Mitochondria-Targeted Antioxidants for Treatment of Hearing Loss: A Systematic Review
Abstract
1. Introduction
2. Effect of Mitochondrial Dysfunction on Disease
3. Overview of Hearing Loss Associated with Mitochondrial Dysfunction
3.1. Hearing Loss Due to Mitochondrial Gene Mutation
3.2. ARHL
3.3. NIHL
3.4. Ototoxic Hearing Loss
4. Mitochondria-Targeted Antioxidants for Treatment of Hearing Loss
4.1. Treatment Effect of Antioxidants on Hearing Loss
4.2. Mitochondria-Targeted Antioxidants for Treatment of Hearing Loss
Author Contributions
Funding
Conflicts of Interest
References
- Balaban, R.S.; Nemoto, S.; Finkel, T. Mitochondria, oxidants and aging. Cell 2005, 120, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef]
- Bottger, E.C.; Schacht, J. The mitochondrion: A perpetrator of acquired hearing loss. Hear. Res. 2013, 303, 12–19. [Google Scholar] [CrossRef]
- Wong, A.C.; Ryan, A.F. Mechanisms of sensorineural cell damage, death and survival in the cochlea. Front. Aging Neurosci. 2015, 7, 58. [Google Scholar] [CrossRef] [PubMed]
- Bonawitz, N.D.; Rodeheffer, M.S.; Shadel, G.S. Defective mitochondrial gene expression results in reactive oxygen species-mediated inhibition of respiration and reduction of yeast life span. Mol. Cell. Biol. 2006, 26, 4818–4829. [Google Scholar] [CrossRef]
- Dhanasekaran, A.; Kotamraju, S.; Kalivendi, S.V.; Matsunaga, T.; Shang, T.; Keszler, A.; Joseph, J.; Kalyanaraman, B. Supplementation of endothelial cells with mitochondria-targeted antioxidants inhibit peroxide-induced mitochondrial iron uptake, oxidative damage and apoptosis. J. Biol. Chem. 2004, 279, 37575–37587. [Google Scholar] [CrossRef]
- Jauslin, M.L.; Meier, T.; Smith, R.A.; Murphy, M.P. Mitochondria-targeted antioxidants protect Friedreich Ataxia fibroblasts from endogenous oxidative stress more effectively than untargeted antioxidants. Faseb. J. 2003, 17, 1972–1974. [Google Scholar] [CrossRef]
- Kelso, G.F.; Porteous, C.M.; Coulter, C.V.; Hughes, G.; Porteous, W.K.; Ledgerwood, E.C.; Smith, R.A.; Murphy, M.P. Selective targeting of a redox-active ubiquinone to mitochondria within cells: Antioxidant and antiapoptotic properties. J. Biol. Chem. 2001, 276, 4588–4596. [Google Scholar] [CrossRef]
- Dirain, C.O.; Ng, M.; Milne-Davies, B.; Joseph, J.K.; Antonelli, P.J. Evaluation of Mitoquinone for Protecting Against Amikacin-Induced Ototoxicity in Guinea Pigs. Otol. Neurotol. 2018, 39, 111–118. [Google Scholar] [CrossRef]
- Jadidian, A.; Antonelli, P.J.; Ojano-Dirain, C.P. Evaluation of apoptotic markers in HEI-OC1 cells treated with gentamicin with and without the mitochondria-targeted antioxidant mitoquinone. Otol. Neurotol. 2015, 36, 526–530. [Google Scholar] [CrossRef]
- Jankauskas, S.S.; Plotnikov, E.Y.; Morosanova, M.A.; Pevzner, I.B.; Zorova, L.D.; Skulachev, V.P.; Zorov, D.B. Mitochondria-targeted antioxidant SkQR1 ameliorates gentamycin-induced renal failure and hearing loss. Biochemistry (Mosc.) 2012, 77, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.R.; Baek, J.I.; Kim, S.H.; Kim, M.A.; Lee, B.; Ryu, N.; Kim, K.H.; Choi, D.G.; Kim, H.M.; Murphy, M.P.; et al. Therapeutic potential of the mitochondria-targeted antioxidant MitoQ in mitochondrial-ROS induced sensorineural hearing loss caused by Idh2 deficiency. Redox. Biol. 2019, 20, 544–555. [Google Scholar] [CrossRef]
- Ng, M.R.; Antonelli, P.J.; Joseph, J.; Dirain, C.O. Assessment of mitochondrial membrane potential in HEI-OC1 and LLC-PK1 cells treated with gentamicin and mitoquinone. Otolaryngol. Head Neck Surg. 2015, 152, 729–733. [Google Scholar] [CrossRef] [PubMed]
- Ojano-Dirain, C.P.; Antonelli, P.J. Prevention of gentamicin-induced apoptosis with the mitochondria-targeted antioxidant mitoquinone. Laryngoscope 2012, 122, 2543–2548. [Google Scholar] [CrossRef] [PubMed]
- Ojano-Dirain, C.P.; Antonelli, P.J.; Le Prell, C.G. Mitochondria-targeted antioxidant MitoQ reduces gentamicin-induced ototoxicity. Otol. Neurotol. 2014, 35, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Tate, A.D.; Antonelli, P.J.; Hannabass, K.R.; Dirain, C.O. Mitochondria-Targeted Antioxidant Mitoquinone Reduces Cisplatin-Induced Ototoxicity in Guinea Pigs. Otolaryngol. Head Neck Surg. 2017, 156, 543–548. [Google Scholar] [CrossRef]
- Carroll, C.J.; Brilhante, V.; Suomalainen, A. Next-generation sequencing for mitochondrial disorders. Br. J. Pharmacol. 2014, 171, 1837–1853. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, A.; Murayama, K.; Mori, M.; Harashima, H.; Yamazaki, T.; Tamaru, S.; Yamashita, Y.; Kishita, Y.; Nakachi, Y.; Kohda, M.; et al. Diagnosis and molecular basis of mitochondrial respiratory chain disorders: exome sequencing for disease gene identification. Biochim. Biophys. Acta 2014, 1840, 1355–1359. [Google Scholar] [CrossRef] [PubMed]
- D’Autreaux, B.; Toledano, M.B. ROS as signalling molecules: mechanisms that generate specificity in ROS homeostasis. Nat. Rev. Mol. Cell Biol. 2007, 8, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Sena, L.A.; Chandel, N.S. Physiological roles of mitochondrial reactive oxygen species. Mol. Cell. 2012, 48, 158–167. [Google Scholar] [CrossRef]
- Chinnery, P.F.; Howell, N.; Lightowlers, R.N.; Turnbull, D.M. Molecular pathology of MELAS and MERRF. The relationship between mutation load and clinical phenotypes. Brain 1997, 120, 1713–1721. [Google Scholar] [CrossRef]
- DiMauro, S.; Schon, E.A. Mitochondrial DNA mutations in human disease. Am. J. Med. Genet. 2001, 106, 18–26. [Google Scholar] [CrossRef]
- Kadowaki, T.; Kadowaki, H.; Mori, Y.; Tobe, K.; Sakuta, R.; Suzuki, Y.; Tanabe, Y.; Sakura, H.; Awata, T.; Goto, Y.; et al. A subtype of diabetes mellitus associated with a mutation of mitochondrial DNA. N. Engl. J. Med. 1994, 330, 962–968. [Google Scholar] [CrossRef]
- Chomyn, A. The myoclonic epilepsy and ragged-red fiber mutation provides new insights into human mitochondrial function and genetics. Am. J. Hum. Genet. 1998, 62, 745–751. [Google Scholar] [CrossRef]
- Schon, E.A.; Bonilla, E.; DiMauro, S. Mitochondrial DNA mutations and pathogenesis. J. Bioenerg. Biomembr. 1997, 29, 131–149. [Google Scholar] [CrossRef]
- Sue, C.M.; Lipsett, L.J.; Crimmins, D.S.; Tsang, C.S.; Boyages, S.C.; Presgrave, C.M.; Gibson, W.P.; Byrne, E.; Morris, J.G. Cochlear origin of hearing loss in MELAS syndrome. Ann. Neurol. 1998, 43, 350–359. [Google Scholar] [CrossRef]
- Kokotas, H.; Petersen, M.B.; Willems, P.J. Mitochondrial deafness. Clin. Genet. 2007, 71, 379–391. [Google Scholar] [CrossRef]
- Gates, G.A.; Mills, J.H. Presbycusis. Lancet 2005, 366, 1111–1120. [Google Scholar] [CrossRef]
- Schuknecht, H.F. Presbycusis. Laryngoscope 1955, 65, 402–419. [Google Scholar] [CrossRef]
- Flood, J.F.; Morley, J.E. Learning and memory in the SAMP8 mouse. Neurosci. Biobehav. Rev. 1998, 22, 1–20. [Google Scholar] [CrossRef]
- Menardo, J.; Tang, Y.; Ladrech, S.; Lenoir, M.; Casas, F.; Michel, C.; Bourien, J.; Ruel, J.; Rebillard, G.; Maurice, T.; et al. Oxidative stress, inflammation and autophagic stress as the key mechanisms of premature age-related hearing loss in SAMP8 mouse Cochlea. Antioxid. Redox. Signal. 2012, 16, 263–274. [Google Scholar] [CrossRef]
- Kamogashira, T.; Hayashi, K.; Fujimoto, C.; Iwasaki, S.; Yamasoba, T. Functionally and morphologically damaged mitochondria observed in auditory cells under senescence-inducing stress. Npj. Aging Mech. Dis. 2017, 3, 2. [Google Scholar] [CrossRef] [PubMed]
- Bai, U.; Seidman, M.D.; Hinojosa, R.; Quirk, W.S. Mitochondrial DNA deletions associated with aging and possibly presbycusis: A human archival temporal bone study. Am. J. Otol. 1997, 18, 449–453. [Google Scholar] [PubMed]
- Fischel-Ghodsian, N.; Bykhovskaya, Y.; Taylor, K.; Kahen, T.; Cantor, R.; Ehrenman, K.; Smith, R.; Keithley, E. Temporal bone analysis of patients with presbycusis reveals high frequency of mitochondrial mutations. Hear. Res. 1997, 110, 147–154. [Google Scholar] [CrossRef]
- Markaryan, A.; Nelson, E.G.; Hinojosa, R. Quantification of the mitochondrial DNA common deletion in presbycusis. Laryngoscope 2009, 119, 1184–1189. [Google Scholar] [CrossRef]
- Someya, S.; Yamasoba, T.; Kujoth, G.C.; Pugh, T.D.; Weindruch, R.; Tanokura, M.; Prolla, T.A. The role of mtDNA mutations in the pathogenesis of age-related hearing loss in mice carrying a mutator DNA polymerase gamma. Neurobiol. Aging 2008, 29, 1080–1092. [Google Scholar] [CrossRef] [PubMed]
- Wenz, T. Mitochondria and PGC-1α in Aging and Age-Associated Diseases. J. Aging Res. 2011, 2011, 810619. [Google Scholar] [CrossRef]
- Zhao, X.Y.; Sun, J.L.; Hu, Y.J.; Yang, Y.; Zhang, W.J.; Hu, Y.; Li, J.; Sun, Y.; Zhong, Y.; Peng, W.; et al. The effect of overexpression of PGC-1α on the mtDNA4834 common deletion in a rat cochlear marginal cell senescence model. Hear. Res. 2013, 296, 13–24. [Google Scholar] [CrossRef]
- Keithley, E.M.; Canto, C.; Zheng, Q.Y.; Wang, X.; Fischel-Ghodsian, N.; Johnson, K.R. Cu/Zn superoxide dismutase and age-related hearing loss. Hear. Res. 2005, 209, 76–85. [Google Scholar] [CrossRef] [PubMed]
- McFadden, S.L.; Ding, D.; Reaume, A.G.; Flood, D.G.; Salvi, R.J. Age-related cochlear hair cell loss is enhanced in mice lacking copper/zinc superoxide dismutase. Neurobiol. Aging 1999, 20, 1–8. [Google Scholar] [CrossRef]
- Coling, D.; Chen, S.; Chi, L.H.; Jamesdaniel, S.; Henderson, D. Age-related changes in antioxidant enzymes related to hydrogen peroxide metabolism in rat inner ear. Neurosci. Lett. 2009, 464, 22–25. [Google Scholar] [CrossRef]
- Ohlemiller, K.K.; McFadden, S.L.; Ding, D.L.; Lear, P.M.; Ho, Y.S. Targeted mutation of the gene for cellular glutathione peroxidase (Gpx1) increases noise-induced hearing loss in mice. J. Assoc. Res. Otolaryngol. 2000, 1, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Someya, S.; Xu, J.; Kondo, K.; Ding, D.; Salvi, R.J.; Yamasoba, T.; Rabinovitch, P.S.; Weindruch, R.; Leeuwenburgh, C.; Tanokura, M.; et al. Age-related hearing loss in C57BL/6J mice is mediated by Bak-dependent mitochondrial apoptosis. Proc. Natl. Acad. Sci. USA 2009, 106, 19432–19437. [Google Scholar] [CrossRef] [PubMed]
- White, K.; Kim, M.J.; Han, C.; Park, H.J.; Ding, D.; Boyd, K.; Walker, L.; Linser, P.; Meneses, Z.; Slade, C.; et al. Loss of IDH2 Accelerates Age-related Hearing Loss in Male Mice. Sci. Rep. 2018, 8, 5039. [Google Scholar] [CrossRef]
- Choi, S.H.; Choi, C.H. Noise-Induced Neural Degeneration and Therapeutic Effect of Antioxidant Drugs. J. Audiol. Otol. 2015, 19, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Spoendlin, H. Primary structural changes in the organ of Corti after acoustic overstimulation. Acta Otolaryngol. 1971, 71, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Fridberger, A.; Flock, A.; Ulfendahl, M.; Flock, B. Acoustic overstimulation increases outer hair cell Ca2+ concentrations and causes dynamic contractions of the hearing organ. Proc. Natl. Acad. Sci. USA 1998, 95, 7127–7132. [Google Scholar] [CrossRef] [PubMed]
- Maurer, J.; Heinrich, U.R.; Mann, W. Morphologic damage and changes of intracellular calcium-binding sites after acute noise trauma in the organ of Corti of the guinea pig. Orl. J. Otorhinolaryngol. Relat. Spec. 1993, 55, 7–12. [Google Scholar] [CrossRef]
- Minami, S.B.; Yamashita, D.; Schacht, J.; Miller, J.M. Calcineurin activation contributes to noise-induced hearing loss. J. Neurosci. Res. 2004, 78, 383–392. [Google Scholar] [CrossRef]
- Vicente-Torres, M.A.; Schacht, J. A BAD link to mitochondrial cell death in the cochlea of mice with noise-induced hearing loss. J. Neurosci. Res. 2006, 83, 1564–1572. [Google Scholar] [CrossRef]
- Rizzuto, R.; De Stefani, D.; Raffaello, A.; Mammucari, C. Mitochondria as sensors and regulators of calcium signalling. Nat. Rev. Mol. Cell Biol. 2012, 13, 566–578. [Google Scholar] [CrossRef] [PubMed]
- Patron, M.; Raffaello, A.; Granatiero, V.; Tosatto, A.; Merli, G.; De Stefani, D.; Wright, L.; Pallafacchina, G.; Terrin, A.; Mammucari, C.; et al. The mitochondrial calcium uniporter (MCU): molecular identity and physiological roles. J. Biol. Chem. 2013, 288, 10750–10758. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, Y.; Long, H.; Pan, S.; Xiong, H.; Fang, Q.; Hill, K.; Lai, R.; Yuan, H.; Sha, S.H. Mitochondrial Calcium Transporters Mediate Sensitivity to Noise-Induced Losses of Hair Cells and Cochlear Synapses. Front. Mol. Neurosci. 2018, 11, 469. [Google Scholar] [CrossRef] [PubMed]
- Ohlemiller, K.K.; Wright, J.S.; Dugan, L.L. Early elevation of cochlear reactive oxygen species following noise exposure. Audiol. Neurootol. 1999, 4, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, D.; Jiang, H.Y.; Schacht, J.; Miller, J.M. Delayed production of free radicals following noise exposure. Brain. Res. 2004, 1019, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Wang, X.; Hill, K.; Chen, J.; Lemasters, J.; Yang, S.M.; Sha, S.H. Autophagy attenuates noise-induced hearing loss by reducing oxidative stress. Antioxid. Redox. Signal. 2015, 22, 1308–1324. [Google Scholar] [CrossRef]
- Gorlach, A.; Bertram, K.; Hudecova, S.; Krizanova, O. Calcium and ROS: A mutual interplay. Redox. Biol. 2015, 6, 260–271. [Google Scholar] [CrossRef]
- Tuerdi, A.; Kinoshita, M.; Kamogashira, T.; Fujimoto, C.; Iwasaki, S.; Shimizu, T.; Yamasoba, T. Manganese superoxide dismutase influences the extent of noise-induced hearing loss in mice. Neurosci. Lett. 2017, 642, 123–128. [Google Scholar] [CrossRef]
- Ohinata, Y.; Miller, J.M.; Altschuler, R.A.; Schacht, J. Intense noise induces formation of vasoactive lipid peroxidation products in the cochlea. Brain. Res. 2000, 878, 163–173. [Google Scholar] [CrossRef]
- Miller, J.M.; Brown, J.N.; Schacht, J. 8-iso-prostaglandin F(2α), a product of noise exposure, reduces inner ear blood flow. Audiol. Neurootol. 2003, 8, 207–221. [Google Scholar] [CrossRef]
- Quirk, W.S.; Seidman, M.D. Cochlear vascular changes in response to loud noise. Am. J. Otol. 1995, 16, 322–325. [Google Scholar] [PubMed]
- Chen, F.Q.; Zheng, H.W.; Hill, K.; Sha, S.H. Traumatic noise activates Rho-family GTPases through transient cellular energy depletion. J. Neurosci. 2012, 32, 12421–12430. [Google Scholar] [CrossRef]
- Nagashima, R.; Yamaguchi, T.; Kuramoto, N.; Ogita, K. Acoustic overstimulation activates 5′-AMP-activated protein kinase through a temporary decrease in ATP level in the cochlear spiral ligament prior to permanent hearing loss in mice. Neurochem. Int. 2011, 59, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Vlajkovic, S.M.; Housley, G.D.; Munoz, D.J.; Robson, S.C.; Sevigny, J.; Wang, C.J.; Thorne, P.R. Noise exposure induces up-regulation of ecto-nucleoside triphosphate diphosphohydrolases 1 and 2 in rat cochlea. Neuroscience 2004, 126, 763–773. [Google Scholar] [CrossRef]
- Dehne, N.; Rauen, U.; de Groot, H.; Lautermann, J. Involvement of the mitochondrial permeability transition in gentamicin ototoxicity. Hear. Res. 2002, 169, 47–55. [Google Scholar] [CrossRef]
- Hyde, G.E.; Rubel, E.W. Mitochondrial role in hair cell survival after injury. Otolaryngol. Head Neck Surg. 1995, 113, 530–540. [Google Scholar] [CrossRef]
- Jensen-Smith, H.C.; Hallworth, R.; Nichols, M.G. Gentamicin rapidly inhibits mitochondrial metabolism in high-frequency cochlear outer hair cells. PLoS ONE 2012, 7, e38471. [Google Scholar] [CrossRef]
- He, Z.; Sun, S.; Waqas, M.; Zhang, X.; Qian, F.; Cheng, C.; Zhang, M.; Zhang, S.; Wang, Y.; Tang, M.; et al. Reduced TRMU expression increases the sensitivity of hair-cell-like HEI-OC-1 cells to neomycin damage in vitro. Sci. Rep. 2016, 6, 29621. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Chen, Y.; Guan, M.X. Mitochondrial DNA mutations associated with aminoglycoside induced ototoxicity. J. Otol. 2017, 12, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hobbie, S.N.; Akshay, S.; Kalapala, S.K.; Bruell, C.M.; Shcherbakov, D.; Bottger, E.C. Genetic analysis of interactions with eukaryotic rRNA identify the mitoribosome as target in aminoglycoside ototoxicity. Proc. Natl. Acad. Sci. USA 2008, 105, 20888–20893. [Google Scholar] [CrossRef]
- Clerici, W.J.; Hensley, K.; DiMartino, D.L.; Butterfield, D.A. Direct detection of ototoxicant-induced reactive oxygen species generation in cochlear explants. Hear. Res. 1996, 98, 116–124. [Google Scholar] [CrossRef]
- Forge, A.; Fradis, M. Structural abnormalities in the stria vascularis following chronic gentamicin treatment. Hear. Res. 1985, 20, 233–244. [Google Scholar] [CrossRef]
- Nakagawa, T.; Yamane, H.; Takayama, M.; Sunami, K.; Nakai, Y. Apoptosis of guinea pig cochlear hair cells following chronic aminoglycoside treatment. Eur. Arch. Otorhinolaryngol. 1998, 255, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Priuska, E.M.; Schacht, J. Formation of free radicals by gentamicin and iron and evidence for an iron/gentamicin complex. Biochem. Pharmacol. 1995, 50, 1749–1752. [Google Scholar] [CrossRef]
- Sha, S.H.; Schacht, J. Formation of reactive oxygen species following bioactivation of gentamicin. Free. Radic. Biol. Med. 1999, 26, 341–347. [Google Scholar] [CrossRef]
- Sha, S.H.; Schacht, J. Stimulation of free radical formation by aminoglycoside antibiotics. Hear. Res. 1999, 128, 112–118. [Google Scholar] [CrossRef]
- Hong, S.H.; Park, S.K.; Cho, Y.S.; Lee, H.S.; Kim, K.R.; Kim, M.G.; Chung, W.H. Gentamicin induced nitric oxide-related oxidative damages on vestibular afferents in the guinea pig. Hear. Res. 2006, 211, 46–53. [Google Scholar] [CrossRef]
- Jiang, H.; Sha, S.H.; Schacht, J. Rac/Rho pathway regulates actin depolymerization induced by aminoglycoside antibiotics. J. Neurosci. Res. 2006, 83, 1544–1551. [Google Scholar] [CrossRef]
- Kopke, R.D.; Liu, W.; Gabaizadeh, R.; Jacono, A.; Feghali, J.; Spray, D.; Garcia, P.; Steinman, H.; Malgrange, B.; Ruben, R.J.; et al. Use of organotypic cultures of Corti’s organ to study the protective effects of antioxidant molecules on cisplatin-induced damage of auditory hair cells. Am. J. Otol. 1997, 18, 559–571. [Google Scholar]
- Banfi, B.; Malgrange, B.; Knisz, J.; Steger, K.; Dubois-Dauphin, M.; Krause, K.H. NOX3, a superoxide-generating NADPH oxidase of the inner ear. J. Biol. Chem. 2004, 279, 46065–46072. [Google Scholar] [CrossRef]
- Lynch, E.D.; Gu, R.; Pierce, C.; Kil, J. Reduction of acute cisplatin ototoxicity and nephrotoxicity in rats by oral administration of allopurinol and ebselen. Hear. Res. 2005, 201, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Mukherjea, D.; Jajoo, S.; Kaur, T.; Sheehan, K.E.; Ramkumar, V.; Rybak, L.P. Transtympanic administration of short interfering (si)RNA for the NOX3 isoform of NADPH oxidase protects against cisplatin-induced hearing loss in the rat. Antioxid. Redox. Signal. 2010, 13, 589–598. [Google Scholar] [CrossRef]
- Hoffman, D.W.; Whitworth, C.A.; Jones-King, K.L.; Rybak, L.P. Potentiation of ototoxicity by glutathione depletion. Ann. Otol. Rhinol. Laryngol. 1988, 97, 36–41. [Google Scholar] [CrossRef]
- Ravi, R.; Somani, S.M.; Rybak, L.P. Mechanism of cisplatin ototoxicity: Antioxidant system. Pharmacol. Toxicol. 1995, 76, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Rybak, L.P.; Ravi, R.; Somani, S.M. Mechanism of protection by diethyldithiocarbamate against cisplatin ototoxicity: Antioxidant system. Fundam. Appl. Toxicol. 1995, 26, 293–300. [Google Scholar] [CrossRef]
- Teranishi, M.; Nakashima, T.; Wakabayashi, T. Effects of α-tocopherol on cisplatin-induced ototoxicity in guinea pigs. Hear. Res. 2001, 151, 61–70. [Google Scholar] [CrossRef]
- Alam, S.A.; Ikeda, K.; Oshima, T.; Suzuki, M.; Kawase, T.; Kikuchi, T.; Takasaka, T. Cisplatin-induced apoptotic cell death in Mongolian gerbil cochlea. Hear. Res. 2000, 141, 28–38. [Google Scholar] [CrossRef]
- Wang, J.; Ladrech, S.; Pujol, R.; Brabet, P.; Van De Water, T.R.; Puel, J.L. Caspase inhibitors but not c-Jun NH2-terminal kinase inhibitor treatment, prevent cisplatin-induced hearing loss. Cancer Res. 2004, 64, 9217–9224. [Google Scholar] [CrossRef]
- Borse, V.; Al Aameri, R.F.H.; Sheehan, K.; Sheth, S.; Kaur, T.; Mukherjea, D.; Tupal, S.; Lowy, M.; Ghosh, S.; Dhukhwa, A.; et al. Epigallocatechin-3-gallate, a prototypic chemopreventative agent for protection against cisplatin-based ototoxicity. Cell. Death. Dis. 2017, 8, e2921. [Google Scholar] [CrossRef]
- Devarajan, P.; Savoca, M.; Castaneda, M.P.; Park, M.S.; Esteban-Cruciani, N.; Kalinec, G.; Kalinec, F. Cisplatin-induced apoptosis in auditory cells: role of death receptor and mitochondrial pathways. Hear. Res. 2002, 174, 45–54. [Google Scholar] [CrossRef]
- Tavanai, E.; Mohammadkhani, G. Role of antioxidants in prevention of age-related hearing loss: A review of literature. Eur. Arch. Otorhinolaryngol. 2017, 274, 1821–1834. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; McMahon, C.M.; Burlutsky, G.; Spankovich, C.; Hood, L.J.; Mitchell, P. Dietary antioxidant intake is associated with the prevalence but not incidence of age-related hearing loss. J. Nutr. Health. Aging 2011, 15, 896–900. [Google Scholar] [CrossRef]
- Kang, J.W.; Choi, H.S.; Kim, K.; Choi, J.Y. Dietary vitamin intake correlates with hearing thresholds in the older population: the Korean National Health and Nutrition Examination Survey. Am. J. Clin. Nutr. 2014, 99, 1407–1413. [Google Scholar] [CrossRef]
- Lasisi, T.J.; Lasisi, A.O. Evaluation of serum antioxidants in age-related hearing loss. Aging Clin. Exp. Res. 2015, 27, 265–269. [Google Scholar] [CrossRef]
- Michikawa, T.; Nishiwaki, Y.; Kikuchi, Y.; Hosoda, K.; Mizutari, K.; Saito, H.; Asakura, K.; Milojevic, A.; Iwasawa, S.; Nakano, M.; et al. Serum levels of retinol and other antioxidants for hearing impairment among Japanese older adults. J. Gerontol. A. Biol. Sci. Med. Sci. 2009, 64, 910–915. [Google Scholar] [CrossRef]
- Spankovich, C.; Hood, L.J.; Silver, H.J.; Lambert, W.; Flood, V.M.; Mitchell, P. Associations between diet and both high and low pure tone averages and transient evoked otoacoustic emissions in an older adult population-based study. J. Am. Acad. Audiol. 2011, 22, 49–58. [Google Scholar] [CrossRef]
- Polanski, J.F.; Cruz, O.L. Evaluation of antioxidant treatment in presbyacusis: prospective, placebo-controlled, double-blind, randomised trial. J. Laryngol. Otol. 2013, 127, 134–141. [Google Scholar] [CrossRef]
- Shargorodsky, J.; Curhan, S.G.; Eavey, R.; Curhan, G.C. A prospective study of vitamin intake and the risk of hearing loss in men. Otolaryngol. Head Neck Surg. 2010, 142, 231–236. [Google Scholar] [CrossRef]
- Seidman, M.D. Effects of dietary restriction and antioxidants on presbyacusis. Laryngoscope 2000, 110, 727–738. [Google Scholar] [CrossRef]
- Seidman, M.D.; Khan, M.J.; Tang, W.X.; Quirk, W.S. Influence of lecithin on mitochondrial DNA and age-related hearing loss. Otolaryngol. Head Neck Surg. 2002, 127, 138–144. [Google Scholar] [CrossRef]
- Takumida, M.; Anniko, M. Radical scavengers: A remedy for presbyacusis. A pilot study. Acta Otolaryngol. 2005, 125, 1290–1295. [Google Scholar] [CrossRef] [PubMed]
- Kashio, A.; Amano, A.; Kondo, Y.; Sakamoto, T.; Iwamura, H.; Suzuki, M.; Ishigami, A.; Yamasoba, T. Effect of vitamin C depletion on age-related hearing loss in SMP30/GNL knockout mice. Biochem. Biophys. Res. Commun. 2009, 390, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Hu, Y.; Zhong, Y.; Chen, B.; Sun, Y.; Yang, Y.; Kong, W. Protective roles of α-lipoic acid in rat model of mitochondrial DNA4834bp deletion in inner ear. J. Huazhong. Univ. Sci. Technolog. Med. Sci. 2010, 30, 514–518. [Google Scholar] [CrossRef] [PubMed]
- Salami, A.; Mora, R.; Dellepiane, M.; Manini, G.; Santomauro, V.; Barettini, L.; Guastini, L. Water-soluble coenzyme Q10 formulation (Q-TER((R))) in the treatment of presbycusis. Acta Otolaryngol. 2010, 130, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Takumida, M.; Anniko, M. Radical scavengers for elderly patients with age-related hearing loss. Acta Otolaryngol. 2009, 129, 36–44. [Google Scholar] [CrossRef]
- Davis, R.R.; Kuo, M.W.; Stanton, S.G.; Canlon, B.; Krieg, E.; Alagramam, K.N. N-Acetyl L-cysteine does not protect against premature age-related hearing loss in C57BL/6J mice: A pilot study. Hear. Res. 2007, 226, 203–208. [Google Scholar] [CrossRef]
- Ding, D.; Jiang, H.; Chen, G.D.; Longo-Guess, C.; Muthaiah, V.P.; Tian, C.; Sheppard, A.; Salvi, R.; Johnson, K.R. N-acetyl-cysteine prevents age-related hearing loss and the progressive loss of inner hair cells in gamma-glutamyl transferase 1 deficient mice. Aging (Albany Ny) 2016, 8, 730–750. [Google Scholar] [CrossRef]
- Durga, J.; Verhoef, P.; Anteunis, L.J.; Schouten, E.; Kok, F.J. Effects of folic acid supplementation on hearing in older adults: A randomized, controlled trial. Ann. Intern. Med. 2007, 146, 1–9. [Google Scholar] [CrossRef]
- Ohinata, Y.; Yamasoba, T.; Schacht, J.; Miller, J.M. Glutathione limits noise-induced hearing loss. Hear. Res. 2000, 146, 28–34. [Google Scholar] [CrossRef]
- Yamasoba, T.; Nuttall, A.L.; Harris, C.; Raphael, Y.; Miller, J.M. Role of glutathione in protection against noise-induced hearing loss. Brain. Res. 1998, 784, 82–90. [Google Scholar] [CrossRef]
- Campbell, K.C.; Meech, R.P.; Klemens, J.J.; Gerberi, M.T.; Dyrstad, S.S.; Larsen, D.L.; Mitchell, D.L.; El-Azizi, M.; Verhulst, S.J.; Hughes, L.F. Prevention of noise- and drug-induced hearing loss with D-methionine. Hear. Res. 2007, 226, 92–103. [Google Scholar] [CrossRef]
- Kil, J.; Pierce, C.; Tran, H.; Gu, R.; Lynch, E.D. Ebselen treatment reduces noise-induced hearing loss via the mimicry and induction of glutathione peroxidase. Hear. Res. 2007, 226, 44–51. [Google Scholar] [CrossRef]
- Lynch, E.D.; Gu, R.; Pierce, C.; Kil, J. Ebselen-mediated protection from single and repeated noise exposure in rat. Laryngoscope 2004, 114, 333–337. [Google Scholar] [CrossRef]
- Pourbakht, A.; Yamasoba, T. Ebselen attenuates cochlear damage caused by acoustic trauma. Hear. Res. 2003, 181, 100–108. [Google Scholar] [CrossRef]
- Seidman, M.; Babu, S.; Tang, W.; Naem, E.; Quirk, W.S. Effects of resveratrol on acoustic trauma. Otolaryngol. Head Neck Surg. 2003, 129, 463–470. [Google Scholar] [CrossRef]
- Heinrich, U.R.; Fischer, I.; Brieger, J.; Rumelin, A.; Schmidtmann, I.; Li, H.; Mann, W.J.; Helling, K. Ascorbic acid reduces noise-induced nitric oxide production in the guinea pig ear. Laryngoscope 2008, 118, 837–842. [Google Scholar] [CrossRef] [PubMed]
- McFadden, S.L.; Woo, J.M.; Michalak, N.; Ding, D. Dietary vitamin C supplementation reduces noise-induced hearing loss in guinea pigs. Hear. Res. 2005, 202, 200–208. [Google Scholar] [CrossRef]
- Fetoni, A.R.; De Bartolo, P.; Eramo, S.L.; Rolesi, R.; Paciello, F.; Bergamini, C.; Fato, R.; Paludetti, G.; Petrosini, L.; Troiani, D. Noise-induced hearing loss (NIHL) as a target of oxidative stress-mediated damage: cochlear and cortical responses after an increase in antioxidant defense. J. Neurosci. 2013, 33, 4011–4023. [Google Scholar] [CrossRef]
- Fetoni, A.R.; Piacentini, R.; Fiorita, A.; Paludetti, G.; Troiani, D. Water-soluble Coenzyme Q10 formulation (Q-ter) promotes outer hair cell survival in a guinea pig model of noise-induced hearing loss (NIHL). Brain. Res. 2009, 1257, 108–116. [Google Scholar] [CrossRef]
- Sergi, B.; Fetoni, A.R.; Paludetti, G.; Ferraresi, A.; Navarra, P.; Mordente, A.; Troiani, D. Protective properties of idebenone in noise-induced hearing loss in the guinea pig. Neuroreport 2006, 17, 857–861. [Google Scholar] [CrossRef]
- Kopke, R.; Bielefeld, E.; Liu, J.; Zheng, J.; Jackson, R.; Henderson, D.; Coleman, J.K. Prevention of impulse noise-induced hearing loss with antioxidants. Acta Otolaryngol. 2005, 125, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Kopke, R.D.; Coleman, J.K.; Liu, J.; Campbell, K.C.; Riffenburgh, R.H. Candidate’s thesis: enhancing intrinsic cochlear stress defenses to reduce noise-induced hearing loss. Laryngoscope 2002, 112, 1515–1532. [Google Scholar] [CrossRef] [PubMed]
- Fetoni, A.R.; Mancuso, C.; Eramo, S.L.; Ralli, M.; Piacentini, R.; Barone, E.; Paludetti, G.; Troiani, D. In vivo protective effect of ferulic acid against noise-induced hearing loss in the guinea-pig. Neuroscience 2010, 169, 1575–1588. [Google Scholar] [CrossRef]
- Bielefeld, E.C.; Kopke, R.D.; Jackson, R.L.; Coleman, J.K.; Liu, J.; Henderson, D. Noise protection with N-acetyl-l-cysteine (NAC) using a variety of noise exposures, NAC doses and routes of administration. Acta Otolaryngol. 2007, 127, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.; Huang, X.; Liu, J.; Kopke, R.; Jackson, R. Dosing study on the effectiveness of salicylate/N-acetylcysteine for prevention of noise-induced hearing loss. Noise. Health. 2010, 12, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Duan, M.; Qiu, J.; Laurell, G.; Olofsson, A.; Counter, S.A.; Borg, E. Dose and time-dependent protection of the antioxidant N-L-acetylcysteine against impulse noise trauma. Hear. Res. 2004, 192, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fetoni, A.R.; Ralli, M.; Sergi, B.; Parrilla, C.; Troiani, D.; Paludetti, G. Protective effects of N-acetylcysteine on noise-induced hearing loss in guinea pigs. Acta Otorhinolaryngol. Ital. 2009, 29, 70–75. [Google Scholar] [PubMed]
- Kopke, R.D.; Weisskopf, P.A.; Boone, J.L.; Jackson, R.L.; Wester, D.C.; Hoffer, M.E.; Lambert, D.C.; Charon, C.C.; Ding, D.L.; McBride, D. Reduction of noise-induced hearing loss using L-NAC and salicylate in the chinchilla. Hear. Res. 2000, 149, 138–146. [Google Scholar] [CrossRef]
- Lorito, G.; Giordano, P.; Petruccelli, J.; Martini, A.; Hatzopoulos, S. Different strategies in treating noiseinduced hearing loss with N-acetylcysteine. Med. Sci. Monit. 2008, 14, BR159–BR164. [Google Scholar] [PubMed]
- Choi, C.H.; Chen, K.; Vasquez-Weldon, A.; Jackson, R.L.; Floyd, R.A.; Kopke, R.D. Effectiveness of 4-hydroxy phenyl N-tert-butylnitrone (4-OHPBN) alone and in combination with other antioxidant drugs in the treatment of acute acoustic trauma in chinchilla. Free. Radic. Biol. Med. 2008, 44, 1772–1784. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.R.; Custer, D.A.; Krieg, E.; Alagramam, K. N-Acetyl L-Cysteine does not protect mouse ears from the effects of noise*. J. Occup. Med. Toxicol. 2010, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Hamernik, R.P.; Qiu, W.; Davis, B. The effectiveness of N-acetyl-L-cysteine (L-NAC) in the prevention of severe noise-induced hearing loss. Hear. Res. 2008, 239, 99–106. [Google Scholar] [CrossRef]
- Doosti, A.; Lotfi, Y.; Moossavi, A.; Bakhshi, E.; Talasaz, A.H.; Hoorzad, A. Comparison of the effects of N-acetyl-cysteine and ginseng in prevention of noise-induced hearing loss in male textile workers. Noise. Health. 2014, 16, 223–227. [Google Scholar] [CrossRef]
- Kramer, S.; Dreisbach, L.; Lockwood, J.; Baldwin, K.; Kopke, R.; Scranton, S.; O’Leary, M. Efficacy of the antioxidant N-acetylcysteine (NAC) in protecting ears exposed to loud music. J. Am. Acad. Audiol. 2006, 17, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Kopke, R.; Slade, M.D.; Jackson, R.; Hammill, T.; Fausti, S.; Lonsbury-Martin, B.; Sanderson, A.; Dreisbach, L.; Rabinowitz, P.; Torre, P., 3rd; et al. Efficacy and safety of N-acetylcysteine in prevention of noise-induced hearing loss: A randomized clinical trial. Hear. Res. 2015, 323, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Kil, J.; Lobarinas, E.; Spankovich, C.; Griffiths, S.K.; Antonelli, P.J.; Lynch, E.D.; Le Prell, C.G. Safety and efficacy of ebselen for the prevention of noise-induced hearing loss: A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2017, 390, 969–979. [Google Scholar] [CrossRef]
- Bock, G.R.; Yates, G.K.; Miller, J.J.; Moorjani, P. Effects of N-acetylcysteine on kanamycin ototoxicity in the guinea pig. Hear. Res. 1983, 9, 255–262. [Google Scholar] [CrossRef]
- Maniu, A.; Perde-Schrepler, M.; Cosgarea, M. Protective effect of L-N-acetylcysteine against gentamycin ototoxicity in the organ cultures of the rat cochlea. Rom. J. Morphol. Embryol. 2011, 52, 159–164. [Google Scholar] [PubMed]
- Somdas, M.A.; Korkmaz, F.; Gurgen, S.G.; Sagit, M.; Akcadag, A. N-acetylcysteine Prevents Gentamicin Ototoxicity in a Rat Model. J. Int. Adv. Otol. 2015, 11, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.C.; Martin, S.M.; Meech, R.P.; Hargrove, T.L.; Verhulst, S.J.; Fox, D.J. D-methionine (D-met) significantly reduces kanamycin-induced ototoxicity in pigmented guinea pigs. Int. J. Audiol. 2016, 55, 273–278. [Google Scholar] [CrossRef]
- Fox, D.J.; Cooper, M.D.; Speil, C.A.; Roberts, M.H.; Yanik, S.C.; Meech, R.P.; Hargrove, T.L.; Verhulst, S.J.; Rybak, L.P.; Campbell, K.C. d-Methionine reduces tobramycin-induced ototoxicity without antimicrobial interference in animal models. J. Cyst. Fibros. 2016, 15, 518–530. [Google Scholar] [CrossRef] [PubMed]
- Turan, M.; Ciger, E.; Arslanoglu, S.; Borekci, H.; Onal, K. Could edaravone prevent gentamicin ototoxicity? An experimental study. Hum. Exp. Toxicol. 2017, 36, 123–127. [Google Scholar] [CrossRef]
- Kalinec, G.M.; Fernandez-Zapico, M.E.; Urrutia, R.; Esteban-Cruciani, N.; Chen, S.; Kalinec, F. Pivotal role of Harakiri in the induction and prevention of gentamicin-induced hearing loss. Proc. Natl. Acad. Sci. USA 2005, 102, 16019–16024. [Google Scholar] [CrossRef] [PubMed]
- Berkiten, G.; Salturk, Z.; Topaloglu, I.; Ugras, H. Protective effect of pentoxifylline on amikacin-induced ototoxicity in rats. Am. J. Otolaryngol. 2012, 33, 689–692. [Google Scholar] [CrossRef] [PubMed]
- El-Anwar, M.W.; Abdelmonem, S.; Nada, E.; Galhoom, D.; Abdelsameea, A.A. Protective effect of pentoxifylline on amikacin-induced ototoxicity. Ear. Nose. Throat. J. 2018, 97, E8–E12. [Google Scholar] [CrossRef]
- Fetoni, A.R.; Eramo, S.L.; Rolesi, R.; Troiani, D.; Paludetti, G. Antioxidant treatment with coenzyme Q-ter in prevention of gentamycin ototoxicity in an animal model. Acta Otorhinolaryngol. Ital. 2012, 32, 103–110. [Google Scholar]
- Noack, V.; Pak, K.; Jalota, R.; Kurabi, A.; Ryan, A.F. An Antioxidant Screen Identifies Candidates for Protection of Cochlear Hair Cells from Gentamicin Toxicity. Front. Cell. Neurosci. 2017, 11, 242. [Google Scholar] [CrossRef]
- Sha, S.H.; Qiu, J.H.; Schacht, J. Aspirin to prevent gentamicin-induced hearing loss. N. Engl. J. Med. 2006, 354, 1856–1857. [Google Scholar] [CrossRef]
- Feldman, L.; Efrati, S.; Eviatar, E.; Abramsohn, R.; Yarovoy, I.; Gersch, E.; Averbukh, Z.; Weissgarten, J. Gentamicin-induced ototoxicity in hemodialysis patients is ameliorated by N-acetylcysteine. Kidney. Int. 2007, 72, 359–363. [Google Scholar] [CrossRef]
- Feldman, L.; Sherman, R.A.; Weissgarten, J. N-acetylcysteine use for amelioration of aminoglycoside-induced ototoxicity in dialysis patients. Semin. Dial. 2012, 25, 491–494. [Google Scholar] [CrossRef]
- Kocyigit, I.; Vural, A.; Unal, A.; Sipahioglu, M.H.; Yucel, H.E.; Aydemir, S.; Yazici, C.; Ilhan Sahin, M.; Oymak, O.; Tokgoz, B. Preventing amikacin related ototoxicity with N-acetylcysteine in patients undergoing peritoneal dialysis. Eur. Arch. Otorhinolaryngol. 2015, 272, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Tokgoz, B.; Ucar, C.; Kocyigit, I.; Somdas, M.; Unal, A.; Vural, A.; Sipahioglu, M.; Oymak, O.; Utas, C. Protective effect of N-acetylcysteine from drug-induced ototoxicity in uraemic patients with CAPD peritonitis. Nephrol. Dial. Transplant. 2011, 26, 4073–4078. [Google Scholar] [CrossRef] [PubMed]
- Choe, W.T.; Chinosornvatana, N.; Chang, K.W. Prevention of cisplatin ototoxicity using transtympanic N-acetylcysteine and lactate. Otol. Neurotol. 2004, 25, 910–915. [Google Scholar] [CrossRef] [PubMed]
- Dickey, D.T.; Muldoon, L.L.; Doolittle, N.D.; Peterson, D.R.; Kraemer, D.F.; Neuwelt, E.A. Effect of N-acetylcysteine route of administration on chemoprotection against cisplatin-induced toxicity in rat models. Cancer Chemother. Pharmacol. 2008, 62, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Otto, W.C.; Brown, R.D.; Gage-White, L.; Kupetz, S.; Anniko, M.; Penny, J.E.; Henley, C.M. Effects of cisplatin and thiosulfate upon auditory brainstem responses of guinea pigs. Hear. Res. 1988, 35, 79–85. [Google Scholar] [CrossRef]
- Dickey, D.T.; Wu, Y.J.; Muldoon, L.L.; Neuwelt, E.A. Protection against cisplatin-induced toxicities by N-acetylcysteine and sodium thiosulfate as assessed at the molecular, cellular and in vivo levels. J. Pharmacol. Exp. Ther. 2005, 314, 1052–1058. [Google Scholar] [CrossRef] [PubMed]
- Berglin, C.E.; Pierre, P.V.; Bramer, T.; Edsman, K.; Ehrsson, H.; Eksborg, S.; Laurell, G. Prevention of cisplatin-induced hearing loss by administration of a thiosulfate-containing gel to the middle ear in a guinea pig model. Cancer Chemother. Pharmacol. 2011, 68, 1547–1556. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.C.; Rybak, L.P.; Meech, R.P.; Hughes, L. D-methionine provides excellent protection from cisplatin ototoxicity in the rat. Hear. Res. 1996, 102, 90–98. [Google Scholar] [CrossRef]
- Korver, K.D.; Rybak, L.P.; Whitworth, C.; Campbell, K.M. Round window application of D-methionine provides complete cisplatin otoprotection. Otolaryngol. Head Neck Surg. 2002, 126, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Rybak, L.P.; Whitworth, C.; Somani, S. Application of antioxidants and other agents to prevent cisplatin ototoxicity. Laryngoscope 1999, 109, 1740–1744. [Google Scholar] [CrossRef]
- Whitworth, C.A.; Ramkumar, V.; Jones, B.; Tsukasaki, N.; Rybak, L.P. Protection against cisplatin ototoxicity by adenosine agonists. Biochem. Pharmacol. 2004, 67, 1801–1807. [Google Scholar] [CrossRef]
- Church, M.W.; Blakley, B.W.; Burgio, D.L.; Gupta, A.K. WR-2721 (Amifostine) ameliorates cisplatin-induced hearing loss but causes neurotoxicity in hamsters: dose-dependent effects. J. Assoc. Res. Otolaryngol. 2004, 5, 227–237. [Google Scholar] [CrossRef]
- Rolland, V.; Meyer, F.; Guitton, M.J.; Bussieres, R.; Philippon, D.; Bairati, I.; Leclerc, M.; Cote, M. A randomized controlled trial to test the efficacy of trans-tympanic injections of a sodium thiosulfate gel to prevent cisplatin-induced ototoxicity in patients with head and neck cancer. J. Otolaryngol. Head Neck Surg. 2019, 48, 4. [Google Scholar] [CrossRef] [PubMed]
- Fouladi, M.; Chintagumpala, M.; Ashley, D.; Kellie, S.; Gururangan, S.; Hassall, T.; Gronewold, L.; Stewart, C.F.; Wallace, D.; Broniscer, A.; et al. Amifostine protects against cisplatin-induced ototoxicity in children with average-risk medulloblastoma. J. Clin. Oncol. 2008, 26, 3749–3755. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, Y.; Ehara, K.; Yamada, T.; Matsuzawa, N.; Arai, S.; Ban, D.; Kudo, A.; Tanabe, M.; Kawashima, Y.; Sakamoto, H. Three-dimensional computed tomography analysis of the vascular anatomy of the splenic hilum for gastric cancer surgery. Surg. Today 2018, 48, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Sastry, J.; Kellie, S.J. Severe neurotoxicity, ototoxicity and nephrotoxicity following high-dose cisplatin and amifostine. Pediatr. Hematol. Oncol. 2005, 22, 441–445. [Google Scholar] [CrossRef]
- Riga, M.G.; Chelis, L.; Kakolyris, S.; Papadopoulos, S.; Stathakidou, S.; Chamalidou, E.; Xenidis, N.; Amarantidis, K.; Dimopoulos, P.; Danielides, V. Transtympanic injections of N-acetylcysteine for the prevention of cisplatin-induced ototoxicity: A feasible method with promising efficacy. Am. J. Clin. Oncol. 2013, 36, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.; Hamilton, S.J.; Angel, D.; Fung, K.; Franklin, J.; Parnes, L.S.; Lewis, D.; Venkatesan, V.; Winquist, E. Cisplatin otoprotection using transtympanic L-N-acetylcysteine: A pilot randomized study in head and neck cancer patients. Laryngoscope 2014, 124, E87–E94. [Google Scholar] [CrossRef]
- Villani, V.; Zucchella, C.; Cristalli, G.; Galie, E.; Bianco, F.; Giannarelli, D.; Carpano, S.; Spriano, G.; Pace, A. Vitamin E neuroprotection against cisplatin ototoxicity: Preliminary results from a randomized, placebo-controlled trial. Head Neck. 2016, 38 (Suppl. 1), E2118–E2121. [Google Scholar] [CrossRef]
- Muratovska, A.; Lightowlers, R.N.; Taylor, R.W.; Wilce, J.A.; Murphy, M.P. Targeting large molecules to mitochondria. Adv. Drug. Deliv. Rev. 2001, 49, 189–198. [Google Scholar] [CrossRef]
- Skulachev, V.P.; Anisimov, V.N.; Antonenko, Y.N.; Bakeeva, L.E.; Chernyak, B.V.; Erichev, V.P.; Filenko, O.F.; Kalinina, N.I.; Kapelko, V.I.; Kolosova, N.G.; et al. An attempt to prevent senescence: A mitochondrial approach. Biochim. Biophys. Acta 2009, 1787, 437–461. [Google Scholar] [CrossRef] [PubMed]
- Weissig, V. Mitochondrial-targeted drug and DNA delivery. Crit. Rev. Ther. Drug Carrier Syst. 2003, 20, 1–62. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.A.; Porteous, C.M.; Coulter, C.V.; Murphy, M.P. Selective targeting of an antioxidant to mitochondria. Eur. J. Biochem. 1999, 263, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P.; Echtay, K.S.; Blaikie, F.H.; Asin-Cayuela, J.; Cocheme, H.M.; Green, K.; Buckingham, J.A.; Taylor, E.R.; Hurrell, F.; Hughes, G.; et al. Superoxide activates uncoupling proteins by generating carbon-centered radicals and initiating lipid peroxidation: studies using a mitochondria-targeted spin trap derived from α-phenyl-N-tert-butylnitrone. J. Biol. Chem. 2003, 278, 48534–48545. [Google Scholar] [CrossRef]
- Filipovska, A.; Kelso, G.F.; Brown, S.E.; Beer, S.M.; Smith, R.A.; Murphy, M.P. Synthesis and characterization of a triphenylphosphonium-conjugated peroxidase mimetic. Insights into the interaction of ebselen with mitochondria. J. Biol. Chem. 2005, 280, 24113–24126. [Google Scholar] [CrossRef] [PubMed]
- Antonenko, Y.N.; Roginsky, V.A.; Pashkovskaya, A.A.; Rokitskaya, T.I.; Kotova, E.A.; Zaspa, A.A.; Chernyak, B.V.; Skulachev, V.P. Protective effects of mitochondria-targeted antioxidant SkQ in aqueous and lipid membrane environments. J. Membr. Biol. 2008, 222, 141–149. [Google Scholar] [CrossRef]
- Rokitskaya, T.I.; Klishin, S.S.; Severina, I.I.; Skulachev, V.P.; Antonenko, Y.N. Kinetic analysis of permeation of mitochondria-targeted antioxidants across bilayer lipid membranes. J. Membr. Biol. 2008, 224, 9–19. [Google Scholar] [CrossRef]
- Zhao, K.; Zhao, G.M.; Wu, D.; Soong, Y.; Birk, A.V.; Schiller, P.W.; Szeto, H.H. Cell-permeable peptide antioxidants targeted to inner mitochondrial membrane inhibit mitochondrial swelling, oxidative cell death and reperfusion injury. J. Biol. Chem. 2004, 279, 34682–34690. [Google Scholar] [CrossRef]
- Yamada, Y.; Nakamura, K.; Abe, J.; Hyodo, M.; Haga, S.; Ozaki, M.; Harashima, H. Mitochondrial delivery of Coenzyme Q10 via systemic administration using a MITO-Porter prevents ischemia/reperfusion injury in the mouse liver. J. Control. Release 2015, 213, 86–95. [Google Scholar] [CrossRef]
- Ernster, L.; Dallner, G. Biochemical, physiological and medical aspects of ubiquinone function. Biochim. Biophys. Acta 1995, 1271, 195–204. [Google Scholar] [CrossRef]
- Matthews, R.T.; Yang, L.; Browne, S.; Baik, M.; Beal, M.F. Coenzyme Q10 administration increases brain mitochondrial concentrations and exerts neuroprotective effects. Proc. Natl. Acad. Sci. USA 1998, 95, 8892–8897. [Google Scholar] [CrossRef]
- Murphy, M.P. Targeting lipophilic cations to mitochondria. Biochim. Biophys. Acta 2008, 1777, 1028–1031. [Google Scholar] [CrossRef]
- Murphy, M.P.; Smith, R.A. Targeting antioxidants to mitochondria by conjugation to lipophilic cations. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 629–656. [Google Scholar] [CrossRef]
- James, A.M.; Cocheme, H.M.; Smith, R.A.; Murphy, M.P. Interactions of mitochondria-targeted and untargeted ubiquinones with the mitochondrial respiratory chain and reactive oxygen species. Implications for the use of exogenous ubiquinones as therapies and experimental tools. J. Biol. Chem. 2005, 280, 21295–21312. [Google Scholar] [CrossRef]
- Murphy, M.P. Understanding and preventing mitochondrial oxidative damage. Biochem. Soc. Trans. 2016, 44, 1219–1226. [Google Scholar] [CrossRef]
- Snow, B.J.; Rolfe, F.L.; Lockhart, M.M.; Frampton, C.M.; O’Sullivan, J.D.; Fung, V.; Smith, R.A.; Murphy, M.P.; Taylor, K.M.; Protect Study, G. A double-blind, placebo-controlled study to assess the mitochondria-targeted antioxidant MitoQ as a disease-modifying therapy in Parkinson’s disease. Mov. Disord. 2010, 25, 1670–1674. [Google Scholar] [CrossRef]
- Gane, E.J.; Weilert, F.; Orr, D.W.; Keogh, G.F.; Gibson, M.; Lockhart, M.M.; Frampton, C.M.; Taylor, K.M.; Smith, R.A.; Murphy, M.P. The mitochondria-targeted anti-oxidant mitoquinone decreases liver damage in a phase II study of hepatitis C patients. Liver Int. 2010, 30, 1019–1026. [Google Scholar] [CrossRef]
- Rossman, M.J.; Santos-Parker, J.R.; Steward, C.A.C.; Bispham, N.Z.; Cuevas, L.M.; Rosenberg, H.L.; Woodward, K.A.; Chonchol, M.; Gioscia-Ryan, R.A.; Murphy, M.P.; et al. Chronic Supplementation With a Mitochondrial Antioxidant (MitoQ) Improves Vascular Function in Healthy Older Adults. Hypertension 2018, 71, 1056–1063. [Google Scholar] [CrossRef]
- Antonenko, Y.N.; Avetisyan, A.V.; Bakeeva, L.E.; Chernyak, B.V.; Chertkov, V.A.; Domnina, L.V.; Ivanova, O.Y.; Izyumov, D.S.; Khailova, L.S.; Klishin, S.S.; et al. Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 1. Cationic plastoquinone derivatives: synthesis and in vitro studies. Biochemistry (Mosc.) 2008, 73, 1273–1287. [Google Scholar] [CrossRef]
- Bakeeva, L.E.; Barskov, I.V.; Egorov, M.V.; Isaev, N.K.; Kapelko, V.I.; Kazachenko, A.V.; Kirpatovsky, V.I.; Kozlovsky, S.V.; Lakomkin, V.L.; Levina, S.B.; et al. Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 2. Treatment of some ROS- and age-related diseases (heart arrhythmia, heart infarctions, kidney ischemia and stroke). Biochemistry (Mosc.) 2008, 73, 1288–1299. [Google Scholar] [CrossRef]
| Antioxidants | Major Findings |
|---|---|
Mito Q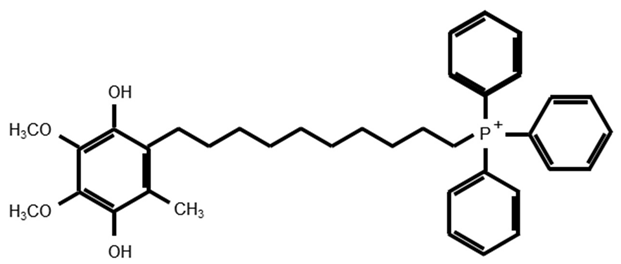 | Reducing cochlear damage and hearing loss (gentamicin, guinea pig) [15]. Decreasing apoptosis as compared to idebenone (gentamicin, HEI-OC1) [14]. Not decreasing apoptosis (gentamicin, HEI-OC1) [10]. Having a potential to disrupt the mitochondrial membrane (gentamicin, HEI-OC1) [13]. Limited protection against hearing loss and cochlear damage (amikacin, guinea pig) [9] Reducing OHC loss and hearing loss (cisplatin, guinea pig) [16] Protecting HCs against ototoxicity (H2O2, IDH2-deficient cochlear explant) [12] |
SkQR1 | Protecting against ototoxicity (gentamicin, rat) [11]. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujimoto, C.; Yamasoba, T. Mitochondria-Targeted Antioxidants for Treatment of Hearing Loss: A Systematic Review. Antioxidants 2019, 8, 109. https://doi.org/10.3390/antiox8040109
Fujimoto C, Yamasoba T. Mitochondria-Targeted Antioxidants for Treatment of Hearing Loss: A Systematic Review. Antioxidants. 2019; 8(4):109. https://doi.org/10.3390/antiox8040109
Chicago/Turabian StyleFujimoto, Chisato, and Tatsuya Yamasoba. 2019. "Mitochondria-Targeted Antioxidants for Treatment of Hearing Loss: A Systematic Review" Antioxidants 8, no. 4: 109. https://doi.org/10.3390/antiox8040109
APA StyleFujimoto, C., & Yamasoba, T. (2019). Mitochondria-Targeted Antioxidants for Treatment of Hearing Loss: A Systematic Review. Antioxidants, 8(4), 109. https://doi.org/10.3390/antiox8040109





