After 100 Years of BCG Immunization against Tuberculosis, What Is New and Still Outstanding for This Vaccine?
Abstract
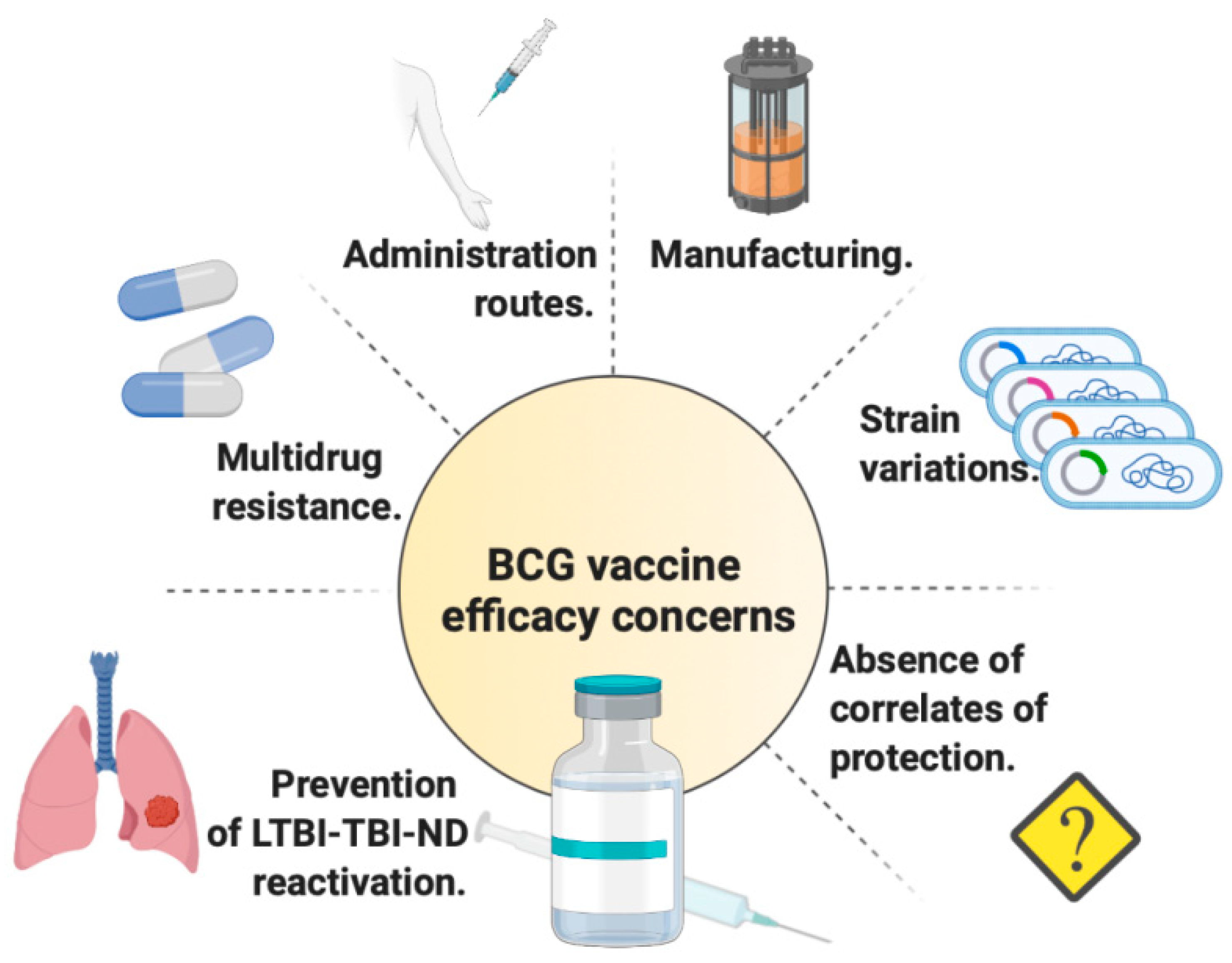
:1. Introduction
2. Benefits and Drawbacks of Current BCG
3. Latest Findings Regarding BCG Efficacy in Clinical Trials and in Advanced Preclinical Models
4. Novel Live, Attenuated, BCG-Based Vaccine Candidates: What Has Been Shown and What Is Still Missing?
5. The Need of Correlates of Protection
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaufmann, S.H.E. Vaccine development against tuberculosis over the last 140 years: Failure as part of success. Front. Microbiol. 2021, 12, 750124. [Google Scholar] [CrossRef]
- Garcia, J.I.; Allue-Guardia, A.; Tampi, R.P.; Restrepo, B.I.; Torrelles, J.B. New developments and insights in the improvement of mycobacterium tuberculosis vaccines and diagnostics within the end tb strategy. Curr. Epidemiol. Rep. 2021, 8, 1–13. [Google Scholar] [CrossRef]
- Ahmed, A.; Rakshit, S.; Adiga, V.; Dias, M.; Dwarkanath, P.; D’Souza, G.; Vyakarnam, A. A century of bcg: Impact on tuberculosis control and beyond. Immunol. Rev. 2021, 301, 98–121. [Google Scholar] [CrossRef]
- Lange, C.; Aaby, P.; Behr, M.A.; Donald, P.R.; Kaufmann, S.H.E.; Netea, M.G.; Mandalakas, A.M. 100 years of mycobacterium bovis bacille calmette-guerin. Lancet Infect. Dis. 2021, 22, 2–12. [Google Scholar] [CrossRef]
- Ferluga, J.; Yasmin, H.; Bhakta, S.; Kishore, U. Vaccination strategies against mycobacterium tuberculosis: Bcg and beyond. Adv. Exp. Med. Biol. 2021, 1313, 217–240. [Google Scholar] [CrossRef] [PubMed]
- Mouhoub, E.; Domenech, P.; Ndao, M.; Reed, M.B. The diverse applications of recombinant bcg-based vaccines to target infectious diseases other than tuberculosis: An overview. Front. Microbiol. 2021, 12, 757858. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, N.; Julian, E.; Torrelles, J.B.; Svatek, R.S. Effects of mycobacterium bovis calmette et guerin (bcg) in oncotherapy: Bladder cancer and beyond. Vaccine 2021, 39, 7332–7340. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Netea, M.G.; Bishai, W.R. Bcg turns 100: Its nontraditional uses against viruses, cancer, and immunologic diseases. J. Clin. Investig. 2021, 131, e148291. [Google Scholar] [CrossRef]
- World Health Organization. Global Tuberculosis Report 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Trauer, J.M.; Kawai, A.; Coussens, A.K.; Datta, M.; Williams, B.M.; McBryde, E.S.; Ragonnet, R. Timing of mycobacterium tuberculosis exposure explains variation in bcg effectiveness: A systematic review and meta-analysis. Thorax 2021, 76, 1131–1141. [Google Scholar] [CrossRef]
- Cernuschi, T.; Malvolti, S.; Nickels, E.; Friede, M. Bacillus calmette-guerin (bcg) vaccine: A global assessment of demand and supply balance. Vaccine 2018, 36, 498–506. [Google Scholar] [CrossRef]
- Antas, P.R.Z.; Flores-Valdez, M.; Shann, F. An opportunity to compare the effects of bcg-moreau and bcg-russia in brazil. Int. J. Tuberc. Lung Dis. 2018, 22, 1108–1109. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, A.S.M.; Albuquerque, L.H.P.; de Ponte, C.G.G.; de Almeida, M.R.; de Faria, S.E.R.; Ribeiro, M.D.S.; Pereira, E.; Antas, P.R.Z. Time to face the proofs: The bcg moreau vaccine promotes superior inflammatory cytokine profile in vitro when compared with russia, pasteur, and danish strains. Hum. Vaccin Immunother. 2021, 12, 1–7. [Google Scholar] [CrossRef]
- Angelidou, A.; Conti, M.-G.; Diray-Arce, J.; Benn, C.S.; Shann, F.; Netea, M.G.; Liu, M.; Potluri, L.-P.; Sanchez-Schmitz, G.; Husson, R.; et al. Licensed bacille calmette-guerin (bcg) formulations differ markedly in bacterial viability, rna content and innate immune activation. Vaccine 2020, 38, 2229–2240. [Google Scholar] [CrossRef]
- Pascoe, J.; Hendon-Dunn, C.L.; Birch, C.P.D.; Williams, G.A.; Chambers, M.A.; Bacon, J. Optimisation of mycobacterium bovis bcg fermentation and storage survival. Pharmaceutics 2020, 12, 900. [Google Scholar] [CrossRef]
- World Health Organization. BCG vaccine: Who position paper, february 2018—Recommendations. Vaccine 2018, 36, 3408–3410. [Google Scholar] [CrossRef]
- Behr, M.A.; Kaufmann, E.; Duffin, J.; Edelstein, P.H.; Ramakrishnan, L. Latent tuberculosis: Two centuries of confusion. Am. J. Respir. Crit. Care Med. 2021, 204, 142–148. [Google Scholar] [CrossRef]
- Emery, J.C.; Richards, A.S.; Dale, K.D.; McQuaid, C.F.; White, R.G.; Denholm, J.T.; Houben, R. Self-clearance of mycobacterium tuberculosis infection: Implications for lifetime risk and population at-risk of tuberculosis disease. Proc. Biol. Sci. 2021, 288, 20201635. [Google Scholar] [CrossRef]
- Harris, R.C.; Sumner, T.; Knight, G.M.; Evans, T.; Cardenas, V.; Chen, C.; White, R.G. Age-targeted tuberculosis vaccination in china and implications for vaccine development: A modelling study. Lancet Glob. Health 2019, 7, e209–e218. [Google Scholar] [CrossRef] [Green Version]
- Harris, R.C.; Sumner, T.; Knight, G.M.; Zhang, H.; White, R.G. Potential impact of tuberculosis vaccines in china, south africa, and india. Sci. Transl. Med. 2020, 12, 1–12. [Google Scholar] [CrossRef]
- Foreman, T.W.; Mehra, S.; LoBato, D.N.; Malek, A.; Alvarez, X.; Golden, N.A.; Bucsan, A.N.; Didier, P.J.; Doyle-Meyers, L.A.; Russell-Lodrigue, K.E.; et al. Cd4+ t-cell-independent mechanisms suppress reactivation of latent tuberculosis in a macaque model of hiv coinfection. Proc. Natl. Acad. Sci. USA 2016, 113, E5636–E5644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuroda, M.J.; Sugimoto, C.; Cai, Y.; Merino, K.M.; Mehra, S.; Arainga, M.; Roy, C.J.; Midkiff, C.C.; Alvarez, X.; Didier, E.S.; et al. High turnover of tissue macrophages contributes to tuberculosis reactivation in simian immunodeficiency virus-infected rhesus macaques. J. Infect. Dis. 2018, 217, 1865–1874. [Google Scholar] [CrossRef] [PubMed]
- Diedrich, C.R.; Rutledge, T.; Maiello, P.; Baranowski, T.M.; White, A.G.; Borish, H.J.; Karell, P.; Hopkins, F.; Brown, J.; Fortune, S.M.; et al. Siv and mycobacterium tuberculosis synergy within the granuloma accelerates the reactivation pattern of latent tuberculosis. PLoS Pathog. 2020, 16, e1008413. [Google Scholar] [CrossRef] [PubMed]
- White, A.D.; Sibley, L.; Gullick, J.; Sarfas, C.; Clark, S.; Fagrouch, Z.; Verschoor, E.; Salguero, F.J.; Dennis, M.; Sharpe, S. Tb and siv coinfection; a model for evaluating vaccine strategies against tb reactivation in asian origin cynomolgus macaques: A pilot study using bcg vaccination. Vaccines 2021, 9, 945. [Google Scholar] [CrossRef]
- Esaulova, E.; Das, S.; Singh, D.K.; Choreno-Parra, J.A.; Swain, A.; Arthur, L.; Rangel-Moreno, J.; Ahmed, M.; Singh, B.; Gupta, A.; et al. The immune landscape in tuberculosis reveals populations linked to disease and latency. Cell Host Microbe 2021, 29, 165–178.e8. [Google Scholar] [CrossRef] [PubMed]
- Gengenbacher, M.; Kaiser, P.; Schuerer, S.; Lazar, D.; Kaufmann, S.H. Post-exposure vaccination with the vaccine candidate bacillus calmette-guerin deltaurec::Hly induces superior protection in a mouse model of subclinical tuberculosis. Microbes Infect. 2016, 18, 364–368. [Google Scholar] [CrossRef]
- Pedroza-Roldan, C.; Guapillo, C.; Barrios-Payan, J.; Mata-Espinosa, D.; Aceves-Sánchez, M.D.J.; Marquina-Castillo, B.; Hernandez-Pando, R.; Flores-Valdez, M.A. The bcgdeltabcg1419c strain, which produces more pellicle in vitro, improves control of chronic tuberculosis in vivo. Vaccine 2016, 34, 4763–4770. [Google Scholar] [CrossRef]
- Nemes, E.; Geldenhuys, H.; Rozot, V.; Rutkowski, K.T.; Ratangee, F.; Bilek, N.; Mabwe, S.; Makhethe, L.; Erasmus, M.; Toefy, A.; et al. Prevention of m. Tuberculosis infection with h4:Ic31 vaccine or bcg revaccination. N. Engl. J. Med. 2018, 379, 138–149. [Google Scholar] [CrossRef]
- Lagranderie, M.; Ravisse, P.; Marchal, G.; Gheorghiu, M.; Balasubramanian, V.; Weigeshaus, E.H.; Smith, D.W. Bcg-induced protection in guinea pigs vaccinated and challenged via the respiratory route. Tuber. Lung Dis. 1993, 74, 38–46. [Google Scholar] [CrossRef]
- Aguilo, N.; Alvarez-Arguedas, S.; Uranga, S.; Marinova, D.; Monzon, M.; Badiola, J.; Martin, C. Pulmonary but not subcutaneous delivery of bcg vaccine confers protection to tuberculosis-susceptible mice by an interleukin 17-dependent mechanism. J. Infect. Dis. 2016, 213, 831–839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaushal, D.; Foreman, T.W.; Gautam, U.S.; Alvarez, X.; Adekambi, T.; Rangel-Moreno, J.; Golden, N.A.; Johnson, A.M.; Phillips, B.L.; Ahsan, M.H.; et al. Mucosal vaccination with attenuated mycobacterium tuberculosis induces strong central memory responses and protects against tuberculosis. Nat. Commun. 2015, 6, 8533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dijkman, K.; Sombroek, C.C.; Vervenne, R.A.W.; Hofman, S.O.; Boot, C.; Remarque, E.J.; Kocken, C.H.M.; Ottenhoff, T.H.M.; Kondova, I.; Khayum, M.A.; et al. Prevention of tuberculosis infection and disease by local bcg in repeatedly exposed rhesus macaques. Nat. Med. 2019, 25, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Sathkumara, H.D.; Muruganandah, V.; Cooper, M.M.; Field, M.A.; Alim, M.A.; Brosch, R.; Ketheesan, N.; Govan, B.; Rush, C.M.; Henning, L.; et al. Mucosal delivery of esx-1-expressing bcg strains provides superior immunity against tuberculosis in murine type 2 diabetes. Proc. Natl. Acad. Sci. USA 2020, 117, 20848–20859. [Google Scholar] [CrossRef] [PubMed]
- White, A.D.; Sarfas, C.; West, K.; Sibley, L.S.; Wareham, A.S.; Clark, S.; Dennis, M.J.; Williams, A.; Marsh, P.D.; Sharpe, S.A. Evaluation of the immunogenicity of mycobacterium bovis bcg delivered by aerosol to the lungs of macaques. Clin. Vaccine Immunol. 2015, 22, 992–1003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, A.D.; Sarfas, C.; Sibley, L.S.; Gullick, J.; Clark, S.; Rayner, E.; Gleeson, F.; Catala, M.; Nogueira, I.; Cardona, P.J.; et al. Protective efficacy of inhaled bcg vaccination against ultra-low dose aerosol m. Tuberculosis challenge in rhesus macaques. Pharmaceutics 2020, 12, 394. [Google Scholar] [CrossRef]
- Darrah, P.A.; Zeppa, J.J.; Maiello, P.; Hackney, J.A.; Wadsworth, M.H., II; Hughes, T.K.; Pokkali, S.; Swanson, P.A., II; Grant, N.L.; Rodgers, M.A.; et al. Prevention of tuberculosis in macaques after intravenous bcg immunization. Nature 2020, 577, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, S.; White, A.; Sarfas, C.; Sibley, L.; Gleeson, F.; McIntyre, A.; Basaraba, R.; Clark, S.; Hall, G.; Rayner, E.; et al. Alternative bcg delivery strategies improve protection against mycobacterium tuberculosis in non-human primates: Protection associated with mycobacterial antigen-specific cd4 effector memory t-cell populations. Tuberculosis 2016, 101, 174–190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irvine, E.B.; O’Neil, A.; Darrah, P.A.; Shin, S.; Choudhary, A.; Li, W.; Honnen, W.; Mehra, S.; Kaushal, D.; Gideon, H.P.; et al. Robust igm responses following intravenous vaccination with bacille calmette-guerin associate with prevention of mycobacterium tuberculosis infection in macaques. Nat. Immunol. 2021, 22, 1515–1523. [Google Scholar] [CrossRef]
- Langermans, J.A.; Andersen, P.; van Soolingen, D.; Vervenne, R.A.; Frost, P.A.; van der Laan, T.; van Pinxteren, L.A.; van den Hombergh, J.; Kroon, S.; Peekel, I.; et al. Divergent effect of bacillus calmette-guerin (bcg) vaccination on mycobacterium tuberculosis infection in highly related macaque species: Implications for primate models in tuberculosis vaccine research. Proc. Natl. Acad. Sci. USA 2001, 98, 11497–11502. [Google Scholar] [CrossRef] [Green Version]
- Grode, L.; Ganoza, C.A.; Brohm, C.; Weiner, J., III; Eisele, B.; Kaufmann, S.H. Safety and immunogenicity of the recombinant bcg vaccine vpm1002 in a phase 1 open-label randomized clinical trial. Vaccine 2013, 31, 1340–1348. [Google Scholar] [CrossRef]
- Loxton, A.G.; Knaul, J.K.; Grode, L.; Gutschmidt, A.; Meller, C.; Eisele, B.; Johnstone, H.; van der Spuy, G.; Maertzdorf, J.; Kaufmann, S.H.E.; et al. Safety and immunogenicity of the recombinant mycobacterium bovis bcg vaccine vpm1002 in hiv-unexposed newborn infants in south africa. Clin. Vaccine Immunol. 2017, 24, 00439-16. [Google Scholar] [CrossRef] [Green Version]
- Gengenbacher, M.; Nieuwenhuizen, N.; Vogelzang, A.; Liu, H.; Kaiser, P.; Schuerer, S.; Lazar, D.; Wagner, I.; Mollenkopf, H.J.; Kaufmann, S.H. Deletion of nuog from the vaccine candidate mycobacterium bovis bcg deltaurec::Hly improves protection against tuberculosis. Mbio 2016, 7, e00679-16. [Google Scholar] [CrossRef] [Green Version]
- Sander, P.; Clark, S.; Petrera, A.; Vilaplana, C.; Meuli, M.; Selchow, P.; Zelmer, A.; Mohanan, D.; Andreu, N.; Rayner, E.; et al. Deletion of zmp1 improves mycobacterium bovis bcg-mediated protection in a guinea pig model of tuberculosis. Vaccine 2015, 33, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Flores-Valdez, M.A.; Pedroza-Roldan, C.; Aceves-Sanchez, M.J.; Peterson, E.J.R.; Baliga, N.S.; Hernandez-Pando, R.; Troudt, J.; Creissen, E.; Izzo, L.; Bielefeldt-Ohmann, H.; et al. The bcgdeltabcg1419c vaccine candidate reduces lung pathology, il-6, tnf-alpha, and il-10 during chronic tb infection. Front. Microbiol. 2018, 9, 1281. [Google Scholar] [CrossRef] [Green Version]
- Aceves-Sanchez, M.J.; Flores-Valdez, M.A.; Pedroza-Roldan, C.; Creissen, E.; Izzo, L.; Silva-Angulo, F.; Dawson, C.; Izzo, A.; Bielefeldt-Ohmann, H.; Segura-Cerda, C.A.; et al. Vaccination with bcgdeltabcg1419c protects against pulmonary and extrapulmonary tb and is safer than bcg. Sci. Rep. 2021, 11, 12417. [Google Scholar] [CrossRef]
- Dannenberg, A.M., Jr.; Collins, F.M. Progressive pulmonary tuberculosis is not due to increasing numbers of viable bacilli in rabbits, mice and guinea pigs, but is due to a continuous host response to mycobacterial products. Tuberculosis 2001, 81, 229–242. [Google Scholar] [CrossRef]
- Horwitz, M.A.; Harth, G. A new vaccine against tuberculosis affords greater survival after challenge than the current vaccine in the guinea pig model of pulmonary tuberculosis. Infect. Immun. 2003, 71, 1672–1679. [Google Scholar] [CrossRef] [Green Version]
- Hoft, D.F.; Blazevic, A.; Abate, G.; Hanekom, W.A.; Kaplan, G.; Soler, J.H.; Weichold, F.; Geiter, L.; Sadoff, J.C.; Horwitz, M.A. A new recombinant bacille calmette-guerin vaccine safely induces significantly enhanced tuberculosis-specific immunity in human volunteers. J. Infect. Dis. 2008, 198, 1491–1501. [Google Scholar] [CrossRef]
- Pym, A.S.; Brodin, P.; Majlessi, L.; Brosch, R.; Demangel, C.; Williams, A.; Griffiths, K.E.; Marchal, G.; Leclerc, C.; Cole, S.T. Recombinant bcg exporting esat-6 confers enhanced protection against tuberculosis. Nat. Med. 2003, 9, 533–539. [Google Scholar] [CrossRef]
- Pym, A.S.; Brodin, P.; Brosch, R.; Huerre, M.; Cole, S.T. Loss of rd1 contributed to the attenuation of the live tuberculosis vaccines mycobacterium bovis bcg and mycobacterium microti. Mol. Microbiol. 2002, 46, 709–717. [Google Scholar] [CrossRef]
- Groschel, M.I.; Sayes, F.; Shin, S.J.; Frigui, W.; Pawlik, A.; Orgeur, M.; Canetti, R.; Honore, N.; Simeone, R.; van der Werf, T.S.; et al. Recombinant bcg expressing esx-1 of mycobacterium marinum combines low virulence with cytosolic immune signaling and improved tb protection. Cell Rep. 2017, 18, 2752–2765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heijmenberg, I.; Husain, A.; Sathkumara, H.D.; Muruganandah, V.; Seifert, J.; Miranda-Hernandez, S.; Kashyap, R.S.; Field, M.A.; Krishnamoorthy, G.; Kupz, A. Esx-5-targeted export of esat-6 in bcg combines enhanced immunogenicity & efficacy against murine tuberculosis with low virulence and reduced persistence. Vaccine 2021, 39, 7265–7276. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.K.; Tran, V.; Leung, A.; Ng, M.; Li, M.; Liu, J. Recombinant bcg overexpressing phop-phor confers enhanced protection against tuberculosis. Mol. Ther. 2018, 26, 2863–2874. [Google Scholar] [CrossRef] [Green Version]
- Penn-Nicholson, A.; Mbandi, S.K.; Thompson, E.; Mendelsohn, S.C.; Suliman, S.; Chegou, N.N.; Malherbe, S.T.; Darboe, F.; Erasmus, M.; Hanekom, W.A.; et al. Risk6, a 6-gene transcriptomic signature of tb disease risk, diagnosis and treatment response. Sci. Rep. 2020, 10, 8629. [Google Scholar] [CrossRef]
- Penn-Nicholson, A.; Hraha, T.; Thompson, E.G.; Sterling, D.; Mbandi, S.K.; Wall, K.M.; Fisher, M.; Suliman, S.; Shankar, S.; Hanekom, W.A.; et al. Discovery and validation of a prognostic proteomic signature for tuberculosis progression: A prospective cohort study. PLoS Med. 2019, 16, e1002781. [Google Scholar] [CrossRef] [PubMed]
- Duffy, F.J.; Weiner, J., III; Hansen, S.; Tabb, D.L.; Suliman, S.; Thompson, E.; Maertzdorf, J.; Shankar, S.; Tromp, G.; Parida, S.; et al. Immunometabolic signatures predict risk of progression to active tuberculosis and disease outcome. Front. Immunol. 2019, 10, 527. [Google Scholar] [CrossRef] [PubMed]
- Weiner, J., III; Maertzdorf, J.; Sutherland, J.S.; Duffy, F.J.; Thompson, E.; Suliman, S.; McEwen, G.; Thiel, B.; Parida, S.K.; Zyla, J.; et al. Metabolite changes in blood predict the onset of tuberculosis. Nat. Commun. 2018, 9, 5208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiner, J., III; Mohney, R.P.; Kaufmann, S.H.E. The potential of metabolic profiling for vaccine development. Semin. Immunol. 2018, 39, 44–51. [Google Scholar] [CrossRef]
- Rozot, V.; Nemes, E.; Geldenhuys, H.; Musvosvi, M.; Toefy, A.; Rantangee, F.; Makhethe, L.; Erasmus, M.; Bilek, N.; Mabwe, S.; et al. Multidimensional analyses reveal modulation of adaptive and innate immune subsets by tuberculosis vaccines. Commun. Biol. 2020, 3, 563. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-Valdez, M.A. After 100 Years of BCG Immunization against Tuberculosis, What Is New and Still Outstanding for This Vaccine? Vaccines 2022, 10, 57. https://doi.org/10.3390/vaccines10010057
Flores-Valdez MA. After 100 Years of BCG Immunization against Tuberculosis, What Is New and Still Outstanding for This Vaccine? Vaccines. 2022; 10(1):57. https://doi.org/10.3390/vaccines10010057
Chicago/Turabian StyleFlores-Valdez, Mario Alberto. 2022. "After 100 Years of BCG Immunization against Tuberculosis, What Is New and Still Outstanding for This Vaccine?" Vaccines 10, no. 1: 57. https://doi.org/10.3390/vaccines10010057
APA StyleFlores-Valdez, M. A. (2022). After 100 Years of BCG Immunization against Tuberculosis, What Is New and Still Outstanding for This Vaccine? Vaccines, 10(1), 57. https://doi.org/10.3390/vaccines10010057





