Chocolate Consumers and Lymphocyte-to-Monocyte Ratio: A Working Hypothesis from a Preliminary Report of a Pilot Study in Celiac Subjects
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of Subjects
2.2. Statitical Analysis
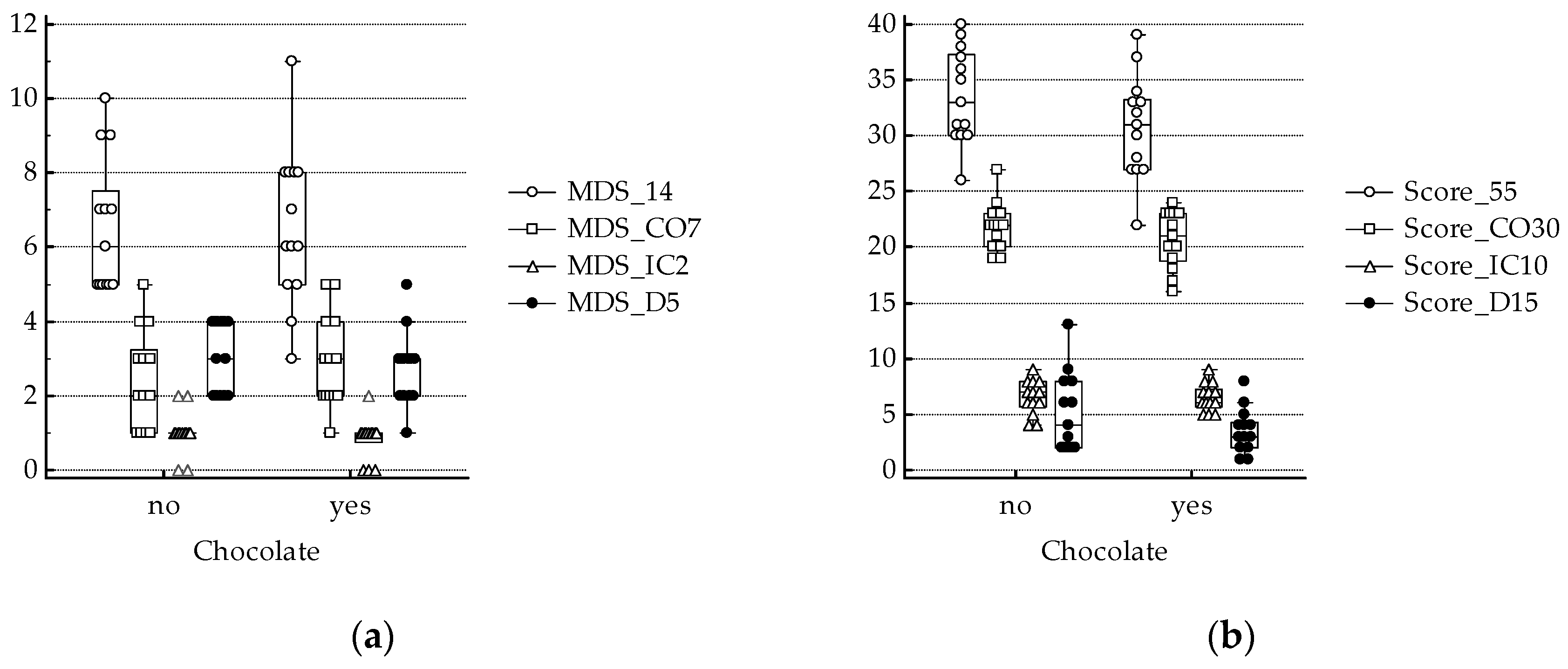
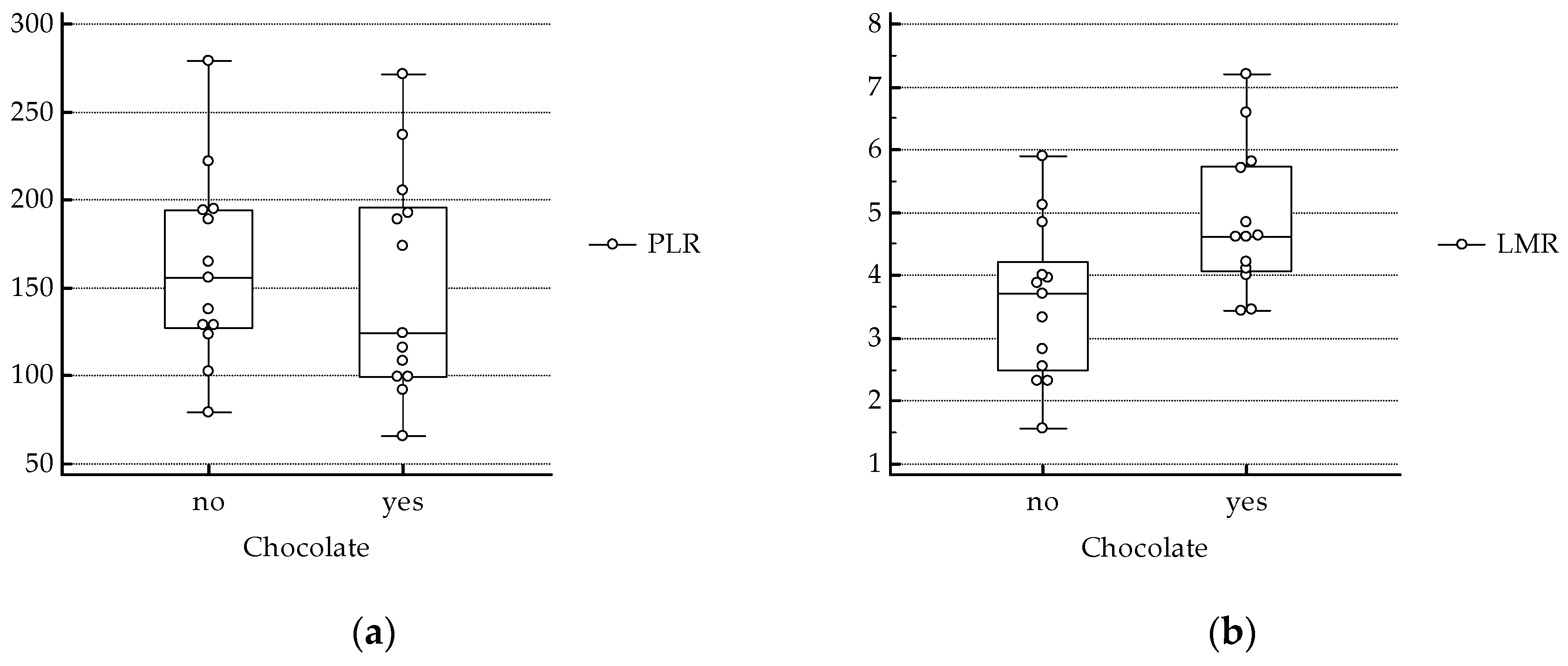
3. Results
4. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Paglieroni, T.G.; Janatpour, K.; Gosselin, R.; Crocker, V.; Dwyre, D.M.; MacKenzie, M.R.; Holland, P.V.; Larkin, E.C. Platelet function abnormalities in qualified whole-blood donors: Effects of medication and recent food intake. Vox Sang. 2004, 86, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Bordeaux, B.; Yanek, L.R.; Moy, T.F.; White, L.W.; Becker, L.C.; Faraday, N.; Becker, D.M. Casual chocolate consumption and inhibition of platelet function. Prev. Cardiol. 2007, 10, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Rull, G.; Mohd-Zain, Z.N.; Shiel, J.; Lundberg, M.H.; Collier, D.J.; Johnston, A.; Warner, T.D.; Corder, R. Effects of high flavanol dark chocolate on cardiovascular function and platelet aggregation. Vasc. Pharmacol. 2015, 71, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.B.; Kim, J.H.; Young, L.; Clark, J.M.; Park, Y. Epigallocatechin gallate (EGCG) alters body fat and lean mass through sex-dependent metabolic mechanisms in Drosophila melanogaster. Int. J. Food Sci. Nutr. 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Wang, D.; Xia, H.; Wang, F.; Yang, X.; Pan, D.; Wang, S.; Yang, L.; Lu, H.; Shu, G.; et al. A comparative study of the effects of palm olein, cocoa butter and extra virgin olive oil on lipid profile, including low-density lipoprotein subfractions in young healthy Chinese people. Int. J. Food Sci. Nutr. 2019, 70, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Dicks, L.; Kirch, N.; Gronwald, D.; Wernken, K.; Zimmermann, B.F.; Helfrich, H.-P.; Ellinger, S. Regular Intake of a Usual Serving Size of Flavanol-Rich Cocoa Powder Does Not Affect Cardiometabolic Parameters in Stably Treated Patients with Type 2 Diabetes and Hypertension-A Double-Blinded, Randomized, Placebo-Controlled Trial. Nutrients 2018, 10, 1435. [Google Scholar] [CrossRef]
- Kirch, N.; Berk, L.; Liegl, Y.; Adelsbach, M.; Zimmermann, B.F.; Stehle, P.; Stoffel-Wagner, B.; Ludwig, N.; Schieber, A.; Helfrich, H.-P.; et al. A nutritive dose of pure (-)-epicatechin does not beneficially affect increased cardiometabolic risk factors in overweight-to-obese adults-a randomized, placebo-controlled, double-blind crossover study. Am. J. Clin. Nutr. 2018, 107, 948–956. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, I.; Li, A.; Manson, J.E.; Sesso, H.D.; Wang, L.; Liu, S. Cocoa Flavanol Intake and Biomarkers for Cardiometabolic Health: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Nutr. 2016, 146, 2325–2333. [Google Scholar] [CrossRef]
- Beale, D.J. Inappropriate Inclusion of 3 Trials Leads to Misleading Results in Meta-Analysis of Cocoa Flavanol Intake and Cardiometabolic Health. J. Nutr. 2017, 147, 976. [Google Scholar] [CrossRef]
- Ellinger, S.; Stehle, P. Impact of Cocoa Consumption on Inflammation Processes-A Critical Review of Randomized Controlled Trials. Nutrients 2016, 8, 321. [Google Scholar] [CrossRef]
- Palmacci, F.; Toti, E.; Raguzzini, A.; Catasta, G.; Aiello, P.; Peluso, I.; Biava, M.; Palmery, M. Neutrophil-to-Lymphocyte Ratio, Mediterranean Diet, and Bone Health in Coeliac Disease Patients: A Pilot Study. Oxidative Med. Cell. Longev. 2019, 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sarikaya, M.; Dogan, Z.; Ergul, B.; Filik, L. Neutrophil-to-lymphocyte ratio as a sensitive marker in diagnosis of celiac disease. Ann. Gastroenterol. 2014, 27, 431–432. [Google Scholar] [PubMed]
- Balaban, D.V.; Popp, A.; Beata, A.; Vasilescu, F.; Jinga, M. Diagnostic accuracy of red blood cell distribution width-to-lymphocyte ratio for celiac disease. Revista Română de Medicină de Laborator 2018, 26, 45–50. [Google Scholar] [CrossRef]
- Sarikaya, M.; Dogan, Z.; Ergul, B.; Filik, L. Platelet-to-lymphocyte ratio for early diagnosis of celiac disease. Indian J. Gastroenterol. 2015, 34, 182–183. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Han, L.; Liu, H.; Wang, L.; Liu, X.; Gao, Y. Decreased Lymphocyte-to-Monocyte Ratio Predicts Poor Prognosis of Acute Ischemic Stroke Treated with Thrombolysis. Med. Sci. Monit. 2017, 23, 5826–5833. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Liu, X.; Wang, L.; Gao, Y. Lymphocyte-to-Monocyte Ratio: A Novel Predictor of the Prognosis of Acute Ischemic Stroke. J. Stroke Cerebrovasc. Dis. 2017, 26, 2595–2602. [Google Scholar] [CrossRef] [PubMed]
- Park, M.G.; Kim, M.K.; Chae, S.H.; Kim, H.K.; Han, J.; Park, K.P. Lymphocyte-to-monocyte ratio on day 7 is associated with outcomes in acute ischemic stroke. Neurol. Sci. 2018, 39, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Gary, T.; Pichler, M.; Belaj, K.; Eller, P.; Hafner, F.; Gerger, A.; Brodmann, M. Lymphocyte-to-monocyte ratio: A novel marker for critical limb ischemia in PAOD patients. Int. J. Clin. Pract. 2014, 68, 1483–1487. [Google Scholar] [CrossRef] [PubMed]
- Duran, M.; Kurtul, A. The correlation between lymphocyte/monocyte ratio and coronary collateral circulation in stable coronary artery disease patients. Biomark. Med. 2017, 11, 43–52. [Google Scholar]
- Silva, N.; Bettencourt, P.; Guimarães, J.T. The lymphocyte-to-monocyte ratio: An added value for death prediction in heart failure. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Chen, S.; Ying, H.; Li, J.; Shi, J.; Zhu, X.; Zhang, Y.; Shen, B. The association between the lymphocyte-monocyte ratio and disease activity in rheumatoid arthritis. Clin. Rheumatol. 2017, 36, 2689–2695. [Google Scholar] [CrossRef] [PubMed]
- Potter, M.D.E.; Brienesse, S.C.; Walker, M.M.; Boyle, A.; Talley, N.J. Effect of the gluten-free diet on cardiovascular risk factors in patients with coeliac disease: A systematic review. J. Gastroenterol. Hepatol. 2018, 33, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Ungprasert, P.; Wijarnpreecha, K.; Tanratana, P. Risk of venous thromboembolism in patients with celiac disease: A systematic review and meta-analysis. J. Gastroenterol. Hepatol. 2016, 31, 1240–1245. [Google Scholar] [CrossRef]
- Hurrell, R.F.; Reddy, M.; Cook, J.D. Inhibition of non-haem iron absorption in man by polyphenolic-containing beverages. Br. J. Nutr. 1999, 81, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Ertem, A.G.; Yayla, C.; Acar, B.; Kirbas, O.; Unal, S.; Sener, M.U.; Akboga, M.K.; Efe, T.H.; Sivri, S.; Sen, F.; et al. Relation between lymphocyte to monocyte ratio and short-term mortality in patients with acute pulmonary embolism. Clin. Respir. J. 2018, 12, 580–586. [Google Scholar] [CrossRef]
- Simons, M.; Scott-Sheldon, L.A.; Risech-Neyman, Y.; Moss, S.F.; Ludvigsson, J.F.; Green, P.H. Celiac Disease and Increased Risk of Pneumococcal Infection: A Systematic Review and Meta-Analysis. Am. J. Med. 2018, 131, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Romero, S.; Perez-Cano, F.J.; Castellote, C.; Castell, M.; Franch, A. Effect of cocoa-enriched diets on lymphocytes involved in adjuvant arthritis in rats. Br. J. Nutr. 2012, 107, 378–387. [Google Scholar] [CrossRef]
- Pioltine, M.B.; De Melo, M.E.; Santos, A.S.; Machado, A.D.; Fernandes, A.E.; Fujiwara, C.T.; Cercato, C.; Mancini, M.C. Genetic Variations in Sweet Taste Receptor Gene Are Related to Chocolate Powder and Dietary Fiber Intake in Obese Children and Adolescents. J. Pers. Med. 2018, 8, 7. [Google Scholar] [CrossRef]
- Melo, S.V.; Agnes, G.; Vitolo, M.R.; Mattevi, V.S.; Campagnolo, P.D.; Almeida, S. Evaluation of the association between the TAS1R2 and TAS1R3 variants and food intake and nutritional status in children. Genet. Mol. Boil. 2017, 40, 415–420. [Google Scholar] [CrossRef]
- Priego, T.; Sanchez, J.; Pico, C.; Ahrens, W.; de Henauw, S.; Kourides, Y.; Lissner, L.; Molnar, D.; Moreno, L.A.; Russo, P.; et al. TAS1R3 and UCN2 Transcript Levels in Blood Cells Are Associated with Sugary and Fatty Food Consumption in Children. J. Clin. Endocrinol. Metab. 2015, 100, 3556–3564. [Google Scholar] [CrossRef]
- Han, P.; Keast, R.S.J.; Roura, E. Salivary leptin and TAS1R2/TAS1R3 polymorphisms are related to sweet taste sensitivity and carbohydrate intake from a buffet meal in healthy young adults. Br. J. Nutr. 2017, 118, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Joseph, P.V.; Reed, D.R.; Mennella, J.A. Individual Differences Among Children in Sucrose Detection Thresholds: Relationship with Age, Gender, and Bitter Taste Genotype. Nurs. Res. 2016, 65, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Harwood, M.L.; Ziegler, G.R.; Hayes, J.E. Tolerance for High Flavanol Cocoa Powder in Semisweet Chocolate. Nutrients 2013, 5, 2258–2267. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, C.; Cicia, G.; Del Giudice, T.; Sacchi, R.; Vecchio, R. Consumers’ Perceptions and Preferences for Bitterness in Vegetable Foods: The Case of Extra-Virgin Olive Oil and Brassicaceae—A Narrative Review. Nutrients 2019, 11, 1164. [Google Scholar] [CrossRef] [PubMed]
- Hwang, L.-D.; Lin, C.; Gharahkhani, P.; Cuellar-Partida, G.; Ong, J.-S.; An, J.; Gordon, S.D.; Zhu, G.; MacGregor, S.; Lawlor, D.A.; et al. New insight into human sweet taste: A genome-wide association study of the perception and intake of sweet substances. Am. J. Clin. Nutr. 2019, 109, 1724–1737. [Google Scholar] [CrossRef] [PubMed]
- Pirastu, N.; Robino, A.; Lanzara, C.; Athanasakis, E.; Esposito, L.; Tepper, B.J.; Gasparini, P. Genetics of Food Preferences: A First View from Silk Road Populations. J. Food Sci. 2012, 77, S413–S418. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Stepaniak, U.; Topór-Mądry, R.; Szafraniec, K.; Pająk, A. Estimated dietary intake and major food sources of polyphenols in the Polish arm of the HAPIEE study. Nutrients 2014, 30, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Marventano, S.; Mistretta, A.; Galvano, F.; Grosso, G. Dietary sources of polyphenols in the Mediterranean healthy Eating, Aging and Lifestyle (MEAL) study cohort. Int. J. Food Sci. Nutr. 2017, 68, 750–756. [Google Scholar] [CrossRef] [PubMed]
| Chocolate | YES (n = 13) | NO (n = 13) |
|---|---|---|
| Men/women | 2/11 | 2/11 |
| Age (years) | 44.0 ± 3.7 | 36.8 ± 3.5 |
| GFD (years) | 2 (1–10) | 2 (1–7) |
| PAL (MET-min-week) | 6166 ± 891 | 7518 ± 1379 |
| BMI (Kg/m2) | 22.1 ± 0.5 | 22.2 ± 0.6 |
| SBP (mmHg) | 116.9 ± 3.1 | 108.4 ± 3.1 |
| DBP (mmHg) | 72.5 ± 2.4 | 69.8 ± 4.0 |
| Chocolate | YES (n = 13) | NO (n = 13) |
|---|---|---|
| WBC (103/microL) | 6.2 (4.5–6.8) | 5.4 (4.5–9.0) |
| N (103/microL) | 3.1 (2.2–4.3) | 3.5 (2.2–5.8) |
| M (103/microL) | 0.36 (0.32–0.54) | 0.44 (0.37–0.66) |
| L (103/microL) | 1.9 (1.4–2.4) | 1.7 (1.1–2.0) |
| P (103/microL) | 275.4 ± 14.8 | 253.8 ± 19.0 |
| RBC (106/microL) | 4.7 ± 0.1 | 4.7 ± 0.1 |
| MCV (fL) | 85.5 (77.5–87.3) | 88.4 (82.9–92.3) |
| Hb (g/dL) | 12.5 ± 0.6 | 13.5 ± 0.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raguzzini, A.; Poce, G.; Consalvi, S.; Toti, E.; Palmacci, F.; Biava, M.; Peluso, I. Chocolate Consumers and Lymphocyte-to-Monocyte Ratio: A Working Hypothesis from a Preliminary Report of a Pilot Study in Celiac Subjects. Antioxidants 2019, 8, 440. https://doi.org/10.3390/antiox8100440
Raguzzini A, Poce G, Consalvi S, Toti E, Palmacci F, Biava M, Peluso I. Chocolate Consumers and Lymphocyte-to-Monocyte Ratio: A Working Hypothesis from a Preliminary Report of a Pilot Study in Celiac Subjects. Antioxidants. 2019; 8(10):440. https://doi.org/10.3390/antiox8100440
Chicago/Turabian StyleRaguzzini, Anna, Giovanna Poce, Sara Consalvi, Elisabetta Toti, Francesca Palmacci, Mariangela Biava, and Ilaria Peluso. 2019. "Chocolate Consumers and Lymphocyte-to-Monocyte Ratio: A Working Hypothesis from a Preliminary Report of a Pilot Study in Celiac Subjects" Antioxidants 8, no. 10: 440. https://doi.org/10.3390/antiox8100440
APA StyleRaguzzini, A., Poce, G., Consalvi, S., Toti, E., Palmacci, F., Biava, M., & Peluso, I. (2019). Chocolate Consumers and Lymphocyte-to-Monocyte Ratio: A Working Hypothesis from a Preliminary Report of a Pilot Study in Celiac Subjects. Antioxidants, 8(10), 440. https://doi.org/10.3390/antiox8100440








