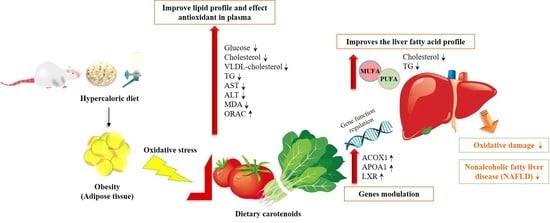
Consumption of Spinach and Tomato Modifies Lipid Metabolism, Reducing Hepatic Steatosis in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Obtaining Spinach and Tomato and Preparation of Experimental Diets
2.2. Chemical Composition of Experimental Diets
2.3. Animals and Experimental Design
2.4. Histopathological Examination
2.5. Plasma Biochemical Parameters
2.6. Determination of Biomarkers of Oxidative Stress
2.7. Carotenoid Analysis in Diets, Liver, Feces and Plasma
2.8. Lipid Analysis in the Liver
2.9. Study of the Expression of Genes Involved in Fatty Liver Metabolism
2.10. Western Blot
2.11. Statistical Analysis
3. Results
3.1. Daily Intake of Feed and Biometric Parameters
3.2. Histopathological Examination and Biochemical Parameters
3.3. Carotenoid Content in the Liver
3.4. Total Fat, Cholesterol, Triglyceride and Fatty Acid Content in the Liver
3.5. Identification of the Genes Related to NAFLD
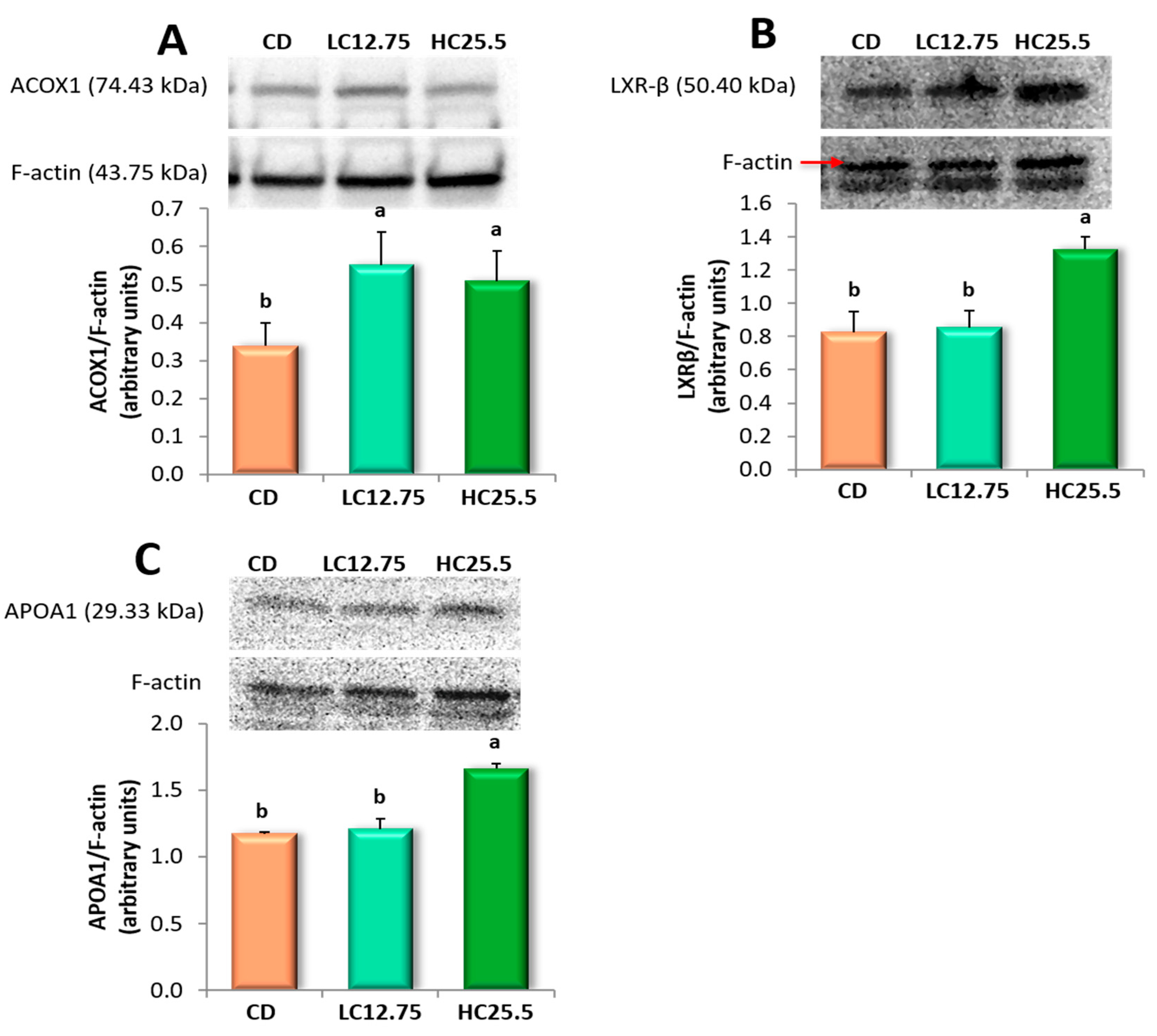
3.6. ACOX1, LXRβ and APOA1 Protein Expressions in the Liver
4. Discussion
4.1. Carotenoid Intake and its Action on Steatosis Biomarkers
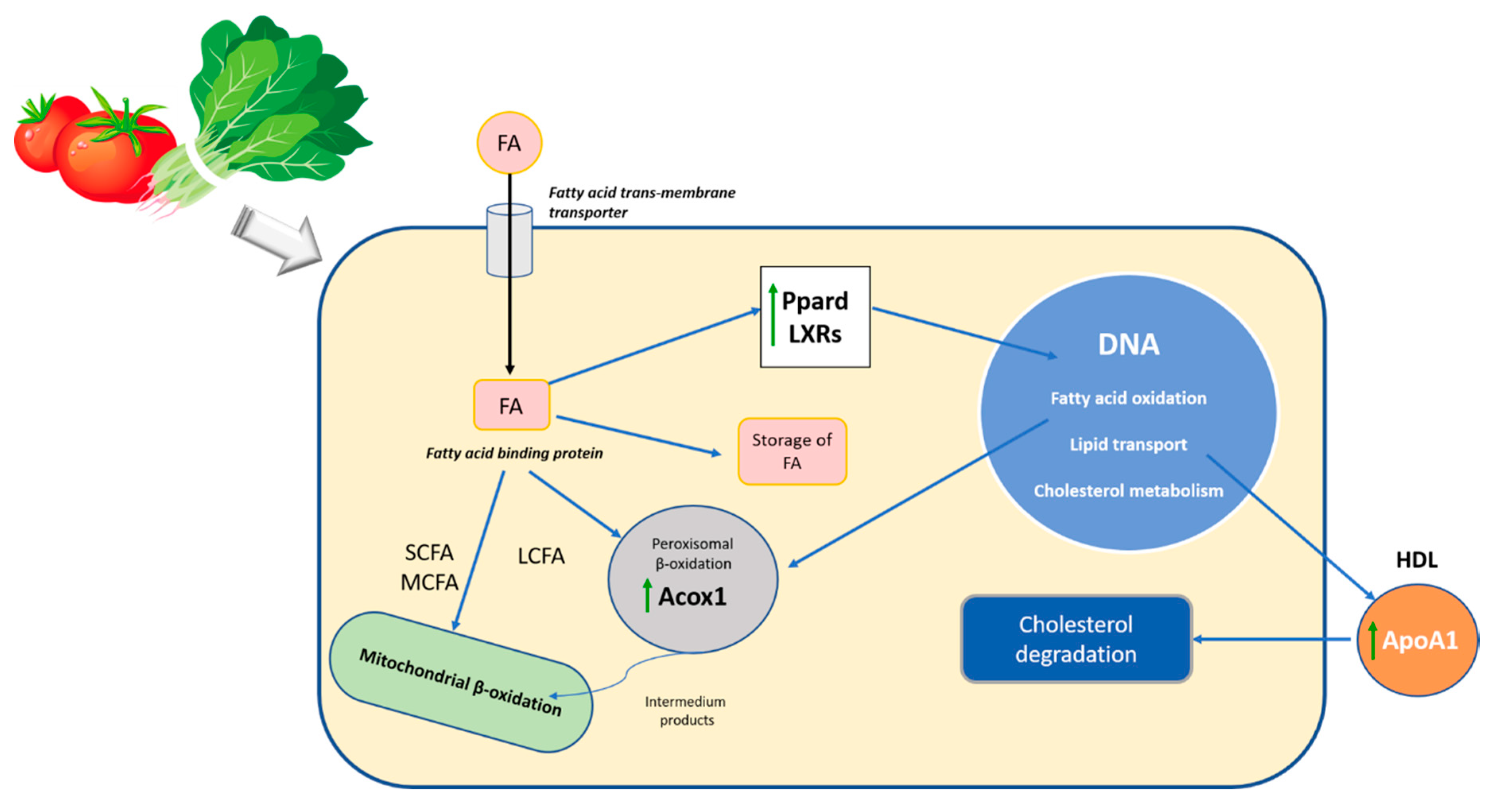
4.2. Bioactive Components of Spinach and Tomato Modulate Gene Expression Related to Steatosis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NF-κB | Nuclear factor kappa B |
| MAPK | Mitogen-activated protein kinase |
| HDL | High-density lipoprotein |
| LDL | Low-density lipoproteins |
| VLDL | Very low-density lipoprotein |
References
- Younossi, Z.; Anstee, Q.M.; Marietti, M.; Hardy, T.; Henry, L.; Eslam, M.; Getorge, J.; Bugialnesi, E. Global burden of NAFLD and NASH: Trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Calzadilla Bertot, L.; Adams, L.A. The natural course of non-alcoholic fatty liver disease. Int. J. Mol. Sci. 2016, 17, 774. [Google Scholar] [CrossRef] [PubMed]
- Mencin, A.A.; Lavine, J.E. Nonalcoholic fatty liver disease in children. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Bataller, R. Alcoholic liver disease: Pathogenesis and new therapeutic targets. Gastroenterology 2011, 141, 1572–1585. [Google Scholar] [CrossRef]
- Yilmaz, B.; Sahin, K.; Bilen, H.; Bahcecioglu, I.H.; Bilir, B.; Ashraf, S.; Halazun, K.J.; Kucuk, O. Carotenoids and non-alcoholic fatty liver disease. Hepatobiliary Surg. Nutr. 2015, 4, 161–171. [Google Scholar]
- Murillo, A.G.; DiMarco, D.M.; Fernandez, M.L. The potential of non-provitamin a carotenoids for the prevention and treatment of non-alcoholic fatty liver disease. Biology 2016, 5, 42. [Google Scholar] [CrossRef] [PubMed]
- Lan, Q.Y.; Zhang, Y.J.; Liao, G.C.; Zhou, R.F.; Zhou, Z.G.; Chen, Y.M.; Zhu, H.L. The association between dietary vitamin A and carotenes and the risk of primary liver cancer: A case-control study. Nutrients 2016, 8, 624. [Google Scholar] [CrossRef]
- Wang, L.; Ding, C.; Zeng, F.; Zhu, H. Low levels of serum β-carotene and β-carotene/retinol ratio are associated with histological severity in nonalcoholic fatty liver disease patients. Ann. Nutr. Metab. 2019, 74, 156–164. [Google Scholar] [CrossRef]
- Maiani, G.; Periago Castón, M.J.; Catasta, G.; Toti, E.; Cambrodón, I.G.; Bysted, A.; Granado-Lorencio, F.; Olmedilla-Alonso, B.; Knuthsen, P.; Valoti, M.; et al. Carotenoids: Actual knowledge on food sources, intakes, stability and bioavailability and their protective role in humans. Mol. Nutr. Food Res. 2009, 53, S194–S218. [Google Scholar] [CrossRef]
- Bernal, C.; Martín-Pozuelo, G.; Lozano, A.B.; Sevilla, A.; García-Alonso, J.; Canovas, M.; Periago, M.J. Lipid biomarkers and metabolic effects of lycopene from tomato juice on liver of rats with induced hepatic steatosis. J. Nutr. Biochem. 2013, 24, 1870–1881. [Google Scholar] [CrossRef]
- Martín-Pozuelo, G.; Navarro-González, I.; González-Barrio, R.; Santaella, M.; García-Alonso, J.; Hidalgo, N.; Gómez-Gallego, C.; Ros, G.; Periago, M.J. The effect of tomato juice supplementation on biomarkers and gene expression related to lipid metabolism in rats with induced hepatic steatosis. Eur. J. Nutr. 2015, 54, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Elvira-Torales, L.I.; Navarro-González, I.; González-Barrio, R.; Martín-Pozuelo, G.; Doménech, G.; Seva, J.; García-Alonso, J.; Periago-Castón, M.J. Tomato juice supplementation influences the gene expression related to steatosis in rats. Nutrients 2018, 10, 1215. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, B.; Miyake, T.; Yamamoto, S.; Furukawa, S.; Hiasa, Y. Usefulness of Beta-cryptoxanthin for nonalcoholic fatty liver diseases. J. Food Nutr. Disord. 2016, 5, 1000196. [Google Scholar]
- Elvira-Torales, L.I.; Martín-Pozuelo, G.; González-Barrio, R.; Navarro-González, I.; Pallarés, F.J.; Santaella, M.; García-Alonso, J.; Sevilla, Á.; Periago-Castón, M.J. Ameliorative effect of spinach on non-alcoholic fatty liver disease induced in rats by a high-fat diet. Int. J. Mol. Sci. 2019, 20, 1662. [Google Scholar] [CrossRef]
- Kim, J.E.; Clark, R.M.; Park, Y.; Lee, J.; Fernandez, M.L. Lutein decreases oxidative stress and inflammation in liver and eyes of guinea pigs fed a hypercholesterolemic diet. Nutr. Res. Pract. 2012, 6, 113–119. [Google Scholar] [CrossRef]
- Qiu, X.; Gao, D.H.; Xiang, X.; Xiong, Y.F.; Zhu, T.S.; Liu, L.G.; Sun, X.-F.; Hao, L.-P. Ameliorative effects of lutein on non-alcoholic fatty liver disease in rats. World J. Gastroenterol. 2015, 21, 8061–8072. [Google Scholar] [CrossRef]
- Chamberlain, S.M.; Hall, J.D.; Patel, J.; Lee, J.R.; Marcus, D.M.; Sridhar, S.; Romero, M.; Labazi, M.; Caldwell, R.W.; Bartoli, M. Protective effects of the carotenoid zeaxanthin in experimental nonalcoholic steatohepatitis. Dig. Dis. Sci. 2009, 54, 1460–1464. [Google Scholar] [CrossRef]
- Xiao, J.; Liong, E.C.; Ching, Y.P.; Chang, R.C.C.; Fung, M.L.; Xu, A.M.; So, K.F.; Tipoe, G.L. Lycium barbarum polysaccharides protect rat liver from non-alcoholic steatohepatitis-induced injury. Nutr. Diabetes 2013, 3, e81. [Google Scholar] [CrossRef]
- Padmore, J.M. Animal Feed. In Official Methods of Analysis of AOAC, 15th ed.; Helrich, K., Ed.; Association of Official Analytical Chemists, Inc.: Arlington, VA, USA, 1990; Volume 1, pp. 70–79. [Google Scholar]
- Prosky, L.; Asp, N.G.; Furda, I.; DeVries, J.W.; Schweizer, T.F.; Harland, B.F. Determination of total dietary fiber in foods and food products: Collaborative study. J. Assoc. Off. Anal. Chem. 1985, 68, 677–679. [Google Scholar] [CrossRef]
- Hirawan, R.; Diehl-Jones, W.; Beta, T. Comparative evaluation of the antioxidant potential of infant cereals produced from purple wheat and red rice grains and LC-MS analysis of their anthocyanins. J. Agric. Food Chem. 2011, 59, 12330–12341. [Google Scholar] [CrossRef]
- Jarukamjorn, K.; Jearapong, N.; Pimson, C.; Chatuphonprasert, W. A high-fat, high-fructose diet induces antioxidant imbalance and increases the risk and progression of nonalcoholic fatty liver disease in mice. Scientifica (Cairo) 2016, 2016, 5029414. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.; Abdelmalek, M.F.; Sullivan, S.; Nadeau, K.J.; Green, M.; Roncal, C.; Nakagawa, T.; Kuwabara, M.; Sato, Y.; Kang, D.-H.; et al. Fructose and sugar: A major mediator of non-alcoholic fatty liver disease. J. Hepatol. 2018, 68, 1063–1075. [Google Scholar] [CrossRef]
- Brunt, E.M.; Janney, C.G.; Di Bisceglie, A.M.; Neuschwander-Tetri, B.A.; Bacon, B.R. Nonalcoholic steatohepatitis: A proposal for grading and staging the histological lesions. Am. J. Gastroenterol. 1999, 94, 2467–2474. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Hoang, H.A.; Gu, L.; Wu, X.; Bacchiocca, M.; Howard, L.; Hampsch-Woodill, M.; Huang, D.; Ou, A.B.; Jacob, R. Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORACFL)) of plasma and other biological and food samples. J. Agric. Food Chem. 2003, 51, 3273–3279. [Google Scholar] [CrossRef]
- Mateos, R.; Lecumberri, E.; Ramos, S.; Goya, L.; Bravo, L. Determination of malondialdehyde (MDA) by high-performance liquid chromatography in serum and liver as a biomarker for oxidative stress. Application to a rat model for hypercholesterolemia and evaluation of the effect of diets rich in phenolic antioxidants from fruits. J. Chromatogr. B 2005, 827, 76–82. [Google Scholar]
- Bensadoun, A.; Weinstein, D. Assay of proteins in the presence of interfering materials. Anal. Biochem. 1976, 70, 241–250. [Google Scholar] [CrossRef]
- Helger, R.; Rindfrey, H.; Hilgenfeldt, J. Direct estimation of creatinine in serum and in urine without deproteinization using a modified Jaffé method. Z. Klin. Chem. Klin. Biochem. 1974, 12, 344–349. [Google Scholar]
- Seybold, C.; Fröhlich, K.; Bitsch, R.; Otto, K.; Böhm, V. Changes in contents of carotenoids and vitamin E during tomato processing. J. Agric. Food Chem. 2004, 52, 7005–7010. [Google Scholar] [CrossRef]
- Arranz, S.; Martínez-Huélamo, M.; Vallverdu-Queralt, A.; Valderas-Martínez, P.; Illán, M.; Sacanella, E.; Escribano, E.; Estruch, R.; Lamuela-Raventos, R.M. Influence of olive on carotenoids adsorption from tomato juice and effects on postpandrial lipemia. Food Chem. 2015, 1, 168–203. [Google Scholar]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- León-Goñi, A.C.; Blanco, D.; Peña, A.; Ronda, M.; González, B.O.; Arteaga, M.E. Hematological and biochemical parameters in Sprague Dawley laboratory rats breed in CENPALAB, Cenp: SPRD. Rev. Electron. Vet. 2011, 12, 1–10. [Google Scholar]
- Aguilar, F.; Autrup, H.; Barlow, S.; Castle, L.; Crebelli, R.; Dekant, W. Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food on request from the Commission on the safety in use of lycopene as a food colour. EFSA J. 2008, 6, 674. [Google Scholar] [CrossRef]
- Bampidis, V.; Azimonti, G.; Bastos, M.L.; Christensen, H.; Dusemund, B.; Kouba, M. EFSA FEEDAP Panel (EFSA Panel on Additives and Products or Substances used in Animal Feed. Scientific Opinion on the safety and efficacy of lutein and lutein/zeaxanthin extracts from Tagetes erecta for poultry for fattening and laying (except turkeys). EFSA J. 2019, 17, 5698. [Google Scholar] [CrossRef]
- Tuomilehto, J.; Lindström, J.; Eriksson, J.G.; Valle, T.T.; Hämäläinen, H.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A.; Rastas, M.; et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med. 2001, 344, 1343–1350. [Google Scholar] [CrossRef]
- Noureddin, M.; Rinella, M.E. Nonalcoholic Fatty liver disease, diabetes, obesity, and hepatocellular carcinoma. Clin. Liver Dis. 2015, 19, 361–379. [Google Scholar] [CrossRef]
- Mantovani, A.; Byrne, C.D.; Bonora, E.; Targher, G. Nonalcoholic fatty liver disease and risk of incident type 2 diabetes: A meta-analysis. Diabetes Care 2018, 41, 372–382. [Google Scholar] [CrossRef]
- Kim, J.S.; Ha, T.Y.; Kim, S.; Lee, S.J.; Ahn, J. Red paprika (Capsicum annuum L.) and its main carotenoid capsanthin ameliorate impaired lipid metabolism in the liver and adipose tissue of high-fat diet-induced obese mice. J. Funct. Foods 2017, 31, 131–140. [Google Scholar] [CrossRef]
- Jiang, W.; Guo, M.H.; Hai, X. Hepatoprotective and antioxidant effects of lycopene on non-alcoholic fatty liver disease in rat. World J. Gastroenterol. 2016, 22, 10180–10188. [Google Scholar] [CrossRef]
- Navarro-González, I.; Pérez-Sánchez, H.; Martín-Pozuelo, G.; García-Alonso, J.; Periago, M.J. The inhibitory effects of bioactive compounds of tomato juice binding to hepatic HMGCR: In vivo study and molecular modelling. PLoS ONE 2014, 9, e83968. [Google Scholar] [CrossRef] [PubMed]
- Alvi, S.S.; Iqbal, D.; Ahmad, S.; Khan, M.S. Molecular rationale delineating the role of lycopene as a potent HMG-CoA reductase inhibitor: In vitro and in silico study. Nat. Prod. Res. 2016, 30, 2111–2114. [Google Scholar] [CrossRef]
- Luvizotto, R.A.; Nascimento, A.F.; Imaizumi, E.; Pierine, D.T.; Conde, S.J.; Correa, C.R.; Yeum, K.-J.; Ferreira, A.L.A. Lycopene supplementation modulates plasma concentrations and epididymal adipose tissue mRNA of leptin, resistin and IL-6 in diet-induced obese rats. Br. J. Nutr. 2013, 110, 1803–1809. [Google Scholar] [CrossRef] [PubMed]
- Kusano, C.; Ferrari, B. Total Antioxidant Capacity: A biomarker in biomedical and nutritional studies. J. Cell Mol. Biol. 2008, 7, 1–15. [Google Scholar]
- Sugiura, M.; Ogawa, K.; Yano, M. Comparison of bioavailability between beta-cryptoxanthin and beta-carotene and tissue distribution in its intact form in rats. Biosci. Biotechnol. Biochem. 2014, 78, 307–310. [Google Scholar] [CrossRef]
- Janero, D.R. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med. 1990, 9, 515–540. [Google Scholar] [CrossRef]
- Ozturk, F.; Gul, M.; Ates, B.; Ozturk, I.C.; Cetin, A.; Vardi, N.; Otlu, A.; Yilmaz, I. Protective effect of apricot (Prunus armeniaca L.) on hepatic steatosis and damage induced by carbon tetrachloride in Wistar rats. Br. J. Nutr. 2009, 102, 1767–1775. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Ogawa, K.; Yano, M. Absorption, storage and distribution of β-cryptoxanthin in rat after chronic administration of Satsuma mandarin (Citrus unshiu MARC.) juice. Biol. Pharm. Bull. 2013, 36, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Latief, U.; Ahmad, R. Role of dietary carotenoids in different etiologies of chronic liver diseases. In Descriptive Food Science, 1st ed.; Valero Díaz, A., García-Gimeno, R.M., Eds.; IntechOpen: London, UK, 2018; pp. 93–112. ISBN 9781789845952. [Google Scholar]
- Shimizu, F.; Ogawa, M.; Fukuba, H. Accumulation and excretion of carotenoids after regular ingestion of carrot juice with a lycopene-free diet. J. Home Econ. Jpn. 2006, 57, 151–157. [Google Scholar]
- Palozza, P.; Simone, R.E.; Catalano, A.; Mele, M.C. Tomato lycopene and lung cancer prevention: From experimental to human studies. Cancers (Basel) 2011, 3, 2333–2357. [Google Scholar] [CrossRef]
- Periago, M.J.; Martín-Pozuelo, G.; Gonzalez-Barrio, R.; Santaella, M.; Gómez, V.; Vazquez, N.; Navarro-González, I.; García-Alonso, J. Effect of tomato juice consumption on the plasmatic lipid profile, hepatic HMGCR activity, and fecal short chain fatty acids content of rats. Food Funct. 2016, 7, 4460–4467. [Google Scholar] [CrossRef]
- Vinué, Á.; Herrero-Cervera, A.; González-Navarro, H. Understanding the impact of dietary cholesterol on chronic metabolic diseases through studies in rodent models. Nutrients 2018, 10, 939. [Google Scholar] [CrossRef]
- Piña-Zentella, R.M.; Rosado, J.L.; Gallegos-Corona, M.A.; Madrigal-Pérez, L.A.; García, O.P.; Ramos-Gomez, M. Lycopene improves diet-mediated recuperation in rat model of nonalcoholic fatty liver disease. J. Med. Food. 2016, 19, 607–614. [Google Scholar] [CrossRef]
- Jia, Y.; Wu, C.; Kim, J.; Kim, B.; Lee, S.J. Astaxanthin reduces hepatic lipid accumulations in high-fat-fed C57BL/6J mice via activation of peroxisome proliferator-activated receptor (PPAR) alpha and inhibition of PPAR gamma and Akt. J. Nutr. Biochem. 2016, 28, 9–18. [Google Scholar] [CrossRef]
- Chiang, J.Y. Bile acids: Regulation of synthesis. J. Lipid Res. 2009, 50, 1955–1966. [Google Scholar] [CrossRef]
- Juárez-Hernández, E.; Chávez-Tapia, N.C.; Uribe, M.; Barbero-Becerra, V.J. Role of bioactive fatty acids in nonalcoholic fatty liver disease. Nutr. J. 2016, 15, 72. [Google Scholar] [CrossRef]
- Ferramosca, A.; Zara, V. Modulation of hepatic steatosis by dietary fatty acids. World J. Gastroenterol. 2014, 20, 1746–1755. [Google Scholar] [CrossRef]
- Reyes-Quiroz, M.E.; Alba, G.; Saenz, J.; Santa-María, C.; Geniz, I.; Alba, G.; Ramírez, R.; Martín-Nieto, J.; Pintado, E.; Sobrino, F. Oleic acid modulates mRNA expression of liver X receptor (LXR) and its target genes ABCA1 and SREBP1c in human neutrophils. Eur. J. Nutr. 2014, 53, 1707–1717. [Google Scholar] [CrossRef]
- Pardo, V.; Gonzalez-Rodriguez, A.; Muntane, J.; Kozma, S.C.; Valverde, A.M. Role of hepatocyte S6K1 in palmitic acid-induced endoplasmic reticulum stress, lipotoxicity, insulin resistance and in oleic acid-induced protection. Food Chem. Toxicol. 2015, 80, 298–309. [Google Scholar] [CrossRef]
- Tapia, G.; Valenzuela, R.; Espinosa, A.; Romanque, P.; Dossi, C.; Gonzalez-Mañán, D.; Videla, L.A.; D’Espessailles, A. N-3 long-chain PUFA supplementation prevents high fat diet induced mouse liver steatosis and inflammation in relation to PPAR-α upregulation and NF-κB DNA binding abrogation. Mol. Nutr. Food Res. 2014, 58, 1333–1341. [Google Scholar] [CrossRef]
- Burri, L.; Berge, K.; Wibrand, K.; Berge, R.K.; Barger, J.L. Differential effects of krill oil and fish oil on the hepatic transcriptome in mice. Front. Genet. 2011, 2, 45. [Google Scholar] [CrossRef]
- Sharoni, Y.; Linnewiel-Hermoni, K.; Khanin, M.; Salman, H.; Veprik, A.; Danilenko, M.; Levy, J. Carotenoids and apocarotenoids in cellular signaling related to cancer: A review. Mol. Nutr. Food Res. 2016, 56, 259–269. [Google Scholar] [CrossRef]
- Ford, N.A.; Elsen, A.C.; Erdman, J.W., Jr. Genetic ablation of carotene oxygenases and consumption of lycopene or tomato powder diets modulate carotenoid and lipid metabolism in mice. Nutr. Res. 2013, 33, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.; Jia, Y.; Cao, W.Q.; Kashireddy, P.; Rao, M.S. Expression of peroxisome proliferator-activated receptor alpha, and PPARalpha regulated genes in spontaneously developed hepatocellular carcinomas in fatty acyl-CoA oxidase null mice. Int. J. Oncol. 2002, 21, 1175–1180. [Google Scholar] [PubMed]
- Poll-The, B.T.; Roels, F.; Ogier, H.; Scotto, J.; Vamecq, J.; Schutgens, R.B.; Wanders, R.J.A.; van Roermund, C.W.T.; van Wijland, M.J.A.; Schram, A.W. A new peroxisomal disorder with enlarged peroxisomes and a specific deficiency of acyl-CoA oxidase (pseudo-neonatal adrenoleukodystrophy). Am. J. Hum. Genet. 1988, 42, 422–434. [Google Scholar]
- Thorpe, C.; Kim, J.J. Structure and mechanism of action of the acyl-CoA dehydrogenases. FASEB J. 1995, 9, 718–725. [Google Scholar] [CrossRef]
- Chistiakov, D.A.; Orekhov, A.N.; Bobryshev, Y.V. ApoA1 and ApoA1-specific self-antibodies in cardiovascular disease. Lab. Investig. 2016, 96, 708–718. [Google Scholar] [CrossRef]
- Georgila, K.; Vyrla, D.; Drakos, E. Apolipoprotein A-I (ApoA-I), Immunity, Inflammation and Cancer. Cancers (Basel) 2019, 11, 1097. [Google Scholar] [CrossRef]
- Mustafa, M.G.; Petersen, J.R.; Ju, H.; Cicalese, L.; Synder, N.; Haidacher, S.J.; Denner, L.; Elferink, C. Biomarker discovery for early detection of hepatocellular carcinoma in hepatitis C-infected patients. Mol. Cell. Proteomics 2013, 12, 3640–3652. [Google Scholar] [CrossRef] [PubMed]
- Fye, H.K.; Wright-Drakesmith, C.; Kramer, H.B.; Camey, S.; Nogueira da Costa, A.; Jeng, A.; Bah, A.; Kirk, G.D.; Sharif, M.I.F.; Ladep, N.G.; et al. Protein profiling in hepatocellular carcinoma by label-free quantitative proteomics in two west African populations. PLoS ONE 2013, 8, e68381. [Google Scholar] [CrossRef]
- Ma, X.L.; Gao, X.H.; Gong, Z.J.; Wu, J.; Tian, L.; Zhang, C.Y.; Zhou, Y.; Sun, Y.-F.; Hu, B.; Qiu, S.-J.; et al. Apolipoprotein A1: A novel serum biomarker for predicting the prognosis of hepatocellular carcinoma after curative resection. Oncotarget 2016, 7, 70654–70668. [Google Scholar] [CrossRef]
- Karavia, E.A.; Papachristou, D.J.; Liopeta, K.; Triantaphyllidou, I.E.; Dimitrakopoulos, O.; Kypreos, K.E. Apolipoprotein A-I modulates processes associated with diet-induced nonalcoholic fatty liver disease in mice. Mol. Med. 2012, 18, 901–912. [Google Scholar] [CrossRef]
- Rajavashisth, T.B.; Taylor, A.K.; Andalibi, A.; Svenson, A.J.; Lusis, A.J. Identification of a zinc finger protein that binds to the sterol regulatory element. Science 1989, 245, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Gambino, R.; Cassader, M. Recent insights into hepatic lipid metabolism in non-alcoholic fatty liver disease (NAFLD). Prog. Lipid Res. 2009, 48, 1–26. [Google Scholar] [CrossRef]
- Becares, N.; Gage, M.C.; Voisin, M.; Shrestha, E.; Martin-Gutierrez, L.; Liang, N.; Louie, R.; Pourcet, B.; Pello, O.M.; Luong, T.V.; et al. Impaired LXRα phosphorylation attenuates progression of fatty liver disease. Cell Rep. 2019, 26, 984–995.e6. [Google Scholar] [CrossRef]
- Chen, K.-T.; Pernelle, K.; Tsai, Y.-H.; Wu, Y.-H.; Hsieh, J.-Y.; Liao, K.-H.; Guguen-Guillouzo, C.; Wang, H.-W. Liver X receptor α (LXRα/NR1H3) regulates differentiation of hepatocyte-like cells via reciprocal regulation of HNF4α. J. Hepatol. 2014, 61, 1276–1286. [Google Scholar] [CrossRef]
- Babeu, J.P.; Boudreau, F. Hepatocyte nuclear factor 4-alpha involvement in liver and intestinal inflammatory networks. World J. Gastroenterol. 2014, 20, 22–30. [Google Scholar] [CrossRef] [PubMed]

| Parameters | CD | LC12.75 | HC25.5 |
|---|---|---|---|
| Initial body weight (g) | 554 ± 63.7 | 547 ± 68.4 | 546 ± 82.5 |
| Final body weight (g) | 551 ± 64.1 | 523 ± 49.2 | 506 ± 60.9 |
| Body weight decrease (g) | 2.83 ± 1.94 b | 27.5 ± 10.5 a | 50.1 ± 23.6 a |
| Food intake (g/day) | 14.7 ± 4.81 | 15.0 ± 5.11 | 15.2 ± 3.23 |
| Water intake (mL/day) | 32.4 ± 5.70 | 31.6 ± 1.43 | 32.4 ± 8.66 |
| Excreted feces (g/day) | 2.46 ± 0.69 | 3.05 ± 0.82 | 3.49 ± 0.92 |
| Excreted urine (mL/day) | 14.6 ± 1.13 | 13.9 ± 2.62 | 16.1 ± 5.4 |
| Carotenoid intake (mg/day) | - | 4.00 ± 1.35 b | 8.07 ± 1.71 a |
| Carotenoids excreted in feces (mg/day) | - | 1.80 ± 0.48 b | 3.64 ± 0.96 a |
| Apparent carotenoids absorption (%) | - | 54.9 ± 14.1 | 69.0 ± 13.3 |
| TDF intake (g/day) | 3.11 ± 1.02 | 3.21 ± 1.09 | 3.67 ± 1.03 |
| TPC intake (mg GAE/day) | 32.8 ± 9.8 | 36.7 ± 12.4 | 37.9 ± 10.6 |
| Parameters | CD | LC12.75 | HC25.5 |
|---|---|---|---|
| Glucose (mg/dL) | 205 ± 15.5 a | 156 ± 26.2 b | 140 ± 23.7 b |
| Proteins (g/dL) | 7.17 ± 0.22 a | 6.36 ± 0.33 b | 5.29 ± 0.32 c |
| Total cholesterol (mg/dL) | 111 ± 8.26 a | 90.5 ± 5.61 b | 78.0 ± 8.16 c |
| HDL-cholesterol (mg/dL) | 33.9 ± 3.21 | 30.6 ± 2.46 | 31.7 ± 5.00 |
| LDL-cholesterol (mg/dL) | 13.1 ± 1.29 | 12.3 ± 1.78 | 11.8 ± 1.77 |
| VLDL-cholesterol (mg/dL) | 63.5 ± 8.94 a | 47.5 ± 3.51 b | 35.7 ± 7.36 c |
| Triglycerides (mg/dL) | 81.9 ± 12.4 a | 52.8 ± 14.7 b | 43.1 ± 9.85 b |
| ALT (U/L) | 36.8 ± 8.50 a | 25.4 ± 2.72 b | 21.6 ± 1.99 b |
| AST (U/L) | 112 ± 13.3 a | 69.3 ± 7.27 b | 58.4 ± 3.38 c |
| Lycopene in plasma (μg/mL) | nd | 0.013 ± 0.001 b | 0.016 ± 0.001 a |
| ORAC in plasma (mmoles TE/L) | 7.56 ± 0.33 c | 10.4 ± 0.27 b | 13.1 ± 0.74 a |
| MDA plasma (nmol MDA/mL) | 7.42 ± 0.76 a | 3.88 ± 0.47 b | 3.39 ± 0.44 b |
| MDA liver (nmol MDA/g protein) | 1.05 ± 0.14 a | 0.59 ± 0.16 b | 0.51 ± 0.08 b |
| MDA urine (nmol MDA/mg creatinine) | 14.0 ± 1.75 a | 6.20 ± 2.43 b | 2.97 ± 1.13 c |
| Carotenoids | CD | LC12.75 | HC25.5 |
|---|---|---|---|
| Neoxanthin | nd | 5.58 ± 2.21 b | 10.2 ± 1.73 a |
| Violaxanthin | nd | 3.82 ± 1.54 b | 7.35 ± 1.46 a |
| Lutein | nd | 63.6 ± 8.86 b | 127 ± 19.0 a |
| Zeaxanthin | nd | 9.94 ± 1.15 b | 13.68 ± 1.96 a |
| Phytoene | nd | 66.8 ± 18.4 b | 103 ± 15.3 a |
| α-carotene | nd | 8.82 ± 2.61 b | 28.0 ± 5.46 a |
| β-carotene | nd | 51.2 ± 10.1 b | 76.3 ± 13.7 a |
| Lycopene | nd | 379 ± 108 b | 676 ± 64.9 a |
| Total | nd | 589 ± 142 b | 1043 ± 107 a |
| Parameters | CD | LC12.75 | HC25.5 |
|---|---|---|---|
| Total fat (%) | 12.9 ± 0.12 a | 12.2 ± 0.35 a,b | 11.9 ± 0.00 b |
| Total cholesterol (mg/g) | 8.37 ± 0.65 a | 4.96 ± 0.47 b | 4.01 ± 0.44 c |
| Triglycerides (mg/g) | 13.81 ± 1.47 a | 9.89 ± 1.93 b | 7.43 ± 1.39 c |
| Caprylic acid (C8:0) | 0.013 ± 0.003 a | 0.011 ± 0.004 a | 0.006 ± 0.001 b |
| Capric acid (C10:0) | 0.033 ± 0.002 a | 0.012 ± 0.007 b | 0.007 ± 0.001 b |
| Undecanoic acid (C11:0) | 0.042 ± 0.007 a | 0.019 ± 0.004 b | 0.014 ± 0.001 c |
| Lauric acid (C12:0) | 0.024 ± 0.008 a | 0.014 ± 0.005 a | 0.007 ± 0.002 b |
| Myristic acid (C14:0) | 0.035 ± 0.003 a | 0.020 ± 0.006 b | 0.018 ± 0.002 b |
| Pentadecanoic acid (C15:0) | 0.015 ± 0.001 a | 0.010 ± 0.001 b | 0.010 ± 0.001 b |
| Palmitic acid (C16:0) | 0.038 ± 0.010 a | 0.020 ± 0.004 b | 0.016 ± 0.004 b |
| Margaric acid (C17:0) | 0.020 ± 0.003 a | 0.016 ± 0.002 b | 0.015 ± 0.001 b |
| Stearic acid (C18:0) | 0.810 ± 0.035 a | 0.540 ± 0.045 b | 0.535 ± 0.032 b |
| Arachidic acid (C20:0) | 0.046 ± 0.015 a | 0.026 ± 0.006 b | 0.019 ± 0.005 b |
| Docosanoic acid (C22:0) | 0.048 ± 0.016 a | 0.028 ± 0.005 b | 0.020 ± 0.005 b |
| Tetracosanoic acid (C24:0) | nd | nd | 0.032 ± 0.006 |
| Myristoleic acid (C14:1) | 0.052 ± 0.015 a | 0.022 ± 0.001 b | 0.017 ± 0.004 c |
| Cis Pentadecanoic acid (C15:1) | nd | 0.007 ± 0.001 b | 0.016 ± 0.004 a |
| Palmitoleic acid (C16:1) | 0.116 ± 0.008 a | 0.067 ± 0.008 b | 0.047 ± 0.002 c |
| Cis Heptadecenoic acid (C17:1) | nd | nd | 0.010 ± 0.004 |
| Elaidic acid (C18:1n9t) | 0.106 ± 0.014 b | 0.119 ± 0.010 b | 0.167 ± 0.041 a |
| Oleic acid (C18:1n9c) | 0.372 ± 0.078 c | 0.562 ± 0.090 b | 0.916 ± 0.067 a |
| Eicosenoic acid (C20:1n9) | nd | nd | 0.006 ± 0.001 |
| Nervonic acid (C24:1n9) | 0.036 ± 0.003 b | 0.051 ± 0.009 a | 0.057 ± 0.010 a |
| Linolelaidic acid (C18:2tn-6) | nd | 0.087 ± 0.005 b | 0.096 ± 0.007 a |
| Linoleic acid (C18:2cn-6) | 0.358 ± 0.040 c | 0.526 ± 0.062 b | 0.858 ± 0.021 a |
| γ-Linolenic acid (C18:3n-6) | 0.014 ± 0.001 a | 0.009 ± 0.001 b | 0.009 ± 0.002 b |
| α-Linolenic acid (C18:3n-3) | nd | 0.019 ± 0.003 b | 0.026 ± 0.004 a |
| Eicosadienoic acid (C20:2n-6) | nd | nd | 0.008 ± 0.001 |
| Dihomo-γ-linolenic acid (C20:3n-6) | 0.029 ± 0.006 | 0.022 ± 0.003 | 0.029 ± 0.010 |
| Arachidonic acid (C20:4n-6) | 0.401 ± 0.050 b | 0.540 ± 0.044 a | 0.556 ± 0.054 a |
| Dihomo-α-linolenic acid (C20:3n-3) | nd | 0.012 ± 0.001 b | 0.077 ± 0.002 a |
| Eicosapentaenoic acid (C20:5n-3) | nd | 0.023 ± 0.004 b | 0.026 ± 0.003 a |
| Docosahexaenoic acid (C22:6n-3) | 0.125 ± 0.004 c | 0.142 ± 0.017 b | 0.163 ± 0.014 a |
| SAFA/TFA ratio (%) | 41.1 ± 2.08 a | 24.49 ± 2.06 b | 18.50 ± 1.10 c |
| MUFA/TFA ratio (%) | 24.9 ± 2.34 b | 28.3 ± 3.44 b | 32.6 ± 2.02 a |
| PUFA/TFA ratio (%) | 34.0 ± 1.59 b | 47.2 ± 2.57 a | 48.0 ± 0.96 a |
| n-6/n-3 ratio | 6.43 ± 0.43 a | 6.07 ± 0.46 a | 5.34 ± 0.26 b |
| Symbol | Gene Name | Fold Change | |
|---|---|---|---|
| CD-LC12.75 | CD-HC25.5 | ||
| β-oxidation of fatty acids | |||
| Acox1 | Acyl-Coenzyme A oxidase 1, palmitoyl | 4.48 | 3.39 |
| Acadl | Acyl-Coenzyme A dehydrogenase, long-chain | 6.32 | - |
| Cholesterol transport and metabolism | |||
| ApoA1 | Apolipoprotein A-1 | - | 4.64 |
| Cyp2e1 | Cytochrome P450, family 2, subfamily e, polypeptide 1 | 6.92 | - |
| Cnbp | CCHC-type zinc finger, nucleic acid-binding protein | - | 3.11 |
| Nr1h2 | Nuclear receptor subfamily 1, group H, member 2 | - | 1.75 |
| Ppard | Peroxisome proliferator-activated receptor delta | - | 9.55 |
| Other lipid transport and metabolism | |||
| Acsm3 | Acyl-CoA synthetase medium-chain family member 3 | 3.32 | - |
| Hnf4a | Hepatocyte nuclear factor 4, Alpha | 4.97 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elvira-Torales, L.I.; Navarro-González, I.; Rodrigo-García, J.; Seva, J.; García-Alonso, J.; Periago-Castón, M.J. Consumption of Spinach and Tomato Modifies Lipid Metabolism, Reducing Hepatic Steatosis in Rats. Antioxidants 2020, 9, 1041. https://doi.org/10.3390/antiox9111041
Elvira-Torales LI, Navarro-González I, Rodrigo-García J, Seva J, García-Alonso J, Periago-Castón MJ. Consumption of Spinach and Tomato Modifies Lipid Metabolism, Reducing Hepatic Steatosis in Rats. Antioxidants. 2020; 9(11):1041. https://doi.org/10.3390/antiox9111041
Chicago/Turabian StyleElvira-Torales, Laura Inés, Inmaculada Navarro-González, Joaquín Rodrigo-García, Juan Seva, Javier García-Alonso, and María Jesús Periago-Castón. 2020. "Consumption of Spinach and Tomato Modifies Lipid Metabolism, Reducing Hepatic Steatosis in Rats" Antioxidants 9, no. 11: 1041. https://doi.org/10.3390/antiox9111041
APA StyleElvira-Torales, L. I., Navarro-González, I., Rodrigo-García, J., Seva, J., García-Alonso, J., & Periago-Castón, M. J. (2020). Consumption of Spinach and Tomato Modifies Lipid Metabolism, Reducing Hepatic Steatosis in Rats. Antioxidants, 9(11), 1041. https://doi.org/10.3390/antiox9111041