Comparison of the Total Polyphenol Content and Antioxidant Activity of Chocolate Obtained from Roasted and Unroasted Cocoa Beans from Different Regions of the World
Abstract
:1. Introduction
2. Methodology
2.1. Experimental Material
2.2. Analytical Methods
3. Results and Discussion
3.1. Determination of Total Polyphenols in Cocoa Beans
3.2. Determination of Total Polyphenols in Chocolates
3.3. Determination of the Ability of Extracts to Inactivate Stable DPPH Radicals in Cocoa Beans and Chocolates Derived Therefrom
4. Summary and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Caligiani, A.; Marseglia, A.; Prandi, B.; Palla, G.; Sforza, S. Influence of fermentation level and geographical origin on cocoa bean oligopeptide pattern. Food Chem. 2016, 211, 431–439. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific Opinion on the substantiation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilation pursuant to Article 13(5) of Regulation (EC) No 1924/2006. EFSA J. 2012, 10, 2809–2830. [Google Scholar] [CrossRef]
- Bertazzo, A.; Agnolin, F.; Comai, S.; Zancato, M.; Costa, C.V.; Seraglia, R.; Traldi, P. The protein profile of Theobroma cacao L. seeds as obtained by matrix-assisted laser desorption/ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2011, 25, 2035–2042. [Google Scholar] [CrossRef] [PubMed]
- Kruszewski, B.; Obiedziński, M.W. Multivariate analysis of essential elements in raw cocoa and processed chocolate mass materials from three different manufacturers. LWT 2018, 98, 113–123. [Google Scholar] [CrossRef]
- Khan, N.; Nicod, N.M. Biomarkers of cocoa consumption. In Chocolate and Health; Springer: Milano, Italy, 2012; pp. 33–40. [Google Scholar]
- Khan, N.; Khymenets, O.; Urpí-Sardà, M.; Tulipani, S.; Garcia-Aloy, M.; Monagas, M.; Mora-Cubillos, X.; Llorach, R.; Andres-Lacueva, C. Cocoa polyphenols and inflammatory markers of cardiovascular disease. Nutrients 2014, 6, 844–880. [Google Scholar] [CrossRef] [PubMed]
- Ioannone, F.; Di Mattia, C.D.; De Gregorio, M.; Sergi, M.; Serafini, M.; Sacchetti, G. Flavanols, proanthocyanidins and antioxidant activity changes during cocoa (Theobroma cacao L.) roasting as affected by temperature and time of processing. Food Chem. 2015, 174, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Medeiros, N.; Marder, R.K.; Farias Wohlenberg, M.; Funchal, C.; Dani, C. Total Phenolic Content and Antioxidant Activity of Different Types of Chocolate, Milk, Semisweet, Dark, and Soy, in Cerebral Cortex, Hippocampus, and Cerebellum of Wistar Rats. Biochem. Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Cinquanta, L.; Di Cesare, C.; Manoni, R.; Piano, A.; Roberti, P.; Salvatori, G. Mineral essential elements for nutrition in different chocolate products. Int. J. Food Sci. Nutr. 2016, 67, 773–778. [Google Scholar] [CrossRef]
- Giacometti, J.; Muhvić, D.; Pavletić, A.; Dudarić, L. Cocoa polyphenols exhibit antioxidant, anti-inflammatory, anticancerogenic, and anti-necrotic activity in carbon tetrachloride-intoxicated mice. J. Funct. Foods 2016, 23, 177–187. [Google Scholar] [CrossRef]
- Tsang, C.; Hodgson, L.; Bussu, A.; Farhat, G.; Al-Dujaili, E. Effect of Polyphenol-Rich Dark Chocolate on Salivary Cortisol and Mood in Adults. Antioxidants 2019, 8, 149. [Google Scholar] [CrossRef]
- Rusconi, M.; Conti, A. Theobroma cacao L., the Food of the Gods: A scientific approach beyond myths and claims. Pharmacol. Res. 2010, 61, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.L.; Doughty, K.; Ali, A. Cocoa and Chocolate in Human Health and Disease. Antioxid. Redox Sign. 2011, 15, 2779–2811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kothe, L.; Zimmermann, B.F.; Galensa, R. Temperature influences epimerization and composition of flavanol monomers, dimers and trimers during cocoa bean roasting. Food Chem. 2013, 141, 3656–3663. [Google Scholar] [CrossRef] [PubMed]
- Żyżelewicz, D.; Krysiak, W.; Oracz, J.; Sosnowska, D.; Budryn, G.; Nebesny, E. The influence of the roasting process conditions on the polyphenol content in cocoa beans, nibs and chocolates. Food Res. Int. 2016, 89, 918–929. [Google Scholar] [CrossRef]
- Dabas, D. Polyphenols as colorants. Adv. Food Technol. Nutr. Sci. Open J. 2016, SE, S1–S6. [Google Scholar] [CrossRef]
- Giacometti, J.; Jolić, S.M.; Josić, D. Cocoa Processing and Impact on Composition. In Processing and Impact on Active Components in Food; Elsevier Inc.: Amsterdam, Netherlands, 2014; pp. 605–612. [Google Scholar] [CrossRef]
- Zyzelewicz, D.; Budryn, G.; Oracz, J.; Antolak, H.; Kregiel, D.; Kaczmarska, M. The effect on bioactive components and characteristics of chocolate by functionalization with raw cocoa beans. Food Res. Int. 2018, 113, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Afoakwa, E.O.; Quao, J.; Takrama, J.; Budu, A.S.; Saalia, F.K. Chemical composition and physical quality characteristics of Ghanaian cocoa beans as affected by pulp pre-conditioning and fermentation. J. Food Sci. Technol. 2013, 50, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Farah, D.M.H.; Zaibunnisa, A.H.; Misnawi, J.; Zainal, S. Effect of roasting process on the concentration of acrylamide and pyrazines in roasted cocoa beans from different origins. APCBEE Procedia 2012, 4, 204–208. [Google Scholar] [CrossRef]
- Ramli, N.; Hassan, O.; Said, M.; Samsudin, W.; Idris, N.A. Influence of roasting conditions on volatile flavor ofroasted Malaysian cocoa beans. J. Food Process. Preserv. 2006, 30, 280–298. [Google Scholar] [CrossRef]
- Cadena Cala, T.; Herrera Ardila, Y.M. Evaluación del Efecto del Procesamiento del Cacao Sobre el Contenido de Polifenoles y su Actividad Antioxidant; Trabajo de Grado, Escuela de Química, Universidad Industrial de Santander: Bucaramanga, Colombia, 2008. [Google Scholar]
- Hoskin, J.C.; Dimick, P.S. Role ofnonenzymatic browning during the processing ofchocolate—A review. Process Biochem. 1984, 19, 92–104. [Google Scholar]
- Oracz, J.; Nebesny, E. Effect of roasting parameters on the physicochemical characteristics of high-molecularweight Maillard reaction products isolated from cocoa beans of different Theobroma cacao L. groups. Eur. Food Res. Technol. 2019, 245, 111–128. [Google Scholar] [CrossRef]
- Oracz, J.; Nebesny, E. Influence of roasting conditions on the biogenic amine content in cocoa beans of different Theobroma cacao cultivars. Food Res. Int. 2014, 55, 1–10. [Google Scholar] [CrossRef]
- Albak, F.; Tekin, A.R. The effect of addıtıon of ıngredıents on physıcal propertıes of dark chocolate durıng conchıng. Basic Res. J. Food Sci. Technol. 2014, 1, 51–59. [Google Scholar]
- García-Alamilla, P.; Salgado-Cervantes, M.A.; Barel, M.; Berthomieu, G.; Rodríguez-Jimenes, G.C.; García-Alvarado, M.A. Moisture, acidity and temperature evolution during cacao drying. J. Food Eng. 2007, 79, 1159–1165. [Google Scholar] [CrossRef]
- Di Mattia, C.D.; Sacchetti, G.; Mastrocola, D.; Serafini, M. From Cocoa to Chocolate: The impact of Processing on In Vitro Antioxidant Activity and the Effects of Chocolate on Antioxidant Markers in Vivo. Front. Immunol. 2017, 8, 1207. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomo- lybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Salvador, I.; Massarioli, A.P.; Silva, A.P.S.; Malaguetta, H.; Melo, P.S.; Alencar, S.M. Can we conserve trans-resveratrol content and antioxidant activity during industrial production of chocolate? J. Sci. Food Agric. 2019, 99, 83–89. [Google Scholar] [CrossRef]
- Gültekin-Özgüven, M.; Berktaş, I.; Özçelik, B. Influence of processing conditions on procyanidin profiles and antioxidant capacity of chocolates: Optimization of dark chocolate manufacturing by response surface methodology. LWT Food Sci. Technol. 2016, 66, 252–259. [Google Scholar] [CrossRef]
- Miller, K.B.; Stuart, D.A.; Smith, M.L.; Lee, C.Y.; McHale, N.L.; Flanagan, J.A.; Du, B.; Hurst, W.J. Antioxidant activity and polyphenol and procyanidin contents of selected commercially available cocoa containing and chocolate products in the United States. J. Agric. Food Chem. 2006, 54, 4062–4068. [Google Scholar] [CrossRef]
- Payne, M.J.; Hurst, W.J.; Miller, K.B.; Rank, C.; Stuart, D.A. Impact of fermentation, drying, roasting, and Dutch processing on epicatechin and catechin content of cacao beans and cocoa ingredients. J. Agric. Food Chem. 2010, 58, 10518–10527. [Google Scholar] [CrossRef]
- Carrillo, L.C.; Londoño-Londoño, J.; Gil, A. Comparison of polyphenol, methylxanthines and antioxidant activity in Theobroma cacao beans from different cocoa-growing areas in Colombia. Food Res. Int. 2010, 60, 273–280. [Google Scholar] [CrossRef]
- Jabłońska-Ryś, E. Zawartość polifenoli w czekoladach. Nauka Przyr. Technol. 2012, 6, #30. [Google Scholar]
- Kowalska, J.; Sidorczuk, A. Analysis of the effect of technological processing on changes in antioxidant properties of cocoa processed products. Pol. J. Food Sci. 2007, 57, 96–99. [Google Scholar]
- Meng, C.C.; Jalil, A.M.M.; Ismail, A. Phenolic and theobromine contents of commercial dark, milk and white chocolates on the Malaysian market. Molecules 2009, 14, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.A.; Donovan, J.L.; Waterhouse, A.I.; Williamson, G. Cocoa and health: A decade of research. Br. J. Nutr. 2008, 99, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Godočiková, L.; Ivanišová, E.; Árvay, J.; Petrová, J.; Kačániová, M. The comparison of biological activity of chocolates made by different technological procedures. Potravinarstvo 2016, 10, 316–322. [Google Scholar] [CrossRef] [Green Version]
- Todorovic, V.; Vidovic, B.; Todorovic, Z.; Sobajic, S. Correlation between Antimicrobial, Antioxidant Activity, and Polyphenols of Alkalized/Nonalkalized Cocoa Powders. J. Food Sci. 2017, 82, 1020–1027. [Google Scholar] [CrossRef]
- Jalil, A.M.M.; Ismail, A. Polyphenols in Cocoa and Cocoa Products: Is There a Link between Antioxidant Properties and Health? Molecules 2008, 13, 2190–2219. [Google Scholar] [CrossRef] [Green Version]
- Benayad, Z.; Gómez-Cordovés, C.; Es-Safi, N.E. Characterization of Flavonoid Glycosides from Fenugreek (Trigonella foenum-graecum) Crude Seeds by HPLC–DAD–ESI/MS Analysis. Int. J. Mol. Sci. 2014, 15, 20668–20685. [Google Scholar] [CrossRef]
- Cheng, V.J.; Bekhit, A.E.-D.A.; McConnell, M.; Mros, S.; Zhao, J. Effect of extraction solvent, waste fraction and grape variety on the antimicrobial and antioxidant activities of extracts from wine residue from cool climate. Food Chem. 2012, 134, 474–482. [Google Scholar] [CrossRef]
- Boulekbache-Makhlouf, L.; Slimani, S.; Madani, K. Total phenolic content, antioxidant and antibacterial activities of fruits of Eucalyptus globulus cultivated in Algeria. Ind. Crops Prod. 2013, 41, 85–89. [Google Scholar] [CrossRef]
- Djikeng, F.T.; Teyomnou, W.T.; Tenyang, N.; Tiencheu, B.; Morfor, A.T.; Touko, B.A.H.; Houketchang, S.N.; Boungo, G.T.; Karuna, M.S.L.; Ngoufack, F.Z. Effect of traditional and oven roasting on the physicochemical properties of fermented cocoa beans. Heliyon 2018, 4, e00533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bauer, D.; de Abreu, J.P.; Oliveira, H.S.S.; Goes-Neto, A.; Koblitz, M.G.B.; Teodoro, A.J. Antioxidant Activity and Cytotoxicity Effect of Cocoa Beans Subjected to Different Processing Conditions in Human Lung Carcinoma Cells. Oxid. Med. Cell. Longev. 2016, 2016, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arlorio, M.; Locatelli, M.; Travaglia, F.; Coïsson, J.; Del Grosso, E.; Minassi, A.; Appendino, G.; Martelli, A. Roasting impact on the contents of clovamide (N-caffeoyl-L-DOPA) and the antioxidant activity of cocoa beans (Theobroma cacao L.). Food Chem. 2008, 106, 967–975. [Google Scholar] [CrossRef]
- Hu, S.; Kim, B.Y.; Baik, M.Y. Physicochemical properties and antioxidant capacity of raw, roasted and puffed cacao beans. Food Chem. 2016, 194, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Zyzelewicz, D.; Bojczuk, M.; Budryn, G.; Jurgonski, A.; Zdunczyk, Z.; Juskiewicz, J.; Oracz, J. Influence of diet enriched with cocoa bean extracts on physiological indices of laboratory rats. Molecules 2019, 24, 825. [Google Scholar] [CrossRef] [PubMed]
- Wollgast, J.; Anklam, E. Review on polyphenols in Theobroma cacao: Changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Res. Int. 2000, 33, 423–447. [Google Scholar] [CrossRef]

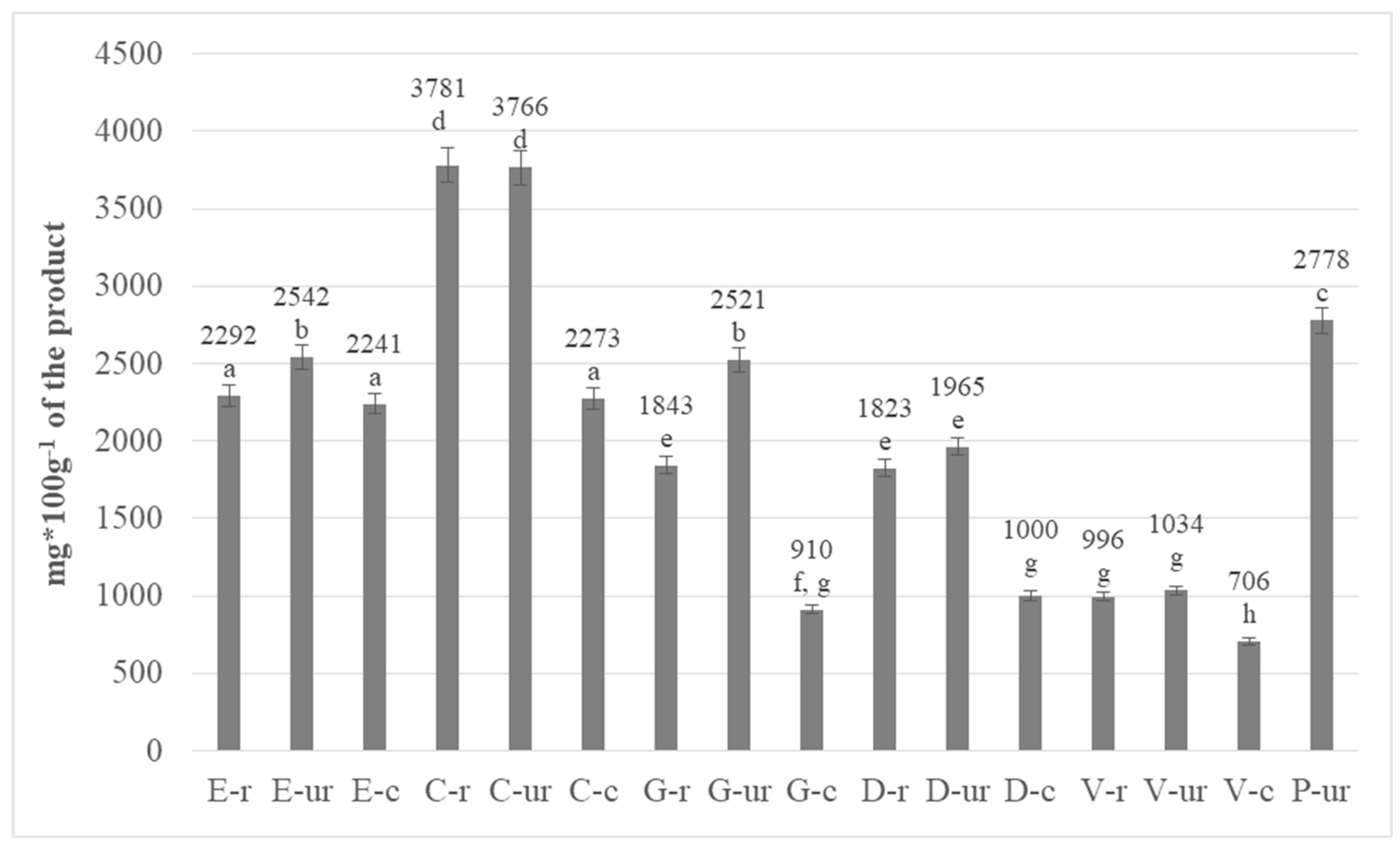
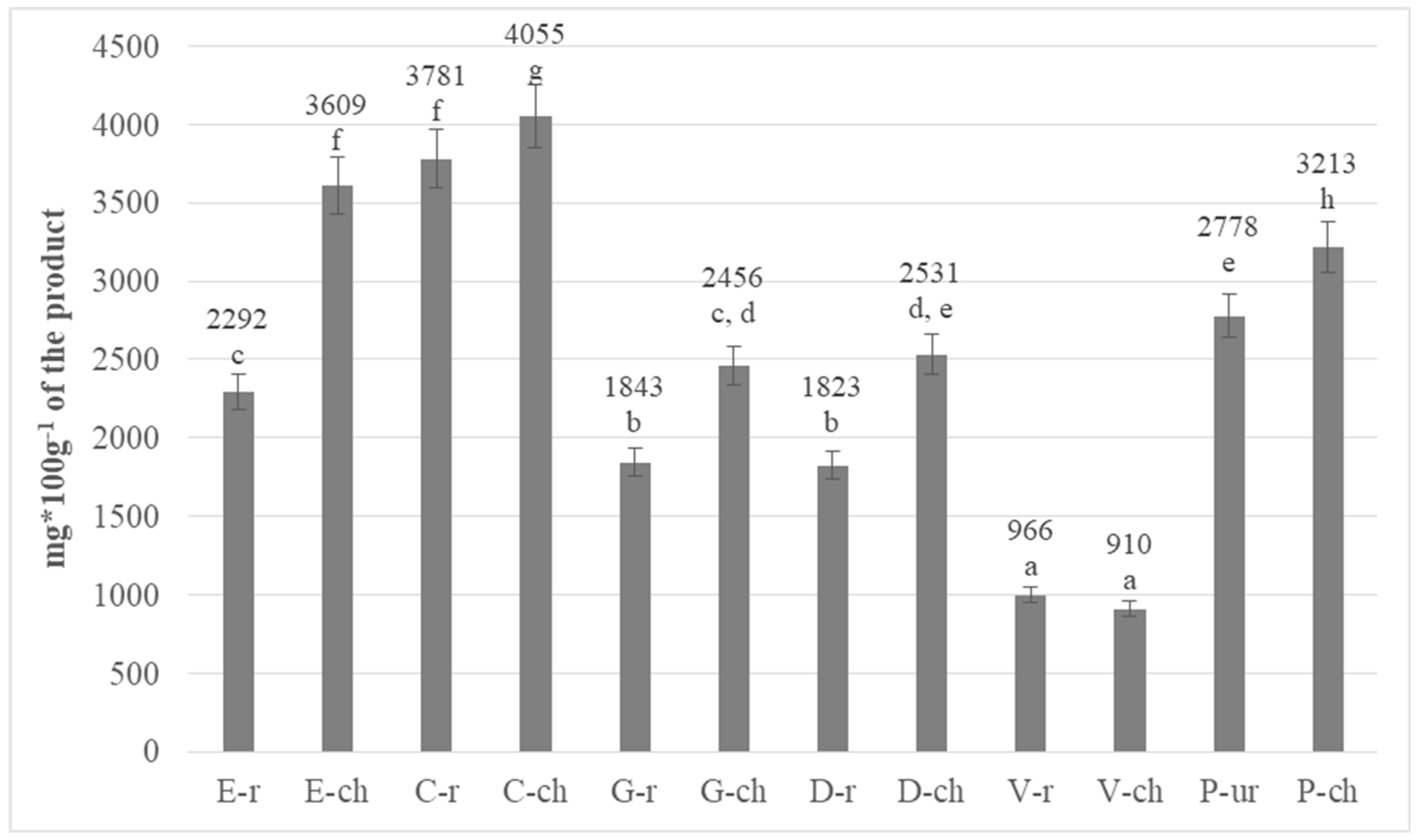
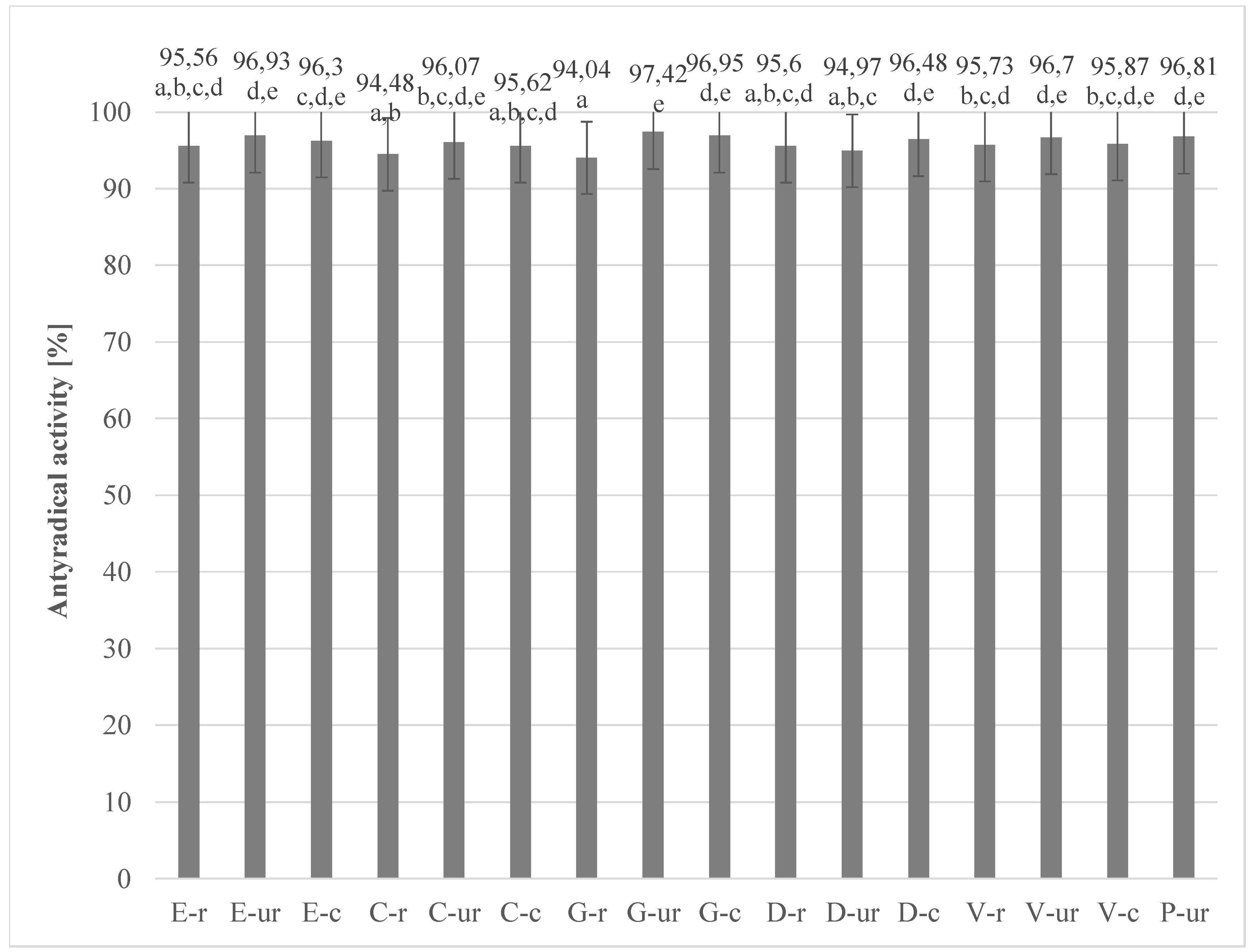
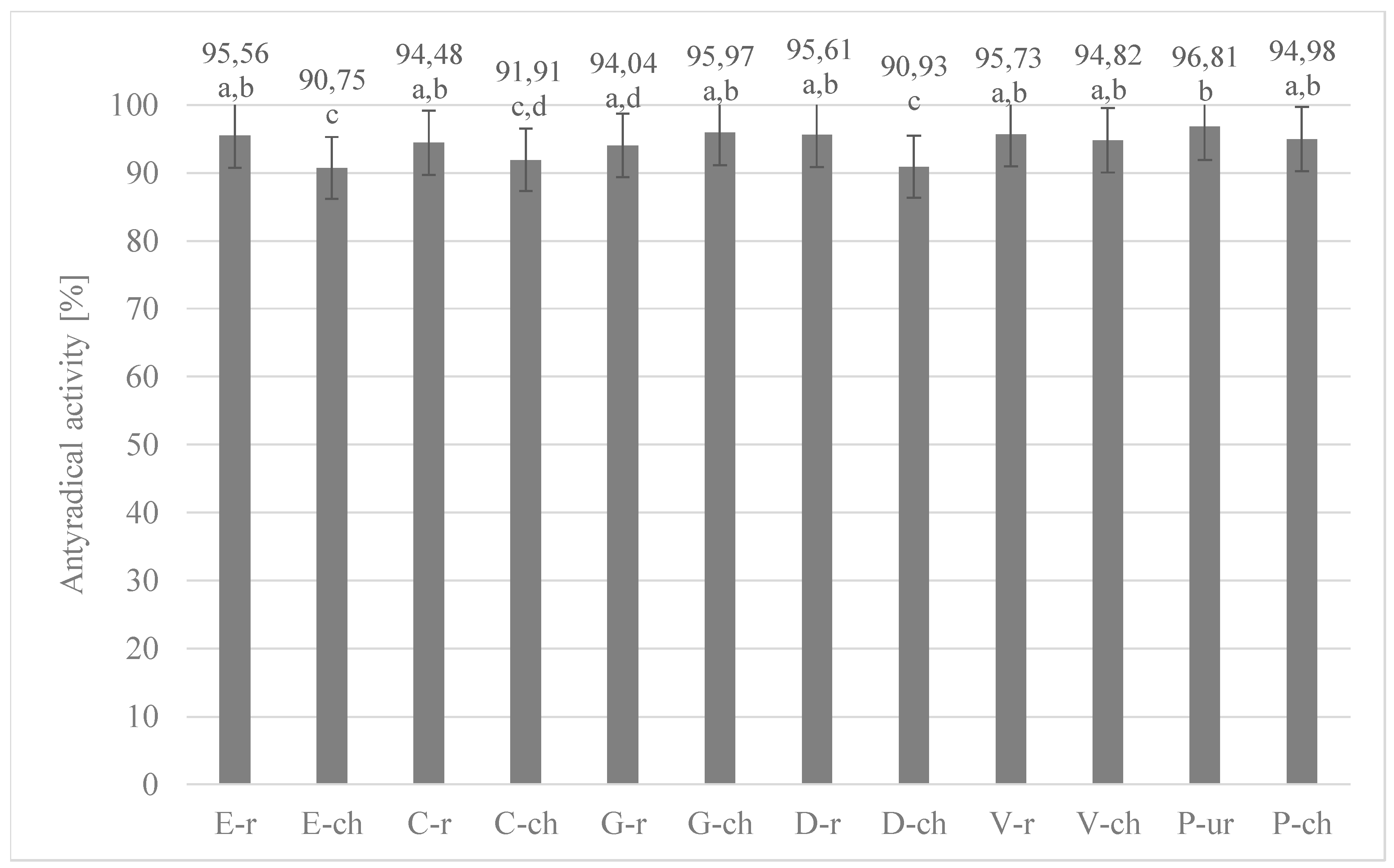
| No | Abbreviation | Full Name |
|---|---|---|
| 1 | E-r | Ecuador-roasted beans |
| 2 | E-ur | Ecuador-unroasted beans |
| 3 | E-c | Ecuador-roasted and crushed beanss |
| 4 | E-ch | Ecuador-chocolate-roasted beans |
| 5 | C-r | Colombia-roasted beans |
| 6 | C-ur | Colombia-unroasted beans |
| 7 | C-c | Colombia-roasted and crushed beanss |
| 8 | C-ch | Colombia-chocolate-roasted beans |
| 9 | G-r | Ghana-roasted beans |
| 10 | G-ur | Ghana-unroasted beans |
| 11 | G-c | Ghana-roasted and crushed beanss |
| 12 | G-ch | Ghana-chocolate-roasted beans |
| 13 | D-r | Dominican-roasted beans |
| 14 | D-ur | Dominican-unroasted beans |
| 15 | D-c | Dominican-roasted and crushed beanss |
| 16 | D-ch | Dominican-chocolate-roasted beans |
| 17 | V-r | Venezuela-roasted beans |
| 18 | V-ur | Venezuela-unroasted beans |
| 19 | V-c | Venezuela-roasted and crushed beanss |
| 20 | V-ch | Venezuela-chocolate-roasted beans |
| 21 | P-ur | Peru-unroasted beans |
| 22 | P-ch | Peru-chocolate-unroasted beans |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbańska, B.; Kowalska, J. Comparison of the Total Polyphenol Content and Antioxidant Activity of Chocolate Obtained from Roasted and Unroasted Cocoa Beans from Different Regions of the World. Antioxidants 2019, 8, 283. https://doi.org/10.3390/antiox8080283
Urbańska B, Kowalska J. Comparison of the Total Polyphenol Content and Antioxidant Activity of Chocolate Obtained from Roasted and Unroasted Cocoa Beans from Different Regions of the World. Antioxidants. 2019; 8(8):283. https://doi.org/10.3390/antiox8080283
Chicago/Turabian StyleUrbańska, Bogumiła, and Jolanta Kowalska. 2019. "Comparison of the Total Polyphenol Content and Antioxidant Activity of Chocolate Obtained from Roasted and Unroasted Cocoa Beans from Different Regions of the World" Antioxidants 8, no. 8: 283. https://doi.org/10.3390/antiox8080283
APA StyleUrbańska, B., & Kowalska, J. (2019). Comparison of the Total Polyphenol Content and Antioxidant Activity of Chocolate Obtained from Roasted and Unroasted Cocoa Beans from Different Regions of the World. Antioxidants, 8(8), 283. https://doi.org/10.3390/antiox8080283





