Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies
Abstract
1. Introduction
2. Methodology
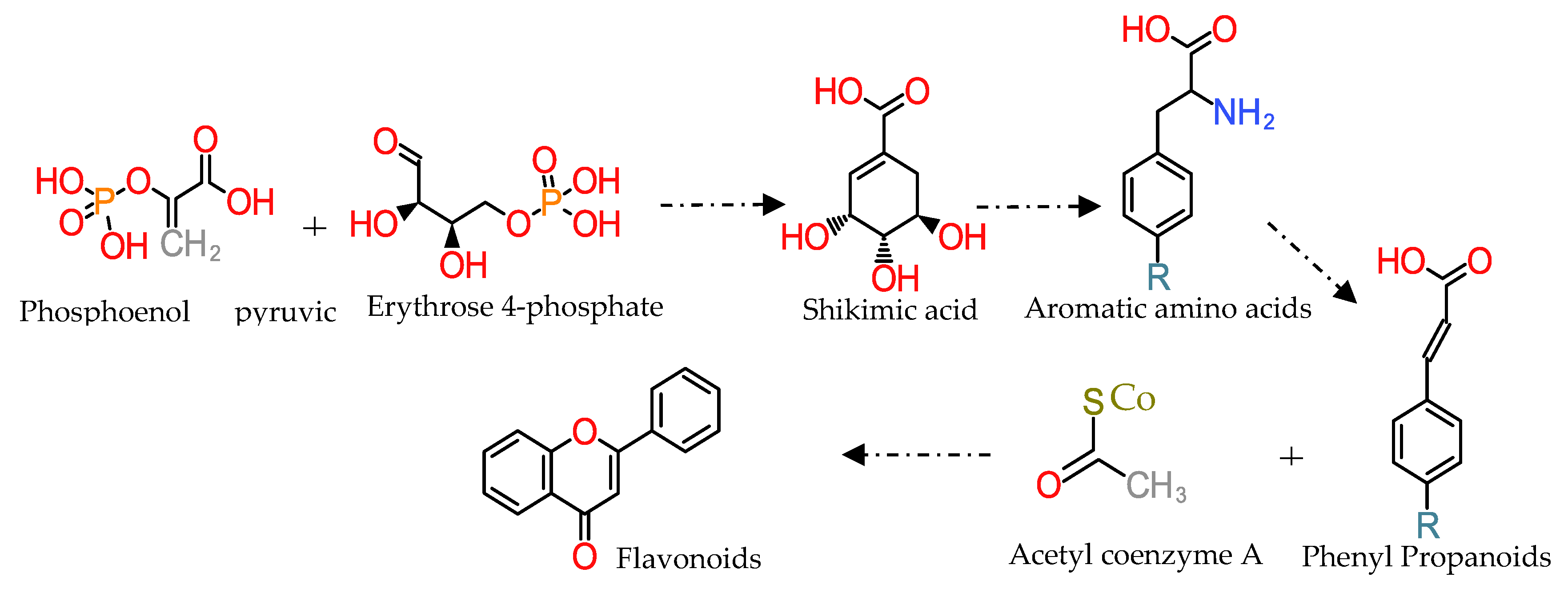
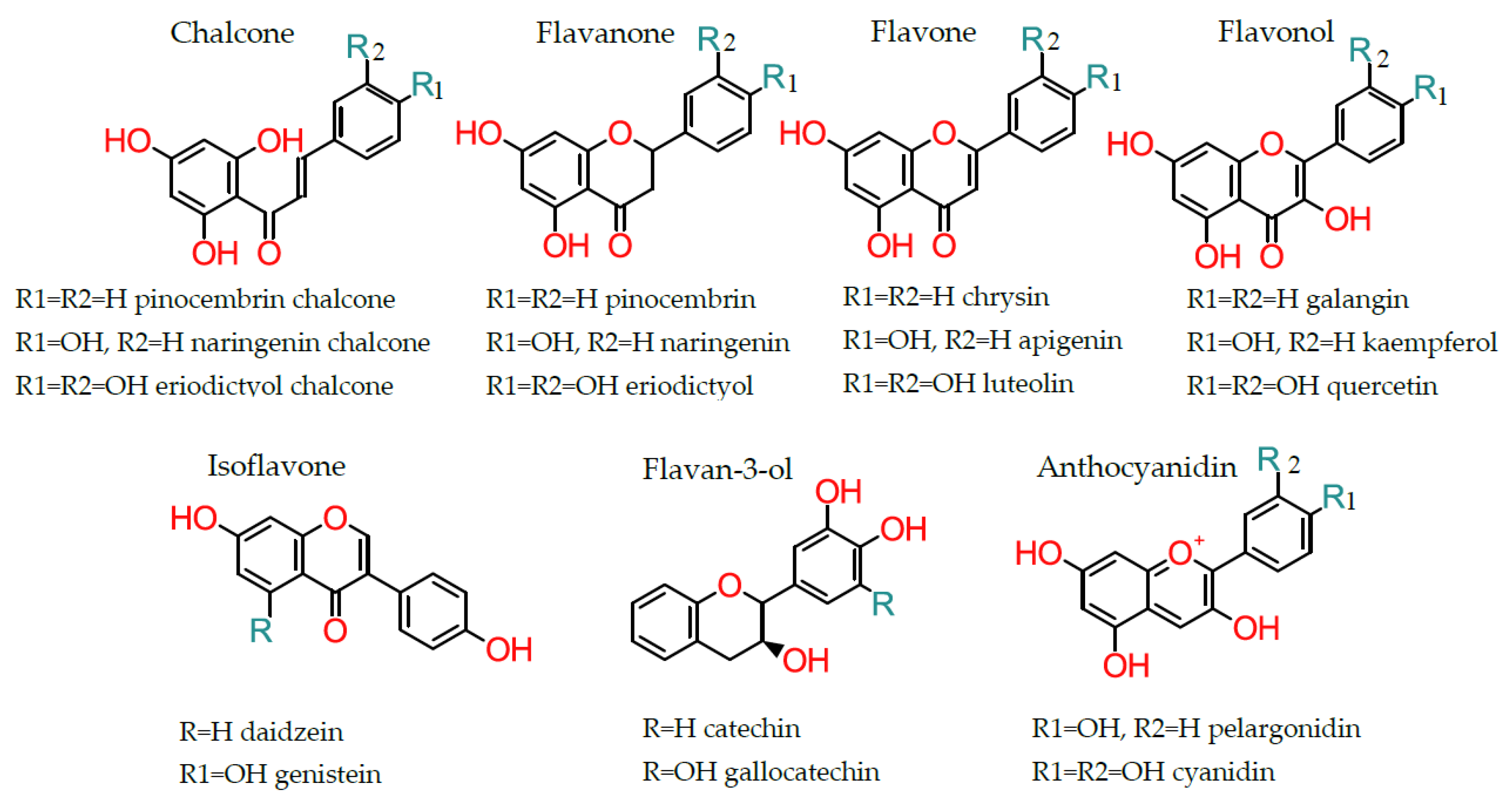
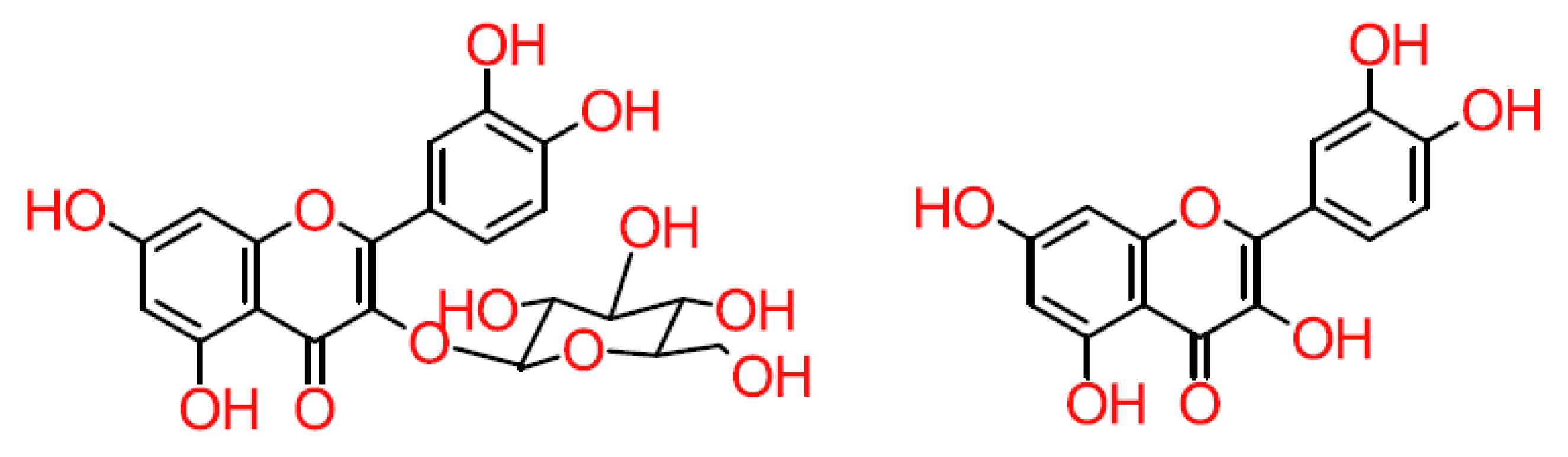
3. Biosynthesis and Subclasses of Flavonoids
4. Dietary Flavonoids
5. Pharmacokinetics
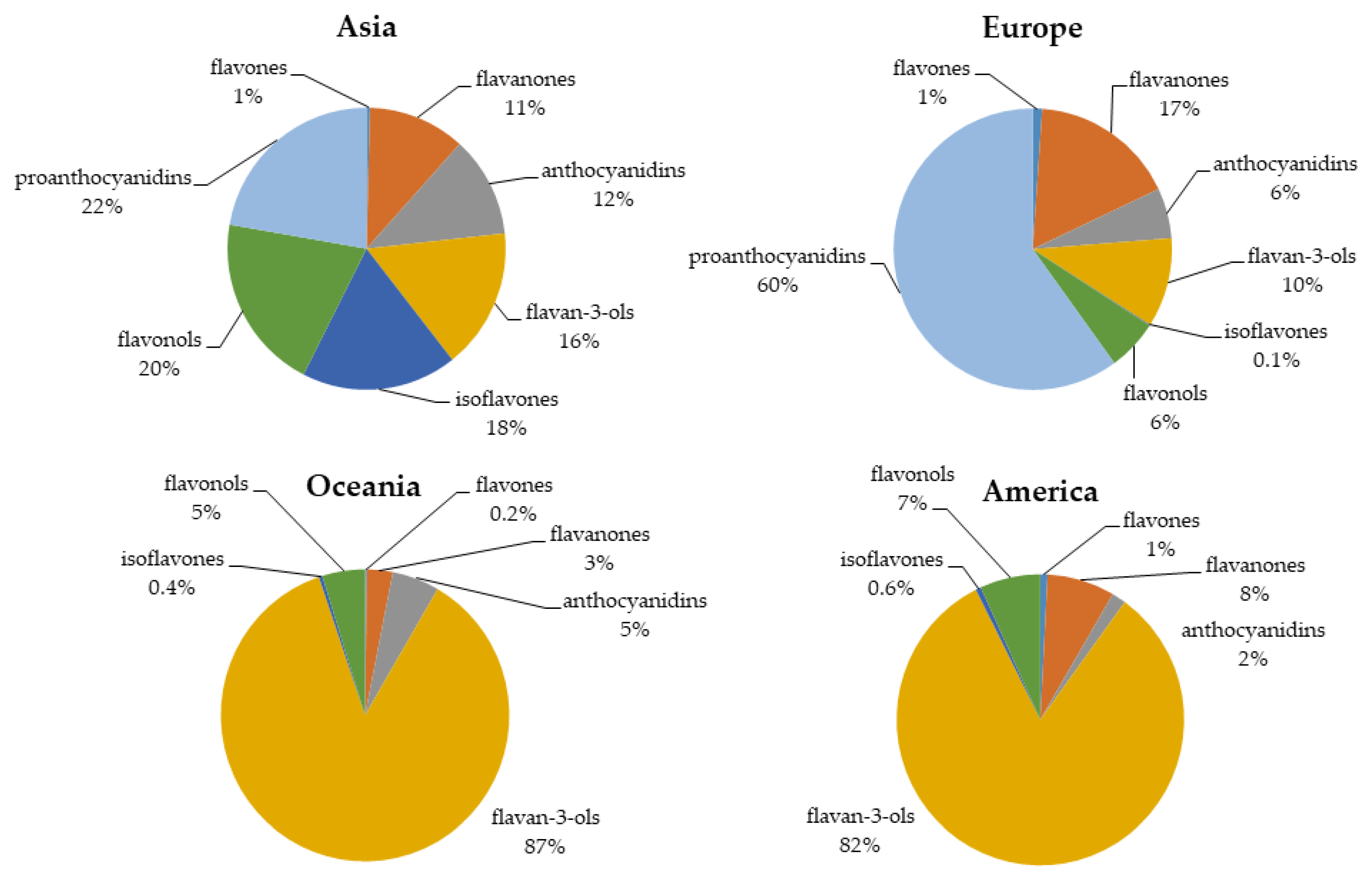
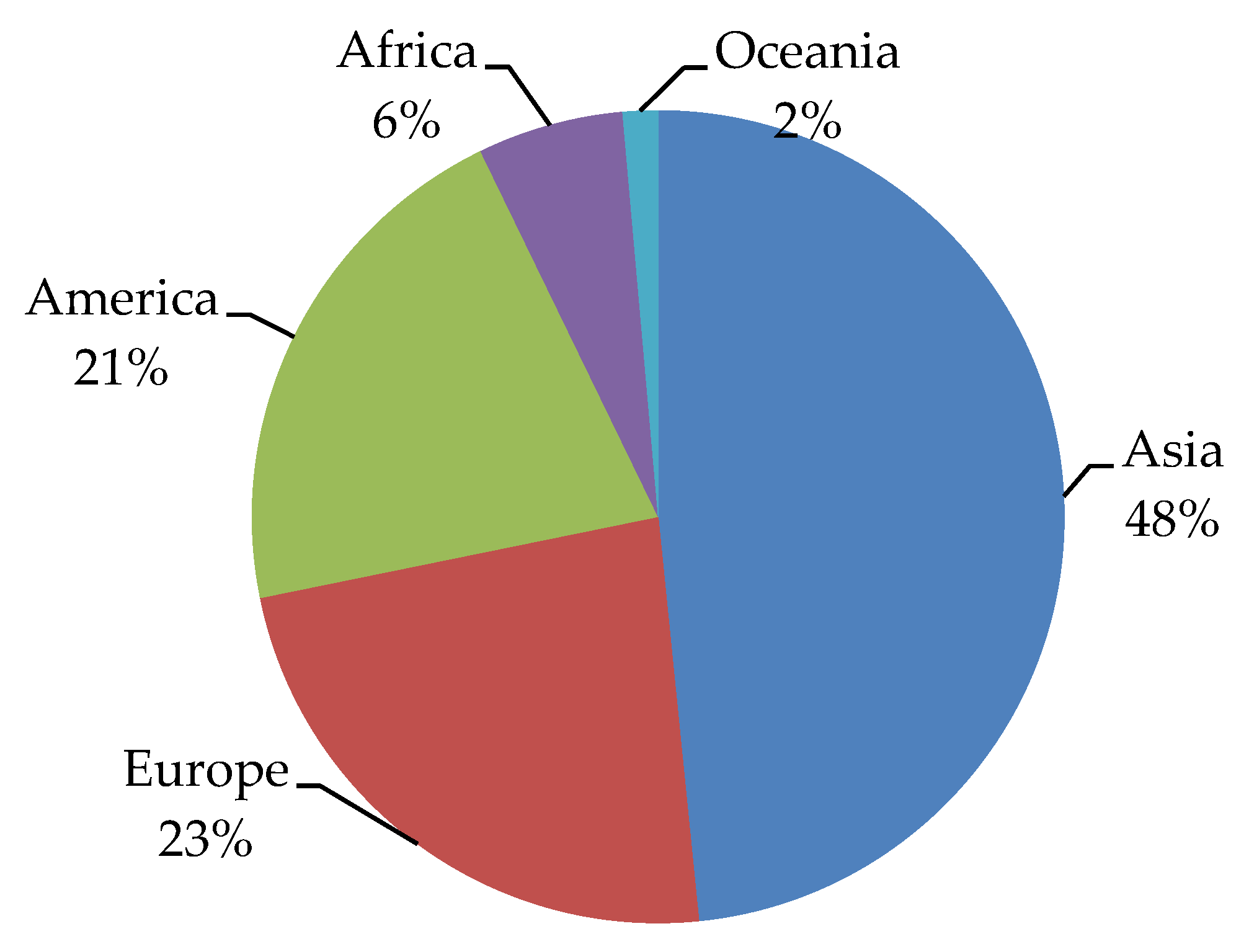
6. Worldwide Flavonoid Intake
6.1. Asia
6.2. Europe
6.3. Oceania
6.4. North America
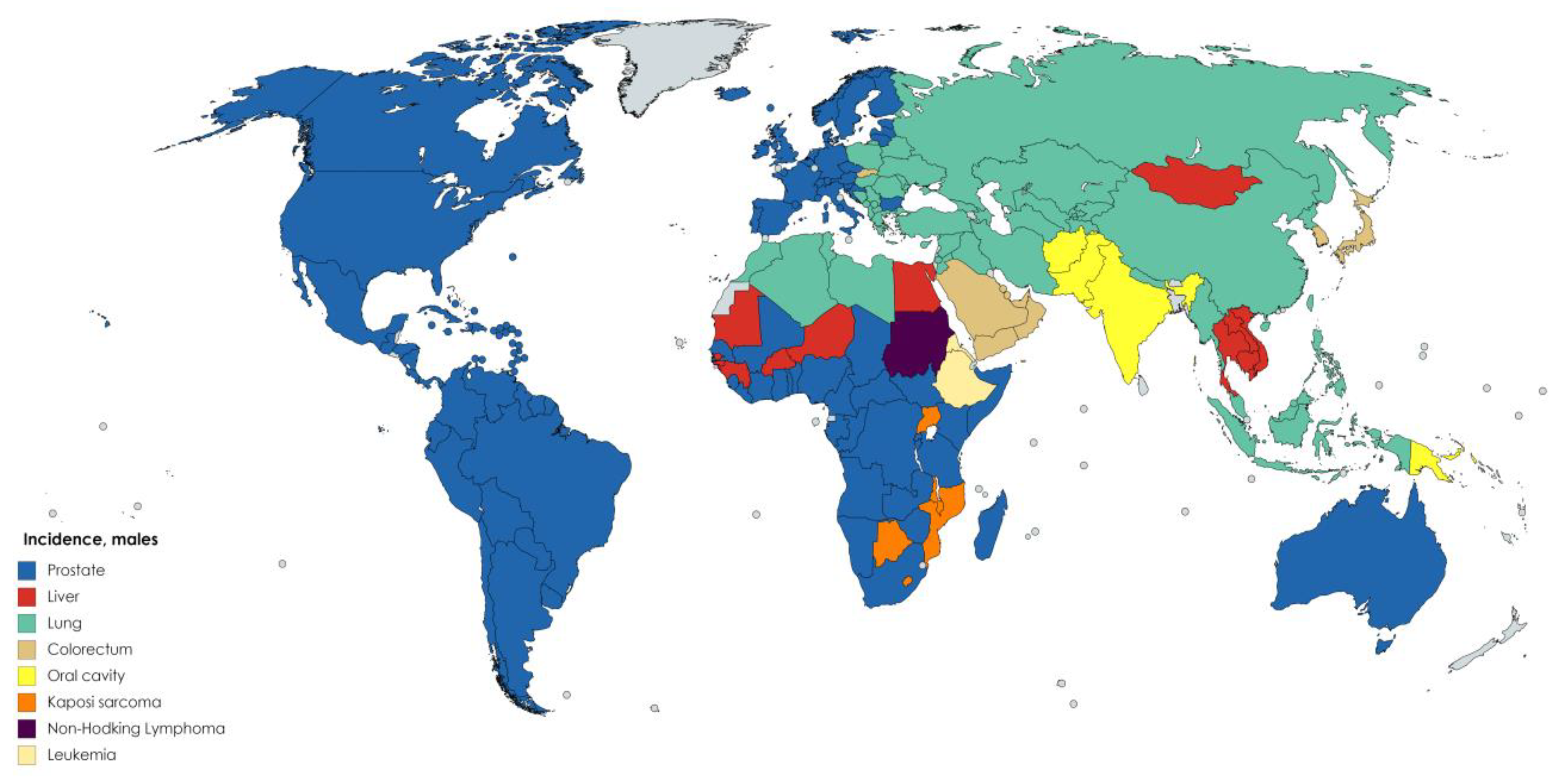
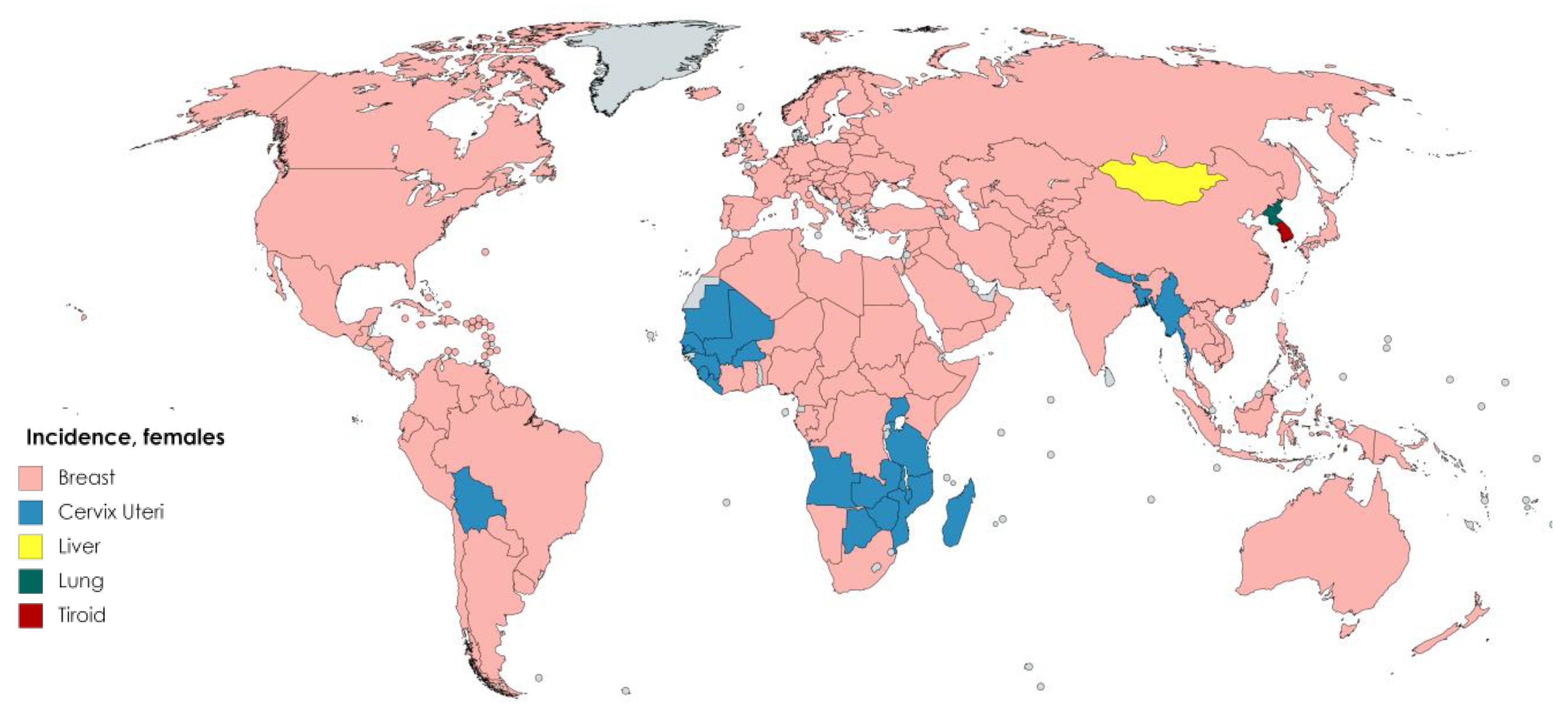
7. Antioxidant Activity of Dietary Flavonoids and Cancer Incidence
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeem, M.; Shariati, M.A.; Khan, I.A.; Imran, A.; Orhan, I.E.; Rizwan, M.; Atif, M.; et al. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Theodoratou, E.; Timofeeva, M.; Li, X.; Meng, X.; Ioannidis, J.P.A. Nature, nurture, and cancer risks: Genetic and nutritional contributions to cancer. Annu. Rev. Nutr. 2017, 37, 293–320. [Google Scholar] [CrossRef] [PubMed]
- Afshin, A.; Sur, P.J.; Fay, K.A.; Cornaby, L.; Ferrara, G.; Salama, J.S.; Mullany, E.C.; Abate, K.H.; Abbafati, C.; Abebe, Z.; et al. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the global burden of disease study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- Darband, S.G.; Kaviani, M.; Yousefi, B.; Sadighparvar, S.; Pakdel, F.G.; Attari, J.A.; Mohebbi, I.; Naderi, S.; Majidinia, M. Quercetin: A functional dietary flavonoid with potential chemo-preventive properties in colorectal cancer. J. Cell Physiol. 2018, 233, 6544–6560. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. Nutritional epidemiology and cancer: A tale of two cities. Cancer Causes Control 2018, 29, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.J.; Li, H.B. Natural antioxidants in foods and medicinal plants: extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Shashirekha, M.N.; Mallikarjuna, S.E.; Rajarathnam, S. Status of bioactive compounds in foods, with focus on fruits and vegetables. Crit. Rev. Food Sci. Nutr. 2015, 55, 1324–1339. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.M.; Samec, D.; Tomczyk, M.; Milella, L.; Russo, D.; Habtemariam, S.; Suntar, I.; Rastrelli, L.; Daglia, M.; Xiao, J.; et al. Flavonoid biosynthetic pathways in plants: Versatile targets for metabolic engineering. Biotechnol. Adv. 2018. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, X.; Yong, H.; Kan, J.; Jin, C. Recent advances in flavonoid-grafted polysaccharides: Synthesis, structural characterization, bioactivities and potential applications. Int. J. Biol. Macromol. 2018, 116, 1011–1025. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Lin, P.H.; Shih, Y.H.; Wang, K.L.; Hong, Y.H.; Shieh, T.M.; Huang, T.C.; Hsia, S.M. Natural antioxidant resveratrol suppresses uterine fibroid cell growth and extracellular matrix formation in vitro and in vivo. Antioxidants 2019, 8, 99. [Google Scholar] [CrossRef]
- Sudhakaran, M.; Sardesai, S.; Doseff, A.I. Flavonoids: new frontier for immuno-regulation and breast cancer control. Antioxidants 2019, 8, 103. [Google Scholar] [CrossRef]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radic. Biol. Med. 2006, 41, 1727–1746. [Google Scholar] [CrossRef]
- Cutler, G.J.; Nettleton, J.A.; Ross, J.A.; Harnack, L.J.; Jacobs, D.R., Jr.; Scrafford, C.G.; Barraj, L.M.; Mink, P.J.; Robien, K. Dietary flavonoid intake and risk of cancer in postmenopausal women: The Iowa Women’s Health Study. Int. J. Cancer 2008, 123, 664–671. [Google Scholar] [CrossRef]
- PubMed. Available online: https://www.ncbi.nlm.nih.gov/pubmed/ (accessed on 9 May 2019).
- Web of Science Database. Available online: https://apps.webofknowledge.com/WOS_GeneralSearch_input.do?product=WOS&search_mode=GeneralSearch&SID=D6uwPoN96kn8K5NwIjw&preferencesSaved= (accessed on 9 May 2019).
- Scarano, A.; Chieppa, M.; Santino, A. Looking at flavonoid biodiversity in horticultural crops: A colored mine with nutritional benefits. Plants 2018, 7, 98. [Google Scholar] [CrossRef]
- Maatta-Riihinen, K.R.; Kamal-Eldin, A.; Torronen, A.R. Identification and quantification of phenolic compounds in berries of Fragaria and Rubus species (family Rosaceae). J. Agric. Food Chem. 2004, 52, 6178–6187. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Gu, L.; Prior, R.L.; McKay, S. Characterization of anthocyanins and proanthocyanidins in some cultivars of Ribes, Aronia, and Sambucus and their antioxidant capacity. J. Agric. Food Chem. 2004, 52, 7846–7856. [Google Scholar] [CrossRef]
- de Pascual-Teresa, S.; Santos-Buelga, C.; Rivas-Gonzalo, J.C. Quantitative analysis of flavan-3-ols in spanish foodstuffs and beverages. J. Agric. Food Chem. 2000, 48, 5331–5337. [Google Scholar] [CrossRef] [PubMed]
- Arts, I.C.; van de Putte, B.; Hollman, P.C. Catechin contents of foods commonly consumed in The Netherlands. 1. Fruits, vegetables, staple foods, and processed foods. J. Agric. Food Chem. 2000, 48, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- Vrhovsek, U.; Rigo, A.; Tonon, D.; Mattivi, F. Quantitation of polyphenols in different apple varieties. J. Agric. Food Chem. 2004, 52, 6532–6538. [Google Scholar] [CrossRef]
- Landberg, R.; Naidoo, N.; van Dam, R.M. Diet and endothelial function: From individual components to dietary patterns. Curr. Opin. Lipidol. 2012, 23, 147–155. [Google Scholar] [CrossRef]
- Medina-Remón, A.; Manach, C.; Knox, C.; Wishart, D.S.; Perez-Jimenez, J.; Rothwell, J.A.; M’Hiri, N.; García-Lobato, P.; Eisner, R.; Neveu, V.; et al. Phenol-Explorer 3.0: A major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database 2013, 2013. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.A.; Garcia-Grau, M.M.; Tomas-Lorente, F. Flavonoid concentration changes in maturing broad bean pods. J. Agric. Food Chem. 1991, 39, 255–258. [Google Scholar] [CrossRef]
- Romani, A.; Mulinacci, N.; Pinelli, P.; Vincieri, F.F.; Cimato, A. Polyphenolic content in five tuscany cultivars of Olea europaea L. J. Agric. Food Chem. 1999, 47, 964–967. [Google Scholar] [CrossRef]
- Rodriguez Galdon, B.; Pena-Mendez, E.; Havel, J.; Rodriguez, E.M.; Romero, C.D. Cluster analysis and artificial neural networks multivariate classification of onion varieties. J. Agric. Food Chem. 2010, 58, 11435–11440. [Google Scholar] [CrossRef]
- Howard, L.R.; Pandjaitan, N.; Morelock, T.; Gil, M.I. Antioxidant capacity and phenolic content of spinach as affected by genetics and growing season. J. Agric. Food Chem. 2002, 50, 5891–5896. [Google Scholar] [CrossRef]
- Fattorusso, E.; Iorizzi, M.; Lanzotti, V.; Taglialatela-Scafati, O. Chemical composition of shallot (Allium ascalonicum Hort.). J. Agric. Food Chem. 2002, 50, 5686–5690. [Google Scholar] [CrossRef]
- Rienks, J.; Barbaresko, J.; Nothlings, U. Association of isoflavone biomarkers with risk of chronic disease and mortality: A systematic review and meta-analysis of observational studies. Nutr. Rev. 2017, 75, 616–641. [Google Scholar] [CrossRef]
- Steadman, K.J.; Burgoon, M.S.; Lewis, B.A.; Edwardson, S.E.; Obendorf, R.L. Minerals, phytic acid, tannin and rutin in buckwheat seed milling fractions. J. Sci. Food Agric. 2001, 81, 1094–1100. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.A.; Cienfuegos-Jovellanos, E.; Marin, A.; Muguerza, B.; Gil-Izquierdo, A.; Cerda, B.; Zafrilla, P.; Morillas, J.; Mulero, J.; Ibarra, A.; et al. A new process to develop a cocoa powder with higher flavonoid monomer content and enhanced bioavailability in healthy humans. J. Agric. Food Chem. 2007, 55, 3926–3935. [Google Scholar] [CrossRef]
- Miller, K.B.; Stuart, D.A.; Smith, N.L.; Lee, C.Y.; McHale, N.L.; Flanagan, J.A.; Ou, B.; Hurst, W.J. Antioxidant activity and polyphenol and procyanidin contents of selected commercially available cocoa-containing and chocolate products in the United States. J. Agric. Food Chem. 2006, 54, 4062–4068. [Google Scholar] [CrossRef]
- Medina, E.; de Castro, A.; Romero, C.; Brenes, M. Comparison of the concentrations of phenolic compounds in olive oils and other plant oils: Correlation with antimicrobial activity. J. Agric. Food Chem. 2006, 54, 4954–4961. [Google Scholar] [CrossRef]
- Carrasco-Pancorbo, A.; Gomez-Caravaca, A.M.; Cerretani, L.; Bendini, A.; Segura-Carretero, A.; Fernandez-Gutierrez, A. Rapid quantification of the phenolic fraction of Spanish virgin olive oils by capillary electrophoresis with UV detection. J. Agric. Food Chem. 2006, 54, 7984–7991. [Google Scholar] [CrossRef]
- Uysal, U.D.; Aturki, Z.; Raggi, M.A.; Fanali, S. Separation of catechins and methylxanthines in tea samples by capillary electrochromatography. J. Sep. Sci. 2009, 32, 1002–1010. [Google Scholar] [CrossRef]
- Khokhar, S.; Magnusdottir, S.G. Total phenol, catechin, and caffeine contents of teas commonly consumed in the United Kingdom. J. Agric. Food Chem. 2002, 50, 565–570. [Google Scholar] [CrossRef]
- Guo, J.; Yue, T.; Yuan, Y.; Wang, Y. Chemometric classification of apple juices according to variety and geographical origin based on polyphenolic profiles. J. Agric. Food Chem. 2013, 61, 6949–6963. [Google Scholar] [CrossRef]
- Belajová, E.; Suhaj, M. Determination of phenolic constituents in citrus juices: Method of high performance liquid chromatography. Food Chem. 2004, 86, 339–343. [Google Scholar] [CrossRef]
- Jandera, P.; Skeifikova, V.; Rehova, L.; Hajek, T.; Baldrianova, L.; Skopova, G.; Kellner, V.; Horna, A. RP-HPLC analysis of phenolic compounds and flavonoids in beverages and plant extracts using a CoulArray detector. J. Sep. Sci. 2005, 28, 1005–1022. [Google Scholar] [CrossRef]
- La Torre, G.L.; Saitta, M.; Vilasi, F.; Pellicanò, T.; Dugo, G. Direct determination of phenolic compounds in Sicilian wines by liquid chromatography with PDA and MS detection. Food Chem. 2006, 94, 640–650. [Google Scholar] [CrossRef]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef]
- Sesink, A.L.; O’Leary, K.A.; Hollman, P.C. Quercetin glucuronides but not glucosides are present in human plasma after consumption of quercetin-3-glucoside or quercetin-4’-glucoside. J. Nutr. 2001, 131, 1938–1941. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Vauzour, D.; Krueger, C.G.; Shanmuganayagam, D.; Reed, J.; Calani, L.; Mena, P.; del Rio, D.; Crozier, A. Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: An update. Arch. Toxicol. 2014, 88, 1803–1853. [Google Scholar] [CrossRef]
- Day, A.J.; Gee, J.M.; DuPont, M.S.; Johnson, I.T.; Williamson, G. Absorption of quercetin-3-glucoside and quercetin-4′-glucoside in the rat small intestine: The role of lactase phlorizin hydrolase and the sodium-dependent glucose transporter. Biochem. Pharmacol. 2003, 65, 1199–1206. [Google Scholar] [CrossRef]
- Walgren, R.A.; Lin, J.T.; Kinne, R.K.; Walle, T. Cellular uptake of dietary flavonoid quercetin 4’-beta-glucoside by sodium-dependent glucose transporter SGLT1. J. Pharmacol. Exp. Ther. 2000, 294, 837–843. [Google Scholar]
- Erlund, I.; Kosonen, T.; Alfthan, G.; Maenpaa, J.; Perttunen, K.; Kenraali, J.; Parantainen, J.; Aro, A. Pharmacokinetics of quercetin from quercetin aglycone and rutin in healthy volunteers. Eur. J. Clin. Pharmacol. 2000, 56, 545–553. [Google Scholar] [CrossRef]
- Mullen, W.; Edwards, C.A.; Crozier, A. Absorption, excretion and metabolite profiling of methyl-, glucuronyl-, glucosyl- and sulpho-conjugates of quercetin in human plasma and urine after ingestion of onions. Br. J. Nutr. 2006, 96, 107–116. [Google Scholar] [CrossRef]
- Clarke, D.B.; Lloyd, A.S.; Botting, N.P.; Oldfield, M.F.; Needs, P.W.; Wiseman, H. Measurement of intact sulfate and glucuronide phytoestrogen conjugates in human urine using isotope dilution liquid chromatography-tandem mass spectrometry with [13C(3)]isoflavone internal standards. Anal. Biochem. 2002, 309, 158–172. [Google Scholar] [CrossRef]
- Nakamura, T.; Murota, K.; Kumamoto, S.; Misumi, K.; Bando, N.; Ikushiro, S.; Takahashi, N.; Sekido, K.; Kato, Y.; Terao, J. Plasma metabolites of dietary flavonoids after combination meal consumption with onion and tofu in humans. Mol. Nutr. Food Res. 2014, 58, 310–317. [Google Scholar] [CrossRef]
- Arts, I.C.; Sesink, A.L.; Faassen-Peters, M.; Hollman, P.C. The type of sugar moiety is a major determinant of the small intestinal uptake and subsequent biliary excretion of dietary quercetin glycosides. Br. J. Nutr. 2004, 91, 841–847. [Google Scholar] [CrossRef]
- Williamson, G.; Kay, C.D.; Crozier, A. The bioavailability, transport, and bioactivity of dietary flavonoids: A review from a historical perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef]
- Jakobek, L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef]
- Gonzales, G.B.; Smagghe, G.; Grootaert, C.; Zotti, M.; Raes, K.; van Camp, J. Flavonoid interactions during digestion, absorption, distribution and metabolism: A sequential structure-activity/property relationship-based approach in the study of bioavailability and bioactivity. Drug Metab. Rev. 2015, 47, 175–190. [Google Scholar] [CrossRef]
- Hollman, P.C.H. Absorption, bioavailability, and metabolism of flavonoids. Pharm. Biol. 2004, 42, 74–83. [Google Scholar] [CrossRef]
- Swieca, M.; Gawlik-Dziki, U.; Dziki, D.; Baraniak, B.; Czyz, J. The influence of protein-flavonoid interactions on protein digestibility in vitro and the antioxidant quality of breads enriched with onion skin. Food Chem. 2013, 141, 451–458. [Google Scholar] [CrossRef]
- Graefe, E.U.; Wittig, J.; Mueller, S.; Riethling, A.K.; Uehleke, B.; Drewelow, B.; Pforte, H.; Jacobasch, G.; Derendorf, H.; Veit, M. Pharmacokinetics and bioavailability of quercetin glycosides in humans. J. Clin. Pharmacol. 2001, 41, 492–499. [Google Scholar] [CrossRef]
- Donovan, J.L.; Bell, J.R.; Kasim-Karakas, S.; German, J.B.; Walzem, R.L.; Hansen, R.J.; Waterhouse, A.L. Catechin is present as metabolites in human plasma after consumption of red wine. J. Nutr. 1999, 129, 1662–1668. [Google Scholar] [CrossRef]
- Van het Hof, K.H.; Wiseman, S.A.; Yang, C.S.; Tijburg, L.B. Plasma and lipoprotein levels of tea catechins following repeated tea consumption. Proc. Soc. Exp. Biol. Med. 1999, 220, 203–209. [Google Scholar]
- Holt, R.R.; Lazarus, S.A.; Sullards, M.C.; Zhu, Q.Y.; Schramm, D.D.; Hammerstone, J.F.; Fraga, C.G.; Schmitz, H.H.; Keen, C.L. Procyanidin dimer B2 [epicatechin-(4beta-8)-epicatechin] in human plasma after the consumption of a flavanol-rich cocoa. Am. J. Clin. Nutr. 2002, 76, 798–804. [Google Scholar] [CrossRef]
- Erlund, I.; Meririnne, E.; Alfthan, G.; Aro, A. Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice. J. Nutr. 2001, 131, 235–241. [Google Scholar] [CrossRef]
- Manach, C.; Morand, C.; Gil-Izquierdo, A.; Bouteloup-Demange, C.; Remesy, C. Bioavailability in humans of the flavanones hesperidin and narirutin after the ingestion of two doses of orange juice. Eur. J. Clin. Nutr. 2003, 57, 235–242. [Google Scholar] [CrossRef]
- Nielsen, I.L.; Dragsted, L.O.; Ravn-Haren, G.; Freese, R.; Rasmussen, S.E. Absorption and excretion of black currant anthocyanins in humans and watanabe heritable hyperlipidemic rabbits. J. Agric. Food Chem. 2003, 51, 2813–2820. [Google Scholar] [CrossRef]
- Haytowitz, D.B.; Bhagwat, S.; Holden, J.M. Sources of variability in the flavonoid content of foods. Procedia Food Sci. 2013, 2, 46–51. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y.; Cao, C.; Cao, J.; Chen, W.; Zhang, Y.; Wang, C.; Wang, J.; Zhang, X.; Zhao, X. Dietary flavonol and flavone intakes and their major food sources in Chinese adults. Nutr. Cancer 2010, 62, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Wang, H.; Wang, D.; Chen, Y.; Zhao, Y.; Xia, W. Using an FFQ to assess intakes of dietary flavonols and flavones among female adolescents in the Suihua area of northern China. Public Health Nutr. 2015, 18, 632–639. [Google Scholar] [CrossRef]
- Kim, Y.J.; Park, M.Y.; Chang, N.; Kwon, O. Intake and major sources of dietary flavonoid in Korean adults: Korean National Health and Nutrition Examination Survey 2010–2012. Asia Pac. J. Clin. Nutr. 2015, 24, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Jun, S.; Shin, S.; Joung, H. Estimation of dietary flavonoid intake and major food sources of Korean adults. Br. J. Nutr. 2016, 115, 480–489. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Agudo, A.; Lujan-Barroso, L.; Romieu, I.; Ferrari, P.; Knaze, V.; Bueno-de-Mesquita, H.B.; Leenders, M.; Travis, R.C.; Navarro, C.; et al. Dietary flavonoid and lignan intake and gastric adenocarcinoma risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Am. J. Clin. Nutr. 2012, 96, 1398–1408. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Andres-Lacueva, C.; Lamuela-Raventos, R.M.; Berenguer, T.; Jakszyn, P.; Barricarte, A.; Ardanaz, E.; Amiano, P.; Dorronsoro, M.; Larranaga, N.; et al. Estimation of dietary sources and flavonoid intake in a Spanish adult population (EPIC-Spain). J. Am. Diet. Assoc. 2010, 110, 390–398. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Knaze, V.; Lujan-Barroso, L.; Slimani, N.; Romieu, I.; Touillaud, M.; Kaaks, R.; Teucher, B.; Mattiello, A.; Grioni, S.; et al. Estimation of the intake of anthocyanidins and their food sources in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Br. J. Nutr. 2011, 106, 1090–1099. [Google Scholar] [CrossRef]
- Knaze, V.; Zamora-Ros, R.; Lujan-Barroso, L.; Romieu, I.; Scalbert, A.; Slimani, N.; Riboli, E.; van Rossum, C.T.; Bueno-de-Mesquita, H.B.; Trichopoulou, A.; et al. Intake estimation of total and individual flavan-3-ols, proanthocyanidins and theaflavins, their food sources and determinants in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Br. J. Nutr. 2012, 108, 1095–1108. [Google Scholar] [CrossRef]
- Vogiatzoglou, A.; Mulligan, A.A.; Lentjes, M.A.; Luben, R.N.; Spencer, J.P.; Schroeter, H.; Khaw, K.T.; Kuhnle, G.G. Flavonoid intake in European adults (18 to 64 years). PLoS ONE 2015, 10, e0128132. [Google Scholar] [CrossRef] [PubMed]
- Johannot, L.; Somerset, S.M. Age-related variations in flavonoid intake and sources in the Australian population. Public Health Nutr. 2006, 9, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Somerset, S.M.; Johannot, L. Dietary flavonoid sources in Australian adults. Nutr. Cancer 2008, 60, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.J.; Walker, K.M.; Dyer, K.A.; Bryan, J. Estimation of daily intake of flavonoids and major food sources in middle-aged Australian men and women. Nutr. Res. 2019, 61, 64–81. [Google Scholar] [CrossRef] [PubMed]
- Grotto, D.; Zied, E. The Standard American Diet and its relationship to the health status of Americans. Nutr. Clin. Pract. 2010, 25, 603–612. [Google Scholar] [CrossRef]
- Bertoia, M.L.; Rimm, E.B.; Mukamal, K.J.; Hu, F.B.; Willett, W.C.; Cassidy, A. Dietary flavonoid intake and weight maintenance: Three prospective cohorts of 124,086 US men and women followed for up to 24 years. BMJ 2016, 352, i17. [Google Scholar] [CrossRef]
- Chun, O.K.; Song, W.O.; Chung, S.J. Estimated dietary flavonoid intake and major food sources of U.S. adults. J. Nutr. 2007, 137, 1244–1252. [Google Scholar] [CrossRef]
- Kim, K.; Vance, T.M.; Chun, O.K. Estimated intake and major food sources of flavonoids among US adults: Changes between 1999–2002 and 2007–2010 in NHANES. Eur. J. Nutr. 2016, 55, 833–843. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Biessy, C.; Rothwell, J.A.; Monge, A.; Lajous, M.; Scalbert, A.; Lopez-Ridaura, R.; Romieu, I. Dietary polyphenol intake and their major food sources in the Mexican Teachers’ Cohort. Br. J. Nutr. 2018, 120, 353–360. [Google Scholar] [CrossRef]
- Saha, S.K.; Lee, S.B.; Won, J.; Choi, H.Y.; Kim, K.; Yang, G.-M.; Dayem, A.A.; Cho, S.-G. Correlation between oxidative stress, nutrition, and cancer initiation. Int. J. Mol. Sci. 2017, 18, 1544. [Google Scholar] [CrossRef]
- Kim, M.; Jung, J. The natural plant flavonoid apigenin is a strong antioxidant that effectively delays peripheral neurodegenerative processes. Anat. Sci. Int. 2019. [Google Scholar] [CrossRef]
- Hwang, I.W.; Chung, S.K. Isolation and identification of myricitrin, an antioxidant flavonoid, from daebong persimmon peel. Prev. Nutr. Food Sci. 2018, 23, 341–346. [Google Scholar] [CrossRef]
- Jayaraman, R.; Subramani, S.; Abdullah, S.H.S.; Udaiyar, M. Antihyperglycemic effect of hesperetin, a citrus flavonoid, extenuates hyperglycemia and exploring the potential role in antioxidant and antihyperlipidemic in streptozotocin-induced diabetic rats. Biomed. Pharmacother. 2018, 97, 98–106. [Google Scholar] [CrossRef]
- Aloud, A.A.; Veeramani, C.; Govindasamy, C.; Alsaif, M.A.; el Newehy, A.S.; Al-Numair, K.S. Galangin, a dietary flavonoid, improves antioxidant status and reduces hyperglycemia-mediated oxidative stress in streptozotocin-induced diabetic rats. Redox. Rep. 2017, 22, 290–300. [Google Scholar] [CrossRef]
- Kanimozhi, S.; Bhavani, P.; Subramanian, P. Influence of the flavonoid, quercetin on antioxidant status, lipid peroxidation and histopathological changes in hyperammonemic Rats. Indian J. Clin. Biochem. 2017, 32, 275–284. [Google Scholar] [CrossRef]
- Abarikwu, S.O.; Olufemi, P.D.; Lawrence, C.J.; Wekere, F.C.; Ochulor, A.C.; Barikuma, A.M. Rutin, an antioxidant flavonoid, induces glutathione and glutathione peroxidase activities to protect against ethanol effects in cadmium-induced oxidative stress in the testis of adult rats. Andrologia 2017, 49. [Google Scholar] [CrossRef]
- Torres Castaneda, G.H.; Dulcey, A.J.C.; Martinez, J.H.I. Flavonoid glycosides from Siparuna gigantotepala leaves and their antioxidant activity. Chem. Pharm. Bull. 2016, 64, 502–506. [Google Scholar] [CrossRef][Green Version]
- Alipour, B.; Rashidkhani, B.; Edalati, S. Dietary flavonoid intake, total antioxidant capacity and lipid oxidative damage: A cross-sectional study of Iranian women. Nutrition 2016, 32, 566–572. [Google Scholar] [CrossRef]
- Olayinka, E.T.; Ore, A.; Adeyemo, O.A.; Ola, O.S.; Olotu, O.O.; Echebiri, R.C. Quercetin, a flavonoid antioxidant, ameliorated procarbazine-induced oxidative damage to murine tissues. Antioxidants 2015, 4, 304–321. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Ferrari, P.; Gonzalez, C.A.; Tjonneland, A.; Olsen, A.; Bredsdorff, L.; Overvad, K.; Touillaud, M.; Perquier, F.; Fagherazzi, G.; et al. Dietary flavonoid and lignan intake and breast cancer risk according to menopause and hormone receptor status in the European Prospective Investigation into Cancer and Nutrition (EPIC) Study. Breast Cancer Res. Treat. 2013, 139, 163–176. [Google Scholar] [CrossRef]
- Boggs, D.A.; Palmer, J.R.; Stampfer, M.J.; Spiegelman, D.; Adams-Campbell, L.L.; Rosenberg, L. Tea and coffee intake in relation to risk of breast cancer in the Black Women’s Health Study. Cancer Causes Control 2010, 21, 1941–1948. [Google Scholar] [CrossRef]
- Oh, J.K.; Sandin, S.; Strom, P.; Lof, M.; Adami, H.O.; Weiderpass, E. Prospective study of breast cancer in relation to coffee, tea and caffeine in Sweden. Int. J. Cancer 2015, 137, 1979–1989. [Google Scholar] [CrossRef]
- Luo, J.; Gao, Y.T.; Chow, W.H.; Shu, X.O.; Li, H.; Yang, G.; Cai, Q.; Rothman, N.; Cai, H.; Shrubsole, M.J.; et al. Urinary polyphenols and breast cancer risk: Results from the Shanghai Women’s Health Study. Breast Cancer Res. Treat. 2010, 120, 693–702. [Google Scholar] [CrossRef]
- Wang, Q.; Li, H.; Tao, P.; Wang, Y.P.; Yuan, P.; Yang, C.X.; Li, J.Y.; Yang, F.; Lee, H.; Huang, Y. Soy isoflavones, CYP1A1, CYP1B1, and COMT polymorphisms, and breast cancer: A case-control study in southwestern China. DNA Cell Biol. 2011, 30, 585–595. [Google Scholar] [CrossRef]
- Hui, C.; Qi, X.; Qianyong, Z.; Xiaoli, P.; Jundong, Z.; Mantian, M. Flavonoids, flavonoid subclasses and breast cancer risk: A meta-analysis of epidemiologic studies. PLoS ONE 2013, 8, e54318. [Google Scholar] [CrossRef]
- Gardeazabal, I.; Romanos-Nanclares, A.; Martinez-Gonzalez, M.A.; Sanchez-Bayona, R.; Vitelli-Storelli, F.; Gaforio, J.J.; Aramendia-Beitia, J.M.; Toledo, E. Total polyphenol intake and breast cancer risk in the SUN cohort. Br. J. Nutr. 2018, 1–23. [Google Scholar] [CrossRef]
- Baeza, I.; de la Fuente, M. The Role of Polyphenols in Menopause. In Nutrition and Diet in Menopause; Martin, C.J.H., Watson, R.R., Preedy, V.R., Eds.; Humana Press: Totowa, NJ, USA, 2013; pp. 51–63. [Google Scholar] [CrossRef]
- Christensen, K.Y.; Naidu, A.; Parent, M.E.; Pintos, J.; Abrahamowicz, M.; Siemiatycki, J.; Koushik, A. The risk of lung cancer related to dietary intake of flavonoids. Nutr. Cancer 2012, 64, 964–974. [Google Scholar] [CrossRef]
- Reale, G.; Russo, G.I.; di Mauro, M.; Regis, F.; Campisi, D.; Giudice, A.L.; Marranzano, M.; Ragusa, R.; Castelli, T.; Cimino, S.; et al. Association between dietary flavonoids intake and prostate cancer risk: A case-control study in Sicily. Complement. Ther. Med. 2018, 39, 14–18. [Google Scholar] [CrossRef]
- Woo, H.D.; Lee, J.; Choi, I.J.; Kim, C.G.; Lee, J.Y.; Kwon, O.; Kim, J. Dietary flavonoids and gastric cancer risk in a Korean population. Nutrients 2014, 6, 4961–4973. [Google Scholar] [CrossRef]
- Sun, L.; Subar, A.F.; Bosire, C.; Dawsey, S.M.; Kahle, L.L.; Zimmerman, T.P.; Abnet, C.C.; Heller, R.; Graubard, B.I.; Cook, M.B.; et al. Dietary flavonoid intake reduces the risk of head and neck but not esophageal or gastric cancer in US men and women. J. Nutr. 2017, 147, 1729–1738. [Google Scholar] [CrossRef]
- Molina-Montes, E.; Sanchez, M.J.; Zamora-Ros, R.; Bueno-de-Mesquita, H.B.; Wark, P.A.; Obon-Santacana, M.; Kuhn, T.; Katzke, V.; Travis, R.C.; Ye, W.; et al. Flavonoid and lignan intake and pancreatic cancer risk in the European prospective investigation into cancer and nutrition cohort. Int. J. Cancer 2016, 139, 1480–1492. [Google Scholar] [CrossRef]
- Nimptsch, K.; Zhang, X.; Cassidy, A.; Song, M.; O’Reilly, E.J.; Lin, J.H.; Pischon, T.; Rimm, E.B.; Willett, W.C.; Fuchs, C.S.; et al. Habitual intake of flavonoid subclasses and risk of colorectal cancer in 2 large prospective cohorts. Am. J. Clin. Nutr. 2016, 103, 184–191. [Google Scholar] [CrossRef]
- Xu, M.; Chen, Y.M.; Huang, J.; Fang, Y.J.; Huang, W.Q.; Yan, B.; Lu, M.S.; Pan, Z.Z.; Zhang, C.X. Flavonoid intake from vegetables and fruits is inversely associated with colorectal cancer risk: A case-control study in China. Br. J. Nutr. 2016, 116, 1275–1287. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Not, C.; Guino, E.; Lujan-Barroso, L.; Garcia, R.M.; Biondo, S.; Salazar, R.; Moreno, V. Association between habitual dietary flavonoid and lignan intake and colorectal cancer in a Spanish case-control study (the Bellvitge Colorectal Cancer Study). Cancer Causes Control 2013, 24, 549–557. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Guinó, E.; Alonso, M.H.; Vidal, C.; Barenys, M.; Soriano, A.; Moreno, V. Dietary flavonoids, lignans and colorectal cancer prognosis. Sci. Rep. 2015, 5, 14148. [Google Scholar] [CrossRef]
| Fruits | ANT | DYC | FVA | FVO | Total |
|---|---|---|---|---|---|
| Berries | |||||
| Aestivalis grape | 79.74 | - | - | 1.7 | 81.44 |
| American cranberry | 49.89 | - | - | 43.84 | 93.73 |
| Black chokeberry | 878.11 | - | - | 134.87 | 1012.98 |
| Black elderberry | 1316.66 | - | - | 42 | 1358.66 |
| Black raspberry | - | - | - | 19 | 19 |
| Blackberry | 172.59 | - | 13.87 | 16.87 | 203.33 |
| Blackcurrant | 593.58 | - | 1.17 | 13.68 | 608.43 |
| Black grape | 72.15 | - | 14.03 | 4.01 | 90.19 |
| Green grape | - | - | 3.78 | 2.49 | 6.27 |
| Green currant | - | - | - | 11.07 | 11.07 |
| Highbush blueberry | 156.6 | - | 1.11 | 54.77 | 212.48 |
| Lingonberry | 60.21 | - | - | 48.98 | 109.19 |
| Lowbush blueberry | 204.56 | - | - | - | 204.56 |
| Red raspberry | 72.47 | - | 5.73 | 16.26 | 94.46 |
| Redcurrant | 33.13 | - | 4.68 | 0.77 | 38.58 |
| Strawberry | 26.87 | - | 9.1075 | 2.32 | 38.29 |
| Drupes | |||||
| Nectarine | 0.86 | - | 17.65 | 1.35 | 19.86 |
| Peach | 0.28 | - | 45.18 | 1.42 | 46.88 |
| Plum | 47.79 | - | 46.9 | 6.98 | 101.67 |
| Sour cherry | 54.43 | - | 0.2 | - | 54.63 |
| Sweet cherry | 170.18 | - | 14.87 | - | 185.05 |
| Pomes | |||||
| Apple | 0.93 | 5.38 | 39.42 | 10.62 | 56.35 |
| Pear | - | - | 4.98 | 0.84 | 5.82 |
| Quince | - | - | 7.49 | 0.67 | 8.16 |
| Tropical Fruits | |||||
| Banana | - | - | 1.55 | - | 1.55 |
| Kiwi | - | - | 0.7 | - | 0.7 |
| Mango | - | - | 1.72 | - | 1.72 |
| Persimmon | - | - | 1.28 | - | 1.28 |
| Pomegranate | - | - | 1.1 | - | 1.1 |
| Vegetables | ANT | CHA | FVA | FNE | FVE | FVO | Total |
|---|---|---|---|---|---|---|---|
| Cabbages | |||||||
| Broccoli | - | - | - | - | - | 27.8 | 27.8 |
| Fruit Vegetables | |||||||
| Avocado | - | - | 0.55 | - | - | - | 0.55 |
| Black olive | 82.97 | - | - | - | 27.43 | 49.43 | 159.83 |
| Green olive | - | - | - | - | 0.56 | - | 0.56 |
| Green sweet pepper | - | - | - | - | 2.11 | 5.49 | 7.6 |
| Red sweet pepper | - | - | - | - | 0.05 | 0.24 | 0.29 |
| Tomato | - | - | - | 0.14 | - | 0.014 | 0.15 |
| Leaf Vegetables | |||||||
| Curly | - | - | - | - | - | 24.06 | 24.06 |
| Escarole | - | - | - | - | - | 18.23 | 18.23 |
| Green lettuce | - | - | - | - | 0.4 | 3.99 | 4.39 |
| Red lettuce | 3.53 | - | - | 2.51 | 16.74 | 22.78 | |
| Spinach | - | - | - | - | - | 119.27 | 119.27 |
| Onion-Family Vegetables | |||||||
| Red onion | 9 | - | - | - | - | 122.51 | 131.51 |
| White onion | - | - | - | - | - | 5.4 | 5.4 |
| Yellow onion | - | - | - | - | - | 59.1 | 59.1 |
| Shallot | - | - | - | - | - | 112.22 | 112.22 |
| Pod Vegetables | |||||||
| Broad bean pod | - | 0.08 | 154.45 | - | 0.37 | 34.64 | 189.54 |
| Green bean | - | - | 2.42 | - | - | 5.55 | 7.97 |
| Shoot Vegetables | |||||||
| Asparagus | - | - | - | - | - | 23.19 | 23.19 |
| Globe artichoke, heads | - | - | - | - | 57.8 | - | 57.8 |
| Seeds | ANT | FVA | FNE | FVE | FVO | IFA | Total |
|---|---|---|---|---|---|---|---|
| Nuts | |||||||
| Almond | - | 4.93 | 0.5 | - | 3.81 | 0.06 | 9.3 |
| Cashew nut | - | 1.1 | - | - | - | - | 1.1 |
| Chestnut | - | 0.05 | - | - | - | - | 0.05 |
| Hazelnut | - | 5.7 | - | - | - | - | 5.7 |
| Peanut | - | - | - | - | - | 0.51 | 0.51 |
| Pecan nut | - | 16.7 | - | - | - | - | 16.7 |
| Pistachio | 6.9 | 0.12 | 0.103 | 0.07 | 7.193 | ||
| Common Bean | |||||||
| Black common bean | 41.05 | - | - | - | 10 | 1.4 | 52.45 |
| Others common bean | 7.42 | - | - | - | 69.58 | 0.2 | 77.2 |
| White common bean | 0.13 | - | - | - | 49.96 | 0.5 | 50.59 |
| Other Beans | |||||||
| Broad bean seed whole | - | 49.37 | - | - | - | - | 49.37 |
| Sunflower seed meal | - | - | - | - | - | 0.02 | 0.02 |
| LENTILS | |||||||
| Lentils | - | 5.17 | 0.95 | 1.09 | - | 7.21 | |
| Soy Products | |||||||
| Soy paste miso | - | - | - | - | - | 63.09 | 63.09 |
| Soy tempeh | - | - | - | - | - | 147.74 | 147.74 |
| Soy tofu | - | - | - | - | - | 39.24 | 39.24 |
| Soybean roasted | - | - | - | - | - | 253.11 | 253.11 |
| Cereals | FVA | FVE | FVO | Total |
|---|---|---|---|---|
| Cereals | ||||
| Barley, whole grain flour | 35.2 | - | - | 35.2 |
| Buckwheat groats, thermally treated | - | - | 8.96 | 8.96 |
| Buckwheat, refined flour | - | - | 5.86 | 5.86 |
| Buckwheat, whole grain flour | - | 0.9 | 36.14 | 37.04 |
| Common wheat, refined flour | - | 18.4 | 0.08 | 18.48 |
| Common wheat, whole grain flour | - | 77.29 | 0.11 | 77.4 |
| Cocoa | FVA | FVO | Total |
|---|---|---|---|
| Cocoa | |||
| Chocolate dark | 212.36 | 25 | 237.36 |
| Chocolate milk | 19.22 | - | 19.22 |
| Cocoa powder | 511.62 | - | 511.62 |
| Oils | FVE | Total |
|---|---|---|
| Oils | ||
| Extra virgin olive oil | 1.53 | 1.53 |
| Virgin olive oil | 0.23 | 0.23 |
| Refined olive oil | 0.15 | 0.15 |
| Non-Alcoholic Beverages | ANT | DYC | FVA | FNE | FVE | FVO | IFA | Total |
|---|---|---|---|---|---|---|---|---|
| Cocoa Beverage | ||||||||
| Chocolate, milk | - | - | 20.33 | - | - | - | - | 20.33 |
| Fruit Juices | ||||||||
| Berry Juices | ||||||||
| Fox grape juice | - | - | 5.9 | - | - | - | - | 5.9 |
| Green grape juice | - | - | 3.88 | - | - | - | - | 3.88 |
| Grapefruit juice | - | - | - | 46.44 | - | 0.68 | - | 47.12 |
| Citrus Juices | ||||||||
| Lemon juice | - | - | - | 32.66 | 4.77 | - | - | 37.43 |
| Lime juice | - | - | - | 19.61 | - | - | - | 19.61 |
| Orange juice | 3.17 | - | - | 37.63 | 6.14 | 1.08 | - | 48.02 |
| Pummelo juice | - | - | - | 8.48 | - | - | - | 8.48 |
| Red raspberry juice | - | - | - | - | - | 9.58 | - | 9.58 |
| Rowanberry | - | - | - | - | - | 7.04 | - | 7.04 |
| Drupe Juices | ||||||||
| Plum juice | - | 5.85 | 24.7 | - | - | - | - | 30.55 |
| Pome Juices | ||||||||
| Apple juice | - | 4.39 | 48.45 | - | - | 2.15 | - | 54.99 |
| Apple (cider) juice | - | 4.78 | 22.66 | - | - | - | - | 27.44 |
| Pear juice | - | - | 3.24 | - | - | - | - | 3.24 |
| Tropical Juices | ||||||||
| Kiwi juice | - | - | 0.38 | - | - | 0.09 | - | 0.47 |
| Pomegranate juice | 10.13 | 0.1 | - | - | - | 0.25 | - | 10.48 |
| Herb Infusions | ||||||||
| German chamomile, tea | - | - | 2.07 | - | - | - | - | 2.07 |
| Lemon verbena | - | - | 10.6 | - | - | - | - | 10.6 |
| Peppermint, tea | - | - | 10.23 | - | - | - | - | 10.23 |
| Tea Infusion | ||||||||
| Fennel tea | - | - | - | - | - | 3.26 | - | 3.26 |
| Black tea | - | - | 73.29 | - | - | 10.06 | - | 83.35 |
| Green tea | - | - | 71.18 | - | - | 6.26 | - | 77.44 |
| Oolong tea | - | - | 35.72 | - | - | - | - | 35.72 |
| Soy Products | ||||||||
| Soy milk | - | - | - | - | - | - | 18 | 18 |
| Alcoholic Beverages | ANT | DYC | DYF | FVA | FNE | FVE | FVO | IFA | Total |
|---|---|---|---|---|---|---|---|---|---|
| Beer | |||||||||
| Beer (alcohol free) | - | 0.0003 | - | 0.11 | 0.01 | - | - | - | 0.12 |
| Beer (ale) | - | 0.01 | - | 0.38 | 0.24 | - | - | 0.02 | 0.65 |
| Beer (dark) | - | 0.03 | - | 0.03 | 0.15 | - | - | - | 0.21 |
| Beer (regular) | - | 0.001 | - | 0.61 | 0.04 | 0.004 | 0.09 | 0.02 | 0.77 |
| Wines | |||||||||
| Red wine | 23.3 | - | 5.44 | 47.02 | 0.85 | - | 7.35 | - | 83.96 |
| Rosé wine | - | - | 0.38 | 2 | - | - | - | - | 2.38 |
| White wine | 0.04 | - | 0.57 | 2.07 | 0.23 | - | 0.695 | - | 3.61 |
| Country | Intake (mg/d) | Subclass | Food Sources |
|---|---|---|---|
| Asia | 107 | Protanthocyanidins > flavonols > isoflavons > flavan-3-ols > anthocyanidins > flavanones > flavones | Kimchi, green tea, persimmons, soybeans, onions |
| Southern europe | 313 | Proanthocyanidins > flavanones > flavan-3-ols > flavonols > anthocyanidins > flavones > isoflavones | Apples, red wine, oranges, beans, pears, peaches |
| Northern europe | 348 | Flavan-3-ols > flavones > anthocyanidins > flavonols > flavanones > isoflavones | Tea, citrus fruits, berries |
| Central europe | 506 | Flavan-3-ols > anthocyanidins > proanthocyanidins > flavanones > flavonols > flavones > isoflavones | Tea, non-citrus fruits, wine |
| Oceania | 351 | Flavan-3-ols > anthocyanidins > flavonols > flavanones > isoflavones > flavones | Black tea, oranges, grapes, wine, apples |
| North America | 230 | Flavan-3-ols > flavanones > flavonols > anthocyanidins > flavones > isoflavones | Apples, wine, citrus fruit juices and tea. |
| Authors | Methods | Results |
|---|---|---|
| Breast | ||
| Itziar Gardeazabal et al. (2018) [97] | Prospective Cohort Study 10,713 Spanish Women Food Frequency Questionnaire (FFQ) Phenol-Explorer database HPLC | There was not a statistically significant association between total flavonoids and breast cancer risk. However, in postmenopausal women, the data indicate an inverse association between breast risk cancer and total polyphenol intake (HR: 0.31, 95% CI: 0.13–0.77) |
| Oh, J.K et al. (2015) [93] | Prospective Cohort Study 42,099 Swedish Women 30–49 years FFQ | Data showed that compared with no consumption, women who consumed >1 cup tea/day had an increased breast cancer risk (Relative Risk (RR): 1.19, 95% Confidence Interval (CI): 1.00–1.42), but women with a high intake of coffee (3–4 cups/day) had a decreased breast cancer risk (RR: 0.87, 95% CI: 0.76–1.00). |
| Raul Zamora-Ros et al. (2013) [91] | Prospective Cohort Study 334,850 women, 35–70 years 11.5 years follow up FFQ Phenol-Explorer database | There was no statistically significant association between total flavonoid (Hazard Ratio (HR) 0.97, 95% CI: 0.90–1.07) and isoflavone (HR 1, 95%CI: 0.91–1.10) intake and breast cancer risk. |
| Wang et al. (2011) [95] | Case-Control Study. 400 cases and 400 controls. Daily intake of soy isoflavones data Gene sequencing | They suggested a protective role of high soy isoflavone intake against breast cancer risk based on the relation of oestrogen metabolites, breast cancer, and isoflavone metabolism. |
| Boggs et al. (2010) [92] | Prospective Cohort Study 52,062 women, 21–69 years 12 years follow up FFQ | Data showed that the intake of coffee (Internal Rate of Return (IRR): 1.03, 95% CI: 0.77–1.39) or tea (IRR: 1.13, 95% CI: 0.78–1.63) was not associated with risk of breast cancer in participants. |
| Luo JF et al. (2010) [94] | Case-Control Study 353 cases, 701 controls, 40–70 years FFQ Liquid chromatography | There was an inverse association between breast cancer risk and urinary excretion of epicatechin (Odds Ratio (OR) 0.59, 95% CI: 0.39–0.88). |
| Lung | ||
| Christensen KY et al. (2012) [99] | Case-Control Study 1061 cases and 1425 controls FFQ | A low intake of flavonoids was related to an increased risk of lung cancer. OR: 0.63, 95% CI: 0.47–0.85. |
| Cutler et al. (2008) [14] | Prospective Cohort Study USDA database FFQ | There was an inverse association between isoflavone intake and cancer incidence (HR: 0.93, 95% CI: 0.86–1.00) and an inverse association between proanthocyanidin (HR: 0.75, 95% CI: 0.57–0.97) and flavanone (HR: 0.68, 95% CI: 0.53–0.86) intake with lung cancer incidence. |
| Prostate | ||
| Giulio Reale et al. (2018) [100] | Case-Control study 118 cases and 222 controls FFQ Prostate Specific Antigen | High intake of some subclasses of flavonoids (catechin (OR: 0.12, 95% CI: 0.04–0.36) and flavonol (OR: 0.19, 95% CI: 0.06–0.56) significantly reduces the risk of prostate cancer. |
| Authors, Year | Methods | Results |
|---|---|---|
| Gastric Adenocarcinoma | ||
| Sun L et al. (2017) [102] | Prospective cohort study 469,008 participants 12 years follow up FFQ | Data suggested that total flavonoid intake was associated with a reduced risk of neck and head cancer (HR: 0.76, 95% CI: 0.66–0.86). |
| Hae Donw Woo et al. (2014) [101] | Case-Control study. 334 cases and 334 controls, aged 35–75 years, from Korea. FFQ USDA database | Total flavonoids and their subclasses were significantly associated with a reduced risk of gastric cancer in women (OR 0.33, 95% CI 0.15–0.73) but not in men. |
| Zamora-Ros et al. (2012) [68] | Observational study 477,312 subjects, aged 35–70 years, from 10 European countries. Average Follow-up of 11 years FFQ USDA and Phenol Explorer Databases. | Total dietary intake was associated with a significant reduction in the risk of gastric adenocarcinoma in women (HR 0.81, 95% CI 0.70, 0.94). |
| Pancreatic Cancer | ||
| Molina-Montes E. et al. (2016) [103] | Prospective cohort 477,309 participants Average Follow-up of 11 years FFQ USDA and Phenol Explorer Databases. | There was no association with pancreatic cancer risk and dietary flavonoid intake (HR: 1.09, 95% CI: 0.95–1.11); however, there was an inverse association, but not statistically significant, between prostate cancer risk and flavanone intake. |
| Colorectal Cancer | ||
| Nimptsch K et al. (2016) [104] | Prospective cohort 42,478 male 76,364 female 26 years follow up FFQ | Data did not show an association between colorectal cancer risk and flavonoid subclass intake. RR (95% IC) values were 0.98 (0.81, 1.19) for anthocyanins, 1.07 (0.95, 1.21) for flavan-3-ols, 0.96 (0.84, 1.10) for flavanones, 1.01 (0.89, 1.15) for flavones, and 1.04 (0.91, 1.18) for flavonols. |
| Xu M. et al. (2016) [105] | Case-Control study 1632 cases and 1632 controls FFQ | There was an inverse association between colorectal cancer risk and flavone (OR: 0.54, 95% CI 0.43, 0·67), flavanone (OR: 0.28, 95% CI: 0.22–0.36), and anthocyanidin (OR: 0.80, 95% CI: 0.64–1.00) intake. |
| Zamora Ros et al. (2015) [107] | Case-control study 523 participants FFQ | Flavonoid intake was not associated with colorectal cancer survival or recurrence. |
| Zamora Ros et al. (2013) [108] | Case-Control study 424 cases and 401 hospital-based controls FFQ Phenol Explorer Database | Data showed an inverse correlation between risk of colorectal cancer and intake of total flavonoids (OR: 0.59, 95% CI, 0.35–0.99) and some flavonoid subclasses (such as proanthocyanidins and flavones) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-García, C.; Sánchez-Quesada, C.; Gaforio, J.J. Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies. Antioxidants 2019, 8, 137. https://doi.org/10.3390/antiox8050137
Rodríguez-García C, Sánchez-Quesada C, Gaforio JJ. Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies. Antioxidants. 2019; 8(5):137. https://doi.org/10.3390/antiox8050137
Chicago/Turabian StyleRodríguez-García, Carmen, Cristina Sánchez-Quesada, and José J. Gaforio. 2019. "Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies" Antioxidants 8, no. 5: 137. https://doi.org/10.3390/antiox8050137
APA StyleRodríguez-García, C., Sánchez-Quesada, C., & Gaforio, J. J. (2019). Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies. Antioxidants, 8(5), 137. https://doi.org/10.3390/antiox8050137







