The Modern Use of an Ancient Plant: Exploring the Antioxidant and Nutraceutical Potential of the Maltese Mushroom (Cynomorium Coccineum L.)
Abstract
:1. Introduction
2. Botany and Folk Medicine
3. Phytochemistry
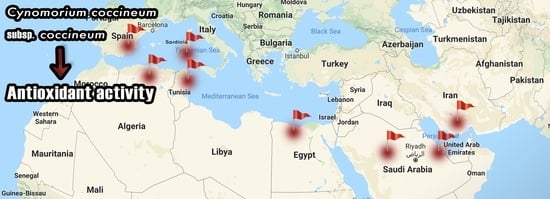
4. Antioxidant Activity
5. Biological Activity
5.1. Inhibition of Cancer Cell Proliferation
5.2. Activity on the Reproductive System
5.3. Anti-Tyrosinase Activity
5.4. Effects on the Cardiovascular System
5.5. Antimicrobial Activity
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Traka, M.H.; Mithen, R.F. Plant science and human nutrition: Challenges in assessing health-promoting properties of phytochemicals. Plant Cell 2011, 23, 2483–2497. [Google Scholar] [CrossRef] [PubMed]
- Lall, N.; Kishore, N. Are plants used for skin care in South Africa fully explored? J. Ethnopharmacol. 2014, 153, 61–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ballero, M. Le piante e la Bibbia; Carlo Delfino Editore: Sassari, Italy, 2016; p. 370. [Google Scholar]
- Abd El-Rahman, H.A.; El-Badry, A.A.; Mahmoud, O.M.; Harraz, F.A. The effect of the aqueous extract of Cynomorium coccineum on the epididymal sperm pattern of the rat. Phytother. Res. 1999, 13, 248–250. [Google Scholar] [CrossRef]
- Al-humaidi, J. Phytochemical screening, total phenolic and antioxidant activity of crude and fractionated extracts of Cynomorium coccineum growing in Saudi Arabia. Eur. J. Med. Plants 2016, 11, 1–9. [Google Scholar] [CrossRef]
- Al-Qarawi, A.A.; Abdel-Rahman, H.A.; El-Badry, A.A.; Harraz, F.; Razig, N.A.; Abdel-Magied, E.M. The effect of extracts of Cynomorium coccineum and Withania somnifera on gonadotrophins and ovarian follicles of immature wistar rats. Phytother. Res. 2000, 14, 288–290. [Google Scholar] [CrossRef]
- Zucca, P.; Rosa, A.; Tuberoso, C.I.G.; Piras, A.; Rinaldi, A.C.; Sanjust, E.; Dessì, M.A.; Rescigno, A. Evaluation of antioxidant potential of “Maltese mushroom” (Cynomorium coccineum) by means of multiple chemical and biological assays. Nutrients 2013, 5, 149–161. [Google Scholar] [CrossRef] [PubMed]
- WCSP. World Checklist of Selected Plant Families. Facilitated by the Royal Botanic Gardens, Kew. 2019. Available online: http://wcsp.science.kew.org/prepareChecklist.do?checklist=selected_families%40%40218060820190942213 (accessed on 6 August 2019).
- Cui, Z.; Guo, Z.; Miao, J.; Wang, Z.; Li, Q.; Chai, X.; Li, M. The genus Cynomorium in China: An ethnopharmacological and phytochemical review. J. Ethnopharmacol. 2013, 147, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bellot, S.; Cusimano, N.; Luo, S.; Sun, G.; Zarre, S.; Gröger, A.; Temsch, E.; Renner, S.S. Assembled Plastid and Mitochondrial Genomes, as well as Nuclear Genes, Place the Parasite Family Cynomoriaceae in the Saxifragales. Genome Biol. Evol. 2016, 8, 2214–2230. [Google Scholar] [CrossRef] [Green Version]
- Cusimano, N.; Renner, S.S. Sequential horizontal gene transfers from different hosts in a widespread Eurasian parasitic plant, Cynomorium coccineum. Am. J. Bot. 2019, 106, 679–689. [Google Scholar] [CrossRef]
- Leonard, J. Observations sur le genre Cynomorium L. en Asie (Cynomoriaceae). Bull. Jard. Bot. Natl. Belg. 1986, 56, 301–304. [Google Scholar] [CrossRef]
- Nickrent, D.L.; Der, J.P.; Anderson, F.E. Discovery of the photosynthetic relatives of the “Maltese mushroom” Cynomorium. BMC Evol. Biol. 2005, 5. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B.; Saito, N.; Detoni, C.H. Anthocyanins of Cephaelis, Cynomorium, Euterpe, Lavatera and Pinanga. Biochem. Syst. Ecol. 1994, 22, 835–836. [Google Scholar] [CrossRef]
- Flannery, M. Avicenna—Persian Philosopher and Scientist. Available online: https://www.britannica.com/biography/Avicenna#accordion-article-history (accessed on 15 May 2019).
- Avicenna. The Canon of Medicine (al-Qānūn fī’l-tibb). The Law of Natural Healing; Great Books of the Islamic World Inc.: Chicago, IL, USA, 2014; Volume 5. [Google Scholar]
- Lanfranco, G.G. Cynomorium coccineum Linn., a Maltese historical plant. J. Malta. Historic Soc. 1960, 3, 53–70. [Google Scholar]
- Boccone, P. Icones & descriptiones rariorum plantarum Siciliae, Melitae, Galliae, & Italiae; E. Theatro Sheldoniano: Madrid, Spain, 1674. [Google Scholar]
- Ghrabi, Z. Cynomorium coccineum L. In A guide to medicinal plants in North Africa; IUCN, I.U.f.C.o.N.a.N.R., Centre for Mediterranean Cooperation: Malaga, Spain, 2005; pp. 113–114. [Google Scholar]
- Hakim, I.-S. Avicenna Canon of Medicine. In Book II Materia Medica. English Translation of the Critical Arabic Text; Department of Islamic Studies, Hamdard University: New Delhi, India, 1998. [Google Scholar]
- Borg, J. Descriptive Flora of the Maltese islands; Government Printing Office: Valeta, Malta, 1927.
- De Soldanis, G.P.F. Della Lingua Punica Presentemente usata da Maltesi; per Generoso Salomoni alla Piazza di, S. Ignazio; Si vendono in Malta: Roma, Italy, 1750; p. 208. [Google Scholar]
- De-Boisgelin, P.M.L. Malte Ancienne et Moderne; R. Phillips: London, UK, 1805; Volume 1. [Google Scholar]
- Duke, J.A. Duke’s Handbook of Medicinal Plants of the Bible, 1st ed.; CRC Press: Boca Raton, FL, USA, 2008; p. 528. [Google Scholar]
- Harraz, F.M.; Pedersen, A.T.; Andersen, O.M. Anthocyanins from the Parasitic Medicinal Plant Cynomarium coccineum. Alex. J. Pharm. Sci. 1996, 10, 159–160. [Google Scholar]
- Meng, H.-C.; Wang, S.; Li, Y.; Kuang, Y.-Y.; Ma, C.-M. Chemical constituents and pharmacologic actions of Cynomorium plants. Chin. J. Nat. Med. 2013, 11, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Rached, W.; Benamar, H.; Bennaceur, M.; Marouf, A. Screening of the antioxidant potential of some Algerian indigenous plants. J. Biol. Sci. 2010, 10, 316–324. [Google Scholar] [CrossRef]
- Zucca, P.; Argiolas, A.; Nieddu, M.; Pintus, M.; Rosa, A.; Sanna, F.; Sollai, F.; Steri, D.; Rescigno, A. Biological Activities and Nutraceutical Potentials of Water Extracts from Different Parts of Cynomorium coccineum L. (Maltese Mushroom). Pol. J. Food Nutr. Sci. 2016, 66, 179–188. [Google Scholar] [CrossRef]
- Jabli, M. Extraction of Eco-Friendly Natural Dyes from Tradescantia pallida Purpurea and Cynomorium coccineum Growing Naturally in Tunisia. Trends Textile Eng. Fashion Technol. 2018, 1, 1–4. [Google Scholar] [CrossRef]
- Zucca, P.; Rescigno, A.; Pintus, M.; Rinaldi, A.C.C.; Sanjust, E. Degradation of textile dyes using immobilized lignin peroxidase-like metalloporphines under mild experimental conditions. Chem. Cent. J. 2012, 6. [Google Scholar] [CrossRef]
- Nayeem, N.; Karvekar, M.D. Stability studies and evaluation of the semi solid dosage form of the rutin, quercitin, ellagic acid, gallic acid and sitosterol isolated from the leaves of Tectona grandis for wound healing activity. Arch. Appl. Sci. Res. 2011, 3, 43–51. [Google Scholar]
- Sheng, Z.; Yan, X.; Zhang, R.; Ni, H.; Cui, Y.; Ge, J.; Shan, A. Assessment of the antidiarrhoeal properties of the aqueous extract and its soluble fractions of Chebulae Fructus (Terminalia chebula fruits). Pharm. Biol. 2016, 54, 1847–1856. [Google Scholar] [CrossRef]
- Ben Attia, I.; Zucca, P.; Marincola, F.C.; Piras, A.; Rosa, A.; Chaieb, M.; Rescigno, A. Chemical composition and antioxidant potential differences between Cynomorium coccineum L. growing in Italy and in Tunisia: Effect of environmental stress. Diversity 2018, 10. [Google Scholar] [CrossRef]
- Rosa, A.; Rescigno, A.; Piras, A.; Atzeri, A.; Scano, P.; Porcedda, S.; Zucca, P.; Assunta Dessì, M. Chemical composition and effect on intestinal Caco-2 cell viability and lipid profile of fixed oil from Cynomorium coccineum L. Food Chem. Toxicol. 2012, 50, 3799–3807. [Google Scholar] [CrossRef]
- Ghanbari, F.; Sayyari, M. Controlled drought stress affects the chilling-hardening capacity of tomato seedlings as indicated by changes in phenol metabolisms, antioxidant enzymes activity, osmolytes concentration and abscisic acid accumulation. Sci. Hortic. 2018, 229, 167–174. [Google Scholar] [CrossRef]
- Benjamin, J.J.; Lucini, L.; Jothiramshekar, S.; Parida, A. Metabolomic insights into the mechanisms underlying tolerance to salinity in different halophytes. Plant Physiol. Biochem. 2019, 135, 528–545. [Google Scholar] [CrossRef]
- Rosa, A.; Nieddu, M.; Piras, A.; Atzeri, A.; Putzu, D.; Rescigno, A. Maltese mushroom (Cynomorium coccineum L.) as source of oil with potential anticancer activity. Nutrients 2015, 7, 849–864. [Google Scholar] [CrossRef]
- Zhou, Y.B.; Ye, R.R.; Lu, X.F.; Lin, P.C.; Yang, S.B.; Yue, P.P.; Zhang, C.X.; Peng, M. GC-MS analysis of liposoluble constituents from the stems of Cynomorium songaricum. J. Pharm. Biomed. Anal. 2009, 49, 1097–1100. [Google Scholar] [CrossRef]
- Dharmananda, S. Cynomorium—Parasitic Plant Widely Used in Traditional Medicine. Available online: http://www.itmonline.org/arts/cynomorium.htm (accessed on 18 June 2015).
- Salehi, B.; Zucca, P.; Orhan, I.E.; Azzini, E.; Adetunji, C.O.; Mohammed, S.A.; Banerjee, S.K.; Sharopov, F.; Rigano, D.; Sharifi-Rad, J.; et al. Allicin and health: A comprehensive review. Trends Food Sci. Technol. 2019, 86, 502–516. [Google Scholar] [CrossRef]
- Davies, K.J. Oxidative stress: The paradox of aerobic life. Biochem. Soc. Symp. 1995, 61, 1–31. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Orrù, R.; Zucca, P.; Falzoi, M.; Atzori, E.; Rescigno, A.; Padiglia, A. First step towards the biomolecular characterization of Pompia, an endemic Citrus-like fruit from Sardinia (Italy). Plant Biosyst. 2017, 151, 464–473. [Google Scholar] [CrossRef]
- Belkhir, M.; Dhaouadi, K.; Rosa, A.; Atzeri, A.; Nieddu, M.; Tuberoso, C.I.G.; Rescigno, A.; Amri, M.; Fattouch, S. Protective effects of azarole polyphenolic extracts against oxidative damage using in vitro biomolecular and cellular models. Ind. Crop. Prod. 2016, 86, 239–250. [Google Scholar] [CrossRef]
- Salehi, B.; Armstrong, L.; Rescigno, A.; Yeskaliyeva, B.; Seitimova, G.; Beyatli, A.; Sharmeen, J.; Mahomoodally, F.M.; Sharopov, F.; Durazzo, A.; et al. Lamium Plants—A Comprehensive Review on Health Benefits and Biological Activities. Molecules 2019, 24, 1913. [Google Scholar] [CrossRef]
- De la Rosa, L.A.; Alvarez-Parrilla, E.; Gonzalez-Aguilar, G.A. Fruit and Vegetable Phytochemicals: Chemistry, Nutritional Value and Stability; Wiley-Blackwell: Singapore, 2010. [Google Scholar]
- Zucca, P.; Sanjust, E.; Trogu, E.; Sollai, F.; Rescigno, A. Evaluation of antioxidant capacity of antioxidant-declared beverages marketed in Italy. It. J. Food Sci. 2010, 22, 313–319. [Google Scholar]
- Aires, A. Phenolics in Foods: Extraction, Analysis and Measurements. In Phenolic Compounds—Natural Sources, Importance and Applications; Soto-Hernández, M., Ed.; IntechOpen: London, UK, 2017. [Google Scholar] [Green Version]
- Salehi, B.; Fokou, V.P.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The Therapeutic Potential of Naringenin: A Review of Clinical Trials. Pharmaceuticals 2019, 12, 11. [Google Scholar] [CrossRef]
- Gonçalves, M.J.; Piras, A.; Porcedda, S.; Marongiu, B.; Falconieri, D.; Cavaleiro, C.; Rescigno, A.; Rosa, A.; Salgueiro, L. Antifungal activity of extracts from Cynomorium coccineum growing wild in Sardinia island (Italy). Nat. Prod. Res. 2015, 29, 2247–2250. [Google Scholar] [CrossRef]
- Griffin, S.; Alkhayer, R.; Mirzoyan, S.; Turabyan, A.; Zucca, P.; Sarfraz, M.; Nasim, M.; Trchounian, A.; Rescigno, A.; Keck, C.; et al. Nanosizing Cynomorium: Thumbs up for Potential Antifungal Applications. Inventions 2017, 2, 24. [Google Scholar] [CrossRef]
- Abdel-Magied, E.M.; Abdel-Rahman, H.A.; Harraz, F.M. The effect of aqueous extracts of Cynomorium coccineum and Withania somnifera on testicular development in immature Wistar rats. J. Ethnopharmacol. 2001, 75, 1–4. [Google Scholar] [CrossRef]
- Sdiri, M.; Li, X.; Du, W.; El-Bok, S.; Xie, Y.-Z.; Ben-Attia, M.; Yang, B. Anticancer Activity of Cynomorium coccineum. Cancers 2018, 10, 354. [Google Scholar] [CrossRef]
- Ashour, O.M.; Abdel-Naim, A.B.; Abdallah, H.M.; Nagy, A.A.; Mohamadin, A.M.; Abdel-Sattar, E.A. Evaluation of the potential cardioprotective activity of some Saudi plants against Doxorubicin toxicity. Z. Naturforsch. C J. Biosci. 2012, 67, 297–307. [Google Scholar] [CrossRef]
- Ikram, M.; Dar, M.S.; Fakouhi, T. Hypotensive agent from Cynomorium coccineum. Pahlavi Med. J. 1978, 9, 167–181. [Google Scholar]
- Liu, C.; Lin, J.-J.; Yang, Z.-Y.; Tsai, C.-C.; Hsu, J.-L.; Wu, Y.-J. Proteomic study reveals a co-occurrence of gallic acid-induced apoptosis and glycolysis in B16F10 melanoma cells. J. Agric. Food Chem. 2014, 62, 11672–11680. [Google Scholar] [CrossRef]
- Badhani, B.; Sharma, N.; Kakkar, R. Gallic acid: a versatile antioxidant with promising therapeutic and industrial applications. RSC Adv. 2015, 5, 27540–27557. [Google Scholar] [CrossRef]
- Flurkey, A.; Cooksey, J.; Reddy, A.; Spoonmore, K.; Rescigno, A.; Inlow, J.; Flurkey, W.H. Enzyme, protein, carbohydrate, and phenolic contaminants in commercial tyrosinase preparations: Potential problems affecting tyrosinase activity and inhibition studies. J. Agric. Food Chem. 2008, 56, 4760–4768. [Google Scholar] [CrossRef]
- Rescigno, A.; Casañola-Martin, G.M.; Sanjust, E.; Zucca, P.; Marrero-Ponce, Y. Vanilloid derivatives as tyrosinase inhibitors driven by virtual screening-based QSAR models. Drug Test. Anal. 2011, 3, 176–181. [Google Scholar] [CrossRef]
- Rescigno, A.; Zucca, P.; Flurkey, A.; Inlow, J.; Flurkey, W.H. Identification and discrimination between some contaminant enzyme activities in commercial preparations of mushroom tyrosinase. Enzyme Microb. Technol. 2007, 41, 620–627. [Google Scholar] [CrossRef]
- Rescigno, A.; Bruyneel, F.; Padiglia, A.; Sollai, F.; Salis, A.; Marchand-Brynaert, J.; Sanjust, E. Structure-activity relationships of various amino-hydroxy-benzenesulfonic acids and sulfonamides as tyrosinase substrates. Biochim. Biophys. Acta Gen. Subj. 2011, 1810, 799–807. [Google Scholar] [CrossRef]
- Casañola-Martin, G.M.; Le-Thi-Thu, H.; Marrero-Ponce, Y.; Castillo-Garit, J.A.; Torrens, F.; Rescigno, A.; Abad, C.; Khan, M.T.H. Tyrosinase enzyme: 1. An overview on a pharmacological target. Curr. Top. Med. Chem. 2014, 14, 1494–1501. [Google Scholar] [CrossRef]
- Burzio, L.A.; Burzio, V.A.; Pardo, J.; Burzio, L.O. In vitro polymerization of mussel polyphenolic proteins catalyzed by mushroom tyrosinase. Comp. Biochem. Physiol. B 2000, 126, 383–389. [Google Scholar] [CrossRef]
- Diana, A.; Reguzzoni, M.; Congiu, T.; Rescigno, A.; Sollai, F.; Raspanti, M. The byssus threads of Pinna nobilis: A histochemical and ultrastructural study. Eur. J. Histochem. 2017, 61, 280–296. [Google Scholar] [CrossRef]
- Le-Thi-Thu, H.; Casañola-Martín, G.M.; Marrero-Ponce, Y.; Rescigno, A.; Saso, L.; Parmar, V.S.; Torrens, F.; Abad, C. Novel coumarin-based tyrosinase inhibitors discovered by OECD principles-validated QSAR approach from an enlarged, balanced database. Mol. Divers. 2011, 15, 507–520. [Google Scholar] [CrossRef]
- Marrero-Ponce, Y.; Casañola-Martín, G.M.; Khan, M.T.H.; Torrens, F.; Rescigno, A.; Abad, C. Ligand-based computer-aided discovery of tyrosinase inhibitors. applications of the TOMOCOMD-CARDD method to the elucidation of new compounds. Curr. Pharm. Des. 2010, 16, 2601–2624. [Google Scholar] [CrossRef]
- Schlich, M.; Fornasier, M.; Nieddu, M.; Sinico, C.; Murgia, S.; Rescigno, A. 3-Hydroxycoumarin loaded vesicles for recombinant human tyrosinase inhibition in topical applications. Colloids Surf. B Biointerfaces 2018, 171, 675–681. [Google Scholar] [CrossRef]
- Lachowicz, J.I.; Nurchi, V.M.; Crisponi, G.; Pelaez, M.D.G.J.; Rescigno, A.; Stefanowicz, P.; Cal, M.; Szewczuk, Z. Metal coordination and tyrosinase inhibition studies with Kojic-βAla-Kojic. J. Inorg. Biochem. 2015, 151, 36–43. [Google Scholar] [CrossRef]
- Le-Thi-Thu, H.; Cardoso, G.C.; Casañola-Martin, G.M.; Marrero-Ponce, Y.; Puris, A.; Torrens, F.; Rescigno, A.; Abad, C. QSAR models for tyrosinase inhibitory activity description applying modern statistical classification techniques: A comparative study. Chemom. Intell. Lab. Syst. 2010, 104, 249–259. [Google Scholar] [CrossRef]
- Zucca, P.; Pintus, M.; Manzo, G.; Nieddu, M.; Steri, D.; Rinaldi, A.C. Antimicrobial, antioxidant and anti-tyrosinase properties of extracts of the Mediterranean parasitic plant Cytinus hypocistis. BMC Res. Notes 2015, 8. [Google Scholar] [CrossRef]
- Bradford, E. The Great Siege; Penguin Books: Harmondsworth, UK, 1964. [Google Scholar]
- Marín, L.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: Antimicrobial properties. BioMed. Res. Int. 2015, 2015. [Google Scholar] [CrossRef]
- Kang, M.S.; Oh, J.S.; Kang, I.C.; Hong, S.J.; Choi, C.H. Inhibitory effect of methyl gallate and gallic acid on oral bacteria. J. Microbiol. 2008, 46, 744–750. [Google Scholar] [CrossRef]
- Maisetta, G.; Batoni, G.; Caboni, P.; Esin, S.; Rinaldi, A.C.; Zucca, P. Tannin profile, antioxidant properties, and antimicrobial activity of extracts from two Mediterranean species of parasitic plant Cytinus. BMC Complement. Altern. Med. 2019, 19, 82. [Google Scholar] [CrossRef]

| Folk Medical Uses | Geographical Area | Preparation | Ref. |
|---|---|---|---|
| Hemorrhoids | Mediterranean Basin | decoction | [18,19] |
| Dysentery; diarrhea | Mediterranean Basin | decoction; infused in white wine | [20,21,22] |
| Bleeding | Malta Island | dried up | [17] |
| Blood vomiting | Italy | decoction | [18,22] |
| Healing | Italy | dried up | [23] |
| Blood pressure | Italy | decoction | [18] |
| Venereal diseases | Mediterranean Basin | infusion | [17] |
| To strengthen teeth | Italy | powdered | [18,22] |
| Compound Class | Name | Extraction Method | Ref. |
|---|---|---|---|
| Flavonoids | Cyanidin 3-O-glucopyranoside | EtOH-H2O-HOAc; H2O | [14] |
| Flavonoids | Cyanidin 3-O-rhamnosylglucoside | not available | [25] |
| Organic acids | Gallic acid | H2O | [7] |
| Organic acids | Betaine | MetOH, and H2O | [33] |
| Organic acids | Bicarboxylic acids | MetOH, and H2O | [33] |
| Saccharides | Monosaccharides | MetOH, and H2O | [33] |
| Lipids | Lipid profile | Fixed oil | [33,34] |
| Lipids | Lipid profile | CHCl3:MetOH | [28] |
| Other compounds | Aminoacidic profile | MetOH, and H2O | [33] |
| Other compounds | Protein content | H2O | [28] |
| Method of Extraction | Part of the Plant | [DPPH•] IC50 (µg·mL−1) | TPC (mg GA·g−1 of fde | FC (mg CE·g−1 of fde) | [ABTS•+] IC50 (µg·mL−1) | Ref. |
|---|---|---|---|---|---|---|
| EtOH | Aerial part | 4.09 ± 0.6 | 406.38 ± 2 | 109.47 ± 33 | n.a. | [27] |
| Water | Aerial part | 13.47 ± 2.2 | 75.69 ± 3.6 | 39.19 ± 2 | n.a. | [27] |
| MetOH | Whole plant (crude extract) | 40.0 | 259.30 ± 6.8 | d.n.q. | 30.0 | [5] |
| ButOH | Whole plant (crude extract) | 5.60 | 201.36 ± 7.4 | N.A. | 6.0 | [5] |
| MetOH | Aerial part | 54.20 ± 2.1 | 173.50 ± 5.1 | 40.34 ± 0.7 | 910 ± 0.1 | [7] |
| Water | Aerial part | 51.60 ± 3.2 | 108.87 ± 3.7 | 37.15 ± 0.6 | 890 ± 0.1 | [7] |
| Biological Activity | The Parts of the Plant Used and Mode of Administration | Type of Study | Origin of the Plant | Ref. |
|---|---|---|---|---|
| (a) Antioxidant | Aerial part (ethanol extract) | in vitro | Algeria | [27] |
| Whole plant (polar and non-polar extract) | in vitro | Saudi Arabia | [5] | |
| External layer (water extract) | in vitro | Italy (Sardinia) | [7] | |
| Aerial part (increasing polarity solvents extract) | in vitro | Italy; Tunisia | [33] | |
| (b) Antimicrobial | Whole plant (methanol extract; fixed oil) | in vitro | Italy (Sardinia) | [50] |
| Whole plant (nanoparticles preparation) | in vitro | Italy (Sardinia) | [51] | |
| Whole plant (water extract) | in vitro | Italy (Sardinia) | [28] | |
| (c) Anti-tyrosinase | Whole plant | in vitro | Italy (Sardinia) | [28] |
| (d) Spermatogenesis and sperm motility | Inner pulp of the plant (water extract) | in vivo | Saudi Arabia | [4] |
| (d) Ovarian increase and folliculogenesis | Whole plant (water extract) | in vivo | Saudi Arabia | [6] |
| (e) Testicular development and spermatogenesis | Whole plant (water extract through a stomach tube) | in vivo | Saudi Arabia | [52] |
| (f) Pro-erectile | Aerial part (water extract subcutaneously) | in vivo | Italy (Sardinia) | [28] |
| (g) Inhibitory effect on the growth of colon cancer Caco-2 cells | Aerial plant (CO2 SFE) | in vitro | Italy (Sardinia) | [34] |
| (h) Inhibitory effect on the growth of B16F10 cells | Aerial part | in vitro | Italy (Sardinia) | [28] |
| (i) Anticancer | Whole plant fixed oil) | in vitro | Italy (Sardinia) | [37] |
| Aerial part (water extract by peritoneal injection) | in vivo | Tunisia (Bizerte) | [53] | |
| (l) Anti-tyrosinase | Whole plant | in vitro | Italy (Sardinia) | [28] |
| (m) Cardioprotective | Aerial part (methanol extract by oral administration) | in vivo | Saudi Arabia | [54] |
| (n) Blood pressure | Whole plant (fresh juice by oral administration) | in vivo | Iran | [55] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zucca, P.; Bellot, S.; Rescigno, A. The Modern Use of an Ancient Plant: Exploring the Antioxidant and Nutraceutical Potential of the Maltese Mushroom (Cynomorium Coccineum L.). Antioxidants 2019, 8, 289. https://doi.org/10.3390/antiox8080289
Zucca P, Bellot S, Rescigno A. The Modern Use of an Ancient Plant: Exploring the Antioxidant and Nutraceutical Potential of the Maltese Mushroom (Cynomorium Coccineum L.). Antioxidants. 2019; 8(8):289. https://doi.org/10.3390/antiox8080289
Chicago/Turabian StyleZucca, Paolo, Sidonie Bellot, and Antonio Rescigno. 2019. "The Modern Use of an Ancient Plant: Exploring the Antioxidant and Nutraceutical Potential of the Maltese Mushroom (Cynomorium Coccineum L.)" Antioxidants 8, no. 8: 289. https://doi.org/10.3390/antiox8080289
APA StyleZucca, P., Bellot, S., & Rescigno, A. (2019). The Modern Use of an Ancient Plant: Exploring the Antioxidant and Nutraceutical Potential of the Maltese Mushroom (Cynomorium Coccineum L.). Antioxidants, 8(8), 289. https://doi.org/10.3390/antiox8080289