Polyphenols from Lycium barbarum (Goji) Fruit European Cultivars at Different Maturation Steps: Extraction, HPLC-DAD Analyses, and Biological Evaluation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
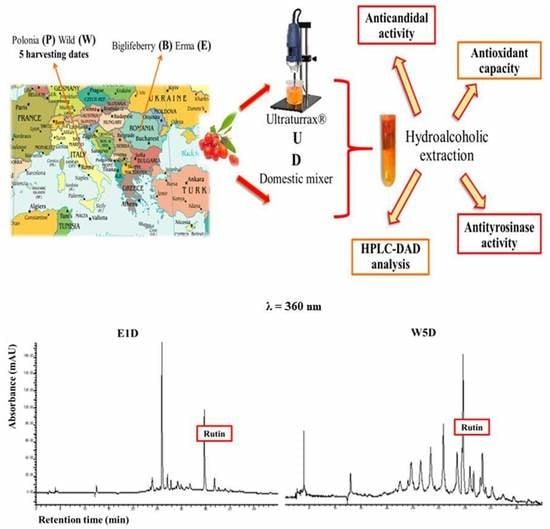
2.2. Sample Preparation
2.3. Extraction of Hydroalcoholic Fractions
2.4. HPLC Analysis
2.5. Total Phenolic Content (TPC)
2.6. Total Flavonoid Content (TFC)
2.7. Antioxidant Activity
2.7.1. DPPH Radical Scavenging Assay
2.7.2. Trolox Equivalent Antioxidant Capacity (TEAC) Assay
2.7.3. FRAP Assay
2.8. Tyrosinase Inhibitory Activity
2.9. Antifungal Susceptibility Testing
3. Results and Discussion
3.1. Polyphenols Extraction and HPLC Analysis
3.2. HPLC Analysis at 278 nm
3.3. Total Phenolic and Flavonoid Content
3.4. Antioxidant Activity
3.4.1. Trolox Equivalent Antioxidant Capacity (TEAC) Assay
3.4.2. DPPH Radical Scavenging Assay
3.4.3. FRAP Assay
3.5. Enzyme Inhibitory Activity
3.6. Anti-Candida Activity of Extracts
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yao, R.; Heinrich, M.; Weckerle, C.S. The genus Lycium as food and medicine: A botanical, ethnobotanical and historical review. J. Ethnopharmacol. 2018, 212, 50–66. [Google Scholar] [CrossRef]
- Kafkaletou, M.; Christopoulos, M.V.; Tsaniklidis, G.; Papadakis, I.; Ioannou, D.; Tzoutzoukou, C.; Tsantili, E. Nutritional value and consumer-perceived quality of fresh goji berries (Lycium barbarum L. and L. chinense L.) from plants cultivated in Southern Europe. Fruits 2018, 73, 5–12. [Google Scholar] [CrossRef]
- Dong, J.Z.; Wang, S.H.; Zhu, L.; Wang, Y. Analysis on the main active components of Lycium barbarum fruits and related environmental factors. J. Med. Plants Res. 2012, 6, 2276–2283. [Google Scholar]
- Potterat, O. Goji (Lycium barbarum and L. chinense): Phytochemistry, pharmacology and safety in the perspective of traditional uses and recent popularity. Planta Med. 2010, 76, 7–19. [Google Scholar] [CrossRef]
- Wang, C.C.; Chang, S.C.; Inbaraj, B.S.; Chen, B.H. Isolation of carotenoids, flavonoids and polysaccharides from Lycium barbarum L. and evaluation of antioxidant activity. Food Chem. 2010, 120, 184–192. [Google Scholar] [CrossRef]
- Amagase, H.; Farnsworth, N.R. A review of botanical characteristics, phytochemistry, clinical relevance in efficacy and safety of Lycium barbarum fruit (Goji). Food Res. Int. 2011, 44, 1702–1717. [Google Scholar] [CrossRef]
- Zhao, R.; Li, Q.; Xiao, B. Effect of Lycium barbarum polysaccharide on the improvement of insulin resistance in NIDDM rats. Yakugaku Zasshi 2005, 125, 981–988. [Google Scholar] [CrossRef]
- Masci, A.; Carradori, S.; Casadei, M.A.; Paolicelli, P.; Petralito, S.; Ragno, R.; Cesa, S. Lycium barbarum polysaccharides: Extraction, purification, structural characterisation and evidence about hypoglycaemic and hypolipidaemic effects. A review. Food Chem. 2018, 254, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Ma, C.; Li, Y.; Leung, K.S.; Jiang, Z.H.; Zhao, Z. Quantification of zeaxanthin dipalmitate and total carotenoids in Lycium fruits (Fructus Lycii). Plant Foods Hum. Nutr. 2005, 60, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Patsilinakos, A.; Ragno, R.; Carradori, S.; Petralito, S.; Cesa, S. Carotenoid content of Goji berries: CIELAB, HPLC-DAD analyses and quantitative correlation. Food Chem. 2018, 2698, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.; Chung, W.Y.; Wang, J.; Richelle, M.; Bucheli, P. Enhanced bioavailability of zeaxanthin in a milk-based formulation of wolfberry (Gou Qi Zi; Fructus barbarum L.). Br. J. Nutr. 2006, 96, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Pascolini, D.; Mariotti, S.P. Global estimates of visual impairment: 2010. Br. J. Ophthalmol. 2012, 96, 614–618. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Kim, H.; Kwon, O.; Chang, N. Associations between fruits, vegetables, vitamin A, β-carotene and flavonol dietary intake, and age-related macular degeneration in elderly women in Korea: The Fifth Korea National Health and Nutrition Examination Survey. Eur. J. Clin. Nutr. 2018, 72, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Donno, D.; Beccaro, G.L.; Mellano, M.G.; Cerutti, A.K.; Bounous, G. Goji berry fruit (Lycium spp.): Antioxidant compound fingerprint and bioactivity evaluation. J. Funct. Foods 2015, 18, 1070–1085. [Google Scholar] [CrossRef]
- Mocan, A.; Moldovan, C.; Zengin, G.; Bender, O.; Locatelli, M.; Simirgiotis, M.; Atalay, A.; Vodnar, D.C.; Rohn, S.; Crișan, G. UHPLC-QTOF-MS analysis of bioactive constituents from two Romanian Goji (Lycium barbarum L.) berries cultivars and their antioxidant, enzyme inhibitory, and real-time cytotoxicological evaluation. Food Chem. Toxicol. 2018, 115, 414–424. [Google Scholar] [CrossRef]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Schmitzer, V.; Slatnar, A.; Stampar, F.; Veberic, R. Composition of sugars, organic acids, and total phenolics in 25 wild or cultivated berry species. J. Food Sci. 2012, 77, C1064–C1070. [Google Scholar] [CrossRef]
- Sharma, S.; Narang, J.K.; Ali, J.; Baboota, S. Synergistic antioxidant action of vitamin E and rutin SNEDDS in ameliorating oxidative stress in a Parkinson’s disease model. Nanotechnol 2016, 27, 375101. [Google Scholar] [CrossRef]
- Fabre, G.; Bayach, I.; Berka, K.; Paloncýová, M.; Starok, M.; Rossi, C.; Duroux, J.L.; Otyepka, M.; Trouillas, P. Synergism of antioxidant action of vitamins E, C and quercetin is related to formation of molecular associations in biomembranes. Chem. Commun. 2015, 51, 7713–7716. [Google Scholar] [CrossRef]
- García-Borrón, J.C.; Solano, F. Molecular anatomy of tyrosinase and its related proteins: Beyond the histidine-bound metal catalytic center. Pigment Cell Res. 2002, 15, 162–173. [Google Scholar] [CrossRef]
- Mocan, A.; Schafberg, M.; Crisan, G.; Rohn, S. Determination of lignans and phenolic components of Schisandra chinensis (Turcz.) Baill. using HPLC-ESI-ToF-MS and HPLC-online TEAC: Contribution of individual components to overall antioxidant activity and comparison with traditional antioxidant assays. J. Funct. Food 2016, 24, 579–594. [Google Scholar] [CrossRef]
- Zolghadri, S.; Bahrami, A.; Hassan Khan, M.T.; Munoz-Munoz, J.; Garcia-Molina, F.; Garcia-Canovas, F.; Saboury, A.A. A comprehensive review on tyrosinase inhibitors. J. Enzyme Inhib. Med. Chem. 2019, 34, 279–309. [Google Scholar] [CrossRef] [PubMed]
- Dahech, I.; Farah, W.; Trigui, M.; Ben Hssouna, A.; Belghith, H.; Belghith, K.S.; Ben Abdallah, F. Antioxidant and antimicrobial activities of Lycium shawii fruits extract. Int. J. Biol. Macromol. 2013, 60, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Simonetti, G.; D’Auria, F.D.; Mulinacci, N.; Milella, R.A.; Antonacci, D.; Innocenti, M.; Pasqua, G. Phenolic content and in vitro antifungal activity of unripe grape extracts from agro-industrial wastes. Nat. Prod. Res. 2019, 33, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Simonetti, G.; Santamaria, A.R.; D’Auria, F.D.; Mulinacci, N.; Innocenti, M.; Cecchini, F.; Pericolini, E.; Gabrielli, E.; Panella, S.; Antonacci, D.; et al. Evaluation of anti-Candida activity of Vitis vinifera L. seed extracts obtained from wine and table cultivars. BioMed Res. Int. 2014, 2014, 127021. [Google Scholar] [CrossRef]
- Simonetti, G.; Valletta, A.; Kolesova, O.; Pasqua, G. Plant products with antifungal activity: From field to biotechnology strategies. In Natural Products as Source of Molecules with Therapeutic Potential; Springer: Cham, Switzerland, 2018; pp. 35–71. [Google Scholar]
- Can Baser, K.H. Biological and pharmacological activities of carvacrol and carvacrol bearing essential oils. Curr. Pharm. Des. 2008, 14, 3106–3119. [Google Scholar] [CrossRef]
- Chaillot, J.; Tebbji, F.; Remmal, A.; Boone, C.; Brown, G.W.; Bellaoui, M.; Sellam, A. The monoterpene carvacrol generates endoplasmic reticulum stress in the pathogenic fungus Candida albicans. Antimicrob. Agents Chemother. 2015, 59, 4584–4592. [Google Scholar] [CrossRef]
- Locatelli, M.; Ferrante, C.; Carradori, S.; Secci, D.; Leporini, L.; Chiavaroli, A.; Leone, S.; Recinella, L.; Orlando, G.; Martinotti, S.; et al. Optimization of aqueous extraction and biological activity of Harpagophytum procumbens root on ex vivo rat colon inflammatory model. Phytother. Res. 2017, 31, 937–944. [Google Scholar] [CrossRef]
- Inbaraj, B.S.; Lu, H.; Kao, T.H.; Chen, B.H. Simultaneous determination of phenolic acids and flavonoids in Lycium barbarum Linnaeus by HPLC–DAD–ESI-MS. J. Pharm. Biomed. Anal. 2010, 51, 549–556. [Google Scholar] [CrossRef]
- Mocan, A.; Crişan, G.; Vlase, L.; Crişan, O.; Vodnar, D.C.; Raita, O.; Gheldiu, A.M.; Toiu, A.; Oprean, R.; Tilea, I. Comparative studies on polyphenolic composition, antioxidant and antimicrobial activities of Schisandra chinensis leaves and fruits. Molecules 2014, 19, 15162–15179. [Google Scholar] [CrossRef]
- Martins, N.; Barros, L.; Dueñas, M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Characterization of phenolic compounds and antioxidant properties of Glycyrrhiza glabra L. rhizomes and roots. RSC Adv. 2015, 5, 26991–26997. [Google Scholar] [CrossRef]
- Damiano, S.; Forino, M.; De, A.; Vitali, L.A.; Lupidi, G.; Taglialatela-Scafati, O. Antioxidant and antibiofilm activities of secondary metabolites from Ziziphus jujuba leaves used for infusion preparation. Food Chem. 2017, 230, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Yamashita, D.; Takeda, Y.; Yonemori, S. Screening for tyrosinase inhibitors among extracts of seashore plants and identification of potent inhibitors from Garcinia subelliptica. Biosci. Biotechnol. Biochem. 2005, 69, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, 4th ed.; CLSI document M27; CLSI: Wayne, PA, USA, 2017. [Google Scholar]
- Le, K.; Chiu, F.; Ng, K. Identification and quantification of antioxidants in Fructus lycii. Food Chem. 2007, 105, 353–363. [Google Scholar] [CrossRef]
- De Moura, C.; dos Reis, A.S.; da Silva, L.D.; de Lima, V.A.; Oldoni, T.L.C.; Pereira, C.; Carpes, S.T. Optimization of phenolic compounds extraction with antioxidant activity from açaí, blueberry and goji berry using response surface methodology. Emir. J. Food Agr. 2018, 30, 180–189. [Google Scholar]
- Zhang, Q.; Chen, W.; Zhao, J.; Xi, W. Functional constituents and antioxidant activities of eight Chinese native goji genotypes. Food Chem. 2016, 200, 230–236. [Google Scholar] [CrossRef]
- Dong, J.Z.; Lu, D.Y.; Wang, Y. Analysis of flavonoids from leaves of cultivated Lycium barbarum L. Plant Foods Hum. Nutr. 2009, 64, 199–204. [Google Scholar] [CrossRef]
- Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L.D.; Hidalgo, F.J.; Miyashita, K.; Camp, J.V.; Alasalvar, C.; Ismail, A.B.; et al. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem. 2018, 264, 471–475. [Google Scholar] [CrossRef]
- Ceccarini, M.R.; Vannini, S.; Cataldi, S.; Moretti, M.; Villarini, M.; Fioretti, B.; Albi, E.; Beccari, T.; Codini, M. In vitro protective effects of Lycium barbarum berries cultivated in Umbria (Italy) on human hepatocellular carcinoma cells. Biomed Res. Int. 2016, 2016, 7529521. [Google Scholar] [CrossRef]
- Abeywickrama, G.; Debnath, S.C.; Ambigaipalan, P.; Shahidi, F. Phenolics of selected cranberry genotypes (Vaccinium macrocarpon Ait.) and their antioxidant efficacy. J. Agric. Food Chem. 2016, 64, 9342–9351. [Google Scholar] [CrossRef]
- Abdennacer, B.; Karim, M.; Yassine, M.; Nesrine, R.; Mouna, D.; Mohamed, B. Determination of phytochemicals and antioxidant activity of methanol extracts obtained from the fruit and leaves of Tunisian Lycium intricatum Boiss. Food Chem. 2015, 174, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Food 2015, 18, 757–781. [Google Scholar] [CrossRef]
- Orhan, D.D.; Ozçelik, B.; Ozgen, S.; Ergun, F. Antibacterial, antifungal, and antiviral activities of some flavonoids. Microbiol. Res. 2010, 165, 496–504. [Google Scholar] [CrossRef] [PubMed]
| Compound | Concentration (μg/mg ± SD) | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P1D | P2D | P3D | P4D | P5D | P1U | P2U | P3U | P4U | P5U | W1D | W2D | W3D | W4D | W5D | W1U | W1U | W3U | W4U | W5U | E1D | B1D | |
| Gallic acid | 0.77 ± 0.07 | 0.03 ± 0.01 | tr | |||||||||||||||||||
| Catechin | 0.23 ± 0.01 | 0.23 ± 0.01 | tr | 0.37 ± 0.01 | 0.03 ± 0.01 | 0.31 ± 0.01 | 0.25 ± 0.01 | |||||||||||||||
| Chlorogenic acid | 0.19 ± 0.01 | 0.16 ± 0.01 | 0.55 ± 0.02 | 0.54 ± 0.02 | 0.56 ± 0.01 | 0.11 ± 0.01 | 0.13 ± 0.01 | 0.57 ± 0.02 | 0.37 ± 0.02 | 0.47 ± 0.02 | 0.10 ± 0.01 | 0.77 ± 0.03 | 0.43 ± 0.02 | 0.22 ± 0.01 | 0.12 ± 0.01 | 0.28 ± 0.01 | 0.43 ± 0.02 | 0.45 ± 0.02 | ||||
| Epicatechin | 0.07 ± 0.01 | |||||||||||||||||||||
| Syringic acid | 0.06 ± 0.01 | 0.14 ± 0.01 | 0.09 ± 0.01 | 0.04 ± 0.01 | 0.21 ± 0.01 | 0.27 ± 0.01 | ||||||||||||||||
| 3-Hydroxy benzoic acid | 0.08 ± 0.01 | |||||||||||||||||||||
| Sinapinic acid | tr | 0.03 ± 0.01 | 0.03 ± 0.01 | 0.03 ± 0.01 | tr | 0.03 ± 0.01 | 0.03 ± 0.01 | tr | 0.03 ± 0.01 | 0.03 ± 0.01 | tr | 0.03 ± 0.01 | 0.03 ± 0.01 | |||||||||
| Naringenin | 0.06 ± 0.01 | |||||||||||||||||||||
| Carvacrol | 0.03 ± 0.01 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.08 ± 0.01 | 0.05 ± 0.01 | 0.04 ± 0.01 | 0.05 ± 0.01 | 0.04 ± 0.01 | 0.04 ± 0.01 | 0.06 ± 0.01 | 0.05 ± 0.01 | 0.04 ± 0.01 | 0.07 ± 0.01 | 0.04 ± 0.01 | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.04 ± 0.01 | 0.05 ± 0.01 | 0.23 ± 0.02 | |
| Total (μg/mg) | 0.03 | 1.07 | 0.21 | 0.63 | 0.62 | 0.93 | 0.16 | 0.17 | 0.95 | 0.50 | 0.51 | 0.16 | 0.18 | 0.93 | 0.98 | 0.27 | 0.24 | 0.54 | 1.09 | 0.78 | 0.23 | |
| Sample | HA Extract (mg/g dw) | Rutin (± 3%) (µg/g dw) | Flavonoids (± 5%) (µg/g dw) | Rutin (± 3%) (µg/mg Extract) | Flavonoids (± 5%) (µg/mg Extract) |
|---|---|---|---|---|---|
| P1D | 467 | 210 | 790 | 0.45 | 1.69 |
| P2D | 548 | 263 | 661 | 0.48 | 1.28 |
| P3D | 528 | 360 | 658 | 0.68 | 1.25 |
| P4D | 410 | 330 | 950 | 0.80 | 2.31 |
| P5D | 340 | 205 | 564 | 0.60 | 1.66 |
| W1D | 422 | 371 | 995 | 0.88 | 2.36 |
| W2D | 472 | 274 | 699 | 0.58 | 1.48 |
| W3D | 493 | 256 | 874 | 0.52 | 1.77 |
| W4D | 430 | 268 | 1033 | 0.62 | 2.40 |
| W5D | 448 | 336 | 925 | 0.75 | 2.06 |
| P1U | 450 | 184 | 591 | 0.41 | 1.31 |
| P2U | 558 | 335 | 735 | 0.60 | 1.32 |
| P3U | 589 | 316 | 705 | 0.54 | 1.20 |
| P4U | 414 | 317 | 1598 | 0.77 | 3.86 |
| P5U | 345 | 238 | 428 | 0.69 | 1.24 |
| W1U | 456 | 405 | 1134 | 0.89 | 2.49 |
| W2U | 506 | 309 | 642 | 0.61 | 1.27 |
| W3U | 494 | 242 | 1237 | 0.49 | 2.50 |
| W4U | 464 | 198 | 1183 | 0.43 | 2.55 |
| W5U | 413 | 250 | 374 | 0.60 | 0.90 |
| E1D | 528 | 288 | 1605 | 0.54 | 3.04 |
| B1D | 420 | 255 | 752 | 0.61 | 1.79 |
| Sample | TPC (mg GAE/g dw Extract) | TFC (mg QE/g dw Extract) | FRAP (mg TE/g dw Extract) | DPPH Scavenging (mg TE/g dw Extract) | ABTS Scavenging (mg TE/g dw Extract) | Tyrosinase Inhibition (mg KAE/g dw Extract) |
|---|---|---|---|---|---|---|
| P1D | 19.22 ± 0.52 | 4.05 ± 0.61 | 23.84 ± 0.31 | 26.29 ± 1.65 | 40.45 ± 0.26 | 2.12 ± 0.25 |
| P2D | 17.09 ± 1.59 | 3.58 ± 0.11 | 19.47 ± 0.99 | 24.62 ± 0.24 | 34.79 ± 1.13 | 2.50 ± 0.86 |
| P3D | 18.09 ± 0.13 | 3.87 ± 0.76 | 20.80 ± 0.74 | 16.77 ± 4.38 | 36.85 ± 0.39 | 2.39 ± 0.53 |
| P4D | 22.64 ± 0.97 | 2.89 ± 0.13 | 36.63 ± 0.84 | 36.47 ± 1.42 | 44.97 ± 0.48 | 9.69 ± 0.19 |
| P5D | 19.50 ± 0.67 | 2.64 ± 0.17 | 33.28 ± 2.03 | 35.58 ± 2.14 | 44.91 ± 0.61 | 6.54 ± 0.78 |
| W1D | 12.28 ± 1.87 | 3.18 ± 0.21 | 16.76 ± 1.03 | 18.66 ± 2.22 | 31.88 ± 0.58 | 2.59 ± 0.35 |
| W2D | 20.29 ± 0.88 | 4.26 ± 0.13 | 21.24 ± 1.46 | 16.77 ± 2.83 | 39.10 ± 1.68 | 3.17 ± 0.83 |
| W3D | 15.33 ± 0.16 | 3.45 ± 0.13 | 20.96 ± 0.75 | 17.99 ± 3.98 | 37.71 ± 0.33 | 3.33 ± 0.70 |
| W4D | 15.27 ± 0.69 | 2.39 ± 0.06 | 29.90 ± 1.57 | 32.97 ± 2.60 | 37.84 ± 0.74 | 8.03 ± 0.85 |
| W5D | 19.54 ± 0.70 | 3.56 ± 0.24 | 32.14 ± 3.49 | 40.32 ± 4.01 | 42.03 ± 0.20 | 8.42 ± 0.75 |
| P1U | 18.86 ± 0.45 | 3.42 ± 0.10 | 28.54 ± 0.59 | 32.58 ± 2.70 | 40.67 ± 1.16 | 5.05 ± 0.58 |
| P2U | 18.39 ± 1.57 | 3.87 ± 0.26 | 25.93 ± 1.79 | 21.78 ± 3.53 | 39.10 ± 0.46 | 3.50 ± 0.29 |
| P3U | 19.59 ± 0.73 | 3.61 ± 0.14 | 23.86 ± 0.59 | 21.11 ± 3.94 | 38.02 ± 0.63 | 3.72 ± 0.63 |
| P4U | 7.69 ± 1.78 | 11.03 ± 1.24 | 23.89 ± 2.09 | 10.75 ± 0.06 | 45.24 ± 4.69 | 57.60 ± 0.01 |
| P5U | 17.71 ± 0.93 | 2.77 ± 0.17 | 30.86 ± 0.86 | 26.23 ± 2.15 | 39.43 ± 0.46 | 6.48 ± 0.16 |
| W1U | 17.43 ± 0.30 | 4.02 ± 0.02 | 20.90 ± 0.49 | 19.89 ± 1.65 | 37.57 ± 1.13 | 4.33 ± 0.72 |
| W2U | 11.83 ± 0.67 | 3.13 ± 0.24 | 15.71 ± 0.05 | 12.20 ± 2.53 | 27.81 ± 0.46 | 4.66 ± 0.99 |
| W3U | 17.00 ± 0.65 | 3.24 ± 0.12 | 20.49 ± 0.62 | 17.99 ± 0.77 | 35.96 ± 0.38 | 1.56 ± 0.74 |
| W4U | 23.49 ± 3.36 | 10.54 ± 0.12 | 51.93 ± 2.97 | 14.98 ± 4.40 | 63.58 ± 13.07 | 62.79 ± 0.66 |
| W5U | 15.10 ± 1.41 | 2.58 ± 0.09 | 28.59 ± 2.55 | 29.13 ± 0.94 | 35.76 ± 0.30 | 9.30 ± 0.44 |
| E1D | 5.76 ± 0.24 | 10.47 ± 1.83 | 23.00 ± 3.66 | 2.18 ± 0.84 | 51.14 ± 4.27 | 59.49 ± 1.33 |
| B1D | 3.31 ± 0.66 | 9.29 ± 0.60 | 21.56 ± 2.28 | na | 76.78 ± 7.91 | 49.35 ± 4.33 |
| Sample | MIC (μg/mL)* at 24 h | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Candida albicans ATCC 24433 | Candida albicans ATCC 10261 | Candida albicans ATCC 90028 | |||||||
| 50 | 90 | 100 | 50 | 90 | 100 | 50 | 90 | 100 | |
| P1D | 125 | 250 | 500 | 281.25 | 312.5 | 625 | 93.75 | 250 | 500 |
| P2D | 250 | 250 | 500 | 375 | 375 | 750 | 125 | 250 | 500 |
| P3D | 250 | 250 | 500 | 375 | 500 | 1000 | 187.5 | 250 | 500 |
| P4D | 125 | 1000 | >1000 | 500 | 1000 | >1000 | 500 | 1000 | >1000 |
| P5D | 250 | 1000 | >1000 | 500 | 1000 | >1000 | 500 | 1000 | >1000 |
| P1U | 125 | 187.5 | 375 | 375 | 500 | 1000 | 250 | 375 | 750 |
| P2U | 250 | 250 | 500 | 375 | 500 | 1000 | 250 | 250 | 500 |
| P3U | 250 | 250 | 500 | 375 | 500 | 1000 | 250 | 250 | 500 |
| P4U | 125 | 250 | 1000 | 250 | 500 | 1000 | 250 | 500 | 1000 |
| P5U | 250 | 500 | 1000 | 250 | 500 | 1000 | 500 | 1000 | 1000 |
| W1D | 250 | 250 | 500 | 500 | 500 | 1000 | 187.5 | 250 | 500 |
| W2D | 250 | 250 | 500 | 500 | 500 | 1000 | 187.5 | 250 | 500 |
| W3D | 250 | 250 | 500 | 500 | 500 | 1000 | 250 | 250 | 500 |
| W4D | 125 | 1000 | >1000 | 500 | 1000 | >1000 | 500 | 1000 | >1000 |
| W5D | 125 | 1000 | >1000 | 500 | 1000 | >1000 | 500 | 1000 | >1000 |
| W1U | 187.5 | 250 | 500 | 500 | 500 | 1000 | 250 | 375 | 500 |
| W2U | 250 | 375 | 500 | 500 | 500 | 1000 | 375 | 375 | 750 |
| W3U | 250 | 250 | 500 | 500 | 500 | 1000 | 125 | 250 | 500 |
| W4U | 250 | 250 | >1000 | 500 | 1000 | >1000 | 500 | 1000 | >1000 |
| W5U | 250 | 1000 | >1000 | 500 | 1000 | >1000 | 500 | 1000 | >1000 |
| E1D | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 |
| B1D | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 | >1000 |
| Rutin | 64 | >64 | >64 | 64 | >64 | >64 | 64 | >64 | >64 |
| Carvacrol | 0.25 | 1 | 2 | 0.25 | 1 | 2 | 0.125 | 0.25 | 0.5 |
| Fluconazole | 0.5 | 2 | 8 | 1 | 4 | >64 | 0.5 | 2 | 8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mocan, A.; Cairone, F.; Locatelli, M.; Cacciagrano, F.; Carradori, S.; Vodnar, D.C.; Crișan, G.; Simonetti, G.; Cesa, S. Polyphenols from Lycium barbarum (Goji) Fruit European Cultivars at Different Maturation Steps: Extraction, HPLC-DAD Analyses, and Biological Evaluation. Antioxidants 2019, 8, 562. https://doi.org/10.3390/antiox8110562
Mocan A, Cairone F, Locatelli M, Cacciagrano F, Carradori S, Vodnar DC, Crișan G, Simonetti G, Cesa S. Polyphenols from Lycium barbarum (Goji) Fruit European Cultivars at Different Maturation Steps: Extraction, HPLC-DAD Analyses, and Biological Evaluation. Antioxidants. 2019; 8(11):562. https://doi.org/10.3390/antiox8110562
Chicago/Turabian StyleMocan, Andrei, Francesco Cairone, Marcello Locatelli, Francesco Cacciagrano, Simone Carradori, Dan C. Vodnar, Gianina Crișan, Giovanna Simonetti, and Stefania Cesa. 2019. "Polyphenols from Lycium barbarum (Goji) Fruit European Cultivars at Different Maturation Steps: Extraction, HPLC-DAD Analyses, and Biological Evaluation" Antioxidants 8, no. 11: 562. https://doi.org/10.3390/antiox8110562
APA StyleMocan, A., Cairone, F., Locatelli, M., Cacciagrano, F., Carradori, S., Vodnar, D. C., Crișan, G., Simonetti, G., & Cesa, S. (2019). Polyphenols from Lycium barbarum (Goji) Fruit European Cultivars at Different Maturation Steps: Extraction, HPLC-DAD Analyses, and Biological Evaluation. Antioxidants, 8(11), 562. https://doi.org/10.3390/antiox8110562