SARS-COV-2 Vaccination Response in Non-Domestic Species Housed at the Toronto Zoo
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Vaccination and Inclusion Criteria
2.2. Surrogate Virus Neutralization Test (sVNT)
2.3. Plaque-Reduction Neutralization Test 70% (PRNT70)
2.4. Data Analysis and Descriptive Statistics
3. Results
3.1. Animal Demographics and Serum Samples
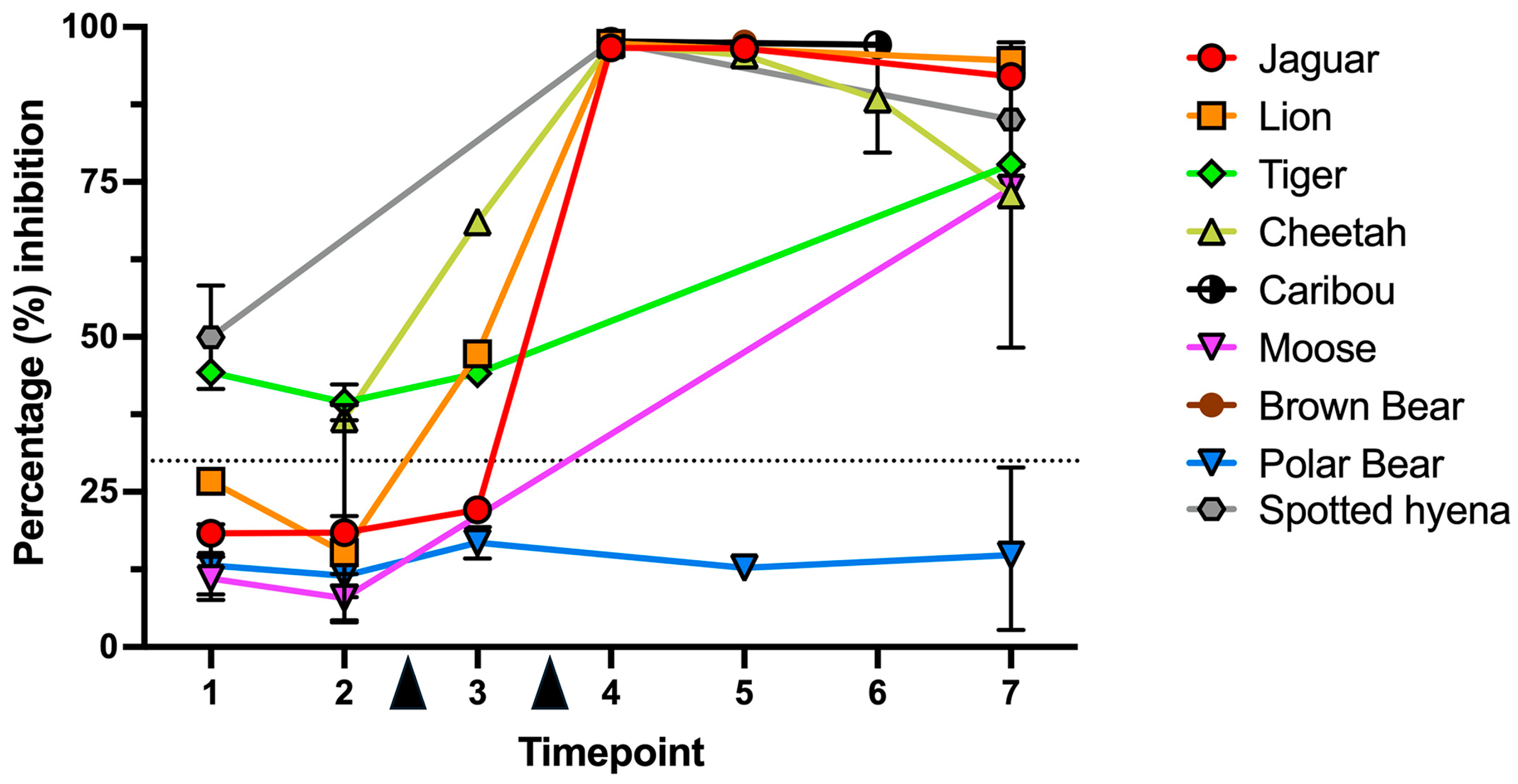
3.2. Surrogate Virus Neutralization Test (sVNT)
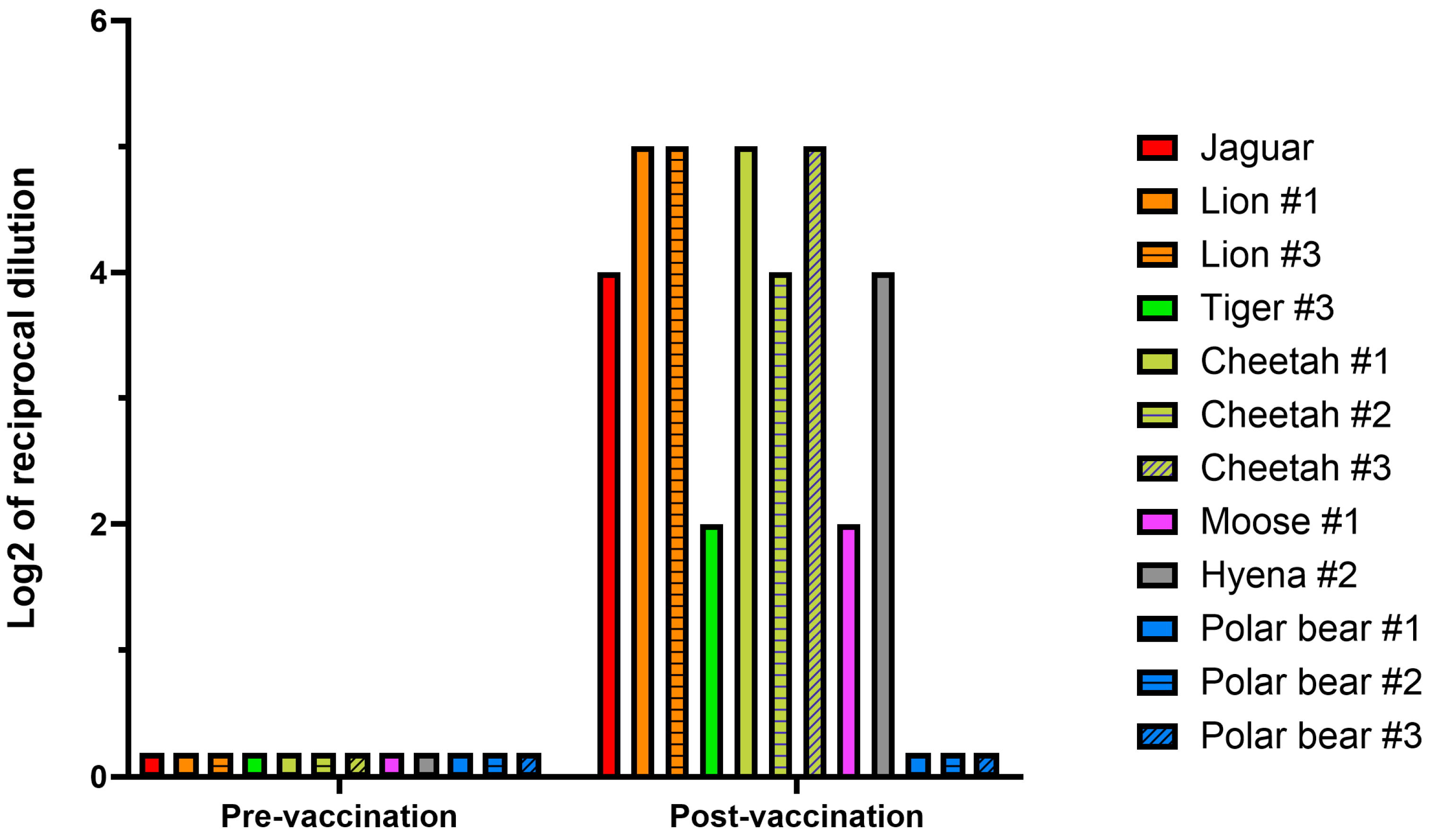
3.3. Plaque-Reduction Neutralization Test 70% (PRNT70)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACE2 | Angiotensin converting enzyme 2 |
| CFIA | Canadian Food Inspection Agency |
| COVID-19 | Coronavirus disease 2019 |
| PRNT70 | Plaque-reduction neutralization test 70% |
| RBD | Receptor-binding domain |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| sVNT | Surrogate virus neutralization test |
| TMPRSS-2 | Transmembrane serine protease 2 |
| TP | Timepoint |
| TZ | Toronto Zoo |
References
- Allender, M.C.; Adkesson, M.J.; Langan, J.N.; Delk, K.W.; Meehan, T.; Aitken-Palmer, C.; McEntire, M.M.; Killian, M.L.; Torchetti, M.; Morales, S.A.; et al. Multi-species outbreak of SARS-CoV-2 Delta variant in a zoological institution, with detection in two new families of carnivores. Transbound. Emerg. Dis. 2022, 69, e3060–e3075. [Google Scholar] [CrossRef] [PubMed]
- European Association of Zoo and Wildlife Veterinarians Infectious Diseases Working Group. Transmissible Disease Handbook. Science-Based Facts & Knowledge About Wild Animals, Zoos and SARS-CoV-2 Virus (Chapter 3.6). European Association of Zoo and Wildlife Veterinarians. 2022. Available online: https://www.eazwv.org/page/inf_handbook (accessed on 27 August 2025).
- Palmer, M.V.; Martins, M.; Falkenberg, S.; Buckley, A.; Caserta, L.C.; Mitchell, P.K.; Cassmann, E.D.; Rollins, A.; Zylich, N.C.; Renshaw, R.W.; et al. Susceptibility of white-tailed deer (Odocoileus virginianus) to SARS-CoV-2. J. Virol. 2021, 95, e00083-21. [Google Scholar] [CrossRef] [PubMed]
- Zoetis’ Emerging Infectious Disease Capabilities Support COVID-19 Solutions for Great Apes and Minks. Zoetis. 2021. Available online: https://www.zoetis.com/news-and-insights/featured-stories/zoetis-emerging-infectious-disease-capabilities-support-covid-19-solutions-for-great-apes-and-minks (accessed on 27 August 2025).
- Hoyte, A.; Webster, M.; Ameiss, K.; Conlee, D.A.; Hainer, N.; Hutchinson, K.; Burakova, Y.; Dominowski, P.J.; Baima, E.T.; King, V.L.; et al. Experimental veterinary SARS-CoV-2 vaccine cross neutralization of the Delta (B.1.617.2) variant virus in cats. Vet. Microbiol. 2022, 268, 109395. [Google Scholar] [CrossRef] [PubMed]
- Rooney, T.A.; Cerveny, S.; Eustace, R.; Colburn, R.; Gerdes, R.S.; Diel, D.G.; Hardham, J.; Thompson, K. Subcutaneous and intramuscular administration of a SARS-CoV-2 vaccine are similarly effective in generating a humoral response in domestic goats (Capra hircus). Am. J. Vet. Res. 2023, 84, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Damas, J.; Hughes, G.M.; Keough, K.C.; Painter, C.A.; Persky, N.S. Broad host range of SARS-CoV-2 predicted by comparative and structural analysis of ACE2 in vertebrates. Proc. Natl. Acad. Sci. USA 2020, 117, 22311–22322. [Google Scholar] [CrossRef] [PubMed]
- Cavaretta, M.; Bronson, E.; Terio, K.; McAloose, D.; Diel, D.G. Examination of the effects of Zoetis mink SARS-CoV-2 vaccination in non-domestic felids. In Proceedings of the 55th Annual AAZV Conference, Nashville, TN, USA, 16 September 2023; p. 65. [Google Scholar]
- McAloose, D.; Laverack, M.; Wang, L.; Killian, M.L.; Caserta, L.C.; Yuan, F.; Mitchell, P.K.; Queen, K.; Mauldin, M.R.; Cronk, B.D.; et al. From people to Panthera: Natural SARS-CoV-2 infection in tigers and lions at the Bronx Zoo. mBio 2020, 11, e02220-20. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zheng, M.; Liu, Y.; Wang, Y.; Xu, Y.; Zhou, Y. Analysis of intermediate hosts and susceptible animals of SARS-CoV-2 by computational methods. Zoonoses 2021, 1, 1–12. [Google Scholar] [CrossRef]
- Abe, K.T.; Li, Z.; Samson, R.; Samavarchi-Tehrani, P.; Valcourt, E.J.; Wood, H.; Budylowski, P.; Dupuis, A.P.; Girardin, R.C.; Rathod, B.; et al. A simple protein-based surrogate neutralization assay for SARS-CoV-2. JCI Insight 2020, 5, e142362. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.W.; Chia, W.N.; Qin, X.; Liu, P.; Chen, M.I.; Tiu, C.; Hu, Z.; Chen, V.C.; Young, B.E.; Sia, W.R.; et al. SARS-CoV-2 surrogate virus neutralization test based on antibody-mediated blockage of ACE2–spike protein–protein interaction. Nat. Biotechnol. 2020, 38, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Kassardjian, A.; Sun, E.; Sookhoo, J.; Muthuraman, K.; Boligan, K.F.; Kucharska, I.; Rujas, E.; Jetha, A.; Branch, D.R.; Babiuk, S.; et al. Modular adjuvant-free pan-HLA-DR-immunotargeting subunit vaccine against SARS-CoV-2 elicits broad sarbecovirus-neutralizing antibody responses. Cell Rep. 2023, 42, 112391. [Google Scholar] [CrossRef] [PubMed]
- Dimeglio, C.; Migueres, M.; Bouzid, N.; Chapuy-Regaud, S.; Gernigon, C.; Da-Silva, I.; Porcheron, M.; Martin-Blondel, G.; Herin, F.; Izopet, J. Antibody titers and protection against Omicron (BA.1 and BA.2) SARS-CoV-2 infection. Vaccines 2022, 10, 1548. [Google Scholar] [CrossRef] [PubMed]
- Sekirov, I.; Petric, M.; Carruthers, E.; Lawrence, D.; Pidduck, T.; Kustra, J.; Laley, J.; Lee, M.-K.; Chahil, N.; Mak, A.; et al. Performance comparison of micro-neutralization assays based on surrogate SARS-CoV-2 and WT SARS-CoV-2 in assessing virus-neutralizing capacity of anti-SARS-CoV-2 antibodies. Access Microbiol. 2021, 3, 000257. [Google Scholar] [CrossRef] [PubMed]
- Dépéry, L.; Bally, I.; Amen, A.; Némoz, B.; Buisson, M.; Grossi, L.; Truffot, A.; Germi, R.; Guilligay, D.; Veloso, M.; et al. Anti-SARS-CoV-2 serology based on ancestral RBD antigens does not correlate with the presence of neutralizing antibodies against Omicron variants. Microbiol. Spectr. 2025, 13, e0156824. [Google Scholar] [CrossRef] [PubMed]
- Nevitt, B.; Lamberski, N. Vaccination of great apes at San Diego Zoo with experimental SARS-CoV-2 veterinary vaccine: Preliminary safety and titers. In Proceedings of the 53rd Annual AAZV Conference, Online, 4 October 2021. [Google Scholar]
- McLelland, D.J.; Lynch, M.; Vogelnest, L.; Eden, P.; Wallace, A.; Weller, J.; Young, S.; Vaughan-Higgins, R.; Antipov, A.; Honda-Okubo, Y.; et al. Safety and immunogenicity of an adjuvanted recombinant spike protein-based severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccine, SPIKEVET™, in selected Carnivora, Primates and Artiodactyla in Australian zoos. J. Vet. Pharmacol. Ther. 2024, 47, 308–321. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, J.K.; Edison, L.K.; Rowe-Haas, D.K.; Takano, T.; Gilor, C.; Crews, C.D.; Tuanyok, A.; Arukha, A.P.; Shiomitsu, S.; Walden, H.D.S.; et al. Both feline coronavirus serotypes 1 and 2 infected domestic cats develop cross-reactive antibodies to SARS-CoV-2 receptor binding domain: Its implication to pan-CoV vaccine development. Viruses 2023, 15, 914. [Google Scholar] [CrossRef] [PubMed]
- Day, M.J.; Horzinek, M.C.; Schultz, R.D.; Squires, R.A. WSAVA guidelines for the vaccination of dogs and cats. J. Small Anim. Pract. 2016, 57, E1–E45. [Google Scholar] [CrossRef] [PubMed]
- Polar Bear International. Adaptations & Characteristics. Available online: https://polarbearsinternational.org/polar-bears-changing-arctic/polar-bear-facts/adaptions-characteristics/ (accessed on 27 August 2025).
- Poland, G.A.; Borrud, A.; Jacobson, R.M.; McDermott, K.; Wollan, P.C.; Brakke, D.; Charboneau, J.W. Determination of deltoid fat pad thickness: Implications for needle length in adult immunization. JAMA 1997, 277, 1709–1711. [Google Scholar] [CrossRef] [PubMed]
- ‘Omicron Has Been a Tsunami’: 17M+ Canadians Have Had Omicron Since December. CityNews. 2022. Available online: https://vancouver.citynews.ca/2022/07/06/omicron-infections-canada/ (accessed on 21 September 2025).
| Family | Common Name | Species | Individual Identification | Sex | Age (Y) | Weight (kg) | TZ Risk-Based Assessment * | Calculated Affinity of ACE2 Receptor Binding ** |
|---|---|---|---|---|---|---|---|---|
| Felidae | Jaguar | Panthera onca | Jg1 | M | 17 | 76 | High risk | Medium |
| Lion | Panthera leo | Ln1 | M | 10 | 200 | High risk | Medium | |
| Ln2 | F | 10 | 120 | High risk | ||||
| Ln3 | F | 11 | 112 | High risk | ||||
| Cheetah | Acinonyx jubatus | Ch1 | M | 6 | 50 | High risk | Medium | |
| Ch2 | M | 6 | 51 | High risk | ||||
| Ch3 | F | 10 | 41 | High risk | ||||
| Tiger (Amur, Sumatran) | Panthera tigris | Tg1 | F | 15 | 82 | High risk | Medium | |
| Tg2 | M | 14 | 120 | High risk | ||||
| Tg3 | M | 10 | 200 | High risk | ||||
| Cervidae | Moose | Alces alces | Mo1 | F | 9 | 300 | High risk | High |
| Mo2 | F | 9 | 250 | High risk | ||||
| Caribou | Rangifer tarandus | Ca1 | F | 12 | 105 | High risk | High | |
| Ca2 | F | 12 | 112 | High risk | ||||
| Ursidae | Polar bear | Ursus maritimus | Pb1 | F | 21 | 365 | Medium risk | Low |
| Pb2 | M | 11 | 465 | Medium risk | ||||
| Pb3 | M | 9 | 475 | Medium risk | ||||
| Pb4 | F | 7 | 340 | Medium risk | ||||
| Pb5 | F | 21 | 320 | Medium risk | ||||
| Brown bear | Ursus arctos | Bb1 | F | 23 | 240 | Medium risk | Medium | |
| Bb2 | M | 24 | 550 | Medium risk | ||||
| Hyenidae | Spotted hyena | Crocuta crocuta | Sh1 | M | 13 | 50 | High risk | High |
| Sh2 | M | 23 | 70 | High risk | ||||
| Sh3 | F | 10 | 60 | High risk |
| Species | ID | TP1 | TP2 | TP3 | TP4 | TP5 | TP6 | TP7 | Fold Change * |
|---|---|---|---|---|---|---|---|---|---|
| Jaguar | Jg1 | 18.31 | 18.45 | 22.07 | 96.63 | 96.50 | 92.04 | 5.24 | |
| Lion | Ln1 | 27.11 ** (15.22/39.00) | 97.25 | 92.56 | 2.50 | ||||
| Ln2 ° | 26.71 | ||||||||
| Ln3 | 4.30 | 47.30 | 97.51 | 96.63 | 22.70 | ||||
| Cheetah | Ch1 | 9.90 | 68.58 | 97.56 | 97.17 | 96.95 | 48.25 | 9.85 | |
| Ch2 | 36.78 | 96.12 | 93.78 | 79.75 | 97.51 | 2.65 | |||
| Ch3 | 39.08 | 97.13 | 2.49 | ||||||
| Tiger | Tg1 ° | 43.77 | |||||||
| Tg2 ° | 44.80 | ||||||||
| Tg3 | 39.44 ** (42.33/36.55) | 44.09 | 77.87 | 1.97 | |||||
| Moose | Mo1 | 14.52 | 11.76 | 75.04 | 5.10 | ||||
| Mo2 ° | 7.60 | 3.99 | |||||||
| Caribou | Cb1 † | 97.46 | |||||||
| Cb2 † | 97.91 | 97.14 | |||||||
| Polar bear | Pb1 | 16.71 | 14.27 | 12.74 | 14.82 | 0.89 | |||
| Pb2 | 9.57 | 8.02 | 20.75 ** (28.91/12.58) | 2.16 | |||||
| Pb3 | 19.77 | 8.98 | 19.31 | 8.84 (14.93/2.74) | 0.98 | ||||
| Pb4 ° | 17.53 ** (21.1/13.96) | ||||||||
| Pb5 ° | 8.47 | ||||||||
| Brown bear | Bb1 † | 97.58 | |||||||
| Bb2 † | 96.95 | ||||||||
| Spotted hyena | Sh1 ° | 53.31 | |||||||
| Sh2 | 41.62 | 77.52 | 1.86 | ||||||
| Sh3 † | 97.51 | 92.64 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pagliarani, S.; Tuling, J.; Pham, P.H.; Leacy, A.; Delnatte, P.; Lillie, B.N.; Masters, N.; Sookhoo, J.; Babiuk, S.; Wootton, S.K.; et al. SARS-COV-2 Vaccination Response in Non-Domestic Species Housed at the Toronto Zoo. Vaccines 2025, 13, 1037. https://doi.org/10.3390/vaccines13101037
Pagliarani S, Tuling J, Pham PH, Leacy A, Delnatte P, Lillie BN, Masters N, Sookhoo J, Babiuk S, Wootton SK, et al. SARS-COV-2 Vaccination Response in Non-Domestic Species Housed at the Toronto Zoo. Vaccines. 2025; 13(10):1037. https://doi.org/10.3390/vaccines13101037
Chicago/Turabian StylePagliarani, Sara, Jaime Tuling, Phuc H. Pham, Alexander Leacy, Pauline Delnatte, Brandon N. Lillie, Nicholas Masters, Jamie Sookhoo, Shawn Babiuk, Sarah K. Wootton, and et al. 2025. "SARS-COV-2 Vaccination Response in Non-Domestic Species Housed at the Toronto Zoo" Vaccines 13, no. 10: 1037. https://doi.org/10.3390/vaccines13101037
APA StylePagliarani, S., Tuling, J., Pham, P. H., Leacy, A., Delnatte, P., Lillie, B. N., Masters, N., Sookhoo, J., Babiuk, S., Wootton, S. K., & Susta, L. (2025). SARS-COV-2 Vaccination Response in Non-Domestic Species Housed at the Toronto Zoo. Vaccines, 13(10), 1037. https://doi.org/10.3390/vaccines13101037






