The Use of Antioxidants in the Treatment of Migraine
Abstract
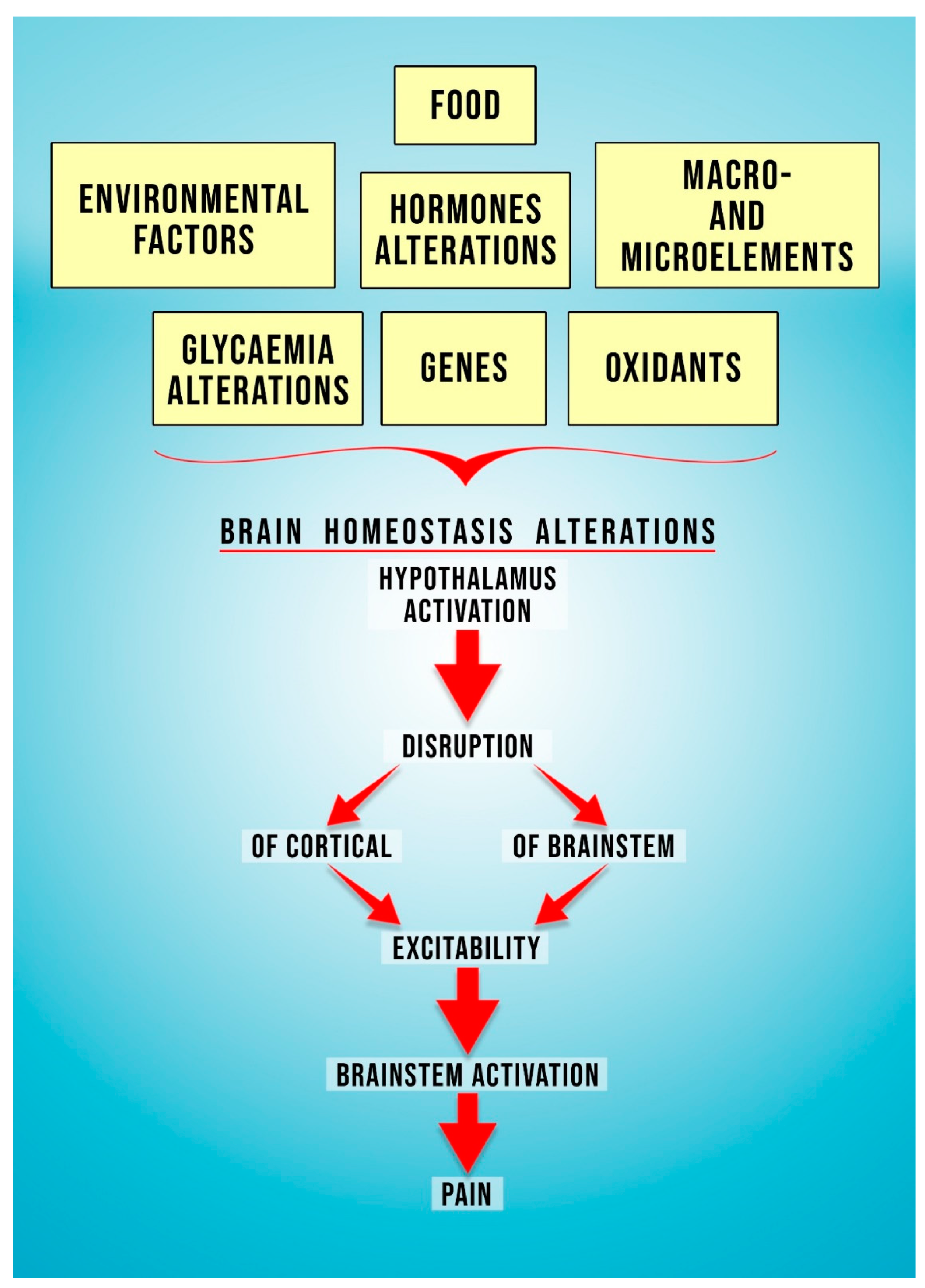
1. Characteristics of Migraine
2. The Role of Oxidative Stress in the Pathophysiology of Migraine
3. Migraine and Plant Antioxidants
4. Vitamin C
5. Curcumin
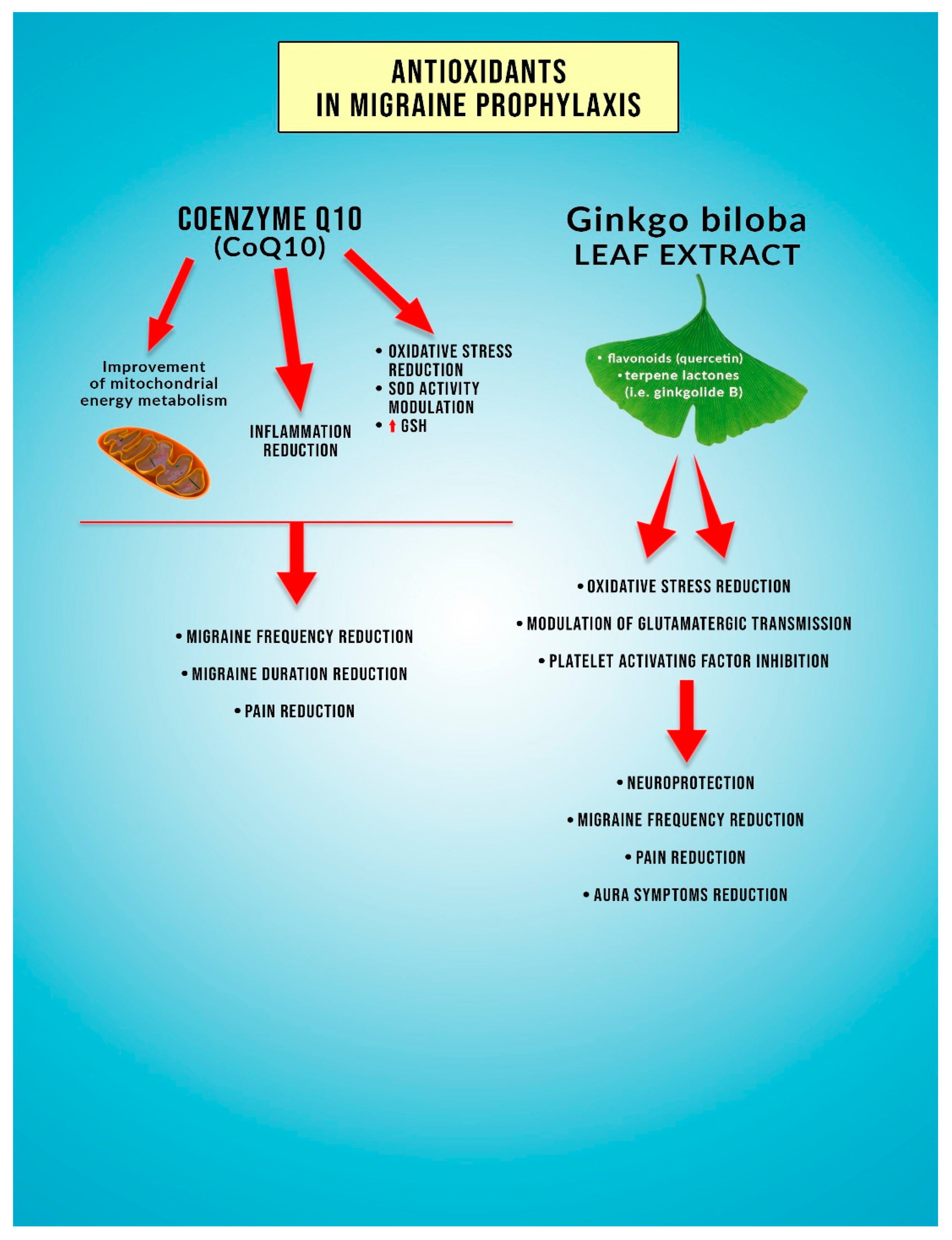
6. Coenzyme Q10
7. Ginkgolide B
8. Feverfew (Tanacetum parthenium)
| Antioxidants in Migraine Treatment | |||
|---|---|---|---|
| Agent | Mechanism of Action | Authors of the Study | Results Obtained |
| Vitamin C | Antioxidant ROS scavenger | Chayasirisobhon [56] Chayasirisobhon [57] | Improvement in MIDAS score, number of headaches days, and headache severity reduction |
| Curcumin | Antioxidant Anti-inflammatory properties Anti-apoptotic properties | Bulboacă et al. [66] Bulboacă et al. [67] Bulboacă et al. [68] | Decrease in MDA concentration Decrease in RNS synthesis Decrease in TOS Increase in TAS Reduction of nociception Analgesic effect |
| Parohan et al. [70] | Decrease in migraine attacks: Frequency Duration Severity | ||
| Coenzyme Q10 | Antioxidant Anti-inflammatory properties Mitochondrial energy metabolism maintenance | Dahri et al. [80] | Decrease in CGRP level |
| Hershey et al. [81] | Increase in CoQ10 plasma concentration Improvement in PedMIDAS score Reduction of headaches episodes frequency | ||
| Zeng et al. [82] | Decrease in migraine attacks: Frequency Duration | ||
| Slater et al. [83] | Decreased severity of headache in the four first weeks Decreasing trend in terms of the frequency of headache episodes | ||
| Guilbot et al. [84] | Reduced number of days with migraine per month Reduced signs of anxiety and depressive symptoms Reduced photosensitivity and nausea Life quality improvement | ||
| Gaul et al. [78] | Reduced severity of migraine | ||
| Shoeibi [85] | Reduction of the migraine episodes frequency Reduction of the single episode duration | ||
| Ginkgolide B | Glutamatergic Transmission modulator Suggested as antioxidant | D’Andrea et al. [95] | Reduction of the migraine episodes frequency |
| Allais et al. [11] | Significant shortening of aura duration | ||
| Feverfew | Antioxidant Anti-inflammation properties | Pfaffenrath et al. [101] | Lack of overall significant effect |
| Diener et al. [102] | Decrease in the frequency of the migraine episodes | ||
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CAT | catalase |
| CGRP | calcitonin-gene related peptide |
| CSD | cortical spreading depression |
| CoQ10 | coenzyme Q10 |
| COX | cyclooxygenase |
| CRPS | complex regional pain syndrome |
| GB | Ginkgo biloba |
| GSH | glutathione |
| HPLC | high performance liquid chromatography |
| LAA | L-ascorbic-acid |
| MDA | malondialdehyde |
| MIDAS | Migraine Disability Assessment Scale |
| NF κB | nuclear transcription factor κB |
| OSI | oxidative stress index |
| PAF | platelet activating factor |
| PedMIDAS | Migraine Disability Assessment Scale in pediatric and adolescent patients |
| QVM | Qualité de Vie et Migraine questionnaire |
| RNS | reactive nitrogen species |
| ROS | reactive oxygen species |
| SOD | superoxide dismutase |
| ST | sumatriptan |
| TAC | total antioxidant capacity |
| TAS | total antioxidant status |
| TOS | total oxidant status |
| TGA | transient global ischemia |
References
- Tottene, A.; Favero, M.; Pietrobon, M. Enhanced thalamocortical synaptic transmission and dysregulation of the excitatory-inhibitory balance at the thalamocortical feed-forward inhibitory microcircuit in a genetic mouse model of migraine. J. Neurosci. 2019, 39, 9841–9851. [Google Scholar] [CrossRef]
- Burch, R.C.; Buse, D.C.; Lipton, R.B. Migraine: Epidemiology, burden, and comorbidity. Neurol. Clin. 2019, 37, 631–649. [Google Scholar] [CrossRef] [PubMed]
- Burstein, R.; Noseda, R.; Borsook, D. Migraine multiple processes, complex pathophysiology. J. Neurosci. 2015, 35, 6619–6629. [Google Scholar] [CrossRef] [PubMed]
- Noseda, R.; Burstein, R. Migraine pathophysiology: Anatomy of the trigeminovascular pathway and associated neurological symptoms, cortical spreading depression, sensitization, and modulation of pain. Pain 2013, 154 (Suppl. 1), S44–S53. [Google Scholar] [CrossRef]
- Stewart, W.F.; Wood, C.; Reed, M.L.; Roy, J.; Lipton, R.B. Cumulative lifetime migraine incidence in women and men. Cephalgia 2008, 28, 1170–1178. [Google Scholar] [CrossRef]
- Stovner, L.J.; Hagen, K. Prevalence, burden, and cost of headache disorders. Curr. Opin. Neurol. 2006, 19, 281–285. [Google Scholar] [CrossRef]
- Launer, L.J.; Terwindt, G.M.; Ferrari, M.D. The prevalence and characteristics of migraine in a population based cohort: The GEM study. Neurology 1999, 53, 537–542. [Google Scholar] [CrossRef]
- Viana, M.; Sances, G.; Linde, M.; Nappi, G.; Khaliq, F.; Goadsby, P.J.; Tassorelli, C. Prolonged migraine aura: New insights from a prospective diary-aided study. J. Headache Pain 2018, 19, 77. [Google Scholar] [CrossRef]
- D’Andrea, G.; Bonavita, V.; Rigamonti, A.; Bussone, G. Treatment of migraine with aura: Comment and prospectives. Neurol. Sci. 2003, 23, 271–278. [Google Scholar] [CrossRef]
- Asher, J.M.; O’Hare, L.; Romei, V. Typical lateral interactions, but increased contrast sensitivity, in migraine-with-aura. Vision 2018, 2, 7. [Google Scholar] [CrossRef]
- Allais, G.; D’Andrea, G.; Maggio, M.; Benedotto, C. The efficiacy of ginkgolide B in the acute treatment of migraine aura: An open preliminary trial. Neurol. Sci. 2013, 34 (Suppl. 1), S161–S163. [Google Scholar] [CrossRef] [PubMed]
- Bolay, H.; Reuter, U.; Dunn, A.K.; Huang, Z.; Boas, D.A.; Moskowitz, M.A. Intrinsic brain activity triggers trigeminal meningeal afferents in a migraine model. Nat. Med. 2002, 8, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Wolthausen, J.; Sternberg, S.; Gerloff, C.; May, A. Are cortical spreading depression and headache in migraine casually linked? Cephalgia 2009, 29, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, H.G.; Albury, C.L.; Griffiths, L.R. Advances in genetics of migraine. J. Headache Pain 2019, 20, 72. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J. Pathophysiology of migraine. Ann. Indian Acad. Neurol. 2012, 15 (Suppl. 1), S15–S22. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, G.; Granella, F.; Cataldini, M.; Verdelli, F.; Balbi, T. GABA and glutamate in migraine. J. Headache Pain 2001, 2, S57–S60. [Google Scholar] [CrossRef]
- Puppe, A.; Limmroth, V. GABAergic drugs for the treatment of migraine. CNS Neurol. Disord. Drug Targets 2007, 6, 247–250. [Google Scholar] [CrossRef]
- D’Andrea, G.; Cananzi, A.R.; Ferro-Milone, F.; Joseph, R.; Grunfeld, S.; Welch, K.M.A. Platelet levels of glutamate and aspartate in normal subjects. Stroke 1989, 2, 299–300. [Google Scholar] [CrossRef]
- Welch, K.M.A.; D’Andrea, G.; Tepley, N.; Barkley, G.L.; Ramadan, N.M. The concept of migraine as a state of central neuronal hyperexcitability. Neurol. Clin. 1990, 8, 817–828. [Google Scholar] [CrossRef]
- Yorns, W.R., Jr.; Hardison, H.H. Mitochondrial dysfunction in migraine. Semin. Pediatr. Neurol. 2013, 20, 188–193. [Google Scholar] [CrossRef]
- Colombo, B.; Saraceno, L.; Comi, G. Riboflavin and migraine the bridge over troubled mitochondria. Neurol. Sci. 2014, 35, 141–144. [Google Scholar] [CrossRef]
- Gross, E.C.; Klement, R.J.; Schenen, J.; D’Agostino, D.P.; Fischer, D. Potential protective mechanisms of ketone bodies in migraine prevention. Nutrients 2019, 11, 811. [Google Scholar] [CrossRef]
- Neri, M.; Frustaci, A.; Milic, M.; Valdiglesias, V.; Fini, M.; Bonassi, S.; Barbanti, P. A meta-analysis of biomarkers related to oxidative stress and nitric oxide pathway in migraine. Cephalgia 2015, 35, 931–937. [Google Scholar] [CrossRef]
- Borkum, J.M. Migraine triggers and oxidative stress: A narrative review and synthesis. Headache 2015, 56, 12–35. [Google Scholar] [CrossRef]
- Goschorska, M.; Baranowska-Bosiacka, I.; Gutowska, I.; Metryka, E.; Skórka- Majewicz, M.; Chlubek, D. Potential role of fluoride in etiopathogenesis of the Alzheimer’s Disease. Int. J. Mol. Sci. 2018, 19, 3956. [Google Scholar] [CrossRef]
- Aboonabi, A.; Meyer, R.R.; Singh, I. The association between metabolic syndrome components and the development of atherosclerosis. J. Hum. Hypertens 2019, 33, 844–855. [Google Scholar] [CrossRef]
- Jabarpour, M.; Rashtchizadeh, N.; Argani, H.; Ghorbanihaghjo, A.; Ranjbarzadhag, M.; Sanajou, D.; Panah, F.; Alirezaei, A. The impact of dyslipidemia and oxidative stress on vasoactive mediators in patients with renal dysfunction. Int. Urol. Nephrol. 2019, 51, 2235–2242. [Google Scholar] [CrossRef]
- Rodrigo, R.; Fernández-Gajardo, R.; Gutiérrez, R.; Matamala, J.M.; Carrasco, R.; Miranda-Merchak, A.; Feuerhake, W. Oxidative stress and pathophysiology of ischemic stroke: Novel therapeutic opportunities. CNS Neurol. Disord. Drug Targets 2013, 12, 698–714. [Google Scholar] [CrossRef]
- Tobore, T.O. Towards a Comprehensive Etiopathogenetic and Pathophysiological Theory of Multiple Sclerosis. Int. J. Neurosci. 2019, 7, 1–41. [Google Scholar] [CrossRef]
- Pohanka, M. Oxidative stress in Alzheimer disease as a target for therapy. Bratisl. Lek. Listy 2018, 119, 535–543. [Google Scholar] [CrossRef]
- Tozzi-Ciancarelli, M.G.; De Matteis, G.; Di Massimo, C.; Marini, C.; Ciancarelli, I.; Carolei, A. Oxidative stress and platelet responsiveness in migraine. Cephalgia 1997, 17, 580–584. [Google Scholar] [CrossRef]
- Alp, R.; Selek, S.; Alp, S.I.; Taşkin, A.; Koçyiğit, A. Oxidative and antioxidative balance in patients with migraine. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 877–882. [Google Scholar] [CrossRef]
- Geyik, S.; Altunisik, E.; Neyal, A.M.; Taysi, S. Oxidative stress and DNA damage in patients with migraine. J. Headache Pain 2016, 17, 10. [Google Scholar] [CrossRef]
- Yigit, M.; Sogut, O.; Tataroglu, Ö.; Yamanoglu, A.; Yigit, E.; Güler, E.M.; Ozer, O.F.; Kocyigit, A. Oxidative/antioxidative status, lymphocyte DNA damage, and urotensin-2 receptor level in patients in migraine attacks. Neuropsychiatr. Dis. Treat. 2018, 14, 367–374. [Google Scholar] [CrossRef]
- Aytaç, B.; Coşkun, Ö.; Alioğlu, B.; Durak, Z.E.; Büber, S.; Tapçi, E.; Ocal, R.; Inan, L.E.; Durak, İ.; Yoldaş, T.K. Decreased antioxidant status in migraine patients with brain white matter hyperintensities. Neurol. Sci. 2014, 35, 1925–1929. [Google Scholar] [CrossRef]
- Ferrari, M.D.; Roon, K.I.; Lipton, R.B.; Goadsby, P.J. Oral triptans (serotonin-5HT1B/1D agonists in acute migraine treatment: A meta-analysis of 53 trials. Lancet 2001, 358, 1668–1675. [Google Scholar] [CrossRef]
- Lipton, R.B.; Silberstein, S.D. Episodic and chronic migraine headache: Breaking down barriers to optimal treatment and prevention. Headache 2015, 55 (Suppl. 2), 103–122. [Google Scholar] [CrossRef]
- Evers, S.; Afra, J.; Frese, A.; Goadsby, P.J.; Linde, M.; May, A.; Sándor, P.S.; European Federation of Neurological Societies. EFNS guideline on the drug treatment of migraine–revised report of an EFNS task force. Eur. J. Neurol. 2009, 16, 968–981. [Google Scholar] [CrossRef]
- Diener, H.C. Headache: Insight, understanding, treatment and patient management. Int. J. Clin. Pract. Suppl. 2013, 178, 33–36. [Google Scholar] [CrossRef]
- Rajapakse, T.; Davenport, W.J. Phytomedicines in the treatment of migraine. CNS Drugs 2019, 33, 399–415. [Google Scholar] [CrossRef]
- Orr, S.L. The evidence for the role of nutraceuticals in the menagement of pediatric migraine: A review. Curr. Pain Headache Rep. 2018, 22, 37. [Google Scholar] [CrossRef]
- D’Onofrio, F.; Raimo, S.; Spitaleri, D.; Casucci, G.; Bussone, G. Usefulness of nutraceuticals in migraine prophylaxis. Neurol. Sci. 2017, 38, S117–S120. [Google Scholar] [CrossRef]
- Ferroni, P.; Barbanti, P.; Della-Morte, D.; Palmirotta, R.; Jirillo, E.; Guadagni, F. Redox Mechanisms in Migraine: Novel Therapeutics and Dietary Interventions. Antioxid. Redox Signal. 2018, 28, 1144–1183. [Google Scholar] [CrossRef]
- Baiano, A.; del Nobile, M.A. Antioxidant compounds from vegetable matrices: Biosynthesis, occurrence, and extraction systems. Crit. Rev. Food Sci. Nutr. 2015, 56, 2053–2068. [Google Scholar] [CrossRef]
- Deng, G.F.; Shen, C.; Xu, X.R.; Kuang, R.D.; Guo, Y.J.; Zeng, L.S.; Gao, L.L.; Lin, X.; Xie, J.F.; Xia, E.Q. Potential of fruit wastes as natural resources of bioactive compounds. Int. J. Mol. Sci. 2012, 13, 8308–8323. [Google Scholar] [CrossRef]
- Carlsen, M.H.; Halversen, B.L.; Holte, K.; Bohn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; et al. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutr. J. 2010, 9, 3. [Google Scholar] [CrossRef]
- Chiaiese, P.; Corrado, G.; Minutolo, M.; Barone, A.; Errico, A. Transcriptional Regulation of Ascorbic Acid During Fruit Ripening in Pepper (Capsicum annuum) Varieties with Low and High Antioxidants Content. Plants 2019, 8, 206. [Google Scholar] [CrossRef]
- Xu, D.P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.J.; Li, H.B. Natural Antioxidants in Foods and Medicinal Plants: Extraction, Assessment and Resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Ramstead, A.G.; Kirpotina, L.N.; Voyich, J.M.; Jutila, M.A.; Quinn, M.T. Therapeutic potential of polyphenols from Epilobium angustifolium (Fireweed). Phytother. Res. 2016, 30, 1287–1297. [Google Scholar] [CrossRef]
- Ravetti, S.; Clemente, C.; Brignone, S.; Hergert, L.; Allemandi, D.; Palma, S. Ascorbic acid in skin health. Cosmetics 2019, 6, 58. [Google Scholar] [CrossRef]
- Du, J.; Cullen, J.J.; Buettner, G.R. Ascorbic acid: Chemistry, biology and the treatment of cancer. Biochim. Biophys. Acta 2012, 1826, 443–457. [Google Scholar] [CrossRef]
- Covarrubias-Pinto, A.; Acuña, A.I.; Beltrán, F.A.; Torres-Díaz, L.; Castro, M.A. Old Things New View: Ascorbic Acid Protects the Brain in Neurodegenerative Disorders. Int. J. Mol. Sci. 2015, 16, 28194–28217. [Google Scholar] [CrossRef]
- Mandl, J.; Szarka, A.; Banhegyi, E. Vitamin C: Update on physiology and pharmacology. Br. J. Pharmacol. 2009, 157, 1097–1110. [Google Scholar] [CrossRef]
- Shaik, M.M.; Gan, S.H. Vitamin Supplementation as Possible Prophylactic Treatment against Migraine with Aura and Menstrual Migraine. Biomed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef]
- Kim, M.S.; Kim, D.J.; Na, C.H.; Shin, B.S. A study of intravenous administartion of vitamin C in the treatment of acute herpetic pain and postherpetic neuralgia. Ann. Dermatol. 2016, 28, 677–683. [Google Scholar] [CrossRef]
- Chayasirisobohon, S. Use of a pink bark extract and antioxidant vitamin combination product as therapy for migraine in patients refractory to pharmacologic medication. Headache 2006, 46, 788–793. [Google Scholar] [CrossRef]
- Chayasirisobhon, S. Efficiacy of Pinus radiata bark extract and vitamin combination product as a prophylactic therapy for recalcitrant migraine and long-term results. Acta Neurol. 2013, 22, 13–21. [Google Scholar]
- Kunnumakkara, A.B.; Bordoloi, D.; Padmavathi, G.; Monisha, J.; Roy, N.K.; Prasad, S.; Aggarwal, B.B. Curcumin, the golden nutraceutical: Multitargeting for multiple chronic diseases. Br. J. Pharmacol. 2017, 174, 1325–1348. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Kumar, A.; Bharti, A.C. Anticancer potential of curcumin: Preclinical and clinical studies. Anticancer Res. 2003, 23, 363–398. [Google Scholar]
- Xu, X.Y.; Meng, X.; Li, S.; Gan, R.Y.; Li, Y.; Li, H.B. Bioactivity, Health Benefits, and Related Molecular Mechanisms of Curcumin: Current Progress, Challenges, and Perspectives. Nutrients 2018, 10, 1553. [Google Scholar] [CrossRef]
- Lee, W.H.; Loo, C.Y.; Bebawy, M.; Luk, F.; Mason, R.S.; Rohanizadeh, R. Curcumin and its derivatives: Their application in neuropharmacology and neuroscience in the 21st century. Curr. Neuropharmacol. 2013, 11, 338–378. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Li, J.B.; Zhao, Y.L.; Li, Y.P.; Yin, L. Investigating the therapeutic potential and mechanism of curcumin in breast cancer based on RNA sequencing and bioinformatics analysis. Breast Cancer 2018, 25, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Cole, G.M.; Teter, B.; Frautschy, S.A. Neuroprotective effects of curcumin. In The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease, Advances in Experimental Medicine and Biology; Aggarwal, B.B., Surh, Y.J., Shishodia, S., Eds.; Springer: Boston, MA, USA, 2007; Volume 595. [Google Scholar]
- Novak Kujundžić, R.; Stepanić, V.; Milković, L.; Čipak Gašparović, A.; Tomljanović, M.; Gall Trošelj, K. Curcumin and its Potential for Systemic Targeting of Inflamm-Aging and Metabolic Reprogramming in Cancer. Int. J. Mol. Sci. 2019, 20, 1180. [Google Scholar] [CrossRef] [PubMed]
- Pulido-Moran, M.; Moreno-Fernandez, J.; Ramirez-Tortosa, C.; Ramirez-Tortosa, C. Curcumin and Health. Molecules 2016, 21, 264. [Google Scholar] [CrossRef]
- Bulboacă, A.E.; Bolboacă, S.D.; Stănescu, I.C.; Sfrângeu, C.A.; Porfire, A.; Tefas, L.; Bulboacă, A.C. The effect of intravenous administration of liposomal curcumin in addition to sumatriptan treatment in an experimental migraine model in rats. Int. J. Nanomed. 2018, 13, 3093–3103. [Google Scholar] [CrossRef]
- Bulboacă, A.E.; Bolboacă, S.D.; Bulboacă, A.C.; Porfire, A.S.; Tefas, L.R.; Suciu, S.M.; Dogaru, G.; Stănescu, I.C. Liposomal Curcumin Enhances the Effect of Naproxen in a Rat Model of Migraine. Med. Sci. Monit. 2019, 25, 5087–5097. [Google Scholar] [CrossRef]
- Bulboacă, A.E.; Bolboacă, S.D.; Stănescu, I.C.; Sfrângeu, C.A.; Bulboacă, A.C. Preemptive Analgesic and Antioxidative Effect of Curcumin for Experimental Migraine. Biomed. Res. Int. 2017, 2017. [Google Scholar] [CrossRef]
- Abdolahi, M.; Tafakhori, A.; Togha, M.; Okhovat, A.A.; Siassi, F.; Eshraghian, M.R.; Sedighiyan, M.; Djalali, M.; Mohammadzadeh Honarvar, N.; Djalali, M. The synergistic effects of ω-3 fatty acids and nano-curcumin supplementation on tumor necrosis factor (TNF)-α gene expression and serum level in migraine patients. Immunogenetics 2017, 69, 371–378. [Google Scholar] [CrossRef]
- Parohan, M.; Sarraf, P.; Javanbakht, M.H.; Foroushani, A.R.; Ranji-Burachaloo, S.; Djalali, M. The synergistic effects of nano-curcumin and coenzyme Q10 supplementation in migraine prophylaxis: A randomized, placebo-controlled, double-blind trial. Nutr. Neurosci. 2019, 26, 1–10. [Google Scholar] [CrossRef]
- Mancuso, M.; Orsucci, D.; Calsolaro, V.; Choub, A.; Siciliano, G. Coenzyme Q10 and Neurological Diseases. Pharmaceuticals 2009, 2, 134–149. [Google Scholar] [CrossRef]
- Liu, H.T.; Cheng, S.B.; Huang, Y.C.; Huang, Y.T.; Lin, P.T. Coenzyme Q10 and Oxidative Stress: Inflammation Status in Hepatocellular Carcinoma Patients after Surgery. Nutrients 2017, 9, 29. [Google Scholar] [CrossRef] [PubMed]
- Mantle, D.; Hargreaves, I. Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview. Antioxidants 2019, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, I.P. Coenzyme Q10 as a therapy for mitochondrial disease. Int. J. Biochem. Cell Biol. 2014, 49, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Moccia, M.; Capacchione, A.; Lanzillo, R.; Carbone, F.; Micillo, T.; Matarese, G.; Palladino, R.; Brescia, V. Sample Size for Oxidative Stress and Inflammation When Treating Multiple Sclerosis with Interferon-β1a and Coenzyme Q10. Brain Sci. 2019, 9, 259. [Google Scholar] [CrossRef]
- Rajapakse, T.; Pringsheim, T. Nutraceuticals in migraine: A summary of existing guidelines for use. Headache 2016, 56, 808–816. [Google Scholar] [CrossRef]
- Sawaddiruk, P.; Apaijai, N.; Paiboonworachat, S.; Kaewchur, T.; Kasitanon, N.; Jaiwongkan, T.; Kerdphoo, S.; Chatipakorn, N.; Chattipakorn, S.C. Coenzyme Q10 supplementation alleviates pain in pregabalin-treated fibromyalgia patients via reducing brain activity and mitochondrial dysfunction. Free Radic. Res. 2019, 53, 901–909. [Google Scholar] [CrossRef]
- Gaul, C.; Diener, H.C.; Danesch, U.; the Migravent® Study Group. Improvement of migraine symptoms with a proprietary supplement containing riboflavin, magnesium and Q10: A randomized, placebo-controlled, double-blind, multicenter trial. J. Headache Pain 2015, 16, 32. [Google Scholar] [CrossRef]
- Schwedt, T.J. Preventive Therapy of Migraine. Continuum (Minneap Minn). Headache 2018, 24, 1052–1065. [Google Scholar] [CrossRef]
- Dahri, M.; Tarighat-Esfajani, A.; Asghari-Jafarabadi, M.; Hashemilar, M. Oral coenzyme Q10 supplementation in patients with migraine effects on clinical features and inflammation markers. Nutr. Neurosci. 2019, 22, 607–615. [Google Scholar] [CrossRef]
- Hershey, A.D.; Powers, S.W.; Vockell, A.L.; Lecates, S.L.; Ellinor, P.L.; Segers, A.; Burdine, D.; Manning, P.; Kabbouche, M.A. Coenzyme Q10 deficiency and response to supplementation in pediatric and adolescent migraine. Headache 2007, 47, 73–80. [Google Scholar] [CrossRef]
- Zeng, Z.; Li, Y.P.; Lu, S.Y.; Huang, W.S.; Di, W. Efficiacy of CoQ10 as a supplementation for migraine: A meta-analysis. Acta Neurol. Scand. 2019, 139, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Slater, S.K.; Nelson, T.D.; Kabbouche, M.A.; LeCates, S.L.; Horn, P.; Segers, A.; Manning, P.; Powers, S.W.; Hershey, A.D. A randomized double-blinded, placebo-controlled, crossover, add-on study of Coenzyme Q10 in the prevention of pediatric and adolescent migraine. Cephalgia 2011, 331, 897–905. [Google Scholar] [CrossRef] [PubMed]
- Guilbot, A.; Bangratz, M.; Ait Abdellah, S.; Lukas, C. A combination of coenzyme Q10, feverfew and magnesium for migraine prophylaxis: A prospective observational study. Complement. Altern. Med. 2017, 17, 433. [Google Scholar] [CrossRef] [PubMed]
- Shoeibi, A.; Olfati, N.; Salehi, M.; Mali, S.; Oryani, M.A. Effectiveness of coenzyme Q10 in prophylactic treatment of migraine headache: An open-label, add-on, controlled trial. Acta Neurol. Belg. 2017, 117, 103–109. [Google Scholar] [CrossRef]
- Kim, M.S.; Bang, J.H.; Lee, J.; Han, J.S.; Baik, T.G.; Jeon, W.K. Ginkgo biloba L. extract protects against chronic cerebral hypoperfusion by modulating neuroinflammation and the cholinergic system. Phytomedicine 2016, 23, 1356–1364. [Google Scholar] [CrossRef]
- Shah, Z.A.; Nada, S.E.; Dore, S. Heme oxygenase 1, beneficial role in permnent ischemic stroke and in Ginkgo Biloba (EGB761) neuroprotection. Neuroscience 2011, 180, 248–255. [Google Scholar] [CrossRef]
- Zhang, Z.; Peng, D.; Zhu, H.; Wang, X. Experimental evidence of Ginkgo biloba extract EGB as a neuroprotective agent in ischemia stroke rats. Brain Res. Bull. 2012, 87, 193–198. [Google Scholar] [CrossRef]
- Tulsulkar, J.; Shah, Z.A. Ginkgo biloba prevents transient global ischemia-induced delayed hippocampal neuronal death through antioxidant and anti-inflammatory mechanism. Neurochem. Int. 2013, 62, 189–197. [Google Scholar] [CrossRef]
- Choi, M.S.; Kim, J.K.; Kim, D.H.; Yoo, H.H. Effects of gut microbiota on the bioavailability of bioactive compounds from ginkgo leaf extracts. Metabolites 2019, 9, 132. [Google Scholar] [CrossRef]
- Shi, C.; Zhao, L.; Zhu, B.; Li, Q.; Yew, D.T.; Yao, Z.; Xu, J. Protective effects of Ginkgo biloba extract (EGb761) and its constituents quercetin and ginkgolide B against β-amyloid peptide-induced toxicity in SH-SY5Y cells. Chem. Biol. Interact. 2009, 181, 115–123. [Google Scholar] [CrossRef]
- D’Andrea, G.; Cevoli, S.; Cologno, D. Herbal therapy in migraine. Neurol. Sci. 2014, 35 (Suppl. 1), S135–S140. [Google Scholar] [CrossRef]
- Li, R.; Chen, B.; Wu, W.; Bao, L.; Li, J.; Qi, R. Ginkgolide B suppresses intercellular adhesion molecule-1 expression via blocking nuclear factor-kB activation in human vascular endothelial cells stimulated by oxidized low-density lipoprotein. J. Pharmacol. Sci. 2009, 110, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Liu, X.; Zhao, Y.; Chen, B.; Li, X.; Qi, R. Ginkgolide B reduces LOX-1 expression by inhibiting Akt phosphorylation and increasing Sirt1 expression in oxidized LDL-stimulated human umbilical vein endothelial cells. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, G.; Bussone, G.; Allais, G.; Aggugia, M.; D’Onofrio, F.; Maggio, M.; Moschiano, F.; Saracco, M.G.; Terzi, M.G.; Petretta, V.; et al. Efficiacy of ginkgolide B in the prophylaxis of migraine with aura. Neurol. Sci. 2009, 30 (Suppl. 1), S121–S124. [Google Scholar] [CrossRef]
- Wu, C.; Chen, F.; Wang, X.; Kim, H.J.; He, G.Q.; Haley-Zitlin, V.; Huang, G. Antioxidant constituents in feverfew (Tanacetu Parthenium) extract and their chromatographic quantification. Food Chem. 2006, 96, 220–227. [Google Scholar] [CrossRef]
- Pareek, A.; Suthar, M.; Rathore, G.S.; Bansal, V. Feverfew (Tanacetum Parthenium L.): A systematic review. Pharmacogn. Rev. 2011, 5, 103–110. [Google Scholar] [CrossRef]
- Hwang, S.H.; Kim, H.Y.; Quispe, Y.N.G.; Wang, Z.; Zuo, G.; Lim, S.S. Aldose Reductase, Protein Glycation Inhibitory and Antioxidant of Peruvian Medicinal Plants: The Case of Tanacetum parthenium L. and Its Constituents. Molecules 2019, 24, 2010. [Google Scholar] [CrossRef]
- Amir, L.H.; Pirotta, M.V.; Raval, M. Breastfeeding—Evidence based guidelines for the use of medicines. Aust. Fam. Physician 2011, 40, 684–690. [Google Scholar]
- Sprenger, T.; Viana, M.; Tassorelli, C. Current prophylactic medications for migraine and their potential mechanisms of action. Neurotherapeutics 2018, 15, 313–323. [Google Scholar] [CrossRef]
- Pfaffenrath, V.; Diener, H.C.; Fischer, M.; Friede, M.; Henneicke-von Zepelin, H.H.; Investigators. The efficacy and safety of Tanacetum parthenium (feverfew) in migraine prophylaxis—A double-blind, multicentre, randomized placebo-controlled dose-response study. Cephalgia 2002, 7, 523–532. [Google Scholar] [CrossRef]
- Diener, H.C.; Pfaffenrath, V.; Schnitker, J.; Friede, M.; Henneicke-von Zepelin, H.H. Efficacy and Safety of 6.25 mg t.i.d. Feverfew CO2-Extract (MIG-99) in Migraine Prevention—A Randomized, Double-Blind, Multicentre, Placebo-Controlled Study. Cephalgia 2005, 25, 1031–1041. [Google Scholar] [CrossRef] [PubMed]
- Volta, G.D.; Zavarise, P.; Perego, L.; Savi, L.; Pezzini, A. Comparison of the Effect of Tanacethum Parthenium, 5-Hydroxy Tryptophan, and Magnesium (Aurastop) versus Magnesium Alone on Aura Phenomenon and Its Evolution. Pain Res. Manag. 2019. [Google Scholar] [CrossRef] [PubMed]
- Pittler, M.H.; Ernst, E. Feverfew for preventing migraine. Cochrane Database Syst. Rev. 2004, 1, CD002286. [Google Scholar]
- Daniel, O.; Mauskop, A. Nutraceuticals in acute and prophylactic treatment of migraine. Curr. Treat. Options Neurol. 2016, 18, 14. [Google Scholar] [CrossRef]
| No. | Authors | Number of Patients | Migraine | Results |
|---|---|---|---|---|
| 1. | Tozzi-Ciancarelli et al. [31] | 23 | With aura | Increased concentration of substances reacting with thiobarbituric acid during attack-free periods |
| 2. | Alp et al. [32] | 75 | Without aura | Decrease in total antioxidant status (TAS) Increase in total oxidant status (TOS) Increase in oxidative stress index (OSI) During attack-free period |
| 3. | Geyik et al. [33] | 50 | With- and without aura | Lack of differences in TOS, TAS, and OSI values between the migraineurs and the control group No differences in TOS, TAS, and OSI values between the groups of migraineurs (with and without aura) Higher 8-OHdG plasma concentration in the migraine patients Higher 8-OHdG plasma concentration in patients with migraine without aura in comparison with the group of patients with migraine with aura |
| 4. | Yigit et al. [34] | 40 | Without aura | Increase in lymphocyte DNA-damage Elevated values of TOS and OSI in migraineurs Elevated MDA concentration in plasma of migraineurs Decreased concentration of UTS2R in plasma of migraineurs Lower TAS level and decrease in CAT activity in migraineurs |
| 5. | Aytaç et al. [35] | 32 (18 with white matter hyperintensities and 14 without white matter hyperintensities) –WHM) | migraine with or without WHM | Decreased CAT activity and increased plasma MDA concentration in migraineurs Decreased CAT activity in migraine patients with WHM-type lesions in comparison with migraine patients without WHM-type lesions Elevated MDA concentration in migraine patients with WHM-type lesions in comparison with migraine patients without WHM-type lesions |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goschorska, M.; Gutowska, I.; Baranowska-Bosiacka, I.; Barczak, K.; Chlubek, D. The Use of Antioxidants in the Treatment of Migraine. Antioxidants 2020, 9, 116. https://doi.org/10.3390/antiox9020116
Goschorska M, Gutowska I, Baranowska-Bosiacka I, Barczak K, Chlubek D. The Use of Antioxidants in the Treatment of Migraine. Antioxidants. 2020; 9(2):116. https://doi.org/10.3390/antiox9020116
Chicago/Turabian StyleGoschorska, Marta, Izabela Gutowska, Irena Baranowska-Bosiacka, Katarzyna Barczak, and Dariusz Chlubek. 2020. "The Use of Antioxidants in the Treatment of Migraine" Antioxidants 9, no. 2: 116. https://doi.org/10.3390/antiox9020116
APA StyleGoschorska, M., Gutowska, I., Baranowska-Bosiacka, I., Barczak, K., & Chlubek, D. (2020). The Use of Antioxidants in the Treatment of Migraine. Antioxidants, 9(2), 116. https://doi.org/10.3390/antiox9020116





