Virus-like Particles as Preventive and Therapeutic Cancer Vaccines
Abstract
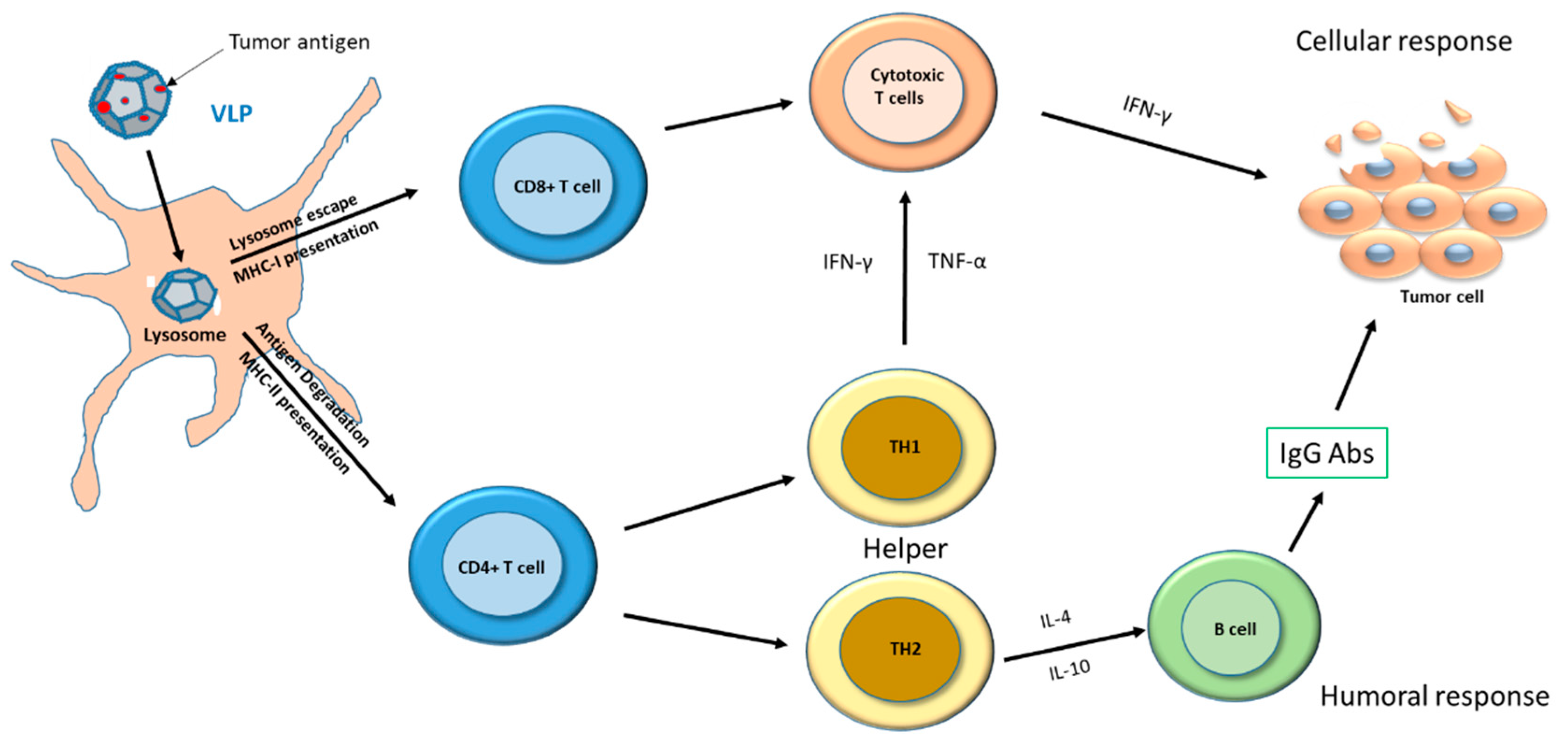
1. Introduction
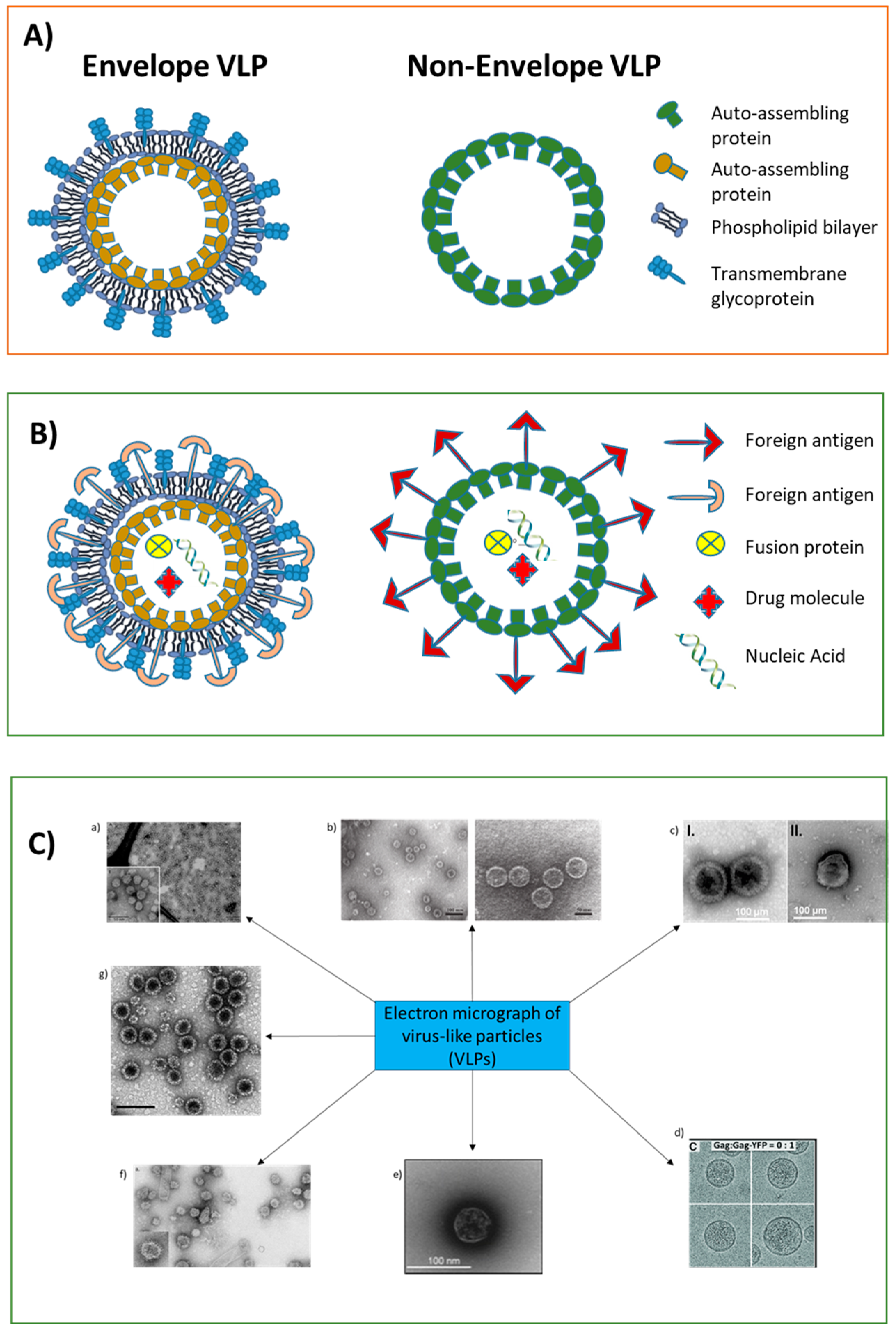
2. VLPs: Chemical and Biological Characteristics
3. VLPs Vaccine for Cancer Prevention
3.1. VLPs in the Prevention of Cancer Caused by Hepatitis B Virus (HBV)
3.2. VLPs in the Prevention of Cancer Caused by HPV
3.3. VLP Vaccines against Human Herpesvirus Type 8 (HHV-8) to Prevent Kaposi Sarcoma Development
3.4. VLP Vaccines to Prevent Cancers Caused by the Human T-Lymphotropic Virus 1 (HTLV-1)
3.5. VLP Vaccines to Prevent Epstein–Barr Virus (EBV)-Related Cancers
3.6. VLPs in the Prevention of Cancer Caused by Hepatitis C Virus (HCV)
3.7. VLPs in the Prevention of Cancer Caused by Merkel Cell Polyomavirus (MCPyV)
4. VLPs Vaccine for Cancer Therapy
4.1. VLPs for Breast Cancer Therapy
4.2. VLPs for Melanoma
4.3. VLPs for Pancreatic Cancer
4.4. VLPs for Cervical Cancer
4.5. VLPs for Hepatocellular Carcinoma
5. VLPs for Delivery of Small Molecule for Cancer Therapy or in Combination with Radiotherapy
6. VLPs for Cancer Vaccine: Critical Issue
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Roldão, A.; Mellado, M.C.; Castilho, L.R.; Carrondo, M.J.; Alves, P.M. Virus-like particles in vaccine development. Expert Rev. Vaccines 2010, 9, 1149–1176. [Google Scholar] [CrossRef] [PubMed]
- Böttcher, J.P.; Reis e Sousa, C. The Role of Type 1 Conventional Dendritic Cells in Cancer Immunity. Trends Cancer 2018, 4, 784–792. [Google Scholar] [CrossRef]
- Laidlaw, B.J.; Craft, J.E.; Kaech, S.M. The multifaceted role of CD4(+) T cells in CD8(+) T cell memory. Nat. Rev. Immunol. 2016, 16, 102–111. [Google Scholar] [CrossRef]
- Braun, M.; Jandus, C.; Maurer, P.; Hammann-Haenni, A.; Schwarz, K.; Bachmann, M.F.; Speiser, D.E.; Romero, P. Virus-like particles induce robust human T-helper cell responses. Eur. J. Immunol. 2012, 42, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Cimica, V.; Galarza, J.M. Adjuvant formulations for virus-like particle (VLP) based vaccines. Clin. Immunol. 2017, 183, 99–108. [Google Scholar] [CrossRef]
- zur Hausen, H. Viruses in human cancers. Science 1991, 254, 1167–1173. [Google Scholar] [CrossRef]
- Moore, P.S.; Chang, Y. Why do viruses cause cancer? Highlights of the first century of human tumour virology. Nat. Rev. Cancer 2010, 10, 878–889. [Google Scholar] [CrossRef]
- Schiller, J.T.; Lowy, D.R. Vaccines to prevent infections by oncoviruses. Annu. Rev. Microbiol. 2010, 64, 23–41. [Google Scholar] [CrossRef]
- Stanley, M. Tumour virus vaccines: Hepatitis B virus and human papillomavirus. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160268. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, J.C.; Perrine, M.; Pericle, F.; Cavallo, F. Virus-Like Particles as an Immunogenic Platform for Cancer Vaccines. Viruses 2020, 12, 488. [Google Scholar] [CrossRef]
- Donaldson, B.; Al-Barwani, F.; Pelham, S.J.; Young, K.; Ward, V.K.; Young, S.L. Multi-target chimaeric VLP as a therapeutic vaccine in a model of colorectal cancer. J. Immunother. Cancer 2017, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.; Young, V.L.; Donaldson, B.C.; Woodall, M.J.; Shields, N.J.; Walker, G.F.; Ward, V.K.; Young, S.L. Delivering Two Tumour Antigens Survivin and Mucin-1 on Virus-Like Particles Enhances Anti-Tumour Immune Responses. Vaccines 2021, 9, 463. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Jing, Z.; Wang, S.; Li, Q.; Xing, Y.; Shi, H.; Li, S.; Hong, Z. P22 virus-like particles as an effective antigen delivery nanoplatform for cancer immunotherapy. Biomaterials 2021, 271, 120726. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, M.O.; Speiser, D.E.; Knuth, A.; Bachmann, M.F. Virus-like particles for vaccination against cancer. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1579. [Google Scholar] [CrossRef]
- Zepeda-Cervantes, J.; Ramírez-Jarquín, J.O.; Vaca, L. Interaction Between Virus-Like Particles (VLPs) and Pattern Recognition Receptors (PRRs) From Dendritic Cells (DCs): Toward Better Engineering of VLPs. Front. Immunol. 2020, 11, 1100. [Google Scholar] [CrossRef]
- Ungaro, F.; Conte, C.; Quaglia, F.; Tornesello, M.L.; Buonaguro, F.M.; Buonaguro, L. VLPs and particle strategies for cancer vaccines. Expert Rev. Vaccines 2013, 12, 1173–1193. [Google Scholar] [CrossRef][Green Version]
- Prasad, B.V.; Schmid, M.F. Principles of virus structural organization. Adv. Exp. Med. Biol. 2012, 726, 17–47. [Google Scholar] [CrossRef] [PubMed]
- Nooraei, S.; Bahrulolum, H.; Hoseini, Z.S.; Katalani, C.; Hajizade, A.; Easton, A.J.; Ahmadian, G. Virus-like particles: Preparation, immunogenicity and their roles as nanovaccines and drug nanocarriers. J. Nanobiotechnol. 2021, 19, 59. [Google Scholar] [CrossRef]
- Manolova, V.; Flace, A.; Bauer, M.; Schwarz, K.; Saudan, P.; Bachmann, M.F. Nanoparticles target distinct dendritic cell populations according to their size. Eur. J. Immunol. 2008, 38, 1404–1413. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, M.O.; Zha, L.; Cabral-Miranda, G.; Bachmann, M.F. Major findings and recent advances in virus-like particle (VLP)-based vaccines. Semin. Immunol. 2017, 34, 123–132. [Google Scholar] [CrossRef]
- Rohovie, M.J.; Nagasawa, M.; Swartz, J.R. Virus-like particles: Next-generation nanoparticles for targeted therapeutic delivery. Bioeng. Transl. Med. 2017, 2, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Lunsdorf, H.; Gurramkonda, C.; Adnan, A.; Khanna, N.; Rinas, U. Virus-like particle production with yeast: Ultrastructural and immunocytochemical insights into Pichia pastoris producing high levels of the hepatitis B surface antigen. Microb. Cell Factories 2011, 10, 48. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.C.; Patkar, K.K.; Basu, A.; Mohandas, K.M.; Mukhopadhyaya, R. Production of immunogenic human papillomavirus-16 major capsid protein derived virus like particles. Indian J. Med. Res. 2009, 130, 213–218. [Google Scholar] [PubMed]
- Barasa, A.K.; Ye, P.; Phelps, M.; Arivudainambi, G.T.; Tison, T.; Ogembo, J.G. BALB/c mice immunized with a combination of virus-like particles incorporating Kaposi sarcoma-associated herpesvirus (KSHV) envelope glycoproteins gpK8.1, gB, and gH/gL induced comparable serum neutralizing antibody activity to UV-inactivated KSHV. Oncotarget 2017, 8, 34481–34497. [Google Scholar] [CrossRef]
- Maldonado, J.O.; Angert, I.; Cao, S.; Berk, S.; Zhang, W.; Mueller, J.D.; Mansky, L.M. Perturbation of Human T-Cell Leukemia Virus Type 1 Particle Morphology by Differential Gag Co-Packaging. Viruses 2017, 9, 191. [Google Scholar] [CrossRef]
- Ogembo, J.G.; Muraswki, M.R.; McGinnes, L.W.; Parcharidou, A.; Sutiwisesak, R.; Tison, T.; Avendano, J.; Agnani, D.; Finberg, R.W.; Morrison, T.G.; et al. A chimeric EBV gp350/220-based VLP replicates the virion B-cell attachment mechanism and elicits long-lasting neutralizing antibodies in mice. J. Transl. Med. 2015, 13, 50. [Google Scholar] [CrossRef]
- Clayton, R.F.; Owsianka, A.; Aitken, J.; Graham, S.; Bhella, D.; Patel, A.H. Analysis of antigenicity and topology of E2 glycoprotein present on recombinant hepatitis C virus-like particles. J. Virol. 2002, 76, 7672–7682. [Google Scholar] [CrossRef]
- Coursaget, P.; Samimi, M.; Nicol, J.T.; Gardair, C.; Touze, A. Human Merkel cell polyomavirus: Virological background and clinical implications. APMIS 2013, 121, 755–769. [Google Scholar] [CrossRef]
- Sapsford, K.E.; Algar, W.R.; Berti, L.; Gemmill, K.B.; Casey, B.J.; Oh, E.; Stewart, M.H.; Medintz, I.L. Functionalizing nanoparticles with biological molecules: Developing chemistries that facilitate nanotechnology. Chem. Rev. 2013, 113, 1904–2074. [Google Scholar] [CrossRef]
- Crisci, E.; Bárcena, J.; Montoya, M. Virus-like particles: The new frontier of vaccines for animal viral infections. Vet. Immunol. Immunopathol. 2012, 148, 211–225. [Google Scholar] [CrossRef]
- Vicente, T.; Roldão, A.; Peixoto, C.; Carrondo, M.J.; Alves, P.M. Large-scale production and purification of VLP-based vaccines. J. Invertebr. Pathol. 2011, 107, S42–S48. [Google Scholar] [CrossRef] [PubMed]
- Zeltins, A. Construction and characterization of virus-like particles: A review. Mol. Biotechnol. 2013, 53, 92–107. [Google Scholar] [CrossRef] [PubMed]
- Weiss, V.U.; Pogan, R.; Zoratto, S.; Bond, K.M.; Boulanger, P.; Jarrold, M.F.; Lyktey, N.; Pahl, D.; Puffler, N.; Schelhaas, M.; et al. Virus-like particle size and molecular weight/mass determination applying gas-phase electrophoresis (native nES GEMMA). Anal. Bioanal. Chem. 2019, 411, 5951–5962. [Google Scholar] [CrossRef] [PubMed]
- Mateu, M.G. Virus engineering: Functionalization and stabilization. Protein Eng. Des. Sel. 2011, 24, 53–63. [Google Scholar] [CrossRef]
- Murphy, T.L.; Murphy, K.M. Dendritic cells in cancer immunology. Cell Mol. Immunol. 2021, 19, 3–13. [Google Scholar] [CrossRef]
- Hoebe, K.; Janssen, E.; Beutler, B. The interface between innate and adaptive immunity. Nat. Immunol. 2004, 5, 971–974. [Google Scholar] [CrossRef]
- Leung, C.S. Endogenous Antigen Presentation of MHC Class II Epitopes through Non-Autophagic Pathways. Front. Immunol. 2015, 6, 464. [Google Scholar] [CrossRef]
- Waldman, A.D.; Fritz, J.M.; Lenardo, M.J. A guide to cancer immunotherapy: From T cell basic science to clinical practice. Nat. Rev. Immunol. 2020, 20, 651–668. [Google Scholar] [CrossRef]
- Gun, S.Y.; Lee, S.W.L.; Sieow, J.L.; Wong, S.C. Targeting immune cells for cancer therapy. Redox Biol. 2019, 25, 101174. [Google Scholar] [CrossRef]
- Saxena, M.; van der Burg, S.H.; Melief, C.J.M.; Bhardwaj, N. Therapeutic cancer vaccines. Nat. Rev. Cancer 2021, 21, 360–378. [Google Scholar] [CrossRef]
- Buonaguro, L.; Petrizzo, A.; Tornesello, M.L.; Buonaguro, F.M. Translating tumor antigens into cancer vaccines. Clin. Vaccine Immunol. 2011, 18, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Cubas, R.; Li, M.; Chen, C.; Yao, Q. Virus-like particle vaccine activates conventional B2 cells and promotes B cell differentiation to IgG2a producing plasma cells. Mol. Immunol. 2009, 46, 1988–2001. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.K.; Tan, W.S.; Ho, K.L. Virus like particles as a platform for cancer vaccine development. PeerJ 2017, 5, e4053. [Google Scholar] [CrossRef] [PubMed]
- Tornesello, M.L.; Buonaguro, L.; Izzo, F.; Buonaguro, F.M. Molecular alterations in hepatocellular carcinoma associated with hepatitis B and hepatitis C infections. Oncotarget 2016, 7, 25087–25102. [Google Scholar] [CrossRef]
- Valenzuela, P.; Medina, A.; Rutter, W.J.; Ammerer, G.; Hall, B.D. Synthesis and assembly of hepatitis B virus surface antigen particles in yeast. Nature 1982, 298, 347–350. [Google Scholar] [CrossRef]
- Blumberg, B.S.; Alter, H.J.; Visnich, S. A “NEW” Antigen in Leukemia Sera. JAMA 1965, 191, 541–546. [Google Scholar] [CrossRef]
- Bayer, M.E.; Blumberg, B.S.; Werner, B. Particles associated with Australia antigen in the sera of patients with leukaemia, Down’s Syndrome and hepatitis. Nature 1968, 218, 1057–1059. [Google Scholar] [CrossRef]
- Millman, I.; Loeb, L.A.; Bayer, M.E.; Blumberg, B.S. Australia antigen (a hepatitis-associated antigen): Purification and physical properties. J. Exp. Med. 1970, 131, 1190–1199. [Google Scholar] [CrossRef]
- Gerber, M.A.; Hadziyannis, S.; Vissoulis, C.; Schaffner, F.; Paronetto, F.; Popper, H. Electron microscopy and immunoelectronmicroscopy of cytoplasmic hepatitis B antigen in hepatocytes. Am. J. Pathol. 1974, 75, 489–502. [Google Scholar]
- Gavilanes, F.; Gonzalez-Ros, J.M.; Peterson, D.L. Structure of hepatitis B surface antigen. Characterization of the lipid components and their association with the viral proteins. J. Biol. Chem. 1982, 257, 7770–7777. [Google Scholar] [CrossRef]
- Qian, C.; Liu, X.; Xu, Q.; Wang, Z.; Chen, J.; Li, T.; Zheng, Q.; Yu, H.; Gu, Y.; Li, S.; et al. Recent Progress on the Versatility of Virus-Like Particles. Vaccines 2020, 8, 139. [Google Scholar] [CrossRef]
- Krugman, S. The newly licensed hepatitis B vaccine. Characteristics and indications for use. JAMA 1982, 247, 2012–2015. [Google Scholar] [CrossRef] [PubMed]
- Shouval, D.; Roggendorf, H.; Roggendorf, M. Enhanced immune response to hepatitis B vaccination through immunization with a Pre-S1/Pre-S2/S vaccine. Med. Microbiol. Immunol. 2015, 204, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Champion, C.R. Heplisav-B: A Hepatitis B Vaccine With a Novel Adjuvant. Ann. Pharmacother. 2021, 55, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Joe, C.C.D.; Chatterjee, S.; Lovrecz, G.; Adams, T.E.; Thaysen-Andersen, M.; Walsh, R.; Locarnini, S.A.; Smooker, P.; Netter, H.J. Glycoengineered hepatitis B virus-like particles with enhanced immunogenicity. Vaccine 2020, 38, 3892–3901. [Google Scholar] [CrossRef]
- Spice, A.J.; Aw, R.; Bracewell, D.G.; Polizzi, K.M. Synthesis and Assembly of Hepatitis B Virus-Like Particles in a Pichia pastoris Cell-Free System. Front. Bioeng. Biotechnol. 2020, 8, 72. [Google Scholar] [CrossRef] [PubMed]
- Gheit, T. Mucosal and Cutaneous Human Papillomavirus Infections and Cancer Biology. Front. Oncol. 2019, 9, 355. [Google Scholar] [CrossRef]
- Frazer, I.H. The HPV Vaccine Story. ACS Pharmacol. Transl. Sci. 2019, 2, 210–212. [Google Scholar] [CrossRef]
- Hagensee, M.E.; Yaegashi, N.; Galloway, D.A. Self-assembly of human papillomavirus type 1 capsids by expression of the L1 protein alone or by coexpression of the L1 and L2 capsid proteins. J. Virol. 1993, 67, 315–322. [Google Scholar] [CrossRef]
- Kirnbauer, R.; Booy, F.; Cheng, N.; Lowy, D.R.; Schiller, J.T. Papillomavirus L1 major capsid protein self-assembles into virus-like particles that are highly immunogenic. Proc. Natl. Acad. Sci. USA 1992, 89, 12180–12184. [Google Scholar] [CrossRef]
- Human papillomavirus vaccines: WHO position paper, May 2017-Recommendations. Vaccine 2017, 35, 5753–5755. [CrossRef]
- Schiller, J.T.; Castellsagué, X.; Garland, S.M. A review of clinical trials of human papillomavirus prophylactic vaccines. Vaccine 2012, 30 (Suppl. 5), F123–F138. [Google Scholar] [CrossRef] [PubMed]
- Kuter, B.J.; Garland, S.M.; Giuliano, A.R.; Stanley, M.A. Current and future vaccine clinical research with the licensed 2-, 4-, and 9-valent VLP HPV vaccines: What’s ongoing, what’s needed? Prev. Med. 2021, 144, 106321. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.; Zhai, L.; Tumban, E. Virus-like Particle-Based L2 Vaccines against HPVs: Where Are We Today? Viruses 2019, 12, 18. [Google Scholar] [CrossRef] [PubMed]
- Pouyanfard, S.; Spagnoli, G.; Bulli, L.; Balz, K.; Yang, F.; Odenwald, C.; Seitz, H.; Mariz, F.C.; Bolchi, A.; Ottonello, S.; et al. Minor Capsid Protein L2 Polytope Induces Broad Protection against Oncogenic and Mucosal Human Papillomaviruses. J. Virol. 2018, 92, 92. [Google Scholar] [CrossRef]
- Tumban, E.; Muttil, P.; Escobar, C.A.; Peabody, J.; Wafula, D.; Peabody, D.S.; Chackerian, B. Preclinical refinements of a broadly protective VLP-based HPV vaccine targeting the minor capsid protein, L2. Vaccine 2015, 33, 3346–3353. [Google Scholar] [CrossRef]
- DeCaprio, J.A.; Garcea, R.L. A cornucopia of human polyomaviruses. Nat. Rev. Microbiol. 2013, 11, 264–276. [Google Scholar] [CrossRef]
- Salunke, D.M.; Caspar, D.L.; Garcea, R.L. Self-assembly of purified polyomavirus capsid protein VP1. Cell 1986, 46, 895–904. [Google Scholar] [CrossRef]
- Rayne, F.; Bouamr, F.; Lalanne, J.; Mamoun, R.Z. The NH2-terminal domain of the human T-cell leukemia virus type 1 capsid protein is involved in particle formation. J. Virol. 2001, 75, 5277–5287. [Google Scholar] [CrossRef]
- Maldonado, J.O.; Cao, S.; Zhang, W.; Mansky, L.M. Distinct Morphology of Human T-Cell Leukemia Virus Type 1-Like Particles. Viruses 2016, 8, 132. [Google Scholar] [CrossRef]
- Martin, J.L.; Mendonça, L.M.; Marusinec, R.; Zuczek, J.; Angert, I.; Blower, R.J.; Mueller, J.D.; Perilla, J.R.; Zhang, W.; Mansky, L.M. Critical Role of the Human T-Cell Leukemia Virus Type 1 Capsid N-Terminal Domain for Gag-Gag Interactions and Virus Particle Assembly. J. Virol. 2018, 92, e00333-18. [Google Scholar] [CrossRef]
- Ruiss, R.; Jochum, S.; Wanner, G.; Reisbach, G.; Hammerschmidt, W.; Zeidler, R. A virus-like particle-based Epstein-Barr virus vaccine. J. Virol. 2011, 85, 13105–13113. [Google Scholar] [CrossRef] [PubMed]
- Germi, R.; Effantin, G.; Grossi, L.; Ruigrok, R.W.H.; Morand, P.; Schoehn, G. Three-dimensional structure of the Epstein-Barr virus capsid. J. Gen. Virol. 2012, 93, 1769–1773. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baumert, T.F.; Ito, S.; Wong, D.T.; Liang, T.J. Hepatitis C virus structural proteins assemble into viruslike particles in insect cells. J. Virol. 1998, 72, 3827–3836. [Google Scholar] [CrossRef] [PubMed]
- Janitzek, C.M.; Peabody, J.; Thrane, S.; Carlsen, P.H.R.; Theander, T.G.; Salanti, A.; Chackerian, B.; Nielsen, M.A.; Sander, A.F. A proof-of-concept study for the design of a VLP-based combinatorial HPV and placental malaria vaccine. Sci. Rep. 2019, 9, 5260. [Google Scholar] [CrossRef] [PubMed]
- Mulama, D.H.; Mutsvunguma, L.Z.; Totonchy, J.; Ye, P.; Foley, J.; Escalante, G.M.; Rodriguez, E.; Nabiee, R.; Muniraju, M.; Wussow, F.; et al. A multivalent Kaposi sarcoma-associated herpesvirus-like particle vaccine capable of eliciting high titers of neutralizing antibodies in immunized rabbits. Vaccine 2019, 37, 4184–4194. [Google Scholar] [CrossRef]
- Perez, E.M.; Foley, J.; Tison, T.; Silva, R.; Ogembo, J.G. Novel Epstein-Barr virus-like particles incorporating gH/gL-EBNA1 or gB-LMP2 induce high neutralizing antibody titers and EBV-specific T-cell responses in immunized mice. Oncotarget 2017, 8, 19255–19273. [Google Scholar] [CrossRef]
- Escalante, G.M.; Foley, J.; Mutsvunguma, L.Z.; Rodriguez, E.; Mulama, D.H.; Muniraju, M.; Ye, P.; Barasa, A.K.; Ogembo, J.G. A Pentavalent Epstein-Barr Virus-Like Particle Vaccine Elicits High Titers of Neutralizing Antibodies against Epstein-Barr Virus Infection in Immunized Rabbits. Vaccines 2020, 8, 169. [Google Scholar] [CrossRef]
- Zhang, T.Y.; Guo, X.R.; Wu, Y.T.; Kang, X.Z.; Zheng, Q.B.; Qi, R.Y.; Chen, B.B.; Lan, Y.; Wei, M.; Wang, S.J.; et al. A unique B cell epitope-based particulate vaccine shows effective suppression of hepatitis B surface antigen in mice. Gut 2020, 69, 343–354. [Google Scholar] [CrossRef]
- Sominskaya, I.; Skrastina, D.; Dislers, A.; Vasiljev, D.; Mihailova, M.; Ose, V.; Dreilina, D.; Pumpens, P. Construction and immunological evaluation of multivalent hepatitis B virus (HBV) core virus-like particles carrying HBV and HCV epitopes. Clin. Vaccine Immunol. 2010, 17, 1027–1033. [Google Scholar] [CrossRef]
- Kumar, A.; Das, S.; Mullick, R.; Lahiri, P.; Tatineni, R.; Goswami, D.; Bhat, P.; Torresi, J.; Gowans, E.J.; Karande, A.A.; et al. Immune responses against hepatitis C virus genotype 3a virus-like particles in mice: A novel VLP prime-adenovirus boost strategy. Vaccine 2016, 34, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- Earnest-Silveira, L.; Christiansen, D.; Herrmann, S.; Ralph, S.A.; Das, S.; Gowans, E.J.; Torresi, J. Large scale production of a mammalian cell derived quadrivalent hepatitis C virus like particle vaccine. J. Virol. Methods 2016, 236, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, D.; Earnest-Silveira, L.; Grubor-Bauk, B.; Wijesundara, D.K.; Boo, I.; Ramsland, P.A.; Vincan, E.; Drummer, H.E.; Gowans, E.J.; Torresi, J. Pre-clinical evaluation of a quadrivalent HCV VLP vaccine in pigs following microneedle delivery. Sci. Rep. 2019, 9, 9251. [Google Scholar] [CrossRef] [PubMed]
- Mesri, E.A.; Cesarman, E.; Boshoff, C. Kaposi’s sarcoma and its associated herpesvirus. Nat. Rev. Cancer 2010, 10, 707–719. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, D.P.; Damania, B. Kaposi sarcoma-associated herpesvirus: Immunobiology, oncogenesis, and therapy. J. Clin. Investig. 2016, 126, 3165–3175. [Google Scholar] [CrossRef]
- Cesarman, E.; Damania, B.; Krown, S.E.; Martin, J.; Bower, M.; Whitby, D. Kaposi sarcoma. Nat. Rev. Dis. Prim. 2019, 5, 9. [Google Scholar] [CrossRef]
- Gonçalves, P.H.; Uldrick, T.S.; Yarchoan, R. HIV-associated Kaposi sarcoma and related diseases. Aids 2017, 31, 1903–1916. [Google Scholar] [CrossRef]
- Weill, M.E.; Flament, P.; Gouesbet, G. Diameters and number densities of soot particles in premixed laminar flat flame propane/oxygen. Appl. Opt. 1983, 22, 2407–2409. [Google Scholar] [CrossRef]
- Poiesz, B.J.; Ruscetti, F.W.; Reitz, M.S.; Kalyanaraman, V.S.; Gallo, R.C. Isolation of a new type C retrovirus (HTLV) in primary uncultured cells of a patient with Sézary T-cell leukaemia. Nature 1981, 294, 268–271. [Google Scholar] [CrossRef]
- Ko, Y.H. EBV and human cancer. Exp. Mol. Med. 2015, 47, e130. [Google Scholar] [CrossRef]
- Lopes, V.; Young, L.S.; Murray, P.G. Epstein-Barr virus-associated cancers: Aetiology and treatment. Herpes 2003, 10, 78–82. [Google Scholar] [PubMed]
- Schiller, J.T.; Lowy, D.R. An Introduction to Virus Infections and Human Cancer. Recent Results Cancer Res. 2021, 217, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Habtamu, K.; Alem, A.; Hanlon, C. Conceptualizing and contextualizing functioning in people with severe mental disorders in rural Ethiopia: A qualitative study. BMC Psychiatry 2015, 15, 34. [Google Scholar] [CrossRef]
- Tabata, K.; Neufeldt, C.J.; Bartenschlager, R. Hepatitis C Virus Replication. Cold Spring Harb. Perspect. Med. 2020, 10, a037093. [Google Scholar] [CrossRef] [PubMed]
- Saik, O.V.; Ivanisenko, T.V.; Demenkov, P.S.; Ivanisenko, V.A. Interactome of the hepatitis C virus: Literature mining with ANDSystem. Virus Res. 2016, 218, 40–48. [Google Scholar] [CrossRef]
- Banerjee, A.; Ray, R.B.; Ray, R. Oncogenic potential of hepatitis C virus proteins. Viruses 2010, 2, 2108–2133. [Google Scholar] [CrossRef]
- Feng, H.; Shuda, M.; Chang, Y.; Moore, P.S. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science 2008, 319, 1096–1100. [Google Scholar] [CrossRef]
- Li, T.C.; Iwasaki, K.; Katano, H.; Kataoka, M.; Nagata, N.; Kobayashi, K.; Mizutani, T.; Takeda, N.; Wakita, T.; Suzuki, T. Characterization of self-assembled virus-like particles of Merkel cell polyomavirus. PLoS ONE 2015, 10, e0115646. [Google Scholar] [CrossRef]
- Touzé, A.; Gaitan, J.; Arnold, F.; Cazal, R.; Fleury, M.J.; Combelas, N.; Sizaret, P.Y.; Guyetant, S.; Maruani, A.; Baay, M.; et al. Generation of Merkel cell polyomavirus (MCV)-like particles and their application to detection of MCV antibodies. J. Clin. Microbiol. 2010, 48, 1767–1770. [Google Scholar] [CrossRef]
- Palladini, A.; Thrane, S.; Janitzek, C.M.; Pihl, J.; Clemmensen, S.B.; de Jongh, W.A.; Clausen, T.M.; Nicoletti, G.; Landuzzi, L.; Penichet, M.L.; et al. Virus-like particle display of HER2 induces potent anti-cancer responses. OncoImmunology 2018, 7, e1408749. [Google Scholar] [CrossRef]
- Salazar-González, J.A.; Ruiz-Cruz, A.A.; Bustos-Jaimes, I.; Moreno-Fierros, L. Expression of Breast Cancer-Related Epitopes Targeting the IGF-1 Receptor in Chimeric Human Parvovirus B19 Virus-Like Particles. Mol. Biotechnol. 2019, 61, 742–753. [Google Scholar] [CrossRef] [PubMed]
- Rolih, V.; Caldeira, J.; Bolli, E.; Salameh, A.; Conti, L.; Barutello, G.; Riccardo, F.; Magri, J.; Lamolinara, A.; Parra, K.; et al. Development of a VLP-Based Vaccine Displaying an xCT Extracellular Domain for the Treatment of Metastatic Breast Cancer. Cancers 2020, 12, 1492. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, M.O.; Heath, M.D.; Cabral-Miranda, G.; Lipp, C.; Zeltins, A.; Sande, M.; Stein, J.V.; Riether, C.; Roesti, E.; Zha, L.; et al. Vaccination with nanoparticles combined with micro-adjuvants protects against cancer. J. Immunother. Cancer 2019, 7, 114. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Lee, P.W.; Wang, C.; Thomas, L.D.; Stewart, P.L.; Steinmetz, N.F.; Pokorski, J.K. Freeze-Drying To Produce Efficacious CPMV Virus-like Particles. Nano Lett. 2019, 19, 2099–2105. [Google Scholar] [CrossRef]
- Cheng, K.; Du, T.; Li, Y.; Qi, Y.; Min, H.; Wang, Y.; Zhang, Q.; Wang, C.; Zhou, Y.; Li, L.; et al. Dual-Antigen-Loaded Hepatitis B Virus Core Antigen Virus-like Particles Stimulate Efficient Immunotherapy Against Melanoma. ACS Appl. Mater. Interfaces 2020, 12, 53682–53690. [Google Scholar] [CrossRef]
- Cubas, R.; Zhang, S.; Li, M.; Chen, C.; Yao, Q. Chimeric Trop2 virus-like particles: A potential immunotherapeutic approach against pancreatic cancer. J. Immunother. 2011, 34, 251–263. [Google Scholar] [CrossRef]
- Zhang, S.; Yong, L.K.; Li, D.; Cubas, R.; Chen, C.; Yao, Q. Mesothelin virus-like particle immunization controls pancreatic cancer growth through CD8+ T cell induction and reduction in the frequency of CD4+ foxp3+ ICOS- regulatory T cells. PLoS ONE 2013, 8, e68303. [Google Scholar] [CrossRef]
- Schumacher, J.; Bacic, T.; Staritzbichler, R.; Daneschdar, M.; Klamp, T.; Arnold, P.; Jägle, S.; Türeci, Ö.; Markl, J.; Sahin, U. Enhanced stability of a chimeric hepatitis B core antigen virus-like-particle (HBcAg-VLP) by a C-terminal linker-hexahistidine-peptide. J. Nanobiotechnol. 2018, 16, 39. [Google Scholar] [CrossRef]
- Turashvili, G.; Brogi, E. Tumor Heterogeneity in Breast Cancer. Front. Med. 2017, 4, 227. [Google Scholar] [CrossRef]
- Terao, Y.; Kawabata, S.; Nakata, M.; Nakagawa, I.; Hamada, S. Molecular characterization of a novel fibronectin-binding protein of Streptococcus pyogenes strains isolated from toxic shock-like syndrome patients. J. Biol. Chem. 2002, 277, 47428–47435. [Google Scholar] [CrossRef]
- Thrane, S.; Janitzek, C.M.; Matondo, S.; Resende, M.; Gustavsson, T.; de Jongh, W.A.; Clemmensen, S.; Roeffen, W.; van de Vegte-Bolmer, M.; van Gemert, G.J.; et al. Bacterial superglue enables easy development of efficient virus-like particle based vaccines. J. Nanobiotechnol. 2016, 14, 30. [Google Scholar] [CrossRef] [PubMed]
- Eddy, K.; Shah, R.; Chen, S. Decoding Melanoma Development and Progression: Identification of Therapeutic Vulnerabilities. Front. Oncol. 2020, 10, 626129. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Beiss, V.; Steinmetz, N.F. Cowpea Mosaic Virus Nanoparticles and Empty Virus-Like Particles Show Distinct but Overlapping Immunostimulatory Properties. J. Virol. 2019, 93, e00129-19. [Google Scholar] [CrossRef] [PubMed]
- Huber, B.; Wang, J.W.; Roden, R.B.S.; Kirnbauer, R. RG1-VLP and Other L2-Based, Broad-Spectrum HPV Vaccine Candidates. J. Clin. Med. 2021, 10, 1044. [Google Scholar] [CrossRef]
- Greenstone, H.L.; Nieland, J.D.; de Visser, K.E.; De Bruijn, M.L.; Kirnbauer, R.; Roden, R.B.; Lowy, D.R.; Kast, W.M.; Schiller, J.T. Chimeric papillomavirus virus-like particles elicit antitumor immunity against the E7 oncoprotein in an HPV16 tumor model. Proc. Natl. Acad. Sci. USA 1998, 95, 1800–1805. [Google Scholar] [CrossRef]
- Monroy-García, A.; Gómez-Lim, M.A.; Weiss-Steider, B.; Hernández-Montes, J.; Huerta-Yepez, S.; Rangel-Santiago, J.F.; Santiago-Osorio, E.; Mora García Mde, L. Immunization with an HPV-16 L1-based chimeric virus-like particle containing HPV-16 E6 and E7 epitopes elicits long-lasting prophylactic and therapeutic efficacy in an HPV-16 tumor mice model. Arch. Virol. 2014, 159, 291–305. [Google Scholar] [CrossRef] [PubMed]
- Ghelardi, A.; Parazzini, F.; Martella, F.; Pieralli, A.; Bay, P.; Tonetti, A.; Svelato, A.; Bertacca, G.; Lombardi, S.; Joura, E.A. SPERANZA project: HPV vaccination after treatment for CIN2. Gynecol. Oncol. 2018, 151, 229–234. [Google Scholar] [CrossRef]
- Jentschke, M.; Kampers, J.; Becker, J.; Sibbertsen, P.; Hillemanns, P. Prophylactic HPV vaccination after conization: A systematic review and meta-analysis. Vaccine 2020, 38, 6402–6409. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, B.; Ding, M.; Song, S.; Kang, Y.; Yu, Y.; Xu, M.; Xiang, T.; Gao, L.; Feng, Q.; et al. A novel vaccine candidate based on chimeric virus-like particle displaying multiple conserved epitope peptides induced neutralizing antibodies against EBV infection. Theranostics 2020, 10, 5704–5718. [Google Scholar] [CrossRef]
- Ashley, C.E.; Carnes, E.C.; Phillips, G.K.; Durfee, P.N.; Buley, M.D.; Lino, C.A.; Padilla, D.P.; Phillips, B.; Carter, M.B.; Willman, C.L.; et al. Cell-specific delivery of diverse cargos by bacteriophage MS2 virus-like particles. ACS Nano 2011, 5, 5729–5745. [Google Scholar] [CrossRef]
- Chung, Y.H.; Cai, H.; Steinmetz, N.F. Viral nanoparticles for drug delivery, imaging, immunotherapy, and theranostic applications. Adv. Drug Deliv. Rev. 2020, 156, 214–235. [Google Scholar] [CrossRef] [PubMed]
- Finbloom, J.A.; Aanei, I.L.; Bernard, J.M.; Klass, S.H.; Elledge, S.K.; Han, K.; Ozawa, T.; Nicolaides, T.P.; Berger, M.S.; Francis, M.B. Evaluation of Three Morphologically Distinct Virus-Like Particles as Nanocarriers for Convection-Enhanced Drug Delivery to Glioblastoma. Nanomaterials 2018, 8, 1007. [Google Scholar] [CrossRef] [PubMed]
- Franke, C.E.; Czapar, A.E.; Patel, R.B.; Steinmetz, N.F. Tobacco Mosaic Virus-Delivered Cisplatin Restores Efficacy in Platinum-Resistant Ovarian Cancer Cells. Mol. Pharm. 2018, 15, 2922–2931. [Google Scholar] [CrossRef] [PubMed]
- Czapar, A.E.; Zheng, Y.R.; Riddell, I.A.; Shukla, S.; Awuah, S.G.; Lippard, S.J.; Steinmetz, N.F. Tobacco Mosaic Virus Delivery of Phenanthriplatin for Cancer therapy. ACS Nano 2016, 10, 4119–4126. [Google Scholar] [CrossRef] [PubMed]
- Shoeb, E.; Hefferon, K. Future of cancer immunotherapy using plant virus-based nanoparticles. Future Sci. OA 2019, 5, Fso401. [Google Scholar] [CrossRef] [PubMed]
- Hoopes, P.J.; Wagner, R.J.; Duval, K.; Kang, K.; Gladstone, D.J.; Moodie, K.L.; Crary-Burney, M.; Ariaspulido, H.; Veliz, F.A.; Steinmetz, N.F.; et al. Treatment of Canine Oral Melanoma with Nanotechnology-Based Immunotherapy and Radiation. Mol. Pharm. 2018, 15, 3717–3722. [Google Scholar] [CrossRef] [PubMed]
| Commercial Name | Virus | Cancer Type | Manufacturers | Tumor Antigens | Adjuvants | Expression System |
|---|---|---|---|---|---|---|
| Engerix-B | HBV | Hepatocellular carcinoma | GSK (UK) | SHBs | Aluminum hydroxide | Yeast: Saccharomyces cerevisiae |
| Recombivax HB (H-B-Vax®II) | HBV | Hepatocellular carcinoma | Merck Vaccine (Canada) | SHBs | Aluminum sulfate | Yeast: Saccharomyces cerevisiae |
| Sci-B-Vac® (Bio-Hep-B®) | HBV | Hepatocellular carcinoma | SCIgen (Israel Ltd.) | SHBs, MHBs, LHBs | Aluminum hydroxide | Mammalian: CHO cells |
| Heplisav-B | HBV | Hepatocellular carcinoma | Dynavax (Oakland-USA) | SHBs | 1018 ISS | Yeast: H. polymorpha |
| Fendrix | HBV | Hepatocellular carcinoma | GSK (Belgium) | SHBs | AS04 (Aluminum hydroxide and MPL) | Yeast: Saccharomyces cerevisiae |
| Hepavax-Gene | HBV | Hepatocellular carcinoma | Crucell (Dusseldorf-Germany) | SHBs, MHBs | Aluminum hydroxide | Yeast: H. polymorpha |
| Cervarix (bivalent vaccine) | HPV | Cervical cancer, anal cancer, penis cancer, head and neck carcinoma | GSK (UK) | L1 HPV 16 L1 HPV 18 | ASO4:Al(OH)3 MPL | Baculovirus: Hi-5 baculovirus |
| Gardasil (quadrivalent vaccine) | HPV | Cervical cancer, anal cancer, penis cancer, head and neck carcinoma | Merck Sharp & Dohme Corp (USA) | L1 HPV6 L1 HPV11 L1 HPV16 L1 HPV18 | Amorphous, aluminum, hydroxyphosphate sulfate | Yeast: Saccharomyces cerevisiae |
| Gardasil 9 (nonavalent vaccine) | HPV | Cervical cancer, anal cancer, penis cancer, head and neck carcinoma | Merck Inc. (Canada) | L1 HPV6 L1 HPV11 L1 HPV16 L1 HPV18 L1 HPV31 L1 HPV33 L1 HPV45 L1 HPV52 L1 HPV58 | Amorphous, aluminum, hydroxyphosphate sulfate | Yeast: Saccharomyces cerevisiae |
| Virus | Cancer Type | Tumor Antigens | Adjuvants | Expression System | References |
|---|---|---|---|---|---|
| Human papillomavirus | Cervical cancer | L2 (epitope 20–38) | Montanide | Pyrococcus furiosus (PfTrx) | [65] |
| Human papillomavirus | Cervical cancer | L2 (epitope 17–31) | alum hydroxide | MS2 and PP7 | [66] |
| Human papillomavirus | Cervical cancer | L2 RG1 epitope and VAR2CSA PM antigen | alum hydroxide | AP205 capsid | [75] |
| Human herpes virus-8 | Kaposi’s sarcoma | gpK8.1 ED fused to NDV fusion protein (F) | alum/MPL | Plasmid transfected into CHO or HEK-293 cells | [76] |
| Epstein–Barr virus | Lymphomas, gastric carcinoma, and nasopharyngeal carcinoma | gp350 and CD21 | none | 293-VII+ cells or exosomes from HEK293 cells | [72] |
| Epstein–Barr virus | Lymphomas, gastric carcinoma, and nasopharyngeal carcinoma | gp350/220 | none | Plasmid transfected in CHO cells | [26] |
| Epstein–Barr virus | Lymphomas, gastric carcinoma, and nasopharyngeal carcinoma | EBNA1 and LMP2 | none | Plasmid transfected in CHO cells | [77] |
| Epstein–Barr virus | Lymphomas, gastric carcinoma, and nasopharyngeal carcinoma | gp350, gB, gp42, and gH/gL | aluminum hydroxide and monophosphoryl lipid A | Plasmid transfected in AGS-EBV-eGFP cells | [78] |
| Epstein–Barr virus | Lymphomas, gastric carcinoma, and nasopharyngeal carcinoma | HBc149, gp350 | aluminum hydroxide and AS04 | E. coli | [79] |
| HCV Hepatitis C virus | Hepatocellular carcinoma | HBV pre-S1 (20-47) | none | E. coli | [80] |
| HCV Hepatitis C virus | Hepatocellular carcinoma | E1, E2 | Alhydrogel | Baculovirus | [81,82] |
| HCV Hepatitis C virus | Hepatocellular carcinoma | E1, E2 | none | Adenovirus | [83] |
| HBV Hepatitis B virus | Hepatocellular carcinoma | HBsAgS | aluminum hydroxide | Plasmid transfected in human embryonic kidney (HEK) FreeStyle-293F cells | [55] |
| HBV Hepatitis B virus | Hepatocellular carcinoma | HBc | none | Pichia pastoris | [56] |
| Cancer Type | Tumor Antigens | Adjuvants | Expression System | Type of Vaccine | References |
|---|---|---|---|---|---|
| Breast cancer | HER2 | none | AP205 phage | Preventive and therapeutic | [100] |
| Breast cancer | IGF-1R | none | Human Parvovirus B19 | Preventive and Therapeutic | [101] |
| Breast cancer | xCT | none | MS2 | Therapeutic | [102] |
| Melanoma | LCMV-TT830–843 | Microcrystalline tyrosine (MCT) | CuMVT | Therapeutic | [103] |
| Melanoma | - | none | Nicotiana bethamiana (CPMV) | Therapeutic | [104] |
| Melanoma | gp100 | none | E. coli | Therapeutic | [105] |
| Pancreatic cancer | mMSLN | none | Baculovirus (SHIV) | Therapeutic | [106] |
| Pancreatic cancer | hMSLN | none | Baculovirus (SHIV) | Therapeutic | [107] |
| Hepatocellular carcinoma | CLDN18.2 | none | E. coli | Therapeutic | [108] |
| Cancer Type | Tumor Antigens | Adjuvants | Expression System | Type of Vaccine | Clinical Trial Identifier | Clinical Stage |
|---|---|---|---|---|---|---|
| Vulva and anal cancer | L1 | Amorphous, aluminum, hydroxyphosphate sulfate | yeast: Saccharomyces cerevisiae | Therapeutic | NCT03051516 | Phase IV |
| HPV infections | L1 | Amorphous, aluminum, hydroxyphosphate sulfate | yeast: Saccharomyces cerevisiae | Prophylactic | NCT03903562 | Phase III |
| Cervical cancer | L1 | Amorphous, aluminum, hydroxyphosphate sulfate | yeast: Saccharomyces cerevisiae | Therapeutic | NCT03284866 | Phase III |
| HPV infections | L1 | Amorphous, aluminum, hydroxyphosphate sulfate | yeast: Saccharomyces cerevisiae | Prophylactic | NCT04235257 | Phase IV |
| Cervical cancer | L1 | Amorphous, aluminum, hydroxyphosphate sulfate | yeast: Saccharomyces cerevisiae | Therapeutic | NCT00092534 | Phase III |
| Pancreatic cancer | - | none | Qβbacteriophage | Therapeutic | NCT04387071 | Phase I/II |
| Melanoma | - | none | Qβbacteriophage | Preventive and Therapeutic | NCT04708418 | Phase II |
| Melanoma | Pembrolizumab (anti-PD-1) | Qβbacteriophage | Therapeutic | NCT02680184 | Phase I |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tornesello, A.L.; Tagliamonte, M.; Buonaguro, F.M.; Tornesello, M.L.; Buonaguro, L. Virus-like Particles as Preventive and Therapeutic Cancer Vaccines. Vaccines 2022, 10, 227. https://doi.org/10.3390/vaccines10020227
Tornesello AL, Tagliamonte M, Buonaguro FM, Tornesello ML, Buonaguro L. Virus-like Particles as Preventive and Therapeutic Cancer Vaccines. Vaccines. 2022; 10(2):227. https://doi.org/10.3390/vaccines10020227
Chicago/Turabian StyleTornesello, Anna Lucia, Maria Tagliamonte, Franco M. Buonaguro, Maria Lina Tornesello, and Luigi Buonaguro. 2022. "Virus-like Particles as Preventive and Therapeutic Cancer Vaccines" Vaccines 10, no. 2: 227. https://doi.org/10.3390/vaccines10020227
APA StyleTornesello, A. L., Tagliamonte, M., Buonaguro, F. M., Tornesello, M. L., & Buonaguro, L. (2022). Virus-like Particles as Preventive and Therapeutic Cancer Vaccines. Vaccines, 10(2), 227. https://doi.org/10.3390/vaccines10020227










