The Influence of Vinification Methods and Cultivars on the Volatile and Phenolic Profiles of Fermented Alcoholic Beverages from Cranberry
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Cranberry Wine
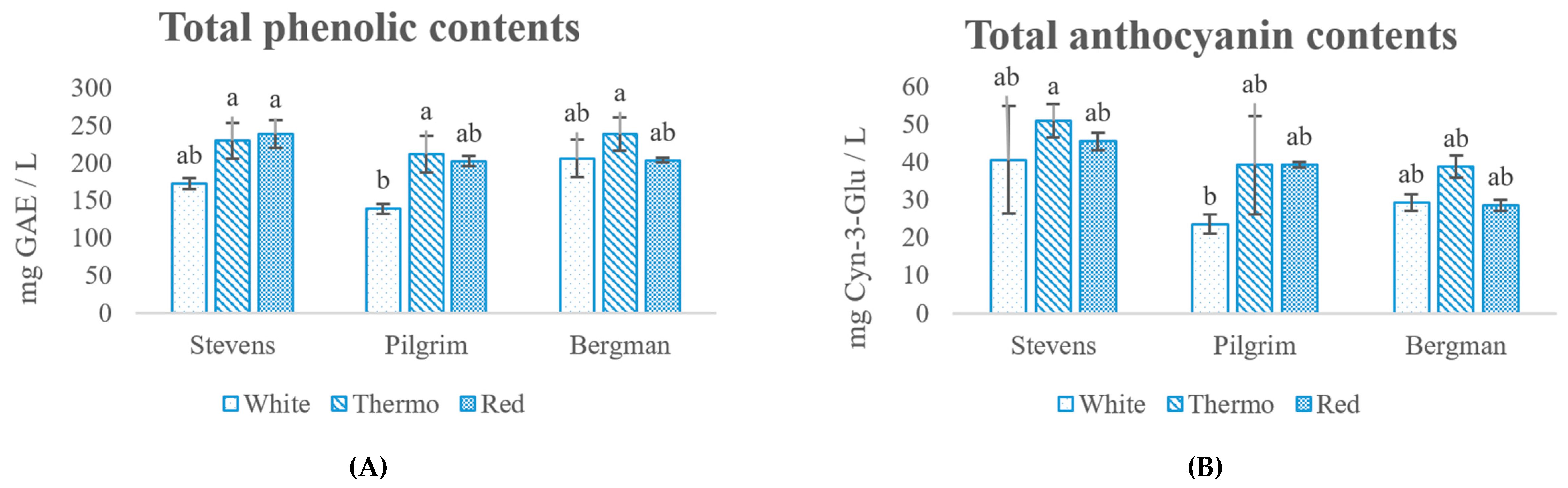
2.3. Determination of Total Phenolic Content (TPC), Proanthocyanin (PAC) and Monomeric Anthocyanin (ANC) Contents
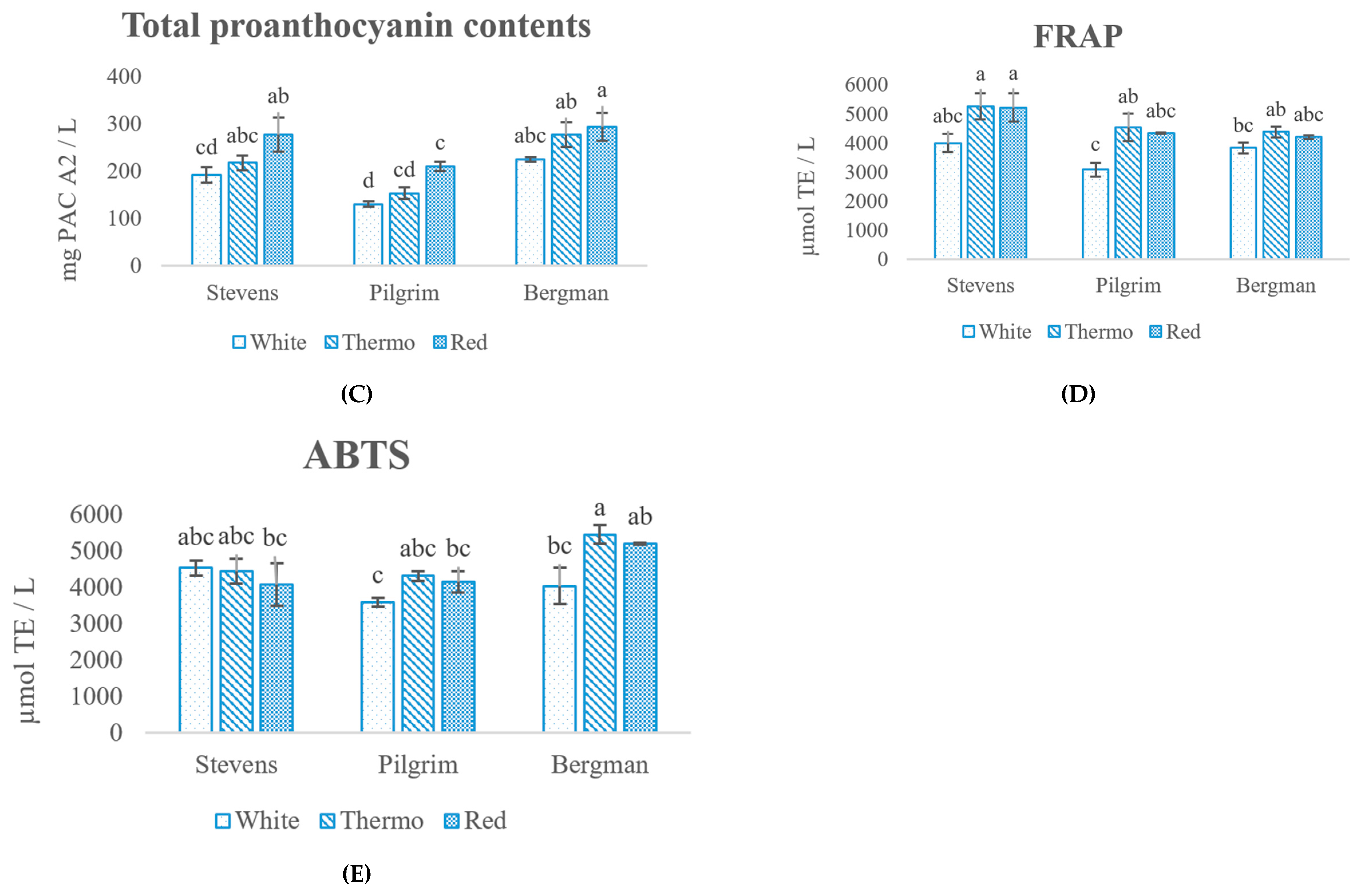
2.4. Determination of Antioxidant Capacity (AOC)
2.5. Quantification of Individual Anthocyanins
2.6. Quantification of Proanthocyanins, Flavonols, and Phenolic Acids
2.7. Quantification of Volatile Compounds
2.8. Odour Activity Values (OAV)
2.9. Statistical Analysis
3. Results and Discussion
3.1. Chemical Analysis
3.2. Identification and Quantification of Polyphenols in Cranberry Wine
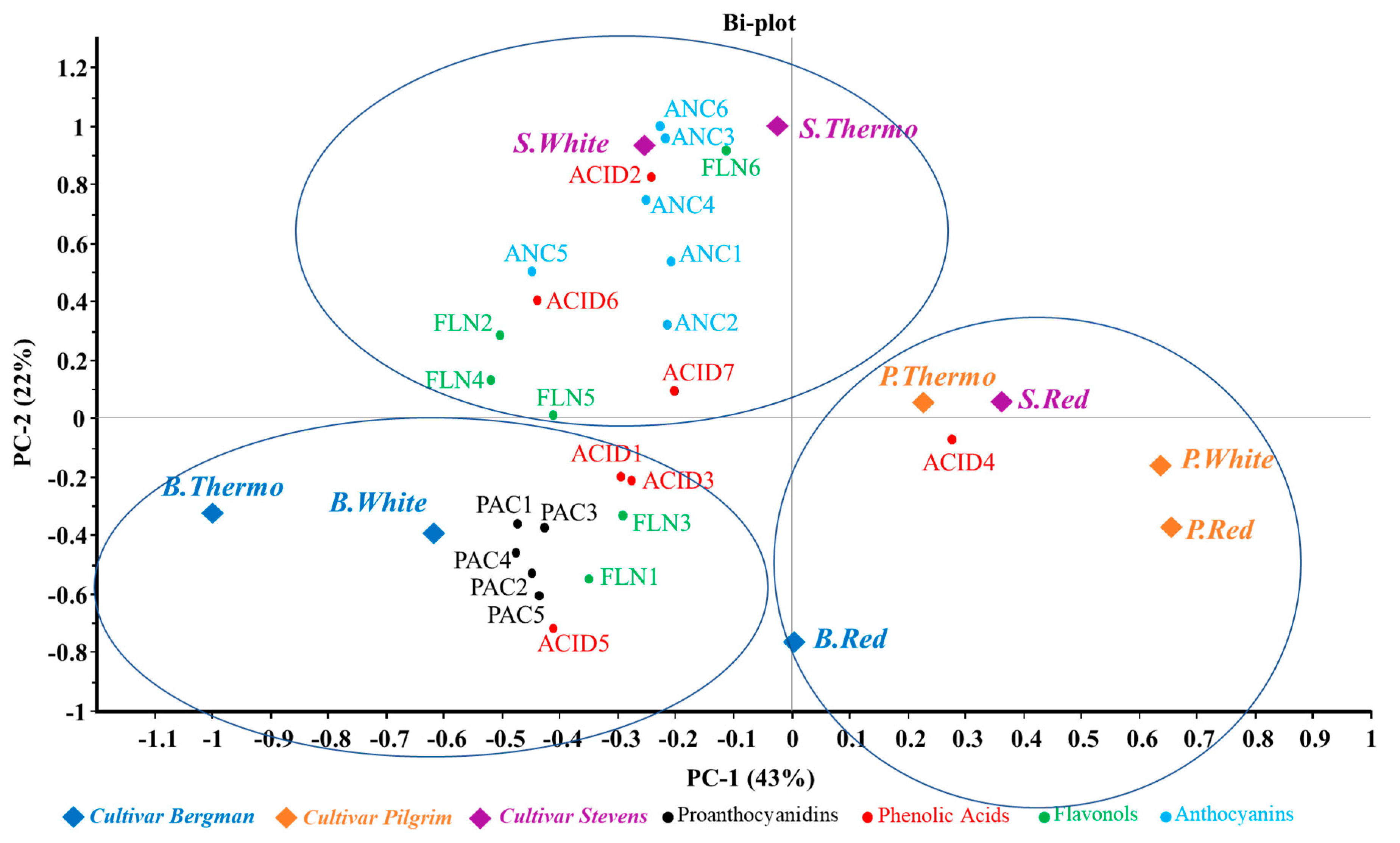
3.3. Distribution of Phenolic Compounds in Different Samples
3.4. Quantitative Analysis of Volatile Compounds
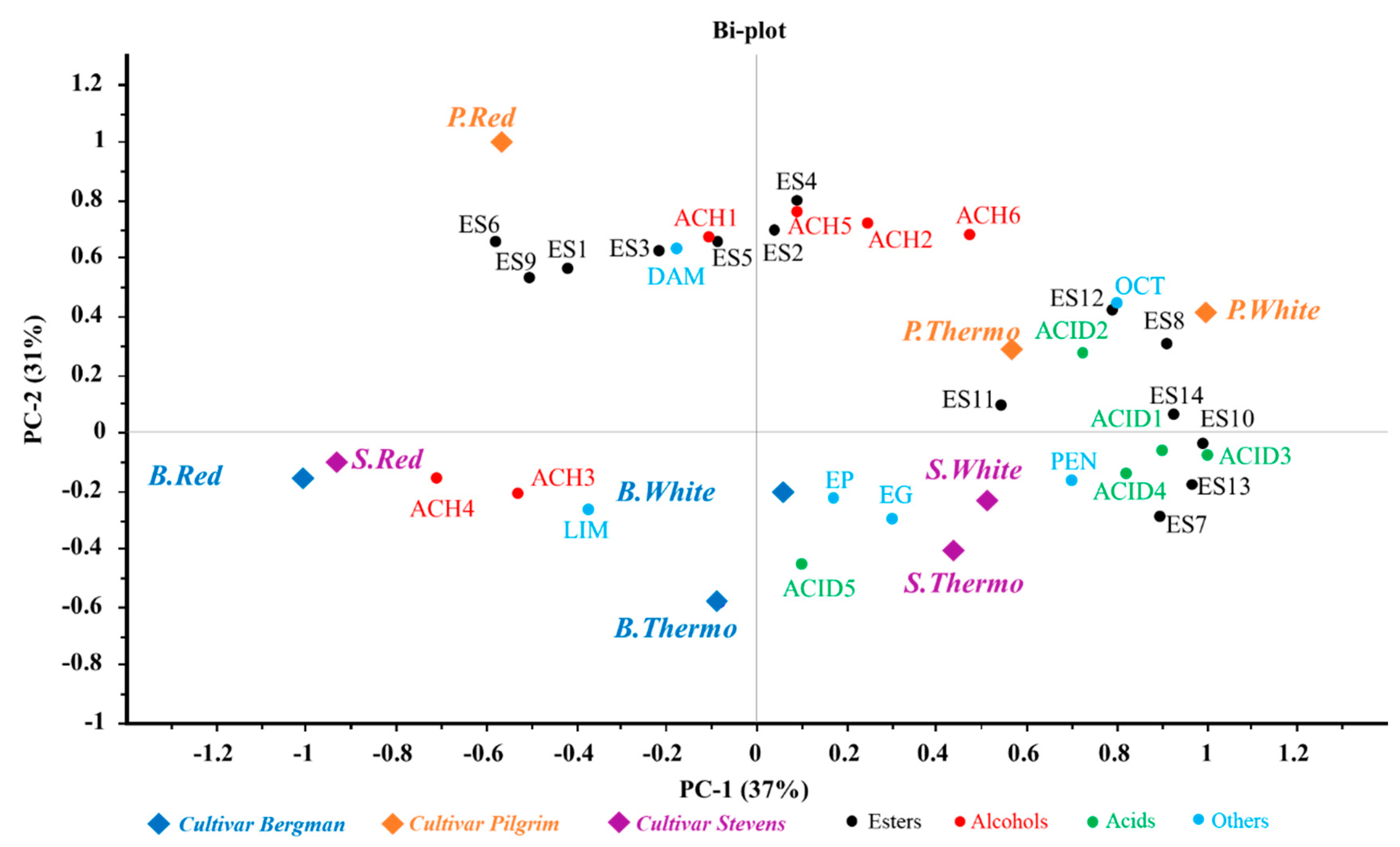
3.5. Distribution of Volatile Compounds in Different Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cote, J.; Caillet, S.; Doyon, G.; Sylvain, J.F.; Lacroix, M. Analyzing cranberry bioactive compounds. Crit. Rev. Food Sci. Nutr. 2010, 50, 872–888. [Google Scholar] [CrossRef] [PubMed]
- Skrovankova, S.; Sumczynski, D.; Mlcek, J.; Jurikova, T.; Sochor, J. Bioactive compounds and antioxidant activity in different types of berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P. Berry fruits: Compositional elements, biochemical activities, and the impact of their intake on human health, performance, and disease. J. Agric. Food Chem. 2008, 56, 627–629. [Google Scholar] [CrossRef]
- Howell, A.B. Bioactive compounds in cranberries and their role in prevention of urinary tract infections. Mol. Nutr. Food Res. 2007, 51, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, J.B.; Camesano, T.A.; Cassidy, A.; Kris-Etherton, P.; Howell, A.; Manach, C.; Ostertag, L.M.; Sies, H.; Skulas-Ray, A.; Vita, J.A. Cranberries and their bioactive constituents in human health. Adv. Nutr. 2013, 4, 618–632. [Google Scholar] [CrossRef]
- De Souza, V.R.; Pereira, P.A.P.; da Silva, T.L.T.; de Oliveira Lima, L.C.; Pio, R.; Queiroz, F. Determination of the bioactive compounds, antioxidant activity and chemical composition of Brazilian blackberry, red raspberry, strawberry, blueberry and sweet cherry fruits. Food Chem. 2014, 156, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Grace, M.H.; Massey, A.R.; Mbeunkui, F.; Yousef, G.G.; Lila, M.A. Comparison of health-relevant flavonoids in commonly consumed cranberry products. J. Food Sci. 2012, 77, H176–H183. [Google Scholar] [CrossRef] [PubMed]
- Khomych, G.; Matsuk, Y.; Nakonechnaya, J.; Oliynyk, N.; Medved, L. Research of cranberry main indicators of chemical composition and its processing products. EUREKA Life Sci. 2017, 6, 29–36. [Google Scholar] [CrossRef]
- Duarte, W.F.; Dias, D.R.; Oliveira, J.M.; Vilanova, M.; Teixeira, J.A.; e Silva, J.B.A.; Schwan, R.F. Raspberry (Rubus idaeus L.) wine: Yeast selection, sensory evaluation and instrumental analysis of volatile and other compounds. Food Res. Int. 2010, 43, 2303–2314. [Google Scholar] [CrossRef]
- Andreu-Sevilla, A.J.; Mena, P.; Martí, N.; García Viguera, C.; Carbonell-Barrachina, Á.A. Volatile composition and descriptive sensory analysis of pomegranate juice and wine. Food Res. Int. 2013, 54, 246–254. [Google Scholar] [CrossRef]
- Rupasinghe, H.P.V.; Clegg, S. Total antioxidant capacity, total phenolic content, mineral elements, and histamine concentrations in wines of different fruit sources. J. Food Compos. Anal. 2007, 20, 133–137. [Google Scholar] [CrossRef]
- Vuong, T.; Martin, L.; Matar, C. Antioxidant activity of fermented berry juices and their effects on nitric oxide and tumor necrosis factor-alpha production in macrophages 264.7 gamma no (–) cell line. J. Food Biochem. 2006, 30, 249–268. [Google Scholar] [CrossRef]
- Dey, G.; Negi, B.; Gandhi, A. Can fruit wines be considered as functional food?—An overview. Nat. Prod. Radiance 2009, 8, 314–322. [Google Scholar]
- Patel, K.D.; Scarano, F.J.; Kondo, M.; Hurta, R.A.; Neto, C.C. Proanthocyanidin-rich extracts from cranberry fruit (Vaccinium macrocarpon Ait.) selectively inhibit the growth of human pathogenic fungi Candida spp. and Cryptococcus neoformans. J. Agric. Food Chem. 2011, 59, 12864–12873. [Google Scholar] [CrossRef] [PubMed]
- Grainger, K.; Tattersall, H. Dry White Wine Making. In Wine Production: Vine To Bottle, 1st ed.; Grainger, K., Tattersall, H., Eds.; John Wiley & Sons: Oxford, UK, 2005; pp. 62–64. [Google Scholar]
- Grainger, K.; Tattersall, H. Red Wine Making. In Wine Production: Vine To Bottle, 1st ed.; Grainger, K., Tattersall, H., Eds.; John Wiley & Sons: Oxford, UK, 2008; pp. 54–61. [Google Scholar]
- Geffroy, O.; Lopez, R.; Feilhes, C.; Violleau, F.; Kleiber, D.; Favarel, J.L.; Ferreira, V. Modulating analytical characteristics of thermovinified Carignan musts and the volatile composition of the resulting wines through the heating temperature. Food Chem. 2018, 257, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Geffroy, O.; Lopez, R.; Serrano, E.; Dufourcq, T.; Gracia-Moreno, E.; Cacho, J.; Ferreira, V. Changes in analytical and volatile compositions of red wines induced by pre-fermentation heat treatment of grapes. Food Chem. 2015, 187, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Atanacković, M.; Petrović, A.; Jović, S.; Bukarica, L.G.; Bursać, M.; Cvejić, J. Influence of winemaking techniques on the resveratrol content, total phenolic content and antioxidant potential of red wines. Food Chem. 2012, 131, 513–518. [Google Scholar] [CrossRef]
- Yuan, Y. Blueberry wine fermentation and its effect on phenolic compounds. PhD Thesis, Nanjing Agricultral University, Nanjing, China, 2012. Available online: http://cdmd.cnki.com.cn/Article/CDMD-10307-1015569871.htm (accessed on 22 May 2019).
- Tang, Y.; Li, X.; Zhang, B.; Chen, P.X.; Liu, R.; Tsao, R. Characterisation of phenolics, betanins and antioxidant activities in seeds of three Chenopodium quinoa Willd. genotypes. Food Chem. 2015, 166, 380–388. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar]
- Ozgen, M.; Reese, R.N.; Tulio, A.Z.; Scheerens, J.C.; Miller, A.R. Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2‘-diphenyl-1-picrylhydrazyl (DPPH) methods. J. Agric. Food Chem. 2006, 54, 1151–1157. [Google Scholar] [CrossRef]
- Vagiri, M.; Ekholm, A.; Andersson, S.C.; Johansson, E.; Rumpunen, K. An optimized method for analysis of phenolic compounds in buds, leaves, and fruits of black currant (Ribes nigrum L.). J. Agric. Food Chem. 2012, 60, 10501–10510. [Google Scholar] [CrossRef]
- Zhao, X.J.; Chen, D.; Kilmartin, P.A.; Jiao, B.N. Simultaneous Determination of Phenolics and Polymethoxylated Flavones in Citrus Fruits by Ultra-High Performance Liquid Chromatography Coupled with Triple-Quadrupole Mass Spectrometry (UHPLC-QqQ-MS). Anal. Lett. 2019, 1–13. [Google Scholar] [CrossRef]
- Jouanneau, S.; Weaver, R.; Nicolau, L.; Herbst-Johnstone, M.; Benkwitz, F.; Kilmartin, P.A. Subregional survey of aroma compounds in Marlborough Sauvignon Blanc wines. Aust. J. Grape Wine Res. 2012, 18, 329–343. [Google Scholar] [CrossRef]
- Burdock, G.A. Fenaroli’s Handbook of Flavor Ingredients, 6th ed.; Burdock, G.A., Ed.; CRC Press: Orlando, FL, USA, 2016. [Google Scholar]
- Borowska, E.J.; Mazur, B.; Kopciuch, R.G.; Buszewski, B. Polyphenol, anthocyanin and resveratrol mass fractions and antioxidant properties of cranberry cultivars. Food Technol. Biotechnol. 2009, 47, 56. [Google Scholar]
- Oszmiański, J.; Lachowicz, S.; Gorzelany, J.; Matłok, N. The effect of different maturity stages on phytochemical composition and antioxidant capacity of cranberry cultivars. Eur. Food Res. Technol. 2017, 244, 705–719. [Google Scholar] [CrossRef]
- Viskelis, P.; Rubinskiene, M.; Jasutiene, I.; Sarkinas, A.; Daubaras, R.; Cesoniene, L. Anthocyanins, antioxidative, and antimicrobial properties of American cranberry (Vaccinium macrocarpon Ait.) and their press cakes. J. Food Sci. 2009, 74, C157–C161. [Google Scholar] [CrossRef]
- Brown, P.N.; Murch, S.J.; Shipley, P. Phytochemical Diversity of Cranberry (Vaccinium macrocarpon Aiton) Cultivars by Anthocyanin Determination and Metabolomic Profiling with Chemometric Analysis. J. Agric. Food Chem. 2012, 60, 261–271. [Google Scholar] [CrossRef]
- Lu, Y.; Pekerti, B.N.; Toh, Z.S.; Broom, F.; Savage, G.; Liu, S.Q.; Huang, D. Physico-chemical parameters and proanthocyanidin profiles of cranberries cultivated in New Zealand. J. Food Compos. Anal. 2017, 63, 1–7. [Google Scholar] [CrossRef]
- Jungfer, E.; Zimmermann, B.F.; Ruttkat, A.; Galensa, R. Comparing procyanidins in selected Vaccinium species by UHPLC-MS2 with regard to authenticity and health effects. J. Agric. Food Chem. 2012, 60, 9688–9696. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Aviram, M.; Zhang, Y.; Henning, S.M.; Feng, L.; Dreher, M.; Heber, D. Comparison of antioxidant potency of commonly consumed polyphenol-rich beverages in the United States. J. Agric. Food Chem. 2008, 56, 1415–1422. [Google Scholar] [CrossRef]
- Saura-Calixto, F. Concept and health-related properties of nonextractable polyphenols: The missing dietary polyphenols. J. Agric. Food Chem. 2012, 60, 11195–11200. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.O.; Trindade, S.C.; Maurer, L.H.; Bersch, A.M.; Sautter, C.K.; Penna, N.G. Physicochemical, antioxidant and sensory quality of brazilian blueberry wine. An. Acad. Bras. Cienc. 2016, 88, 1557–1568. [Google Scholar] [CrossRef] [PubMed]
- Ngamwonglumlert, L.; Devahastin, S.; Chiewchan, N. Natural colorants: Pigment stability and extraction yield enhancement via utilization of appropriate pretreatment and extraction methods. Crit. Rev. Food Sci. Nutr. 2017, 57, 3243–3259. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.; Costa, E.; Calhau, C.; Morais, R.; Pintado, M. Anthocyanin extraction from plant tissues: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 3072–3083. [Google Scholar] [CrossRef]
- Zhang, J.; Singh, R.; Quek, S.Y. Exraction of anthocyanins from natural sources-methods and commercial considerations. In Anthocyanins from Natural Sources: Exploiting Targeted Delivery for Improved Health, 1st ed.; Brooks, M., Celli, G., Eds.; Royal Society of Chemistry: Cambridge, UK, 2019; pp. 81–109. [Google Scholar]
- Wang, B.C.; He, R.; Li, Z.M. The stability and antioxidant activity of anthocyanins from blueberry. Food Technol. Biotechnol. 2010, 48, 42–49. [Google Scholar]
- Ko, M.J.; Cheigh, C.I.; Cho, S.W.; Chung, M.S. Subcritical water extraction of flavonol quercetin from onion skin. J. Food Eng. 2011, 102, 327–333. [Google Scholar] [CrossRef]
- Cote, J.; Caillet, S.; Doyon, G.; Sylvain, J.F.; Lacroix, M. Bioactive compounds in cranberries and their biological properties. Crit. Rev. Food Sci. Nutr. 2010, 50, 666–679. [Google Scholar] [CrossRef]
- Zhu, J.; Chen, F.; Wang, L.; Niu, Y.; Chen, H.; Wang, H.; Xiao, Z. Characterization of the key aroma volatile compounds in cranberry (Vaccinium macrocarpon Ait.) using gas chromatography–olfactometry (GC-O) and odor activity value (OAV). J. Agric. Food Chem. 2016, 64, 4990–4999. [Google Scholar] [CrossRef]
- Anjou, K.; von Sydow, E. The aroma of cranberries. Acta Chem. Scand. 1967, 21, 945–952. [Google Scholar] [CrossRef]
- Ferreira, V.; López, R.; Cacho, J.F. Quantitative determination of the odorants of young red wines from different grape varieties. J. Sci. Food Agric. 2000, 80, 1659–1667. [Google Scholar] [CrossRef]
- Chatonnet, P.; Viala, C.; Dubourdieu, D. Influence of Polyphenolic Components of Red Wines on the Microbial Synthesis of Volatile Phenols. Am. J. Enol. Vitic. 1997, 48, 443–448. [Google Scholar]
- Klinke, H.B.; Thomsen, A.; Ahring, B.K. Inhibition of ethanol-producing yeast and bacteria by degradation products produced during pre-treatment of biomass. Appl. Microbiol. Biotechnol. 2004, 66, 10–26. [Google Scholar] [CrossRef] [PubMed]
| Sample | TPC A | Total ANC B | Total PAC C | Total AOC D | pH | Brix (°) | |
|---|---|---|---|---|---|---|---|
| FRAP | ABTS | ||||||
| Stevens | 882.0 ± 2.5 b | 283.3 ± 21.3 a | 445.8 ± 22.2 b | 9770.0 ± 563.0 a | 9506.9 ± 21.3 a | 2.85 ± 0.04 a | 5.33 ± 0.46 a |
| Pilgrim | 660.1 ± 12.5 c | 168.1 ± 9.8 c | 397.7 ± 21.1 c | 7470.0 ± 264.6 b | 8429.5 ± 307.4 b | 2.98 ± 0.15 a | 5.20 ± 0.20 a |
| Bergman | 938.7 ± 10.3 a | 210.7 ± 5.9 b | 583.9 ± 12.8 a | 9908.9 ± 333.9 a | 9557.9 ± 24.1 a | 2.99 ± 0.18 a | 5.43 ± 0.32 a |
| RT (min) | Compounds | Concentration (µg/mL) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stevens | Pilgrim | Bergman | |||||||||||
| Clear | Thermo | Whole | Clear | Thermo | Whole | Clear | Thermo | Whole | |||||
| A | Anthocyanins | ||||||||||||
| 7.39 | Cyanidin 3-galactoside | 2.50 ± 0.21 b | 2.20 ± 0.16 b | – | 1.17 ± 0.02 c | 3.86 ± 0.05 a | 0.20 ± 0.01 d | 1.34 ± 0.03 c | 2.45 ± 0.38 b | 0.26 ± 0.12 d | |||
| 8.07 | Cyanidin 3-glucoside | 0.85 ± 0.07 ab | 0.77 ± 0.00 abc | 0.95 ± 0.28 a | 0.31 ± 0.01 c | 0.41 ± 0.06 bc | 0.71 ± 0.02 abc | 0.61 ± 0.04 abc | 0.86 ± 0.16 ab | 0.65 ± 0.15 abc | |||
| 8.54 | Cyanidin 3-arabinoside | 28.77 ± 1.49 a | 28.70 ± 0.58 a | 22.26 ± 3.22 ab | 13.11 ± 0.47 b | 15.88 ± 0.40 bcd | 17.23 ± 0.40 bcd | 18.92 ± 0.09 bcd | 21.98 ± 2.77 abc | 14.53 ± 3.34 cd | |||
| 9.18 | Peonidin 3-galactoside | 5.83 ± 0.16 a | 4.81 ± 0.18 bc | 0.24 ± 0.08 e | 2.54 ± 0.14 c | 5.52 ± 0.04 ab | 0.38 ± 0.10 e | 3.06 ± 0.09 d | 4.43 ± 0.50 c | 0.20 ± 0.14 e | |||
| 9.78 | Peonidin 3-glucoside | 4.31 ± 0.09 ab | 3.51 ± 0.10 abc | 2.78 ± 0.50 bcd | 1.76 ± 0.07 b | 1.74 ± 0.05 d | 2.01 ± 0.39 d | 3.82 ± 0.05 ab | 4.09 ± 0.49 ab | 2.17 ± 0.72 cd | |||
| 10.22 | Peonidin 3-arabinoside | 15.89 ± 0.89 a | 15.43 ± 0.22 a | 10.79 ± 1.47 bc | 7.23 ± 0.31 bc | 7.99 ± 0.24 bcd | 8.17 ± 0.56 bcd | 10.26 ± 0.01 bcd | 11.27 ± 1.14 b | 6.68 ± 1.70 d | |||
| Total | 58.15 ± 2.03 a | 55.42 ± 0.56 a | 37.03 ± 5.53 bc | 26.12 ± 1.01 c | 35.40 ± 0.54 bc | 28.69 ± 1.24 c | 38.02 ± 0.11 bc | 45.09 ± 5.44 ab | 24.50 ± 6.18 c | ||||
| B | Proanthocyanidins | ||||||||||||
| 3.51 | PAC B1 | 0.75 ± 0.14 a | 0.29 ± 0.01 a | 0.38 ± 0.07 a | 0.38 ± 0.04 a | 0.20 ± 0.05 a | 0.33 ± 0.17 a | 1.10 ± 0.35 a | 1.12 ± 0.40 a | 0.71 ± 0.41 a | |||
| 4.68 | Catechin | 2.63 ± 0.03 abc | 1.73 ± 0.04 c | 1.70 ± 0.20 c | 2.12 ± 0.30 bc | 1.29 ± 0.20 c | 2.12 ± 0.66 bc | 4.19 ± 0.88 ab | 4.74 ± 0.57 a | 3.33 ± 0.92 abc | |||
| 6.30 | PAC B2 | 3.74 ± 0.29 abc | 1.44 ± 0.10 bc | 1.73 ± 0.51 bc | 1.51 ± 0.02 bc | 0.88 ± 0.04 c | 1.87 ± 0.52 bc | 4.74 ± 1.63 ab | 5.94 ± 1.86 a | 3.56 ± 1.39 abc | |||
| 7.61 | Epicatechin | 8.41 ± 0.60 bc | 6.34 ± 0.30 c | 5.77 ± 0.77 c | 5.79 ± 0.56 c | 4.47 ± 0.31 c | 5.48 ± 1.65 c | 16.27 ± 2.94 a | 15.48 ± 2.37 ab | 11.24 ± 3.83 abc | |||
| 11.52 | PAC A2 | 3.70 ± 0.11 bc | 3.83 ± 0.09 bc | 3.80 ± 0.30 bc | 3.08 ± 0.04 c | 3.50 ± 0.38 bc | 4.00 ± 0.02 bc | 5.87 ± 1.41 ab | 7.88 ± 1.10 a | 6.25 ± 0.95 ab | |||
| Total | 19.24 ± 0.96 abc | 13.62 ± 0.33 c | 13.39 ± 1.85 c | 12.89 ± 0.88 c | 10.33 ± 0.22 c | 13.80 ± 3.03 bc | 37.01 ± 12.42 a | 35.16 ± 6.29 ab | 25.08 ± 7.50 abc | ||||
| C | Phenolic Acids | ||||||||||||
| 1.28 | Gallic acid | 0.41 ± 0.11 bcde | 0.37 ± 0.04 bcd | 0.35 ± 0.01 bcd | 0.30 ± 0.03 bcde | 0.20 ± 0.01 de | 0.12 ± 0.01 e | 2.06 ± 0.07 a | 0.48 ± 0.06 b | 0.25 ± 0.01 cde | |||
| 2.16 | Protocatechuic acid | 6.46 ± 0.89 a | 5.47 ± 0.05 ab | 4.61 ± 0.04 bc | 4.44 ± 0.00 bc | 3.64 ± 0.28 cd | 3.21 ± 0.40 cd | 5.62 ± 0.05 ab | 4.52 ± 0.09 bc | 2.96 ± 0.39 d | |||
| 4.59 | Gentisic acid | 0.89 ± 0.01 ab | 0.89 ± 0.01 ab | 0.94 ± 0.02 ab | 0.75 ± 0.05 b | 0.83 ± 0.01 ab | 0.86 ± 0.07 ab | 0.88 ± 0.10 ab | 0.95 ± 0.02 ab | 1.00 ± 0.05 ab | |||
| 5.09 | β-Resorcylic acid | − | − | − | − | − | 0.43 ± 0.19 a | − | − | − | |||
| 5.21 | Chlorogenic acid | 0.87 ± 0.08 bc | 0.74 ± 0.12 c | 1.04 ± 0.10 bc | 0.97 ± 0.42 bc | 1.90 ± 0.31 ab | 1.14 ± 0.03 bc | 2.80 ± 1.07 ab | 3.95 ± 0.85 a | 2.64 ± 0.48 abc | |||
| 5.71 | Caffeic acid | 2.72 ± 0.28 a | 2.35 ± 0.03 a | 2.05 ± 0.10 a | 1.87 ± 0.24 a | 1.87 ± 0.40 a | 1.80 ± 0.01 a | 2.32 ± 0.00 a | 2.55 ± 0.19 a | 2.34 ± 0.44 a | |||
| 8.45 | p-Coumaric acid | 10.11 ± 0.16 ab | 10.99 ± 0.35 ab | 8.83 ± 0.43 ab | 10.93 ± 0.59 ab | 10.21 ± 0.52 ab | 8.42 ± 0.86 b | 9.38 ± 0.36 ab | 11.24 ± 0.39 a | 11.19 ± 1.56 ab | |||
| Total | 21.47 ± 1.35 abc | 20.86 ± 0.48 abc | 17.94 ± 0.78 bc | 19.26 ± 1.32 abc | 18.64 ± 1.51 abc | 15.98 ± 0.69 c | 23.06 ± 1.51 ab | 23.69 ± 0.67 a | 20.37 ± 2.91 abc | ||||
| D | Flavonols | ||||||||||||
| 9.86 | Myricetin-3-galactoside | 1.08 ± 0.04 d | 1.44 ± 0.01 d | 1.62 ± 0.07 d | 1.53 ± 0.23 d | 4.52 ± 0.06 b | 2.46 ± 0.07 cd | 3.82 ± 1.04 bc | 7.02 ± 0.75 a | 2.76 ± 0.26 bcd | |||
| 10.09 | Myricetin-3-glucoside | 0.47 ± 0.05 ab | 0.48 ± 0.02 ab | 0.40 ± 0.04 ab | 0.31 ± 0.03 b | 0.45 ± 0.06 ab | 0.31 ± 0.01 b | 0.49 ± 0.15 ab | 0.61 ± 0.05 ab | 0.38 ± 0.02 ab | |||
| 11.39 | Quercetin-3-galactoside | 0.17 ± 0.03 c | 0.20 ± 0.00 c | 0.07 ± 0.01 c | 0.69 ± 0.08 bc | 3.27 ± 0.21 a | 0.46 ± 0.14 c | 1.69 ± 0.60 b | 3.78 ± 0.41 a | 0.34 ± 0.16 c | |||
| 12.90 | Quercetin-3-rhamnoside | 6.56 ± 0.28 ab | 6.64 ± 0.03 ab | 5.88 ± 0.13 ab | 5.06 ± 0.47 b | 5.79 ± 0.93 ab | 5.60 ± 0.19 ab | 6.68 ± 0.80 ab | 7.63 ± 0.62 a | 6.31 ± 0.74 ab | |||
| 14.76 | Phloridzin | 0.93 ± 0.00 a | 1.00 ± 0.03 a | 0.84 ± 0.03 a | 0.78 ± 0.06 a | 1.13 ± 0.18 a | 0.82 ± 0.02 a | 1.02 ± 0.40 a | 1.17 ± 0.27 a | 0.96 ± 0.15 a | |||
| 18.68 | Quercetin | 15.38 ± 1.88 bcd | 24.37 ± 0.10 a | 9.58 ± 0.05 def | 12.39 ± 0.03 cde | 17.31 ± 0.53 bcd | 8.90 ± 1.55 ef | 12.18 ± 1.45 cde | 13.95 ± 1.37 bcd | 7.29 ± 1.33 f | |||
| Total | 24.59 ± 2.28 bc | 34.13 ± 0.08 a | 18.38 ± 0.11 c | 20.75 ± 0.85 c | 32.49 ± 0.92 ab | 18.55 ± 1.94 c | 25.87 ± 4.42 abc | 34.16 ± 3.47 a | 18.02 ± 2.66 c | ||||
| No. | Compound | CAS | Codes | Concentration (µg/L) | p-value | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stevens | Pilgrim | Bergman | |||||||||||||||||||||||
| White | SD% | Thermo | SD% | Red | SD% | White | SD% | Thermo | SD% | Red | SD% | White | SD% | Thermo | SD% | Red | SD% | Cultivar | Vinification | ||||||
| Esters | |||||||||||||||||||||||||
| 1 | Ethyl Acetate | 141-78-6 | ES1 | 5,716 ab | 11 | 4811 bc | 3 | 5,648 ab | 9 | 5714 ab | 2 | 4,929 bc | 9 | 6904 a | 6 | 5582 abc | 4 | 4106 c | 17 | 6345 a | 5 | 0.1 | <0.0001 * | ||
| 2 | Methyl butanoate | 623-42-7 | ES2 | 0.72 de | 13 | 0.52 e | 12 | 0.49 e | 9 | 1.62 b | 6 | 1.17 c | 3 | 2.15 a | 7 | 1.16 c | 11 | 1.00 cd | 23 | 1.08 cd | 16 | <0.0001 * | 0.0223 * | ||
| 3 | Isobutyl acetate | 110-19-0 | ES3 | 10.02 a | 12 | 10.45 a | 2 | 11.00 a | 4 | 10.75 a | 2 | 11.23 a | 4 | 11.33 a | 6 | 10.55 a | 16 | 9.35 a | 2 | 10.81 a | 2 | 0.1 | 0.3 | ||
| 4 | Ethyl butyrate | 105-54-4 | ES4 | 35.76 bc | 9 | 33.55 bc | 4 | 38.97 abc | 7 | 44.96 abc | 13 | 46.93 ab | 11 | 53.08 a | 14 | 39.04 abc | 24 | 30.40 c | 5 | 34.47 bc | 3 | <0.0001 * | 0.2 | ||
| 5 | Ethyl methylbutyrate | 7452-79-1 | ES5 | 11.09 a | 6 | 9.11 a | 13 | 11.65 a | 7 | 10.71 a | 20 | 11.53 a | 23 | 13.24 a | 26 | 11.27 a | 31 | 7.85 a | 28 | 9.43 a | 13 | 0.2 | 0.3 | ||
| 6 | Isoamyl acetate | 123-92-2 | ES6 | 27.89 ab | 15 | 25.71 c | 3 | 38.49 b | 5 | 32.70 bc | 7 | 31.05 bc | 7 | 58.39 a | 17 | 29.65 bc | 15 | 26.45 bc | 5 | 40.14 b | 2 | 0.0108 * | 0.0011 * | ||
| 7 | Methyl hexanoate | 106-70-7 | ES7 | 7.87 ab | 3 | 8.39 a | 3 | 4.37 d | 3 | 8.44 a | 5 | 7.54 ab | 10 | 4.86 d | 8 | 6.58 bc | 11 | 8.14 a | 1 | 5.23 cd | 7 | 0.9 | <0.0001 * | ||
| 8 | Ethyl hexanoate | 123-66-0 | ES8 | 35.86 ab | 18 | 29.70 bc | 12 | 16.40 d | 2 | 42.18 a | 11 | 37.85 ab | 2 | 28.44 bc | 13 | 22.95 cd | 19 | 22.48 cd | 6 | 12.66 d | 3 | <0.0001 * | <0.0001 * | ||
| 9 | Methyl benzoate | 93-58-3 | ES9 | 83.25 bcd | 11 | 57.52 cd | 14 | 101.03 abc | 15 | 106.65 ab | 15 | 48.11 d | 4 | 145.55 a | 20 | 74.95 bcd | 5 | 69.83 bcd | 20 | 110.20 ab | 23 | 0.2 | 0.0003 * | ||
| 10 | Ethyl octanoate | 106-32-1 | ES10 | 43.79 ab | 5 | 34.94 bc | 14 | 6.25 e | 7 | 49.37 a | 18 | 40.39 abc | 10 | 14.88 de | 18 | 26.67 cd | 18 | 31.18 bc | 5 | 4.98 e | 4 | 0.1 | <0.0001 * | ||
| 11 | Ethyl benzoate | 93-89-0 | ES11 | 3,752 ab | 11 | 2,701 bc | 16 | 1631 c | 20 | 4,928 a | 17 | 1,730 c | 7 | 3122 bc | 18 | 4,128 ab | 23 | 3068 bc | 17 | 2412 bc | 26 | 0.4 | <0.0001* | ||
| 12 | β-phenylethyl acetate | 103-45-7 | ES12 | 267.79 a | 2 | 262.27 ab | 0 | 252.14 b | 2 | 271.28 a | 1 | 271.15 a | 2 | 267.32 a | 3 | 263.57 ab | 2 | 260.15 ab | 0 | 257.77 ab | 1 | 0.0049 * | 0.0275* | ||
| 13 | Ethyl decanoate | 110-38-3 | ES13 | 5.18 a | 8 | 4.43 a | 9 | 1.82 b | 1 | 5.03 a | 5 | 4.80 a | 15 | 2.16 b | 9 | 4.15 a | 1 | 4.09 a | 18 | 1.80 b | 2 | 0.5 | <0.0001* | ||
| 14 | Ethyl cinnamate (trans) | 103-36-6 | ES14 | 93.11 b | 10 | 99.84 b | 20 | 22.73 f | 7 | 170.71 a | 4 | 83.96 bc | 13 | 36.94 def | 7 | 62.05 cd | 2 | 59.35 cde | 13 | 27.83 ef | 5 | 0.1 | 0.0011 * | ||
| Alcohols | |||||||||||||||||||||||||
| 15 | Isobutanol | 78-83-1 | ACH1 | 11,764 bc | 22 | 11,797 bc | 7 | 13,601 ab | 3 | 15,844 a | 3 | 15,020 ab | 11 | 16,475 a | 5 | 11,010 bc | 17 | 7,515 c | 8 | 15,331 ab | 9 | <0.0001 * | 0.0044 * | ||
| 16 | Isoamylalcohol | 123-51-3 | ACH2 | 26,280 a | 14 | 25,705 a | 6 | 26,720 a | 3 | 28,911 a | 3 | 26,997 a | 6 | 30,073 a | 2 | 25,320 a | 7 | 25,281 a | 18 | 23,904 a | 11 | 0.0087 * | 0.5 | ||
| 17 | α-terpineol | 98-55-5 | ACH3 | 288.42 bc | 10 | 228.89 c | 6 | 293.06 b | 7 | 286.13 bc | 3 | 272.78 bc | 4 | 319.28 b | 5 | 423.71 a | 11 | 415.03 a | 3 | 473.69 a | 0 | <0.0001 * | 0.0006 * | ||
| 18 | Benzyl alcohol | 100-51-6 | ACH4 | 1,842 ab | 4 | 1768 b | 9 | 2604 a | 2 | 918 cd | 4 | 752.67 d | 4 | 1,843 a | 13 | 1181 bcd | 31 | 1527 bcd | 10 | 1785 abc | 7 | 0.0002 * | 0.0007 * | ||
| 19 | Geraniol (trans) | 106-24-1 | ACH5 | 68.50 a | 1 | 58.97 a | 16 | 64.67 a | 10 | 84.70 a | 14 | 85.71 a | 15 | 95.74a | 23 | 75.40 a | 33 | 65.46 a | 26 | 73.03 a | 14 | 0.0084 * | 0.5 | ||
| 20 | Phenylethyl alcohol | 60-12-8 | ACH6 | 3,116 bc | 13 | 2904 c | 8 | 2859 c | 5 | 4108 a | 2 | 3,308 bc | 8 | 3778ab | 6 | 2990 bc | 5 | 2775 c | 14 | 2752 c | 7 | <0.0001 * | 0.0225 * | ||
| Acids | |||||||||||||||||||||||||
| 21 | Methylbutyric acid | 116-53-0 | ACID1 | 523.23 ab | 10 | 429.26 abcde | 21 | 292.55 e | 6 | 541.72 a | 5 | 456.33 abcd | 13 | 317.79 de | 17 | 496.39 abc | 2 | 366.73 bcde | 15 | 330.94 cde | 1 | 0.9 | 0.0001 * | ||
| 22 | Hexanoic acid | 142-62-1 | ACID2 | 306.54 cde | 6 | 475.47 ab | 16 | 272.14 de | 6 | 451.20 abc | 8 | 579.65 a | 12 | 345.68 bcd | 12 | 300.40 cde | 33 | 255.80 de | 2 | 178.86 e | 11 | <0.0001 * | 0.0001 * | ||
| 23 | Octanoic acid | 124-07-2 | ACID3 | 272.72 ab | 12 | 250.15 ab | 20 | 54.32 c | 8 | 325.28 a | 7 | 255.01 ab | 17 | 91.84 c | 10 | 200.62 b | 6 | 195.79 b | 14 | 41.50 c | 8 | 0.2 | <0.0001 * | ||
| 24 | Decanoic acid | 334-48-5 | ACID4 | 18.73 b | 4 | 20.77 b | 25 | 4.68 d | 8 | 30.21ab | 6 | 8.57 cd | 26 | 6.42 cd | 3 | 21.22 ab | 2 | 15.37bc | 2 | 4.22 d | 4 | 0.9 | 0.0004 * | ||
| 25 | Benzoic acid | 65-85-0 | ACID5 | 65,564 ab | 8 | 59,455 abcd | 13 | 53162 bcd | 10 | 60,490abcd | 7 | 47,267 d | 10 | 49,584 cd | 4 | 72,760 a | 3 | 59,848abcd | 3 | 64,506 abc | 9 | 0.0003 * | 0.0019 * | ||
| Others | |||||||||||||||||||||||||
| 26 | Pentanal | 110-62-3 | PEN | 11.55 a | 2 | 13.65 a | 5 | 6.76 a | 8 | 9.41a | 5 | 15.47 a | 10 | 6.43 a | 14 | 6.76 a | 8 | 7.80ab | 7 | 7.06 ab | 7 | 0.9 | 0.1 | ||
| 27 | 3-Octanone | 106-68-3 | OCT | 0.82 bc | 24 | 0.67 bcd | 16 | 0.16 e | 15 | 1.21a | 13 | 0.92 ab | 17 | 0.90 ab | 13 | 0.49 cde | 2 | 0.58bc | 6 | 0.30 c | 16 | <0.0001 * | 0.0032 * | ||
| 28 | D-Limonene | 5989-27-5 | LIM | 7.47 cd | 2 | 6.45 e | 6 | 6.51 de | 1 | 6.87cde | 2 | 6.42 e | 7 | 7.66 bc | 2 | 8.72 ab | 4 | 9.36a | 8 | 8.97 a | 2 | <0.0001 * | 0.2 | ||
| 29 | β-damascenone | 23696-85-7 | DAM | 1.40 b | 8 | 0.93 b | 11 | 0.75 b | 30 | 1.32b | 9 | 0.95 b | 35 | 4.96 a | 15 | 1.18 b | 4 | 1.23b | 19 | 0.75 b | 24 | 0.0494 * | 0.3 | ||
| 30 | 4-ethylguaiacol | 2785-89-9 | EG | 8.46 a | 10 | 8.35 a | 16 | 6.79 ab | 4 | 5.67 b | 6 | 0.52 d | 16 | 1.47 cd | 9 | 3.07 bc | 3 | 2.23 c | 2 | 1.56 d | 2 | <0.0001 * | 0.0042 * | ||
| 31 | 4-ethylphenol | 123-07-9 | EP | 20.31 a | 12 | 11.32 b | 10 | 11.82 b | 4 | 6.25 c | 2 | 2.49 d | 3 | 4.05 cd | 3 | 3.16 cd | 1 | 3.15 c | 1 | 3.88 cd | 1 | <0.0001 * | 0.0012 * | ||
| No. | Compounds | Descriptor a | OAV | Thresholds (µg/kg) b | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stevens | Pilgrim | Bergman | ||||||||||||
| White | Thermo | Red | White | Thermo | Red | White | Thermo | Red | ||||||
| 1 | Ethyl Acetate | floral | 1143 | 962 | 1130 | 1143 | 986 | 1381 | 1116 | 821 | 1269 | 5 | ||
| 2 | Methyl butyrate | apple | <1 | <1 | <1 | 2 | 1 | 2 | 1 | 1 | 1 | 1 | ||
| 3 | Ethyl butyrate | fruity | 358 | 336 | 390 | 450 | 469 | 531 | 390 | 304 | 345 | 0.1 | ||
| 4 | Ethyl methylbutyrate | fruity | 1109 | 911 | 1165 | 1071 | 1153 | 1324 | 1127 | 785 | 943 | 0.01 | ||
| 5 | Isoamyl acetate | banana | 14 | 13 | 19 | 16 | 16 | 29 | 15 | 13 | 20 | 2 | ||
| 6 | Ethyl hexanoate | fruity | 120 | 99 | 55 | 141 | 126 | 95 | 76 | 75 | 42 | 0.3 | ||
| 7 | Methyl benzoate | herb | <1 | <1 | <1 | <1 | <1 | 1 | <1 | <1 | 1 | 110 | ||
| 8 | Ethyl octanoate | fruity | 9 | 7 | 1 | 10 | 8 | 3 | 5 | 6 | 1 | 5 | ||
| 9 | Ethyl benzoate | floral, fruity | 38 | 27 | 16 | 49 | 17 | 31 | 41 | 31 | 24 | 100 | ||
| 10 | Ethyl cinnamate (trans) | floral | 5 | 6 | 1 | 10 | 5 | 2 | 4 | 3 | 2 | 17 | ||
| 11 | Isobutanol | wine | 33 | 33 | 38 | 44 | 42 | 46 | 31 | 21 | 43 | 360 | ||
| 12 | Isoamylalcohol | whiskey, pungent | 105 | 103 | 107 | 116 | 108 | 120 | 101 | 101 | 96 | 250 | ||
| 13 | α-terpineol | fresh, minty | 10 | 8 | 10 | 10 | 10 | 11 | 15 | 15 | 17 | 28 | ||
| 14 | Benzyl alcohol | floral | 1535 | 1473 | 2170 | 765 | 627 | 1536 | 985 | 1273 | 1487 | 1.2 | ||
| 15 | Geraniol (trans) | rose | 17 | 15 | 16 | 21 | 21 | 24 | 19 | 16 | 18 | 4 | ||
| 16 | Phenylethyl alcohol | green, floral | 207,752 | 193,581 | 190,631 | 273,855 | 220,509 | 251,881 | 199,320 | 185,010 | 183,453 | 0.015 | ||
| 17 | Methylbutyric acid | cheese, sweat | 52 | 43 | 29 | 54 | 46 | 32 | 50 | 37 | 33 | 10 | ||
| 18 | Hexanoic acid | sweat | 3 | 5 | 3 | 5 | 6 | 4 | 3 | 3 | 2 | 93 | ||
| 19 | Pentanal | pungent | <1 | 1 | <1 | <1 | 1 | <1 | 1 | <1 | <1 | 12 | ||
| 20 | D-Limonene | citrus, mint | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 4 | ||
| 21 | β-damascenone | apple, rose, honey | 1993 | 1328 | 1069 | 1888 | 1353 | 7085 | 1683 | 1763 | 1074 | 0.0007 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Chen, D.; Chen, X.; Kilmartin, P.; Quek, S.Y. The Influence of Vinification Methods and Cultivars on the Volatile and Phenolic Profiles of Fermented Alcoholic Beverages from Cranberry. Antioxidants 2019, 8, 144. https://doi.org/10.3390/antiox8050144
Zhang J, Chen D, Chen X, Kilmartin P, Quek SY. The Influence of Vinification Methods and Cultivars on the Volatile and Phenolic Profiles of Fermented Alcoholic Beverages from Cranberry. Antioxidants. 2019; 8(5):144. https://doi.org/10.3390/antiox8050144
Chicago/Turabian StyleZhang, Jingying, Donglin Chen, Xiao Chen, Paul Kilmartin, and Siew Young Quek. 2019. "The Influence of Vinification Methods and Cultivars on the Volatile and Phenolic Profiles of Fermented Alcoholic Beverages from Cranberry" Antioxidants 8, no. 5: 144. https://doi.org/10.3390/antiox8050144
APA StyleZhang, J., Chen, D., Chen, X., Kilmartin, P., & Quek, S. Y. (2019). The Influence of Vinification Methods and Cultivars on the Volatile and Phenolic Profiles of Fermented Alcoholic Beverages from Cranberry. Antioxidants, 8(5), 144. https://doi.org/10.3390/antiox8050144