Myrciaria tenella (DC.) O. Berg (Myrtaceae) Leaves as a Source of Antioxidant Compounds
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Leaf Extract Preparation
2.3. Total Sugar, Protein, and Phenolic Compounds
2.4. Phytochemistry Screening by Thin Layer Chromatography (TLC)
2.5. Antioxidant Activity In Vitro
2.6. Cell Viability
2.7. Annexin V-FITC Apoptotic Activity
2.8. Antioxidant Activity In Vivo
2.8.1. Caenorhabditis elegans Maintenance and Extract Treatment
2.8.2. Evaluation of Reactive Oxygen Species (ROS) Production in C. elegans
2.8.3. Oxidative Stress Assay
2.8.4. Effect of CEE on C. elegans Development
2.9. Ultra-Performance Liquid Chromatography (UPLC)
2.10. Statistical Analysis
3. Results
3.1. Phytochemical Composition
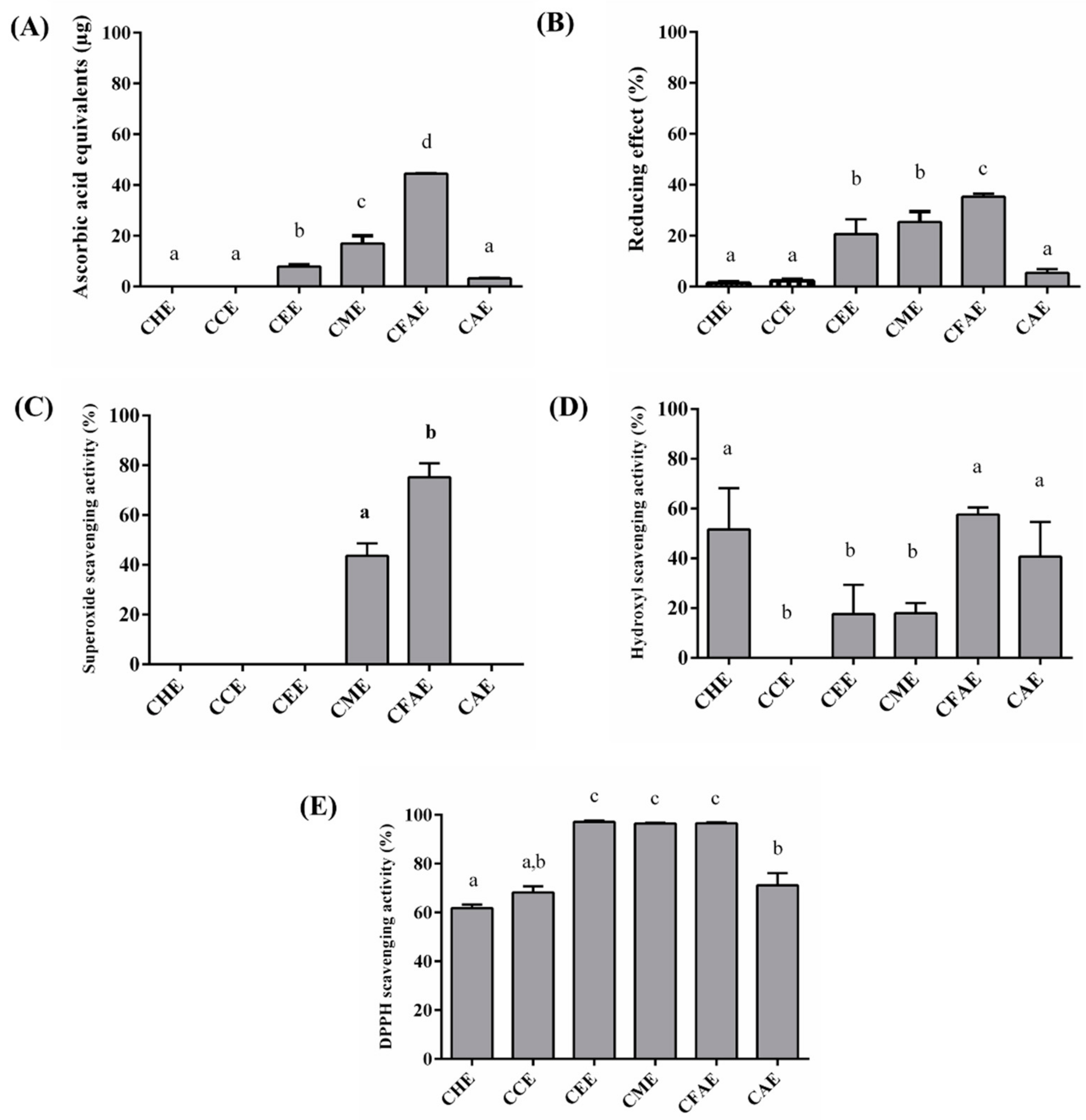
3.2. In Vitro Antioxidant Assays
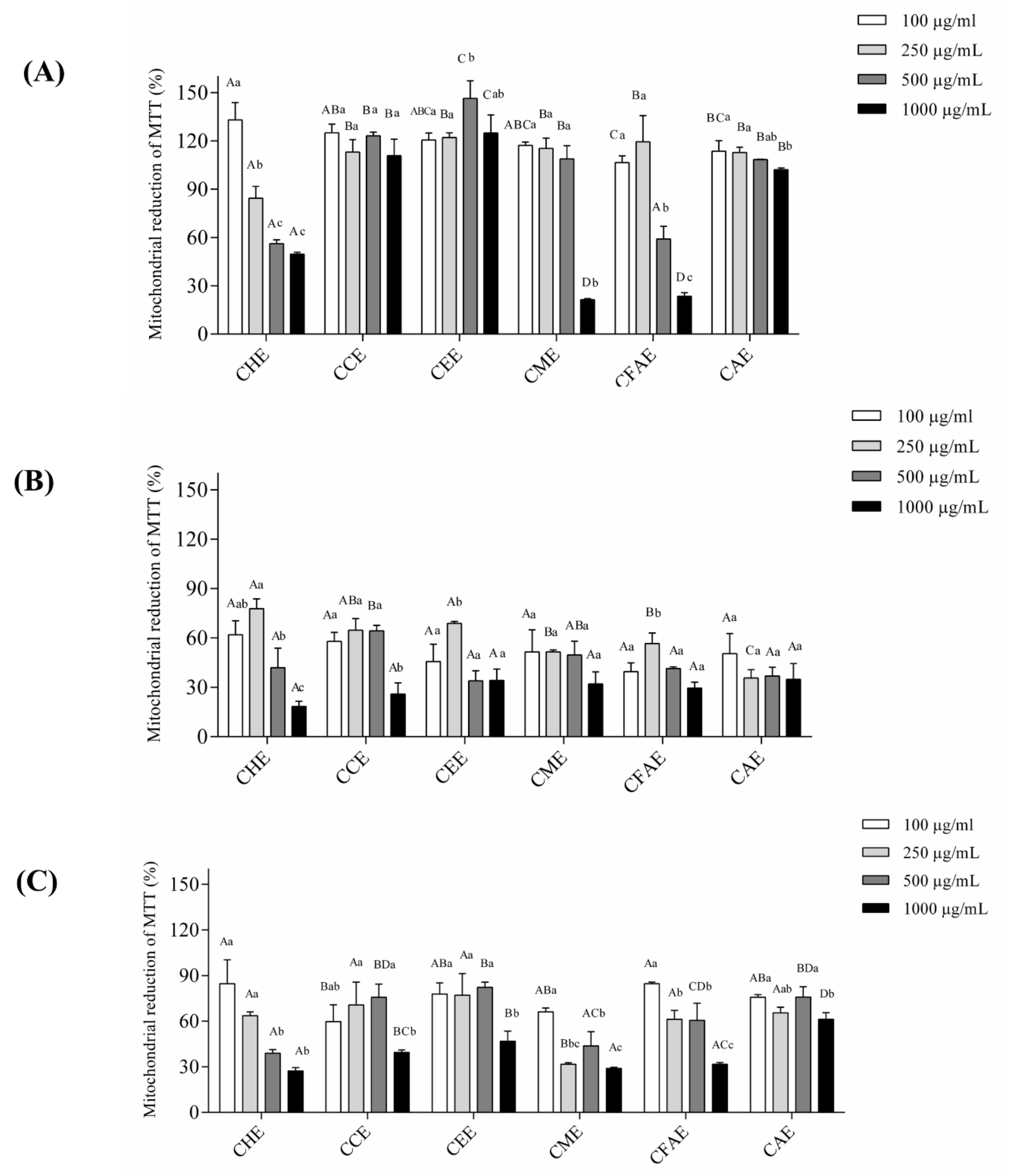
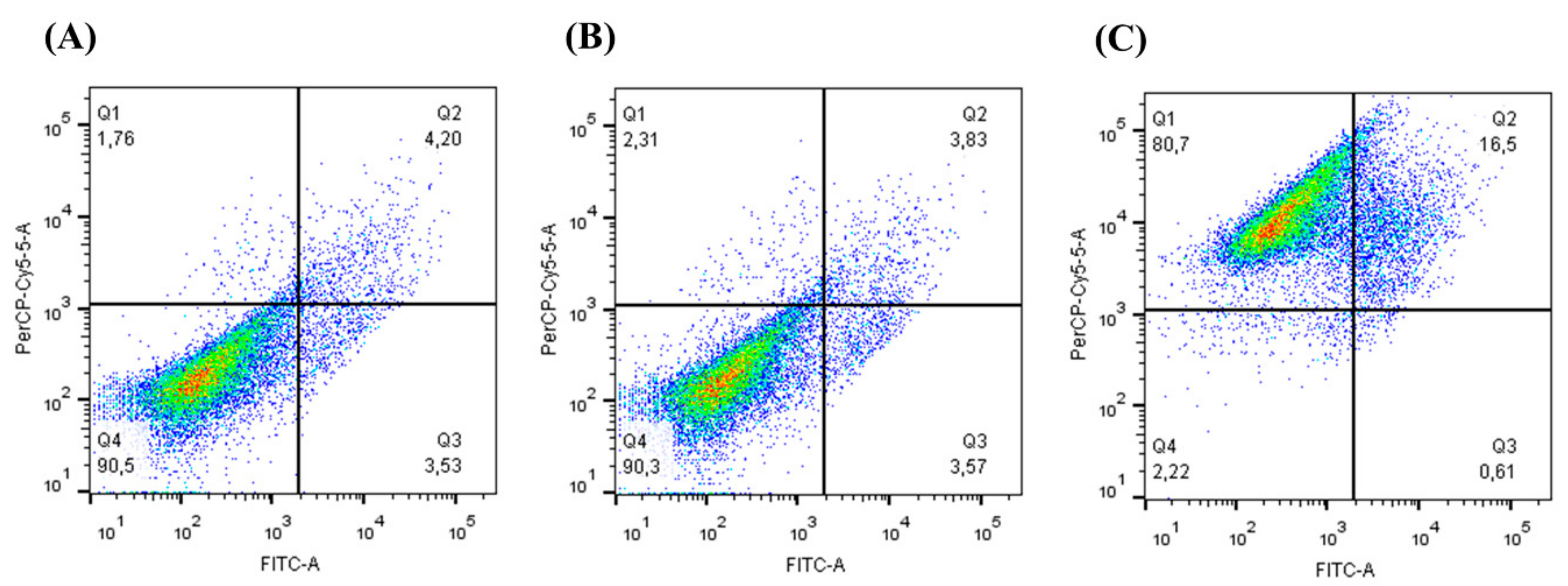
3.3. Cell Viability
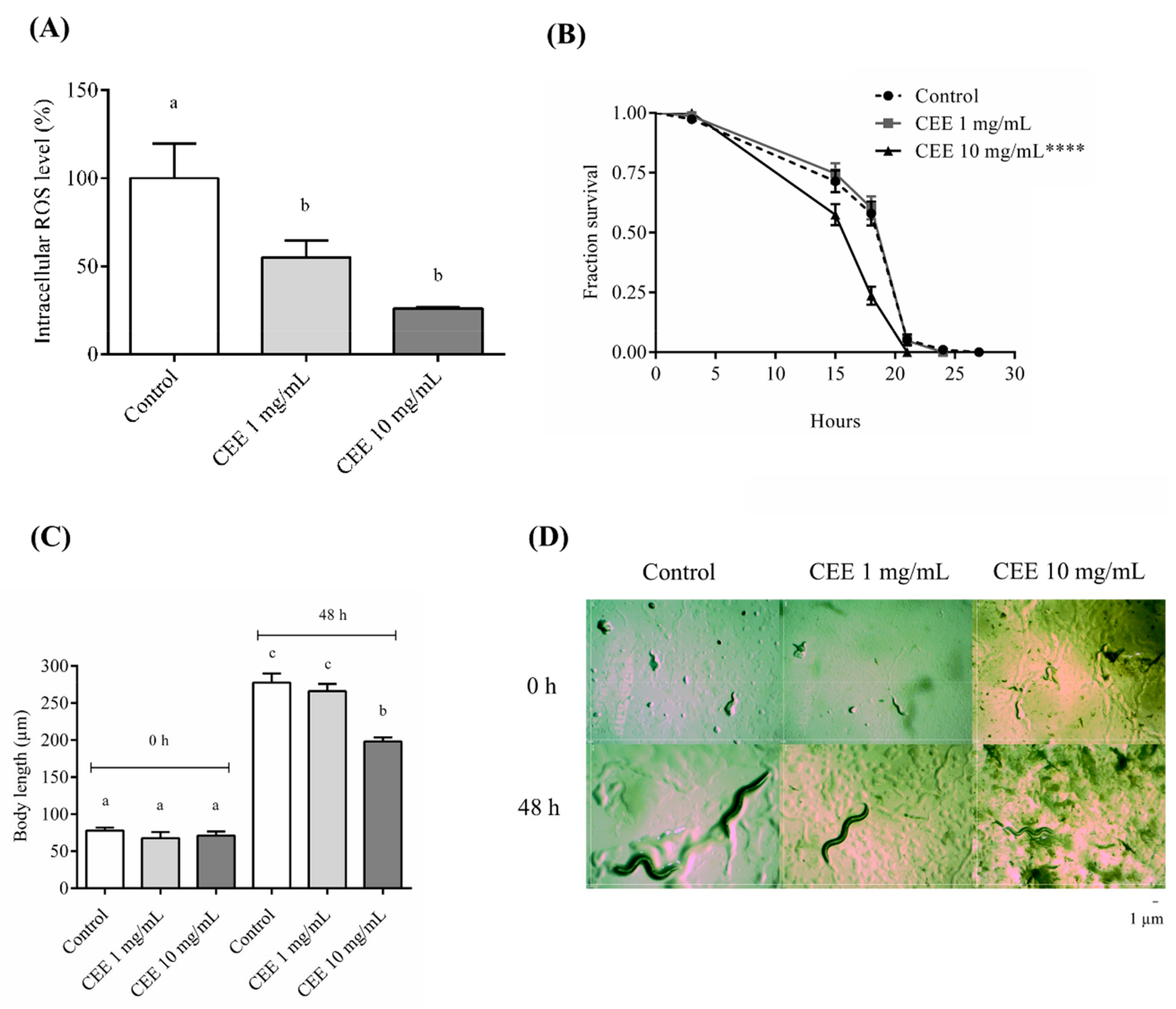
3.4. Antioxidant Assays In Vivo Using C. elegans
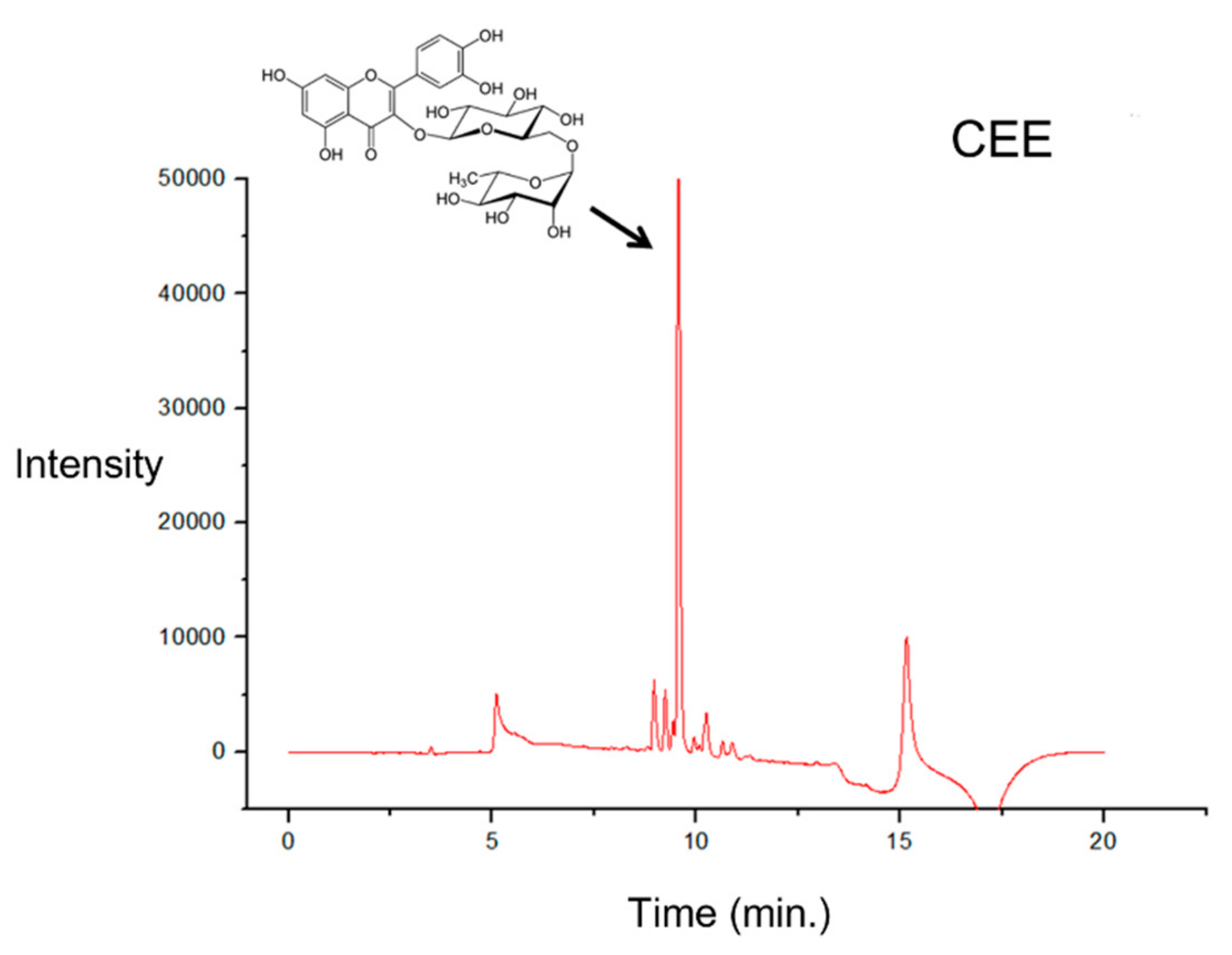
3.5. UPLC Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sponchiado, G.; Adam, M.L.; Silva, C.D.; Soley, B.S.; de Mello-Sampayo, C.; Cabrini, D.A.; Correr, C.J.; Otuki, M.F. Quantitative genotoxicity assays for analysis of medicinal plants: A systematic review. J. Ethnopharmacol. 2016, 178, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Armendáriz-Barragán, B.; Zafar, N.; Badri, W.; Galindo-Rodríguez, S.A.; Kabbaj, D.; Fessi, H.; Elaissari, A. Plant extracts: From encapsulation to application. Expert Opin. Drug Deliv. 2016, 13, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
- Rubió, L.; Motilva, M.J.; Romero, M.P. Recent Advances in Biologically Active Compounds in Herbs and Spices: A Review of the Most Effective Antioxidant and Anti-Inflammatory Active Principles. Crit. Rev. Food Sci. Nutr. 2013, 53, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Borges, L.L.; Conceição, E.C.; Silveira, D. Active compounds and medicinal properties of Myrciaria genus. Food Chem. 2014, 153, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Persson, T.; Popescu, B.O.; Cedazo-Minguez, A. Oxidative stress in Alzheimer’s disease: Why did antioxidant therapy fail. Oxid. Med. Cell Longev. 2014, 1–11. [Google Scholar] [CrossRef]
- Roleira, F.M.F.; Tavares-Da-Silva, E.J.; Varela, C.L.; Costa, S.C.; Silva, T.; Garrido, J.; Borges, F. Plant derived and dietary phenolic antioxidants: Anticancer properties. Food Chem. 2015, 183, 235–258. [Google Scholar] [CrossRef]
- Olas, B. Berry phenolic antioxidants—implications for human health? Front. Pharmacol. 2018, 9. [Google Scholar] [CrossRef]
- Chirinos, R.; Galarza, J.; Betalleluz-Pallardel, I.; Pedreschi, R.; Campos, D. Antioxidant compounds and antioxidant capacity of Peruvian camu camu (Myrciaria dubia (H.B.K.) McVaugh) fruit at different maturity stages. Food Chem. 2010, 120, 1019–1024. [Google Scholar] [CrossRef]
- Morales, P.; Barros, L.; Dias, M.I.; Santos-Buelga, C.; Ferreira, I.C.; Asquieri, E.R.; Berrios, J. Non-fermented and fermented jabuticaba (Myrciaria cauliflora Mart.) pomaces as valuable sources of functional ingredients. Food Chem. 2016, 208, 220–227. [Google Scholar] [CrossRef]
- Tietbohl, L.A.C.; Oliveira, A.P.; Esteves, R.S.; Albuquerque, R.D.D.G.; Folly, D.; Machado, F.P.; Corrêa, A.L.; Santos, M.G.; Ruiz, A.L.G.; Rocha, L. Antiproliferative activity in tumor cell lines, antioxidant capacity and total phenolic, flavonoid and tannin contents of Myrciaria floribunda. An. Acad. Bras. Ciênc. 2017, 1–10. [Google Scholar] [CrossRef]
- Oliveira, L.M.; Porte, A.; Godoy, R.L.O.; Souza, M.C.; Pacheco, S.; Santiago, M.C.P.A.; Gouvêa, A.C.M.S.; Nascimento, L.S.M.; Borguini, R.G. Chemical characterization of Myrciaria floribunda (H. West ex Willd) fruit. Food Chem. 2018, 248, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Sobral, M.; Grippa, C.R.; Souza, M.C.; Aguiar, O.T.; Bertoncello, R.; Guimarães, T.B. Fourteen new species and two taxonomic notes on Brazilian Myrtaceae. Phytotaxa 2012, 50, 19–50. [Google Scholar] [CrossRef]
- Alice, C.B.; Vargas, V.M.; Silva, G.A.; Siqueira, N.C.; Schapoval, E.E.; Gleye, J.; Henriques, J.A.; Henriques, A.T. Screening of plants used in South Brazil folk medicine. J. Ethnopharmacol. 1991, 35, 165–171. [Google Scholar] [CrossRef]
- Apel, M.A.; Lima, M.E.; Sobral, M.; Young, M.C.; Cordeiro, I.; Schapoval, E.E.; Henriques, A.T.; Moreno, P.R. Anti-inflammatory activity of essential oil from leaves of Myrciaria tenella and Calycorectes sellowianus. Pharm. Biol. 2010, 48, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Athukorala, Y.; Kim, K.N.; Jeon, Y.J. Antiproliferative and antioxidant properties of an enzymatic hydrolysate from brown alga, Ecklonia cava. Food Chem. Toxicol. 2006, 44, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Wagner, H.M.; Bladt, S.; Zgainski, E.M. Plant Drug Analysis; Springer: New York, NY, USA, 1984; 320p. [Google Scholar]
- Nascimento, A.K.L.; Melo-Silveira, R.F.; Dantas-Santos, N.; Fernandes, J.M.; Zucolotto, S.M.; Rocha, H.A.O.; Scortecci, K.C. Antioxidant and Antiproliferative Activities of Leaf Extracts from Plukenetia volubilis Linneo (Euphorbiaceae). Evid.-Based Complement. Altern. Med. 2013, 1–10. [Google Scholar] [CrossRef]
- Melo-Silveira, R.F.; Viana, R.L.S.; Sabry, D.A.; da Silva, R.A.; Machado, D.; Nascimento, A.K.L.; Scortecci, K.C.; Ferreira-Halder, C.V.; Sassaki, G.L.; Rocha, H.A.O. Antiproliferative xylan from corn cobs induces apoptosis in tumor cells. Carbohydr. Polym. 2019, 210, 245–253. [Google Scholar] [CrossRef]
- Brenner, S. The genetics of Caenorhabditis elegans. Genetics 1974, 77, 71–94. [Google Scholar]
- Fracassetti, D.; Costa, C.; Moulay, L.; Tomás-Barberán, F.A. Ellagic acid derivatives, ellagitannins, proanthocyanidins and other phenolics, vitamin C and antioxidant capacity of two powder products from camu-camu fruit (Myrciaria dubia). Food Chem. 2013, 139, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Losada-Barreiro, S.; Bravo-Díaz, C. Free radicals and polyphenols: The redox chemistry of neurodegenerative diseases. Eur. J. Med. Chem. 2017, 133, 379–402. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Schneider, N.F.; Moura, N.F.; Colpo, T.; Marins, K.; Marangoni, C.; Flach, A. Estudo dos compostos voláteis e atividade antimicrobiana da Myrciaria tenella (cambuí). Rev. Bras. Farmacol. 2008, 89, 131–133. [Google Scholar]
- Hussain, H.; Green, I.R.; Ali, I.; Khan, I.A.; Ali, Z.; Al-Sadi, A.M.; Ahmed, I. Ursolic acid derivatives for pharmaceutical use: A patent review (2012–2016). Expert Opin. Ther. Pat. 2017. [Google Scholar] [CrossRef] [PubMed]
- Pironi, A.M.; de Araújo, P.R.; Fernandes, M.A.; Salgado, H.R.N.; Chorilli, M. Characteristics, biological properties and analytical methods of ursolic acid: A review. Crit. Rev. Anal. Chem. 2018, 48, 86–93. [Google Scholar] [CrossRef]
- Tan, W.; Lu, J.; Huang, M.; Li, Y.; Chen, M.; Wu, G.; Gong, J.; Zhong, Z.; Xu, Z.; Dang, Y.; et al. Anti-cancer natural products isolated from chinese medicinal herbs. Chin. Med. J. 2011, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.Y.; Lee, S.R.; Heo, J.W.; No, M.H.; Rhee, B.D.; Ko, K.S.; Kwak, H.B.; Han, J. Ursolic acid in health and disease. Korean J. Physiol. Pharmacol. 2018, 22, 235–248. [Google Scholar] [CrossRef] [Green Version]
- Lorent, J.H.; Quetin-Leclercq, J.; Mingeot-Leclercq, M.P. The amphiphilic nature of saponins and their effects on artificial and biological membranes and potential consequences for red blood and cancer cells. Org. Biomol. Chem. 2014, 12, 8803–8822. [Google Scholar] [CrossRef] [PubMed]
- Reynertson, K.A.; Yang, H.; Jiang, B.; Basile, M.J.; Kennelly, E.J. Quantitative analysis of antiradical phenolic constituents from fourteen edible Myrtaceae fruits. Food Chem. 2008, 109, 883–890. [Google Scholar] [CrossRef] [Green Version]
- Souza, C.G.; Andrade, D.M.L.; Jordão, J.B.R.; Ávila, R.I.; Borges, L.L.; Vaz, B.G.; Valadares, M.C.; Gil, E.S.; Conceição, E.C.; Rocha, M.L. Radical scavenger capacity of jabuticaba fruit (Myrciaria cauliflora) and its biological effects in hypertensive rats. Oxid. Med. Cell Longev. 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Somogyi, A.; Rosta, K.; Pusztai, P.; Tulassay, Z.; Nagy, G. Antioxidant measurements. Physiol. Meas. 2007, 28, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Dusek, P.; Schneider, S.A.; Aaseth, J. Iron chelation in the treatment of neurodegenerative diseases. J. Trace Elem. Med. Biol. 2016, 38, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Giampietro, R.; Spinelli, F.; Contino, M.; Colabufo, N.A. The pivotal role of copper in neurodegeneration: A new strategy for the therapy of neurodegenerative disorders. Mol. Pharm. 2018, 15, 808–820. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.H.; Tyan, Y.C.; Chen, Z.S.; Lin, C.L.; Yang, M.H.; Yuan, S.S.; Tsai, W.C. Evaluation of the antioxidant activity and antiproliferative effect of the jaboticaba (Myrciaria cauliflora) seed extracts in oral carcinoma cells. Biomed Res. Int. 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Benfeitas, R.; Uhlen, M.; Nielsen, J.; Mardinoglu, A. New challenges to study heterogeneity in cancer redox metabolism. Front. Cell Dev. Biol. 2017, 5, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Alu’datt, M.H.; Rababah, T.; Alhamad, M.N.; Al-Rabadi, G.J.; Tranchant, C.C.; Almajwal, A.; Kubow, S.; Alli, I. Occurrence, types, properties and interactions of phenolic compounds with other food constituents in oil-bearing plants. Crit. Rev. Food Sci. Nutr. 2017, 13, 1–10. [Google Scholar] [CrossRef]
- Meng, M.B.; Wang, H.H.; Cui, Y.L.; Wu, Z.Q.; Shi, Y.Y.; Zaorsky, N.G.; Deng, L.; Yuan, Z.Y.; Lu, Y.; Wang, P. Necroptosis in tumorigenesis, activation of anti-tumor immunity, and cancer therapy. Oncotarget 2016, 7, 57391–57413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Philipp, S.; Sosna, J.; Adam, D. Cancer and necroptosis: Friend or foe? Cell. Mol. Life Sci. 2016, 73, 2183–2193. [Google Scholar] [CrossRef]
- Ganeshpurkar, A.; Saluja, A.K. The pharmacological potential of rutin. Saudi Pharm. J. 2017, 25, 149–164. [Google Scholar] [CrossRef]
- Gautam, R.; Singh, M.; Gautam, S.; Rawat, J.K.; Saraf, S.A.; Kaithwas, G. Rutin attenuates intestinal toxicity induced by Methotrexate linked with anti-oxidative and anti-inflammatory effects. BMC Complement. Altern. Med. 2016, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Neves, N.A.; Stringheta, P.C.; Gómez-Alonso, S.; Hermosín-Gutiérrez, I. Flavonols and ellagic acid derivatives in peels of different species of jabuticaba (Plinia spp.) identified by HPLC-DAD-ESI/MS. Food Chem. 2018, 252, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Alajmi, M.F.; Alam, P.; Rehman, M.T.; Husain, F.M.; Khan, A.A.; Siddiqui, N.A.; Hussain, A.; Kalam, M.A.; Parvez, M.K. Interspecies anticancer and antimicrobial activities of genus Solanum and estimation of rutin by validated UPLC-PDA Method. Evid.-Based Complement. Altern. Med. 2018, 2018, 6040815. [Google Scholar] [CrossRef] [PubMed]
- Grünz, G.; Haas, K.; Soukup, S.; Klingenspor, M.; Kulling, S.E.; Daniel, H.; Spanier, B. Structural features and bioavailability of four flavonoids and their implications for lifespan-extending and antioxidant actions in C. elegans. Mech. Ageing Dev. 2012, 133, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kampkötter, A.; Nkwonkam, C.G.; Zurawski, R.F.; Timpel, C.; Chovolou, Y.; Wätjen, W.; Kahl, R. Investigations of protective effects of the flavonoids quercetin and rutin on stress resistance in the model organism Caenorhabditis elegans. Toxicology 2007, 234, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H. Bacteria and the Aging and Longevity of Caenorhabditis elegans. Annu. Rev. Genet. 2013, 47, 233–246. [Google Scholar] [CrossRef]
- Kalmobé, J.; Ndjonka, D.; Boursou, D.; Vildina, J.D.; Liebau, E. Phytochemical analysis and in vitro anthelmintic activity of Lophira lanceolata (Ochnaceae) on the bovine parasite Onchocerca ochengi and on drug resistant strains of the free-living nematode Caenorhabditis elegans. BMC Complement. Altern. Med. 2017, 17, 1–12. [Google Scholar] [CrossRef]
- Massamha, B.; Gadzirayi, C.T.; Mukutirwa, I. Efficacy of Allium sativum (garlic) controlling nematode parasites in sheep. Int. J. Appl. Res. Vet. Med. 2010, 8, 161–169. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, A.R.C.; Cordeiro, M.L.d.S.; Silva, L.M.P.; Cadavid, C.O.M.; Caland, R.B.d.O.; Fernandes-Negreiros, M.M.; Queiroz, M.F.; Barbosa, J.d.S.; Aragão, C.F.S.; Zucolotto, S.M.; et al. Myrciaria tenella (DC.) O. Berg (Myrtaceae) Leaves as a Source of Antioxidant Compounds. Antioxidants 2019, 8, 310. https://doi.org/10.3390/antiox8080310
Ribeiro ARC, Cordeiro MLdS, Silva LMP, Cadavid COM, Caland RBdO, Fernandes-Negreiros MM, Queiroz MF, Barbosa JdS, Aragão CFS, Zucolotto SM, et al. Myrciaria tenella (DC.) O. Berg (Myrtaceae) Leaves as a Source of Antioxidant Compounds. Antioxidants. 2019; 8(8):310. https://doi.org/10.3390/antiox8080310
Chicago/Turabian StyleRibeiro, Ana Raquel Carneiro, Maria Lúcia da Silva Cordeiro, Larissa Marina Pereira Silva, Cesar Orlando Munoz Cadavid, Ricardo Basílio de Oliveira Caland, Marília Medeiros Fernandes-Negreiros, Moacir Fernandes Queiroz, Jefferson da Silva Barbosa, Cicero Flavio Soares Aragão, Silvana Maria Zucolotto, and et al. 2019. "Myrciaria tenella (DC.) O. Berg (Myrtaceae) Leaves as a Source of Antioxidant Compounds" Antioxidants 8, no. 8: 310. https://doi.org/10.3390/antiox8080310
APA StyleRibeiro, A. R. C., Cordeiro, M. L. d. S., Silva, L. M. P., Cadavid, C. O. M., Caland, R. B. d. O., Fernandes-Negreiros, M. M., Queiroz, M. F., Barbosa, J. d. S., Aragão, C. F. S., Zucolotto, S. M., Oliveira, R. d. P., Rocha, H. A. O., & Scortecci, K. C. (2019). Myrciaria tenella (DC.) O. Berg (Myrtaceae) Leaves as a Source of Antioxidant Compounds. Antioxidants, 8(8), 310. https://doi.org/10.3390/antiox8080310






