A Narrative Review of COVID-19 Vaccines
Abstract
:1. Introduction
2. SARS-CoV-2 Variants
3. Clinical Overview of COVID-19
4. Traditional Vaccine Discovery
5. Classes of COVID-19 Vaccines
5.1. Whole Virus COVID-19 Vaccines
5.1.1. Inactivated Vaccines
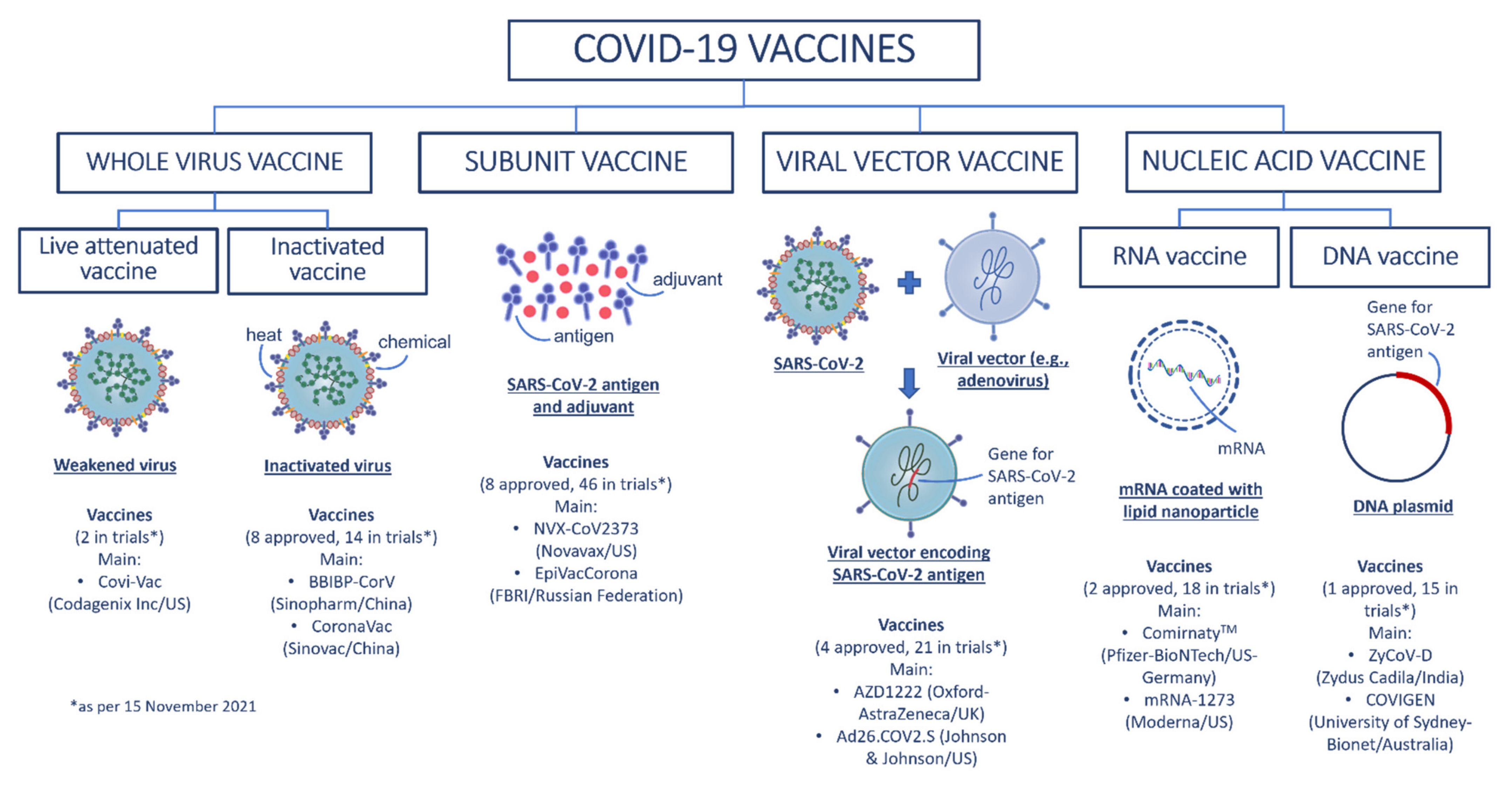
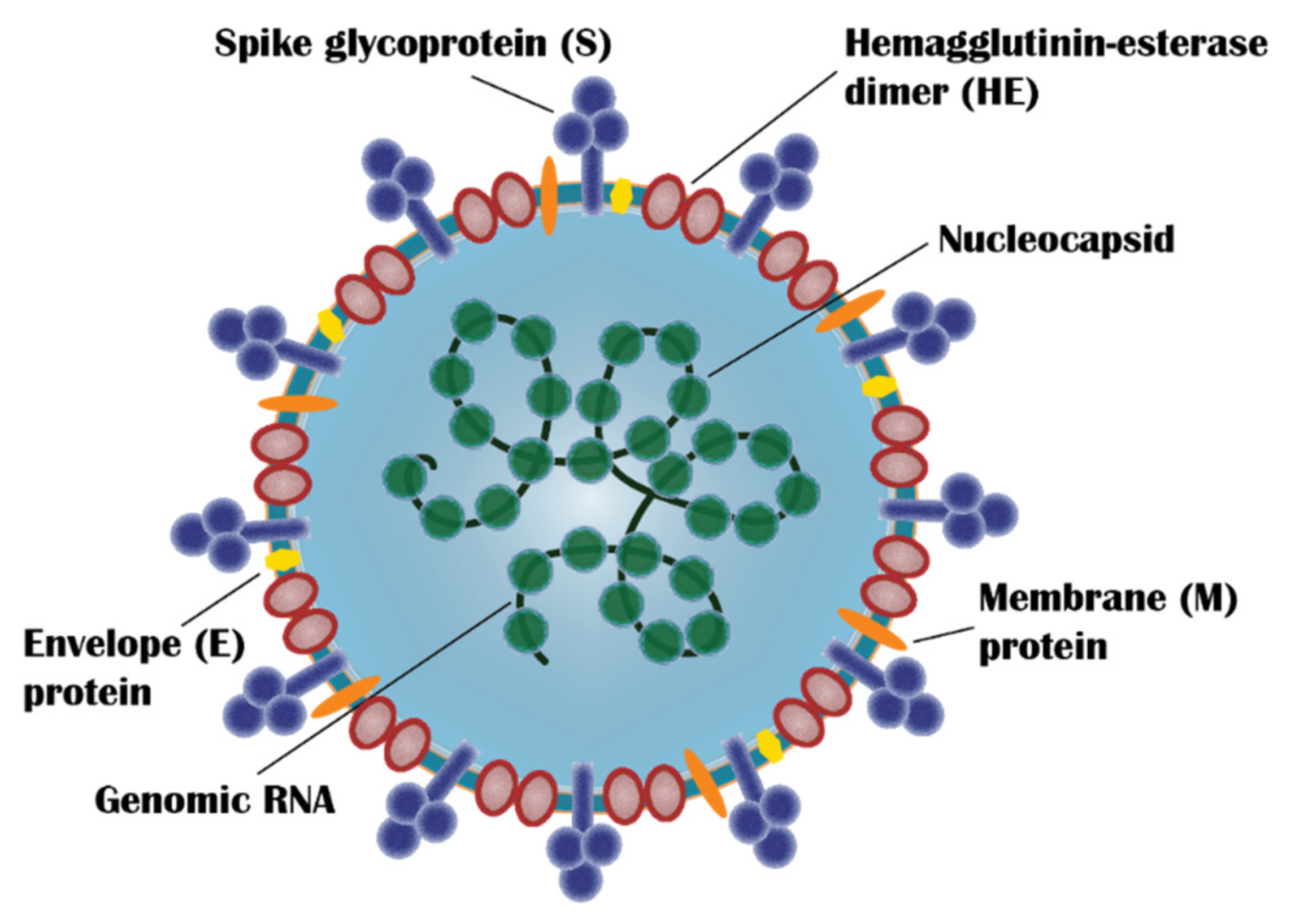
5.1.2. Live Attenuated Vaccines
5.2. Subunit Vaccines
5.3. Viral Vector Vaccines
5.4. Nucleic Acid COVID-19 Vaccines
5.4.1. DNA Vaccines
5.4.2. RNA Vaccines
| No | Vaccine Name | Status | Developer | Vaccine Type | Efficacy | Dose | Storage | Price (per Dose) | Source |
|---|---|---|---|---|---|---|---|---|---|
| 1 | ComirnatyTM (BNT162b2) | Approved in several countries, emergency use in US, elsewhere | Pfizer-BioNTech Germany-US | RNA based vaccine | 95% [83] | 2 dose, 3 weeks apart [84] | −70 °C [85] | €19.50 (US$23.15) [86] | [56] |
| 2 | Moderna mRNA-1273 and mRNA-1273.351 | Approved in Switzerland, emergency use in US, elsewhere | ModernaTX, Inc US | RNA based vaccine | 94.1% [87] | 2 doses, 4 weeks apart [88] | −25 °C [89] | US$25.50 [86] | [56] |
| 3 | AstraZeneca AZD1222 | Approved in Brazil, emergency use in EU, elsewhere | The University of Oxford-AstraZeneca UK | Viral vector (non-replicating) | 76% [90] | 2 doses, between four and 12 weeks apart [91] | 2–8 °C [85] | US$2.15 (EU), US$5.25 (others) [92] | [56] |
| 4 | ConvideciaTM (Ad5-nCoV) | Approved in China, emergency use in other countries | CanSino Biologics China | Viral vector (non-replicating) | 65.28% | Single dose | 2–8 °C [93] | US$27.15 (Pakistan) [94] | [56] |
| 5 | Ad26.COV2.S | Emergency use in US, elsewhere | Janssen (Johnson & Johnson) US | Viral vector (non-replicating) | 66.9% [95] | Single dose [95] | 2–8 °C [85] | US$10 [96] | [56] |
| 6 | BBIBP-CorV | Approved in China, Bahrain, UAE, emergency use in other countries | Sinopharm (Beijing) China | Inactivated virus | 79% [97] | 2 doses, 3 weeks apart [98] | 2–8 °C [99] | US$37.50 (Hungary) [100] | [56] |
| 7 | Inactivated SARS-CoV-2 (vero cell) | Approved in China, Limited use in UAE | Sinopharn + Wuhan Institute of Biological Products China | Inactivated virus | 72.8% | 2 doses, 3 weeks apart | 2–8 °C | N/A | [56] |
| 8 | CoronaVac | Approved in China, emergency use in other countries | Sinovac China | Inactivated virus | 51% in Brazil trial, 84% in Turkey trial [99] | 2 doses, 2 weeks apart [99] | 2–8 °C [89] | US$13.60 (Indonesia) [101] | [56] |
| 9 | Sputnik V | Emergency use in Russia, elsewhere | The Gamaleya Research Institute Russia | Viral vector (non-replicating) | 91.6% | 2 doses, 3 weeks apart | −18 °C [89] | Less than US$10 [102] | [103] |
| 10 | EpiVacCorona | Approved in Turkmenistan, early use in Russia | FBRI Russia | Protein subunit | N/A | 2 doses, 3 weeks apart | 2–8 °C | US$11 [104] | [103] |
| 11 | ZF2001/RBD-Dimer | Emergency use in China, Uzbekistan | Anhui Zhifei Longcom China | Protein subunit | N/A | 3 doses, 4 weeks apart | 2–8 °C | N/A | [103] |
| 12 | Soberana 2/Pasteur | Emergency use in Iran, Cuba | Instituto Finlay de Vacunas Cuba | Protein subunit | 62% two doses, 91.2% with Soberana Plus | 2 doses, 4 weeks apart | 2–8 °C | N/A | [103] |
| 13 | Abdala/CIGB-66 | Emergency use in Cuba | Center for Genetic Engineering and Biotechnology (CIGB) Cuba | Protein subunit | 92.28% | 3 doses, 2 weeks apart | 2–8 °C | N/A | [103] |
| 14 | Medigen | Emergency use in Taiwan | Medigen Vaccine Biologics Taiwan | Protein subunit | N/A | 2 doses, 4 weeks apart | 2–8 °C | N/A | [56] |
| 15 | Covaxin® | Emergency use in India, elsewhere | Bharat Biotech India | Inactivated virus | 77.8% | 2 doses, 4 weeks apart | At least a week at room temperature | US$16.42 (India) [105] | [103] |
| 16 | QazCovid-in® | Early use in Kazakhstan | Research Institute for Biological Safety Problems Kazakhstan | Inactivated virus | N/A | 1 or 2 doses, 3 weeks apart | 2–8 °C | US$4.7 [106] | [103] |
| 17 | Inactivated (Vero Cells) | Emergency use in China | Shenzhen Kangtai Biological Products Co., Ltd. China | Inactivated virus | N/A | 2 doses, 4 weeks apart | 2–8 °C | N/A | [103] |
| 18 | COVIran Barekat | Emergency use in Iran | Shifa Pharmed ParsIran | Inactivated virus | N/A | 2 doses | 2–8 °C | N/A | [103] |
| 19 | CoviVac | Early use in Russia | Chumakov Cente Russia | Inactivated virus | N/A | N/A | 2–8 °C | N/A | [103] |
| 20 | NVX-CoV2373 | Emergency use in Indonesia | Novavax US | Protein subunit | 89.7% | 2 doses, 3 weeks apart | 2–8 °C | US$20.90 (Denmark) [86] | [103] |
| 21 | ZyCoV-D | Emergency use in India | Zydus CadilaIndia | DNA based vaccine | 66.6% | 3 doses, 4 weeks apart | 2–8 °C | N/A | [103] |
| 22 | COVAX-19® | Emergency use in Iran | Vaxine Pty.Ltd/Cinnagen Co. Australia | Protein Subunit | N/A | 2 doses, 3 weeks apart | N/A | N/A | [103] |
| 23 | Soberana Plus | Emergency use in Cuba | Instituto Finlay de Vacunas Cuba | Protein subunit | N/A | N/A | N/A | N/A | [103] |
6. COVID-19 Vaccines—Future Outlook
6.1. Combination COVID-19 Vaccines
6.2. Booster COVID-19 Vaccines
6.3. Mandatory COVID-19 Vaccinations
7. COVID-19 Vaccination Consideration
7.1. Uniform Vaccine Availability and Affordability
7.2. Vaccine Hesitancy
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| No | Vaccine Name | Age Group | Common Side Effect(s) | Main User–Country |
|---|---|---|---|---|
| 1 | ComirnatyTM (BNT162b2) | 5 years of age and older [84] | Pain at the injection site, tiredness, headache, muscle pain, chills, joint pain, and fever | US, UK, South Korea, Singapore, Saudi Arabia, New Zealand, Japan, Israel, Hungary, Germany, France, Canada, Australia |
| 2 | Moderna COVID-19 VaccinemRNA-1273 and mRNA-1273.351 | 18 years of age and older [88] | Pain at the injection site, tiredness, headache, muscle pain, chills, joint pain, swollen lymph nodes in the same arm as the injection, nausea and vomiting, and fever | US, UK, Singapore, France |
| 3 | COVID-19 Vaccine AstraZeneca (AZD1222) | 18 years of age and older [91] | Injection site pain or tenderness, tiredness, headachemuscle pain, fever and chills | UK, South Korea, Saudi Arabia, Phillipines, India, Germany, France, Canada, Brazil, Australia |
| 4 | ConvideciaTM (Ad5-nCoV) | 18 years of age and older [155] | Injection site pain, mild to severe fever (up to grade 3), headache, mild to severe fatigue (up to grade 3), muscle and joint pain, throat pain and cough. | China, Pakistan |
| 5 | Ad26.COV2.S | 18 years of age and older [95] | Arm (pain, redness, swelling), body (tiredness, headache, muscle pain, chills, fever, nausea) | US, South Africa, The Netherlands |
| 6 | BBIBP-CorV | 18 years of age and older [98] | Headaches, fatigue, injection site reactions | China, Hungary, UAE |
| 7 | Inactivated SARS-CoV-2 (vero cell) | 18 years of age and older | Injection site pain, followed by fever, which were mild and self-limiting | China, UAE |
| 8 | CoronaVac | 18 years of age and older [99] | Injection site reactions, fatigue, diarrhea, and muscle pain | China, Brazil, Turkey, Indonesia, Phillipines |
| 9 | Sputnik V | N/A | Headaches, pain at injection site | Russia |
| 10 | EpiVacCorona | N/A | N/A | Turkmenistan, Russia |
| 11 | ZF2001/RBD-Dimer | N/A | Common mild side-effects including injection pain, redness and swelling | China, Uzbekistan |
| 12 | Soberana 2/Pasteur | N/A | Pain and redness at the injection site, general malaise | Iran |
| 13 | Abdala/CIGB-66 | N/A | No serious adverse side effect | Cuba |
| 14 | Medigen | N/A | No vaccine-related serious adverse effects | Taiwan |
| 15 | Covaxin® | 18 years of age and old-er [156] | Fever, headaches, irritability, pain, swelling, or both at the site of injection | India |
| 16 | QazCovid-in® | N/A | No serious side effects | Kazakhstan |
| 17 | Inactivated (Vero Cells) | N/A | N/A | China |
| 18 | COVIran Barekat | N/A | N/A | Iran |
| 19 | CoviVac | N/A | N/A | Russia |
| 20 | NVX-CoV2373 | N/A | N/A | Indonesia, Denmark |
| 21 | ZyCoV-D | N/A | N/A | India |
| 22 | COVAX-19® | N/A | N/A | Iran |
| 23 | Soberana Plus | N/A | N/A | Cuba |
Appendix B
| No | Vaccine Candidate | Developer | Vaccine Type | Developer Country | Trial Phase | Clinical Trial ID | Country | Enrolment Target | Actual/Estimated Trial Dates | Primary Outcome Measures |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Gam-COVID-Vac Sputnik V | Gamaleya Research Institute | Viral vector (non-replicating) | Russia | 3 | NCT04530396 | Russia | 33,758 | 7 September 2020–1 May 2021 | Percentage of trial subjects with COVID-19 developed within 6 months after the first dose |
| 2 | NVX-CoV2373 | Novavax | Protein subunit | US | 3 | NCT04611802 | Mexico, Puerto Rico, US | 33,000 | 27 December 2020–30 June 2023 | Participants with symptoms; reactogenicity incidence and severity; incidence and severity of MAAEs, UnSoAEs, SAEs, AESIs; antibodies to SARS-CoV-2 Nucleoprotein (NP); deaths due to any cause |
| 3 | ZF2001/RBD-Dimer | Anhui Zhifei Longcom | Protein subunit | China | 3 | NCT04646590 | China, Ecuador, Indonesia, Pakistan, Uzbekistan | 29,000 | 16 December 2020–April 2022 | Endpoints of efficacy and safety |
| 4 | CVnCoV | Curevac | RNA based vaccine | Germany | 3 | NCT04652102 | Germany | 36,500 | 14 December 2020–15 May 2022 | Participants with virologically confirmed PCR positive cases of COVID-19 of any severity; participant with MAAEs, SAEs, AESIs |
| 5 | Inactivated (Vero Cells) | Chinese Academy of Medical Sciences | Inactivated virus | China | 3 | NCT04659239 | Brazil, Malaysia | 34,020 | 28 January 2021–July 2022 | Incidence of COVID-19 cases after two-doses of vaccination, the incidence of SoAEs |
| 6 | QazCovid-in® | Research Institute for Biological Safety Problems Kazakhstan | Inactivated virus | Kazakhstan | 3 | NCT04691908 | Kazakhstan | 3000 | 25 December 2020–30 July 2021 | Seroconversion; vaccine immunogenicity versus placebo, frequency of confirmed COVID-19 cases |
| 7 | ZyCoV-D | Zydus Cadila | DNA based vaccine | India | 3 | CTRI/2021/01/030416 | India | 28,216 | N/A | To demonstrate the efficacy of ZyCoV-D in the prevention of virologically confirmed symptomatic COVID-19 cases as compared to placebo |
| 8 | Covaxin | Bharat Biotech | Inactivated virus | India | 3 | NCT04641481 | India | 25,800 | 16 November 2020–December 2022 | First occurrence of virologically confirmed (RT-PCR positive) symptomatic COVID-19 cases |
| 9 | VAT00002: with adjuvant | Sanofi/GSK | Protein subunit | US | 3 | NCT04904549 | US | 37,430 | 26 May 2021–13 January 2023 | Occurrence of symptomatic COVID-19, presence of injection site or systemic reactions, non-serious UnSoAEs, immediate AEs, MAAEs, SAEs, AESIs, and virologically confirmed SARS-CoV-2 infections and/or symptomatic COVID-19 |
| 10 | Inactivated (Vero Cells) | Shenzhen Kangtai Biological Products Co., Ltd. | Inactivated virus | China | 3 | NCT04852705 | China | 28,000 | May 2021–November 2022 | Incidence density of symptomatic COVID-19 cases |
| 11 | FINLAY-FR-2 anti-SARS-CoV-2 Vaccine | Instituto Finlay de Vacunas | Protein subunit | Cuba | 3 | IFV/COR/09 | Cuba | 44,010 | N/A | Virologically confirmed symptomatic COVID-19 infection |
| 12 | EpiVacCorona | FBRI | Protein subunit | Russia | 3 | NCT04780035 | Russia | 3000 | 18 November 2020–September 2021 | The proportion of vaccinated volunteers with no laboratory confirmed symptoms caused by SARS-CoV-2, within 6 months post vaccination versus placebo, the prophylactic efficacy of the vaccine |
| 13 | Recombinant (Sf9 cell) | West China Hospital | Protein subunit | China | 3 | NCT04904471 | China | 40,000 | 1 June 2021–31 December 2022 | Virologically confirmed (polymerase chain reaction [PCR] positive) symptomatic COVID-19 cases at first appearance, regardless of severity; incidence of SAEs, AESIs, MAAEs, SoAEs, UnSoAEs |
| 14 | mRNA vaccine (ARCoV) | Academy of Military Science (AMS), Walvax Biotechnology | RNA based vaccine | China | 3 | NCT04847102 | N/A | 28,000 | 28 May 2021–30 May 2023 | Incidence rate (person-year) of COVID-19 cases, AEs, and SAEs |
| 15 | CIGB-66 | Center for Genetic Engineering and Biotechnology (CIGB) | Protein subunit | Cuba | 3 | RPCEC00000359 | Cuba | 48,000 | N/A | Vaccine efficacy (number of symptomatic COVID-19 subjects with no evidence of previous exposure to viral infection) |
| 16 | VLA2001 | Valneva | Inactivated virus | France | 3 | NCT04864561 | UK | 4000 | 26 April 2021–30 June 2022 | Immune response measured after completion of a 2-dose immunization schedule, as determined by the GMT of SARS-CoV-2-specific neutralizing antibodies, frequency and severity of any AEs |
| 17 | Nanocovax | Nanogen Pharmaceutical Biotechnology | Protein Subunit | Viet Nam | 3 | NCT04922788 | Viet Nam | 13,000 | 7 June 2021–7 August 2022 | Participants who experience a first episode of virologically confirmed case of COVID-19; any severity SAEs, MAAEs; geometric mean of anti-S IgG concentrations; geometric mean of SARS-CoV-2 serum neutralizing titres by plaque reduction neutralization test (PRNT) |
| 18 | ERUCOV-VAC (Turkovac) | Erciyes University | Inactivated virus | Turkey | 3 | NCT04942405 | Turkey | 40,800 | 21 June 2021–31 March 2023 | Protection indexes of two vaccine doses for symptomatic COVID-19, 2 weeks after the second dose of vaccination |
| 19 | ARCT-154 | Arcturus Therapeutics Inc | RNA based vaccine | US | 3 | ISRCTN15779782 | Switzerland | N/A | 3 August 2021–1 September 2023 | Percentage of participants with virologically confirmed COVID-19 |
| 20 | INO-4800 | Inovio Pharmaceuticals | DNA based vaccine | US | 3 | ISRCTN15779782 | Switzerland | N/A | 3 August 2021–1 September 2023 | Percentage of participants with virologically confirmed COVID-19 |
| 21 | SCB-2019 | Clover Biopharmaceuticals/GSK/Dynavax | Protein subunit | Australia | 3 | NCT05012787 | South Africa, Ukraine | 300 | 13 September 2021–16 December 2022 | Participants with AEs, UnSoAEs, SAEs, MAAEs, AESIs, any confirmed relapse of immune-mediated disease |
| 22 | CoVLP | Medicago | Virus like particle | Canada | 3 | NCT04636697 | Canada, US | 900 | 22 November 2021–31 May 2022 | GMTs of the three vaccine lots |
| 23 | COVAX-19® | Vaxine Pty.Ltd/Cinnagen Co. | Protein Subunit | Australia | 2 | IRCT20150303021315N24 | Iran | 16,876 | N/A | Evaluation of COVID-19 incidence |
| 24 | DelNS1-2019-nCoV-RBD-OPT1 | The University of Hong Kong and Xiamen University | Viral vector (Replicating) | Hong Kong | 2 | ChiCTR2100051391 | Hong Kong | N/A | N/A | N/A |
| 25 | BECOV2 | Biological E Limited | Protein subunit | India | 3 | CTRI/2021/08/036074 | India | 2140 | N/A | Immune response measured after completion of 2-dose immunization schedule, as determined by GMT/C |
| 26 | GBP510 | SK Bioscience Co., Ltd. and CEPI | Protein subunit | South Korea | 3 | NCT05007951 | South Korea | 3990 | 30 August 2021–September 2022 | GMT of SARS-CoV-2 neutralizing antibody |
| 27 | COVI-VAC | Codagenix Inc | Live-Attenuated | US | 3 | ISRCTN15779782 | Switzerland | N/A | 3 August 2021–1 September 2023 | Percentage of participants with virologically confirmed COVID-19 |
| 28 | Razi Cov Pars | Razi Vaccine and Serum Research Institute | Protein subunit | Iran | 2 | IRCT20210206050259N3 | Iran | 41,128 | N/A | Occurrence of confirmed symptomatic COVID-19 disease two weeks after the second vaccine dose |
| 29 | AG0301-COVID19 | AnGes + Takarabio + Osaka University | DNA based vaccine | Japan | 2/3 | NCT04655625 | Japan | 500 | 23 November 2020–31 March 2022 | Incidence of treatment-emergent AEs; Immunogenicity |
| 30 | GRAd-COV2 | ReiThera | Viral vector (non-replicating) | Italy | 2/3 | NCT04791423 | Italy | 10,300 | 15 March 2021–30 April 2022 | Participants with symptomatic laboratory confirmed COVID-19, incidence of AEs, SAEs, MAAEs, and AESIs, local and systemic SoAEs, post-treatment GMTs and GMFRs in SARS-CoV-2 S and/or RBD antibodies |
| 31 | UB-612 | Vaxxinity | Protein subunit | US | 2/3 | NCT04683224 | N/A | 7320 | 1 February 2021–22 March 2023 | The incidence of local reactions solicited systemic events, AEs, MAAEs, SAEs and AESIs, change in safety chemistry and hematology blood lab values for assessment of risk in Phase 3, prevention of SARS-CoV-2 infection in adults, change after second dose through to the end of study in antibody titres |
| 32 | GX-19 | Genexine Consortium | DNA based vaccine | South Korea | 2/3 | NCT05067946 | N/A | 14,000 | October 2021–October 2023 | First occurrence of COVID-19 at least 14 days after the second vaccination. Incidence of SoAEs, UnSoAEs, SAEs |
| 33 | rVSV-SARS-CoV-2-S Vaccine | Israel Institute for Biological Research | Viral vector (Replicating) | Israel | 2/3 | NCT04990466 | Israel | 20,000 | 30 September 2021–28 February 2022 | Prevention of Serology-confirmed SARS-CoV-2 infection |
| 34 | COVIran Barekat | Shifa Pharmed Industrial Co | Inactivated virus | Iran | 2/3 | IRCT20201202049567N3 | Iran | 20,000 | N/A | Vaccine efficacy of Shifa-Pharmed inactivated SARS-CoV-2 vaccine |
| 35 | ReCOV | Jiangsu Rec-Biotechnology Co Ltd. | Protein subunit | China | 2/3 | NCT05084989 | N/A | 20,301 | 31 December 2021–31 December 2021 | Number of Participants with Occurrence of COVID-19 cases, AEs, SAEs and AESIs |
| 36 | mRNA-1273.211 | ModernaTX.Inc | RNA based vaccine | US | 2/3 | NCT04927065 | US | 896 | 24 March 2021–22 December 2021 | Vaccine efficacy against SARS-CoV-2 infection; effect of vaccine on peak nasal viral load |
| 37 | AZD2816 | AstraZeneca and The University of Oxford | Viral vector (non-replicating) | UK | 2/3 | NCT04973449 | UK | 2475 | 27 June 2021–15 June 2022 | Safety and tolerability of 1 dose of AZD2816 in seronegative participants previously vaccinated with AZD1222, and 2 doses in unvaccinated seronegative participants |
| 38 | SCTV01C | Sinocelltech Ltd. | Protein Subunit | China | 2/3 | NCT05043311 | N/A | 12,420 | 30 October 2021–1 October 2022 | The incidence of COVID-19 infections |
| 39 | FINLAY-FR-1 | Instituto Finlay de Vacunas Cuba | Protein subunit | Cuba | 2 | IFV/COR/04 | Cuba | 676 | 13 August 2020–11 January 2021 | SAEs, titre of specific anti-RBD IgG antibodies at baseline and 14, 28 and 56 days |
| 40 | LUNAR-COV19/ARCT-021 | Arcturus Therapeutics Inc | RNA based vaccine | US | 2 | NCT04668339 | Singapore, US | 600 | 7 January 2021–30 April 2022 | Local and systemic SoAEs, AEs, SAEs, MAAEs, new onset of chronic disease, abnormal chemistry and hematology values; GMT and GMFR of neutralizing antibody |
| 41 | VXA-CoV2-1 | Vaxart | Viral vector (non-replicating) | US | 2 | NCT05067933 | US | 896 | October 2020–June 2023 | Rate of UnSoAEs, frequency of SAEs and MAAEs |
| 42 | Dendritic cell vaccine AV-COVID-19 | Aivita Biomedical, Inc + Ministry of Health Republic of Indonesia | Viral vector (Replicating) + APC | US | 2 | NCT05007496 | Indonesia | 145 | April 2021–May 2021 | Efficacy based on T-cell-induced immune response |
| 43 | MRT5500 | Sanofi Pasteur | RNA based vaccine | US | 2 | NCT04798027 | US | 333 | 12 March 2021–July 2022 | Presence of immediate AEs, solicited injection site reactions and systemic reactions, UnSoAEs, MAAEs, and AESIs; Presence of out-of-range biological test results, neutralizing antibody titre, seroconversion |
| 44 | SARS-CoV-2 VLP Vaccine | The Scientific and Technological Research Council of Turkey | Virus like particle | Turkey | 2 | NCT04962893 | Turkey | 330 | 26 June 2021–September 2022 | Comparison of efficacy, specific IgG, neutralizing antibody, and cellular immune response |
| 45 | Recombinant SARS-CoV-2 Fusion Protein Vaccine (V-01) | Guangdong Provincial Center for Disease Control and Prevention | Protein subunit | China | 2 | ChiCTR2100045107 | China | 880 | 28 March 2021–30 July 2022 | Positive conversion rate of serum anti-SARS-CoV-2 RBD protein antibody, and its GMT and GMI; positive conversion rate of serum anti-SARS-CoV-2 neutralizing antibody, and its GMI |
| 46 | SCB-2020S | Clover Biopharmaceuticals AUS Pty Ltd. | Protein subunit | Australia | 2 | NCT04950751 | N/A | 150 | August 2021–April 2020 | GMT and GMFR of SARS-CoV-2 neutralising antibodies to B.1.351 variant, proportion of subjects achieving seroconversion of SARS-CoV-2 neutralising antibodies to B.1.351 variant |
| 47 | SC-Ad6-1 | Tetherex Pharmaceuticals Corporation | Viral vector (non-replicating) | US | 2 | NCT05077267 | Germany | 210 | 19 August 2021–1 February 2024 | SARS-CoV-2 neutralizing antibody titers |
| 48 | Recombinant RBD Protein Vaccine | Bagheiat-allah University of Medical Sciences | Protein subunit | Iran | 2 | IRCT20210620051639N2 | Iran | 300 | N/A | IgG antibody against Receptor Binding Domain (RBD) protein |
| 49 | KBP-201 | Kentucky Bioprocessing | Protein subunit | US | 1/2 | NCT04473690 | US | 180 | 31 July 2021–31 July 2022 | Solicited administration site reactions and systemic events |
| 50 | RBD SARS-CoV-2 HBsAg VLP | SpyBiotech + Serum Institute of India | Virus like particle | UK | 1/2 | ACTRN12620000817943 | Australia | 280 | N/A | To assess the immune response, safety and reactogenicity, of a two-dose schedule of two dose amounts of RBD SARS-CoV-2 HBsAg VLP vaccine as compared with two-dose administration of placebo |
| 51 | IMP CoVac-1 | University Hospital Tuebingen | Protein subunit | Germany | 1/2 | NCT04954469 | Germany | 68 | 30 June 2021–31 March 2022 | Safety- Eastern Cooperative Oncology Group (ECOG) status, vital signs, blood chemistry and coagulation, and hematology |
| 52 | LV-SMENP-DC | Shenzhen Geno-Immune Medical Institute | Viral vector (non-replicating) + APC | China | 1/2 | NCT04276896 | China | 100 | 24 March 2020–31 December 2024 | Clinical improvement based on the 7-point scale, lower Murray lung injury score |
| 53 | hAd5-S+N bivalent vaccine | ImmunityBio Inc | Viral vector (non-replicating) | US | 1/2 | NCT04843722 | US | 540 | May 2021–August 2022 | Efficacy: percent of subjects that show an increase in N-reactive T cells |
| 54 | CIGB-669 | Center for Genetic Engineering and Biotechnology (CIGB) | Protein subunit | Cuba | 1/2 | RPCEC00000345 | Cuba | 88 | N/A | Safety: occurrence and intensity of AEs, subjects with seroconversion of anti-RBD IgG antibodies to SARS-CoV-2 |
| 55 | AdCLD-CoV19 | Cellid Co., Ltd. | Viral vector (non-replicating) | South Korea | 1/2 | NCT04666012 | South Korea | 150 | 29 December 2020–April 2022 | Incidence of SoAEs and UnSoAEs |
| 56 | GLS-5310 | GeneOne Life Science Inc | DNA based vaccine | South Korea | 1/2 | NCT04673149 | South Korea | 345 | 23 December 2020–31 December 2022 | Incidence of AEs; GMT of antigen-specific binding antibody titres |
| 57 | S-268019 | Shionogi | Protein subunit | Japan | 1/2 | jRCT2051200092 | Japan | 214 | N/A | AEs, adverse reactions, SAEs, local and systemic reactogenicity SoAEs, GMT of SARS-CoV-2 neutralizing antibody |
| 58 | SARS-CoV-2-RBD-Fc-fusion protein | University Medical Center Groningen + Akston Biosciences Inc. | Protein subunit | The Netherlands | 1/2 | NCT04681092 | The Netherlands | 130 | 12 April 2021–30 June 2021 | Safety / Tolerability (35 days) |
| 59 | COVAC-1 and COVAC-2 | University of Saskatchewan | Protein subunit | Canada | 1/2 | NCT04702178 | Canada | 108 | 10 February 2021–February 2023 | AEs during 28 days after each injection |
| 60 | COVID-eVax | Takis + Rottapharm Biotech | DNA based vaccine | Italy | 1/2 | NCT04788459 | Italy | 160 | 25 February 2021–June 2022 | Local and systemic SoAEs, UnSoAEs, quantitative antibody titres, binding to the specific SARS-CoV-2 antigen, SARS-CoV-2 neutralizing antibody titre, change from baseline in antigen-specific cellular immune responses to SARS-CoV-2, percentage of subjects who seroconverted |
| 61 | Inactivated (NDV based) chimeric vaccine | Mahidol University and Government Pharmaceutical Organization | Inactivated virus | Thailand | 1/2 | NCT04764422 | Thailand | 460 | 20 March 2021–April 2023 | Frequency of reportable local and systemic SoAEs after each vaccination; measurement of changes in hemoglobin, white blood cells, platelet count, creatinine, AST, ALT, bilirubin |
| 62 | VBI-2902a | VBI Vaccines Inc | Virus like particle | Canada | 1/2 | NCT04773665 | Canada | 780 | 15 March 2021–June 2022 | Rate and severity of local and systemic SoAEs, UnSoAEs, MAAEs, SAEs, and laboratory abnormalities; AEs leading to discontinuation of study vaccination |
| 63 | EuCorVac-19 | POP Biotechnologies and EuBiologics Co Ltd. | Protein subunit | South Korea | 1/2 | NCT04783311 | South Korea | 280 | 23 February 2021–January 2023 | Immediate AEs, local and systemic AEs, UnSoAEs, SAEs, AESIs. |
| 64 | DS-5670a | Daiichi Sankyo Co Ltd. | RNA based vaccine | Japan | 1/2 | NCT04821674 | Japan | 152 | 15 March 2021–31 December 2022 | Number of participants reporting treatment-emergent AEs, local and systemic AEs, and SAEs; GMT, GMFR, and SCR of SARS-CoV-2 specific neutralizing antibody |
| 65 | COVIVAC | Institute of Vaccines and Medical Biologicals | Viral vector (non-replicating) | Viet Nam | 1/2 | NCT04830800 | Viet Nam | 420 | 10 March 2021–30 September 2022 | Number and severity of local and systemic SoAEs, UnSoAEs, SAEs, MAAEs, AESIs, and clinically significant hematological and biochemical measurements |
| 66 | Recombinant SARS-CoV-2 Vaccine (CHO Cell) | National Vaccine and Serum Institute | Protein subunit | China | 1/2 | NCT04869592 | China | 3580 | 25 April 2021–25 October 2022 | Incidence and severity of any adverse reactions/events and abnormal blood biochemistry, blood routine, blood coagulation function and urine routine, SAEs, AESIs, GMT of SARS-CoV-2 neutralizing antibody |
| 67 | EXG-5003 | Elixirgen Therapeutics Inc | RNA based vaccine | Japan | 1/2 | NCT04863131 | Japan | 60 | 28 April 2021–31 January 2023 | Number of participants reporting local and systemic AEs |
| 68 | KD-414 | KM Biologics Co Ltd. | Inactivated virus | Japan | 1/2 | jRCT2071200106 | Japan | 210 | N/A | Safety: all adverse events and immunogenicity: neutralizing antibody conversion rate against SARS-CoV-2 |
| 69 | MVA vector expressing stabilized S Protein | German Centre for Infection Research | Viral vector (non-replicating) | Germany | 1/2 | NCT04895449 | Germany | 240 | 1 June 2021–1 March 2022 | Percentage of participants experiencing solicited local or systemic reactogenicity as defined by the study protocol |
| 70 | QazCovac | Research Institute for Biological Safety Problem | Protein subunit | Kazakhstan | 1/2 | NCT04930003 | Kazakhstan | 244 | 15 June 2021–December 2021 | Frequency of AEs for up to 7 and 21 days after immunization. The proportion of volunteers with increased levels of the immune response of specific neutralizing antibody titres using ELISA following the vaccination, compared with placebo |
| 71 | AG0302-COVID19 | AnGes, Inc/Osaka University | DNA based vaccine | Japan | 1/2 | NCT04993586 | Japan | 400 | 29 July 2021–31 December 2021 | Incidence of Treatment-Emergent AEs, immunogenicity |
| 72 | Hipra | Laboratorios Hipra, S.A. | Protein subunit | Spain | 1/2 | NCT05007509 | Spain | 30 | 16 August 2021–September 2022 | Local and systemic SoAEs and UnSoAEs |
| 73 | Versamune-CoV-2FC vaccine | Farmacore Biotecnologia Ltd.a | Protein subunit | Brazil | 1/2 | NCT05016934 | N/A | N/A | 1 November 2021–20 April 2022 | Frequency and severity of local and systemic AEs and AESIs. |
| 74 | ARCT-165 | Arcturus Therapeutics Inc | RNA based vaccine | US | 1/2 | NCT05037097 | Singapore, US | 72 | 30 August 2021–March 2023 | Local and systemic SoAEs, AEs, SAEs, MAAEs, new onset of chronic disease, abnormal chemistry and hematology values; GMT and GMFR of neutralizing antibody |
| 75 | ARCT-021 | Arcturus Therapeutics Inc | RNA based vaccine | US | 1/2 | NCT05037097 | Singapore, US | 72 | 30 August 2021–March 2023 | Local and systemic SoAEs, AEs, SAEs, MAAEs, new onset of chronic disease, abnormal chemistry and hematology values; GMT and GMFR of neutralizing antibody |
| 76 | SII B.1.351, a monovalent (Beta) variant | Novavax | Protein subunit | US | 1/2 | NCT05029856 | Australia | 240 | February 2022–August 2022 | MN50 GMTs to the SARS-CoV-2 B.1.351 (Beta) and B.1.617.2 (Delta), expressed as GMT and SCRs/SRRs. Local and systemic SoAEs, UnSoAEs, MAAEs |
| 77 | SII Bivalent: (ancestral strain and (Beta) variant) | Novavax | Protein subunit | US | 1/2 | NCT05029856 | Australia | 240 | February 2022–August 2022 | MN50 GMTs to the SARS-CoV-2 B.1.351 (Beta) and B.1.617.2 (Delta), expressed as GMT and SCRs/SRRs. Local and systemic SoAEs, UnSoAEs, MAAEs |
| 78 | SII B.1.617.2, monovalent (Delta) variant | Novavax | Protein subunit | US | 1/2 | NCT05029856 | Australia | 240 | February 2022–August 2022 | MN50 GMTs to the SARS-CoV-2 B.1.351 (Beta) and B.1.617.2 (Delta), expressed as GMT and SCRs/SRRs. Local and systemic SoAEs, UnSoAEs, MAAEs |
| 79 | AAV5-RBD-S vaccine (BCD-250) | Biocad | Viral vector (non-replicating) | Russia | 1/2 | NCT05037188 | Russia | 160 | 10 August 2021–December 2022 | Percentage of subjects with ≥ 4 fold rise of serum SARS-CoV-2-specific IgG titer from baseline |
| 80 | CoviVac | Chumakov Federal Scientific Center for Research and Development of Immune-and-Biological Products | Inactivated virus | Russia | 1/2 | NCT05046548 | Russia | 400 | 3 October 2020–1 October 2021 | Geometric mean titer (GMT) |
| 81 | VB10.2129, encoding RBD | Vaccibody AS | DNA based vaccine | Norway | 1/2 | NCT05069623 | Norway | 160 | 27 October 2021–October 2023 | Local and systemic SoAEs, UnSoAEs, SAEs |
| 82 | VB10.2210 | Vaccibody AS | DNA based vaccine | Norway | 1/2 | NCT05069623 | Norway | 160 | 27 October 2021–October 2023 | Local and systemic SoAEs, UnSoAEs, SAEs |
| 83 | SARS-CoV-2 Protein Subunit Recombinant Vaccine | Biofarma | Protein subunit | Indonesia | 1/2 | NCT05067894 | Indonesia | 780 | November 2021–March 2022 | Safety (phase I) and immunogenicity (phase II) of the SARS-CoV-2 protein subunit recombinant vaccine. |
| 84 | MVA-SARS-2-S | University of Munich | Viral vector (non-replicating) | Germany | 1 | NCT04569383 | Germany | 30 | October 2020–May 2021 | Percentage of participants experiencing solicited local or systemic reactogenicity |
| 85 | LNP-nCoVsaRNA | Imperial College London | RNA based vaccine | UK | 1 | ISRCTN17072692 | UK | 320 | 1 April 2020–31 July 2021 | Solicited local injection site reactions, solicited systemic and laboratory reactions, UnSoAEs, SAEs, titres of neutralizing antibody and IgG |
| 86 | Covid-19/aAPC | Shenzhen Geno-Immune Medical Institute | Viral vector (Replicating) | China | 1 | NCT04299724 | China | 100 | 15 February 2020–31 December 2024 | Frequency of vaccine events and serious vaccine events, proportion of subjects with positive T cell response |
| 87 | AdimrSC-2f | Adimmune Corporation | Protein subunit | Taiwan | 1 | NCT04522089 | Taiwan | 70 | 20 August 2020–20 March 2021 | SoAEs and incidence of abnormal laboratory tests results |
| 88 | Covigenix VAX-001 | Entos Pharmaceuticals Inc | DNA based vaccine | Canada | 1 | NCT04591184 | Canada | 72 | 7 April 2021–August 2022 | Safety of a 2-dose regimen of VAX-001 when doses are given 14 days apart, Mean change from baseline in safety laboratory measures, frequency of treatment-emergent SAEs throughout the study and up to 12 months post-second dose |
| 89 | CORVax | Providence Health & Services | DNA based vaccine | US | 1 | NCT04627675 | US | 36 | 30 December 2020–May 2022 | Toxicity and MAAEs at various time frames |
| 90 | ChulaCov19 | Chulalongkorn University | RNA based vaccine | Thailand | 1 | NCT04566276 | Thailand | 96 | January 2021–June 2021 | Frequency and grade of AEs, reportable local and systemic SoAEs |
| 91 | bacTRL-Spike | Symvivo corporation | DNA based vaccine | Canada | 1 | NCT04334980 | Australia | 24 | 2 November 2020–28 February 2022 | Frequency of adverse events up to 12 months post-vaccination |
| 92 | COH04S1 | City of Hope Medical Center | Viral vector (non-replicating) | US | 1 | NCT04639466 | US | 129 | 11 December 2020–10 November 2022 | Incidence of adverse events for up to 365 days |
| 93 | MF59 | The University of Queensland | Protein Subunit | Australia | 1 | NCT04495933 | Australia | 216 | 13 July 2020–8 November 2021 | Local and systemic SoAEs, UnSoAEs, SAEs, GMT of the serum antibody and Nab response |
| 94 | COVIGEN | The University of Sydney Bionet Co., Ltd. | DNA based vaccine | Australia | 1 | NCT04742842 | Australia | 150 | 15 February 2021–31 December 2022 | Frequency of solicited local and systemic reactogenicity AEs, UnSoAEs, SAEs, MAAEs, change in safety laboratory values from baseline |
| 95 | BBV154 | Bharat Biotech | Viral vector (non-replicating) | India | 1 | NCT04751682 | India | 175 | 1 March 2021–30 November 2021 | Incidence of immediate AEs, local and systemic SoAEs, SAEs, and UnSoAEs |
| 96 | PTX-COVID19-B | Providence Therapeutics Holdings Inc | RNA based vaccine | Canada | 1 | NCT04765436 | Canada | 60 | 14 January 2021–14 February 2022 | Occurrence of AEs after each vaccination, assessments of AEs (days 1-42) and safety (days 1-395), immunogenicity analysis |
| 97 | CoV2 SAM (LNP) | GlaxoSmithKline | RNA based vaccine | UK | 1 | NCT04758962 | US | 40 | 15 February 2021–9 May 2022 | Participants with at least 1 solicited administration site event and solicited systemic event, during 7-day follow-up period, with any UnSoAEs during 30-day follow-up period after each vaccination, and with any hematological and biochemical laboratory abnormality at screening, day 1, 2, 8, 31, 32, and 38. |
| 98 | NBP2001 | SK Bioscience Co Ltd. | Protein subunit | South Korea | 1 | NCT04760743 | South Korea | 50 | 17 December 2020–April 2022 | Occurrence of immediate systemic reactions, local and systemic SoAEs, UnSoAEs, SAEs, MAAEs, and AESIs, GMT and GMFR of IgG and neutralizing antibody to the SARS-CoV-2 |
| 99 | ChAdV68-S and AM-LNP-S | Gritstone Oncology | Viral vector (non-replicating) | US | 1 | NCT04776317 | US | 130 | 25 March 2021–19 September 2022 | Frequency by grade of solicited local reactogenicity AEs, solicited systemic reactogenicity AEs, UnSoAEs, AESIs, clinical safety laboratory AEs by severity grade and SAEs |
| 100 | SpFN COVID-19 Vaccine | Walter Reed Army Institute of Research | Protein subunit | US | 1 | NCT04784767 | US | 72 | 5 April 2021–30 October 2023 | Participants with local and systemic reactions, incidents of treatment-adverse events as assessed by FDA toxicity grading scale, and participants with humoral immune response |
| 101 | FAKHRAVAC (MIVAC) | Organization of Defensive Innovation and Research | Inactivated virus | Iran | 1 | IRCT20210206050259N1 | Iran | 135 | N/A | Abnormal vital signs and anaphylactic reactions immediately after vaccination, local and systemic AEs within the first week post-vaccination, abnormal laboratory findings |
| 102 | MV-014-212 | Meissa Vaccines Inc | Live-Attenuated | US | 1 | NCT04798001 | US | 130 | 12 April 2021–31 October 2022 | SoAEs, UnSoAEs, SAEs, MAAEs, and change in serum neutralizing antibody titres against vaccine-encoded SARS-CoV-2 S protein |
| 103 | Koçak-19 Inaktif Adjuvanlı COVID-19 Vaccine | Kocak Farma | Inactivatedviral vecviral virus | Turkey | 1 | NCT04838080 | Turkey | 38 | 19 March 2021–20 October 2021 | AEs, from day 0 until the end of follow up period of 6 months |
| 104 | ABNCoV2 | Radboud University | Virus like particle | The Netherlands | 1 | NCT04839146 | The Netherlands | 42 | 11 March 2021–20 December 2021 | Safety: possibly related Grade 3 AEs and SAEs, Immunogenicity endpoint: concentration of ABNCoV2-specific antibodies |
| 105 | HDT-301 | SENAI CIMATEC | RNA based vaccine | Brazil | 1 | NCT04844268 | N/A | 78 | May 2021–July 2022 | Safety and tolerability of two doses of HDT-1 vaccine (day 1, 29 and 57) |
| 106 | Adjuvanted Inactivated Vaccine | The Scientific and Technological Research Council of Turkey | Inactivated virus | Turkey | 1 | NCT04866069 | Turkey | 50 | 25 April 2021–April 2022 | AAEs, local and systemic SoAEs and UnSoAEs |
| 107 | mRNA-1283 | ModernaTX, Inc | RNA based vaccine | US | 1 | NCT04813796 | US | 125 | 11 March 2021–13 April 2022 | Number of participants with solicited local and systemic reactogenicity Adverse Reactions (ARs), UnSoAEs, and MAAEs, AESIs, and SAEs |
| 108 | Recombinant NDV Vectored Vaccine | Laboratorio Avi-Mex | Inactivated virus | Mexico | 1 | NCT04871737 | Mexico | 90 | 20 May 2021–June 2022 | AEs, pregnancy test, urinalysis, oxygen saturation |
| 109 | mRNACOVID-19 Vaccine | Stemirna Therapeutics Co Ltd. and Shanghai East Hospital | RNA based vaccine | China | 1 | ChiCTR2100045984 | China | 30 | 25 March 2021–25 May 2022 | Safety and immunogenicity |
| 110 | CoVepiT | OSE Immunotherapeutic | Protein subunit | Belgium | 1 | NCT04885361 | Belgium | 48 | 26 May 2021–31 March 2022 | The incidence of solicited local and systemic reactogenicity signs and symptoms, UnSoAEs, SAEs, AESIs, subjects with significantly increased CD8+ T cells responding to SARS-CoV-2 |
| 111 | CoV2-OGEN1 | USSF/Vaxform | Protein subunit | US | 1 | NCT04893512 | N/A | 45 | 7 June 2021–15 September 2022 | Safety evaluation of 2-dose vaccination schedule of orally administered CoV2-OGEN1 by following local and systemic adverse events |
| 112 | LNP-nCOV saRNA-02 Vaccine | MRC/UVRI and LSHTM Uganda Research Unit | RNA based vaccine | Uganda | 1 | NCT04934111 | Uganda | 42 | September 2021–August 2022 | Participant with solicited local injection site and systemic reactions, UnSoAEs, SAEs, titre of neutralizing antibody and IgG |
| 113 | Baiya SARS-CoV-2 Vax 1 Vaccine | Baiya Phytopharm Co Ltd. | Protein subunit | Thailand | 1 | NCT04953078 | N/A | 96 | September 2021–November 2022 | Frequency and grade SoAEs and AEs, change in blood pressure, pulse rate, respiratory rate, and physical condition, safety laboratory value |
| 114 | PIV5-SARS CoV-2 | CyanVac LLC | Viral vector (non-replicating) | US | 1 | NCT04954287 | US | 80 | July 2021–November 2022 | SoAEs, UnSoAEs |
| 115 | 202-CoV | Shanghai Zerun Biotechnology, Walvax Biotechnology | Protein subunit | China | 1 | NCT04982068 | China | 144 | 12 July 2021–October 2022 | SoAEs, UnSoAEs |
| 116 | COVIDITY | Scancell Ltd. | DNA based vaccine | UK | 1 | NCT05047445 | South Africa | 40 | 30 September 2021–June 2022 | Safety and tolerability of COVIDITY as assessed by AEs, vital signs, and physical examination |
| 117 | PIKA-adjuvanted vaccine | Yisheng Biopharma | Protein subunit | Singapore | 1 | ACTRN12621001009808 | Australia, New Zealand | 45 | 24 September 2021–13 June 2022 | To assess the safety and tolerability of the PIKA COVID-19 vaccine by monitoring AEs and clinical laboratory tests |
| 118 | SARS-CoV-2 DNA vaccine | The University of Hong Kong; Immuno Cure 3 Limited | DNA based vaccine | Hong Kong | 1 | NCT05102643 | Hong Kong | 30 | November 2021–December 2022 | Reactogenicity and AEs |
| 119 | Ad5-triCoV/Mac or ChAd-triCoV/Mac | McMaster University | Viral vector (non-replicating) | Canada | 1 | NCT05094609 | Canada | 30 | 9 November 2021–30 June 2023 | Number of participants reporting AEs and severity of AEs following vaccination |
| 120 | T-cell priming specific peptides on a gold nanoparticle | Emergex Vaccines | Protein subunit | Switzerland | 1 | NCT05113862 | Switzerland | 26 | December 2021–November 2022 | Local and systemic SoAEs, UnSoAEs, SAEs, AESIs, SAEs, MAAEs, AESIs. GMT and GMFR of Anti-SAS-CoV-2 RBD IgG and neutralizing anti-SARS-CoV-2 |
| 121 | IN-B009 | HK inno.N Corporation | Protein subunit | South Korea | 1 | NCT05113849 | South Korea | 40 | 16 September 2021–February 2023 | Occurrence of IAR, local and systemic AE, UnSoAE, |
References
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. WHO Virtual Press Conference on COVID-19. Available online: https://www.who.int/docs/default-source/coronaviruse/transcripts/who-audio-emergencies-coronavirus-press-conference-full-and-final-11mar2020.pdf?sfvrsn=cb432bb3_2 (accessed on 11 March 2020).
- World Health Organization. Novel Coronavirus 2019. Available online: https://covid19.who.int/ (accessed on 29 November 2021).
- Sørensen, M.D.; Sørensen, B.; Gonzalez-Dosal, R.; Melchjorsen, C.J.; Weibel, J.; Wang, J.; Jun, C.W.; Huanming, Y.; Kristensen, P. Severe acute respiratory syndrome (SARS): Development of diagnostics and antivirals. Ann. N. Y. Acad. Sci. 2006, 1067, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Hui, D.S.; Perlman, S. Middle East respiratory syndrome. Lancet 2015, 386, 995–1007. [Google Scholar] [CrossRef] [Green Version]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef] [Green Version]
- Gordon, D.E.; Jang, G.M.; Bouhaddou, M.; Xu, J.; Obernier, K.; White, K.M.; O’Meara, M.J.; Rezelj, V.V.; Guo, J.Z.; Swaney, D.L.; et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature 2020, 583, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Bosch, B.J.; van der Zee, R.; de Haan, C.A.; Rottier, P.J. The coronavirus spike protein is a class I virus fusion protein: Structural and functional characterization of the fusion core complex. J. Virol. 2003, 77, 8801–8811. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; Yang, C.; Xu, X.-f.; Xu, W.; Liu, S.-w. Structural and functional properties of SARS-CoV-2 spike protein: Potential antivirus drug development for COVID-19. Acta Pharmacol. Sin. 2020, 41, 1141–1149. [Google Scholar] [CrossRef]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, S.; Zhu, Y.; Liu, M.; Lan, Q.; Xu, W.; Wu, Y.; Ying, T.; Liu, S.; Shi, Z.; Jiang, S.; et al. Fusion mechanism of 2019-nCoV and fusion inhibitors targeting HR1 domain in spike protein. Cell. Mol. Immunol. 2020, 17, 765–767. [Google Scholar] [CrossRef]
- Ju, B.; Zhang, Q.; Ge, J.; Wang, R.; Sun, J.; Ge, X.; Yu, J.; Shan, S.; Zhou, B.; Song, S.; et al. Human neutralizing antibodies elicited by SARS-CoV-2 infection. Nature 2020, 584, 115–119. [Google Scholar] [CrossRef]
- Abbasi-Oshaghi, E.; Mirzaei, F.; Farahani, F.; Khodadadi, I.; Tayebinia, H. Diagnosis and treatment of coronavirus disease 2019 (COVID-19): Laboratory, PCR, and chest CT imaging findings. Int. J. Surg. 2020, 79, 143–153. [Google Scholar] [CrossRef]
- Lauring, A.S.; Hodcroft, E.B. Genetic Variants of SARS-CoV-2—What Do They Mean? JAMA 2021, 325, 529–531. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). SARS-CoV-2 Variant Classifications and Definitions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/variants/variant-info.html (accessed on 22 November 2021).
- WHO. Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. Available online: https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern (accessed on 29 November 2021).
- Wink, P.L.; Zempulski Volpato, F.C.; Monteiro, F.L.; Willig, J.B.; Zavascki, A.P.; Luís Barth, A.; Martins, A.F. First identification of SARS-CoV-2 Lambda (C.37) variant in Southern Brazil. Infect. Control. Hosp. Epidemiol. 2021, 9, 1–2. [Google Scholar] [CrossRef]
- WHO. Tracking SARS-CoV-2 Variants. Available online: https://www.who.int/en/activities/tracking-SARS-CoV-2-variants/ (accessed on 22 November 2021).
- Rambaut, A.; Holmes, E.C.; O’Toole, Á.; Hill, V.; McCrone, J.T.; Ruis, C.; du Plessis, L.; Pybus, O.G. A dynamic nomenclature proposal for SARS-CoV-2 lineages to assist genomic epidemiology. Nat. Microbiol. 2020, 5, 1403–1407. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Jonathan, K.; Kosinski-Collins, M.; Sundberg, E. Coronavirus Structure, Vaccine and Therapy Development. Available online: https://web.mit.edu/fnl/volume/324/king_etal.html (accessed on 6 September 2021).
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Guan, X.; Wu, P.; Wang, X.; Zhou, L.; Tong, Y.; Ren, R.; Leung, K.S.M.; Lau, E.H.Y.; Wong, J.Y.; et al. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N. Engl. J. Med. 2020, 382, 1199–1207. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; De Siati, D.R.; Horoi, M.; Le Bon, S.D.; Rodriguez, A.; Dequanter, D.; Blecic, S.; El Afia, F.; Distinguin, L.; et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): A multicenter European study. Eur. Arch. Oto-Rhino-Laryngol. 2020, 277, 2251–2261. [Google Scholar] [CrossRef] [PubMed]
- Lescure, F.X.; Bouadma, L.; Nguyen, D.; Parisey, M.; Wicky, P.H.; Behillil, S.; Gaymard, A.; Bouscambert-Duchamp, M.; Donati, F.; Le Hingrat, Q.; et al. Clinical and virological data of the first cases of COVID-19 in Europe: A case series. Lancet. Infect. Dis. 2020, 20, 697–706. [Google Scholar] [CrossRef] [Green Version]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 180, 934–943. [Google Scholar] [CrossRef] [Green Version]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; Cohen, S.L.; et al. Presenting Characteristics, Comorbidities, and Outcomes among 5700 Patients Hospitalized with COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef] [PubMed]
- Nath, A. Long-Haul COVID. Neurology 2020, 95, 559–560. [Google Scholar] [CrossRef] [PubMed]
- Carfì, A.; Bernabei, R.; Landi, F.; for the Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J. Infect. 2020, 81, e4–e6. [Google Scholar] [CrossRef] [PubMed]
- Jabri, A.; Kalra, A.; Kumar, A.; Alameh, A.; Adroja, S.; Bashir, H.; Nowacki, A.S.; Shah, R.; Khubber, S.; Kanaa’N, A.; et al. Incidence of Stress Cardiomyopathy During the Coronavirus Disease 2019 Pandemic. JAMA Netw. Open 2020, 3, e2014780. [Google Scholar] [CrossRef] [PubMed]
- Puntmann, V.O.; Carerj, M.L.; Wieters, I.; Fahim, M.; Arendt, C.; Hoffmann, J.; Shchendrygina, A.; Escher, F.; Vasa-Nicotera, M.; Zeiher, A.M.; et al. Outcomes of Cardiovascular Magnetic Resonance Imaging in Patients Recently Recovered From Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- Vincent, M.J.; Bergeron, E.; Benjannet, S.; Erickson, B.R.; Rollin, P.E.; Ksiazek, T.G.; Seidah, N.G.; Nichol, S.T. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol. J. 2005, 2, 69. [Google Scholar] [CrossRef] [Green Version]
- Elavarasi, A.; Prasad, M.; Seth, T.; Sahoo, R.K.; Madan, K.; Nischal, N.; Soneja, M.; Sharma, A.; Maulik, S.K.; Shalimar; et al. Chloroquine and Hydroxychloroquine for the Treatment of COVID-19: A Systematic Review and Meta-analysis. J. Gen. Intern. Med. 2020, 35, 3308–3314. [Google Scholar] [CrossRef]
- Horby, P.; Mafham, M.; Linsell, L.; Bell, J.L.; Staplin, N.; Emberson, J.R.; Wiselka, M.; Ustianowski, A.; Elmahi, E.; Prudon, B.; et al. Effect of Hydroxychloroquine in Hospitalized Patients with COVID-19: Preliminary results from a multi-centre, randomized, controlled trial. medRxiv 2020. [Google Scholar] [CrossRef]
- FDA. Coronavirus (COVID-19) Update: FDA Revokes Emergency Use Authorization for Chloroquine and Hydroxychloroquine. Available online: https://www.fda.gov/news-events/press-announcements/coronavirus-COVID-19-update-fda-revokes-emergency-use-authorization-chloroquine-and (accessed on 22 June 2021).
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Hohmann, E.; Chu, H.Y.; Luetkemeyer, A.; Kline, S.; et al. Remdesivir for the Treatment of COVID-19—Final Report. N. Engl. J. Med. 2020, 383, 1813–1826. [Google Scholar] [CrossRef] [PubMed]
- First COVID-19 Treatment Recommended for EU Authorisation. Available online: https://www.ema.europa.eu/en/news/first-COVID-19-treatment-recommended-eu-authorisation (accessed on 22 June 2021).
- FDA Approves First Treatment for COVID-19. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-COVID-19 (accessed on 22 June 2021).
- The WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group; Sterne, J.A.C.; Murthy, S.; Diaz, J.V.; Slutsky, A.S.; Villar, J.; Angus, D.C.; Annane, D.; Azevedo, L.C.P.; Berwanger, O.; et al. Association Between Administration of Systemic Corticosteroids and Mortality Among Critically Ill Patients With COVID-19: A Meta-analysis. JAMA 2020, 324, 1330–1341. [Google Scholar] [CrossRef]
- NIH. Therapeutic Management of Adults With COVID-19. Available online: https://www.covid19treatmentguidelines.nih.gov/management/therapeutic-management/ (accessed on 22 June 2021).
- Merck and Ridgeback’s Investigational Oral Antiviral Molnupiravir Reduced the Risk of Hospitalization or Death by Approximately 50 Percent Compared to Placebo for Patients with Mild or Moderate COVID-19 in Positive Interim Analysis of Phase 3 Study. Available online: https://www.merck.com/news/merck-and-ridgebacks-investigational-oral-antiviral-molnupiravir-reduced-the-risk-of-hospitalization-or-death-by-approximately-50-percent-compared-to-placebo-for-patients-with-mild-or-moderat/ (accessed on 15 November 2021).
- First Oral Antiviral for COVID-19, Lagevrio (Molnupiravir), Approved by MHRA. Available online: https://www.gov.uk/government/news/first-oral-antiviral-for-COVID-19-lagevrio-molnupiravir-approved-by-mhra (accessed on 15 November 2021).
- Bartoli, A.; Gabrielli, F.; Alicandro, T.; Nascimbeni, F.; Andreone, P. COVID-19 treatment options: A difficult journey between failed attempts and experimental drugs. Intern. Emerg. Med. 2021, 16, 281–308. [Google Scholar] [CrossRef]
- Wolff, J.A.; Malone, R.W.; Williams, P.; Chong, W.; Acsadi, G.; Jani, A.; Felgner, P.L. Direct gene transfer into mouse muscle in vivo. Science 1990, 247, 1465–1468. [Google Scholar] [CrossRef]
- Pfizer’s Novel COVID-19 Oral Antiviral Treatment Candidate Reduced Risk of Hospitalization or Death by 89% in Interim Analysis of Phase 2/3 Epic-Hr Study. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizers-novel-COVID-19-oral-antiviral-treatment-candidate (accessed on 15 November 2021).
- Kayser, V.R.I. A Concise History of Vaccines and Vaccination. Hum. Vaccin. Immunother. 2021; in press. [Google Scholar] [CrossRef]
- Willis, N.J. Edward Jenner and the eradication of smallpox. Scott. Med. J. 1997, 42, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.C.; DeVit, M.; Johnston, S.A. Genetic immunization is a simple method for eliciting an immune response. Nature 1992, 356, 152–154. [Google Scholar] [CrossRef] [PubMed]
- Pronker, E.S.; Weenen, T.C.; Commandeur, H.; Claassen, E.H.J.H.M.; Osterhaus, A.D.M.E. Risk in Vaccine Research and Development Quantified. PLoS ONE 2013, 8, e57755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plotkin, S.; Robinson, J.M.; Cunningham, G.; Iqbal, R.; Larsen, S. The complexity and cost of vaccine manufacturing—An overview. Vaccine 2017, 35, 4064–4071. [Google Scholar] [CrossRef] [PubMed]
- Regulatory Approval of Pfizer/BioNTech Vaccine for COVID-19. Available online: https://www.gov.uk/government/publications/regulatory-approval-of-pfizer-biontech-vaccine-for-COVID-19 (accessed on 17 June 2021).
- FDA Takes Key Action in Fight Against COVID-19 By Issuing Emergency Use Authorization for First COVID-19 Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/fda-takes-key-action-fight-against-COVID-19-issuing-emergency-use-authorization-first-COVID-19 (accessed on 21 June 2021).
- WHO COVID-19 Vaccine Tracker and Landscape. Available online: https://www.who.int/publications/m/item/draft-landscape-of-COVID-19-candidate-vaccines (accessed on 6 August 2021).
- The New York Times Coronavirus Vaccine Tracker. Available online: https://www.nytimes.com/interactive/2020/science/coronavirus-vaccine-tracker.html (accessed on 6 August 2021).
- Sabbaghi, A.; Miri, S.M.; Keshavarz, M.; Zargar, M.; Ghaemi, A. Inactivation methods for whole influenza vaccine production. Rev. Med. Virol. 2019, 29, e2074. [Google Scholar] [CrossRef] [PubMed]
- Sanders, B.; Koldijk, M.; Schuitemaker, H. Inactivated Viral Vaccines. In Vaccine Analysis: Strategies, Principles, and Control; Nunnally, B.K., Turula, V.E., Sitrin, R.D., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 45–80. [Google Scholar]
- Ramesh, N. Types of Vaccines Infographics. Available online: https://sites.bu.edu/covid-corps/projects/science-communication/types-of-vaccines-infographics/ (accessed on 6 September 2021).
- WHO. Status of COVID-19 Vaccines within WHO EUL/PQ Evaluation Process. Available online: https://www.who.int/teams/regulation-prequalification/eul/COVID-19 (accessed on 28 June 2021).
- Al Kaabi, N.; Zhang, Y.; Xia, S.; Yang, Y.; Al Qahtani, M.M.; Abdulrazzaq, N.; Al Nusair, M.; Hassany, M.; Jawad, J.S.; Abdalla, J.; et al. Effect of 2 Inactivated SARS-CoV-2 Vaccines on Symptomatic COVID-19 Infection in Adults: A Randomized Clinical Trial. JAMA 2021, 326, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.-X.; Zhang, T.-T.; Shi, G.-F.; Cheng, F.-M.; Zheng, Y.-M.; Tung, T.-H.; Chen, H.-X. Safety of an inactivated SARS-CoV-2 vaccine among healthcare workers in China. Expert Rev. Vaccines 2021, 20, 891–898. [Google Scholar] [CrossRef]
- Sinovac. Responses to the SAGE Working Group on COVID-19 Vaccines. Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-vaccines-SAGE_recommendation-Sinovac-CoronaVac-background-2021.1 (accessed on 21 April 2021).
- Hansson, M.; Nygren, P.A.; Ståhl, S. Design and production of recombinant subunit vaccines. Biotechnol. Appl. Biochem. 2000, 32, 95–107. [Google Scholar] [CrossRef]
- Christensen, D. Vaccine adjuvants: Why and how. Hum. Vaccines Immunother 2016, 12, 2709–2711. [Google Scholar] [CrossRef] [Green Version]
- Oxford. Types of Vaccine. Available online: https://vk.ovg.ox.ac.uk/vk/types-of-vaccine (accessed on 12 August 2021).
- NIH. Vaccine Types. Available online: https://www.niaid.nih.gov/research/vaccine-types (accessed on 12 August 2021).
- Dai, L.; Gao, G.F. Viral targets for vaccines against COVID-19. Nat. Rev. Immunol. 2021, 21, 73–82. [Google Scholar] [CrossRef]
- Ura, T.; Okuda, K.; Shimada, M. Developments in Viral Vector-Based Vaccines. Vaccines 2014, 2, 624–641. [Google Scholar] [CrossRef] [Green Version]
- Jackson, D.A.; Symons, R.H.; Berg, P. Biochemical Method for Inserting New Genetic Information into DNA of Simian Virus 40: Circular SV40 DNA Molecules Containing Lambda Phage Genes and the Galactose Operon of Escherichia coli. Proc. Natl. Acad. Sci. USA 1972, 69, 2904–2909. [Google Scholar] [CrossRef] [Green Version]
- Vrba, S.M.; Kirk, N.M.; Brisse, M.E.; Liang, Y.; Ly, H. Development and Applications of Viral Vectored Vaccines to Combat Zoonotic and Emerging Public Health Threats. Vaccines 2020, 8, 680. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: A pooled analysis of four randomised trials. Lancet 2021, 397, 881–891. [Google Scholar] [CrossRef]
- Pottegård, A.; Lund, L.C.; Karlstad, Ø.; Dahl, J.; Andersen, M.; Hallas, J.; Lidegaard, Ø.; Tapia, G.; Gulseth, H.L.; Ruiz, P.L.-D.; et al. Arterial events, venous thromboembolism, thrombocytopenia, and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-S in Denmark and Norway: Population based cohort study. BMJ 2021, 373, n1114. [Google Scholar] [CrossRef]
- Wise, J. COVID-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ 2021, 372, n699. [Google Scholar] [CrossRef]
- Baker, A.T.; Boyd, R.J.; Sarkar, D.; Teijeira-Crespo, A.; Chan, C.K.; Bates, E.; Waraich, K.; Vant, J.; Wilson, E.; Truong, C.D.; et al. ChAdOx1 interacts with CAR and PF4 with implications for thrombosis with thrombocytopenia syndrome. Sci. Adv. 2021, 7, eabl8213. [Google Scholar] [CrossRef] [PubMed]
- Vogel, F.R.; Sarver, N. Nucleic acid vaccines. Clin. Microbiol. Rev. 1995, 8, 406–410. [Google Scholar] [CrossRef]
- Zhang, C.; Maruggi, G.; Shan, H.; Li, J. Advances in mRNA Vaccines for Infectious Diseases. Front. Immunol. 2019, 10, 594. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Gascón, A.; del Pozo-Rodríguez, A.; Solinís, M.Á. Development of nucleic acid vaccines: Use of self-amplifying RNA in lipid nanoparticles. Int. J. Nanomed. 2014, 9, 1833–1843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silveira, M.M.; Moreira, G.M.S.G.; Mendonça, M. DNA vaccines against COVID-19: Perspectives and challenges. Life Sci. 2021, 267, 118919. [Google Scholar] [CrossRef] [PubMed]
- Pardi, N.; Hogan, M.J.; Porter, F.W.; Weissman, D. mRNA vaccines—A new era in vaccinology. Nat. Rev. Drug Discov. 2018, 17, 261–279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, M.A. A Comparison of Plasmid DNA and mRNA as Vaccine Technologies. Vaccines 2019, 7, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- FDA. U.S. Pfizer-BioNTech COVID-19 Vaccine EUA Letter of Authorization Reissued. Available online: https://www.fda.gov/media/144412/download (accessed on 5 October 2021).
- FDA. U.S. Pfizer-BioNTech COVID-19 Vaccine. Available online: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-COVID-19/pfizer-biontech-COVID-19-vaccine (accessed on 18 May 2021).
- Holm, M.R.; Poland, G.A. Critical aspects of packaging, storage, preparation, and administration of mRNA and adenovirus-vectored COVID-19 vaccines for optimal efficacy. Vaccine 2021, 39, 457–459. [Google Scholar] [CrossRef] [PubMed]
- Kollewe, J. COVID-19 Vaccines: The Contracts, Prices and Profits. Available online: https://www.theguardian.com/world/2021/aug/11/COVID-19-vaccines-the-contracts-prices-and-profits (accessed on 12 August 2021).
- FDA. U.S. Moderna COVID-19 Vaccine EUA Letter of Authorization. Available online: https://www.fda.gov/media/144636/download (accessed on 18 May 2021).
- FDA. U.S. Moderna COVID-19 Vaccine. Available online: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-COVID-19/moderna-COVID-19-vaccine (accessed on 18 May 2021).
- Santos, A.F.; Gaspar, P.D.; de Souza, H.J.L. Refrigeration of COVID-19 Vaccines: Ideal Storage Characteristics, Energy Efficiency and Environmental Impacts of Various Vaccine Options. Energies 2021, 14, 1849. [Google Scholar] [CrossRef]
- Vogel, G.; Kupferschmidt, K. Side effect worry grows for AstraZeneca vaccine. Science 2021, 372, 14. [Google Scholar] [CrossRef]
- Torjesen, I. COVID-19: AstraZeneca vaccine is approved in EU with no upper age limit. BMJ 2021, 372, n295. [Google Scholar] [CrossRef] [PubMed]
- Dyer, O. COVID-19: Countries are learning what others paid for vaccines. BMJ 2021, 372, n281. [Google Scholar] [CrossRef]
- McDougallScientific. 2021 COVID-19 Vaccines: Summary, Updates and Status. Available online: https://www.mcdougallscientific.com/wp-content/uploads/2021-COVID-19-Vaccine-and-Clicnial-Trials-Update.pdf (accessed on 12 August 2021).
- Farooq, U.; Shahzad, A. Pakistan to Start Private Imports of CanSino COVID-19 Vaccine for Sale. Available online: https://www.reuters.com/article/us-health-coronavirus-pakistan-vaccine-idUSKBN2BE0N3 (accessed on 12 August 2021).
- WHO. The J&J COVID-19 Vaccine: What You Need to Know. Available online: https://www.who.int/news-room/feature-stories/detail/the-j-j-COVID-19-vaccine-what-you-need-to-know (accessed on 18 May 2021).
- Trefis Team. What’s Happening With Johnson & Johnson’s COVID-19 Vaccine? Available online: https://www.forbes.com/sites/greatspeculations/2021/06/09/whats-happening-with-johnson--johnsons-COVID-19-vaccine/?sh=53842c5f1aed (accessed on 12 August 2021).
- WHO. The Sinopharm COVID-19 Vaccine: What You Need to Know. Available online: https://www.who.int/news-room/feature-stories/detail/the-sinopharm-COVID-19-vaccine-what-you-need-to-know#:~:text=A%20large%20multi%2Dcountry%20Phase,efficacy%20against%20hospitalization%20was%2079%25. (accessed on 18 May 2021).
- WHO. Evidence Assessment: Sinopharm/BBIBP COVID-19 Vaccine. Available online: https://cdn.who.int/media/docs/default-source/immunization/sage/2021/april/2_sage29apr2021_critical-evidence_sinopharm.pdf (accessed on 18 May 2021).
- WHO. COVID-19 Vaccine (Vero Cell), Inactivated (Sinopharm). Available online: https://www.who.int/docs/default-source/coronaviruse/v.3_21195_sinopharm-vaccine-explainer-24.pdf?sfvrsn=e7507eb4_19&download=true (accessed on 12 August 2021).
- Reuters. Hungary Publishes Chinese, Russian Vaccine Contracts Amid COVID-19 Surge. Available online: https://www.reuters.com/article/us-health-coronavirus-hungary-idUSKBN2B30YP (accessed on 12 August 2021).
- Reuters. Sinovac Coronavirus Vaccine Offered by Chinese City for Emergency Use Costs $60. Available online: https://www.reuters.com/article/us-health-coronavirus-china-vaccine-idUSKBN2710UQ (accessed on 12 August 2021).
- Sputnik, V. The Cost of One Dose of the Sputnik V Vaccine Will be Less Than $10 for International Market. Available online: https://sputnikvaccine.com/newsroom/pressreleases/the-cost-of-one-dose-will-be-less-than-10-for-international-markets/ (accessed on 11 September 2021).
- Zimmer, C.; Corum, J.; Wee, S.-L. Coronavirus Vaccine Tracker. Available online: https://www.nytimes.com/interactive/2020/science/coronavirus-vaccine-tracker.html? (accessed on 15 November 2021).
- PrecisionVax. EpiVacCorona Vaccine. Available online: https://www.precisionvaccinations.com/vaccines/epivaccorona-vaccine (accessed on 11 September 2021).
- Kapur, M. For All Its “Made in India” Pitch, Covaxin Is the Most Expensive COVID-19 Vaccine in India. Available online: https://qz.com/india/2019375/why-does-covaxin-cost-more-than-covishield-and-sputnik-in-india/ (accessed on 11 September 2021).
- Demesinova, A. Kazakhstan Vaccine against CVI to be Rolled Out in Late April. Available online: https://www.kazpravda.kz/en/news/society/kazakhstan-vaccine-against-cvi-to-be-rolled-out-in-late-april (accessed on 11 September 2021).
- He, Q.; Mao, Q.; An, C.; Zhang, J.; Gao, F.; Bian, L.; Li, C.; Liang, Z.; Xu, M.; Wang, J. Heterologous prime-boost: Breaking the protective immune response bottleneck of COVID-19 vaccine candidates. Emerg. Microbes Infect. 2021, 10, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Callaway, E. Mix-And-Match COVID Vaccines Trigger Potent Immune Response. Nature 2021, 593, 491. [Google Scholar] [CrossRef]
- Liu, X.; Shaw, R.H.; Stuart, A.S.V.; Greenland, M.; Aley, P.K.; Andrews, N.J.; Cameron, J.C.; Charlton, S.; Clutterbuck, E.A.; Collins, A.M.; et al. Safety and immunogenicity of heterologous versus homologous prime-boost schedules with an adenoviral vectored and mRNA COVID-19 vaccine (Com-COV): A single-blind, randomised, non-inferiority trial. Lancet 2021, 398, 856–869. [Google Scholar] [CrossRef]
- Hillus, D.; Schwarz, T.; Tober-Lau, P.; Hastor, H.; Thibeault, C.; Kasper, S.; Helbig, E.T.; Lippert, L.J.; Tscheak, P.; Schmidt, M.L.; et al. Safety, reactogenicity, and immunogenicity of homologous and heterologous prime-boost immunisation with ChAdOx1-nCoV19 and BNT162b2: A prospective cohort study. medRxiv 2021. [Google Scholar] [CrossRef]
- Schmidt, T.; Klemis, V.; Schub, D.; Mihm, J.; Hielscher, F.; Marx, S.; Abu-Omar, A.; Schneitler, S.; Becker, S.L.; Gärtner, B.C.; et al. Immunogenicity and reactogenicity of a heterologous COVID-19 prime-boost vaccination compared with homologous vaccine regimens. medRxiv 2021. medRxiv: 2021.06.13.21258859. [Google Scholar] [CrossRef]
- CDC. Delta Variant: What We Know About the Science? Available online: https://www.cdc.gov/coronavirus/2019-ncov/variants/delta-variant.html (accessed on 15 August 2021).
- Mahase, E. Delta variant: What is happening with transmission, hospital admissions, and restrictions? BMJ 2021, 373, n1513. [Google Scholar] [CrossRef]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of COVID-19 Vaccines against the B.1.617.2 (Delta) Variant. N. Engl. J. Med. 2021, 385, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Bergwerk, M.; Gonen, T.; Lustig, Y.; Amit, S.; Lipsitch, M.; Cohen, C.; Mandelboim, M.; Levin, E.G.; Rubin, C.; Indenbaum, V.; et al. COVID-19 Breakthrough Infections in Vaccinated Health Care Workers. N. Engl. J. Med. 2021, 385, 1474–1484. [Google Scholar] [CrossRef]
- Stephenson, J. COVID-19 Vaccinations in Nursing Home Residents and Staff Give Robust Protection, Though Breakthrough Infections Still Possible. JAMA Health Forum 2021, 2, e211195. [Google Scholar] [CrossRef]
- Shrotri, M.; Navaratnam, A.M.D.; Nguyen, V.; Byrne, T.; Geismar, C.; Fragaszy, E.; Beale, S.; Fong, W.L.E.; Patel, P.; Kovar, J.; et al. Spike-antibody waning after second dose of BNT162b2 or ChAdOx1. Lancet 2021, 398, 385–387. [Google Scholar] [CrossRef]
- Bar-On, Y.M.; Goldberg, Y.; Mandel, M.; Bodenheimer, O.; Freedman, L.; Kalkstein, N.; Mizrahi, B.; Alroy-Preis, S.; Ash, N.; Milo, R.; et al. Protection of BNT162b2 Vaccine Booster against COVID-19 in Israel. N. Engl. J. Med. 2021, 385, 1393–1400. [Google Scholar] [CrossRef] [PubMed]
- Munro, A.P.S.; Janani, L.; Cornelius, V.; Aley, P.K.; Babbage, G.; Baxter, D.; Bula, M.; Cathie, K.; Chatterjee, K.; Dodd, K.; et al. Safety and immunogenicity of seven COVID-19 vaccines as a third dose (booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV-BOOST): A blinded, multicentre, randomised, controlled, phase 2 trial. Lancet 2021, 398, 2258–2276. [Google Scholar] [CrossRef]
- Factbox: Countries Weigh Need for Booster COVID-19 Shots. Available online: https://www.reuters.com/business/healthcare-pharmaceuticals/countries-weigh-need-booster-COVID-19-shots-2021-08-05/ (accessed on 15 August 2021).
- Moscow Begins Booster Vaccine Campaign as Cases Surge. Available online: https://www.reuters.com/business/healthcare-pharmaceuticals/moscow-begins-booster-vaccine-campaign-russias-COVID-19-cases-surge-2021-07-01/ (accessed on 15 August 2021).
- Turkey Offers COVID-19 Booster Shot After Early Use of Chinese Vaccine. Available online: https://www.wsj.com/articles/turkey-offers-COVID-19-booster-shot-after-using-chinese-vaccine-in-early-drive-11625144544 (accessed on 15 August 2021).
- FDA. Coronavirus (COVID-19) Update: FDA Authorizes Additional Vaccine Dose for Certain Immunocompromised Individuals. Available online: https://www.fda.gov/news-events/press-announcements/coronavirus-COVID-19-update-fda-authorizes-additional-vaccine-dose-certain-immunocompromised (accessed on 15 August 2021).
- WHO. Interim Statement on COVID-19 Vaccine Booster Doses. Available online: https://www.who.int/news/item/10-08-2021-interim-statement-on-COVID-19-vaccine-booster-doses (accessed on 16 August 2021).
- Tajikistan Declares Mandatory COVID-19 Vaccination. Available online: https://tass.com/world/1310239 (accessed on 15 August 2021).
- Dyer, O. COVID-19: Turkmenistan becomes first country to make vaccination mandatory for all adults. BMJ 2021, 374, n1766. [Google Scholar] [CrossRef]
- Saudi Arabia’s Vaccination Rate Spikes as Deadline Approaches. Available online: https://www.bloomberg.com/news/articles/2021-07-15/saudi-arabia-s-vaccination-rate-spikes-as-deadline-approaches (accessed on 15 August 2021).
- Factbox: Countries Making COVID-19 Vaccines Mandatory. Available online: https://www.reuters.com/world/countries-make-COVID-19-vaccines-mandatory-2021-07-13/ (accessed on 15 August 2021).
- California and New York City to Mandate Vaccine for Government Workers. Available online: https://www.theguardian.com/us-news/2021/jul/26/covid-california-new-york-city-department-of-veterans-affairs-vaccine-mandate (accessed on 15 August 2021).
- Coronavirus: Thousands Protest against Restrictions across Europe. Available online: https://www.dw.com/en/coronavirus-thousands-protest-against-restrictions-across-europe/a-58627841 (accessed on 15 August 2021).
- COVAX—Working for Global Equitable Access to COVID-19 Vaccines. Available online: https://www.who.int/initiatives/act-accelerator/covax (accessed on 24 August 2021).
- COVAX Vaccine Roll-Out. Available online: https://www.gavi.org/covax-vaccine-roll-out#country-updates (accessed on 24 August 2021).
- CEPI. COVAX: CEPI’s Response to COVID-19. Available online: https://cepi.net/covax/ (accessed on 24 August 2021).
- Director-General’s Opening Remarks at the Media Briefing on COVID-19. Available online: https://www.who.int/director-general/speeches/detail/director-general-s-opening-remarks-at-the-media-briefing-on-COVID-19-9-april-2021 (accessed on 4 September 2021).
- MacDonald, N.E.; Sage Working Group on Vaccine Hesitancy. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 33, 4161–4164. [Google Scholar] [CrossRef]
- Durbach, N. ‘They Might As Well Brand Us’: Working-Class Resistance to Compulsory Vaccination in Victorian England. Soc. Hist. Med. 2000, 13, 45–63. [Google Scholar] [CrossRef]
- Patel, M.K.; Orenstein, W.A. Classification of global measles cases in 2013–17 as due to policy or vaccination failure: A retrospective review of global surveillance data. Lancet Glob. Health 2019, 7, e313–e320. [Google Scholar] [CrossRef] [Green Version]
- de Figueiredo, A.; Simas, C.; Karafillakis, E.; Paterson, P.; Larson, H.J. Mapping global trends in vaccine confidence and investigating barriers to vaccine uptake: A large-scale retrospective temporal modelling study. Lancet 2020, 396, 898–908. [Google Scholar] [CrossRef]
- Smith, T.C. Vaccine Rejection and Hesitancy: A Review and Call to Action. Open Forum Infect. Dis. 2017, 4, ofx146. [Google Scholar] [CrossRef] [Green Version]
- Zucker, J.R.; Rosen, J.B.; Iwamoto, M.; Arciuolo, R.J.; Langdon-Embry, M.; Vora, N.M.; Rakeman, J.L.; Isaac, B.M.; Jean, A.; Asfaw, M.; et al. Consequences of Undervaccination—Measles Outbreak, New York City, 2018–2019. N. Engl. J. Med. 2020, 382, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Wakefield, A.J.; Murch, S.H.; Anthony, A.; Linnell, J.; Casson, D.M.; Malik, M.; Berelowitz, M.; Dhillon, A.P.; Thomson, M.A.; Harvey, P.; et al. RETRACTED: Ileal-lymphoid-nodular hyperplasia, non-specific colitis, and pervasive developmental disorder in children. Lancet 1998, 351, 637–641. [Google Scholar] [CrossRef]
- Taylor, B.; Miller, E.; Farrington, C.; Petropoulos, M.-C.; Favot-Mayaud, I.; Li, J.; Waight, P.A. Autism and measles, mumps, and rubella vaccine: No epidemiological evidence for a causal association. Lancet 1999, 353, 2026–2029. [Google Scholar] [CrossRef] [Green Version]
- Kennedy, J. Populist politics and vaccine hesitancy in Western Europe: An analysis of national-level data. Eur. J. Public Health 2019, 29, 512–516. [Google Scholar] [CrossRef]
- Germani, F.; Biller-Andorno, N. The anti-vaccination infodemic on social media: A behavioral analysis. PLoS ONE 2021, 16, e0247642. [Google Scholar] [CrossRef]
- Zhang, E.J.; Chughtai, A.A.; Heywood, A.; MacIntyre, C.R. Influence of political and medical leaders on parental perception of vaccination: A cross-sectional survey in Australia. BMJ Open 2019, 9, e025866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kata, A. Anti-vaccine activists, Web 2.0, and the postmodern paradigm—An overview of tactics and tropes used online by the anti-vaccination movement. Vaccine 2012, 30, 3778–3789. [Google Scholar] [CrossRef]
- WHO. Ten Threats to Global Health in 2019. Available online: https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019 (accessed on 18 July 2021).
- Sarasty, O.; Carpio, C.E.; Hudson, D.; Guerrero-Ochoa, P.A.; Borja, I. The demand for a COVID-19 vaccine in Ecuador. Vaccine 2020, 38, 8090–8098. [Google Scholar] [CrossRef]
- Sallam, M.; Dababseh, D.; Eid, H.; Al-Mahzoum, K.; Al-Haidar, A.; Taim, D.; Yaseen, A.; Ababneh, N.A.; Bakri, F.G.; Mahafzah, A. High Rates of COVID-19 Vaccine Hesitancy and Its Association with Conspiracy Beliefs: A Study in Jordan and Kuwait among Other Arab Countries. Vaccines 2021, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Dror, A.A.; Eisenbach, N.; Taiber, S.; Morozov, N.G.; Mizrachi, M.; Zigron, A.; Srouji, S.; Sela, E. Vaccine hesitancy: The next challenge in the fight against COVID-19. Eur. J. Epidemiol. 2020, 35, 775–779. [Google Scholar] [CrossRef]
- Robinson, E.; Jones, A.; Lesser, I.; Daly, M. International estimates of intended uptake and refusal of COVID-19 vaccines: A rapid systematic review and meta-analysis of large nationally representative samples. Vaccine 2021, 39, 2024–2034. [Google Scholar] [CrossRef]
- Daly, M.; Robinson, E. Willingness to vaccinate against COVID-19 in the US: Longitudinal evidence from a nationally representative sample of adults from April–October 2020. medRxiv 2020. [Google Scholar] [CrossRef]
- Roozenbeek, J.; Schneider, C.R.; Dryhurst, S.; Kerr, J.; Freeman, A.L.J.; Recchia, G.; van der Bles, A.M.; van der Linden, S. Susceptibility to misinformation about COVID-19 around the world. R. Soc. Open Sci. 2020, 7, 201199. [Google Scholar] [CrossRef]
- Yasmin, F.; Najeeb, H.; Moeed, A.; Naeem, U.; Asghar, M.S.; Chughtai, N.U.; Yousaf, Z.; Seboka, B.T.; Ullah, I.; Lin, C.Y.; et al. COVID-19 Vaccine Hesitancy in the United States: A Systematic Review. Front. Public Health 2021, 9, 770985. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.-C.; Guan, X.-H.; Li, Y.-H.; Huang, J.-Y.; Jiang, T.; Hou, L.-H.; Li, J.-X.; Yang, B.-F.; Wang, L.; Wang, W.-J.; et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2020, 396, 479–488. [Google Scholar] [CrossRef]
- WHO. Strategic Advisory Group of Experts on Immunization (SAGE) COVID-19 Vaccines Technical Documents. Available online: https://www.who.int/groups/strategic-advisory-group-of-experts-on-immunization/COVID-19-materials (accessed on 22 November 2021).

| WHO Terminology | Pangolin * | S Protein Mutations of Interest | Country of First Detection | Time of First Detection | |
|---|---|---|---|---|---|
| VBM | Epsilon | B.1.427/B.1.429 | L452R, D614G, S13I, W152C, L452R, D614G | United States (California) | September 2020 |
| Eta | B.1.525 | A67V, 69del, 70del, 144del, E484K, D614G, Q677H, F888L | United Kingdom/Nigeria | December 2020 | |
| Iota | B.1.526 | L5F, (D80G*), T95I, (Y144-*), (F157S*), D253G, (L452R*), (S477N*), E484K, D614G, A701V, (T859N*), (D950H*), (Q957R*) | United States (New York) | November 2020 | |
| Kappa | B.1.617.1 | (T95I), G142D, E154K, L452R, E484Q, D614G, P681R, Q1071H | India | December 2020 | |
| Zeta | P.2 | E484K, (F565L*), D614G, V1176F | Brazil | April 2020 | |
| Lambda | C.37 | G75V, T76I, 246-252del, L452Q, F490S, D614G and T859N | Peru | December 2020 | |
| Mu | B.1.621, B.1.621.1 | N/A | Colombia | September 2021 | |
| Alpha | B.1.1.7 | 69del, 70del, 144del, (E484K*), (S494P*), N501Y, A570D, D614G, P681H, T716I, S982A, D1118H (K1191N*) | United Kingdom | September 2020 | |
| Beta | B.1.351 | D80A, D215G, 241del, 242del, 243del, K417N, E484K, N501Y, D614G, A701V | South Africa | May 2020 | |
| Gamma | P.1 | L18F, T20N, P26S, D138Y, R190S, K417T, E484K, N501Y, D614G, H655Y, T1027I | Japan/Brazil | November 2020 | |
| VOC | Delta | B.1.617.2 | T19R, (V70F*), T95I, G142D, E156-, F157-, R158G, (A222V*), (W258L*), (K417N*), L452R, T478K, D614G, P681R, D950N | India | October 2020 |
| Omicron | B.1.1.529 | N/A | South Africa | November 2021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eroglu, B.; Nuwarda, R.F.; Ramzan, I.; Kayser, V. A Narrative Review of COVID-19 Vaccines. Vaccines 2022, 10, 62. https://doi.org/10.3390/vaccines10010062
Eroglu B, Nuwarda RF, Ramzan I, Kayser V. A Narrative Review of COVID-19 Vaccines. Vaccines. 2022; 10(1):62. https://doi.org/10.3390/vaccines10010062
Chicago/Turabian StyleEroglu, Barbaros, Rina Fajri Nuwarda, Iqbal Ramzan, and Veysel Kayser. 2022. "A Narrative Review of COVID-19 Vaccines" Vaccines 10, no. 1: 62. https://doi.org/10.3390/vaccines10010062
APA StyleEroglu, B., Nuwarda, R. F., Ramzan, I., & Kayser, V. (2022). A Narrative Review of COVID-19 Vaccines. Vaccines, 10(1), 62. https://doi.org/10.3390/vaccines10010062






