Domestic Sautéing with EVOO: Change in the Phenolic Profile
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Standards
2.2. Samples
2.3. Domestic Sauté Process
2.4. Polyphenol Extraction and Analysis
2.4.1. Polyphenol Extraction
2.4.2. Polyphenol Analysis by UPLC-ESI-QqQ-MS/MS
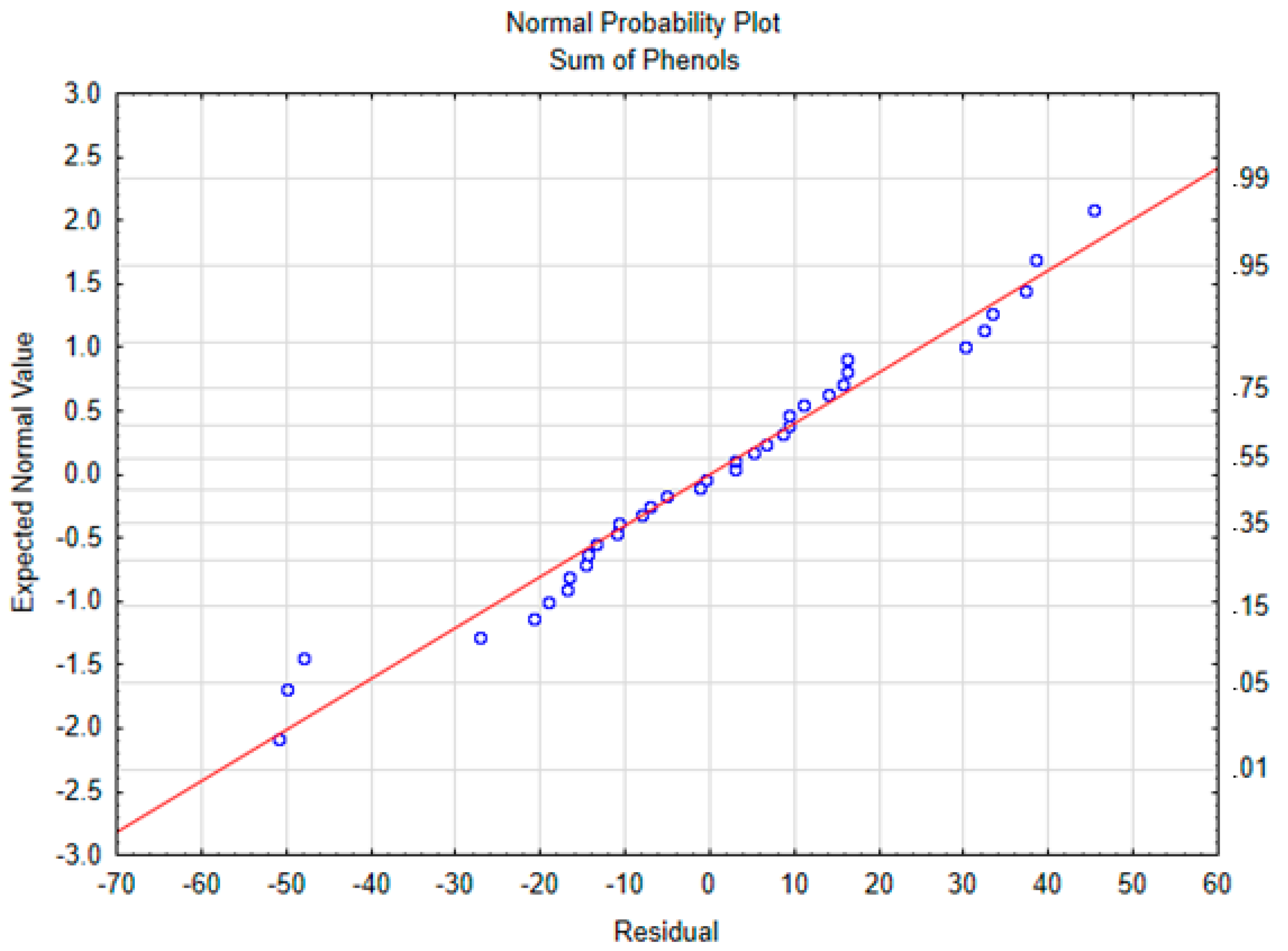
2.5. Statistical Analysis
3. Results and Discussion
3.1. Total Polyphenols
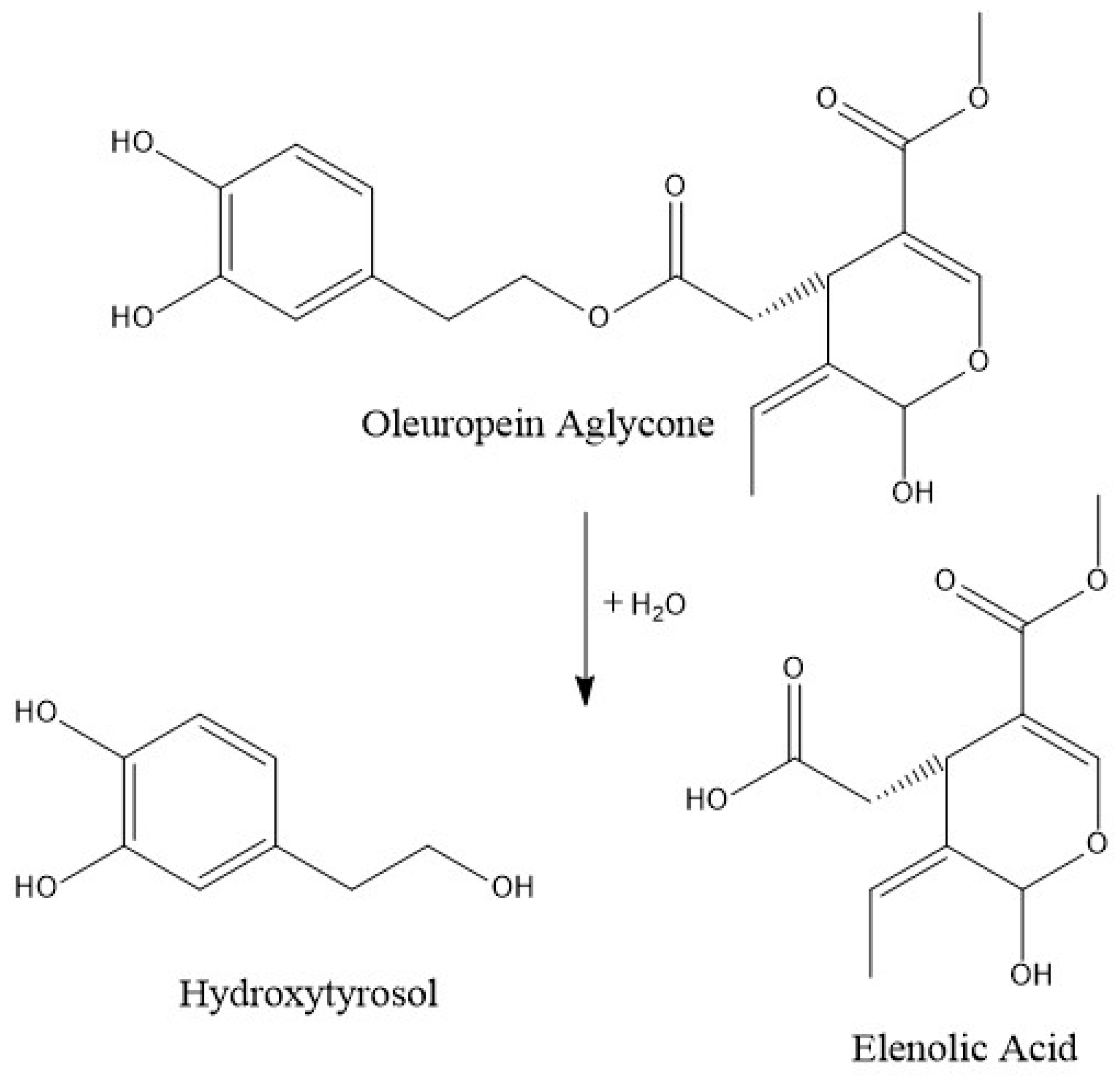
3.2. Secoiridoids
3.3. Phenolic Alcohols and Others
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cicerale, S.; Lucas, L.; Keast, R. Biological activities of phenolic compounds present in virgin olive oil. Int. J. Mol. Sci. 2010, 11, 458–479. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef]
- Gotsis, E.; Anagnostis, P.; Mariolis, A.; Vlachou, A.; Katsiki, N.; Karagiannis, A. Health benefits of the mediterranean diet: An update of research over the last 5 years. Angiology 2015, 66, 304–318. [Google Scholar] [CrossRef]
- Nocella, C.; Cammisotto, V.; Fianchini, L.; D’Amico, A.; Novo, M.; Castellani, V.; Stefanini, L.; Violi, F.; Carnevale, R. Extra Virgin Olive Oil and Cardiovascular Diseases: Benefits for Human Health. Endocr. Metab. Immune Disord. Drug Targets 2017, 18, 4–13. [Google Scholar] [CrossRef]
- Martínez-Lapiscina, E.H.; Clavero, P.; Toledo, E.; Estruch, R.; Salas-Salvadó, J.; San Julián, B.; Sanchez-Tainta, A.; Ros, E.; Valls-Pedret, C.; Martinez-Gonzalez, M.Á. Mediterranean diet improves cognition: The PREDIMED-NAVARRA randomised trial. J. Neurol. Neurosurg. Psychiatry 2013, 84, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Estruch, R.; Ros, E. Prevention of Diabetes with Mediterranean Diets. Ann. Intern. Med. 2014, 161, 157. [Google Scholar] [CrossRef] [PubMed]
- Hussein, Z.; Fawole, O.A.; Opara, U.L. Preharvest factors influencing bruise damage of fresh fruits—A review. Sci. Hortic. 2018, 229, 45–58. [Google Scholar] [CrossRef]
- Goulas, V.; Orphanides, A.; Pelava, E.; Gekas, V. Impact of Thermal Processing Methods on Polyphenols and Antioxidant Activity of Olive Oil Polar Fraction. J. Food Process. Preserv. 2015, 39, 1919–1924. [Google Scholar] [CrossRef]
- Ramírez-Anaya, J.D.P.; Samaniego-Sánchez, C.; Castañeda-Saucedo, M.C.; Villalón-Mir, M.; De La Serrana, H.L.G. Phenols and the antioxidant capacity of Mediterranean vegetables prepared with extra virgin olive oil using different domestic cooking techniques. Food Chem. 2015, 188, 430–438. [Google Scholar] [CrossRef]
- Engelsen, S.B. Explorative Spectrometric Evaluations of Frying Oil Deterioration. JAOCS J. Am. Oil Chem. Soc. 1997, 74, 1495–1508. [Google Scholar] [CrossRef]
- Rannou, C.; Laroque, D.; Renault, E.; Prost, C.; Sérot, T. Mitigation strategies of acrylamide, furans, heterocyclic amines and browning during the Maillard reaction in foods. Food Res. Int. 2016, 90, 154–176. [Google Scholar] [CrossRef]
- Persson, E.; Graziani, G.; Ferracane, R.; Fogliano, V.; Skog, K. Influence of antioxidants in virgin olive oil on the formation of heterocyclic amines in fried beefburgers. Food Chem. Toxicol. 2003, 41, 1587–1597. [Google Scholar] [CrossRef]
- Sordini, B.; Veneziani, G.; Servili, M.; Esposto, S.; Selvaggini, R.; Lorefice, A.; Taticchi, A. A quanti-qualitative study of a phenolic extract as a natural antioxidant in the frying processes. Food Chem. 2019, 279, 426–434. [Google Scholar] [CrossRef]
- Zamora, R.; Hidalgo, F.J. Carbonyl-Phenol Adducts: An Alternative Sink for Reactive and Potentially Toxic Lipid Oxidation Products. J. Agric. Food Chem. 2018, 66, 1320–1324. [Google Scholar] [CrossRef]
- Esposto, S.; Taticchi, A.; Di Maio, I.; Urbani, S.; Veneziani, G.; Selvaggini, R.; Sordini, B.; Servili, M. Effect of an olive phenolic extract on the quality of vegetable oils during frying. Food Chem. 2015, 176, 184–192. [Google Scholar] [CrossRef]
- Attya, M.; Benabdelkamel, H.; Perri, E.; Russo, A.; Sindona, G. Effects of conventional heating on the stability of major olive oil phenolic compounds by tandem mass spectrometry and isotope dilution assay. Molecules 2010, 15, 8734–8746. [Google Scholar] [CrossRef]
- Cerretani, L.; Bendini, A.; Rodriguez-Estrada, M.T.; Vittadini, E.; Chiavaro, E. Microwave heating of different commercial categories of olive oil: Part I. Effect on chemical oxidative stability indices and phenolic compounds. Food Chem. 2009, 115, 1381–1388. [Google Scholar] [CrossRef]
- Taticchi, A.; Esposto, S.; Urbani, S.; Veneziani, G.; Selvaggini, R.; Sordini, B.; Servili, M. Effect of an olive phenolic extract added to the oily phase of a tomato sauce, on the preservation of phenols and carotenoids during domestic cooking. LWT Food Sci. Technol. 2017, 84, 572–578. [Google Scholar] [CrossRef]
- Bendini, A.; Cerretani, L.; Salvador, M.D.; Fregapane, G.; Lercker, G. Stability of the sensory quality of virgin olive oil during storage: An overview. Ital. J. Food Sci. 2009, 21, 389–406. [Google Scholar]
- Kalogeropoulos, N.; Mylona, A.; Chiou, A.; Ioannou, M.S.; Andrikopoulos, N.K. Retention and distribution of natural antioxidants (α-tocopherol, polyphenols and terpenic acids) after shallow frying of vegetables in virgin olive oil. LWT Food Sci. Technol. 2007, 40, 1008–1017. [Google Scholar] [CrossRef]
- Brenes, M.; García, A.; Dobarganes, M.C.; Velasco, J.; Romero, C. Influence of thermal treatments simulating cooking processes on the polyphenol content in virgin olive oil. J. Agric. Food Chem. 2002, 50, 5962–5967. [Google Scholar] [CrossRef]
- Allouche, Y.; Jiménez, A.; Gaforio, J.J.; Uceda, M.; Beltrán, G. How Heating Affects Extra Virgin Olive Oil Quality Indexes and Chemical Composition. J. Agric. Food Chem. 2007, 55, 9646–9654. [Google Scholar] [CrossRef]
- Casal, S.; Malheiro, R.; Sendas, A.; Oliveira, B.P.P.; Pereira, J.A. Olive oil stability under deep-frying conditions. Food Chem. Toxicol. 2010, 48, 2972–2979. [Google Scholar] [CrossRef]
- Aubaile-Sallenave, F. La Méditerranée, une cuisine, des cuisines. In Anthropologie de L’Alimentation; SAGE: London, UK; Thousand Oaks, CA, USA; New Delhi, India, 1996; pp. 139–194. [Google Scholar]
- García, A.; Brenes, M.; García, P.; Romero, C.; Garrido, A. Phenolic content of commercial olive oils. Eur. Food Res. Technol. 2003, 216, 520–525. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Chiou, A.; Mylona, A.; Ioannou, M.S.; Andrikopoulos, N.K. Recovery and distribution of natural antioxidants (α-tocopherol, polyphenols and terpenic acids) after pan-frying of Mediterranean finfish in virgin olive oil. Food Chem. 2007, 100, 509–517. [Google Scholar] [CrossRef]
- Suárez, M.; Macià, A.; Romero, M.-P.; Motilva, M.-J. Improved liquid chromatography tandem mass spectrometry method for the determination of phenolic compounds in virgin olive oil. J. Chromatogr. A 2008, 1214, 90–99. [Google Scholar] [CrossRef]
- López-Yerena, A.; Lozano-Castellón, J.; Olmo-Cunillera, A.; Tresserra-Rimbau, A.; Quifer-Rada, P.; Jiménez, B.; Pérez, M.; Vallverdú-Queralt, A. Effects of organic and conventional growing systems on the phenolic profile of extra-virgin olive oil. Molecules 2019, 24, 1986. [Google Scholar] [CrossRef]
- Sánchez de Medina, V.; Miho, H.; Melliou, E.; Magiatis, P.; Priego-Capote, F.; Luque de Castro, M.D. Quantitative method for determination of oleocanthal and oleacein in virgin olive oils by liquid chromatography–tandem mass spectrometry. Talanta 2017, 162, 24–31. [Google Scholar] [CrossRef]
- Douglas, C. Montgomery: Design and Analysis of Experiments; John Willy Sons: Hoboken, NJ, USA, 2000; p. 734. [Google Scholar]
- Mariod, A.A.; Ibrahim, R.M.; Ismail, M.; Ismail, N. Antioxidant activity of the phenolic leaf extracts from monechma ciliatum in stabilization of corn oil. JAOCS J. Am. Oil Chem. Soc. 2010, 87, 35–43. [Google Scholar] [CrossRef]
- Xiangli, F.; Wei, W.; Chen, Y.; Jin, W.; Xu, N. Optimization of preparation conditions for polydimethylsiloxane (PDMS)/ceramic composite pervaporation membranes using response surface methodology. J. Membr. Sci. 2008, 311, 23–33. [Google Scholar] [CrossRef]
- Ramírez-Anaya, J.; Castañeda-Saucedo, M.C.; Olalla-Herrera, M.; Villalón-Mir, M.; De la Serrana, H.L.G.; Samaniego-Sánchez, C. Changes in the antioxidant properties of extra virgin olive oil after cooking typical mediterranean vegetables. Antioxidants 2019, 8, 246. [Google Scholar]
- Lozano-Castellón, J.; López-Yerena, A.; Rinaldi de Alvarenga, J.F.; Romero del Castillo-alba, J.; Vallverdú-queralt, A.; Escribano-ferrer, E.; Lamuela-raventós, R.M. Health-promoting properties of oleocanthal and oleacein: Two secoiridoids from extra-virgin olive oil. Crit. Rev. Food Sci. Nutr. 2019, 1–17. [Google Scholar] [CrossRef]
- Beauchamp, G.K.; Keast, R.S.J.; Morel, D.; Lin, J.; Pika, J.; Han, Q.; Lee, C.H.; Smith, A.B.; Breslin, P.A.S. Ibuprofen-like activity in extra-virgin olive oil. Nature 2005, 437, 45–46. [Google Scholar] [CrossRef]
- Hansen, K.; Adsersen, A.; Christensen, S.B.; Jensen, S.R.; Nyman, U.; Smitt, U.W. Isolation of an angiotensin converting enzyme (ACE) inhibitor from Olea europaea and Olea lancea. Phytomedicine 2011, 2, 319–325. [Google Scholar] [CrossRef]
- Czerwińska, M.E.; Kiss, A.K.; Naruszewicz, M. Inhibition of human neutrophils NEP activity, CD11b/CD18 expression and elastase release by 3,4-dihydroxyphenylethanol-elenolic acid dialdehyde, oleacein. Food Chem. 2014, 153, 1–8. [Google Scholar] [CrossRef]
- Obied, H.K.; Prenzler, P.D.; Ryan, D.; Servili, M.; Taticchi, A.; Esposto, S.; Robards, K. Biosynthesis and biotransformations of phenol-conjugated oleosidic secoiridoids from Olea europaea L. Nat. Prod. Rep. 2008, 25, 1167. [Google Scholar] [CrossRef]
- Singleton, V.L. Oxygen with phenols and related reactions in musts, wines, and model systems: Observations and practical implications. Am. J. Enol. Vitic. 1987, 38, 69–77. [Google Scholar]
- Hvattum, E.; Ekeberg, D. Study of the collision-induced radical cleavage of flavonoid glycosides using negative electrospray ionization tandem quadrupole mass spectrometry. J. Mass Spectrom. 2003, 38, 43–49. [Google Scholar] [CrossRef]
- Vallverdu-Queralt, A.; Regueiro, J.; Rinaldi de Alvarenga, J.; Torrado, X.; Lamuela-Ravento, R.M. Home Cooking and Phenolics: Effect of Thermal Treatment and Addition of Extra Virgin Olive Oil on the Phenolic Profile of Tomato Sauces. J. Agric. Food Chem. 2014, 62, 3314–3320. [Google Scholar] [CrossRef]
- Krichene, D.; Salvador, M.D.; Fregapane, G. Stability of Virgin Olive Oil Phenolic Compounds during Long-Term Storage (18 Months) at Temperatures of 5–50 °C. J. Agric. Food Chem. 2015, 63, 6779–6786. [Google Scholar] [CrossRef]
- European Commission. European Commission Regulation EC No. 432/2012 establishing a list of permitted health claims made on foods, other than those referring to the reduction of disease risk and to children’s development and health. Off. J. Eur. Union L 2012, 136, 1–40. [Google Scholar]
- Daskalaki, D.; Kefi, G.; Kotsiou, K.; Tasioula-Margari, M. Evaluation of phenolic compounds degradation in virgin olive oil during storage and heating. J. Food Nutr. Res. 2009, 48, 31–41. [Google Scholar]
| Experiment | Temperature (Level) | Time (Level) | Temperature | Time |
|---|---|---|---|---|
| 1 | –1 | –1 | 120 °C | 30 |
| 2 | 1 | –1 | 170 °C | 15 |
| 3 | –1 | 1 | 120 °C | 60 |
| 4 | 1 | 1 | 170 °C | 30 |
| Group/Compound | Raw | ↓T ↓t | ↓T ↑t | ↑T ↓t | ↑T ↑t |
|---|---|---|---|---|---|
| Sum of phenols | 860 ± 22 | 487 ± 29 | 498 ± 32 | 240 ± 19 | 218 ± 12 |
| Secoiridoids | 835 ± 22 | 466 ± 30 | 481 ± 31 | 231 ± 20 | 213 ± 12 |
| Ligstroside aglycone | 368 ± 7 | 190 ± 13 | 193 ± 11 | 94 ± 21 | 97 ± 7 |
| Oleocanthal | 81 ± 4 | 51 ± 3 | 53 ± 5 | 41 ± 3 | 41 ± 3 |
| Oleuropein aglycone | 79 ± 2 | 45 ± 3 | 47 ± 3 | 15 ± 2 | 12 ± 1 |
| Oleacein | 252 ± 9 | 134 ± 13 | 139 ± 15 | 46 ± 6 | 32 ± 4 |
| HDCM-OA | 23.6 ± 0.9 | 21 ± 2 | 22 ± 3 | 16 ± 2 | 9 ± 1 |
| HOA | 3.2 ± 0.2 | 2.3 ± 0.3 | 3.3 ± 0.5 | 2.3 ± 0.3 | 0.7 ± 0.1 |
| Elenolic acid | 25.1 ± 0.2 | 16 ± 2 | 16 ± 1 | 10 ± 1 | 10.9 ± 0.6 |
| Hydroxyelenolic acid | 1.9 ± 0.1 | 8 ± 1 | 8 ± 1 | 6.7 ± 0.8 | 9.1 ± 0.9 |
| Phenolic alcohols | 19.6 ± 0.5 | 18 ± 1 | 14 ± 1 | 5.9 ± 0.6 | 2.8 ± 0.2 |
| Hydroxytyrosol acetate | 4.5 ± 0.2 | 3.9 ± 0.3 | 4.0 ± 0.2 | 1.8 ± 0.2 | 1.4 ± 0.1 |
| Hydroxytyrosol | 15.2 ± 0.7 | 14 ± 1 | 10.0 ± 0.9 | 4.1 ± 0.5 | 1.5 ± 0.2 |
| Flavonoids | 1.8 ± 0.2 | 1.3 ± 0.4 | 1.3 ± 0.4 | 0.79 ± 0.08 | 0.86 ± 0.05 |
| Apigenin | 0.61 ± 0.04 | 0.7 ± 0.4 | 0.7 ± 0.4 | 0.48 ± 0.05 | 0.54 ± 0.03 |
| Luteolin | 1.16 ± 0.15 | 0.54 ± 0.08 | 0.61 ± 0.06 | 0.31 ± 0.04 | 0.32 ± 0.02 |
| Phenolic acids | 3.7 ± 0.3 | 1.4 ± 0.7 | 1.2 ± 0.4 | 1.2 ± 0.3 | 0.8 ± 0.2 |
| Ferulic acid | 3.3 ± 0.3 | 0.9 ± 0.7 | 0.7 ± 0.4 | 0.7 ± 0.3 | 0.4 ± 0.2 |
| p-Coumaric acid | 0.45 ± 0.01 | 0.49 ± 0.06 | 0.47 ± 0.04 | 0.47 ± 0.04 | 0.40 ± 0.02 |
| Lignans | 0.44 ± 0.05 | 0.48 ± 0.07 | 0.49 ± 0.05 | 0.60 ± 0.06 | 0.64 ± 0.07 |
| Pinoresinol | 0.44 ± 0.05 | 0.48 ± 0.07 | 0.49 ± 0.05 | 0.60 ± 0.06 | 0.64 ± 0.07 |
| Group/Compound | R2 | β0 | Temperature | Time | Interaction | |||
|---|---|---|---|---|---|---|---|---|
| F-Value | β1 | F-Value | β2 | F-Value | β3 | |||
| Sum of phenols | 0.968 | 360.8 * | 1038.8 * | −131.9 * | 0.458 | - | 3.87 | −8.05 |
| Secoiridoids | 0.965 | 347.9 * | 946.9 * | −125.8 * | 0.057 | - | 4.04 | −8.22 * |
| Ligstroside aglycone | 0.927 | 143.2 * | 427.4 * | −47.8 * | 0.443 | - | 0.0018 | - |
| Oleocanthal | 0.703 | 46.5 * | 81.0 * | −5.31 * | 0.603 | - | 0.232 | - |
| Oleuropein aglycone | 0.973 | 29.7 * | 1276.1 * | −16.2 * | 0.402 | −0.287 | 8.96 * | −1.35 * |
| Oleacein | 0.955 | 87.9 * | 736.9 * | −48.6 * | 1.59 | −2.26 | 6.83 * | −4.68 * |
| HDCM-OA | 0.845 | 17.2 * | 144.3 * | −4.34 * | 15.5 * | −1.42 * | 33.7 * | −2.10 * |
| HOA | 0.888 | 2.18 * | 369.9 * | −0.660 * | 0.718 | −0.147 * | 142.7 * | −0.645 * |
| Elenolic acid | 0.824 | 13.2 * | 159.8 * | −2.99 * | 4.26 * | 0.488 * | 0.678 | - |
| Hydroxyelenolic acid | 0.444 | 7.92 * | 0.002 | - | 17.1 * | 0.654 * | 11.9 * | 0.546 * |
| Phenolic alcohols | 0.978 | 10.1 * | 1427.7 * | −5.78 * | 124.6 * | −1.71 * | 1.36 | 0.178 |
| Hydroxytyrosol acetate | 0.964 | 2.77 * | 916.5 * | −1.18 * | 3.28 | −0.0705 | 12.6 * | −0.138 * |
| Hydroxytyrosol | 0.971 | 7.37 * | 1026.5 * | −4.60 * | 129.9 * | −1.64 * | 4.86 * | 0.316 * |
| Flavonoids | 0.396 | 1.06 * | 23.1 * | −0.236 * | 0.723 | - | 0.0038 | - |
| Apigenin | 0.121 | 0.615 * | 5.54 * | −0.105 * | 0.265 | - | 0.0648 | - |
| Luteolin | 0.855 | 0.446 * | 202.4 * | −0.131 * | 4.17 * | 0.0187 * | 2.44 | −0.0144 |
| Phenolic acids | 0.090 | 1.15 * | 3.35 | −0.147 | 3.08 | −0.141 | 0.0288 | −0.0136 |
| Ferulic acid | 0.085 | 0.682 * | 3.27 | −0.133 | 2.97 | −0.127 | 0.0158 | −0.00927 |
| p-Coumaric acid | 0.111 | 0.463 * | 3.43 | −0.013 | 3.60 | −0.0138 | 0.354 | −0.00433 |
| Lignans | 0.520 | 0.553 * | 39.2 * | 0.068 * | 1.16 | 0.0116 | 0.583 | 0.00827 |
| Pinoresinol | 0.526 | 0.553 * | 39.2 * | 0.068 * | 1.16 | 0.0116 | 0.583 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lozano-Castellón, J.; Vallverdú-Queralt, A.; Rinaldi de Alvarenga, J.F.; Illán, M.; Torrado-Prat, X.; Lamuela-Raventós, R.M. Domestic Sautéing with EVOO: Change in the Phenolic Profile. Antioxidants 2020, 9, 77. https://doi.org/10.3390/antiox9010077
Lozano-Castellón J, Vallverdú-Queralt A, Rinaldi de Alvarenga JF, Illán M, Torrado-Prat X, Lamuela-Raventós RM. Domestic Sautéing with EVOO: Change in the Phenolic Profile. Antioxidants. 2020; 9(1):77. https://doi.org/10.3390/antiox9010077
Chicago/Turabian StyleLozano-Castellón, Julián, Anna Vallverdú-Queralt, José Fernando Rinaldi de Alvarenga, Montserrat Illán, Xavier Torrado-Prat, and Rosa Maria Lamuela-Raventós. 2020. "Domestic Sautéing with EVOO: Change in the Phenolic Profile" Antioxidants 9, no. 1: 77. https://doi.org/10.3390/antiox9010077
APA StyleLozano-Castellón, J., Vallverdú-Queralt, A., Rinaldi de Alvarenga, J. F., Illán, M., Torrado-Prat, X., & Lamuela-Raventós, R. M. (2020). Domestic Sautéing with EVOO: Change in the Phenolic Profile. Antioxidants, 9(1), 77. https://doi.org/10.3390/antiox9010077








