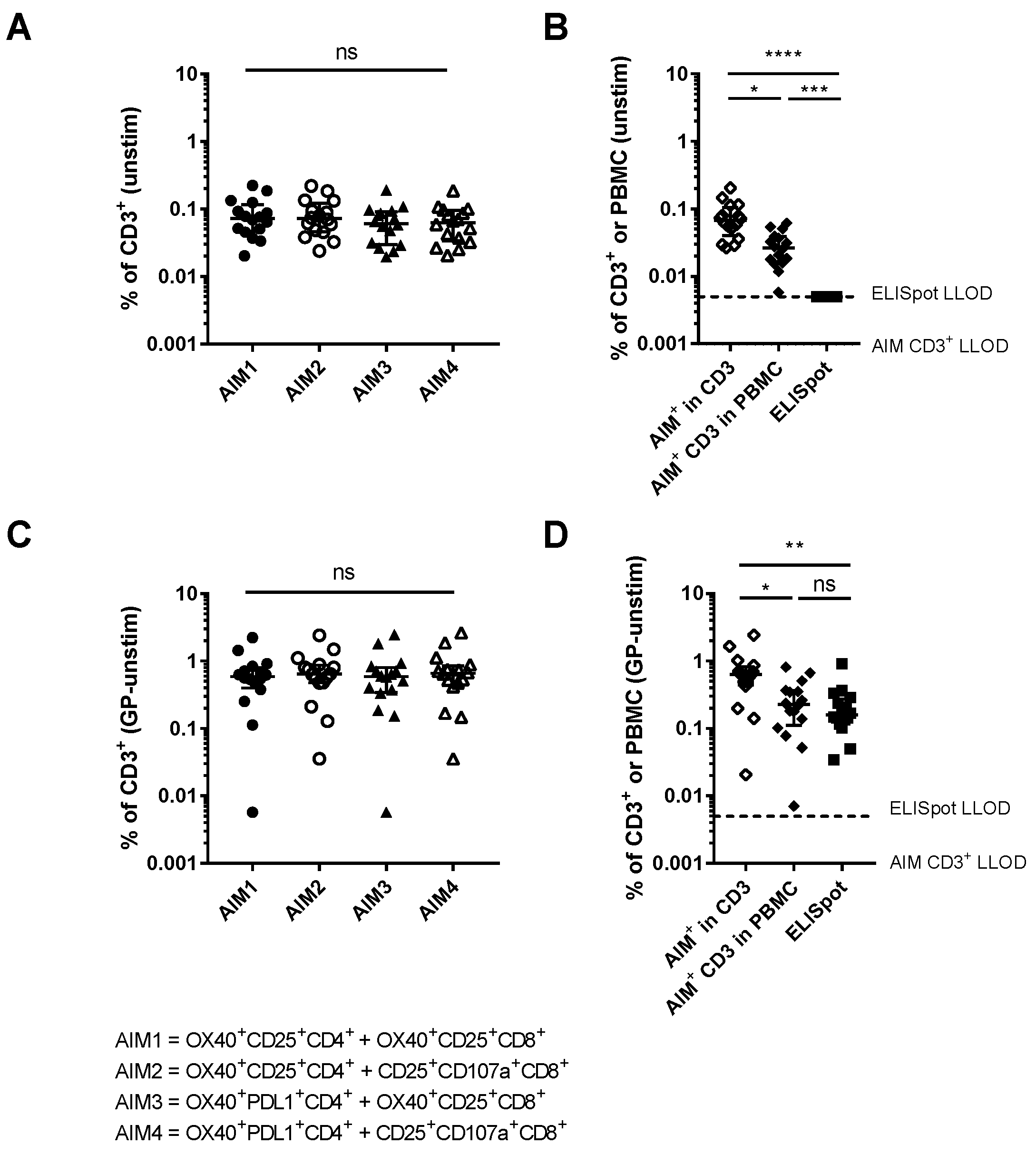
3.2. Comparison of Different Activation-Induced Markers for Detection of Vaccine-Specific T Cells
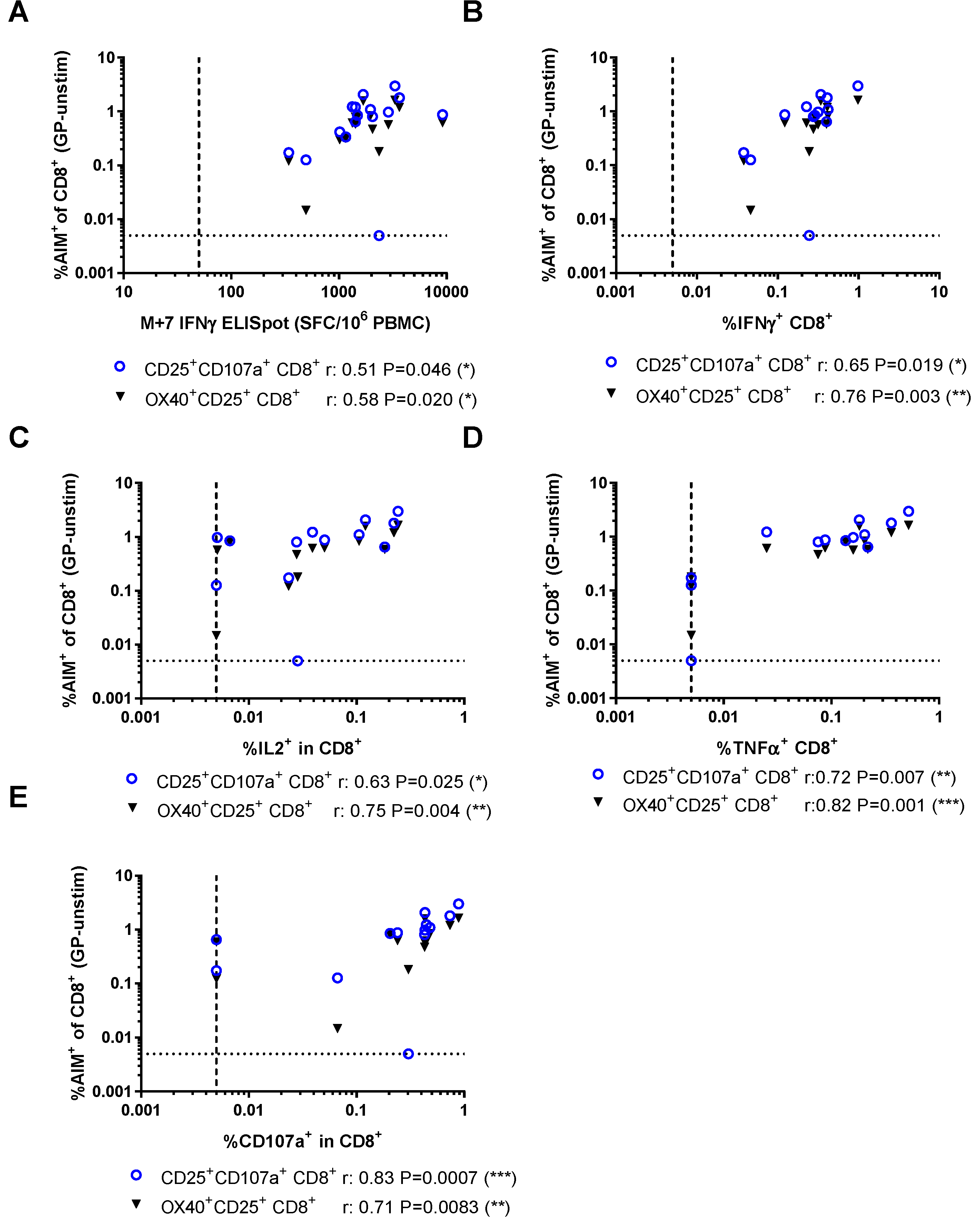
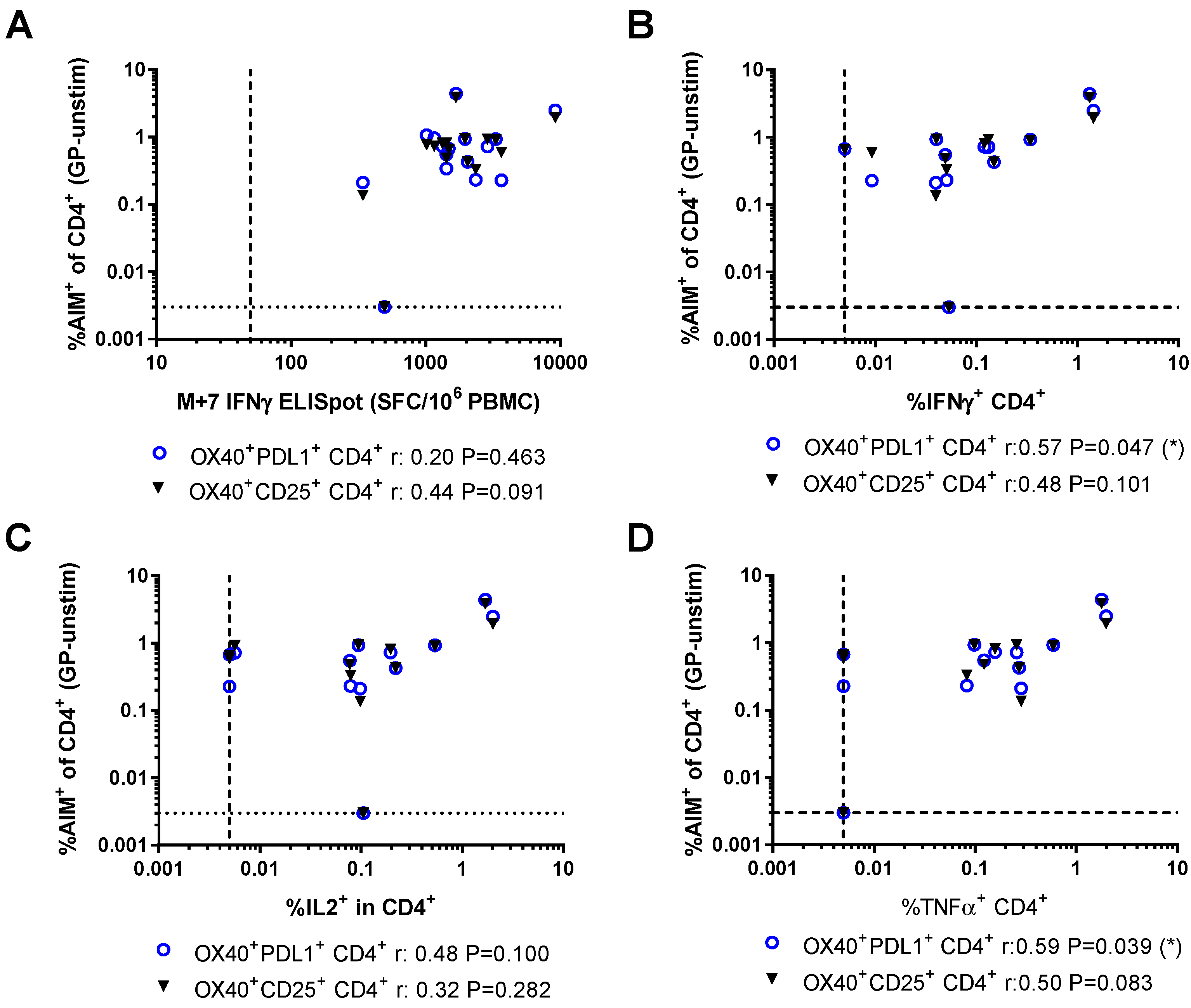
3.3. Relationship Between AIM Assays and Other Measures of Vaccine Immunogenicity
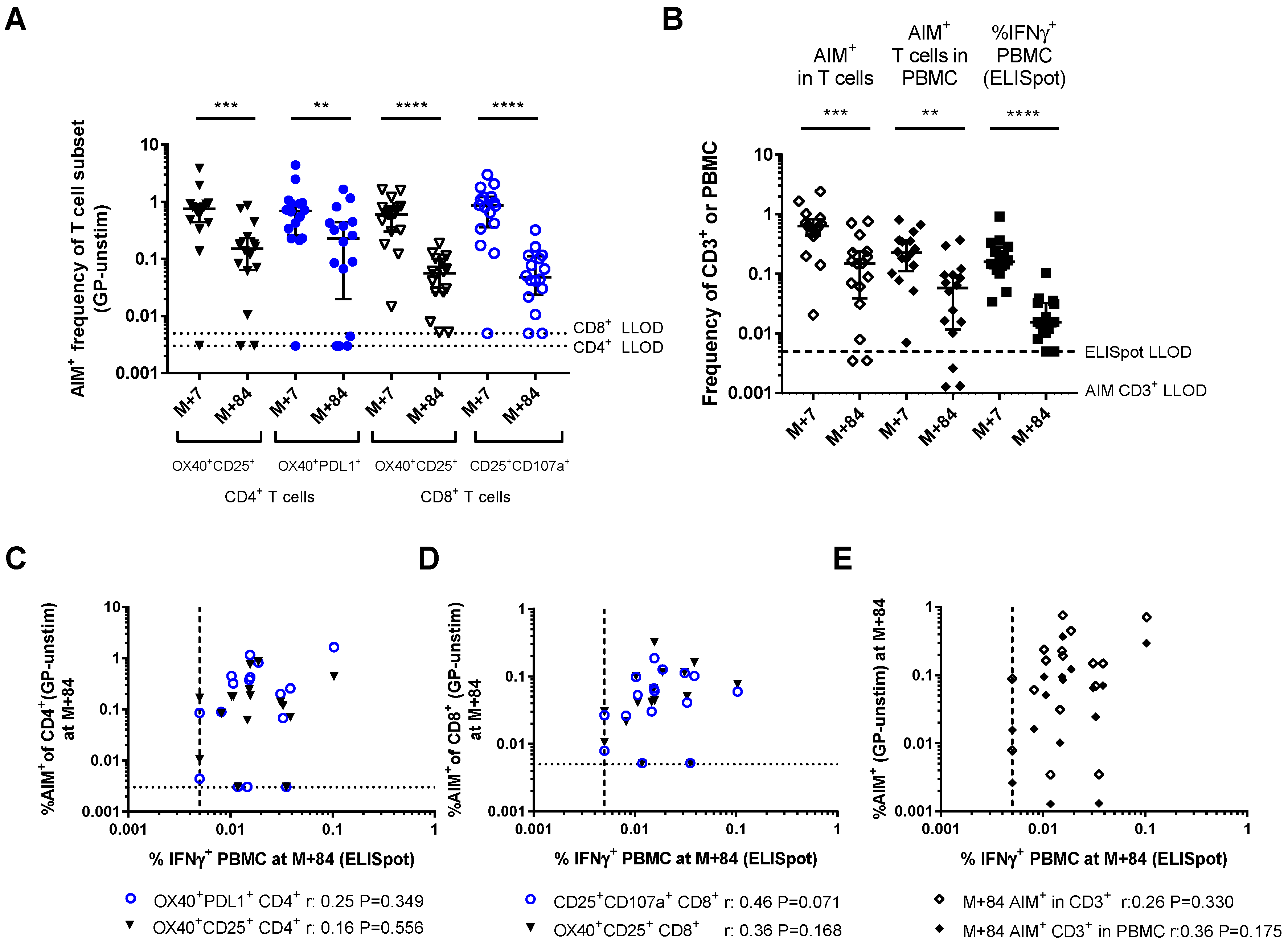
3.4. Activation-Induced Markers as a Measure of Durable Vaccine Responses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Plotkin, S.A. Vaccines: Correlates of vaccine-induced immunity. Clin. Infect. Dis. 2008, 47, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, S.A. Correlates of protection induced by vaccination. Clin. Vaccine Immunol. 2010, 17, 1055–1065. [Google Scholar] [CrossRef] [PubMed]
- Mascola, J.R.; Haynes, B.F. HIV-1 neutralizing antibodies: Understanding nature’s pathways. Immunol. Rev. 2013, 254, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Doria-Rose, N.A. HIV neutralizing antibodies: Clinical correlates and implications for vaccines. J. Infect. Dis. 2010, 201, 981–983. [Google Scholar] [CrossRef] [PubMed]
- Schlingmann, B.; Castiglia, K.R.; Stobart, C.C.; Moore, M.L. Polyvalent vaccines: High-maintenance heroes. PLoS Pathog. 2018, 14, e1006904. [Google Scholar] [CrossRef] [PubMed]
- Voss, G.; Casimiro, D.; Neyrolles, O.; Williams, A.; Kaufmann, S.H.E.; McShane, H.; Hatherill, M.; Fletcher, H.A. Progress and challenges in TB vaccine development. F1000Res 2018, 7, 199. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.V. Vaccines against malaria. Philos. Trans. R Soc. Lond. B Biol. Sci. 2011, 366, 2806–2814. [Google Scholar] [CrossRef] [PubMed]
- Röhn, T.A.; Bachmann, M.F. Vaccines against non-communicable diseases. Curr. Opin. Immunol. 2010, 22, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Fishman, M. Challenges facing the development of cancer vaccines. Methods Mol. Biol. 2014, 1139, 543–553. [Google Scholar] [PubMed]
- Gilbert, S.C. T-cell-inducing vaccines—What’s the future. Immunology 2012, 135, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Ten Brinke, A.; Marek-Trzonkowska, N.; Mansilla, M.J.; Turksma, A.W.; Piekarska, K.; Iwaszkiewicz-Grześ, D.; Passerini, L.; Locafaro, G.; Puñet-Ortiz, J.; van Ham, S.M.; et al. Monitoring T-cell responses in translational studies: Optimization of dye-based proliferation assay for evaluation of antigen-specific responses. Front. Immunol. 2017, 8, 1870. [Google Scholar] [CrossRef] [PubMed]
- Saade, F.; Gorski, S.A.; Petrovsky, N. Pushing the frontiers of T-cell vaccines: Accurate measurement of human T-cell responses. Expert Rev. Vaccines 2012, 11, 1459–1470. [Google Scholar] [CrossRef] [PubMed]
- Slota, M.; Lim, J.B.; Dang, Y.; Disis, M.L. Elispot for measuring human immune responses to vaccines. Expert Rev. Vaccines 2011, 10, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.G.; Smits, K.; Joosten, S.A.; van Meijgaarden, K.E.; Satti, I.; Fletcher, H.A.; Caccamo, N.; Dieli, F.; Mascart, F.; McShane, H.; et al. Intracellular cytokine staining and flow cytometry: Considerations for application in clinical trials of novel tuberculosis vaccines. PLoS ONE 2015, 10, e0138042. [Google Scholar] [CrossRef] [PubMed]
- Shirai, A.; Holmes, K.; Klinman, D. Detection and quantitation of cells secreting IL-6 under physiologic conditions in BALB/c mice. J. Immunol. 1993, 150, 793–799. [Google Scholar] [PubMed]
- Moodie, Z.; Price, L.; Gouttefangeas, C.; Mander, A.; Janetzki, S.; Löwer, M.; Welters, M.J.; Ottensmeier, C.; van der Burg, S.H.; Britten, C.M. Response definition criteria for elispot assays revisited. Cancer Immunol. Immunother. 2010, 59, 1489–1501. [Google Scholar] [CrossRef] [PubMed]
- Britten, C.M.; Janetzki, S.; van der Burg, S.H.; Gouttefangeas, C.; Hoos, A. Toward the harmonization of immune monitoring in clinical trials: Quo vadis? Cancer Immunol. Immunother. 2008, 57, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Janetzki, S.; Panageas, K.S.; Ben-Porat, L.; Boyer, J.; Britten, C.M.; Clay, T.M.; Kalos, M.; Maecker, H.T.; Romero, P.; Yuan, J.; et al. Results and harmonization guidelines from two large-scale international elispot proficiency panels conducted by the cancer vaccine consortium (CVC/SVI). Cancer Immunol. Immunother. 2008, 57, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Coughlan, L.; Lambe, T. Measuring cellular immunity to influenza: Methods of detection, applications and challenges. Vaccines 2015, 3, 293–319. [Google Scholar] [CrossRef] [PubMed]
- Horton, H.; Thomas, E.P.; Stucky, J.A.; Frank, I.; Moodie, Z.; Huang, Y.; Chiu, Y.L.; McElrath, M.J.; de Rosa, S.C. Optimization and validation of an 8-color intracellular cytokine staining (ICS) assay to quantify antigen-specific T cells induced by vaccination. J. Immunol. Methods 2007, 323, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Parra, M.; Herrera, D.; Jácome, M.F.; Mesa, M.C.; Rodríguez, L.S.; Guzmán, C.; Angel, J.; Franco, M.A. Circulating rotavirus-specific T cells have a poor functional profile. Virology 2014, 468–470, 340–350. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, S.C.; Lu, F.X.; Yu, J.; Perfetto, S.P.; Falloon, J.; Moser, S.; Evans, T.G.; Koup, R.; Miller, C.J.; Roederer, M. Vaccination in humans generates broad T cell cytokine responses. J. Immunol. 2004, 173, 5372–5380. [Google Scholar] [CrossRef] [PubMed]
- Roederer, M.; Brenchley, J.M.; Betts, M.R.; de Rosa, S.C. Flow cytometric analysis of vaccine responses: How many colors are enough? Clin. Immunol. 2004, 110, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Seddiki, N.; Cook, L.; Hsu, D.C.; Phetsouphanh, C.; Brown, K.; Xu, Y.; Kerr, S.J.; Cooper, D.A.; Munier, C.M.; Pett, S.; et al. Human antigen-specific CD4+ CD25+ CD134+ CD39+ T cells are enriched for regulatory T cells and comprise a substantial proportion of recall responses. Eur. J. Immunol. 2014, 44, 1644–1661. [Google Scholar] [CrossRef] [PubMed]
- Dan, J.M.; Lindestam Arlehamn, C.S.; Weiskopf, D.; da Silva Antunes, R.; Havenar-Daughton, C.; Reiss, S.M.; Brigger, M.; Bothwell, M.; Sette, A.; Crotty, S. A cytokine-independent approach to identify antigen-specific human germinal center T follicular helper cells and rare antigen-specific CD4+ T cells in blood. J. Immunol. 2016, 197, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Havenar-Daughton, C.; Reiss, S.M.; Carnathan, D.G.; Wu, J.E.; Kendric, K.; Torrents de la Peña, A.; Kasturi, S.P.; Dan, J.M.; Bothwell, M.; Sanders, R.W.; et al. Cytokine-independent detection of antigen-specific germinal center T follicular helper cells in immunized nonhuman primates using a live cell activation-induced marker technique. J. Immunol. 2016, 197, 994–1002. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, P.K.; Yu, J.; Roederer, M. Live-cell assay to detect antigen-specific CD4+ T-cell responses by CD154 expression. Nat. Protoc. 2006, 1, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Frentsch, M.; Arbach, O.; Kirchhoff, D.; Moewes, B.; Worm, M.; Rothe, M.; Scheffold, A.; Thiel, A. Direct access to CD4+ T cells specific for defined antigens according to CD154 expression. Nat. Med. 2005, 11, 1118–1124. [Google Scholar] [CrossRef] [PubMed]
- Porichis, F.; Hart, M.G.; Zupkosky, J.; Barblu, L.; Kwon, D.S.; McMullen, A.; Brennan, T.; Ahmed, R.; Freeman, G.J.; Kavanagh, D.G.; et al. Differential impact of PD-1 and/or IL-10 blockade on HIV-1-specific CD4 T cell and antigen-presenting cell functions. J. Virol. 2014, 88, 2508–2518. [Google Scholar] [CrossRef] [PubMed]
- Betts, M.R.; Brenchley, J.M.; Price, D.A.; de Rosa, S.C.; Douek, D.C.; Roederer, M.; Koup, R.A. Sensitive and viable identification of antigen-specific CD8+ T cells by a flow cytometric assay for degranulation. J. Immunol. Methods 2003, 281, 65–78. [Google Scholar] [CrossRef]
- Wolfl, M.; Kuball, J.; Ho, W.Y.; Nguyen, H.; Manley, T.J.; Bleakley, M.; Greenberg, P.D. Activation-induced expression of CD137 permits detection, isolation, and expansion of the full repertoire of CD8+ T cells responding to antigen without requiring knowledge of epitope specificities. Blood 2007, 110, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Redmond, W.L.; Ruby, C.E.; Weinberg, A.D. The role of OX40-mediated co-stimulation in T-cell activation and survival. Crit. Rev. Immunol. 2009, 29, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Zaunders, J.J.; Munier, M.L.; Seddiki, N.; Pett, S.; Ip, S.; Bailey, M.; Xu, Y.; Brown, K.; Dyer, W.B.; Kim, M.; et al. High levels of human antigen-specific CD4+ T cells in peripheral blood revealed by stimulated coexpression of CD25 and CD134 (OX40). J. Immunol. 2009, 183, 2827–2836. [Google Scholar] [CrossRef] [PubMed]
- Venkatraman, N.; Anagnostou, N.; Bliss, C.; Bowyer, G.; Wright, D.; Lövgren-Bengtsson, K.; Roberts, R.; Poulton, I.; Lawrie, A.; Ewer, K.; et al. Safety and immunogenicity of heterologous prime-boost immunization with viral-vectored malaria vaccines adjuvanted with matrix-m™. Vaccine 2017, 35, 6208–6217. [Google Scholar] [CrossRef] [PubMed]
- Ewer, K.; Rampling, T.; Venkatraman, N.; Bowyer, G.; Wright, D.; Lambe, T.; Imoukhuede, E.B.; Payne, R.; Fehling, S.K.; Strecker, T.; et al. A monovalent chimpanzee adenovirus Ebola vaccine boosted with MVA. N. Engl. J. Med. 2016, 374, 1635–1646. [Google Scholar] [CrossRef] [PubMed]
- Rampling, T.; Ewer, K.J.; Bowyer, G.; Bliss, C.M.; Edwards, N.J.; Wright, D.; Payne, R.O.; Venkatraman, N.; de Barra, E.; Snudden, C.M.; et al. Safety and high level efficacy of the combination malaria vaccine regimen of RTS,S/AS01B with chimpanzee adenovirus 63 and modified vaccinia Ankara vectored vaccines expressing ME-TRAP. J. Infect. Dis. 2016, 214, 772–781. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D.A.; Honko, A.N.; Asiedu, C.; Trefry, J.C.; Lau-Kilby, A.W.; Johnson, J.C.; Hensley, L.; Ammendola, V.; Abbate, A.; Grazioli, F.; et al. Chimpanzee adenovirus vaccine generates acute and durable protective immunity against ebolavirus challenge. Nat. Med. 2014, 20, 1126–1129. [Google Scholar] [CrossRef] [PubMed]
- Bliss, C.M.; Bowyer, G.; Anagnostou, N.A.; Havelock, T.; Snudden, C.M.; Davies, H.; de Cassan, S.C.; Grobbelaar, A.; Lawrie, A.M.; Venkatraman, N.; et al. Assessment of novel vaccination regimens using viral vectored liver stage malaria vaccines encoding ME-TRAP. Sci. Rep. 2018, 8, 3390. [Google Scholar] [CrossRef] [PubMed]
- Reiss, S.; Baxter, A.E.; Cirelli, K.M.; Dan, J.M.; Morou, A.; Daigneault, A.; Brassard, N.; Silvestri, G.; Routy, J.P.; Havenar-Daughton, C.; et al. Comparative analysis of activation induced marker (AIM) assays for sensitive identification of antigen-specific CD4 T cells. PLoS ONE 2017, 12, e0186998. [Google Scholar] [CrossRef] [PubMed]
- Havenar-Daughton, C.; Lindqvist, M.; Heit, A.; Wu, J.E.; Reiss, S.M.; Kendric, K.; Bélanger, S.; Kasturi, S.P.; Landais, E.; Akondy, R.S.; et al. CXCL13 is a plasma biomarker of germinal center activity. Proc. Natl. Acad. Sci. USA. 2016, 113, 2702–2707. [Google Scholar] [CrossRef] [PubMed]
- Abbas, A.K.; Murphy, K.M.; Sher, A. Functional diversity of helper T lymphocytes. Nature 1996, 383, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Langenkamp, A.; Geginat, J.; Lanzavecchia, A. Functional subsets of memory T cells identified by CCR7 expression. Curr. Top. Microbiol. Immunol. 2000, 251, 167–171. [Google Scholar] [PubMed]
- Seder, R.A.; Ahmed, R. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nat. Immunol. 2003, 4, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Cohen, G.B.; Kaur, A.; Johnson, R.P. Isolation of viable antigen-specific CD4 T cells by CD40L surface trapping. J. Immunol. Methods 2005, 302, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Hengel, R.L.; Thaker, V.; Pavlick, M.V.; Metcalf, J.A.; Dennis, G.; Yang, J.; Lempicki, R.A.; Sereti, I.; Lane, H.C. Cutting edge: L-selectin (CD62L) expression distinguishes small resting memory CD4+ T cells that preferentially respond to recall antigen. J. Immunol. 2003, 170, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Macallan, D.C.; Borghans, J.A.; Asquith, B. Human T cell memory: A dynamic view. Vaccines 2017, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Webster, H.K.; Looareesuwan, S.; Supanaranond, W.; Phillips, R.E.; Chanthavanich, P.; Warrell, D.A. Antigen-specific immunosuppression in human malaria due to plasmodium falciparum. J. Infect. Dis. 1986, 153, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Van Braeckel-Budimir, N.; Kurup, S.P.; Harty, J.T. Regulatory issues in immunity to liver and blood-stage malaria. Curr. Opin. Immunol. 2016, 42, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Kurup, S.P.; Obeng-Adjei, N.; Anthony, S.M.; Traore, B.; Doumbo, O.K.; Butler, N.S.; Crompton, P.D.; Harty, J.T. Regulatory T cells impede acute and long-term immunity to blood-stage malaria through CTLA-4. Nat. Med. 2017, 23, 1220–1225. [Google Scholar] [CrossRef] [PubMed]
- Ford, T.; Wenden, C.; Mbekeani, A.; Dally, L.; Cox, J.H.; Morin, M.; Winstone, N.; Hill, A.V.S.; Gilmour, J.; Ewer, K.J. Cryopreservation-related loss of antigen-specific IFNγ producing CD4: Lessons from a malaria vaccine trial substudy. Vaccine 2017, 35, 1898–1906. [Google Scholar] [CrossRef] [PubMed]
- Janetzki, S.; Britten, C.M.; Kalos, M.; Levitsky, H.I.; Maecker, H.T.; Melief, C.J.; Old, L.J.; Romero, P.; Hoos, A.; Davis, M.M. “MIATA”—Minimal information about T cell assays. Immunity 2009, 31, 527–528. [Google Scholar] [CrossRef] [PubMed]
- Maecker, H.T.; Hassler, J.; Payne, J.K.; Summers, A.; Comatas, K.; Ghanayem, M.; Morse, M.A.; Clay, T.M.; Lyerly, H.K.; Bhatia, S.; et al. Precision and linearity targets for validation of an IFNγ ELISPOT, cytokine flow cytometry, and tetramer assay using CMV peptides. BMC Immunol. 2008, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- Nomura, L.; Maino, V.C.; Maecker, H.T. Standardization and optimization of multiparameter intracellular cytokine staining. Cytometry A 2008, 73, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, K.; Verma, S.; Ellner, J.J.; Salgame, P. Quest for correlates of protection against tuberculosis. Clin. Vaccine Immunol. 2015, 22, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Valletta, J.J.; Recker, M. Identification of immune signatures predictive of clinical protection from malaria. PLoS Comput. Biol. 2017, 13, e1005812. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.A.; Angel, J.; Greenberg, H.B. Immunity and correlates of protection for rotavirus vaccines. Vaccine 2006, 24, 2718–2731. [Google Scholar] [CrossRef] [PubMed]
- Lumsden, J.M.; Schwenk, R.J.; Rein, L.E.; Moris, P.; Janssens, M.; Ofori-Anyinam, O.; Cohen, J.; Kester, K.E.; Heppner, D.G.; Krzych, U. Protective immunity induced with the RTS, S/AS vaccine is associated with IL-2 and TNF-α producing effector and central memory CD4 T cells. PLoS ONE 2011, 6, e20775. [Google Scholar] [CrossRef] [PubMed]
| Assay/Markers | Lower Limit of Detection (% of Subset) | Frequency in Unstimulated (% of Subset) | Antigen-Specific Signal at M+7 (GP-Unstim, % of Subset) | Antigen-Specific Signal at M+84 (GP-Unstim, % of Subset) |
|---|---|---|---|---|
| ELISpot | 0.005 | 0.005 | 0.159 (0.120–0.275) | 0.015 (0.010–0.032) |
| ICS: CD4+ “any of three” | 0.005 | 0.064 (0.045–0.096) | 0.265 (0.069–0.527) | NA |
| ICS: CD8+ “any of three” | 0.005 | 0.033 (0.020–0.048) | 0.298 (0.210–0.456) | NA |
| ICS: CD8+ “any of four” | 0.005 | 0.067 (0.053–0.150) | 0.492 (0.341–0.676) | NA |
| AIM: CD4+ OX40+CD25+ | 0.003 | 0.110 (0.069–0.172) | 0.753 (0.445–0.924) | 0.153 (0.064–0.231) |
| AIM: CD4+ OX40+PDL1+ | 0.003 | 0.102 (0.044–0.131) | 0.700 (0.259–0.961) | 0.230 (0.020–0.443) |
| AIM: CD8+ OX40+CD25+ | 0.005 | 0.021 (0.010–0.033) | 0.601 (0.304–0.826) | 0.056 (0.026–0.101) |
| AIM: CD8+ CD25+CD107a+ | 0.005 | 0.020 (0.012–0.036) | 0.861 (0.359–1.219) | 0.048 (0.024–0.112) |
| AIM: CD3+ (average of marker combinations) | 0.001 | 0.067 (0.040–0.104) | 0.633 (0.443–0.820) | 0.150 (0.039–0.236) |
| AIM+CD3+ (average of marker combinations) in PBMC | 0.001 | 0.026 (0.016–0.039) | 0.227 (0.111–0.364) | 0.058 (0.012–0.095) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bowyer, G.; Rampling, T.; Powlson, J.; Morter, R.; Wright, D.; Hill, A.V.S.; Ewer, K.J. Activation-induced Markers Detect Vaccine-Specific CD4+ T Cell Responses Not Measured by Assays Conventionally Used in Clinical Trials. Vaccines 2018, 6, 50. https://doi.org/10.3390/vaccines6030050
Bowyer G, Rampling T, Powlson J, Morter R, Wright D, Hill AVS, Ewer KJ. Activation-induced Markers Detect Vaccine-Specific CD4+ T Cell Responses Not Measured by Assays Conventionally Used in Clinical Trials. Vaccines. 2018; 6(3):50. https://doi.org/10.3390/vaccines6030050
Chicago/Turabian StyleBowyer, Georgina, Tommy Rampling, Jonathan Powlson, Richard Morter, Daniel Wright, Adrian V.S. Hill, and Katie J. Ewer. 2018. "Activation-induced Markers Detect Vaccine-Specific CD4+ T Cell Responses Not Measured by Assays Conventionally Used in Clinical Trials" Vaccines 6, no. 3: 50. https://doi.org/10.3390/vaccines6030050
APA StyleBowyer, G., Rampling, T., Powlson, J., Morter, R., Wright, D., Hill, A. V. S., & Ewer, K. J. (2018). Activation-induced Markers Detect Vaccine-Specific CD4+ T Cell Responses Not Measured by Assays Conventionally Used in Clinical Trials. Vaccines, 6(3), 50. https://doi.org/10.3390/vaccines6030050





