Antioxidants Profiling of By-Products from Eucalyptus Greenboards Manufacture
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
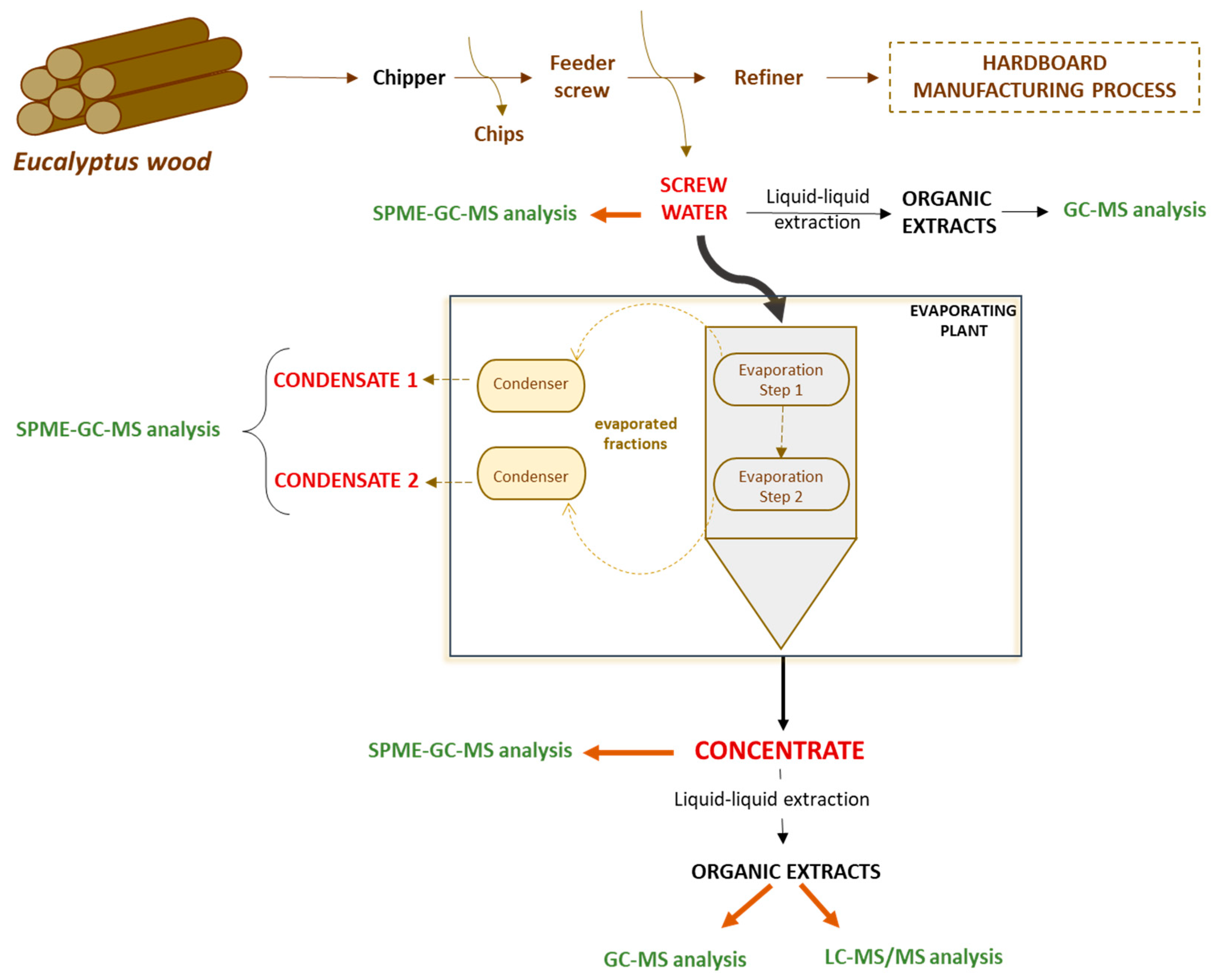
2.2. Studied By-Products
2.3. Liquid-Liquid Extraction
2.4. Total Polyphenols Content and Antioxidant Activity Procedures
2.5. Solid-Phase Microextraction Procedure
2.6. GC-MS Analysis
2.7. LC-MS/MS Analysis
3. Results
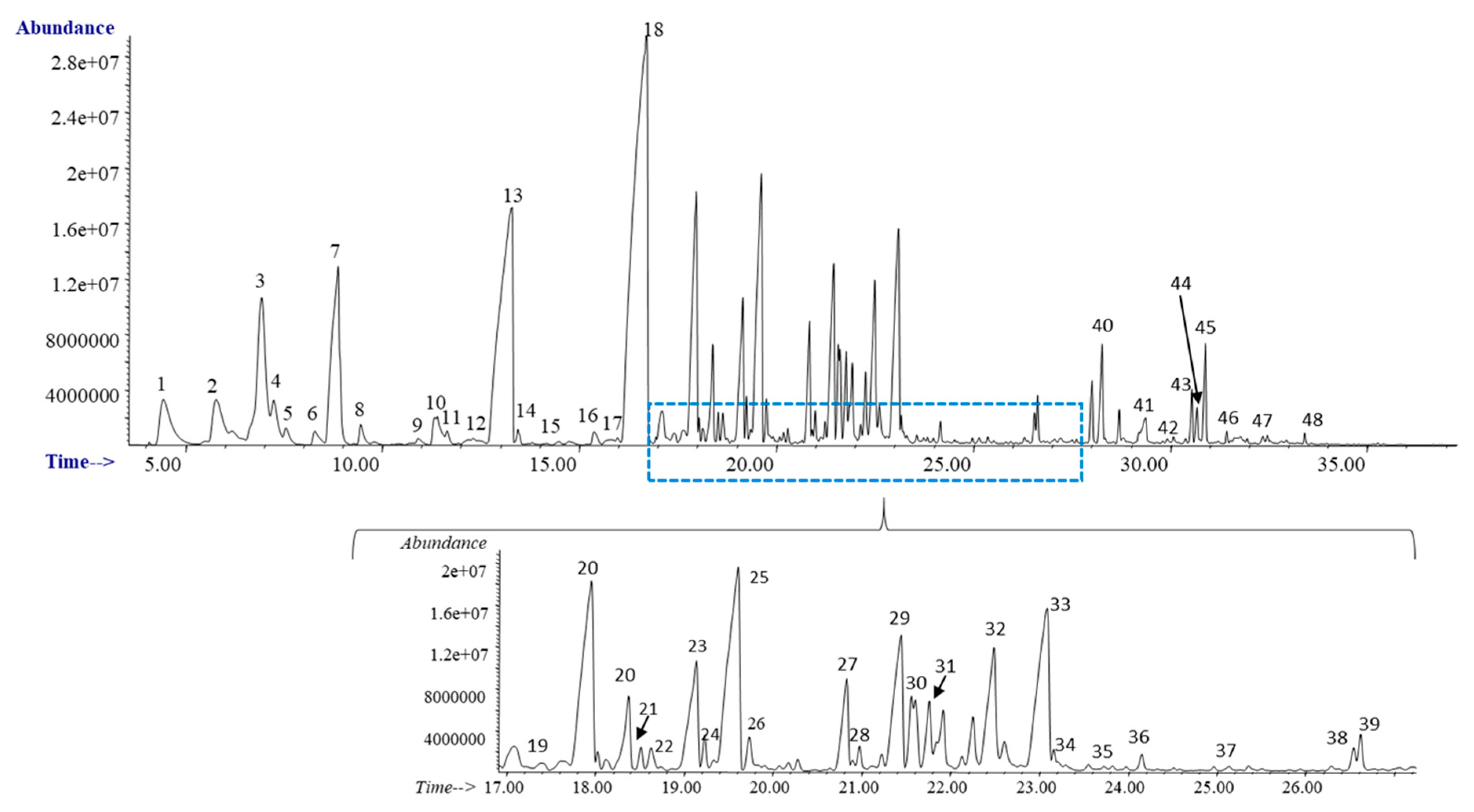
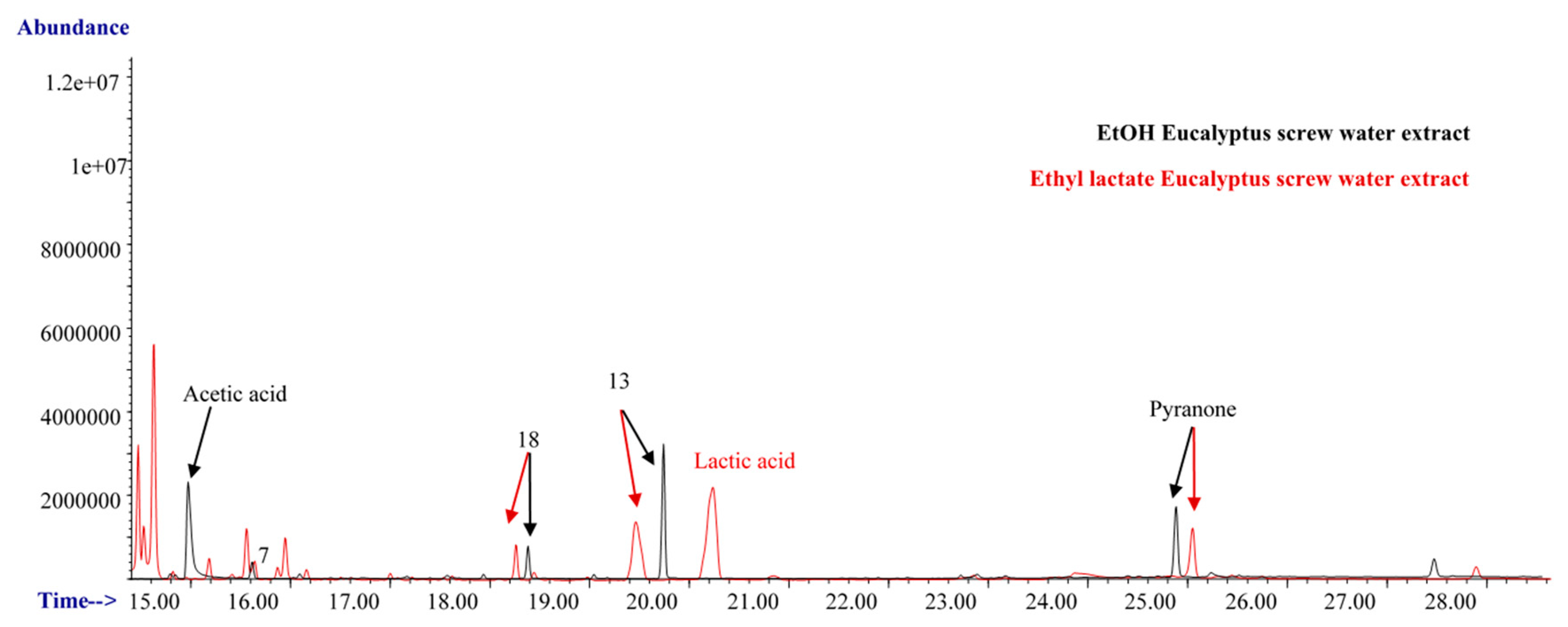
3.1. Eucalyptus Screw Water
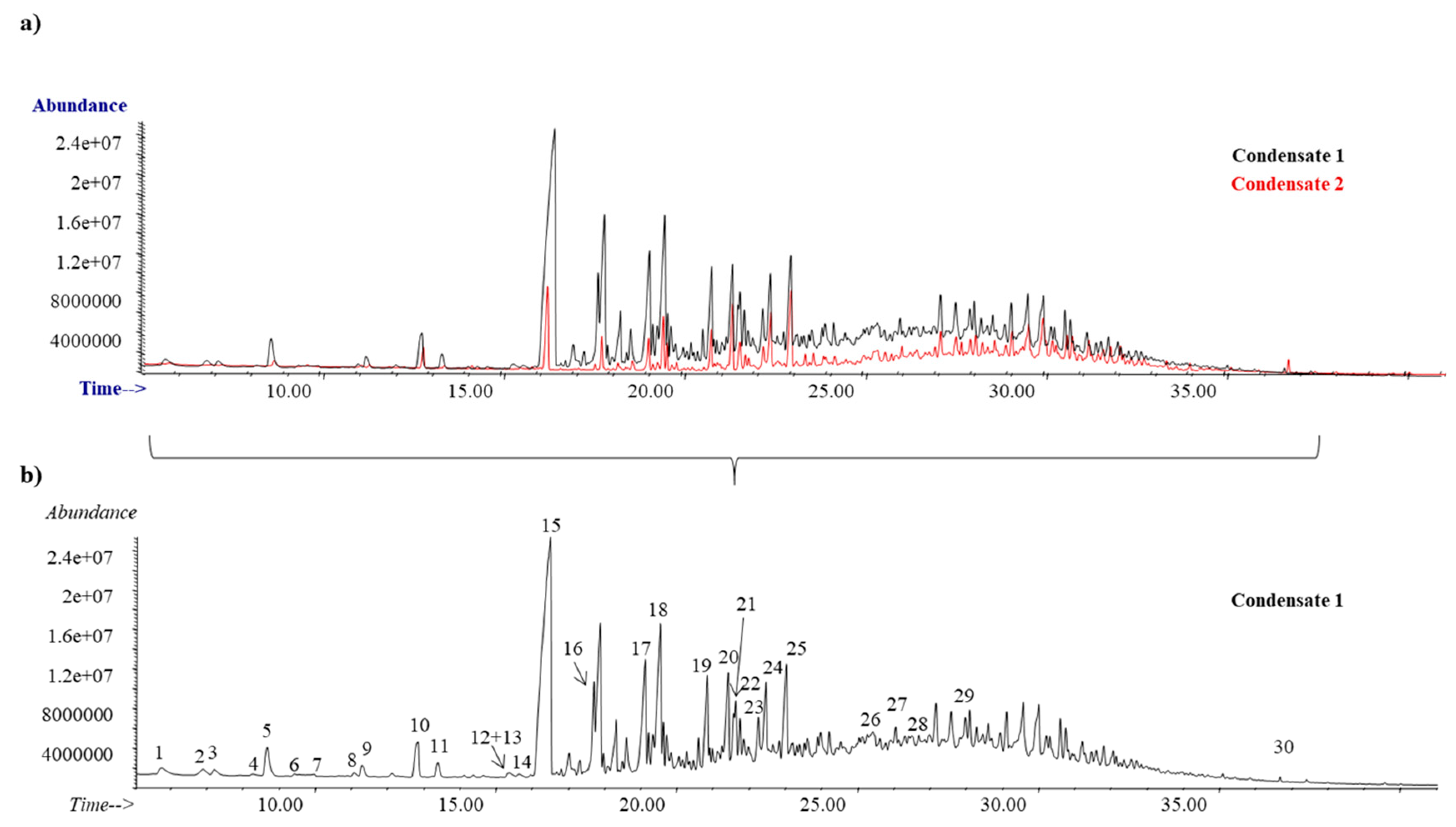
3.2. Eucalyptus Condensates
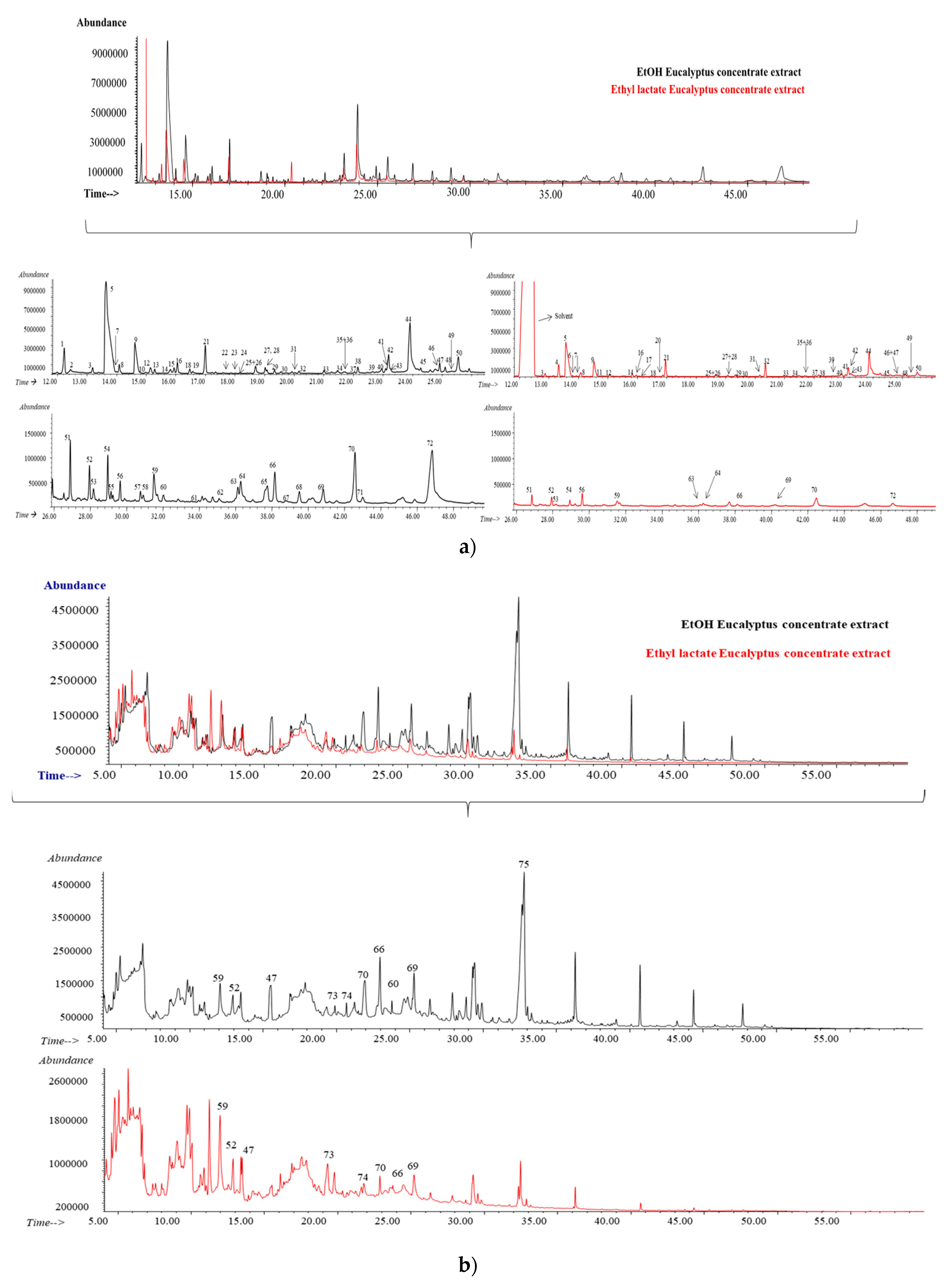
3.3. Concentrate of Eucalyptus
Stability Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cadahía, E.; Conde, E.; García-Vallejo, M.C.; Fernández de Simón, B. High pressure liquid chromatographic analysis of polyphenols in leaves of Eucalyptus camaldulensis, E. globulus and E. rudis: Proanthocyanidins, ellagitannins and flavonol glycosides. Phytochem. Anal. 1997, 8, 78–83. [Google Scholar] [CrossRef]
- Amakura, Y.; Yoshimura, M.; Sugimoto, N.; Yamazaki, T.; Yoshida, T. Marker constituents of the natural antioxidant Eucalyptus leaf extract for the evaluation of food additives. Biosci. Biotechnol. Biochem. 2009, 73, 1060–1065. [Google Scholar] [CrossRef]
- Dezsi, S.; Bãdãrãu, A.S.; Bischin, C.; Vodnar, D.C.; Silaghi-Dumitrescu, R.; Gheldiu, A.M.; Mocan, A.; Vlase, L. Antimicrobial and antioxidant activities and phenolic profile of Eucalyptus globulus Labill. and Corymbia ficifolia (F. Muell.) KD Hill & LAS Johnson leaves. Molecules 2015, 20, 4720–4734. [Google Scholar]
- Luís, A.; Neiva, D.; Pereira, H.; Gominho, J.; Domingues, F.; Duarte, A. Stumps of Eucalyptus globulus as a source of antioxidant and antimicrobial polyphenols. Molecules 2014, 19, 16428–16446. [Google Scholar] [CrossRef]
- Mota, I.; Rodrigues Pinto, P.C.; Novo, C.; Sousa, G.; Guerreiro, O.; Guerra, A.N.R.; Duarte, M.F.; Rodrigues, A.R.E. Extraction of polyphenolic compounds from Eucalyptus globulus bark: Process optimization and screening for biological activity. Ind. Eng. Chem. Res. 2012, 51, 6991–7000. [Google Scholar] [CrossRef]
- Gonzalez, J.; Cruz, J.M.; Domínguez, H.; Parajó, J.C. Production of antioxidants from Eucalyptus globulus wood by solvent extraction of hemicellulose hydrolysates. Food Chem. 2004, 84, 243–251. [Google Scholar] [CrossRef]
- Lin, S.; Zhou, Z.; Yin, W.Q. Three new polyphenolic acids from the leaves of Eucalyptus citriodora with antivirus activity. Chem. Pharmac. Bull. 2016, 64, 1641–1646. [Google Scholar] [CrossRef]
- The Conflict of the Eucalyptus Plantation in Spain and Portugal, Annual Report 2011, Greenpeace Spain. Available online: https://archivo-es.greenpeace.org/espana/Global/espana/report/bosques/InformeEucalipto2011.pdf (accessed on 15 February 2019).
- Neto, C.P.; Belino, E.; Evtuguin, D.; Silvestre, A.J.D. Total fractionation and analysis of organic components of industrial Eucalyptus globulus kraft black liquor. Appita J. 1999, 52, 213–217. [Google Scholar]
- Marques, A.P.; Evtuguin, D.V.; Magina, S.; Amado, F.M.L.; Prates, A. Chemical composition of spent liquors from acidic magnesium-based sulphite pulping of Eucalyptus globulus. J. Wood. Chem. Technol. 2009, 29, 322–336. [Google Scholar] [CrossRef]
- Wang, K.; Xu, F.; Sun, R. Molecular characteristics of kraft-AQ pulping lignin fractionated by sequential organic solvent extraction. Int. J. Mol. Sci. 2010, 11, 2988–3001. [Google Scholar] [CrossRef]
- Radoykova, T.; Nenkova, S.; Valchev, I. Black liquor lignin products, isolation and characterization. J. Chem. Technol. Metall. 2013, 48, 524–529. [Google Scholar]
- Pinto, P.C.R.; Costa, C.E.; Rodrigues, A.R.E.d. Oxidation of lignin from Eucalyptus globulus pulping liquors to produce syringaldehyde and vanillin. Ind. Eng. Chem. Res. 2013, 52, 4421–4428. [Google Scholar] [CrossRef]
- Da Silva, E.A.B.; Zabkova, M.; Araujo, J.D.; Cateto, C.A.; Barreiro, M.F.; Belgacem, M.N.; Rodrigues, A.E. An integrated process to produce vanillin and lignin-based polyurethanes from Kraft lignin. Chem. Eng. Res. Des. 2009, 87, 1276–1292. [Google Scholar] [CrossRef]
- Rodrigues Pinto, P.C.; Borges da Silva, E.A.; Rodrigues, A.R.E.d. Insights into oxidative conversion of lignin to high-added-value phenolic aldehydes. Ind. Eng. Chem. Res. 2010, 50, 741–748. [Google Scholar] [CrossRef]
- Fernández-Agulló, A.; Freire, M.S.; González-Álvarez, J. Effect of the extraction technique on the recovery of bioactive compounds from eucalyptus (Eucalyptus globulus) wood industrial wastes. Ind. Crop. Prod. 2015, 64, 105–113. [Google Scholar] [CrossRef]
- Otero-Pareja, M.A.; Casas, L.; Fernández-Ponce, M.A.; Mantell, C.; Ossa, E. Green extraction of antioxidants from different varieties of red grape pomace. Molecules 2015, 20, 9686–9702. [Google Scholar] [CrossRef] [PubMed]
- Lores, M.; Pájaro, M.; Álvarez-Casas, M.; Domínguez, J.; García-Jares, C. Use of ethyl lactate to extract bioactive compounds from Cytisus scoparius: Comparison of pressurized liquid extraction and medium scale ambient temperature systems. Talanta 2015, 140, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.-E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Swamy, M.K.; Akhtar, M.S.; Sinniah, U.R. Antimicrobial properties of plant essential oils against human pathogens and their mode of action: An updated review. Evid. Based Complement. Altenat. Med. 2016, 2016, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Plengsuriyakarn, T.; Karbwang, J.; Na-Bangchang, K. Anticancer activity using positron emission tomography-computed tomography and pharmacokinetics of β-eudesmol in human cholangiocarcinoma xenografted nude mouse model. Clin. Exp. Pharmacol. Physiol. 2015, 42, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Said, Z.B.-O.S.; Haddadi-Guemghar, H.; Boulekbache-Makhlouf, L.; Rigou, P.; Remini, H.; Adjaoud, A.; Khoudja, N.K.; Madani, K. Essential oils composition, antibacterial and antioxidant activities of hydrodistillated extract of Eucalyptus globulus fruits. Ind. Crops Prod. 2016, 89, 167–175. [Google Scholar] [CrossRef]
- Gullón, B.; Gullón, P.; Lú-Chau, T.A.; Moreira, M.T.; Lema, J.M.; Eibes, G. Optimization of solvent extraction of antioxidants from Eucalyptus globulus leaves by response surface methodology: Characterization and assessment of their bioactive properties. Ind. Crop. Prod. 2017, 108, 649–659. [Google Scholar] [CrossRef]
- Niemelä, K.; Sjöstöm, E. Alkaline degradation of alginates to carboxylic acids. Carbohyd. Res. 1985, 144, 241–249. [Google Scholar] [CrossRef]
- Top Added Chemicals: The Biobased Economy 12 Years Later. American Chemical Society (ACS) Network, 2017. Available online: https://communities.acs.org/community/science/sustainability/green-chemistry-nexus-blog/blog/2017/03/16/top-value-added-chemicals-the-biobased-economy-12-years-later. (accessed on 1 March 2019).
- Peleteiro, S.; Santos, V.N.; Garrote, G.; Parajó, J.C. Furfural production from eucalyptus wood using an acidic ionic liquid. Carbohyd. Polym. 2016, 146, 20–25. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Energy and Efficiency. Top Value-Added Chemicals from Biomass. Volume I—Results of Screening for Potential Candidates from Sugars and Synthesis Gas. 2004. Available online: https://www.energy.gov/eere/bioenergy/downloads/top-value-added-chemicals-biomass-volume-i-results-screening-potential (accessed on 10 July 2019).
- Silva, M.M.; Lidon, F. Food preservatives-An overview on applications and side effects. Emir. J. Food Agric. 2016, 28, 366–373. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, J.; Ou, Y.; Chen, H.; Xiao, S.; Liu, G.; Cao, Y.; Huang, Q. Cellular antioxidant activities of polyphenols isolated from Eucalyptus leaves (Eucalyptus grandis—Eucalyptus urophylla GL9). J. Funct. Food 2014, 7, 737–745. [Google Scholar] [CrossRef]
- Santos, S.N.A.O.; Freire, C.S.R.; Domingues, M.R.R.M.; Silvestre, A.J.D.; Neto, C.P. Characterization of phenolic components in polar extracts of Eucalyptus globulus Labill. bark by high-performance liquid chromatography-mass spectrometry. J. Agric. Food Chem. 2011, 59, 9386–9393. [Google Scholar] [CrossRef]
| By-Product | Density (g mL−1) | pH | TPC (mg GAE L−1) | AA (mM TRE L−1) | |
|---|---|---|---|---|---|
| Eucalyptus screw water | Raw material | 0.9870 | 3.65 | 6652 ± 21 | 52 ± 1 |
| EtOH extract | 0.9928 | 4.27 | 3630 ± 66 | 33 ± 1 | |
| Ethyl lactate extract | 1.002 | 4.03 | 4005 ± 49 | 34 ± 1 | |
| Condensates | 1 | 1.0020 | 2.97 | 6.1 ± 0.06 | ---- |
| 2 | 1.0042 | 2.89 | 7.7 ± 0.2 | ---- | |
| Concentrate of Eucalyptus | Raw material | 1.1059 | 3.69 | 43,702 ± 1413 | 197 ± 1 |
| EtOH extract | 0.9336 | 4.74 | 17,001 ± 377 | 101 ± 3 | |
| Ethyl lactate extract | 1.0320 | 4.32 | 30,287 ± 417 | 165 ± 6 | |
| Code | Compound | Ret. Time (min) | CAS | Screw Water (SPME-GC-MS) | Screw Water Extracts | |
|---|---|---|---|---|---|---|
| EtOH | Ethyl Lactate | |||||
| 1 | α-Pinene | 4.46 | 7785-70-8 | X | ||
| 2 | Myrcene | 5.79 | 123-35-3 | X | ||
| 3 | Eucalyptol | 6.94 | 470-82-6 | X | ||
| 4 | β-Ocimene | 7.26 | 13877-91-3 | X | ||
| 5 | γ-Terpinene | 7.56 | 99-85-4 | X | ||
| 6 | Allo-ocimene | 8.35 | 673-84-7 | X | ||
| 7 | Linalol | 8.82 | 78-70-6 | X | X | X |
| 8 | Neo-allo-ocimene | 9.82 | 7216-56-0 | X | ||
| 9 | 4-Terpineol | 10.95 | 562-74-3 | X | ||
| 10 | α-Terpineol | 11.4 | 98-55-5 | X | ||
| 11 | Isoamyl caproate | 11.69 | 2198-61-0 | X | ||
| 12 | Cis-Geraniol | 12.34 | 106-25-2 | X | ||
| 13 | Trans-Geraniol | 13.26 | 106-24-1 | X | X | X |
| 14 | α-Citral | 13.46 | 141-27-5 | X | ||
| 15 | Bornyl acetate | 13.82 | 76-49-3 | X | ||
| 16 | α-Terpineol acetate | 15.4 | 80-26-2 | X | ||
| 17 | Nerol acetate | 15.79 | 141-12-8 | X | ||
| 18 | Geranyl acetate | 16.52 | 105-87-3 | X | X | X |
| 19 | Caryophyllene | 17.38 | 87-44-5 | X | ||
| 20 | Alloaromadendrene | 17.91/18.41 | 25246-27-9 | X | ||
| 21 | Butanoic acid, 2-methyl-, 3,7-dimethyl-2,6-octadienyl ester, (E) | 18.51 | 68705-63-5 | X | ||
| 22 | 2-Isopropenyl-4a,8-dimethyl-1,2,3,4,4a,5,6,7 -octahydronaphthalene | 18.63 | 192435 (NIST Number) | X | ||
| 23 | Ledene | 19.12 | 21747-46-6 | X | ||
| 24 | α-Selinene | 19.22 | 473-13-2 | X | ||
| 25 | Geranyl butyrate | 19.54 | 106-29-6 | X | ||
| 26 | β-Cadinene | 19.73 | 523-47-7 | X | ||
| 27 | Ledol | 20.81 | 577-27-5 | X | ||
| 28 | 1,2-Dimethyl-5-nitroadamantane | 20.9 | 6588-68-7 | X | ||
| 29 | (−)-Globulol | 21.38 | 489-41-8 | X | ||
| 30 | Viridiflorol | 21.57 | 552-02-3 | X | ||
| 31 | Butanoic acid, 2-methyl-, 3,7-dimethyl-2,6-octadienyl ester, (Z)- | 21.76 | 68705-63-5 | X | ||
| 32 | 1H-Indene, 1-ethylideneoctahydro-7a-methyl-, (1Z,3aα,7aβ)- | 22.24 | 56324-69-7 | X | ||
| 33 | β-Eudesmol | 22.99 | 473-15-4 | X | ||
| 34 | Guai-1(10)-en-11-ol | 23.16 | 22451-73-6 | X | ||
| 35 | Geranyl hexanoate | 23.97 | 10032-02-7 | X | ||
| 36 | Farnesol | 24.15 | 4602-84-0 | X | ||
| 37 | Myristic acid | 25.12 | 544-63-8 | X | ||
| 38 | 2-Propanone, 1-methyl-1-(2,4,6-trimethoxyphenyl) | 26.54 | 26537-69-9 | X | ||
| 39 | Farnesol acetate | 26.62 | 352672 (ID) | X | ||
| 40 | 2-Propanone, 1-methyl-1-(2,4,6-trimethoxyphenyl) | 28.21 | 26537-69-9 | X | ||
| 41 | Hexadecanoic acid | 29.3 | 57-10-3 | X | ||
| 42 | Ethyl hexadecanoate | 29.8 | 628-97-7 | X | ||
| 43 | 3,5-di-tert-Butyl-4-hydroxycinnamic acid | 30.5 | 22014-01-3 | X | ||
| 44 | Falcarinol | 30.66 | 21852-80-2 | X | ||
| 45 | 6,11-dimethyl-2.6,10-dodecatrien-1-ol | 30.89 | 196533 (NIST number) | X | ||
| 46 | Quinazoline, 3-4-dihydro-2-allylthio-4-spirocyclohexane | 31.42 | 271789 (NIST number) | X | ||
| 47 | Linoleic acid | 32.31/32.45 | 60-33-3 | X | ||
| 48 | Geranyl linalool | 33.39 | 1113-21-9 | X | ||
| Code | Compound | Ret. Time (min) | CAS |
|---|---|---|---|
| 1 | β-Myrcene | 5.78 | 123-35-3 |
| 2 | Eucalyptol | 6.91 | 470-82-6 |
| 3 | β-cis-Ocimene | 7.26 | 3338-55-4 |
| 4 | α-Terpinene | 8.33 | 99-86-5 |
| 5 | β-Linalool | 8.62 | 78-70-6 |
| 6 | Trans-allocimene | 9.46 | 7216-56-0 |
| 7 | 3,5,5-trimethyl-hexanoic acid | 10 | 3302-10-1 |
| 8 | Creosol | 11.06 | 93-51-6 |
| 9 | α-Terpineol | 11.28 | 98-55-5 |
| 10 | Geraniol | 12.81 | 106-24-1 |
| 11 | Decan-1-ol | 13.39 | 112-30-1 |
| 12 | α-Terpinyl acetate | 15.37 | 80-26-2 |
| 13 | Citronellol acetate | 15.44 | 150-84-5 |
| 14 | Eugenol | 15.45 | 97-53-0 |
| 15 | Geranyl acetate | 16.18 | 105-87-3 |
| 16 | 2,6-Di-tert-butylphenol | 17.52 | 128-39-2 |
| 17 | (+)-Ledene | 18.98 | 21747-46-6 |
| 18 | Geranyl butyrate | 19.38 | 106-29-6 |
| 19 | Epiglobulol | 20.73 | 150051 (NIST number) |
| 20 | (−)-Globulol | 21.3 | 489-41-8 |
| 21 | Guaiol | 21.52 | 489-86-1 |
| 22 | Geranyl-2-methylbutyrate | 21.66 | 68705-63-5 |
| 23 | 1H-Indene, 1-ethylideneoctahydro-7a-methyl-, (1Z,3aα,7aβ)- | 22.26 | 56324-69-7 |
| 24 | γ-Eudesmol | 22.35 | 1209-71-8 |
| 25 | β-Eudesmol | 22.88 | 473-15-4 |
| 26 | Myristic acid | 25.26 | 544-63-8 |
| 27 | Farnesyl acetate | 26.64 | 4128-17-0 |
| 28 | 4-Methyldibenzothiophene | 27.16 | 7372-88-5 |
| 29 | 3-Ethyldibenzothiophene | 28.94 | 89817-03-8 |
| 30 | Octadecanoic acid, butyl ester | 36.68 | 123-95-5 |
| Code | Compound | Ret. Time (min) | CAS | Polar Column | Non-Polar Column | |||
|---|---|---|---|---|---|---|---|---|
| EtOH | Ethyl Lactate | Ret. Time (min) | EtOH | Ethyl Lactate | ||||
| 1 | Hydroxyacetone | 12.22 | 116-09-6 | X | ||||
| 2 | 1-Methoxyacetone | 12.44 | 5878-19-3 | X | ||||
| 3 | 1-Hydroxy-2-butanone | 12.89 | 5077-67-8 | X | X | |||
| 4 | Butanoic acid, 2-hydroxy-, ethyl ester | 13.16 | 52089-54-0 | X | ||||
| 5 | Acetic acid | 13.67 | 64-19-7 | X | X | |||
| 6 | Acetoxyacetone | 13.93 | 592-20-1 | X | ||||
| 7 | Methyl pyruvate | 13.99 | 600-22-6 | X | X | |||
| 8 | Furfural | 14.1 | 98-01-1 | X | X | |||
| 9 | Formic acid | 14.6 | 64-18-6 | X | X | |||
| 10 | 2-Furfuryl-acetate | 14.99 | 623-17-6 | X | ||||
| 11 | 2-Furyl methyl ketone | 14.8 | 1192-62-7 | X | ||||
| 12 | Propanoic acid | 15.16 | 79-09-4 | X | ||||
| 13 | 2,3-Butanediol | 15.3 | 24347-58-8 | X | ||||
| 14 | 5-Methyl-2-furfural | 15.8 | 620-02-0 | X | X | |||
| 15 | Vinyl 2-butenoate | 15.9 | 14861-06-4 | X | ||||
| 16 | Propylene glycol | 16.06 | 57-55-6 | X | X | |||
| 17 | 4-Cyclopentene-1,3-dione | 16.08 | 930-60-9 | X | ||||
| 18 | Butanoic acid | 16.4 | 107-92-6 | X | X | |||
| 19 | Ethylene glycol | 16.59 | 107-21-1 | X | ||||
| 20 | 4-hydroxy-butanoic acid | 16.87 | 591-81-1 | X | ||||
| 21 | Furfuryl alcohol | 17 | 98-00-0 | X | X | |||
| 22 | 2-Cyclopenten-1-one, 3-ethyl-2-hydroxy- | 17.61 | 21835-01-8 | X | ||||
| 23 | 5-Methyl-2-furfuryl alcohol | 17.88 | 3857-25-8 | X | ||||
| 24 | Pentanoic acid | 18.05 | 109-52-4 | X | ||||
| 25 | 2(5H)-Furanone | 18.66 | 497-23-4 | X | X | |||
| 26 | 2-Cyclopenten-1-one, 2-hydroxy- | 18.7 | 10493-98-8 | X | X | |||
| 27 | 1-(2-Butoxyethoxy)ethanol | 19.03 | 54446-78-5 | X | X | |||
| 28 | 1,2-Diisopropylhydrazine | 19.1 | 3711-34-0 | X | X | |||
| 29 | Furfuryl alcohol | 19.31 | 98-00-0 | X | X | |||
| 30 | Cyclotene | 19.55 | 80-71-7 | X | X | |||
| 31 | 2,5-Dimethyl-4-hydroxy-3(2H)-furanone | 20.02 | 3658-77-3 | X | X | |||
| 32 | N-Butyl-tert-butylamine | 20.17 | 16486-74-1 | X | X | |||
| 33 | Glycerin | 21 | 56-81-5 | X | X | |||
| 34 | Maltol | 21.48 | 118-71-8 | X | X | |||
| 35 | Glutaconic anhydride | 21.68 | 5926-95-4 | X | X | |||
| 36 | Phenol acetate | 21.74 | 122-79-2 | X | X | |||
| 37 | Furyl hydroxymethyl ketone | 22.04 | 17678-19-2 | X | X | |||
| 38 | Furaneol | 22.16 | 3658-77-3 | X | X | |||
| 39 | 2(3H)-Furanone, 5-acetyldihydro- | 22.66 | 29393-32-6 | X | X | |||
| 40 | Dihydroxyacetone | 22.97 | 96-26-4 | X | X | |||
| 41 | Methyl acetoxyacetate | 23.1 | 5837-80-9 | X | X | |||
| 42 | Cyclopropyl carbinol | 23.2 | 2516-33-8 | X | X | |||
| 43 | 4,5-Dimethyl-1,3-dioxol-2-one | 23.24 | 37830-90-3 | X | X | |||
| 44 | Methyltartronic acid | 23.92 | 595-98-2 | X | X | |||
| 45 | 2-Acetylresorcinol | 24.6 | 699-83-2 | X | X | |||
| 46 | 1,1-Ethanediol, diacetate | 24.79 | 542-10-9 | X | X | |||
| 47 | Syringol | 24.93 | 91-10-1 | X | X | X | X | |
| 48 | Pyranone | 25.12 | 28564-83-2 | X | X | |||
| 49 | 2,5-Dihydroxypropiophenone | 25.33 | 938-46-5 | X | X | |||
| 50 | Glycerin | 25.55 | 56-81-5 | X | X | |||
| 51 | 3-Pyridinol | 26.89 | 109-00-2 | X | X | |||
| 52 | 5-Hydroxymethylfurfural | 27.96 | 67-47-0 | X | X | X | X | |
| 53 | 1,2-Dihydroxy-3-methoxybenzene | 28.19 | 934-00-9 | X | X | |||
| 54 | 1-(2-Furyl)-1,2-ethanediol | 28.96 | 19377-75-4 | X | X | |||
| 55 | Vanillin | 29.12 | 121-33-5 | X | ||||
| 56 | Dihydro-4-hydroxy-2-(3H)-Furanone | 29.65 | 5469-16-9 | X | X | |||
| 57 | Vanillyl methyl ketone | 30.78 | 2503-46-0 | X | ||||
| 58 | Myristic acid | 30.94 | 544-63-8 | X | ||||
| 59 | Catechol | 31.53 | 120-80-9 | X | X | X | X | |
| 60 | Methoxyeugenol | 32.07 | 6627-88-9 | X | 23.78 | X | ||
| 61 | 3-Hydroxy-4-methoxybenzyl alcohol | 33.97 | 6/6/4383 | X | ||||
| 62 | Homovanillyl alcohol | 35.10 | 2380-78-1 | X | ||||
| 63 | Ethyl β-d-riboside | 36.14 | 126954 (NIST number) | X | X | |||
| 64 | n-Hexadecanoic acid | 36.25 | 57-10-3 | X | X | |||
| 65 | Palmitoleic acid | 36.65 | 373-49-9 | X | ||||
| 66 | Syringaldehyde | 38.15 | 134-96-3 | X | X | 22.87 | X | X |
| 67 | 1,4-Benzenediol, 2-methoxy- | 38.77 | 824-46-4 | X | ||||
| 68 | Homovanillic acid | 39.53 | 306-08-1 | X | ||||
| 69 | Desaspidinol | 40.82 | 437-72-9 | X | X | 25.26 | X | X |
| 70 | Antiarol | 42.58 | 642-71-7 | X | X | 21.91 | X | X |
| 71 | p-(3-hydroxybutyl)phenol | 43.00 | 501-96-2 | X | ||||
| 72 | Oleic Acid | 46.81 | 112-80-1 | X | X | |||
| 73 | 6-hydroxycoumarin | 2669-94-5 | 19.72 | X | X | |||
| 74 | Acetoguaiacon | 498-02-2 | 21.22 | X | X | |||
| 75 | Linolenic acid | 60-33-3 | 32.61 | X | ||||
| Polyphenols | Concentrate of Eucalyptus | |
|---|---|---|
| EtOH Extract | Ethyl Lactate Extract | |
| Gallic acid | 226 ± 15 | 72 ± 19 |
| Protocatechuic acid | 26 ± 7 | 19 ± 4 |
| Chlorogenic acid | 0.6 ± 0.05 | 0.4 ± 0.02 |
| 3,4-dihydroxybenzaldehyde | 1.3 ± 0.1 | n.d |
| 4-hydroxybenzaldehyde | 0.6 ± 0.03 | n.d |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Celeiro, M.; Lamas, J.P.; Arcas, R.; Lores, M. Antioxidants Profiling of By-Products from Eucalyptus Greenboards Manufacture. Antioxidants 2019, 8, 263. https://doi.org/10.3390/antiox8080263
Celeiro M, Lamas JP, Arcas R, Lores M. Antioxidants Profiling of By-Products from Eucalyptus Greenboards Manufacture. Antioxidants. 2019; 8(8):263. https://doi.org/10.3390/antiox8080263
Chicago/Turabian StyleCeleiro, Maria, J. Pablo Lamas, Rosa Arcas, and Marta Lores. 2019. "Antioxidants Profiling of By-Products from Eucalyptus Greenboards Manufacture" Antioxidants 8, no. 8: 263. https://doi.org/10.3390/antiox8080263
APA StyleCeleiro, M., Lamas, J. P., Arcas, R., & Lores, M. (2019). Antioxidants Profiling of By-Products from Eucalyptus Greenboards Manufacture. Antioxidants, 8(8), 263. https://doi.org/10.3390/antiox8080263







