Role of Clostridium perfringens Necrotic Enteritis B-like Toxin in Disease Pathogenesis
Abstract
1. Introduction
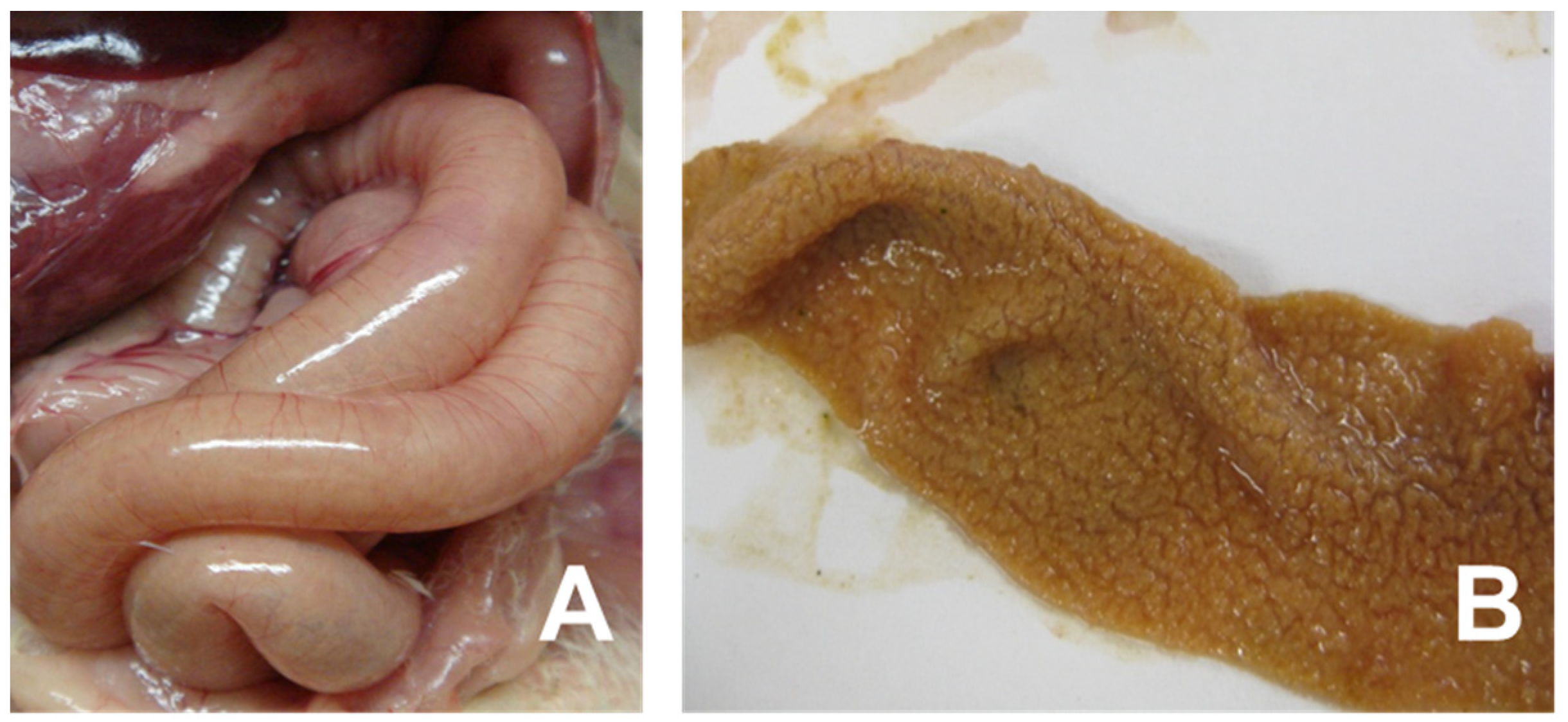
2. Necrotic Enteritis
3. Clostridium perfringens Toxinotypes
4. C. perfringens Toxins
5. C. perfringens NetB Toxin and Its Role in NE
6. Incidence of NetB-Positive C. perfringens Isolated from Broilers
7. In Vitro and In Vivo NetB Production by NetB-Positive C. perfringens Isolates
8. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food security: The challenge of feeding 9 billion people. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-W.; Lillehoj, H.S. An update on direct-fed microbials in broiler chickens in post-antibiotic era. Anim. Prod. Sci. 2017, 57, 1575. [Google Scholar] [CrossRef]
- St-Pierre, N.; Cobanov, B.; Schnitkey, G. Economic losses from heat stress by US livestock industries. J. Dairy Sci. 2003, 86, E52–E77. [Google Scholar] [CrossRef]
- Blake, D.P.; Knox, J.; Dehaeck, B.; Huntington, B.; Rathinam, T.; Ravipati, V.; Ayoade, S.; Gilbert, W.; Adebambo, A.O.; Jatau, I.D.; et al. Re-calculating the cost of coccidiosis in chickens. Vet. Res. 2020, 51, 115. [Google Scholar] [CrossRef]
- Gadde, U.; Kim, W.H.; Oh, S.T.; Lillehoj, H.S. Alternatives to antibiotics for maximizing growth performance and feed efficiency in poultry: A review. Anim. Heal. Res. Rev. 2017, 18, 26–45. [Google Scholar] [CrossRef]
- An, B.-K.; Choi, Y.-I.; Kang, C.-W.; Lee, K.-W. Effects of dietary Corynebacterium ammoniagenes-derived single cell protein on growth performance, blood and tibia bone characteristics, and meat quality of broiler chickens. J. Anim. Feed. Sci. 2018, 27, 140–147. [Google Scholar] [CrossRef]
- Grant, A.; Gay, C.; Lillehoj, H.S. Bacillus spp. as direct-fed microbial antibiotic alternatives to enhance growth, immunity, and gut health in poultry. Avian Pathol. 2018, 47, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Han, S.M.; Keum, M.C.; Lee, S.; An, B.K.; Lee, K.-W. Evaluation of bee venom as a novel feed additive in fast-growing broilers. Br. Poult. Sci. 2018, 59, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Ndazigaruye, G.; Kim, D.-H.; Kang, C.-W.; Kang, K.-R.; Joo, Y.-J.; Lee, S.-R.; Lee, K.-W. Effects of low-protein diets and exogenous protease on growth performance, carcass traits, intestinal morphology, cecal volatile fatty acids and serum parameters in broilers. Animals 2019, 9, 226. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Lillehoj, H.S.; Lee, Y.; Bravo, D.; Lillehoj, E.P. Dietary antibiotic growth promoters down-regulate intestinal inflammatory cytokine expression in chickens challenged with LPS or co-infected with Eimeria maxima and Clostridium perfringens. Front. Vet. Sci. 2019, 6, 420. [Google Scholar] [CrossRef]
- Hofacre, C.L.; Smith, J.A.; Mathis, G.F. An optimist’s view on limiting necrotic enteritis and maintaining broiler gut health and performance in today’s marketing, food safety, and regulatory climate. Poult. Sci. 2018, 97, 1929–1933. [Google Scholar] [CrossRef]
- Van Immerseel, F.; De Buck, J.; Pasmans, F.; Huyghebaert, G.; Haesebrouck, F.; Ducatelle, R. Clostridium perfringens in poultry: An emerging threat for animal and public health. Avian Pathol. 2004, 33, 537–549. [Google Scholar] [CrossRef]
- Tsiouris, V. Poultry management: A useful tool for the control of necrotic enteritis in poultry. Avian Pathol. 2016, 45, 323–325. [Google Scholar] [CrossRef]
- Broom, L. Necrotic enteritis; current knowledge and diet-related mitigation. World’s Poult. Sci. J. 2017, 73, 281–292. [Google Scholar] [CrossRef]
- Cooper, K.; Songer, J.G.; Uzal, F.A. Diagnosing clostridial enteric disease in poultry. J. Vet. Diagn. Investig. 2013, 25, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Caly, D.L.; D’lnca, R.; Auclair, E.; Drider, D. Alternatives to antibiotics to prevent necrotic enteritis in broiler chickens: A microbiologist’s perspective. Front. Microbiol. 2015, 6, 1336. [Google Scholar] [CrossRef]
- Kaldhusdal, M.; Løvland, A. The economical impact of Clostridium perfringens is greater than anticipated. World Poult. 2000, 16, 50–51. [Google Scholar]
- Løvland, A.; Kaldhusdal, K. Liver lesions seen at slaughter as an indicator of necrotic enteritis in broiler flocks. FEMS Microbiol. Immunol. 1999, 24, 345–351. [Google Scholar] [CrossRef]
- Wade, B.; Keyburn, A. The true cost of necrotic enteritis. World Poult. 2015, 31, 16–17. [Google Scholar]
- Li, J.; Adams, V.; Bannam, T.L.; Miyamoto, K.; Garcia, J.P.; Uzal, F.A.; Rood, J.I.; McClane, B.A. Toxin plasmids of Clostridium perfringens. MMBR 2013, 77, 208–233. [Google Scholar] [CrossRef]
- Rood, J.I.; Adams, V.; Lacey, J.; Lyras, D.; McClane, B.A.; Melville, S.B.; Moore, R.J.; Popoff, M.R.; Sarker, M.R.; Songer, J.G.; et al. Expansion of the Clostridium perfringens toxin-based typing scheme. Anaerobe 2018, 53, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Kiu, R.; Hall, L.J. An update on the human and animal enteric pathogen Clostridium perfringens. Emerg. Microbes Infect. 2018, 7, 1–15. [Google Scholar] [CrossRef]
- Ohtani, K.; Shimizu, T. Regulation of toxin production in Clostridium perfringens. Toxins 2016, 8, 207. [Google Scholar] [CrossRef]
- Mi, E.; Li, J.; McClane, B.A. NanR Regulates Sporulation and Enterotoxin Production by Clostridium perfringens Type F Strain F4969. Infect. Immun. 2018, 86, e00416-18. [Google Scholar] [CrossRef]
- Keyburn, A.L.; Bannam, T.L.; Moore, R.J.; Rood, J.I. NetB, a pore-forming toxin from necrotic enteritis strains of Clostridium perfringens. Toxins 2010, 2, 1913–1927. [Google Scholar] [CrossRef]
- Allaart, J.G.; de Bruijn, N.D.; van Asten, A.J.A.M.; Fabri, T.H.F.; Gröne, A. NetB-producing and beta2-producing Clostridium perfringens associated with subclincal necrotic enteritis in laying hens in the Netherlands. Avian Pathol. 2012, 41, 541–546. [Google Scholar] [CrossRef]
- Revitt-Mills, S.A.; Rood, J.I.; Adams, V. Clostridium perfringens extracellular toxins and enzymes: 20 and counting. Microbiol. Aust. 2015, 36, 114. [Google Scholar] [CrossRef]
- Oda, M.; Terao, Y.; Sakurai, J.; Nagahama, M. Membrane-Binding Mechanism of Clostridium perfringens Alpha-Toxin. Toxins 2015, 7, 5268–5275. [Google Scholar] [CrossRef]
- Navarro, M.A.; McClane, B.A.; Uzal, F.A. Mechanisms of Action and Cell Death Associated with Clostridium perfringens Toxins. Toxins 2018, 10, 212. [Google Scholar] [CrossRef] [PubMed]
- Popoff, M.R. Clostridium pore-forming toxins: Powerful virulence factors. Anaerobe 2014, 30, 220–238. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, O.; Yoshihara, K.; Katayama, S.; Minami, J.; Okabe, A. Purification and characterization of Clostridium perfringens 120-kilodalton collagenase and nucleotide sequence of the corresponding gene. J. Bacteriol. 1994, 176, 149–156. [Google Scholar] [CrossRef]
- Wang, Y.-H. Sialidases from Clostridium perfringens and their inhibitors. Front. Cell. Infect. Microbiol. 2020, 9, 462. [Google Scholar] [CrossRef]
- Li, C.; Yan, X.; Lillehoj, H.S. Complete genome sequences of Clostridium perfringens Del1 strain isolated from chickens affected by necrotic enteritis. Gut Pathog 2017, 9, 69. [Google Scholar] [CrossRef]
- Newstead, S.; Potter, J.A.; Wilson, J.C.; Xu, G.; Chien, C.-H.; Watts, A.; Withers, S.G.; Taylor, G. The structure of Clostridium perfringens nani sialidase and its catalytic intermediates. J. Biol. Chem. 2008, 283, 9080–9088. [Google Scholar] [CrossRef]
- Shimizu, T.; Ohtani, K.; Hirakawa, H.; Ohshima, K.; Yamashita, A.; Shihba, T.; Ogasawara, N.; Hattori, M.; Kuhara, S.; Hayashi, H. Complete genome sequence of Clostridium perfringens, an anaerobic flesh-eater. Proc. Natl. Acad. Sci. USA 2002, 99, 996–1001. [Google Scholar] [CrossRef]
- Goossens, E.; Valgaeren, B.R.; Pardon, B.; Haesebrouck, F.; Ducatelle, R.; Deprez, P.R.; Van Immerseel, F. Rethinking the role of alpha toxin in Clostridium perfringens-associated enteric diseases: A review on bovine necro-haemorrhagic enteritis. Vet. Res. 2017, 48, 9. [Google Scholar] [CrossRef] [PubMed]
- Wade, B.; Keyburn, A.; Seemann, T.; Rood, J.; Moore, R. Binding of Clostridium perfringens to collagen correlates with the ability to cause necrotic enteritis in chickens. Vet. Microbiol. 2015, 180, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Savva, C.G.; da Costa, S.P.F.; Bokori-Brown, M.; Naylor, C.E.; Cole, A.R.; Moss, D.S.; Titball, R.W.; Basak, A.K. Molecular architecture and functional analysis of NetB, a pore-forming toxin from Clostridium perfringens. J. Biol. Chem. 2013, 288, 3512–3522. [Google Scholar] [CrossRef] [PubMed]
- Keyburn, A.L.; Boyce, J.D.; Vaz, P.; Bannam, T.L.; Ford, M.E.; Parker, D.; Di Rubbo, A.; Rood, J.I.; Moore, R.J. NetB, a new toxin that is associated with avian necrotic enteritis caused by Clostridium perfringens. PLOS Pathog. 2008, 4, e26. [Google Scholar] [CrossRef] [PubMed]
- Schotte, U.; Truyen, U.; Neubauer, H. Significance of β2-toxigenic Clostridium perfringens infections in animals and their predisposing factors—A review. J. Vet. Med. B 2004, 51, 423–426. [Google Scholar] [CrossRef]
- Chen, J.; McClane, B.A. Characterization of Clostridium perfringens TpeL Toxin Gene Carriage, Production, Cytotoxic Contributions, and Trypsin Sensitivity. Infect. Immun. 2015, 83, 2369–2381. [Google Scholar] [CrossRef] [PubMed]
- Coursodon, C.F.; Glock, R.D.; Moore, K.L.; Cooper, K.K.; Songer, J.G. TpeL-producing strains of Clostridium perfringens type A are highly virulent for broiler chicks. Anaerobe 2012, 18, 117–121. [Google Scholar] [CrossRef]
- Guttenberg, G.; Hornei, S.; Jank, T.; Schwan, C.; Lü, W.; Einsle, O.; Papatheodorou, P.; Aktories, K. Molecular char-acteristics of Clostridium perfringens TpeL toxin and consequences of mono-O-GlcNAcylation of Ras in living cells. J. Biol. Chem. 2012, 287, 24929–24940. [Google Scholar] [CrossRef]
- Crespo, R.; Fisher, D.J.; Shivaprasad, H.L.; Fernández-Miyakawa, M.E.; Uzal, F.A. Toxinotypes of Clostridium perfringens isolated from sick and healthy Avian Species. J. Vet. Diagn. Investig. 2007, 19, 329–333. [Google Scholar] [CrossRef]
- França, M.; Barrios, M.A.; Stabler, L.; Zavala, G.; Shivaprasad, H.L.; Lee, M.D.; Villegas, A.M.; Uzal, F.A. Association of Beta2-PositiveClostridium perfringensType A With Focal Duodenal Necrosis in Egg-Laying Chickens in the United States. Avian Dis. 2016, 60, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Bueschel, D.M.; Jost, B.H.; Billington, S.J.; Trinh, H.T.; Songer, J.G. Prevalence of cpb2, encoding beta2 toxin, in Clostridium perfringens field isolates: Correlation of genotype with phenotype. Vet. Microbiol. 2003, 94, 121–129. [Google Scholar] [CrossRef]
- Gu, C.; Lillehoj, H.S.; Sun, Z.; Lee, Y.; Zhao, H.; Xianyu, Z.; Yan, X.; Wang, Y.; Lin, S.; Liu, L.; et al. Characterization of virulent netB+/tpeL+ Clostridium perfringens strains from necrotic enteritis-afflicted broiler chicken farms. Avian Dis. 2019, 63, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, O.; Okabe, A. Clostridial hydrolytic enzymes degrading extracellular components. Toxicon 2001, 39, 1769–1780. [Google Scholar] [CrossRef]
- Li, J.; Sayeed, S.; Robertson, S.; Chen, J.; McClane, B.A. Sialidases affect the host cell adherence and epsilon tox-in-induced cytotoxicity of Clostridium perfringens type D strain CN3718. PLoS Pathog. 2011, 7, e1002429. [Google Scholar] [CrossRef] [PubMed]
- Olkowski, A.; Wojnarowicz, C.; Chirino-Trejo, M.; Laarveld, B.; Sawicki, G. Sub-clinical necrotic enteritis in broiler chickens: Novel etiological consideration based on ultra-structural and molecular changes in the intestinal tissue. Res. Vet. Sci. 2008, 85, 543–553. [Google Scholar] [CrossRef]
- Wade, B.; Keyburn, A.L.; Haring, V.; Ford, M.; Rood, J.I.; Moore, R.J. The adherent abilities of Clostridium perfringens strains are critical for the pathogenesis of avian necrotic enteritis. Vet. Microbiol. 2016, 197, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Parreira, V.R.; Costa, M.; Eikmeyer, F.; Blom, J.; Prescott, J.F. Sequence of two plasmids from clostridium perfringens chicken necrotic enteritis isolates and comparison with c. perfringens conjugative plasmids. PLoS ONE 2012, 7, e49753. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.-X.; Porter, C.J.; Hardy, S.P.; Steer, D.; Smith, I.; Quinsey, N.S.; Hughes, V.; Cheung, J.K.; Keyburn, A.L.; Kaldhusdal, M.; et al. Structural and functional analysis of the pore-forming toxin NetB from Clostridium perfringens. mBio 2013, 4, e00019-13. [Google Scholar] [CrossRef]
- Yu, Q.; Lepp, D.; Gohari, I.M.; Wu, T.; Zhou, H.; Yin, X.; Yu, H.; Prescott, J.F.; Nie, S.-P.; Xie, M.-Y.; et al. The agr-like quorum sensing system is required for pathogenesis of necrotic enteritis caused by Clostridium perfringens in poultry. Infect. Immun. 2017, 85, e00975-e16. [Google Scholar] [CrossRef]
- Lepp, D.; Roxas, B.; Parreira, V.R.; Marri, P.R.; Rosey, E.L.; Gong, J.; Songer, J.G.; Vedantam, G.; Prescott, J.F. Identification of novel pathogenicity loci in Clostridium perfringens strains that cause avian necrotic enteritis. PLoS ONE 2010, 5, e10795. [Google Scholar] [CrossRef]
- Keyburn, A.L.; Sheedy, S.A.; Ford, M.E.; Williamson, M.M.; Awad, M.M.; Rood, J.I.; Moore, R.J. Alpha-Toxin of Clostridium perfringens is not an essential virulence factor in necrotic enteritis in chickens. Infect. Immun. 2006, 74, 6496–6500. [Google Scholar] [CrossRef]
- Coursodon, C.F.; Trinh, H.T.; Mallozzi, M.; Vedantam, G.; Glock, R.; Songer, J. Clostridium perfringens alpha toxin is produced in the intestines of broiler chicks inoculated with an alpha toxin mutant. Anaerobe 2010, 16, 614–617. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Lepp, D.; Pei, Y.; Liu, M.; Yin, X.; Ma, R.; Prescott, J.F.; Gong, J. Influence of pCP1NetB ancillary genes on the virulence of Clostridium perfringens poultry necrotic enteritis strain CP1. Gut Pathog. 2017, 9, 6. [Google Scholar] [CrossRef]
- Martin, T.G.; Smyth, J.A. Prevalence of netB among some clinical isolates of Clostridium perfringens from animals in the United States. Vet. Microbiol. 2009, 136, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Abildgaard, L.; Søndergaard, T.; Engberg, R.M.; Schramm, A.; Højberg, O. In vitro production of necrotic enteritis toxin B, NetB, by netB-positive and netB-negative Clostridium perfringens originating from healthy and diseased broiler chickens. Vet. Microbiol. 2010, 144, 231–235. [Google Scholar] [CrossRef]
- Park, J.Y.; Kim, S.; Oh, J.Y.; Kim, H.R.; Jang, I.; Lee, H.S.; Kwon, Y.K. Characterization of Clostridium perfringens isolates obtained from 2010 to 2012 from chickens with necrotic enteritis in Korea. Poult. Sci. 2015, 94, 1158–1164. [Google Scholar] [CrossRef]
- Gaucher, M.-L.; Perron, G.G.; Arsenault, J.; Letellier, A.; Boulianne, M.; Quessy, S. Recurring necrotic enteritis outbreaks in commercial broiler chicken flocks strongly influence toxin gene carriage and species richness in the resident Clostridium perfringens population. Front. Microbiol. 2017, 8, 881. [Google Scholar] [CrossRef]
- Yang, W.Y.; Chou, C.H.; Wang, C. Characterization of toxin genes and quantitative analysis of netB in necrotic en-teritis (NE)-producing and non-NE-producing Clostridium perfringens isolated from chickens. Anaerobe 2018, 54, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Mwangi, S.; Timmons, J.; Fitz-Coy, S.; Parveen, S. Characterization of Clostridium perfringens recovered from broiler chicken affected by necrotic enteritis. Poult. Sci. 2019, 98, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Profeta, F.; Di Francesco, C.E.; Di Provvido, A.; Scacchia, M.; Alessiani, A.; Di Giannatale, E.; Marruchella, G.; Orsini, M.; Toscani, T.; Marsilio, F. Prevalence of netB-positive Clostridium perfringens in Italian poultry flocks by environmental sampling. J. Vet. Diagn. 2020, 32, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Brady, J.; Hernandez-Doria, J.D.; Bennett, C.; Guenter, W.; House, J.D.; Rodriguez-Lecompte, J.C. Toxinotyping of necrotic enteritis-producing and commercial isolates of Clostridium perfringens from chickens fed organic diets. Avian Pathol. 2010, 39, 475–481. [Google Scholar] [CrossRef]
- Hibberd, M.C.; Neumann, A.P.; Rehberger, T.G.; Siragusa, G.R. Multilocus sequence typing subtypes of poultry Clostridium perfringens isolates demonstrate disease niche partitioning. J. Clin. Microbiol. 2011, 49, 1556–1567. [Google Scholar] [CrossRef]
- Li, C.; Lillehoj, H.S.; Gadde, U.D.; Ritter, D.; Oh, S. Characterization of Clostridium perfringens strains isolated from healthy and necrotic enteritis-afflicted broiler chickens. Avian Dis. 2017, 61, 178–185. [Google Scholar] [CrossRef]
- Cooper, K.K.; Songer, J.G. Virulence of Clostridium perfringens in an experimental model of poultry necrotic enteritis. Vet. Microbiol. 2010, 142, 323–328. [Google Scholar] [CrossRef]
- Timbermont, L.; Haesebrouck, F.; Ducatelle, R.; Van Immerseel, F. Necrotic enteritis in broilers: An updated review on the pathogenesis. Avian Pathol. 2011, 40, 341–347. [Google Scholar] [CrossRef]
- Prescott, J.F.; Parreira, V.R.; Gohari, I.M.; Lepp, D.; Gong, J. The pathogenesis of necrotic enteritis in chickens: What we know and what we need to know: A review. Avian Pathol. 2016, 45, 288–294. [Google Scholar] [CrossRef]
- Rood, J.I.; Keyburn, A.L.; Moore, R.J. NetB and necrotic enteritis: The hole movable story. Avian Pathol. 2016, 45, 295–301. [Google Scholar] [CrossRef]
- Barbara, A.J.; Trinh, H.T.; Glock, R.D.; Songer, J.G. Necrotic enteritis-producing strains of Clostridium perfringens displace non-necrotic enteritis strains from the gut of chicks. Vet. Microbiol. 2008, 126, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Moran, E.T. Intestinal events and nutritional dynamics predispose Clostridium perfringens virulence in broilers. Poult. Sci. 2014, 93, 3028–3036. [Google Scholar] [CrossRef] [PubMed]
- Kiu, R.; Brown, J.; Bedwell, H.; Leclaire, C.; Caim, S.; Pickard, D.; Dougan, G.; Dixon, R.A.; Hall, L.J. Genomic analysis on broiler-associated Clostridium perfringens strains and exploratory caecal microbiome investigation reveals key factors linked to poultry necrotic enteritis. Anim. Microbiome 2019, 1, 12. [Google Scholar] [CrossRef]
- Cooper, K.K.; Theoret, J.R.; Stewart, B.A.; Trinh, H.T.; Glock, R.D.; Songer, J.G. Virulence for chickens of Clostridium perfringens isolated from poultry and other sources. Anaerobe 2010, 16, 289–292. [Google Scholar] [CrossRef]
- Miller, R.W.; Skinner, E.J.; Sulakvelidze, A.; Mathis, G.F.; Hofacre, C.L. Bacteriophage Therapy for Control of Necrotic Enteritis of Broiler Chickens Experimentally Infected with Clostridium perfringens. Avian Dis. 2010, 54, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Richardson, K.; Hofacre, C.; Mathis, G.; Lumpkins, B.; Phillips, R. Impact of Controlling Bacteria in Feed on Broiler Performance During a Clostridial Challenge. Avian Dis. 2017, 61, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Naseri, K.G.; Kheravii, S.; Keerqin, C.; Morgan, N.; Swick, R.; Choct, M.; Wu, S.-B. Two different Clostridium perfringens strains produce different levels of necrotic enteritis in broiler chickens. Poult. Sci. 2019, 98, 6422–6432. [Google Scholar] [CrossRef] [PubMed]
- Lacey, J.A.; Allnutt, T.R.; Vezina, B.; Van, T.T.H.; Stent, T.; Han, X.; Rood, J.I.; Wade, B.; Keyburn, A.L.; Seemann, T.; et al. Whole genome analysis reveals the diversity and evolutionary relationships between necrotic enteritis-causing strains of Clostridium perfringens. BMC Genom. 2018, 19, 379. [Google Scholar] [CrossRef]
- Yang, W.Y.; Lee, Y.J.; Lu, H.Y.; Branton, S.L.; Chou, C.H.; Wang, C. The netB-positive Clostridium perfringens in the experimental induction of necrotic enteritis with or without predisposing factors. Poult. Sci. 2019, 98, 5297–5306. [Google Scholar] [CrossRef]
- Gholamiandekhordi, A.R.; Ducatelle, R.; Heyndrickx, M.; Haesebrouck, F.; Van Immerseel, F. Molecular and phe-notypical characterization of Clostridium perfringens isolates from poultry flocks with different disease status. Vet. Microbiol. 2006, 113, 143–152. [Google Scholar] [CrossRef]
- Eeckhaut, V.; Wang, J.; Van Parys, A.; Haesebrouck, F.; Joossens, M.; Falony, G.; Raes, J.; Ducatelle, R.; Van Immerseel, F. The probiotic butyricicoccus pullicaecorum reduces feed conversion and protects from potentially harmful intestinal microorganisms and necrotic enteritis in broilers. Front. Microbiol. 2016, 7, 1416. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Ranjitkar, S.; Sharma, N.K.; Engberg, R.M. Influence of feeding crimped kernel maize silage on the course of subclinical necrotic enteritis in a broiler disease model. Anim. Nutr. 2017, 3, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Keyburn, A.L.; Yan, X.X.; Bannam, T.L.; Van Immerseel, F.; Rood, J.I.; Moore, R.J. Association between avian necrotic enteritis and Clostridium perfringens strains expressing NetB toxin. Vet. Res. 2010, 41, 21. [Google Scholar] [CrossRef] [PubMed]
- Llanco, L.A.; Viviane, N.; Ferreira, A.J.P.; Avila-Campos, M.J. Toxinotyping and antimicrobial susceptibility of Clostridium perfringens isolated from broiler chickens with necrotic enteritis. Int. J. Microbiol. Res. 2012, 4, 290–294. [Google Scholar]
- Chalmers, G.; Bruce, H.L.; Hunter, D.B.; Parreira, V.R.; Kulkarni, R.R.; Jiang, Y.-F.; Prescott, J.F.; Boerlin, P. Multilocus sequence typing analysis of Clostridium perfringens isolates from necrotic enteritis outbreaks in broiler chicken populations. J. Clin. Microbiol. 2008, 46, 3957–3964. [Google Scholar] [CrossRef]
- Nowell, V.J.; Poppe, C.; Parreira, V.R.; Jiang, Y.-F.; Reid-Smith, R.; Prescott, J.F. Clostridium perfringens in retail chicken. Anaerobe 2010, 16, 314–315. [Google Scholar] [CrossRef]
- Ronco, T.; Stegger, M.; Ng, K.L.; Lilje, B.; Lyhs, U.; Andersen, P.S.; Pedersen, K. Genome analysis of Clostridium perfringens isolates from healthy and necrotic enteritis infected chickens and turkeys. BMC Res. Notes 2017, 10, 270. [Google Scholar] [CrossRef]
- Ezatkhah, M.; Alimolaei, M.; Shahdadnejad, N. The Prevalence of netB Gene in Isolated Clostridium perfringens From Organic Broiler Farms Suspected to Necrotic Enteritis. Int. J. Enteric Pathog. 2016, 4, 3–35667. [Google Scholar] [CrossRef]
- Drigo, I.; Agnoletti, F.; Bacchin, C.; Guolo, A.; Cocchi, M.; Bonci, M.; Bano, L. Diffusion of Clostridium perfringens NetB positive strains in healthy and diseased chickens. Ital. J. Anim. Sci. 2009, 8, 761–764. [Google Scholar] [CrossRef][Green Version]
- Hu, W.-S.; Kim, H.; Koo, O.K. Molecular genotyping, biofilm formation and antibiotic resistance of enterotoxigenic Clostridium perfringens isolated from meat supplied to school cafeterias in South Korea. Anaerobe 2018, 52, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.; Aspán, A.; Kaldhusdal, M.; Engström, B.E. Genetic diversity and prevalence of netB in Clostridium perfringens isolated from a broiler flock affected by mild necrotic enteritis. Vet. Microbiol. 2010, 144, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Smyth, J.A.; Martin, T.G. Disease producing capability of netB positive isolates of C. perfringens recovered from normal chickens and a cow, and netB positive and negative isolates from chickens with necrotic enteritis. Vet. Microbiol. 2010, 146, 76–84. [Google Scholar] [CrossRef]
- Lee, K.-W.; Kim, W.H.; Li, C.; Lillehoj, H.S. Detection of necrotic enteritis B–like toxin secreted by Clostridium perfringens using capture enzyme-linked immunosorbent assay. Avian Dis. 2020, 64, 490–495. [Google Scholar] [CrossRef]
- Lee, K.W.; Lillehoj, H.S.; Kim, W.H.; Park, I.; Li, C.; Lu, M.; Hofacre, C.L. Research note: First report on the detection of necrotic enteritis (NE) B-like toxin in biological samples from NE-afflicted chickens using capture enzyme-linked immunosorbent assay. Poult. Sci. 2021, 100, 101190. [Google Scholar] [CrossRef]
- Wickramasuriya, S.S.; Park, I.; Lee, Y.; Kim, W.H.; Przybyszewski, C.; Gay, C.G.; van Oosterwijk, J.G.; Lillehoj, H.S. Oral delivery of Bacillus subtilis expressing chicken NK-2 peptide protects against Eimeria acervuline infection in broiler chickens. Front. Vet. Sci. 2021, 8, 684818. [Google Scholar] [CrossRef]
| Type | Toxin A Produced (Structural Gene) | |||||
|---|---|---|---|---|---|---|
| α-Toxin (cpa) | β-Toxin (cpb) | ε-Toxin (etx) | ι-Toxin (iap) | CPE (cpe) | NetB (netB) | |
| A | + | − | − | − | − | − |
| B | + | + | + | − | − | − |
| C | + | + | − | − | ± | − |
| D | + | − | + | − | ± | − |
| E | + | − | − | + | ± | − |
| F | + | − | − | − | + | − |
| G | + | − | − | − | − | + |
| Full Name | Gene | Other Name | Gene Location A | Size (kDa) | Activity | References |
|---|---|---|---|---|---|---|
| Alpha-toxin | cpa, plc | phospholipase C | C | 43 | To hydrolyze cell membrane phospholipids | [22] |
| Perfringolysin O | pfoA | θ-toxin (pore-forming toxin) | C | 54 | Pore formation via binding to cholesterol-comprising cell membrane | [22,30] |
| Collagenase | colA | κ-toxin | C | 120 | To degrade collagen that is main component of connective tissues of the host cells | [22,31] |
| Sialidase | nanI | secreted major neuramidases | C | 77 | Involved in removal of sialic acids from a variety of glycoconjugates on cell membranes | [32,33,34] |
| Sialidase | nanH | non-secreted neuramidases | C | 43 | ||
| Sialidase | nanJ | secreted neuramidases | C | 129 | ||
| Hyaluronidase | nagH | µ-toxin | C | ≈182 | To degrade hyaluronan coating cells allowing direct contact between pathogen and host cells, or to degrade hyaluronan leading to viscosity reduction facilitating increased permeability of the connective tissues | [35,36] |
| Hyaluronidase | nagI | µ-toxin | C | ≈146 | ||
| Hyaluronidase | nagJ | µ-toxin | C | ≈128 | ||
| Hyaluronidase | nagK | µ-toxin | C | ≈131 | ||
| Hyaluronidase | nagL | µ-toxin | C | ≈127 | ||
| Collagen adhesion protein | cnaA | cell surface protein | C | 78 | Binding of the Clostridium perfringens to collagen types IV, V and gelatin | [37] |
| NE B-like toxin | netB | pore-forming toxin | P | 33 | To form heptameric, hydrophilic pores with a central diameter of approximately 26 Å | [38,39] |
| Beta2 toxin | cpb2 | pore-forming toxin | P | 28 | Pore forming leading to cell disruption | [40] |
| Toxin C. perfringens large cytotoxin | tpeL | large clostridial toxin | P | ≈205 | Ras-specific glucosyltransferase activity inactivating the Ras signaling pathway leading to apoptosis | [41,42,43] |
| Strain | Country | Status | Virulent Genes | References | ||||
|---|---|---|---|---|---|---|---|---|
| Plc | NetB | CnaA | TpeL | Cpb2 | ||||
| Del1 | USA | NE A | + | + | + | − | + | [47,68] |
| TpeL17 | USA | NE | + | + | na B | + | + | [47] |
| N11 | USA | Health | + | − | − | − | + | [68,75] |
| CP15 | USA | NE | + | − | − | − | − | [68,75] |
| JGS4143 | USA | NE | + | + | na | − | + | [41,76] |
| CP-6 | USA | NE | + | + | na | na | na | [77,78] |
| EHE-NE18 | Australia | NE | + | + | + | − | + | [37,39,75,79,80] |
| WER-NE36 | Australia | NE | + | + | − | − | + | [37,75,79,80] |
| CP1 | Canada | NE | + | + | na | na | + | [52,81] |
| Strain 56 | Belgium | NE | + | + | + | na | − | [82,83] |
| S48 | Denmark | NE | + | + | na | na | na | [60,84] |
| Country | Study Year A | Detection Method | NE Chicken, n/Total | Healthy Chicken, n/Total | Ref. | ||
|---|---|---|---|---|---|---|---|
| NetB Positive | % | NetB Positive | % | ||||
| Australia | 2010 | PCR | 14/18 | 77.8 | - | - | [25] |
| Australia/Canada/Belgium/Denmark | 2010 | PCR | 31/44 | 70.5 | 2/55 | 3.6 | [85] |
| Brazil | 2012 | PCR | 0/22 | 0.0 | - | - | [86] |
| Canada | 2005–2007 | PCR | 39/41 | 95.1 | 7/20 | 35.0 | [87] |
| Canada | 2011–2012 | PCR | 9/45 | 20.0 | 12/18 | 66.7 | [62] |
| Canada | 2011–2012 | PCR | 41/41 | 100.0 | 26/30 | 86.7 | [62] |
| Canada | 2010 | PCR | 6/6 | 100.0 | 4/5 | 80.0 | [66] |
| Canada B | 2010 | PCR | - | - | 39/183 | 21.3 | [88] |
| Denmark | 1997–2002 | PCR | 13/25 | 52.0 | 14/23 | 60.9 | [60] |
| Denmark/ Finland | 1997–2001 | PCR | 12/22 | 54.5 | 0/8 | 0.0 | [89] |
| Iran | 2016 | PCR | 8/45 | 17.8 | - | - | [90] |
| Italy C | 2015–2017 | qPCR | - | - | 31/151 | 20.5 | [65] |
| Italy | 2009 | PCR | 16/30 | 53.3 | 4/22 | 18.2 | [91] |
| Korea | 2010–2012 | PCR | 8/17 | 47.1 | 2/50 | 4.0 | [61] |
| Korea B | 2018 | PCR | - | - | 4/9 | 44.4 | [92] |
| Netherlands | 2012 | PCR | 43/45 | 95.6 | - | - | [26] |
| Sweden | 2004 | PCR | 31/34 | 91.2 | - D | 25.0 | [93] |
| Sweden | 2004 | PCR | 16/23 | 69.6 | - | - | [93] |
| Sweden | 2004 | PCR | 0/11 | 0.0 | - | - | [93] |
| USA | 2004–2009 | PCR | 17/20 | 85.0 | 10/54 | 18.5 | [67] |
| USA | 2009 | PCR | 7/12 | 58.3 | 7/80 | 8.8 | [59] |
| USA | 2018 | qPCR | 11/15 | 73.3 | 9/15 | 60.0 | [63] |
| USA | 2016 | PCR | 119/145 | 82.1 | 59/85 | 69.4 | [64] |
| USA | 2003–2004 | PCR | 19/19 | 100.0 | - | - | [47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, K.-W.; Lillehoj, H.S. Role of Clostridium perfringens Necrotic Enteritis B-like Toxin in Disease Pathogenesis. Vaccines 2022, 10, 61. https://doi.org/10.3390/vaccines10010061
Lee K-W, Lillehoj HS. Role of Clostridium perfringens Necrotic Enteritis B-like Toxin in Disease Pathogenesis. Vaccines. 2022; 10(1):61. https://doi.org/10.3390/vaccines10010061
Chicago/Turabian StyleLee, Kyung-Woo, and Hyun S. Lillehoj. 2022. "Role of Clostridium perfringens Necrotic Enteritis B-like Toxin in Disease Pathogenesis" Vaccines 10, no. 1: 61. https://doi.org/10.3390/vaccines10010061
APA StyleLee, K.-W., & Lillehoj, H. S. (2022). Role of Clostridium perfringens Necrotic Enteritis B-like Toxin in Disease Pathogenesis. Vaccines, 10(1), 61. https://doi.org/10.3390/vaccines10010061







