Total Oxidant and Antioxidant Capacity of Gingival Crevicular Fluid and Saliva in Patients with Periodontitis: Review and Clinical Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Sample Collection
2.3. Clinical Examination
2.4. Redox Assays
2.5. Statistical Analysis
3. Results
3.1. Total Oxidant Status (TOS)
3.2. Total Antioxidant Capacity (TAC)
3.3. Oxidative Stress Index (OSI)
3.4. FRAP
3.5. Correlations
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Konkel, J.E.; O’Boyle, C.; Krishnan, S. Distal Consequences of Oral Inflammation. Front. Immunol. 2019, 10, 1403. [Google Scholar] [CrossRef]
- Papapanou, P.N.; Sanz, M.; Buduneli, N.; Dietrich, T.; Feres, M.; Fine, D.H.; Flemmig, T.F.; Garcia, R.; Giannobile, W.V.; Graziani, F.; et al. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. Proc. J. Clin. Periodontol. 2018, 45, S162–S170. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Periodontol. 2018, 89, S159–S172. [Google Scholar] [CrossRef]
- Kimura, S.; Yonemura, T.; Kaya, H. Increased oxidative product formation by peripheral blood polymorphonuclear leukocytes in human periodontal diseases. J. Periodontal. Res. 1993, 28, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Andrukhov, O.; Rausch-Fan, X. Oxidative Stress and Antioxidant System in Periodontitis. Front. Physiol. 2017, 8, 910. [Google Scholar] [CrossRef]
- Knaś, M.; Maciejczyk, M.; Waszkiel, D.; Zalewska, A. Oxidative stress and salivary antioxidants. Dent. Med. Probl. 2013, 50, 461–466. [Google Scholar]
- Tóthová, L.; Celec, P. Oxidative Stress and Antioxidants in the Diagnosis and Therapy of Periodontitis. Front. Physiol. 2017, 8, 1055. [Google Scholar] [CrossRef] [PubMed]
- Giannopoulou, C.; Krause, K.-H.; Müller, F. The NADPH oxidase NOX2 plays a role in periodontal pathologies. Semin. Immunopathol. 2008, 30, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Isola, G.; Alibrandi, A.; Currò, M.; Matarese, M.; Ricca, S.; Matarese, G.; Ientile, R.; Kocher, T. Evaluation of salivary and serum ADMA levels in patients with periodontal and cardiovascular disease as subclinical marker of cardiovascular risk. J. Periodontol. 2020. [Epub ahead of print]. [Google Scholar] [CrossRef]
- Isola, G.; Polizzi, A.; Santonocito, S.; Alibrandi, A.; Ferlito, S. Expression of salivary and serum malondialdehyde and lipid profile of patients with periodontitis and coronary heart disease. Int. J. Mol. Sci. 2019, 20, 6061. [Google Scholar] [CrossRef]
- Isola, G.; Polizzi, A.; Muraglie, S.; Leonardi, R.; Lo Giudice, A. Assessment of Vitamin C and Antioxidant Profiles in Saliva and Serum in Patients with Periodontitis and Ischemic Heart Disease. Nutrients 2019, 11, 2956. [Google Scholar] [CrossRef] [PubMed]
- Toczewska, J.; Konopka, T.; Zalewska, A.; Maciejczyk, M. Nitrosative Stress Biomarkers in the Non-Stimulated and Stimulated Saliva, as well as Gingival Crevicular Fluid of Patients with Periodontitis: Review and Clinical Study. Antioxidants 2020, 9, 259. [Google Scholar] [CrossRef] [PubMed]
- Koss-Mikołajczyk, I.; Baranowska, M.; Namieśnik, J.; Bartoszek, A. Determination of antioxidantactivity of phytochemicals in cellular models by fluorescence/luminescence methods. Postepy Hig. Med. Dosw. 2017, 71, 602–617. [Google Scholar] [CrossRef] [PubMed]
- Erel, O. A new automated colorimetric method for measuring total oxidant status. Clin. Biochem. 2005, 38, 1103–1111. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Chung, W.Y. Total antioxidant power of plasma: Male-female differences and effect of anticoagulant used. Ann. Clin. Biochem. 1999, 36, 104–106. [Google Scholar] [CrossRef]
- Choromańska, B.; Myśliwiec, P.; Łuba, M.; Wojskowicz, P.; Myśliwiec, H.; Choromańska, K.; Żendzian-Piotrowska, M.; Dadan, J.; Zalewska, A.; Maciejczyk, M. Impact of Weight Loss on the Total Antioxidant/Oxidant Potential in Patients with Morbid Obesity—A Longitudinal Study. Antioxidants 2020, 9, 376. [Google Scholar] [CrossRef]
- Maciejczyk, M.; Szulimowska, J.; Taranta-Janusz, K.; Werbel, K.; Wasilewska, A.; Zalewska, A. Salivary FRAP as a marker of chronic kidney disease progression in children. Antioxidants 2019, 8, 409. [Google Scholar] [CrossRef]
- Maciejczyk, M.; Taranta-Janusz, K.; Wasilewska, A.; Kossakowska, A.; Zalewska, A. A Case-Control Study of Salivary Redox Homeostasis in Hypertensive Children. Can Salivary Uric Acid be a Marker of Hypertension? J. Clin. Med. 2020, 9, 837. [Google Scholar] [CrossRef] [PubMed]
- Skutnik-Radziszewska, A.; Maciejczyk, M.; Fejfer, K.; Krahel, J.; Flisiak, I.; Kołodziej, U.; Zalewska, A. Salivary Antioxidants and Oxidative Stress in Psoriatic Patients: Can Salivary Total Oxidant Status and Oxidative Status Index Be a Plaque Psoriasis Biomarker? Oxid. Med. Cell. Longev. 2020, 2020, 1–12. [Google Scholar] [CrossRef]
- Klimiuk, A.; Zalewska, A.; Sawicki, R.; Knapp, M.; Maciejczyk, M. Salivary Oxidative Stress Increases with the Progression of Chronic Heart Failure. J. Clin. Med. 2020, 9, 769. [Google Scholar] [CrossRef]
- Maciejczyk, M.; Szulimowska, J.; Taranta-Janusz, K.; Wasilewska, A.; Zalewska, A. Salivary Gland Dysfunction, Protein Glycooxidation and Nitrosative Stress in Children with Chronic Kidney Disease. J. Clin. Med. 2020, 9, 1285. [Google Scholar] [CrossRef] [PubMed]
- Choromańska, M.; Klimiuk, A.; Kostecka-Sochoń, P.; Wilczyńska, K.; Kwiatkowski, M.; Okuniewska, N.; Waszkiewicz, N.; Zalewska, A.; Maciejczyk, M. Antioxidant defence, oxidative stress and oxidative damage in saliva, plasma and erythrocytes of dementia patients. Can salivary AGE be a marker of dementia? Int. J. Mol. Sci. 2017, 18, 2205. [Google Scholar] [CrossRef] [PubMed]
- Maciejczyk, M.; Szulimowska, J.; Skutnik, A.; Taranta-Janusz, K.; Wasilewska, A.; Wiśniewska, N.; Zalewska, A. Salivary Biomarkers of Oxidative Stress in Children with Chronic Kidney Disease. J. Clin. Med. 2018, 7, 209. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, T.J.; Drake, R.B.; Naylor, J.E. The Plaque Control Record. J. Periodontol. 1972, 43, 38. [Google Scholar] [CrossRef]
- Lange, D.E.; Plagmann, H.C.; Eenboom, A.; Promesberger, A. Klinische Bewertungsverahren zur Objektivierung der Mundhygiene. Dtsch. Zahnarztl. Z. 1977, 32, 44–47. [Google Scholar]
- Ainamo, J.; Bay, I. Problems and proposals for recording gingivitis and plaque. Int. Dent. J. 1975, 25, 229–235. [Google Scholar]
- Newbrun, E. Indices to Measure Gingival Bleeding. J. Periodontol. 1996, 67, 555–561. [Google Scholar] [CrossRef]
- Taso, E.; Stefanovic, V.; Stevanovic, I.; Vojvodic, D.; Topic, A.; Petkovic-Curcin, A.; Obradovic-Djuricic, K.; Markovic, A.; Djukic, M.; Vujanovic, D. Influence of dental restorations on oxidative stress in gingival crevicular fluid. Oxid. Med. Cell. Longev. 2018, 2018, 1–17. [Google Scholar] [CrossRef]
- Erel, O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin. Biochem. 2004, 37, 277–285. [Google Scholar] [CrossRef]
- Zalewska, A.; Kossakowska, A.; Taranta-Janusz, K.; Zięba, S.; Fejfer, K.; Salamonowicz, M.; Kostecka-Sochoń, P.; Wasilewska, A.; Maciejczyk, M. Dysfunction of Salivary Glands, Disturbances in Salivary Antioxidants and Increased Oxidative Damage in Saliva of Overweight and Obese Adolescents. J. Clin. Med. 2020, 9, 548. [Google Scholar] [CrossRef]
- Zalewska, A.; Maciejczyk, M.; Szulimowska, J.; Imierska, M.; Błachnio-Zabielska, A. High-Fat Diet Affects Ceramide Content, Disturbs Mitochondrial Redox Balance, and Induces Apoptosis in the Submandibular Glands of Mice. Biomolecules 2019, 9, 877. [Google Scholar] [CrossRef] [PubMed]
- Zalewska, A.; Ziembicka, D.; Żendzian-Piotrowska, M.; Maciejczyk, M. The Impact of High-Fat Diet on Mitochondrial Function, Free Radical Production, and Nitrosative Stress in the Salivary Glands of Wistar Rats. Oxid. Med. Cell. Longev. 2019, 2019, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Zhang, X.L.; Wang, Y.Z.; Yang, C.X.; Chen, G. Lipid peroxidation levels, total oxidant status and superoxide dismutase in serum, saliva and gingival crevicular fluid in chronic periodontitis patients before and after periodontal therapy. Aust. Dent. J. 2010, 55, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Baltacıoğlu, E.; Yuva, P.; Aydın, G.; Alver, A.; Kahraman, C.; Karabulut, E.; Akalın, F.A. Lipid Peroxidation Levels and Total Oxidant/Antioxidant Status in Serum and Saliva from Patients with Chronic and Aggressive Periodontitis. Oxidative Stress Index: A New Biomarker for Periodontal Disease? J. Periodontol. 2014, 85, 1432–1441. [Google Scholar] [CrossRef]
- Zhang, T.; Andrukhov, O.; Haririan, H.; Müller-Kern, M.; Liu, S.; Liu, Z.; Rausch-Fan, X. Total Antioxidant Capacity and Total Oxidant Status in Saliva of Periodontitis Patients in Relation to Bacterial Load. Front. Cell. Infect. Microbiol. 2016, 5, 97. [Google Scholar] [CrossRef]
- Chapple, I.L.C.; Mason, G.I.; Garner, I.; Matthews, J.B.; Thorpe, G.H.; Maxwell, S.R.J.; Whitehead, T.P. Enhanced chemiluminescent assay for measuring the total antioxidant capacity of serum, saliva and crevicular fluid. Ann. Clin. Biochem. 1997, 34, 412–421. [Google Scholar] [CrossRef]
- Chapple, I.L.C.; Brock, G.; Eftimiadi, C.; Matthews, J.B. Glutathione in gingival crevicular fluid and its relation to local antioxidant capacity in periodontal health and disease. J. Clin. Pathol. Mol. Pathol. 2002, 55, 367–373. [Google Scholar] [CrossRef]
- Sculley, D.V.; Langley-Evans, S.C. Periodontal disease is associated with lower antioxidant capacity in whole saliva and evidence of increased protein oxidation. Clin. Sci. 2003, 105, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Diab-Ladki, R.; Pellat, B.; Chahine, R. Decrease in the total antioxidant activity of saliva in patients with periodontal diseases. Clin. Oral Investig. 2003, 7, 103–107. [Google Scholar] [CrossRef]
- Brock, G.R.; Butterworth, C.J.; Matthews, J.B.; Chapple, I.L.C. Local and systemic total antioxidant capacity in periodontitis and health. J. Clin. Periodontol. 2004, 31, 515–521. [Google Scholar] [CrossRef]
- Chapple, I.L.C.; Brock, G.R.; Milward, M.R.; Ling, N.; Matthews, J.B. Compromised GCF total antioxidant capacity in periodontitis: Cause or effect? J. Clin. Periodontol. 2007, 34, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Konopka, T.; Król, K.; Kopeć, W.; Gerber, H. Total antioxidant status and 8-hydroxy-2′-deoxyguanosine levels in gingival and peripheral blood of periodontitis patients. Arch. Immunol. Ther. Exp. 2007, 55, 417–425. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Gornitsky, M.; Velly, A.M.; Yu, H.; Benarroch, M.; Schipper, H.M. Salivary DNA, lipid, and protein oxidation in nonsmokers with periodontal disease. Free Radic. Biol. Med. 2009, 46, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Abou Sulaiman, A.E.; Shehadeh, R.M.H. Assessment of Total Antioxidant Capacity and the Use of Vitamin C in the Treatment of Non-Smokers with Chronic Periodontitis. J. Periodontol. 2010, 81, 1547–1554. [Google Scholar] [CrossRef]
- Dhotre, P.S.; Suryakar, A.N.; Bhogade, R.B. Oxidative Stress in Periodontitis. Electron. J. Gen. Med. 2012, 9, 81–84. [Google Scholar] [CrossRef]
- Konuganti, K.; Seshan, H.; Zope, S.; Silvia, W.D. A comparative evaluation of whole blood total antioxidant capacity using a novel nitroblue tetrazolium reduction test in patients with periodontitis and healthy subjects: A randomized, controlled trial. J. Indian Soc. Periodontol. 2012, 16, 620–622. [Google Scholar] [CrossRef]
- Novakovic, N.; Todorovic, T.; Rakic, M.; Milinkovic, I.; Dozic, I.; Jankovic, S.; Aleksic, Z.; Cakic, S. Salivary antioxidants as periodontal biomarkers in evaluation of tissue status and treatment outcome. J. Periodontal. Res. 2014, 49, 129–136. [Google Scholar] [CrossRef]
- Miricescu, D.; Totan, A.; Calenic, B.; Mocanu, B.; Didilescu, A.; Mohora, M.; Spinu, T.; Greabu, M. Salivary biomarkers: Relationship between oxidative stress and alveolar bone loss in chronic periodontitis. Acta Odontol. Scand. 2014, 72, 42–47. [Google Scholar] [CrossRef]
- Thomas, B.; Madani, S.M.; Prasad, B.R.; Kumari, S. Comparative evaluation of serum antioxidant levels in periodontally diseased patients: An interventional study. Contemp. Clin. Dent. 2014, 5, 340–344. [Google Scholar]
- Baňasová, L.; Kamodyová, N.; Janšáková, K.; Tóthová, Ľ.; Stanko, P.; Turňa, J.; Celec, P. Salivary DNA and markers of oxidative stress in patients with chronic periodontitis. Clin. Oral Investig. 2015, 19, 201–207. [Google Scholar] [CrossRef]
- Acquier, A.B.; De Couto Pita, A.K.; Busch, L.; Sánchez, G.A. Parameters of oxidative stress in saliva from patients with aggressive and chronic periodontitis. Redox Rep. 2017, 22, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Becerik, S.; Öztürk, V.Ö.; Celec, P.; Kamodyova, N.; Atilla, G.; Emingil, G. Gingival crevicular fluid and plasma oxidative stress markers and TGM-2 levels in chronic periodontitis. Arch. Oral Biol. 2017, 83, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi-Motamayel, F.; Goodarzi, M.T.; Jamshidi, Z.; Kebriaei, R. Evaluation of Salivary and Serum Antioxidant and Oxidative Stress Statuses in Patients with Chronic Periodontitis: A Case-Control Study. Front. Physiol. 2017, 8, 189. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.; Singh, S.T.; Sharma, V.; Verma, A.; Singh, C.D.; Gill, J.S. Assessment of lipid peroxidation levels and total antioxidant status in chronic and aggressive periodontitis patients: An in vivo study. J. Contemp. Dent. Pract. 2018, 19, 287–291. [Google Scholar]
- Narendra, S.; Das, U.K.; Tripathy, S.K.; Sahani, N.C. Superoxide dismutase, uric acid, total antioxidant status, and lipid peroxidation assay in chronic and aggressive periodontitis patients. J. Contemp. Dent. Pract. 2018, 19, 874–880. [Google Scholar] [CrossRef]
- Badarinath, A.V.; Mallikarjuna Rao, K.; Madhu Sudhana Chetty, C.; Ramkanth, S.; Rajan, T.V.S.; Gnanaprakash, K. A review on In-vitro antioxidant methods: Comparisions, correlations and considerations. Int. J. PharmTech Res. 2010, 2, 1276–1285. [Google Scholar]
- Akpinar, A.; Toker, H.; Ozdemir, H.; Bostanci, V.; Aydin, H. The effects of non-surgical periodontal therapy on oxidant and anti-oxidant status in smokers with chronic periodontitis. Arch. Oral Biol. 2013, 58, 717–723. [Google Scholar] [CrossRef]
- Bostanci, V.; Toker, H.; Senel, S.; Ozdemir, H.; Aydin, H. Effect of Chronic Periodontitis on Serum and Gingival Crevicular Fluid Oxidant and Antioxidant Status in Patients with Familial Mediterranean Fever Before and After Periodontal Treatment. J. Periodontol. 2014, 85, 706–712. [Google Scholar] [CrossRef]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and Clinical Significance of Biomarkers of Oxidative Stress in Humans. Oxid. Med. Cell. Longev. 2017, 2017, 1–32. [Google Scholar] [CrossRef]
- Peluso, I.; Raguzzini, A. Salivary and Urinary Total Antioxidant Capacity as Biomarkers of Oxidative Stress in Humans. Patholog. Res. Int. 2016, 2016, 1–14. [Google Scholar] [CrossRef]
- Chen, M.; Cai, W.; Zhao, S.; Shi, L.; Chen, Y.; Li, X.; Sun, X.; Mao, Y.; He, B.; Hou, Y.; et al. Oxidative stress-related biomarkers in saliva and gingival crevicular fluid associated with chronic periodontitis: A systematic review and meta-analysis. J. Clin. Periodontol. 2019, 46, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Dhotre, P.S.; Suryakar, A.N.; Bhogade, R.B. Oxidative stress in periodontitis: A critical link to cardiovascular disease. Biomed. Res. 2011, 22, 178–182. [Google Scholar]
- Evin, A.; Gamsiz-Isik, H.; Cifcibasi, E.; Yalcin, F. Plasma and salivary total antioxidant capacity in healthy controls compared with aggressive and chronic periodontitis patients. Saudi Med. J. 2015, 36, 856–861. [Google Scholar]
- Nguyen, T.T.; Ngo, L.Q.; Promsudthi, A.; Surarit, R. Salivary Lipid Peroxidation in Patients with Generalized Chronic Periodontitis and Acute Coronary Syndrome. J. Periodontol. 2016, 87, 134–141. [Google Scholar] [CrossRef]
- Rhemrev, J.P.T.; Van Overveld, F.W.P.C.; Haenen, G.R.M.M.; Teerlink, T.; Bast, A.; Vermeiden, J.P.W. Quantification of the nonenzymatic fast and slow TRAP in a postaddition assay in human seminal plasma and the antioxidant contributions of various seminal compounds. J. Androl. 2000, 21, 913–920. [Google Scholar]
- Liu, Z.; Liu, Y.; Song, Y.; Zhang, X.; Wang, S.; Wang, Z. Systemic Oxidative Stress Biomarkers in Chronic Periodontitis: A Meta-Analysis. Dis. Mark. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Toczewska, J.; Konopka, T. Activity of enzymatic antioxidants in periodontitis: A systematic overview of the literature. Dent. Med. Probl. 2019, 56, 419–426. [Google Scholar] [CrossRef]
- Żukowski, P.; Maciejczyk, M.; Waszkiel, D. Sources of free radicals and oxidative stress in the oral cavity. Arch. Oral Biol. 2018, 92, 8–17. [Google Scholar] [CrossRef]
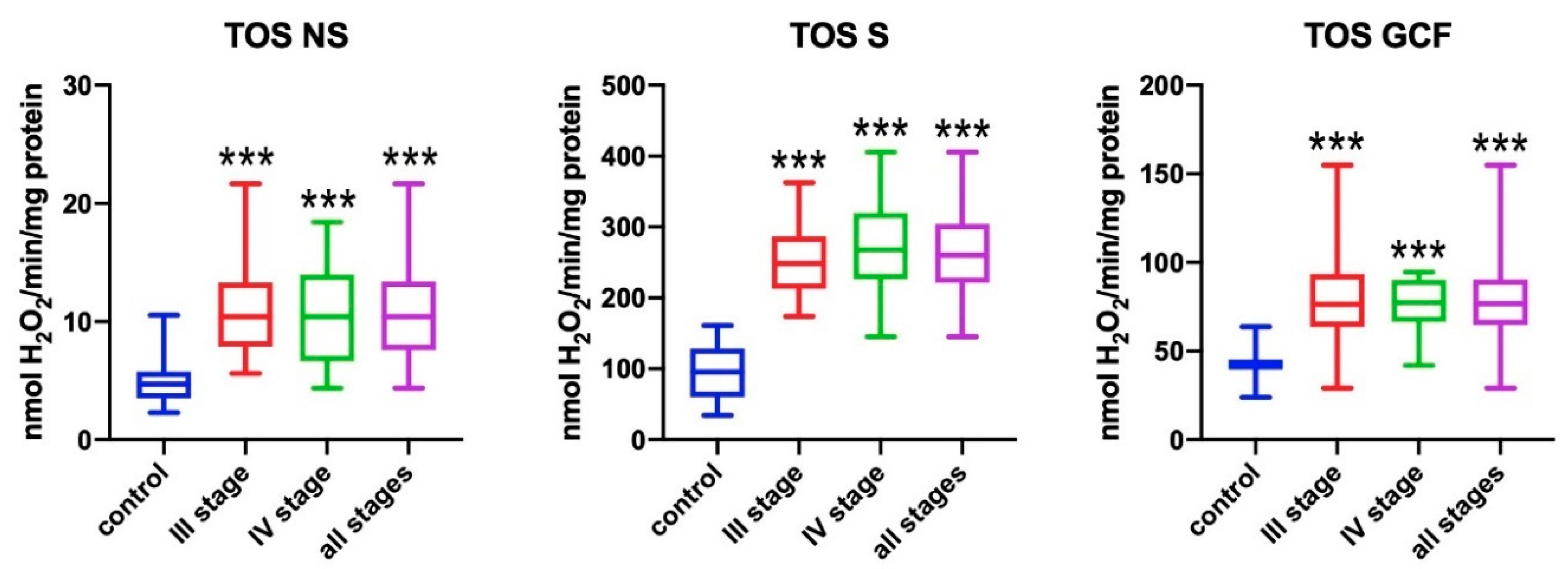
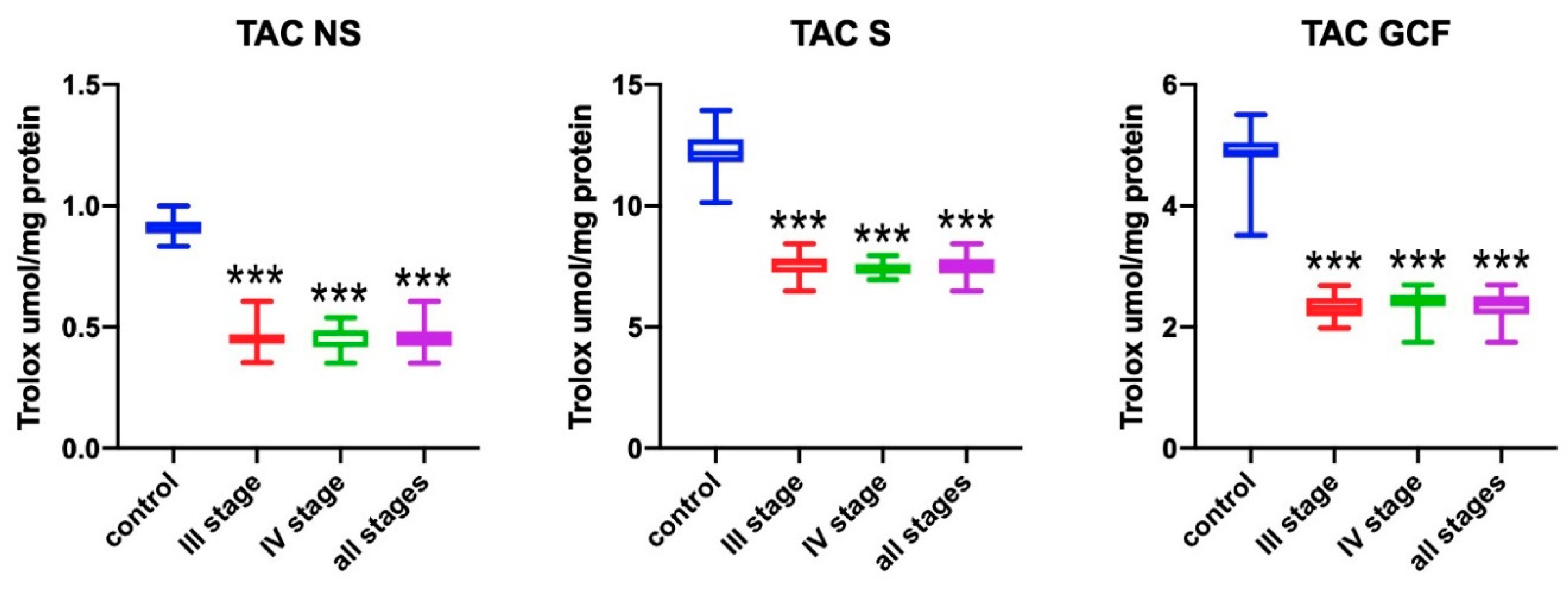
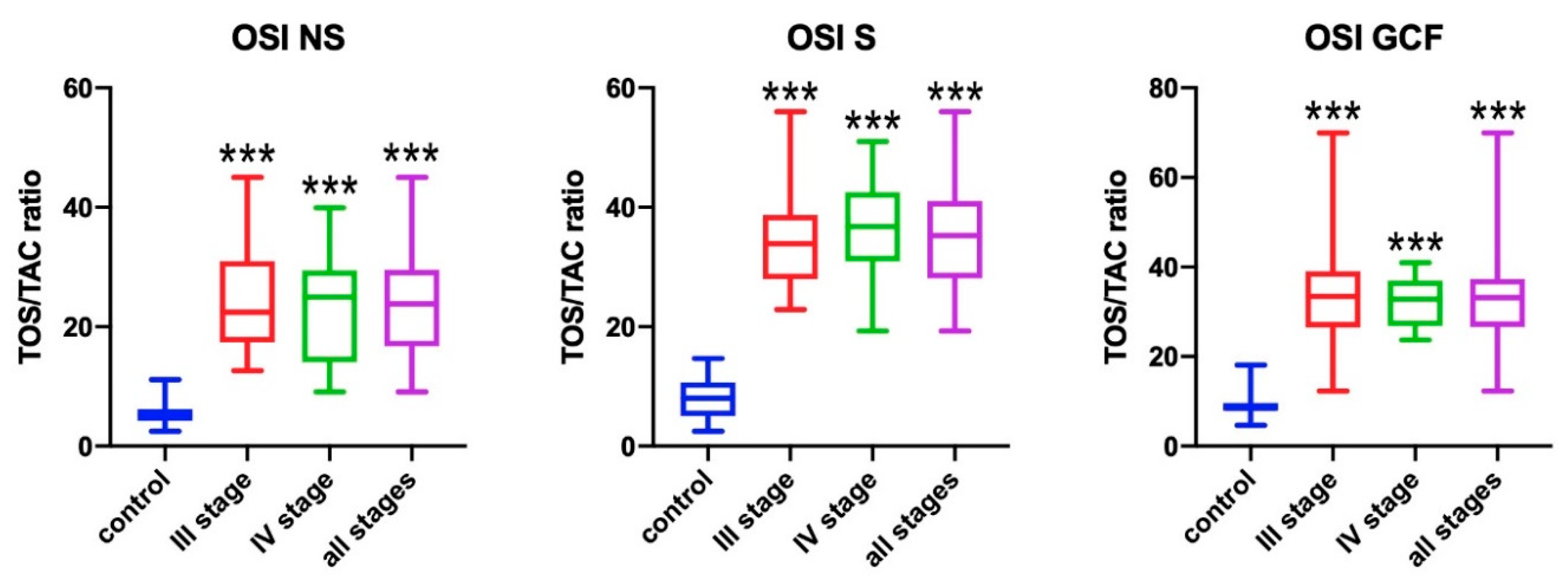
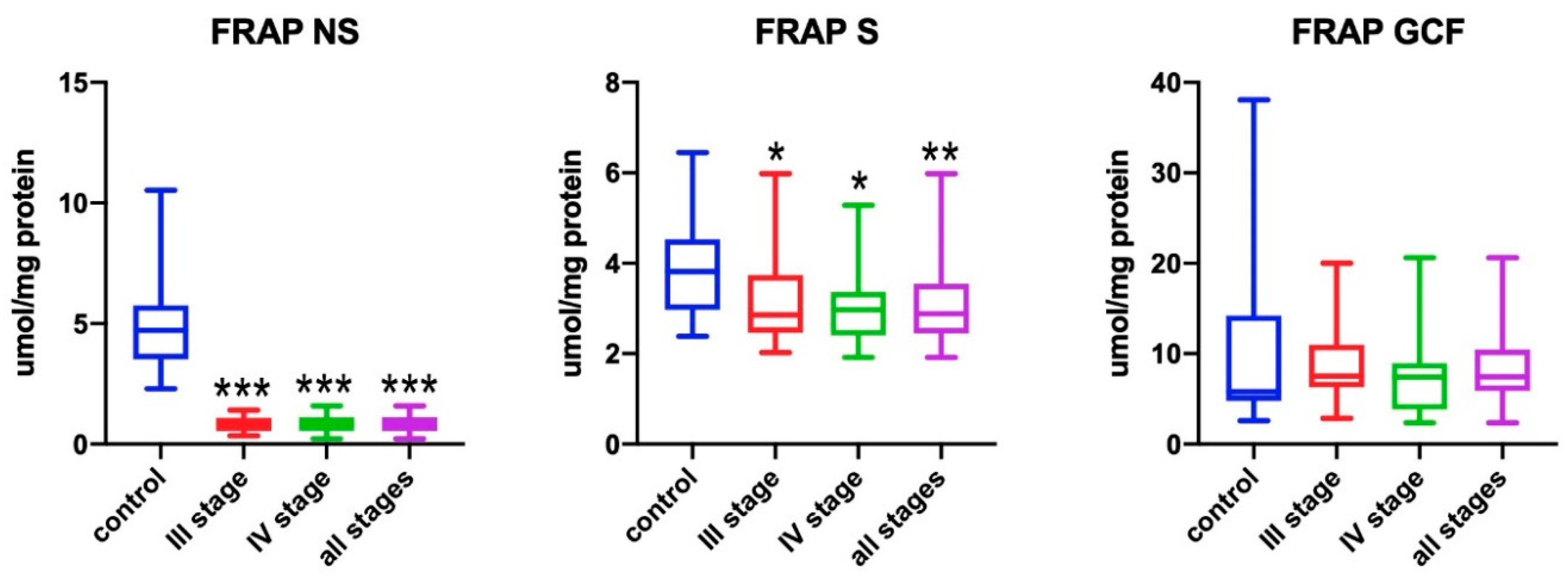
| Parameter | Control | Stage III | Stage IV | All Stages | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | Min | Max | Median | Min | Max | Median | Min | Max | Median | Min | Max | ||
| Age | 39 | 20 | 55 | 44 | 20 | 55 | 45 | 29 | 55 | 45 | 20 | 55 | |
| sex | Women | 17 (59%) | 16 (47%) | 13 (54%) | 29 (50%) | ||||||||
| Men | 12 (41%) | 18 (53%) | 11 (46%) | 29 (50%) | |||||||||
| Unstimulated saliva flow (mL/min) | 0.4 | 0.2 | 1 | 0.4 | 0.1 | 1 | 0.45 | 0.1 | 1.3 | 0.4 | 0.1 | 1.3 | |
| Stimulated saliva flow (mL/min) | 1.6 | 0.4 | 3.4 | 1.4 | 0.3 | 3 | 1.4 | 0.6 | 3 | 1.4 | 0.3 | 3 | |
| Protein in unstimulated saliva (μg/mL) | 589.51 | 300.45 | 1101 | 827.95 * | 481.23 | 1387 | 839.62 * | 23.48 | 1847.1 | 833.79 * | 23.48 | 1847.1 | |
| Protein in stimulated saliva (μg/mL) | 598.98 | 235.74 | 946.29 | 610.32 | 28.91 | 926.41 | 634.78 | 43.74 | 811.92 | 634.78 | 28.91 | 926.41 | |
| Protein in gingival fluid (μg/mL) | 30.94 | 8.44 | 91.66 | 130.43 * | 36.8 | 336.97 | 134.24 * | 45.5 | 445.64 | 131.13 * | 36.8 | 445.64 | |
| Number of teeth | 27 | 19 | 28 | 28 | 24 | 28 | 23* | 15 | 28 | 26 | 15 | 28 | |
| PI | 20 | 0 | 79 | 43 * | 9 | 100 | 43.5 * | 0 | 100 | 43.5 * | 0 | 100 | |
| API | 32 | 7 | 68 | 65 * | 29 | 100 | 86.5 * | 22 | 100 | 72.5 * | 22 | 100 | |
| BoP | 10 | 0.7 | 26 | 41 * | 4 | 100 | 61 * | 17 | 100 | 46.5 * | 4 | 100 | |
| PD | 1.7 | 1.2 | 2.3 | 3.15 * | 2.1 | 5.3 | 4.1 * | 2.7 | 5.4 | 3.5 * | 2.1 | 5.4 | |
| Mean CAL > 0 | 2.1 | 1 | 5.2 | 4.95 * | 2.4 | 8.1 | 6.05* | 3 | 10.1 | 5.4* | 2.4 | 10.1 | |
| Author, Year, and Country | Fluid Method | Study Group Size and Age | p for Perio | Other Data |
|---|---|---|---|---|
| Chapple et al. 1997 [36], Great Britain | NS Serum TAC—Chemiluminescence | CP—18 (>35) HP—16 (>35) | Saliva ↓ p < 0.01 Serum n.s. | |
| Chapple et al. 2002 [37], Great Britain | GCF Plasma TAC—Chemiluminescence | P—10 (mean 46,1) HP—10 (mean 46,9) | GCF ↓ p < 0.015 Plasma n.s. | |
| Sculley and Langley-Evans 2003 [38], Great Britain | NS FRAP—TPTZ Benzie, 1996 | AgP—46 (mean 59,6) HP—46 (mean 60,3) | FRAP n.s. | |
| Diab-Ladki et al. 2003 [39], Libia | S TAC—ABTS Miller, 1997 | AgP—17 (30–45) HP—20 (30–45) | ↓ p < 0.05 (40%) | |
| Brock et al. [40] 2004, Great Britain | S NS GCF Serum TAC—Chemiluminescence | CP—17 (mean 43,5) HP—27 (mean 44,7) | GCF ↓ p < 0.001 NS, S, serum n.s. | No significant correlation in GCF TAC-PD, TAC in saliva lower than GCF p < 0.0005. |
| Chapple et al. [41] 2007, Great Britain | GCF Plasma TAC—Chemiluminescence | CP—35 (32–61) | Significant raise in TAC in GCF after periodontal treatment p < 0.001, plasma n.s. | |
| Konopka et al. [42] 2007, Poland | Serum TAC—ABTS Re, 1999 | CP—30 (mean 44,9) AgP—26 (mean 31,5) HP—25 (mean 33,2) | CP and AgP ↓ p = 0.000, ↓ in CP/AgP | No significant correlation with clinical status. |
| Su et al. [43] 2009, Canada | NS TAC—ABTS Miller, 1997 | P—58 (mean 52,3) HP—234 (mean 45,4) | ↓ p < 0.0001 | Significant correlation with perio disease and CPITN |
| Abou Sulaiman et al. [44] 2010, Siria | Plasma TAC—ABTS Erel, 2004 | CP—30 (mean 41) HP—30 (mean 34) | ↓ p < 0.001 | No significant correlation with clinical status; significant raise after periodontal treatment. |
| Dhotre et al. [45] 2012, India | Serum FRAP—TPTZ Benzie, 1996 | P—25 (no data) HP—25 (no data) | ↓ p < 0.001 | |
| Konuganti et al. [46], 2012, India | Blood TAC—NBT | CP—15 (18–40) HP—15 (18–40) | ↓ p < 0.001 | |
| Novakovic et al. [47] 2014, Serbia | NS TAC—ABTS | CP—21 (mean 39,1) HP—21 (mean 35,2) | TAC-n.s. | No significant correlation with clinical status. |
| Miricescu et al. [48], 2014, Romania | NS TAC—ABTS | CP—20 (mean 51,3) HP—20 (mean 18,6) | ↓ p < 0.05 | |
| Baltacioğlu et al. [34] 2014, Turkey | NS Serum TAC—ABTS Erel, 2004 OSI—Erel, 2004 | CP—33 (>40) AgP—35 (18–40) HP—30 (no data) | Saliva, serum ↓ TAC p = 0.001 ↑ OSI p = 0.001 | Significant correlation: TAC in saliva and serum with PI, GI, PD and CAL. Significant positive correlation with OSI. |
| Thomas et al. [49] 2014, India | Serum TAC—No data | CP—25 (no data) HP—25 (no data) | ↓ p < 0,001 | |
| Baňasova et al. [50] 2015, Slovakia | NS TAC—ABTS Erel, 2004 FRAP—TPTZ, Benzie, 1996 | CP—23 (mean 43) HP—19 (mean 39,1) | ↓TAC in women p < 0.01 ↓ FRAP in women | Significant positive correlation TAC and FRAP with a low value of PD and CAL. |
| Acquier et al. [51] 2016, Argentina | NS TRAP—Lissi and Vargas | CP—20 (mean 37,4) AgP—20 (mean 19,5) HP—20 (17–40) | CP and AgP ↑ p < 0.001 ↑ CP/AgP | No significant correlation with clinical status. |
| Becerik et al. [52] 2017, Turkey | GCF Plasma TAC—ABTS Erel, 2004 FRAP—TPTZ, Benzie, 1996 | CP—20 (mean 43,1) HP—20 (mean 38,4) | GCF TAC n.s. plasma ↓ FRAP TAC n.s. | Significant correlation in GCF: FRAP-PI, PBI, PD, and CAL; positive significant correlation with plasma TAC-PD FRAP-CAL |
| Ahmadi-Motamayel et al. [53] 2017, Iran | NS Serum FRAP—Riviere | CP—55 (30–50) HP—55 (30–50) | Saliva and serum n.s. | |
| Tripathi et al. [54] 2018, India | NS Serum TAC—ELISA OSI—ABTS Erel, 2004 | CP—40 (>18) AgP—40 (18–40) HP—40 (no data) | Saliva ↓ TAC p = 0.04 ↑ OSI (in CP) p = 0.01 Serum ↓ TAC p = 0.03 ↑ OSI p = 0.02 | ↑ OSI saliva in CP p = 0.01 ↑ OSI serum in CP and AgP p = 0.02 |
| Narenda et al. [55] 2018, India | GCF Serum TAC—ABTS Miller, 1997 | CP—46 (mean 47,1) AgP—32 (mean 25,7) HP—50 (mean 36,6) | GCF in AgP ↓ p < 0.001 Serum in AgP ↓ p < 0.001 | |
| Own study | GCF NS S TAC, OSI—ABTS Erel, 2004 FRAP—TPTZ, Benzie, 1996 | P—60 (mean 43,6) HP—30 (mean 40,3) | GCF ↓TAC p < 0.001 ↑OSI p < 0.001 FRAP n.s. Saliva ↓TAC p < 0.001 ↑OSI p < 0.001 ↓ FRAP p < 0.001 | For significant p positive correlations between TAC in GCF—PD > 5 mm, negative correlation with TAC in stimulated saliva with PI; for stage IV negative correlation between OSI in NS—PI. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toczewska, J.; Maciejczyk, M.; Konopka, T.; Zalewska, A. Total Oxidant and Antioxidant Capacity of Gingival Crevicular Fluid and Saliva in Patients with Periodontitis: Review and Clinical Study. Antioxidants 2020, 9, 450. https://doi.org/10.3390/antiox9050450
Toczewska J, Maciejczyk M, Konopka T, Zalewska A. Total Oxidant and Antioxidant Capacity of Gingival Crevicular Fluid and Saliva in Patients with Periodontitis: Review and Clinical Study. Antioxidants. 2020; 9(5):450. https://doi.org/10.3390/antiox9050450
Chicago/Turabian StyleToczewska, Joanna, Mateusz Maciejczyk, Tomasz Konopka, and Anna Zalewska. 2020. "Total Oxidant and Antioxidant Capacity of Gingival Crevicular Fluid and Saliva in Patients with Periodontitis: Review and Clinical Study" Antioxidants 9, no. 5: 450. https://doi.org/10.3390/antiox9050450
APA StyleToczewska, J., Maciejczyk, M., Konopka, T., & Zalewska, A. (2020). Total Oxidant and Antioxidant Capacity of Gingival Crevicular Fluid and Saliva in Patients with Periodontitis: Review and Clinical Study. Antioxidants, 9(5), 450. https://doi.org/10.3390/antiox9050450







