Polyphenolic Profile and Antioxidant Capacity of Extracts from Gordonia axillaris Fruits
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Sample Preparation
2.3. Instruments
2.4. Extraction of Antioxidants
2.4.1. Ultrasonic-Assisted Extraction
2.4.2. Maceration Extraction
2.4.3. Soxhlet Extraction
2.5. Determination of Antioxidant Capacity
2.6. Determination of the Yield of Phenolic Compounds
2.7. Determination of the Yield of Flavonoid Compounds
2.8. Analysis of Phenolic Compounds by UPLC-MS/MS
2.9. Experimental Design
2.9.1. Single-Factor Experiments
2.9.2. Response Surface Methodology (RSM)
2.10. Statistical Analysis
3. Results and Discussion
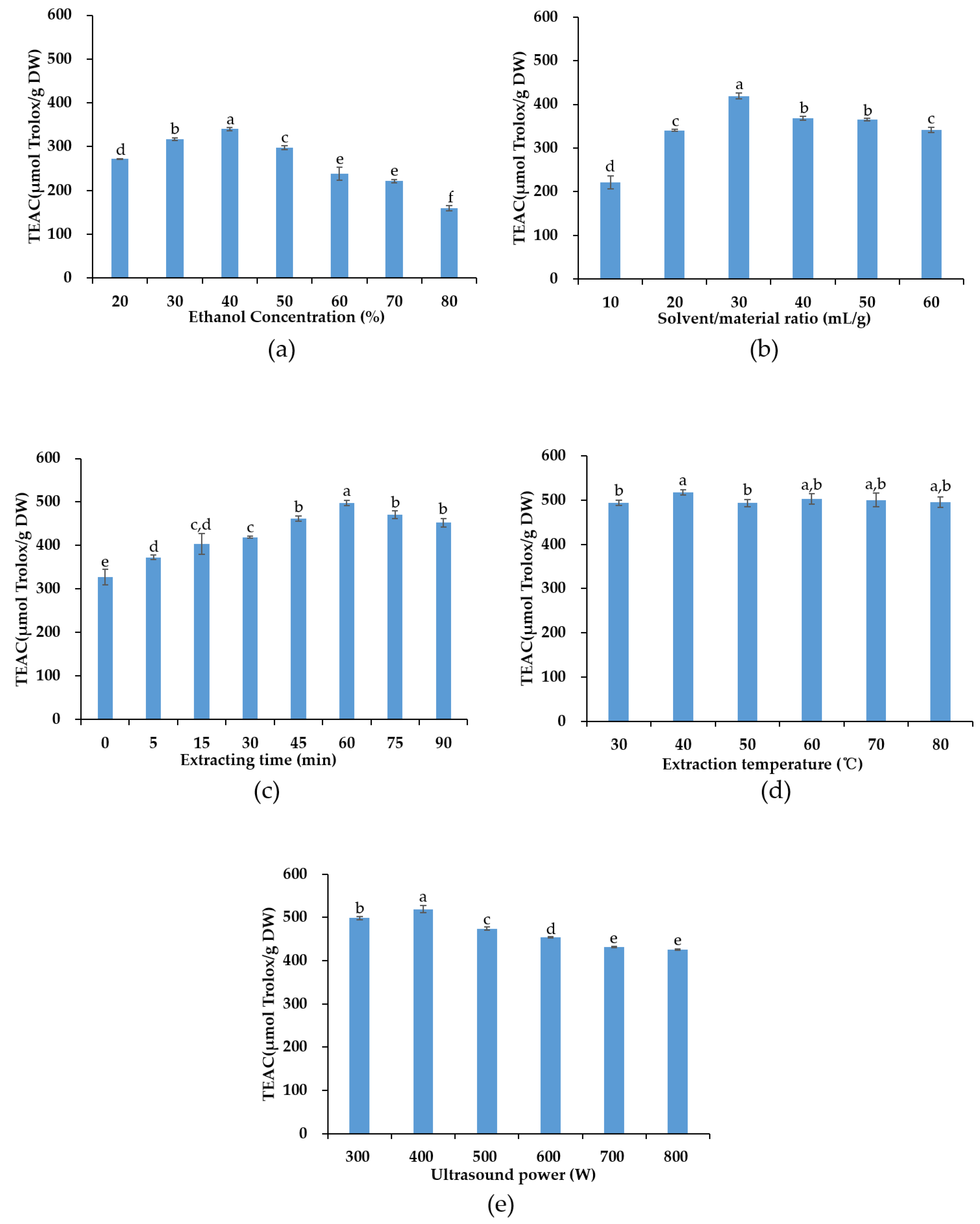
3.1. Results from Single-Factor Experiments
3.2. Results of Response Surface Methodology Experiments
3.2.1. Central Composite Rotatable Design (CCRD) and Results
3.2.2. Fitting the Model
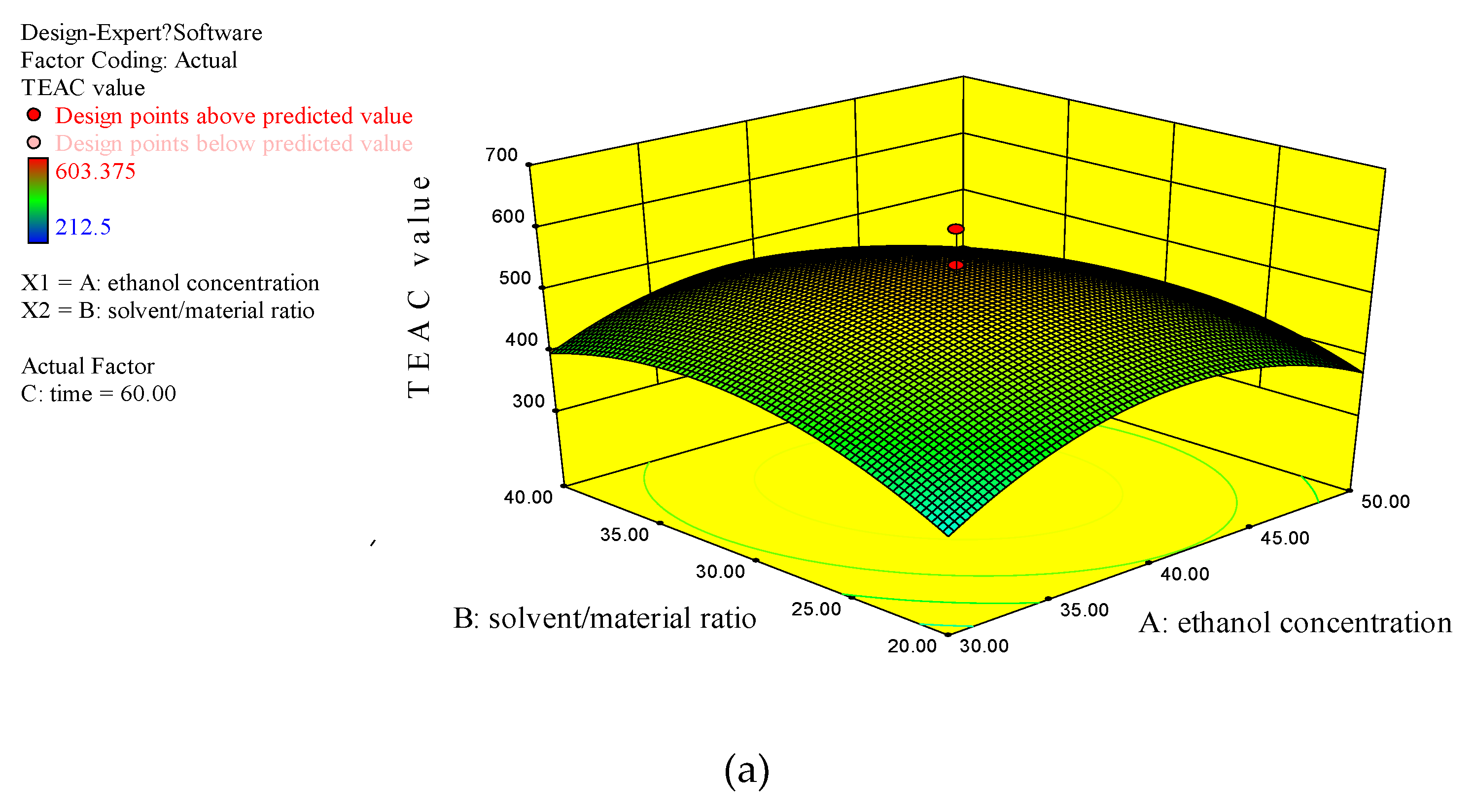
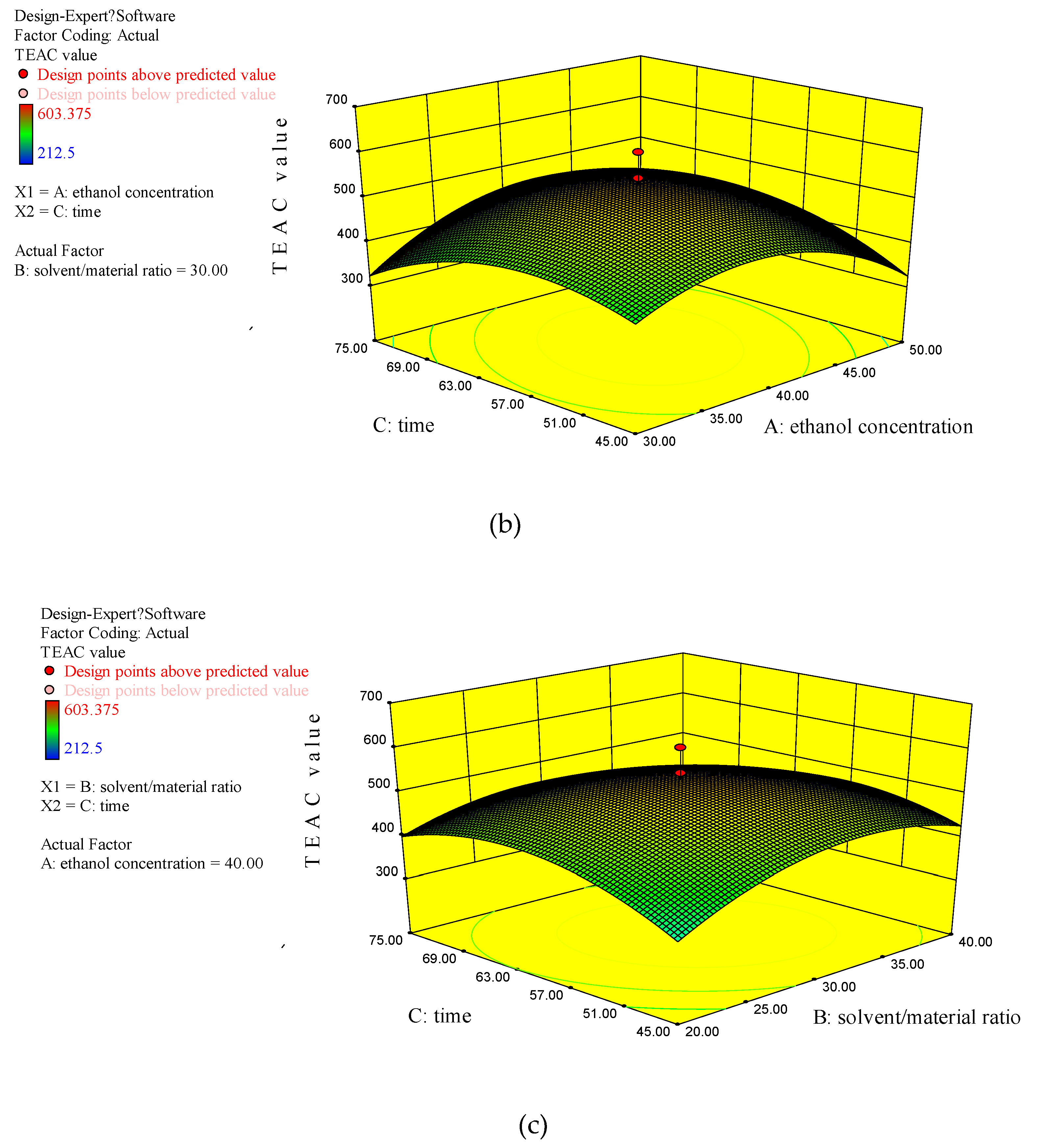
3.2.3. Analysis of Response Surface Plots
3.2.4. Verification of the Predicted Value
3.3. Comparison of UAE with Two Conventional Extraction Methods
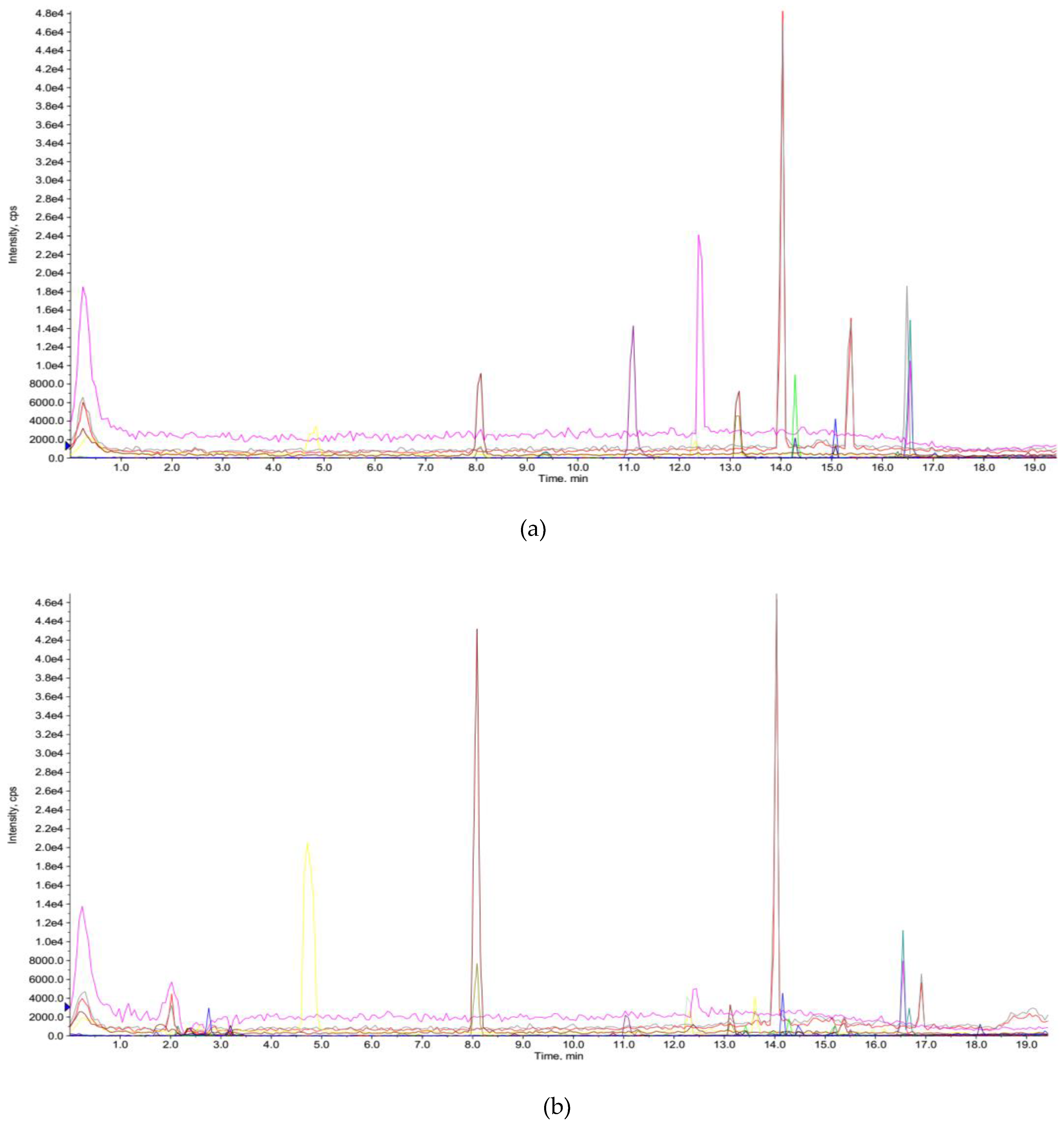
3.4. Phenolic Compounds in Extracts
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, J.J.; Xu, D.P.; Zhou, T.; Zhou, Y.; Li, S.; Li, H.B. Bioactivities and health benefits of wild fruits. Int. J. Mol. Sci. 2016, 17, 1258. [Google Scholar] [CrossRef] [PubMed]
- Li, Y. Extraction of Bioactive Components from Gordonia axillaris Fruit and the Hepatoprotective Effect. Master’s Thesis, Sun Yat Sen University, Guangzhou, China, 2018. [Google Scholar]
- Chang, C.W.; Yang, L.L.; Yen, K.Y.; Hatano, T.; Yoshida, T.; Okuda, T. Tannins from Theaceous plants. VII. new. Gamma-pyrone glucoside, and dimeric ellagitannins from Gordonia axillaris. Chem. Pharm. Bull. 1994, 42, 1922–1923. [Google Scholar] [CrossRef]
- Power, M.; de Thompson, M.C.; Heese, H.V.; Louw, H.H.; Khan, M.B. Priorities for provision of health care services for children in the Cape Province. S. Afr. Med. J. 1991, 80, 481–486. [Google Scholar] [PubMed]
- Wang, C.C.; Chen, L.G.; Yang, L.L. Camelliin B induced apoptosis in HeLa cell line. Toxicology 2001, 168, 231–240. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.T.; Xu, X.R.; Qin, X.S.; Gan, R.Y.; Li, H.B. Antioxidant capacities and total phenolic contents of 56 wild fruits from South China. Molecules 2010, 15, 8602–8617. [Google Scholar] [CrossRef]
- Li, Y.; Li, S.; Lin, S.J.; Zhang, J.J.; Zhao, C.N.; Li, H.B. Microwave-assisted extraction of natural antioxidants from the exotic Gordonia axillaris fruit: Optimization and identification of phenolic compounds. Molecules 2017, 22, 1481. [Google Scholar] [CrossRef]
- Bagheri, H.; Esmaiili, M. Ultrasound-assisted extraction of phenolic compounds from unripe grape (Qora). Erwerbs-Obstbau 2017, 59, 221–226. [Google Scholar] [CrossRef]
- Pereira, M.G.; Hamerski, F.; Andrade, E.F.; Scheer, A.D.; Corazza, M.L. Assessment of subcritical propane, ultrasound-assisted and Soxhlet extraction of oil from sweet passion fruit (Passiflora alata Curtis) seeds. J. Supercrit. Fluid. 2017, 128, 338–348. [Google Scholar] [CrossRef]
- Cravotto, G.; Cintas, P. Power ultrasound in organic systhesis: Moving cavitational chemistry from academia to innovative and large-scale applications. Chem. Soc. Rev. 2006, 35, 180–196. [Google Scholar] [CrossRef]
- Xu, D.P.; Zheng, J.; Zhou, Y.; Li, Y.; Li, S.; Li, H.B. Ultrasound-assisted extraction of natural antioxidants from the flower of Limonium sinuatum: Optimization and comparison with conventional methods. Food Chem. 2017, 217, 552–559. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications: A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.J.; Li, H.B. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.P.; Zhou, Y.; Zheng, J.; Li, S.; Li, A.N.; Li, H.B. Optimization of ultrasound-assisted extraction of natural antioxidants from the flower of Jatropha integerrima by response surface methodology. Molecules 2016, 21, 18. [Google Scholar] [CrossRef] [PubMed]
- Deng, G.F.; Xu, D.P.; Li, S.; Li, H.B. Optimization of ultrasound-assisted extraction of natural antioxidants from sugar apple (Annona squamosa L.) peel using response surface methodology. Molecules 2015, 20, 20448–20459. [Google Scholar] [CrossRef]
- Zhou, T.; Xu, D.P.; Lin, S.J.; Li, Y.; Zheng, J.; Zhou, Y.; Zhang, J.J.; Li, H.B. Ultrasound-assisted extraction and identification of natural antioxidants from the fruit of Melastoma sanguineum Sims. Molecules 2017, 22, 306. [Google Scholar] [CrossRef] [PubMed]
- Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Barroso, C.G.; Barbero, G.F. Optimization of the ultrasound-assisted extraction of anthocyanins and total phenolic compounds in mulberry (Morus nigra) pulp. Food Chem. 2017, 219, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Severini, C.; Derossi, A.; Fiore, A.G. Ultrasound-assisted extraction to improve the recovery of phenols and antioxidants from spent espresso coffee ground: A study by response surface methodology and desirability approach. Eur. Food Res. Technol. 2017, 243, 835–847. [Google Scholar] [CrossRef]
- Shirzad, H.; Niknam, V.; Taheri, M.; Ebrahimzadeh, H. Ultrasound-assisted extraction process of phenolic antioxidants from olive leaves: A nutraceutical study using RSM and LC-ESI-DAD-MS. J. Food Sci. Tech. 2017, 54, 2361–2371. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Li, H.B.; Xu, D.P.; Xu, X.R.; Chen, F. Total phenolic contents and antioxidant capacities of 51 edible and wild flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Kim, D.O.; Chun, O.K.; Kim, Y.J.; Moon, H.Y.; Lee, C.Y. Quantification of polyphenolics and their antioxidant capacity in fresh plums. J. Agric. Food Chem. 2003, 51, 6509–6515. [Google Scholar] [CrossRef]
- Chotphruethipong, L.; Benjakul, S.; Kijroongrojana, K. Optimization of extraction of antioxidative phenolic compounds from cashew (Anacardium occidentale L.) leaves using response surface methodology. J. Food Biochem. 2017, 41, e12379. [Google Scholar] [CrossRef]
- Da Silva, M.F.; Casazza, A.A.; Ferrari, P.F.; Aliakbarian, B.; Converti, A.; Bezerra, R.P.; Porto, A.; Perego, P. Recovery of phenolic compounds of food concern from Arthrospira platensis by green extraction techniques. Algal Res. 2017, 25, 391–401. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Li, Y.; Xu, D.P.; Li, H.B. Optimization of ultrasound-assisted extraction of natural antioxidants from the Osmanthus fragrans flower. Molecules 2016, 21, 218. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Xu, D.P.; Xu, X.R.; Chen, Y.M.; Ling, W.H.; Chen, F.; Li, H.B. Optimization of ultrasound-Assisted extraction of lycopene from papaya processing waste by response surface methodology. Food Anal. Methods 2015, 8, 1207–1214. [Google Scholar] [CrossRef]
- Ghitescu, R.E.; Volf, I.; Carausu, C.; Buhlmann, A.M.; Gilca, I.A.; Popa, V.I. Optimization of ultrasound-assisted extraction of polyphenols from spruce wood bark. Ultrason. Sonochem. 2015, 22, 535–541. [Google Scholar] [CrossRef]
- Xu, Y.; Pan, S.Y. Effects of various factors of ultrasonic treatment on the extraction yield of all-trans-lycopene from red grapefruit (Citrus paradise Macf.). Ultrason. Sonochem. 2013, 20, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Jia, X.; Wang, Y.; Zhu, H.; Chen, Q. Ultrasonically assisted extraction of rutin from Artemisia selengensis Turcz: Comparison with conventional extraction techniques. Food Anal. Methods 2010, 3, 261–268. [Google Scholar] [CrossRef]
- Xia, E.Q.; Yu, Y.Y.; Xu, X.R.; Deng, G.F.; Guo, Y.J.; Li, H.B. Ultrasound-assisted extraction of oleanolic acid and ursolic acid from Ligustrum lucidum Ait. Ultrason. Sonochem. 2012, 19, 772–776. [Google Scholar] [CrossRef]
- Ma, Y.Q.; Ye, X.Q.; Fang, Z.X.; Chen, J.C.; Xu, G.H.; Liu, D.H. Phenolic compounds and antioxidant activity of extracts from ultrasonic treatment of Satsuma mandarin (Citrus unshiu Marc.) peels. J. Agric. Food Chem. 2008, 56, 5682–5690. [Google Scholar] [CrossRef]
- Chen, F.L.; Zhang, Q.; Fei, S.M.; Gu, H.Y.; Yang, L. Optimization of ultrasonic circulating extraction of samara oil from Acer saccharum using combination of Plackett-Burman design and Box-Behnken design. Ultrason. Sonochem. 2017, 35, 161–175. [Google Scholar] [CrossRef]
- Yue, T.L.; Shao, D.Y.; Yuan, Y.H.; Wang, Z.L.; Qiang, C.Y. Ultrasound-assisted extraction, HPLC analysis, and antioxidant activity of polyphenols from unripe apple. J. Sep. Sci. 2012, 35, 2138–2145. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.Q.; Tu, X.J.; Dong, J.T.; Long, P.; Yang, W.C.; Miao, X.Q.; Chen, W.B.; Wu, Z.H. Soxhlet-assisted matrix solid phase dispersion to extract flavonoids from rape (Brassica campestris) bee pollen. J. Chromatogr. B. 2015, 1005, 17–22. [Google Scholar] [CrossRef]
- Subramanian, R.; Subbramaniyan, P.; Ameen, J.N.; Raj, V. Double bypasses soxhlet apparatus for extraction of piperine from Piper nigrum. Arab. J. Chem. 2016, 91, S537–S540. [Google Scholar] [CrossRef]
- Dent, M.; Dragović-Uzelac, V.; Garofulić, I.E.; Bosiljkov, T.; Ježek, D.; Brnčić, M. Comparison of conventional and ultrasound-assisted extraction techniques on mass fraction of phenolic compounds from Sage (Salvia officinalis L.). Chem. Biochem. Eng. Q. 2015, 29, 475–484. [Google Scholar] [CrossRef]
- Hromadkova, Z.; Kost’Alova, Z.; Ebringerova, A. Comparison of conventional and ultrasound-assisted extraction of phenolics-rich heteroxylans from wheat bran. Ultrason. Sonochem. 2008, 15, 1062–1068. [Google Scholar] [CrossRef] [PubMed]

| Variable | Units | Symbol | Coded Levels | ||||
|---|---|---|---|---|---|---|---|
| −1.68 | −1 | 0 | 1 | 1.68 | |||
| Ethanol concentration | % (v/v) | X1 | 23.18 | 30 | 40 | 50 | 56.82 |
| Solvent/material ratio | mL/g | X2 | 13.18 | 20 | 30 | 40 | 46.82 |
| Extraction time | min | X3 | 34.77 | 45 | 60 | 75 | 85.23 |
| Run | X1 (Ethanol Concentration, %) | X2 (Solvent/Material Ratio, mL/g) | X3 (Extraction Time, min) | Y (TEAC, µmol Trolox/g DW) | |
|---|---|---|---|---|---|
| Actual Value | Predicted Value | ||||
| 1 * | 40.00 | 30.00 | 60.00 | 511.85 | 531.11 |
| 2 | 40.00 | 46.82 | 60.00 | 325.69 | 347.03 |
| 3 | 56.82 | 30.00 | 60.00 | 243.01 | 256.81 |
| 4 | 40.00 | 30.00 | 85.23 | 317.37 | 323.43 |
| 5 | 30.00 | 40.00 | 45.00 | 423.16 | 393.22 |
| 6 | 50.00 | 20.00 | 75.00 | 376.83 | 364.21 |
| 7 | 30.00 | 40.00 | 75.00 | 278.25 | 259.29 |
| 8 | 50.00 | 40.00 | 45.00 | 293.50 | 268.04 |
| 9 | 40.00 | 30.00 | 34.77 | 275.42 | 329.54 |
| 10 | 50.00 | 20.00 | 45.00 | 261.15 | 237.55 |
| 11 * | 40.00 | 30.00 | 60.00 | 523.29 | 531.11 |
| 12 * | 40.00 | 30.00 | 60.00 | 508.04 | 531.11 |
| 13 | 40.00 | 13.18 | 60.00 | 267.60 | 306.44 |
| 14 | 50.00 | 40.00 | 75.00 | 283.33 | 287.29 |
| 15 * | 40.00 | 30.00 | 60.00 | 504.23 | 531.11 |
| 16 | 23.18 | 30.00 | 60.00 | 212.50 | 258.88 |
| 17 | 30.00 | 20.00 | 45.00 | 314.54 | 268.02 |
| 18 * | 40.00 | 30.00 | 60.00 | 603.38 | 531.11 |
| 19 * | 40.00 | 30.00 | 60.00 | 546.17 | 531.11 |
| 20 | 30.00 | 20.00 | 75.00 | 258.61 | 241.51 |
| Source | Sum of Squares | df | Mean Square | F Value | p Value | Significant |
|---|---|---|---|---|---|---|
| Model | 2.635 × 105 | 9 | 29279.68 | 15.02 | 0.0001 | significant |
| X1 (Ethanol concentration) | 5.21 | 1 | 5.21 | 2.673 × 10−3 | 0.9598 | |
| X2 (Solvent/material ratio) | 1988.99 | 1 | 1988.99 | 1.02 | 0.3363 | |
| X3 (Time) | 44.99 | 1 | 44.99 | 0.023 | 0.8823 | |
| X1X2 | 4484.03 | 1 | 4484.03 | 2.30 | 0.1604 | |
| X1X3 | 11730.86 | 1 | 11730.86 | 6.02 | 0.0341 | |
| X2X3 | 5768.58 | 1 | 5768.58 | 2.96 | 0.1162 | |
| X12 | 1.345 × 105 | 1 | 1.345 × 105 | 68.98 | <0.0001 | |
| X22 | 75240.48 | 1 | 75240.48 | 38.59 | <0.0001 | |
| X32 | 75423.11 | 1 | 75423.11 | 38.68 | <0.0001 | |
| Residual | 19499.88 | 10 | 1949.99 | |||
| Lack of Fit | 12381.50 | 5 | 2476.30 | 1.74 | 0.2791 | not significant |
| Pure Error | 7118.37 | 5 | 1423.67 | |||
| Cor Total | 2.830 × 105 | 19 | ||||
| R-Squared | 0.9311 | |||||
| Adj R-Squared | 0.8691 |
| Extraction Methods | Ethanol Conc. (%) | Time | Temp. (°C) | TEAC (µmol Trolox/g DW) | TPC (mg GAE/g DW) | TFC (mg QE/g DW) |
|---|---|---|---|---|---|---|
| Maceration | 39.78 | 24 h | 25 | 161.25 ± 2.29 | 13.16 ± 2.43 | 1.86 ± 0.12 |
| Soxhlet | 39.78 | 4 h | 85 | 113.77 ± 2.58 | 9.02 ± 0.78 | 1.77 ± 0.22 |
| UAE | 39.78 | 59.47 min | 40 | 525.05 ± 14.34 | 43.45 ± 3.16 | 3.22 ± 0.44 |
| Phenolic Components | Retention Time (tR, min) | Parent Ion (m/z, [M − H]¯) | Product Ion (m/z) | Contents (µg/g DW) |
|---|---|---|---|---|
| Rutin | 14.77 | 609 | 300, 343 | 21.86 ± 3.38 |
| Gallic acid | 4.76 | 169.1 | 125, 112 | 17.80 ± 2.68 |
| Protocatechuic acid | 8.07 | 153.1 | 109, 108 | 10.81 ± 2.54 |
| Epicatechin | 12.3 | 289 | 203, 245 | 3.89 ± 0.43 |
| 2-Hydrocinnamic acid | 14.03 | 163.1 | 119, 90 | 2.68 ± 0.25 |
| p-Coumaric acid | 14.03 | 162.7 | 119, 90 | 2.67 ± 0.22 |
| Quercetin | 16.54 | 301 | 179, 151 | 2.45 ± 0.21 |
| Epicatechin gallate | 13.13 | 441 | 169, 289.1 | 1.07 ± 0.09 |
| Ferulic acid | 14.28 | 193.1 | 134, 178 | 0.12 ± 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Cao, S.-Y.; Lin, S.-J.; Zhang, J.-R.; Gan, R.-Y.; Li, H.-B. Polyphenolic Profile and Antioxidant Capacity of Extracts from Gordonia axillaris Fruits. Antioxidants 2019, 8, 150. https://doi.org/10.3390/antiox8060150
Li Y, Cao S-Y, Lin S-J, Zhang J-R, Gan R-Y, Li H-B. Polyphenolic Profile and Antioxidant Capacity of Extracts from Gordonia axillaris Fruits. Antioxidants. 2019; 8(6):150. https://doi.org/10.3390/antiox8060150
Chicago/Turabian StyleLi, Ya, Shi-Yu Cao, Sheng-Jun Lin, Jia-Rong Zhang, Ren-You Gan, and Hua-Bin Li. 2019. "Polyphenolic Profile and Antioxidant Capacity of Extracts from Gordonia axillaris Fruits" Antioxidants 8, no. 6: 150. https://doi.org/10.3390/antiox8060150
APA StyleLi, Y., Cao, S.-Y., Lin, S.-J., Zhang, J.-R., Gan, R.-Y., & Li, H.-B. (2019). Polyphenolic Profile and Antioxidant Capacity of Extracts from Gordonia axillaris Fruits. Antioxidants, 8(6), 150. https://doi.org/10.3390/antiox8060150






