Dietary Selenium Supplementation Ameliorates Female Reproductive Efficiency in Aging Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals and Experimental Groups and Diet Regimes
2.3. Blood Analyses
2.3.1. Determination of Whole-Blood Se Concentrations
2.3.2. Determination of Blood Total Antioxidant Capacity
2.4. Ovarian Tissue Specimen Collection
2.4.1. Histological Assessment for Follicle Counting
2.4.2. Immunohistochemistry for GPX4 Expression Analysis
2.4.3. Detection of Apoptosis in Ovarian Tissues by TUNEL Assay
2.5. Total RNA Extraction and cDNA Synthesis
2.6. Quantitative Reverse Transcription PCR (RT-qPCR)
2.7. Superovulation, GV Oocyte Collection and In Vitro Culture
2.7.1. Oocyte Collection and In Vitro Maturation (IVM)
2.7.2. Oocytes Activation and Embryonic Development
2.8. Statistical Analyses
3. Results
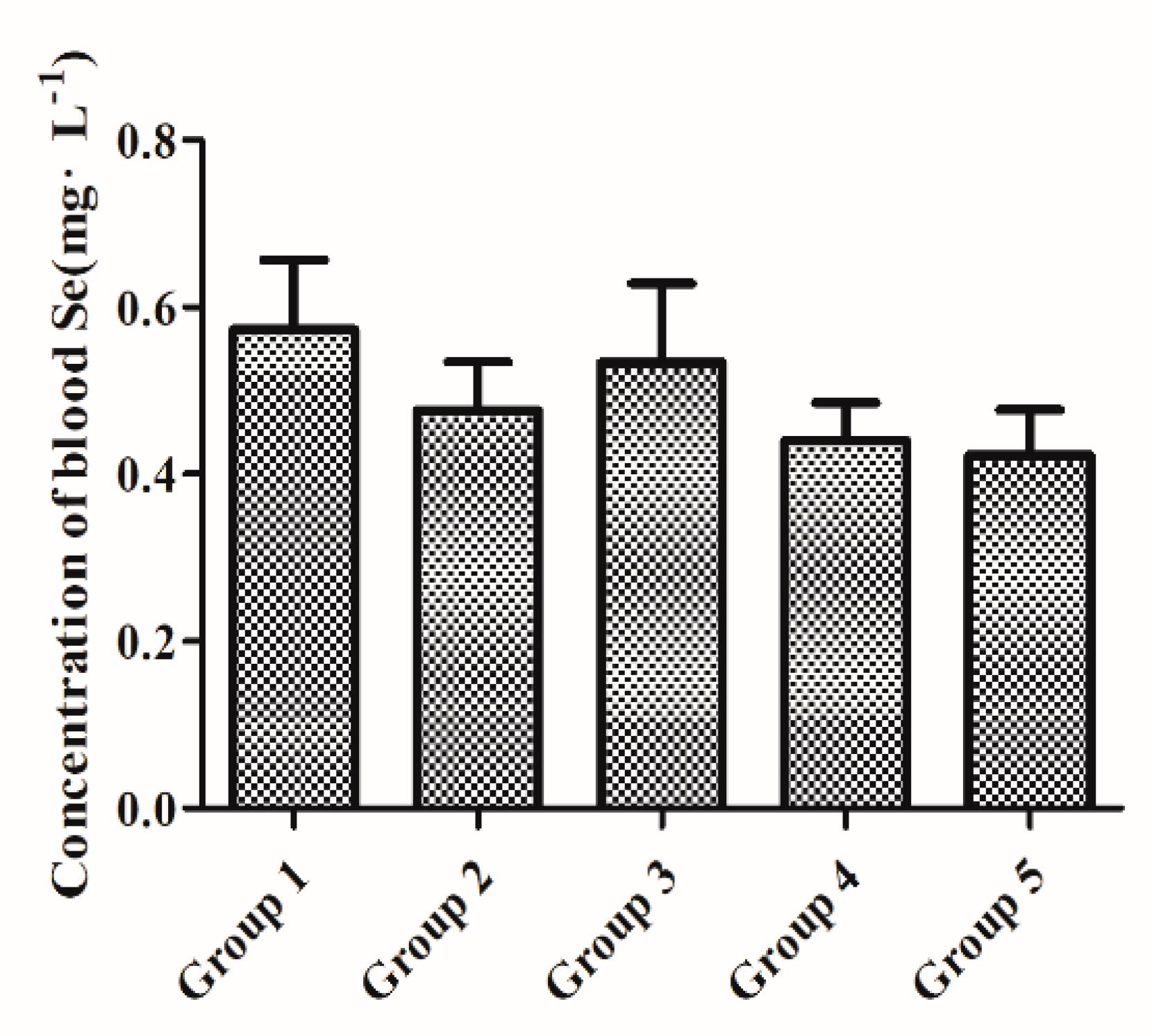
3.1. Baseline Whole-Blood Se Status after Initial Two-Week Feeding with a Low-Se Diet
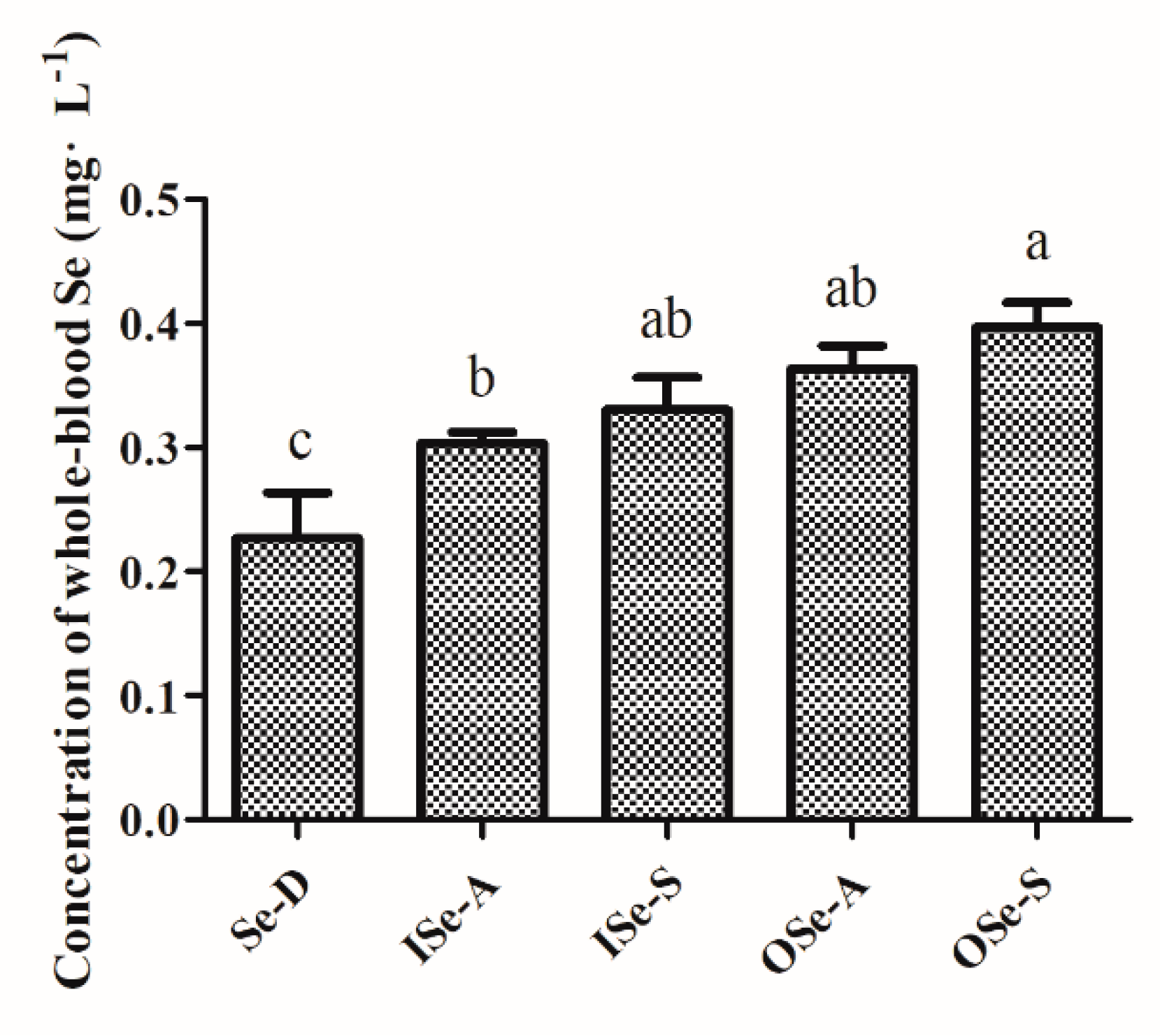
3.2. Endpoint Whole-Blood Se Status in Aging Mice Fed Different Concentrations of Se
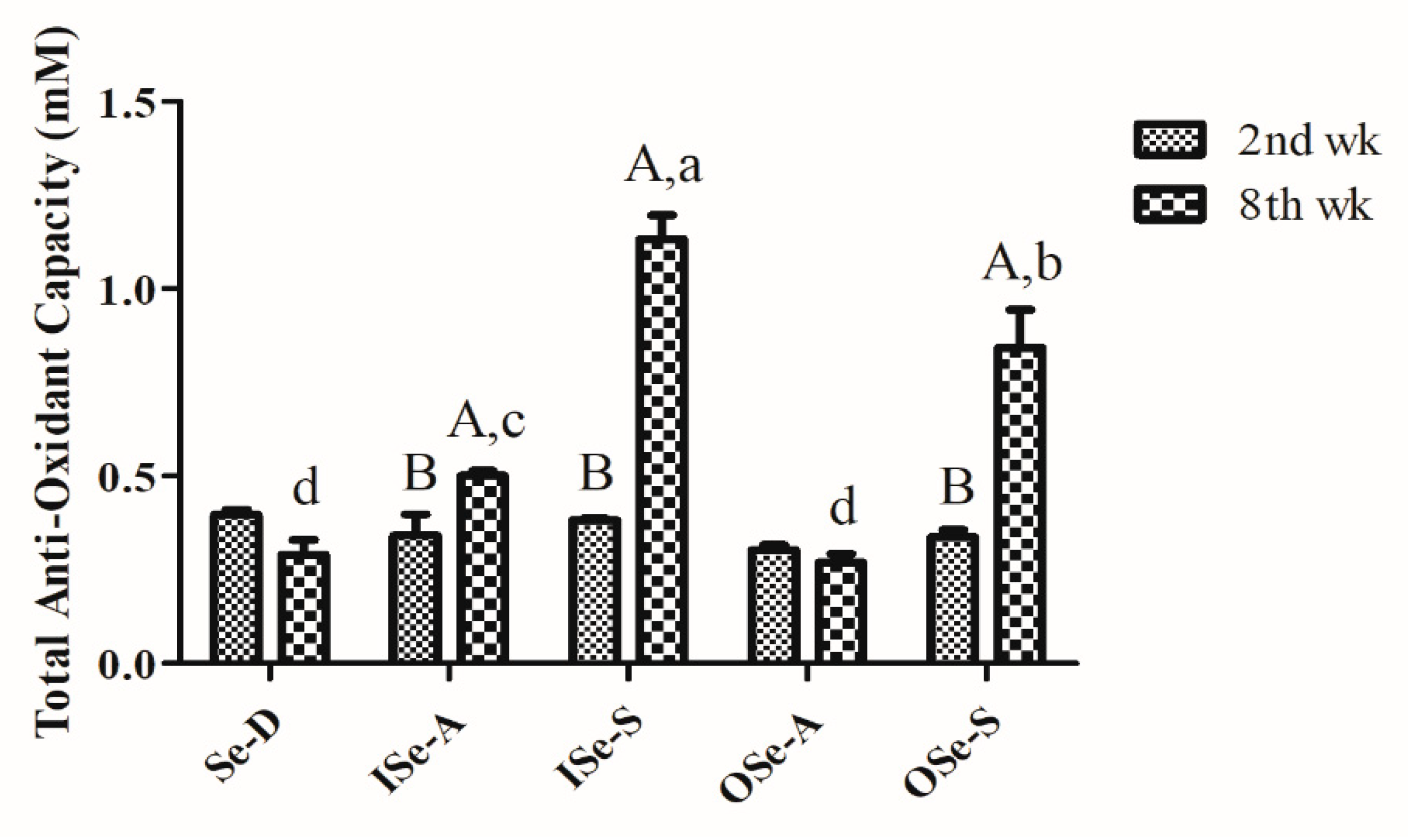
3.3. Dietary Se Supplementation Improves Blood Total Antioxidant Capacity in Aging Mice
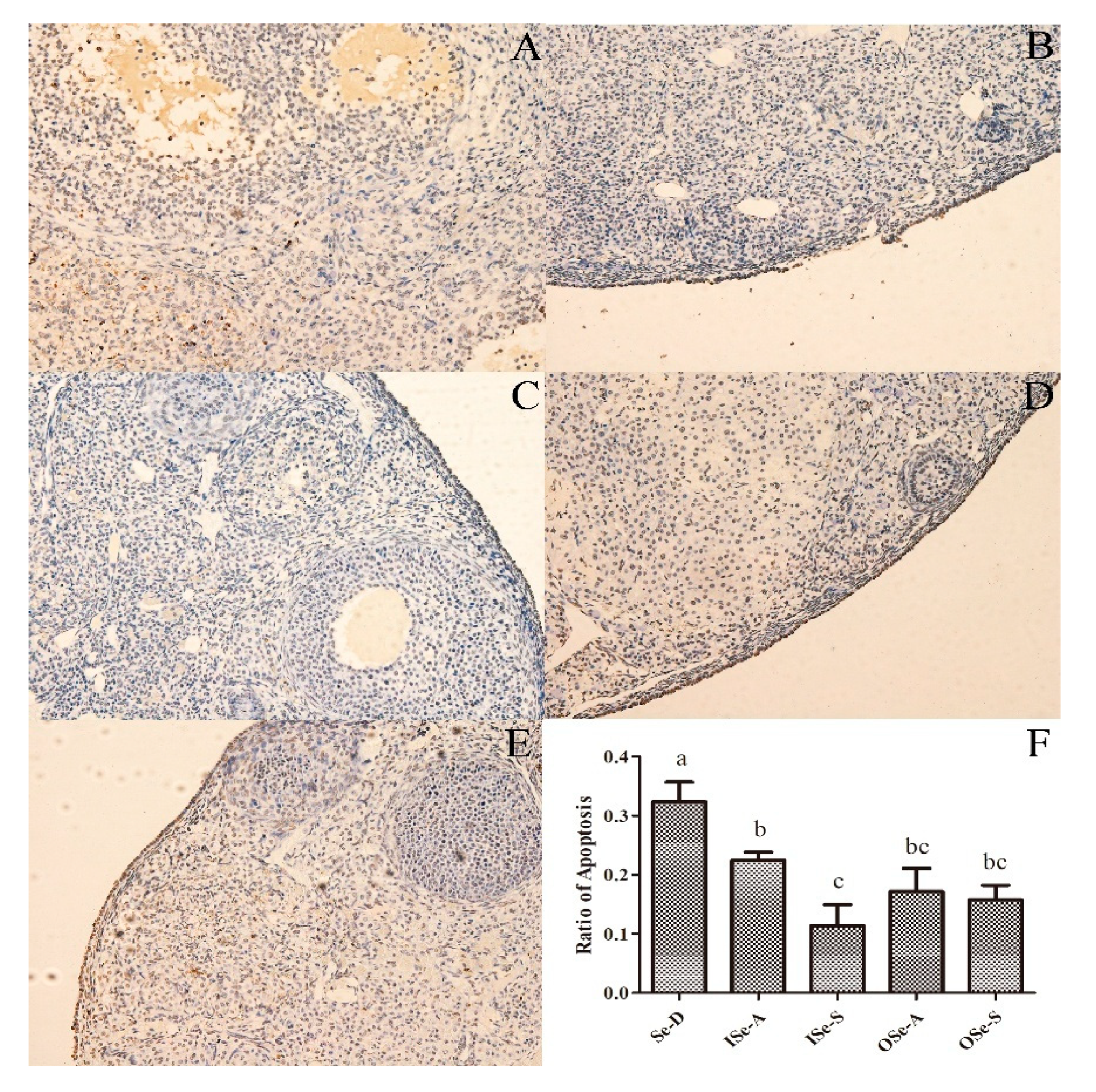
3.4. Dietary Se Supplementation Reduces Rate of Apoptosis in Ovarian Tissues of Aging Mice
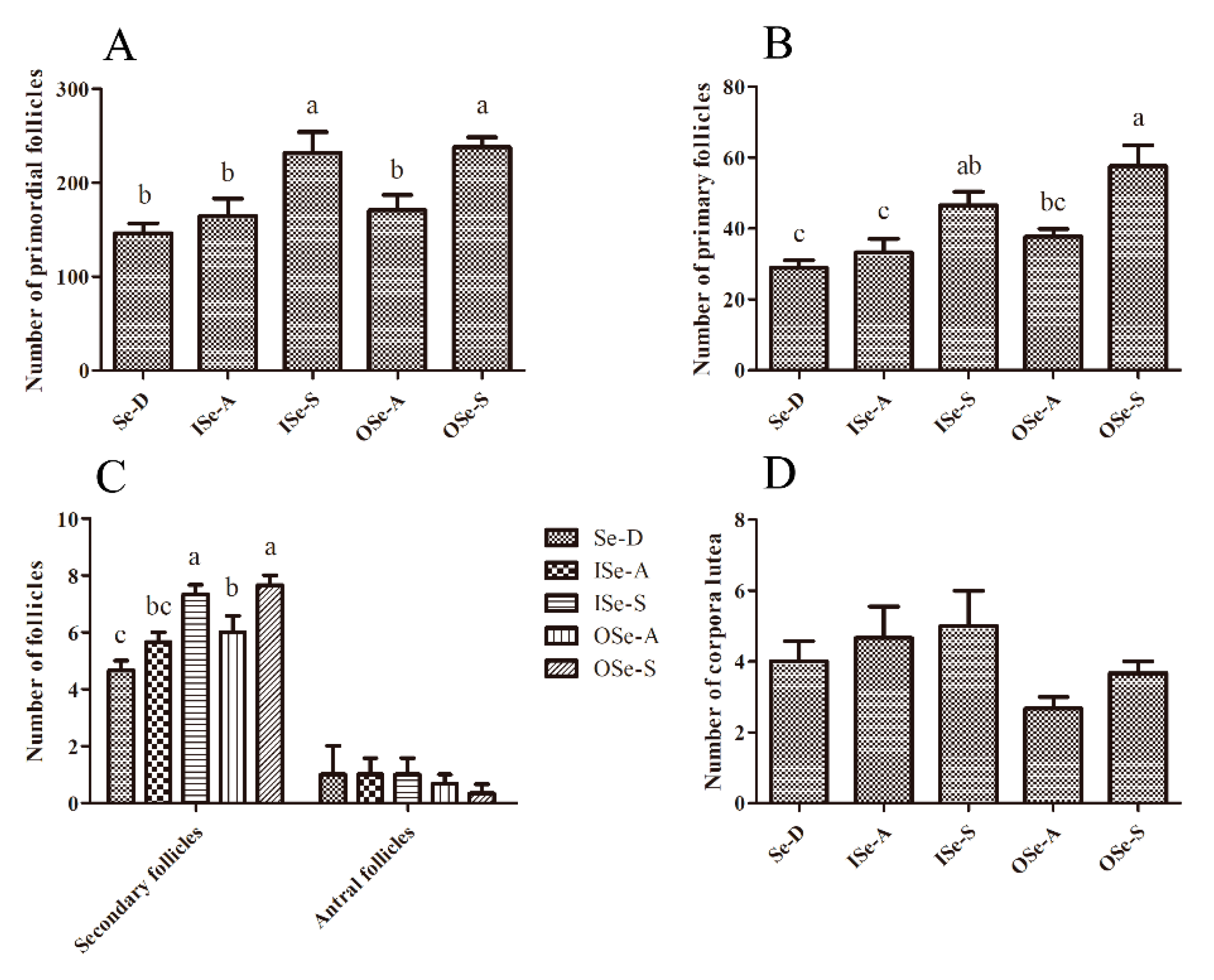
3.5. Effects of Dietary Se Supplementation on Maintenance of Follicle Reserves in Aging Ovaries
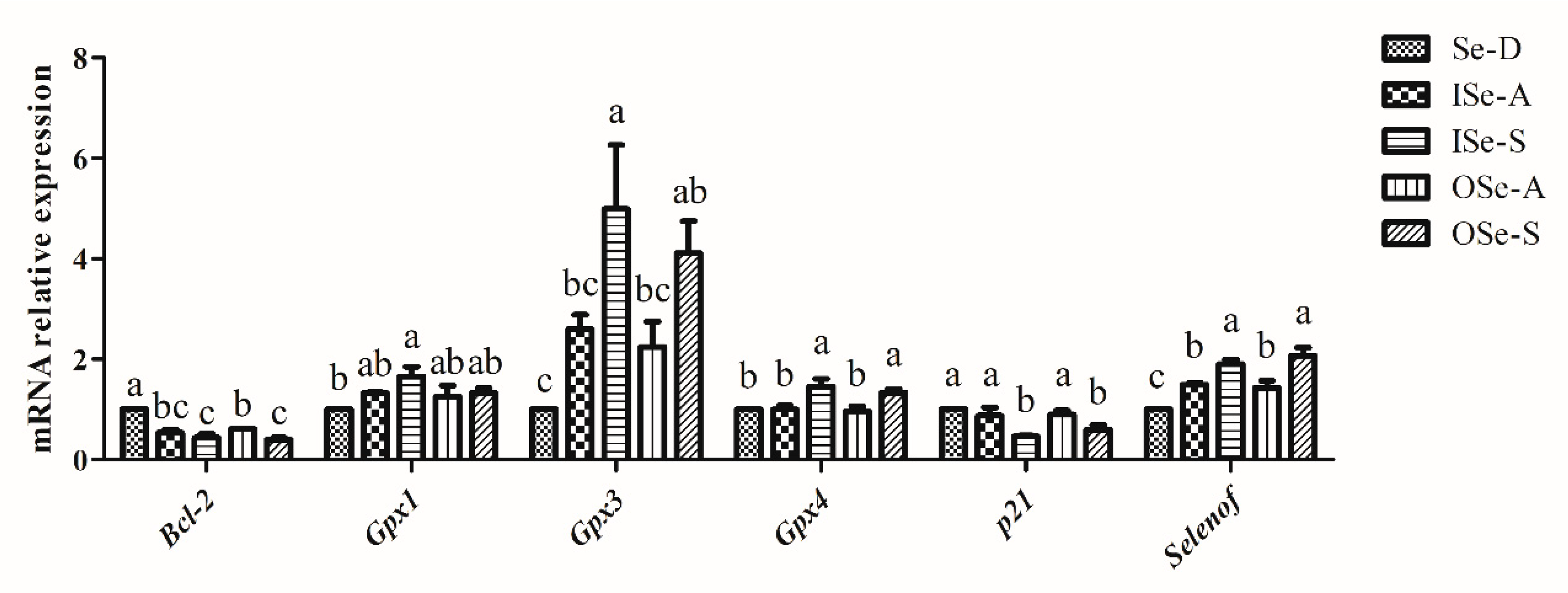
3.6. Effect of Dietary Se on Expression of Selenoprotein, Cell-Cycle, and Apoptosis-Related Genes in Ovaries of Aging Mice
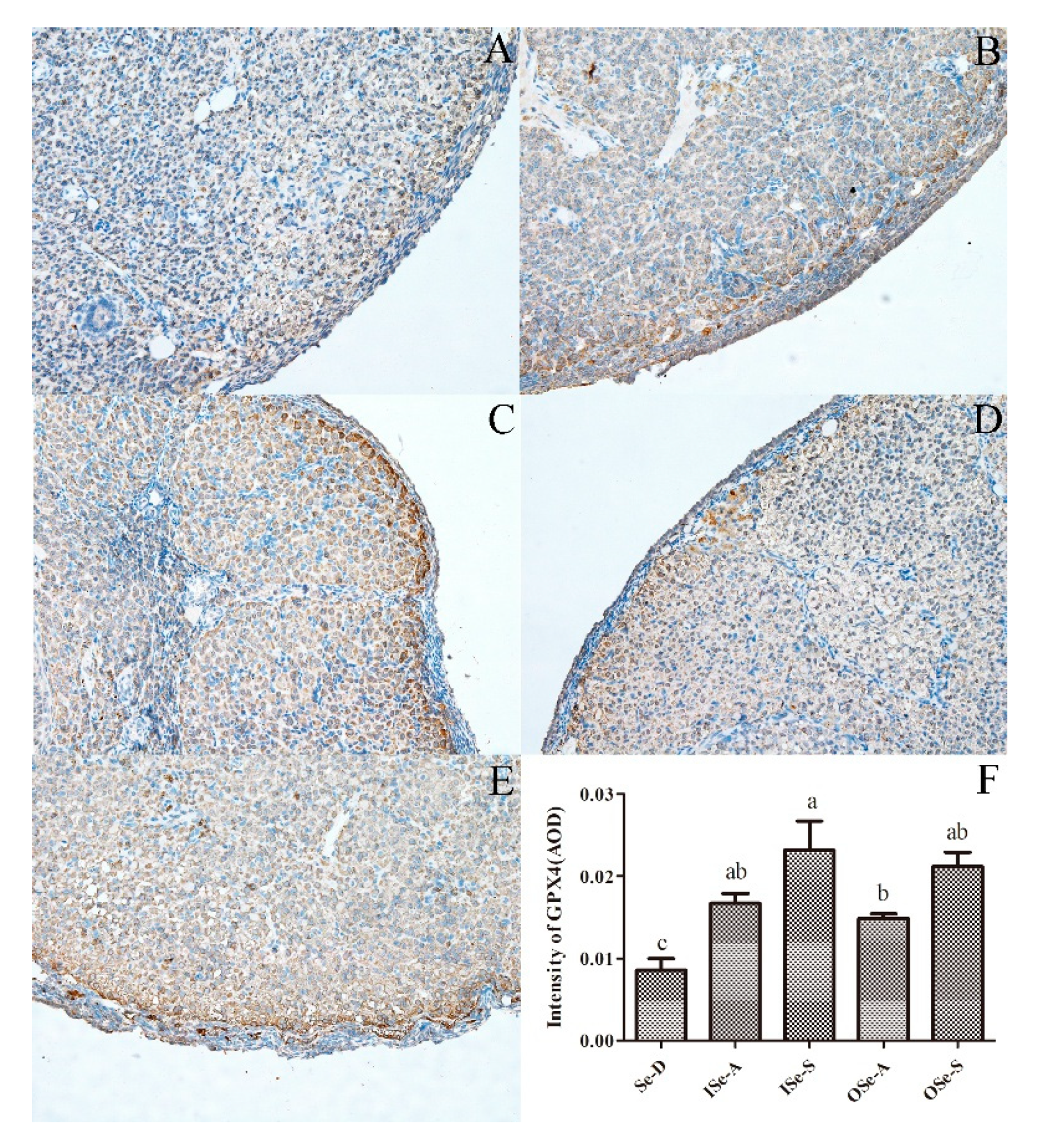
3.7. Expression of GPX4 protien in Ovarian Tissues of Aging Mice
3.8. Effect of Se Supplementation on In Vitro Developmental Potential of Embryos Resulting from GV Oocytes
4. Discussion
4.1. Blood Se Status and Total Antioxidant Capacity
4.2. Effect of Se on Follicle Quantity and Apoptosis in Ovarian Tissue of Aging Mice
4.3. Se Effect on In Vitro Embryo Developmental Potential
4.4. Expression of Gpx1, Gpx3, Gpx4, Selenof, Bcl-2, and p21
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Legrain, Y.; Touat-Hamici, Z.; Chavatte, L. Interplay between selenium levels, selenoprotein expression, and replicative senescence in WI-38 human fibroblasts. J. Biol. Chem. 2014, 289, 6299–6310. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Drake, A.C.; Thomas, N.O.; Ferguson, L.G.; Chappell, P.E.; Shay, K.P. Dietary resveratrol increases mid-life fecundity of female Nothobranchius guentheri. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2018, 208, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Duncan, F.E.; Gerton, J.L. Mammalian oogenesis and female reproductive aging. Aging 2018, 10, 162. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Yin, Y.; Ye, X.; Zeng, M.; Zhao, Q.; Keefe, D.L.; Liu, L. Resveratrol protects against age-associated infertility in mice. Hum. Reprod. 2013, 28, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.M.; Telfer, E.E.; Anderson, R.A. The ageing ovary and uterus: New biological insights. Hum. Reprod. Update 2012, 19, 67–83. [Google Scholar] [CrossRef]
- Liu, J.; Liu, M.; Ye, X.; Liu, K.; Huang, J.; Wang, L.; Ji, G.; Liu, N.; Tang, X.; Baltz, J.M. Delay in oocyte aging in mice by the antioxidant N-acetyl-L-cysteine (NAC). Hum. Reprod. 2012, 27, 1411–1420. [Google Scholar] [CrossRef]
- Lim, J.; Luderer, U. Oxidative damage increases and antioxidant gene expression decreases with aging in the mouse ovary. Biol. Reprod. 2011, 84, 775–782. [Google Scholar] [CrossRef]
- Song, C.; Peng, W.; Yin, S.; Zhao, J.; Fu, B.; Zhang, J.; Mao, T.; Wu, H.; Zhang, Y. Melatonin improves age-induced fertility decline and attenuates ovarian mitochondrial oxidative stress in mice. Sci. Rep. 2016, 6, 35165. [Google Scholar] [CrossRef]
- Qazi, I.; Angel, C.; Yang, H.; Pan, B.; Zoidis, E.; Zeng, C.-J.; Han, H.; Zhou, G.-B. Selenium, selenoproteins, and female reproduction: A review. Molecules 2018, 23, 3053. [Google Scholar] [CrossRef]
- Qazi, I.H.; Angel, C.; Yang, H.; Zoidis, E.; Pan, B.; Wu, Z.; Ming, Z.; Zeng, C.-J.; Meng, Q.; Han, H. Role of Selenium and Selenoproteins in Male Reproductive Function: A Review of Past and Present Evidences. Antioxidants 2019, 8, 268. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Burk, R.F.; Hill, K.E. Regulation of selenium metabolism and transport. Annu. Rev. Nutr. 2015, 35, 109–134. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.E.; Hurst, R. Selenium in human health and disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef] [PubMed]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular pathways and physiological roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef] [PubMed]
- Gladyshev, V.N.; Arnér, E.S.; Berry, M.J.; Brigelius-Flohé, R.; Bruford, E.A.; Burk, R.F.; Carlson, B.A.; Castellano, S.; Chavatte, L.; Conrad, M. Selenoprotein gene nomenclature. J. Biol. Chem. 2016, 291, 24036–24040. [Google Scholar] [CrossRef]
- Papp, L.V.; Lu, J.; Holmgren, A.; Khanna, K.K. From selenium to selenoproteins: Synthesis, identity, and their role in human health. Antioxid. Redox Signal. 2007, 9, 775–806. [Google Scholar] [CrossRef]
- Rayman, M.P. The use of high-selenium yeast to raise selenium status: How does it measure up? Br. J. Nutr. 2004, 92, 557–573. [Google Scholar] [CrossRef]
- Surai, P.F.; Fisinin, V.I. Selenium in Pig Nutrition and reproduction: Boars and semen quality—A Review. Asian Australas J. Anim. Sci. 2015, 28, 730. [Google Scholar] [CrossRef]
- Surai, P.F.; Fisinin, V.I. Selenium in livestock and other domestic animals. In Selenium; Springer: Berlin/Heidelberg, Germany, 2016; pp. 595–606. [Google Scholar]
- Surai, P.F.; Kochish, I.I.; Fisinin, V.I.; Juniper, D.T. Revisiting Oxidative Stress and the Use of Organic Selenium in Dairy Cow Nutrition. Animals 2019, 9, 462. [Google Scholar] [CrossRef]
- Yim, S.H.; Clish, C.B.; Gladyshev, V.N. Selenium Deficiency Is Associated with Pro-longevity Mechanisms. Cell Rep. 2019, 27, 2785–2797. [Google Scholar] [CrossRef]
- Baudry, J.; Kopp, J.F.; Boeing, H.; Kipp, A.P.; Schwerdtle, T.; Schulze, M.B. Changes of trace element status during aging: Results of the EPIC-Potsdam cohort study. Eur. J. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Olivieri, O.; Stanzial, A.M.; Girelli, D.; Trevisan, M.T.; Guarini, P.; Terzi, M.; Caffi, S.; Fontana, F.; Casaril, M.; Ferrari, S. Selenium status, fatty acids, vitamins A and E, and aging: The Nove Study. Am. J. Clin. Nutr. 1994, 60, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zeng, H.; Cheng, W.-H. Beneficial and paradoxical roles of selenium at nutritional levels of intake in healthspan and longevity. Free Radic. Biol. Med. 2018, 127, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.T.; Cao, L.; Mattson, E.; Witwer, K.W.; Cao, J.; Zeng, H.; He, X.; Combs, G.F., Jr.; Cheng, W.H. Opposing impacts on healthspan and longevity by limiting dietary selenium in telomere dysfunctional mice. Aging Cell 2017, 16, 125–135. [Google Scholar] [CrossRef]
- Hammad, G.; Legrain, Y.; Touat-Hamici, Z.; Duhieu, S.; Cornu, D.; Bulteau, A.-L.; Chavatte, L. Interplay between selenium levels and replicative senescence in WI-38 human fibroblasts: A proteomic approach. Antioxidants 2018, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Touat-Hamici, Z.; Legrain, Y.; Sonet, J.; Bulteau, A.-L.; Chavatte, L. Alteration of selenoprotein expression during stress and in aging. In Selenium; Springer: Berlin/Heidelberg, Germany, 2016; pp. 539–551. [Google Scholar]
- Ceko, M.; Hummitzsch, K.; Hatzirodos, N.; Bonner, W.; Aitken, J.; Russell, D.; Lane, M.; Rodgers, R.; Harris, H. X-Ray fluorescence imaging and other analyses identify selenium and GPX1 as important in female reproductive function. Metallomics 2014, 7, 71–82. [Google Scholar] [CrossRef]
- Paszkowski, T.; Traub, A.; Robinson, S.; McMaster, D. Selenium dependent glutathione peroxidase activity in human follicular fluid. Clin. Chim. Acta 1995, 236, 173–180. [Google Scholar] [CrossRef]
- Rayman, M.P. Is Adequate Selenium Important for Healthy Human Pregnancy? In Selenium; Springer: Berlin/Heidelberg, Germany, 2016; pp. 353–364. [Google Scholar]
- Rayman, M.P.; Searle, E.; Kelly, L.; Johnsen, S.; Bodman-Smith, K.; Bath, S.C.; Mao, J.; Redman, C.W. Effect of selenium on markers of risk of pre-eclampsia in UK pregnant women: A randomised, controlled pilot trial. Br. J. Nutr. 2014, 112, 99–111. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium and adverse health conditions of human pregnancy. In Selenium; Springer: New York, NY, USA, 2011; pp. 531–544. [Google Scholar]
- Mesdaghinia, E.; Rahavi, A.; Bahmani, F.; Sharifi, N.; Asemi, Z. Clinical and metabolic response to selenium supplementation in pregnant women at risk for intrauterine growth restriction: Randomized, double-blind, placebo-controlled trial. Biol. Trace Elem. Res. 2017, 178, 14–21. [Google Scholar] [CrossRef]
- Lewandowska, M.; Sajdak, S.; Lubiński, J. The Role of Early Pregnancy Maternal Selenium Levels on the Risk for Small-for-Gestational Age Newborns. Nutrients 2019, 11, 2298. [Google Scholar] [CrossRef]
- Lewandowska, M.; Sajdak, S.; Lubiński, J. Serum Selenium Level in Early Healthy Pregnancy as a Risk Marker of Pregnancy Induced Hypertension. Nutrients 2019, 11, 1028. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, G.; Isidori, A.M.; Moretti, C.; Di Dato, C.; Greco, E.; Ciolli, P.; Bonomi, M.; Petrone, L.; Fumarola, A.; Campagna, G.; et al. Selenium supplementation in the management of thyroid autoimmunity during pregnancy: Results of the “SERENA study”, a randomized, double-blind, placebo-controlled trial. Endocrine 2019. [Google Scholar] [CrossRef] [PubMed]
- Piekutowski, K.; Makarewicz, R.; Zachara, B. The antioxidative role of selenium in pathogenesis of cancer of the female reproductive system. Neoplasma 2007, 54, 374–378. [Google Scholar] [PubMed]
- Wu, W.; Wang, Q.; Yin, F.; Yang, Z.; Zhang, W.; Gabra, H.; Li, L. Identification of proteomic and metabolic signatures associated with chemoresistance of human epithelial ovarian cancer. Int. J. Oncol. 2016, 49, 1651–1665. [Google Scholar] [CrossRef]
- Agnani, D.; Camacho-Vanegas, O.; Camacho, C.; Lele, S.; Odunsi, K.; Cohen, S.; Dottino, P.; Martignetti, J.A. Decreased levels of serum glutathione peroxidase 3 are associated with papillary serous ovarian cancer and disease progression. J. Ovarian Res. 2011, 4, 18. [Google Scholar] [CrossRef]
- Falck, E.; Karlsson, S.; Carlsson, J.; Helenius, G.; Karlsson, M.; Klinga-Levan, K. Loss of glutathione peroxidase 3 expression is correlated with epigenetic mechanisms in endometrial adenocarcinoma. Cancer Cell Int. 2010, 10, 46. [Google Scholar] [CrossRef]
- Wang, F.-H.; Peng, X.; Chen, Y.; Wang, Y.; Yang, M.; Guo, M.-Y. Se Regulates the Contractile Ability of Uterine Smooth Musclevia Selenoprotein N, Selenoprotein T, and Selenoprotein Win Mice. Biol. Trace Elem. Res. 2019, 192, 196–205. [Google Scholar] [CrossRef]
- Zhou, J.; Li, C.; Gu, G.; Wang, Q.; Guo, M. Selenoprotein n was required for the regulation of selenium on the uterine smooth muscle contraction in mice. Biol. Trace Elem. Res. 2018, 183, 138–146. [Google Scholar] [CrossRef]
- Chen, C.-J.; Xiao, P.; Chen, Y.; Fang, R. Selenium Deficiency Affects Uterine Smooth Muscle Contraction Through Regulation of the RhoA/ROCK Signalling Pathway in Mice. Biol. Trace Elem. Res. 2019, 192, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Lan, D.; Li, J.; Lin, Y.; Li, M. Selenium supplementation during in vitro maturation enhances meiosis and developmental capacity of yak oocytes. Anim. Sci. J. 2018, 89, 298–306. [Google Scholar] [CrossRef]
- Yao, X.; Ei-Samahy, M.; Fan, L.; Zheng, L.; Jin, Y.; Zhang, G.; Liu, Z.; Wang, F. In vitro influence of selenium on the proliferation of and steroidogenesis in goat luteinized granulosa cells. Theriogenology 2018, 114, 70–80. [Google Scholar] [CrossRef]
- Grazul-Bilska, A.T.; Caton, J.S.; Arndt, W.; Burchill, K.; Thorson, C.; Borowczyk, E.; Bilski, J.J.; Redmer, D.A.; Reynolds, L.P.; Vonnahme, K.A. Cellular proliferation and vascularization in ovine fetal ovaries: Effects of undernutrition and selenium in maternal diet. Reproduction 2009, 137, 699. [Google Scholar] [CrossRef]
- Dalto, D.B.; Tsoi, S.; Dyck, M.K.; Matte, J.-J. Gene ontology analysis of expanded porcine blastocysts from gilts fed organic or inorganic selenium combined with pyridoxine. BMC Genom. 2018, 19, 836. [Google Scholar] [CrossRef]
- Atef, M.; Abd-Ellatif, R.; Emam, M.; Abo, R.E.G.; Amer, A.; Hafez, Y. Therapeutic potential of sodium selenite in letrozole induced polycystic ovary syndrome rat model: Targeting mitochondrial approach (selenium in PCOS). Arch. Biochem. Biophys. 2019, 671, 245–254. [Google Scholar] [CrossRef]
- Dalto, D.B.; Roy, M.; Audet, I.; Palin, M.-F.; Guay, F.; Lapointe, J.; Matte, J.J. Interaction between vitamin B6 and source of selenium on the response of the selenium-dependent glutathione peroxidase system to oxidative stress induced by oestrus in pubertal pig. J. Trace Elem. Med. Biol. 2015, 32, 21–29. [Google Scholar] [CrossRef]
- Mamon, M.A.C.; Ramos, G.B. Maternal selenium-supplementation at various stages of periconception period: Influence on murine blastocyst morphology and implantation status. J. Anim. Sci. Technol. 2017, 59, 7. [Google Scholar] [CrossRef]
- Mohammadi, S.; Movahedin, M.; Mowla, S.J. Up-regulation of CatSper genes family by selenium. Reprod. Biol. Endocrinol. 2009, 7, 126. [Google Scholar] [CrossRef]
- Wu, Z.; Pan, B.; Qazi, I.H.; Yang, H.; Guo, S.; Yang, J.; Zhang, Y.; Zeng, C.; Zhang, M.; Han, H.; et al. Melatonin Improves In Vitro Development of Vitrified-Warmed Mouse Germinal Vesicle Oocytes Potentially via Modulation of Spindle Assembly Checkpoint-Related Genes. Cells 2019, 8, 1009. [Google Scholar] [CrossRef]
- Zhou, J.C.; Zheng, S.; Mo, J.; Liang, X.; Xu, Y.; Zhang, H.; Gong, C.; Liu, X.L.; Lei, X.G. Dietary Selenium Deficiency or Excess Reduces Sperm Quality and Testicular mRNA Abundance of Nuclear Glutathione Peroxidase 4 in Rats. J. Nutr. 2017, 147, 1947–1953. [Google Scholar] [CrossRef]
- Myers, M.; Britt, K.L.; Wreford, N.G.; Ebling, F.J.; Kerr, J.B. Methods for quantifying follicular numbers within the mouse ovary. Reproduction 2004, 127, 569–580. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Rayman, M.P.; Bath, S.C.; Westaway, J.; Williams, P.; Mao, J.; Vanderlelie, J.J.; Perkins, A.V.; Redman, C.W. Selenium status in U.K. pregnant women and its relationship with hypertensive conditions of pregnancy. Br. J. Nutr. 2015, 113, 249–258. [Google Scholar] [CrossRef]
- Cao, L.; Zhang, L.; Zeng, H.; Wu, R.T.; Wu, T.-L.; Cheng, W.-H. Analyses of selenotranscriptomes and selenium concentrations in response to dietary selenium deficiency and age reveal common and distinct patterns by tissue and sex in telomere-dysfunctional mice. J. Nutr. 2017, 147, 1858–1866. [Google Scholar] [CrossRef]
- Gong, J.; Xiao, M. Effect of organic selenium supplementation on selenium status, oxidative stress, and antioxidant status in selenium-adequate dairy cows during the periparturient period. Biol. Trace Elem. Res. 2018, 186, 430–440. [Google Scholar] [CrossRef]
- Siqueira, I.R.; Fochesatto, C.; de Andrade, A.; Santos, M.; Hagen, M.; Bello-Klein, A.; Netto, C.A. Total antioxidant capacity is impaired in different structures from aged rat brain. Int. J. Dev. Neurosci. 2005, 23, 663–671. [Google Scholar] [CrossRef]
- Zeng, R.; Liang, Y.; Farooq, M.U.; Zhang, Y.; Ei, H.H.; Tang, Z.; Zheng, T.; Su, Y.; Ye, X.; Jia, X. Alterations in transcriptome and antioxidant activity of naturally aged mice exposed to selenium-rich rice. Environ. Sci. Pollut. Res. 2019, 26, 17834–17844. [Google Scholar] [CrossRef]
- Jamilian, M.; Mansury, S.; Bahmani, F.; Heidar, Z.; Amirani, E.; Asemi, Z. The effects of probiotic and selenium co-supplementation on parameters of mental health, hormonal profiles, and biomarkers of inflammation and oxidative stress in women with polycystic ovary syndrome. J. Ovarian Res. 2018, 11, 80. [Google Scholar] [CrossRef]
- Chao, Y.; Yu, B.; He, J.; Huang, Z.; Mao, X.; Luo, J.; Luo, Y.; Zheng, P.; Yu, J.; Chen, D. Effects of different levels of dietary hydroxy-analogue of selenomethionine on growth performance, selenium deposition and antioxidant status of weaned piglets. Arch. Anim. Nutr. 2019, 73, 374–383. [Google Scholar] [CrossRef]
- Chen, J.; Tian, M.; Guan, W.; Wen, T.; Yang, F.; Chen, F.; Zhang, S.; Song, J.; Ren, C.; Zhang, Y. Increasing selenium supplementation to a moderately-reduced energy and protein diet improves antioxidant status and meat quality without affecting growth performance in finishing pigs. J. Trace Elem. Med. Biol. 2019, 56, 38–45. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, F.; Guan, W.; Song, H.; Tian, M.; Cheng, L.; Shi, K.; Song, J.; Chen, F.; Zhang, S. Increasing selenium supply for heat-stressed or actively cooled sows improves piglet preweaning survival, colostrum and milk composition, as well as maternal selenium, antioxidant status and immunoglobulin transfer. J. Trace Elem. Med. Biol. 2019, 52, 89–99. [Google Scholar] [CrossRef]
- Feng, P.; Wei, J.R.; Zhang, Z.-G. Influence of selenium and fluoride on blood antioxidant capacity of rats. Exp. Toxicol. Pathol. 2012, 64, 565–568. [Google Scholar] [CrossRef]
- Feng, P.; Wei, J.; Zhang, Z. Intervention of selenium on chronic fluorosis-induced injury of blood antioxidant capacity in rats. Biol. Trace Elem. Res. 2011, 144, 1024–1031. [Google Scholar] [CrossRef]
- Dipali, S.S.; Ferreira, C.R.; Zhou, L.T.; Pritchard, M.T.; Duncan, F.E. Histologic analysis and lipid profiling reveal reproductive age-associated changes in peri-ovarian adipose tissue. Reprod. Biol. Endocrinol. 2019, 17, 46. [Google Scholar] [CrossRef]
- Basini, G.; Tamanini, C. Selenium stimulates estradiol production in bovine granulosa cells: Possible involvement of nitric oxide. Domest. Anim. Endocrinol. 2000, 18, 1–17. [Google Scholar] [CrossRef]
- Kamada, H.; Ikumo, H. Effect of selenium on cultured bovine luteal cells. Anim. Reprod. Sci. 1997, 46, 203–211. [Google Scholar] [CrossRef]
- Kerr, J.B.; Myers, M.; Anderson, R.A. The dynamics of the primordial follicle reserve. Reproduction 2013, 146, R205–R215. [Google Scholar] [CrossRef]
- Falone, S.; Santini, S.J.; Cordone, V.; Grannonico, M.; Cacchio, M.; Di Emidio, G.; Tatone, C.; Amicarelli, F. Regular and Moderate Exercise Counteracts the Decline of Antioxidant Protection but Not Methylglyoxal-Dependent Glycative Burden in the Ovary of Reproductively Aging Mice. Oxidative Med. Cell. Longev. 2016, 2016, 3837623. [Google Scholar] [CrossRef]
- Demirci, K.; Nazıroğlu, M.; Övey, İ.S.; Balaban, H. Selenium attenuates apoptosis, inflammation and oxidative stress in the blood and brain of aged rats with scopolamine-induced dementia. Metab. Brain Dis. 2017, 32, 321–329. [Google Scholar] [CrossRef]
- Fang, J.; Zheng, Z.; Yang, Z.; Peng, X.; Zuo, Z.; Cui, H.; Ouyang, P.; Shu, G.; Chen, Z.; Huang, C. Ameliorative effects of selenium on the excess apoptosis of the jejunum caused by AFB 1 through death receptor and endoplasmic reticulum pathways. Toxicol. Res. 2018, 7, 1108–1119. [Google Scholar] [CrossRef]
- Wang, J.; Liu, Z.; He, X.; Lian, S.; Liang, J.; Yu, D.; Sun, D.; Wu, R. Selenium deficiency induces duodenal villi cell apoptosis via an oxidative stress-induced mitochondrial apoptosis pathway and an inflammatory signaling-induced death receptor pathway. Metallomics 2018, 10, 1390–1400. [Google Scholar] [CrossRef]
- Khera, A.; Vanderlelie, J.J.; Holland, O.; Perkins, A.V. Overexpression of endogenous anti-oxidants with selenium supplementation protects trophoblast cells from reactive oxygen species-induced apoptosis in a Bcl-2-dependent manner. Biol. Trace Elem. Res. 2017, 177, 394–403. [Google Scholar] [CrossRef]
- Uhm, S.J.; Gupta, M.K.; Yang, J.H.; Lee, S.H.; Lee, H.T. Selenium improves the developmental ability and reduces the apoptosis in porcine parthenotes. Mol. Reprod. Dev. Inc. Gamete Res. 2007, 74, 1386–1394. [Google Scholar] [CrossRef]
- Mihalas, B.P.; Bromfield, E.G.; Sutherland, J.M.; De Iuliis, G.N.; McLaughlin, E.A.; Aitken, R.J.; Nixon, B. Oxidative damage in naturally aged mouse oocytes is exacerbated by dysregulation of proteasomal activity. J. Biol. Chem. 2018, 293, 18944–18964. [Google Scholar] [CrossRef]
- Silber, S.J.; Kato, K.; Aoyama, N.; Yabuuchi, A.; Skaletsky, H.; Fan, Y.; Shinohara, K.; Yatabe, N.; Kobayashi, T. Intrinsic fertility of human oocytes. Fertil. Steril. 2017, 107, 1232–1237. [Google Scholar] [CrossRef]
- Hill, K.E.; Lyons, P.R.; Burk, R.F. Differential regulation of rat liver selenoprotein mRNAs in selenium deficiency. Biochem. Biophys. Res. Commun. 1992, 185, 260–263. [Google Scholar] [CrossRef]
- Bösl, M.R.; Takaku, K.; Oshima, M.; Nishimura, S.; Taketo, M.M. Early embryonic lethality caused by targeted disruption of the mouse selenocysteine tRNA gene (Trsp). Proc. Natl. Acad. Sci. USA 1997, 94, 5531–5534. [Google Scholar] [CrossRef]
- Burk, R.F.; Olson, G.E.; Hill, K.E.; Winfrey, V.P.; Motley, A.K.; Kurokawa, S. Maternal-fetal transfer of selenium in the mouse. Faseb J. 2013, 27, 3249–3256. [Google Scholar] [CrossRef]
- Dalto, D.B.; Tsoi, S.; Audet, I.; Dyck, M.K.; Foxcroft, G.; Matte, J.J. Gene expression of porcine blastocysts from gilts fed organic or inorganic selenium and pyridoxine. Reproduction 2015, 149, 31–42. [Google Scholar] [CrossRef]
- Fortier, M.-E.; Audet, I.; Giguère, A.; Laforest, J.-P.; Bilodeau, J.-F.; Quesnel, H.; Matte, J. Effect of dietary organic and inorganic selenium on antioxidant status, embryo development, and reproductive performance in hyperovulatory first-parity gilts. J. Anim. Sci. 2012, 90, 231–240. [Google Scholar] [CrossRef]
- Dalto, D.B.; Audet, I.; Lapointe, J.; Matte, J.J. The importance of pyridoxine for the impact of the dietary selenium sources on redox balance, embryo development, and reproductive performance in gilts. J. Trace Elem. Med. Biol. 2016, 34, 79–89. [Google Scholar] [CrossRef]
- Abedelahi, A.; Salehnia, M.; Allameh, A. The effects of different concentrations of sodium selenite on the in vitro maturation of preantral follicles in serum-free and serum supplemented media. J. Assist. Reprod. Genet. 2008, 25, 483–488. [Google Scholar] [CrossRef]
- Abedelahi, A.; Salehnia, M.; Allameh, A.; Davoodi, D. Sodium selenite improves the in vitro follicular development by reducing the reactive oxygen species level and increasing the total antioxidant capacity and glutathione peroxide activity. Hum. Reprod. 2010, 25, 977–985. [Google Scholar] [CrossRef]
- Touat-Hamici, Z.; Legrain, Y.; Bulteau, A.-L.; Chavatte, L. Selective up-regulation of human selenoproteins in response to oxidative stress. J. Biol. Chem. 2014, 289, 14750–14761. [Google Scholar] [CrossRef]
- Zahia, T.-H.; Yona, L.; Anne-Laure, B.; Laurent, C. Selective up-regulation of human selenoproteins in response to oxidative stress. Free Radic. Biol. Med. 2014, 75, S25. [Google Scholar] [CrossRef]
- Akahoshi, N.; Anan, Y.; Hashimoto, Y.; Tokoro, N.; Mizuno, R.; Hayashi, S.; Yamamoto, S.; Shimada, K.-I.; Kamata, S.; Ishii, I. Dietary selenium deficiency or selenomethionine excess drastically alters organ selenium contents without altering the expression of most selenoproteins in mice. J. Nutr. Biochem. 2019, 69, 120–129. [Google Scholar] [CrossRef]
- Hatzirodos, N.; Hummitzsch, K.; Irving-Rodgers, H.F.; Harland, M.L.; Morris, S.E.; Rodgers, R.J. Transcriptome profiling of granulosa cells from bovine ovarian follicles during atresia. BMC Genom. 2014, 15, 40. [Google Scholar] [CrossRef]
- Xu, X.; Leng, J.-Y.; Gao, F.; Zhao, Z.-A.; Deng, W.-B.; Liang, X.-H.; Zhang, Y.-J.; Zhang, Z.-R.; Li, M.; Sha, A.-G. Differential expression and anti-oxidant function of glutathione peroxidase 3 in mouse uterus during decidualization. Febs Lett. 2014, 588, 1580–1589. [Google Scholar] [CrossRef]
- Hayashi, K.-G.; Ushizawa, K.; Hosoe, M.; Takahashi, T. Differential genome-wide gene expression profiling of bovine largest and second-largest follicles: Identification of genes associated with growth of dominant follicles. Reprod. Biol. Endocrinol. 2010, 8, 11. [Google Scholar] [CrossRef]
- Green, D.R. An element of life. Cell 2018, 172, 389–390. [Google Scholar] [CrossRef]
- Forcina, G.C.; Dixon, S.J. GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis. Proteomics 2019, 1800311. [Google Scholar] [CrossRef]
- Maiorino, M.; Conrad, M.; Ursini, F. GPx4, lipid peroxidation, and cell death: Discoveries, rediscoveries, and open issues. Antioxid. Redox Signal. 2018, 29, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Ingold, I.; Berndt, C.; Schmitt, S.; Doll, S.; Poschmann, G.; Buday, K.; Roveri, A.; Peng, X.; Freitas, F.P.; Seibt, T. Selenium utilization by GPX4 is required to prevent hydroperoxide-induced ferroptosis. Cell 2018, 172, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Seibt, T.M.; Proneth, B.; Conrad, M. Role of GPX4 in ferroptosis and its pharmacological implication. Free Radic. Biol. Med. 2019, 133, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Friedmann Angeli, J.P.; Krysko, D.V.; Conrad, M. Ferroptosis at the crossroads of cancer-acquired drug resistance and immune evasion. Nat. Rev. Cancer 2019, 19, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Doll, S.; Freitas, F.P.; Shah, R.; Aldrovandi, M.; da Silva, M.C.; Ingold, I.; Grocin, A.G.; Xavier da Silva, T.N.; Panzilius, E.; Scheel, C.H.; et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature 2019. [Google Scholar] [CrossRef]
- Wenzel, S.E.; Tyurina, Y.Y.; Zhao, J.; Croix, C.M.S.; Dar, H.H.; Mao, G.; Tyurin, V.A.; Anthonymuthu, T.S.; Kapralov, A.A.; Amoscato, A.A. PEBP1 wardens ferroptosis by enabling lipoxygenase generation of lipid death signals. Cell 2017, 171, 628–641. [Google Scholar] [CrossRef]
- Labunskyy, V.M.; Yoo, M.-H.; Hatfield, D.L.; Gladyshev, V.N. Sep15, a thioredoxin-like selenoprotein, is involved in the unfolded protein response and differentially regulated by adaptive and acute ER stresses. Biochemistry 2009, 48, 8458–8465. [Google Scholar] [CrossRef]
- Ren, B.; Huang, Y.; Zou, C.; Wu, Y.; Huang, Y.; Ni, J.; Tian, J. Transcriptional Regulation of Selenoprotein F by Heat Shock Factor 1 during Selenium Supplementation and Stress Response. Cells 2019, 8, 479. [Google Scholar] [CrossRef]
- Ren, B.; Liu, M.; Ni, J.; Tian, J. Role of selenoprotein F in protein folding and secretion: Potential involvement in human disease. Nutrients 2018, 10, 1619. [Google Scholar] [CrossRef]
- Yin, N.; Zheng, X.; Zhou, J.; Liu, H.; Huang, K. Knockdown of 15-kDa selenoprotein (Sep15) increases hLE cells’ susceptibility to tunicamycin-induced apoptosis. Jbic J. Biol. Inorg. Chem. 2015, 20, 1307–1317. [Google Scholar] [CrossRef]
- Juszczuk-Kubiak, E.; Bujko, K.; Cymer, M.; Wicińska, K.; Gabryszuk, M.; Pierzchała, M. Effect of inorganic dietary selenium supplementation on selenoprotein and lipid metabolism gene expression patterns in liver and loin muscle of growing lambs. Biol. Trace Elem. Res. 2016, 172, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Kale, J.; Osterlund, E.J.; Andrews, D.W. BCL-2 family proteins: Changing partners in the dance towards death. Cell Death Differ. 2018, 25, 65. [Google Scholar] [CrossRef] [PubMed]
- Pattingre, S.; Tassa, A.; Qu, X.; Garuti, R.; Liang, X.H.; Mizushima, N.; Packer, M.; Schneider, M.D.; Levine, B. Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 2005, 122, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Escobar, M.L.; Echeverria, O.M.; Palacios-Martínez, S.; Juárez-Chavero, S.; Sánchez-Sánchez, L.; Vázquez-Nin, G.H. Beclin 1 Interacts With Active Caspase-3 and Bax in Oocytes From Atretic Follicles in the Rat Ovary. J. Histochem. Cytochem. 2019, 0022155419881127. [Google Scholar] [CrossRef]
- Levine, B.; Klionsky, D.J. Development by self-digestion: Molecular mechanisms and biological functions of autophagy. Dev. Cell 2004, 6, 463–477. [Google Scholar] [CrossRef]
- Nakatogawa, H.; Suzuki, K.; Kamada, Y.; Ohsumi, Y. Dynamics and diversity in autophagy mechanisms: Lessons from yeast. Nat. Rev. Mol. Cell Biol. 2009, 10, 458. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Y.; Hamid, S.; Cai, J.; Liu, Q.; Li, H.; Zhao, R.; Wang, H.; Xu, S.; Zhang, Z. Interplay between autophagy and apoptosis in selenium deficient cardiomyocytes in chicken. J. Inorg. Biochem. 2017, 170, 17–25. [Google Scholar] [CrossRef]
- Sattar, H.; Yang, J.; Zhao, X.; Cai, J.; Liu, Q.; Ishfaq, M.; Yang, Z.; Chen, M.; Zhang, Z.; Xu, S. Selenoprotein-U (SelU) knockdown triggers autophagy through PI3K–Akt–mTOR pathway inhibition in rooster Sertoli cells. Metallomics 2018, 10, 929–940. [Google Scholar] [CrossRef]
- Kaushal, N.; Bansal, M. Inhibition of CDC2/Cyclin B1 in response to selenium-induced oxidative stress during spermatogenesis: Potential role of Cdc25c and p21. Mol. Cell. Biochem. 2007, 298, 139–150. [Google Scholar] [CrossRef]
| Groups | Number of GV Oocytes Collected | M II Oocytes (%) | Activated Oocytes (%) | No. of Embryos Developed to | ||
|---|---|---|---|---|---|---|
| Two-Cell Embryos (%) | Blastocysts (%) | Hatched Blastocysts (%) | ||||
| Se-D | 43 | 26 (60.51 ± 0.89) bc | 26 (100 ± 0) a | 17 (63.89 ± 12.73) | 5 (19.44 ± 4.81) b | 0 (0)d |
| ISe-A | 33 | 17 (51.52 ± 2.62) d | 11 (82.22 ± 1.92) c | 9 (65.00 ± 8.66) | 6 (36.67 ± 15.28) ab | 2 (13.33 ± 11.55) c |
| ISe-S | 59 | 34 (57.49 ± 1.10) cd | 28 (83.09 ± 5.28) bc | 18 (63.13 ± 11.77) | 12 (42.47 ± 4.33) a | 10 (35.73 ± 2.15) a |
| OSe-A | 66 | 46 (69.70 ± 6.94) a | 40 (87.29 ± 5.28) bc | 27 (67.40 ± 5.19) | 11(29.85 ± 6.37) ab | 7 (17.4 ± 3.49) bc |
| OSe-S | 58 | 38 (65.56 ± 5.09) ab | 34 (89.68 ± 3.43) b | 20 (58.59 ± 7.00) | 13 (37.88 ± 11.44) a | 9 (26.52 ± 1.31) ab |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.; Qazi, I.H.; Pan, B.; Angel, C.; Guo, S.; Yang, J.; Zhang, Y.; Ming, Z.; Zeng, C.; Meng, Q.; et al. Dietary Selenium Supplementation Ameliorates Female Reproductive Efficiency in Aging Mice. Antioxidants 2019, 8, 634. https://doi.org/10.3390/antiox8120634
Yang H, Qazi IH, Pan B, Angel C, Guo S, Yang J, Zhang Y, Ming Z, Zeng C, Meng Q, et al. Dietary Selenium Supplementation Ameliorates Female Reproductive Efficiency in Aging Mice. Antioxidants. 2019; 8(12):634. https://doi.org/10.3390/antiox8120634
Chicago/Turabian StyleYang, Haoxuan, Izhar Hyder Qazi, Bo Pan, Christiana Angel, Shichao Guo, Jingyu Yang, Yan Zhang, Zhang Ming, Changjun Zeng, Qingyong Meng, and et al. 2019. "Dietary Selenium Supplementation Ameliorates Female Reproductive Efficiency in Aging Mice" Antioxidants 8, no. 12: 634. https://doi.org/10.3390/antiox8120634
APA StyleYang, H., Qazi, I. H., Pan, B., Angel, C., Guo, S., Yang, J., Zhang, Y., Ming, Z., Zeng, C., Meng, Q., Han, H., & Zhou, G. (2019). Dietary Selenium Supplementation Ameliorates Female Reproductive Efficiency in Aging Mice. Antioxidants, 8(12), 634. https://doi.org/10.3390/antiox8120634







