Evaluation of Antioxidant and Antiproliferative Properties of Cornus mas L. Fruit Juice
Abstract
1. Introduction
2. Materials and Methods
2.1. Compositional Analysis of Cornelian Cherry (Cornus mas)
2.2. Cell Lines and Cell Cultures
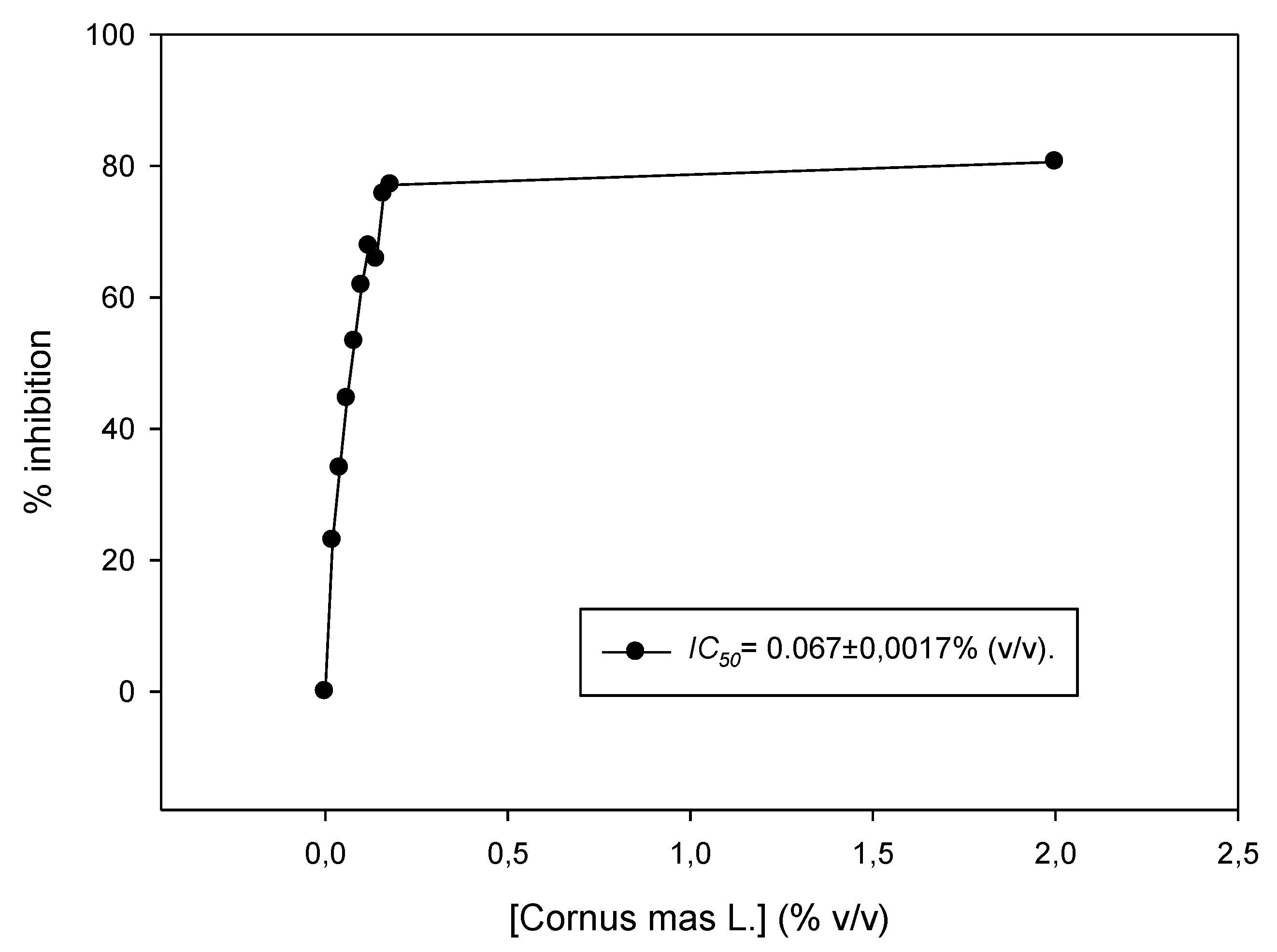
2.3. Determination of Cornus mas L. Juice in Vitro Antioxidant Activity (DPPH Assay)
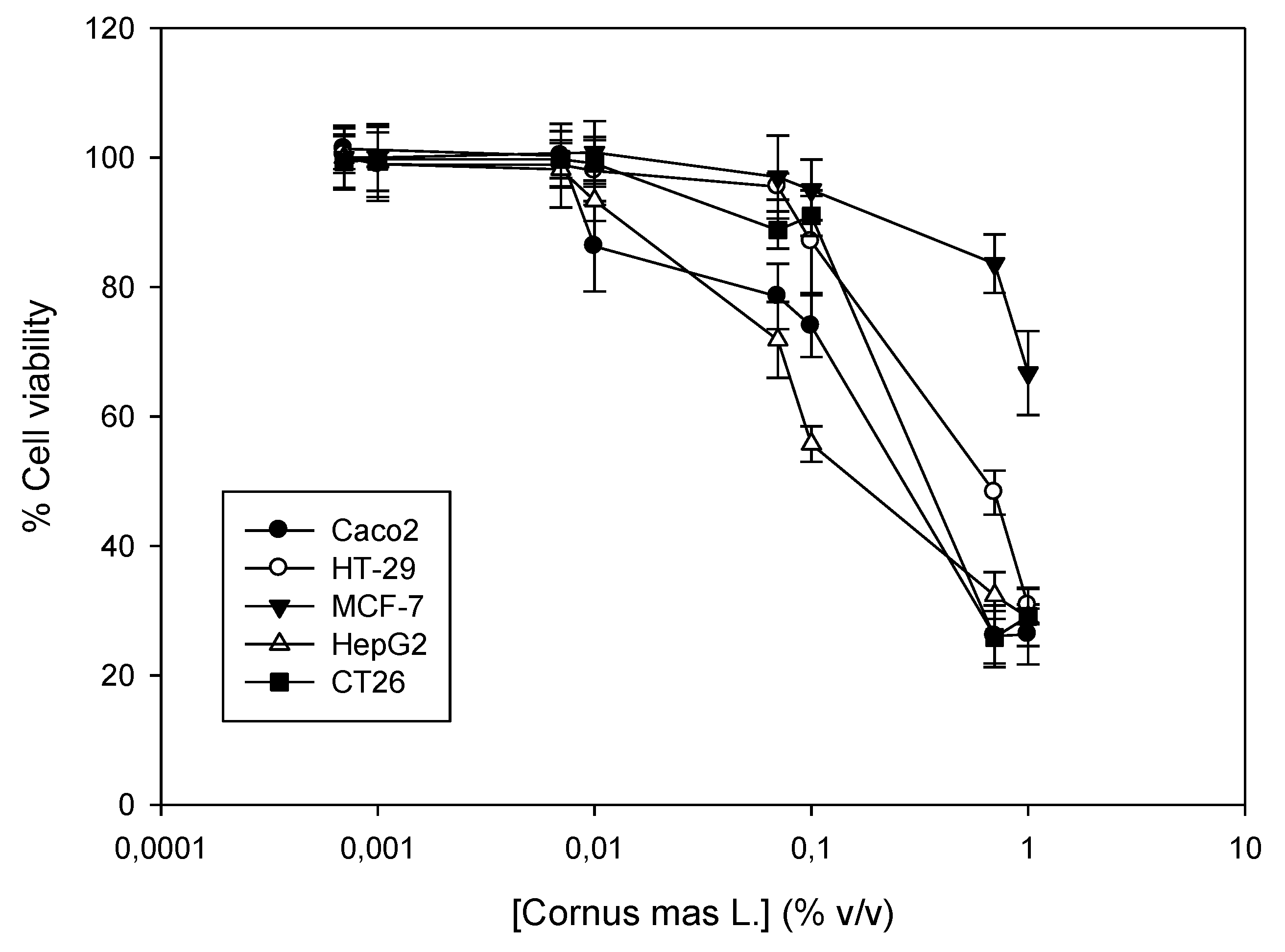
2.4. Determination of the In Vitro Antiproliferative Effect of Cornus mas L. Juice (Sulforhodamine B Assay)
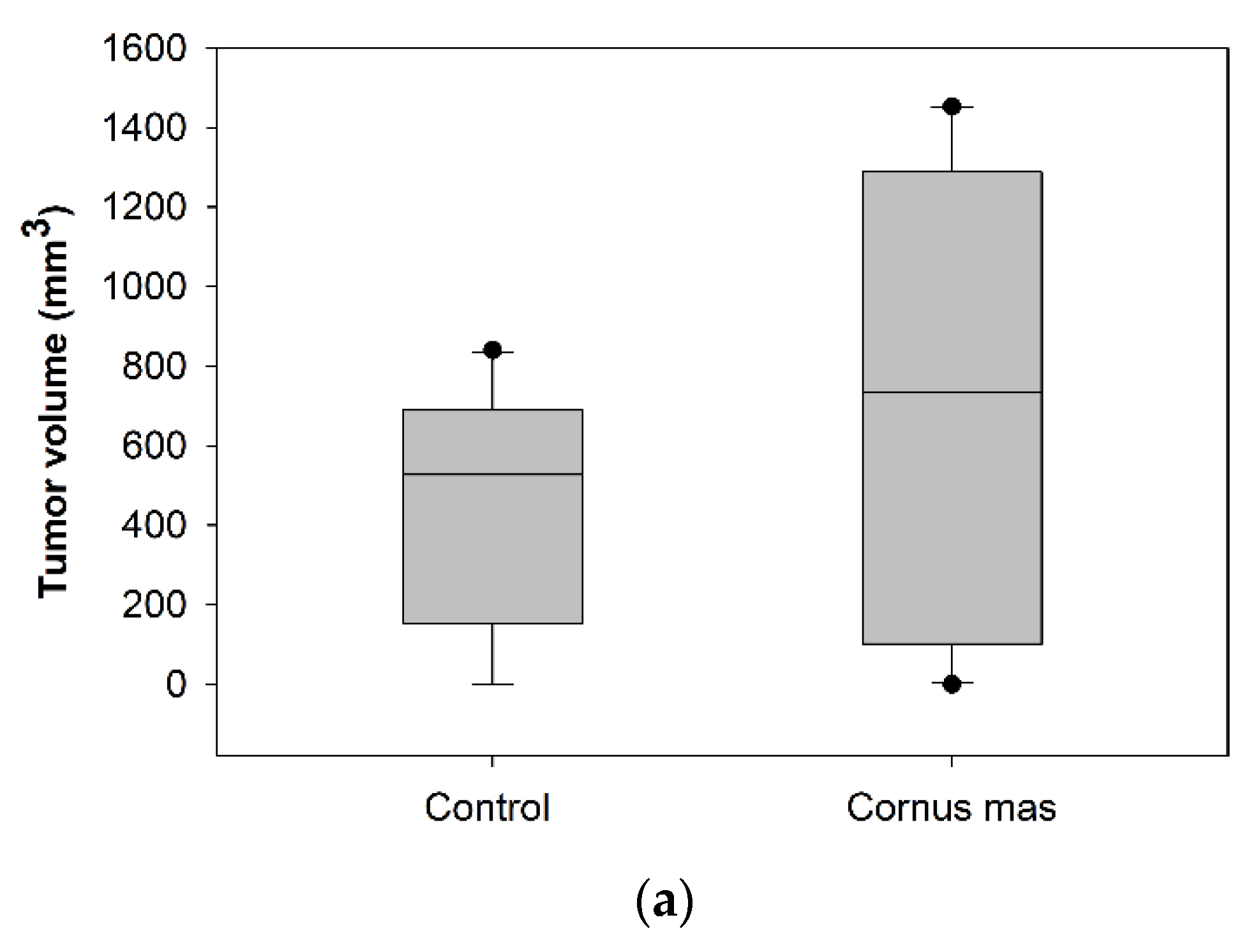
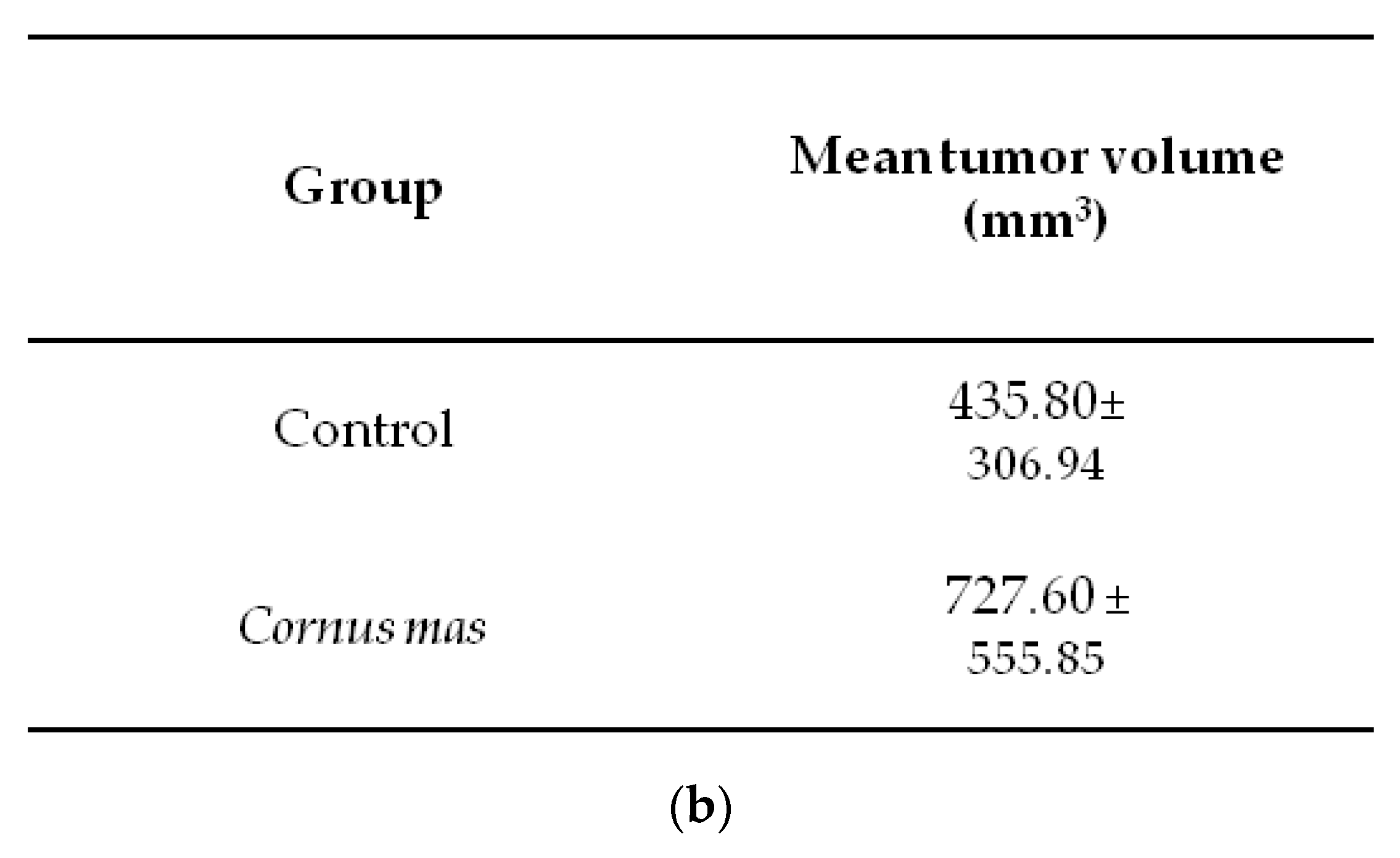
2.5. Animals and CT26 Experimental Tumor Model
2.6. Statistical Evaluation
2.7. Ethics Statement
3. Results and Discussion
3.1. Composition of Cornus mas L. Juice
3.2. Antioxidant Activity of Cornus mas L. Juice In Vitro
3.3. Antiproliferative Effect of Cornus mas L. Juice In Vitro against Cancer Cell Lines
3.4. Effect of Cornus mas L. Juice on the Growth of Syngeneic Colon Carcinoma Tumors
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sozański, T.; Kucharska, A.Z.; Szumny, A.; Magdalan, J.; Bielska, K.; Merwid-Ląd, A.; Woźniak, A.; Dzimira, S.; Piórecki, N.; Trocha, M. The protective effect of the Cornus mas fruits (cornelian cherry) on hypertriglyceridemia and atherosclerosis through PPARα activation in hypercholesterolemic rabbits. Phytomedicine 2014, 21, 1774–1784. [Google Scholar] [CrossRef]
- Hosseinpour-Jaghdani, F.; Shomali, T.; Gholipour-Shahraki, S.; Rahimi-Madiseh, M.; Rafieian-Kopaei, M. Cornus mas: A review on traditional uses and pharmacological properties. J. Complement. Integr. Med. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Tural, S.; Koca, I. Physico-chemical and antioxidant properties of Cornelian cherry fruits (Cornus mas L.) grown in Turkey. Sci. Hortic. 2008, 116, 362–366. [Google Scholar] [CrossRef]
- Mantzourani, I.; Terpou, A.; Alexopoulos, A.; Bezirtzoglou, E.; Bekatorou, A.; Plessas, S. Production of a potentially synbiotic fermented Cornelian cherry (Cornus mas L.) beverage using Lactobacillus paracasei K5 immobilized on wheat bran. Biocatal. Agric. Biotechnol. 2019, 17, 347–351. [Google Scholar] [CrossRef]
- Nouska, C.; Kazakos, S.; Mantzourani, I.; Alexopoulos, A.; Bezirtzoglou, E.; Plessas, S. Fermentation of Cornus mas L. Juice for functional low alcoholic beverage production. Cur. Res. Nutr. Food Sci. 2016, 4, 119–124. [Google Scholar] [CrossRef]
- Deng, S.; West, B.J.; Jensen, C.J. UPLC-TOF-MS Characterization and Identification of Bioactive Iridoids in Cornus mas Fruit. J. Anal. Methods Chem. 2013, 2013, 710972. [Google Scholar] [CrossRef] [PubMed]
- Dinda, B.; Kyriakopoulos, A.M.; Dinda, S.; Zoumpourlis, V.; Thomaidis, N.S.; Velegraki, A.; Markopoulos, C.; Dinda, M. Cornus mas L. (cornelian cherry), an important European and Asian traditional food and medicine: Ethnomedicine, phytochemistry and pharmacology for its commercial utilization in drug industry. J. Ethnopharmacol. 2016, 193, 670–690. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Schutzki, R.; Chandra, A.; Nair, M.G. Characterization, quantification, and bioactivities of anthocyanins in Cornus species. J. Agric. Food Chem. 2002, 50, 2519–2523. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, B.; Abasi, M.; Abbasi, M.M.; Jahanban-Esfahlan, R. Anti-Proliferative Properties of Cornus mas Fruit in Different Human Cancer Cells. Asian Pac. J. Cancer Prev. 2015, 16, 5727–5731. [Google Scholar] [CrossRef] [PubMed][Green Version]
- De Biaggi, M.; Donno, D.; Mellano, M.G.; Riondato, I.; Rakotoniaina, E.N.; Beccaro, G.L. Cornus mas (L.) Fruit as a Potential Source of Natural Health-Promoting Compounds: Physico-Chemical Characterisation of Bioactive Components. Plant Foods Hum. Nutr. 2018, 73, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Cunniff, P. Official methods of Analysis of AOAC. In Official Methods of Analysis of AOAC International, 16th ed.; AOAC International: Gaithersburg, MD, USA, 1997. [Google Scholar]
- The Commission of the European Communities. Regulation No 2568/91. Off. J. Eur. Commun. 1991, 248, 1–83. [Google Scholar]
- Anestopoulos, I.; Kavo, A.; Tentes, I.; Kortsaris, A.; Panayiotidis, M.; Lazou, A.; Pappa, A. Silibinin protects H9c2 cardiac cells from oxidative stress and inhibits phenylephrine-induced hypertrophy: Potential mechanisms. J. Nutr. Biochem. 2013, 24, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Fitsiou, E.; Mitropoulou, G.; Spyridopoulou, K.; Vamvakias, M.; Bardouki, H.; Galanis, A.; Chlichlia, K.; Kourkoutas, Y.; Panayiotidis, M.; Pappa, A. Chemical Composition and Evaluation of the Biological Properties of the Essential Oil of the Dietary Phytochemical Lippia Citriodora. Molecules 2018, 23, 15. [Google Scholar] [CrossRef] [PubMed]
- Vichai, V.; Kirtikara, K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat. Protoc. 2006, 1, 1112–1116. [Google Scholar] [CrossRef] [PubMed]
- Spyridopoulou, K.; Tiptiri-Kourpeti, A.; Lampri, E.; Fitsiou, E.; Vasileiadis, S.; Vamvakias, M.; Bardouki, H.; Goussia, A.; Malamou-Mitsi, V.; Panayiotidis, M.I.; et al. Dietary mastic oil extracted from Pistacia lentiscus var. chia suppresses tumor growth in experimental colon cancer models. Sci. Rep. 2017, 7, 3782. [Google Scholar] [CrossRef] [PubMed]
- Gąstoł, M.; Krośniak, M.; Derwisz, M.; Dobrowolska-Iwanek, J. Cornelian cherry (Cornus mas L.) juice as a potential source of biological compounds. J. Med. Food 2013, 16, 728–732. [Google Scholar] [CrossRef] [PubMed]
- Tarko, T.; Duda-Chodak, A.; Satora, P.; Sroka, P.; Pogoń, P.; Machalica, J. Chaenomeles japonica, Cornus mas, Morus nigra fruits characteristics and their processing potential. J. Food Sci. Technol. 2014, 51, 3934–3941. [Google Scholar] [CrossRef] [PubMed]
- Vidrih, R.; Čejić, Ž.; Hribar, J. Content of certain food components in flesh and stones of the cornelian cherry (Cornus mas L.) genotypes. Croat. J. Food Sci. Technol. 2012, 4, 64–70. [Google Scholar]
- Pantelidis, G.E.; Vasilakakis, M.; Manganaris, G.A.; Diamantidis, G. Antioxidant capacity, phenol, anthocyanin and ascorbic acid contents in raspberries, blackberries, red currants, gooseberries and Cornelian cherries. Food Chem. 2007, 102, 777–783. [Google Scholar] [CrossRef]
- Moldovan, B.; Filip, A.; Clichici, S.; Suharoschi, R.; Bolfa, P.; David, L. Antioxidant activity of Cornelian cherry (Cornus mas L.) fruits extract and the in vivo evaluation of its anti-inflammatory effects. J. Funct. Foods 2016, 26, 77–87. [Google Scholar] [CrossRef]
- David, L.; Danciu, V.; Moldovan, B.; Filip, A. Effects of in vitro gastrointestinal digestionon the antioxidant capacity and anthocyanin content of Cornelian cherry fruit extract. Antioxidants 2019, 8, 114. [Google Scholar] [CrossRef] [PubMed]
- Savikin, K.; Zdunic, G.; Jankovic, T.; Stanojkovic, T.; Juranic, Z.; Menkovic, N. In vitro cytotoxic and antioxidative activity of Cornus mas and Cotinus coggygria. Nat. Prod. Res. 2009, 23, 1731–1739. [Google Scholar] [CrossRef] [PubMed]
- Teller, N.; Thiele, W.; Boettler, U.; Sleeman, J.; Marko, D. Delphinidin inhibits a broad spectrum of receptor tyrosine kinases of the ErbB and VEGFR family. Mol. Nutr. Food Res. 2009, 53, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Thiele, W.; Rothley, M.; Teller, N.; Jung, N.; Bulat, B.; Plaumann, D.; Vanderheiden, S.; Schmaus, A.; Cremers, N.; Göppert, B.; et al. Delphinidin is a novel inhibitor of lymphangiogenesis but promotes mammary tumor growth and metastasis formation in syngeneic experimental rats. Carcinogenesis 2013, 34, 2804–2813. [Google Scholar] [CrossRef] [PubMed]
- Sak, K. Cytotoxicity of dietary flavonoids on different human cancer types. Pharmacogn. Rev. 2014, 8, 122–146. [Google Scholar] [CrossRef]
| Sample | Citric Acid | Malic Acid | Succinic Acid |
|---|---|---|---|
| Fruit | 0.2 | 74.4 | 21.8 |
| Juice | 0.1 | 14.5 | 6.8 |
| Colour * (CIELAB Parameters) | Dry Material | Moisture | Fat | ||
|---|---|---|---|---|---|
| L* | a* | b* | % | % | % |
| 27.1 | 23.3 | 8.1 | 24.75 | 75.25 | 0.25 |
| Brix | pH | Acidity (%) * |
|---|---|---|
| 21.40 | 3.27 | 2.59 |
| Minerals | Sugars (%) | |||||
|---|---|---|---|---|---|---|
| Κ | Fe | Cu | Ca | Μg | Fructose | Glucose |
| 672.44 | 0.58 | 0.38 | 173.41 | 52.02 | 20.3 | 31.2 |
| Fatty Acids | Percentage of Fatty Acids in Cornus mas Kernel Oil |
|---|---|
| Palmitic | 7.0 |
| Stearic | 2.2 |
| Oleic | 23.0 |
| Linoleic (ω-6) | 53.6 |
| α-linolenic (ω-3) | 1.4 |
| Arachidonic | 1.4 |
| Cell Lines | EC50 (% v/v) |
|---|---|
| Caco2 | 0.37 ± 0.04 |
| HT-29 | 0.49 ± 0.09 |
| MCF-7 | n.d.* |
| HepG2 | 0.08 ± 0.004 |
| CT26 | 0.20 ± 0.08 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tiptiri-Kourpeti, A.; Fitsiou, E.; Spyridopoulou, K.; Vasileiadis, S.; Iliopoulos, C.; Galanis, A.; Vekiari, S.; Pappa, A.; Chlichlia, K. Evaluation of Antioxidant and Antiproliferative Properties of Cornus mas L. Fruit Juice. Antioxidants 2019, 8, 377. https://doi.org/10.3390/antiox8090377
Tiptiri-Kourpeti A, Fitsiou E, Spyridopoulou K, Vasileiadis S, Iliopoulos C, Galanis A, Vekiari S, Pappa A, Chlichlia K. Evaluation of Antioxidant and Antiproliferative Properties of Cornus mas L. Fruit Juice. Antioxidants. 2019; 8(9):377. https://doi.org/10.3390/antiox8090377
Chicago/Turabian StyleTiptiri-Kourpeti, Angeliki, Eleni Fitsiou, Katerina Spyridopoulou, Stavros Vasileiadis, Christos Iliopoulos, Alex Galanis, Stavroula Vekiari, Aglaia Pappa, and Katerina Chlichlia. 2019. "Evaluation of Antioxidant and Antiproliferative Properties of Cornus mas L. Fruit Juice" Antioxidants 8, no. 9: 377. https://doi.org/10.3390/antiox8090377
APA StyleTiptiri-Kourpeti, A., Fitsiou, E., Spyridopoulou, K., Vasileiadis, S., Iliopoulos, C., Galanis, A., Vekiari, S., Pappa, A., & Chlichlia, K. (2019). Evaluation of Antioxidant and Antiproliferative Properties of Cornus mas L. Fruit Juice. Antioxidants, 8(9), 377. https://doi.org/10.3390/antiox8090377







