An Immunobridging Study to Evaluate the Neutralizing Antibody Titer in Adults Immunized with Two Doses of Either ChAdOx1-nCov-19 (AstraZeneca) or MVC-COV1901
Abstract
:1. Introduction
2. Methods
2.1. Clinical Trials and Sample Population
2.2. Vaccines
2.3. Immunobridging Study
- Immunogenicity data: immunobridging study to evaluate the immunogenicity of a locally developed vaccine against a comparator vaccine which has already been approved in Taiwan.
- Safety data: at least 3000 subjects were required to be tracked for at least one month after the last dose and all subjects to be followed for a median of two months after the last dose.
- The lower limit of the 95% CI of the geometric mean titer ratio (GMTR) of the prototype strain live virus neutralizing antibodies for the MVC-COV1901 vaccine group to the external control group must be greater than 0.67;
- The sero-response level (the proportion of subjects whose neutralizing antibody titers against the prototype strain live virus at 28 days after receiving the second dose of the MVC-COVID19 vaccine) was defined as the proportion of subjects with neutralizing antibody titers against the prototype strain live virus at 60% of the reverse cumulative distribution curve for the external control group. The lower limit of the 95% CI for the sero-response rate must be greater than 50%.
2.4. Live SARS-CoV-2 Neutralization Assay
2.5. Statistical Analysis
3. Results
3.1. Demographics
3.2. Immunogenicity
4. Sensitivity Analysis
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kuo, T.Y.; Lin, M.Y.; Coffman, R.L.; Campbell, J.D.; Traquina, P.; Lin, Y.J.; Liu, L.T.-C.; Cheng, J.; Wu, Y.-C.; Wu, C.-C.; et al. Development of CpG-adjuvanted stable prefusion SARS-CoV-2 spike antigen as a subunit vaccine against COVID-19. Sci. Rep. 2020, 10, 20085. [Google Scholar] [CrossRef]
- Taiwan Centers for Disease Control. Taiwan National Infectious Disease Statistics System. Available online: https://nidss.cdc.gov.tw/en/nndss/Cdcwnh07?id=19CoV (accessed on 7 October 2021).
- Ministry of Health and Welfare, Taiwan. News Release on 10 June 2021 on EUA Approval Criteria for Locally Developed COVID-19 Vaccine (In Traditional Chinese). 10 June 2021. Available online: https://www.mohw.gov.tw/fp-5017-61305-1.html (accessed on 7 October 2021).
- Huang, Z.; He, J.; Su, J.; Ou, Z.; Liu, G.; Fu, R.; Shou, Q.; Zheng, M.; Group, T.; Luxembourg, A.; et al. Immunogenicity and safety of the quadrivalent human papillomavirus vaccine in Chinese females aged 9 to 26 years: A phase 3, open-label, immunobridging study. Vaccine 2021, 39, 760–766. [Google Scholar] [CrossRef] [PubMed]
- International Coalition of Medicines Regulatory Authorities. ICMRA COVID-19 Vaccine Development: Future Steps Workshop. 24 June 2021. Available online: http://www.icmra.info/drupal/en/covid-19/24june2021 (accessed on 7 October 2021).
- Hsieh, S.M.; Liu, M.C.; Chen, Y.H.; Lee, W.S.; Hwang, S.J.; Cheng, S.H.; Ko, W.C.; Hwang, K.P.; Wang, N.C.; Lee, Y.L.; et al. Safety and immunogenicity of CpG 1018 and aluminium hydroxide-adjuvanted SARS-CoV-2 S-2P protein vaccine MVC-COV1901: Interim results of a large-scale, double-blind, randomised, placebo-controlled phase 2 trial in Taiwan. Lancet Respir. Med. 2021, 9, 1396–1406. [Google Scholar] [CrossRef]
- Blanchard, B.; Lee, Y. Taiwan Approves Medigen’s COVID-19 Vaccine Candidate. 19 July 2021. Available online: https://www.reuters.com/world/asia-pacific/taiwan-approves-production-medigens-covid-19-vaccine-candidate-2021-07-19/” (accessed on 17 September 2021).
- Hannah, B. European Pharmaceutical Review. Immuno-Bridging Studies Are Sufficient for Authorising New COVID-19 Vaccines, Say Regulators. 16 September 2021. Available online: https://www.europeanpharmaceuticalreview.com/news/162653/immuno-bridging-studies-are-sufficient-for-authorising-new-covid-19-vaccines-say-regulators/ (accessed on 7 October 2021).
- Medicines and Healthcare Products Regulatory Agency, UK. Access Consortium: Alignment with ICMRA Consensus on Immunobridging for Authorising New COVID-19 Vaccines. 15 September 2021. Available online: https://www.gov.uk/government/publications/access-consortium-alignment-with-icmra-consensus-on-immunobridging-for-authorising-new-covid-19-vaccines/access-consortium-alignment-with-icmra-consensus-on-immunobridging-for-authorising-new-covid-19-vaccines (accessed on 7 October 2021).
- Reed, G.F.; Meade, B.D.; Steinhoff, M.C. The reverse cumulative distribution plot: A graphic method for exploratory analysis of antibody data. Pediatrics 1995, 96, 600–603. [Google Scholar] [CrossRef] [PubMed]
- Therapeutic Goods Administration, Australia. Access Consortium: Alignment with ICMRA Consensus on Immunobridging for Authorising New COVID-19 Vaccines. 14 September 2021. Available online: https://www.tga.gov.au/access-consortium-alignment-icmra-consensus-immunobridging-authorising-new-covid-19-vaccines (accessed on 15 February 2022).
- Taiwan Centers for Disease Control. V-Watch Bulletin Report 2020. 21 November 2021. Available online: https://www.cdc.gov.tw/File/Get/Xwv8akaYldqmvlSsN3_H5Q (accessed on 25 January 2022).
- Cheng, S.-H.; Lin, Y.-C.; Chen, C.-P.; Cheng, C.-Y. Safety and immunogenicity of a heterologous booster of protein subunit vaccine MVC-COV1901 after two doses of adenoviral vector vaccine AZD1222. medRxiv 2021. [Google Scholar] [CrossRef]
- Hsieh, S.-M.; Chang, S.-C.; Cheng, H.-Y.; Shih, S.-R.; Lien, C.E. The reactogenicity and immunogenicity of a booster dose after the second dose of a protein subunit vaccine MVC-COV1901. medRxiv 2021. [Google Scholar] [CrossRef]
- McDonald, I.; Murray, S.M.; Reynolds, C.J.; Altmann, D.M.; Boyton, R.J. Comparative systematic review and meta-analysis of reactogenicity, immunogenicity and efficacy of vaccines against SARS-CoV-2. NPJ Vaccines 2021, 6, 74. [Google Scholar] [CrossRef] [PubMed]
- Voysey, M.; Costa Clemens, S.A.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: A pooled analysis of four randomised trials. Lancet 2021, 397, 881–891. [Google Scholar] [CrossRef]
- Gilbert, P.B.; Montefiori, D.C.; McDermott, A.; Fong, Y.; Benkeser, D.; Deng, W.; Zhou, H.; Houchens, C.R.; Martins, K.; Jayashankar, L.; et al. Immune Correlates Analysis of the mRNA-1273 COVID-19 Vaccine Efficacy Trial. medRxiv 2021. [Google Scholar] [CrossRef]
- Feng, S.; Phillips, D.J.; White, T.; Sayal, H.; Aley, P.K.; Bibi, S.; Dold, C.; Fuskova, M.; Gilbert, S.C.; Hirsch, I.; et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 2032–2040. [Google Scholar] [CrossRef] [PubMed]

| Item | <65 Years | All Ages | ||
|---|---|---|---|---|
| AZD1222 | MVC-COV1901 | AZD1222 | MVC-COV1901 | |
| n = 194 | PPI Subset n = 682 | n = 200 | PPI Subset n = 903 | |
| Age (years) | ||||
| Mean (SD) | 42.2 (11.1) | 38.02 (11.03) | 42.9 (11.7) | 45.68 (16.64) |
| Median (IQR) | 41(16.75) | 37.0 (17.0) | 41.5(17.2) | 42.0 (32.0) |
| Min–Max | 22.0–64.0 | 20.0–64.0 | 22.0–69.0 | 20.0–87.0 |
| Gender | ||||
| Male | 78 (40.2) | 386 (56.6) | 80 (40.0) | 521 (57.7) |
| Female | 116 (59.8) | 296 (43.4) | 120 (60.0) | 382 (42.3) |
| BMI (kg/m2) | ||||
| Mean (SD) | 25.3 (4.5) | 24.9 (4.3) | 25.2(4.6) | 24.9 (4.1) |
| Median (IQR) | 24.6 (5.97) | 24.3 (5.7) | 24.5(5.97) | 24.4 (5.3) |
| Min–Max | 16.6–39.6 | 14.3–45.2 | 17.01–37.5 | 17.7–36.9 |
| BMI group | ||||
| <30 kg/m2 | 159 (81.96) | 594 (87.1) | 798 (88.4) | 164 (82.0) |
| ≥30 kg/m2 | 35 (18.04) | 88 (12.9) | 105 (11.6) | 36 (18.0) |
| Pre-vaccination neutralizing antibody status | ||||
| Seropositive | 2 (1.03) | 8 (1.17) | 2 (1.0) | 10 (1.11) |
| Seronegative | 192 (98.97) | 674 (98.83) | 198 (99.0) | 893 (98.9) |
| Comorbidity Category | ||||
| At least one comorbidity | 74 (38.1) | 89 (13.0) | 123 (61.5) | 729 (80.7) |
| No comorbidity | 120 (61.9) | 593 (87.0) | 77 (38.5) | 174 (19.3) |
| Item | <65 Years | All Ages | ||||
|---|---|---|---|---|---|---|
| AZD1222 n = 192 | MVC-COV1901 (PPI Subset) n = 674 | p-Value (GMT Ratio) | AZD1222 n = 198 | MVC-COV1901 (PPI Subset) n = 893 | p-Value (GMT Ratio) | |
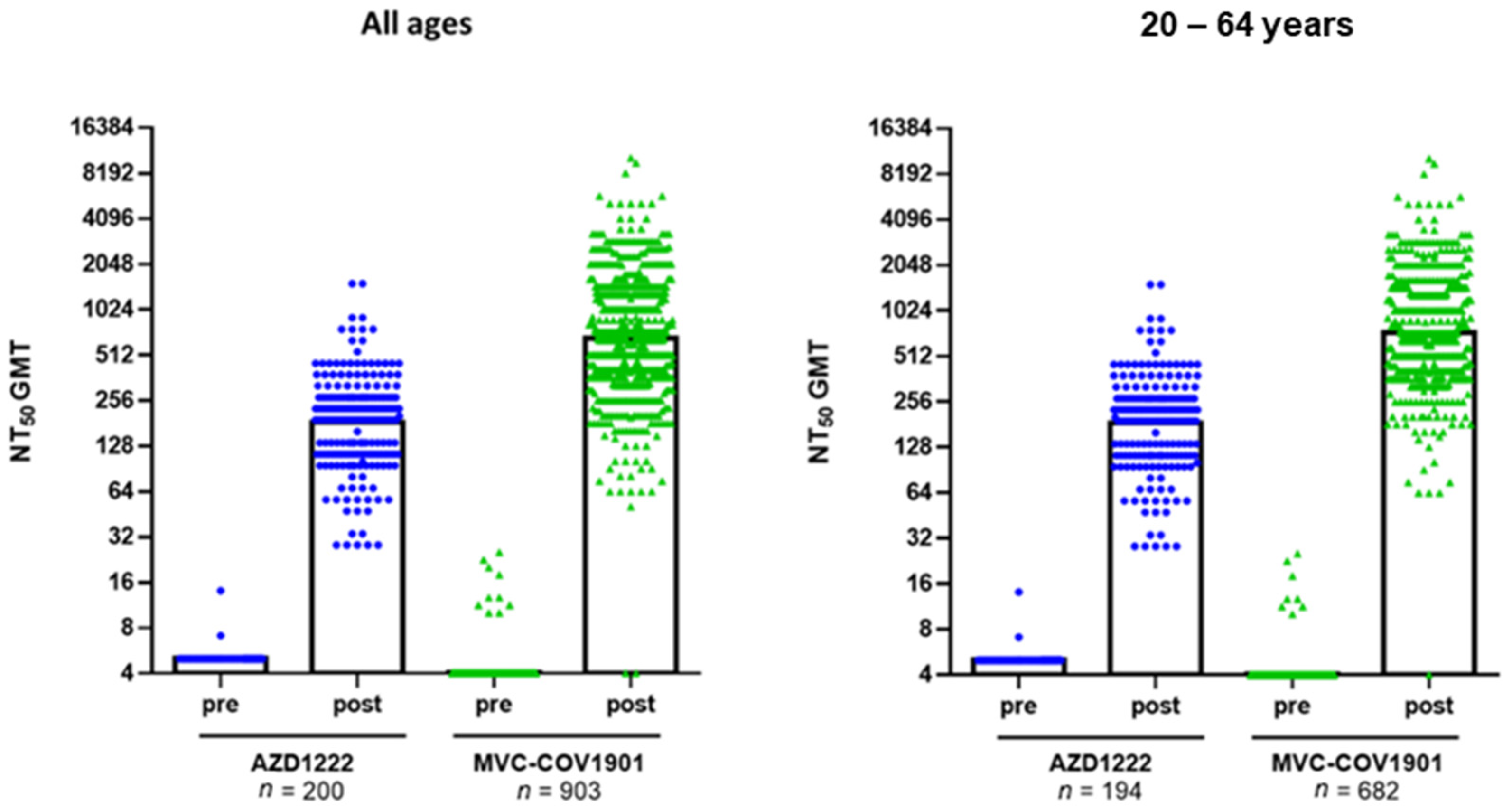
| GMT (95% CI) | 185.97 (167.3–206.7) | 723.6 (683.7–765.8) | 184.05 (166.5–204.7) | 654.07 (620.9–689.03) | ||
| GMT Ratio of MVC/AZ (95% CI) | 3.89 (3.45–4.4) | <0.0001 | 3.55 (3.2–3.97) | <0.0001 | ||
| Adjusted GMT Ratio * (95% CI) | 3.78 (3.3–4.3) | <0.0001 | 3.8 (3.4–4.3) | <0.0001 | ||
| Item | <65 Years | All Ages | ||||
|---|---|---|---|---|---|---|
| AstraZeneca AZD1222 n = 192 | MVC-COV1901 (PPI Subset) n = 458 | p-Value (GMT Ratio) | AstraZeneca AZD1222 n = 198 | MVC-COV1901 (PPI Subset) n = 635 | p-Value (GMT Ratio) | |
| GMT (95% CI) | 185.97 (167.3–206.7) | 492.6 (470.1–516.1) | 184.05 (166.5–204.7) | 453.7 (433.8–474.7) | ||
| GMT Ratio of MVC/AZ (95% CI) | 2.65 (2.4~2.97) | <0.0001 | 2.46 (2.2–2.7) | <0.0001 | ||
| Adjusted GMT Ratio * (95% CI) | 2.6 (2.3–2.9) | <0.0001 | 2.62 (2.3–2.9) | <0.0001 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estrada, J.A.; Cheng, C.-Y.; Ku, S.-Y.; Hu, H.-C.; Yeh, H.-W.; Lin, Y.-C.; Chen, C.-P.; Cheng, S.-H.; Janssen, R.; Lin, I.-F. An Immunobridging Study to Evaluate the Neutralizing Antibody Titer in Adults Immunized with Two Doses of Either ChAdOx1-nCov-19 (AstraZeneca) or MVC-COV1901. Vaccines 2022, 10, 655. https://doi.org/10.3390/vaccines10050655
Estrada JA, Cheng C-Y, Ku S-Y, Hu H-C, Yeh H-W, Lin Y-C, Chen C-P, Cheng S-H, Janssen R, Lin I-F. An Immunobridging Study to Evaluate the Neutralizing Antibody Titer in Adults Immunized with Two Doses of Either ChAdOx1-nCov-19 (AstraZeneca) or MVC-COV1901. Vaccines. 2022; 10(5):655. https://doi.org/10.3390/vaccines10050655
Chicago/Turabian StyleEstrada, Josue Antonio, Chien-Yu Cheng, Shin-Yen Ku, Hui-Chun Hu, Hsiu-Wen Yeh, Yi-Chun Lin, Cheng-Pin Chen, Shu-Hsing Cheng, Robert Janssen, and I-Feng Lin. 2022. "An Immunobridging Study to Evaluate the Neutralizing Antibody Titer in Adults Immunized with Two Doses of Either ChAdOx1-nCov-19 (AstraZeneca) or MVC-COV1901" Vaccines 10, no. 5: 655. https://doi.org/10.3390/vaccines10050655
APA StyleEstrada, J. A., Cheng, C.-Y., Ku, S.-Y., Hu, H.-C., Yeh, H.-W., Lin, Y.-C., Chen, C.-P., Cheng, S.-H., Janssen, R., & Lin, I.-F. (2022). An Immunobridging Study to Evaluate the Neutralizing Antibody Titer in Adults Immunized with Two Doses of Either ChAdOx1-nCov-19 (AstraZeneca) or MVC-COV1901. Vaccines, 10(5), 655. https://doi.org/10.3390/vaccines10050655






